Introduction
Mucocutaneous fungal infections, including oral candidiasis, genital candidiasis, and esophageal candidiasis, are common fungal infections in the pediatric population caused by the Candida species, predominantly Candida albicans. In newborns, these infections assume a significant importance, especially in preterm patients, admitted to the neonatal intensive care unit (NICU) or after major surgery. Reference Warris, Pana and Oletto1,Reference Vila, Sultan, Montelongo-Jauregui and Jabra-Rizk2
The most severe complications of Candida spp. infections can involve especially preterm newborns after major surgery, and rarely healthy newborns. Reference De Rose, Santisi and Ronchetti3,Reference Chow, Linden and Bliss4 However, there is evidence that neonatal oral Candida spp. colonization is a risk factor for developing Candida septicemia and invasive candidiasis. Reference Batista, Krebs, Ruiz, Auler, Hahn and Paula5,Reference Endrigo Tinoco-Araujo, Ferreira and Araújo6 Further, the real epidemiology of Candida spp. infections in term newborns are still unclear. Reference Al-Rusan, Darwazeh and Lataifeh7
The vaginal maternal colonization can lead to possible transmission to the oral and skin of newborns, but even other mechanisms of transmission are described as contaminations of infant supplies and the hospital environment. Reference Roilides, Farmaki and Evdoridou8
We aim to describe the outbreak and the infection prevention and control (IPC) measures applied for neonatal Candida spp. infections in a newborn nursery and neonatal special care unit of a secondary hospital in Veneto and analyze the possible related risk factors for fungal infection in this population.
Methods
Study design and setting
This study was conducted at Santa Maria del Prato Hospital in Feltre, a public facility located in the northeastern Italian region of Veneto. The hospital is one of the main birth centers in the area, with around 750 deliveries recorded in 2022, accounting for 2.3% of all births in the Veneto Region. 9
The study was designed as an observational cohort study, initiated following the identification of a suspected outbreak of neonatal Candida infections between April and May 2024.
Although no structured surveillance data on the incidence of Candida infections were previously available in this facility, clinical staff noted an unusually high number of symptomatic cases within a short time frame.
In response, an active surveillance program was launched, extending screening to both symptomatic and asymptomatic newborns and mothers. The surveillance period lasted from April 1 to July 30, 2024, and aimed to define the extent and characteristics of the outbreak by including all neonates admitted to the unit.
Population and data sources
All newborns born from April 1, 2024, to June 30, 2024, and their mothers were included in the study. Two researchers collected the clinical, demographic, radiological, and microbiological findings of the newborns and the mothers. All clinical, demographic, diagnostic, and prescription data were retrieved from clinical charts and manually collected from electronic medical records through the Redcap® data collection form.
Case definition
A case was defined as a neonatal Candida infection characterized by clinical symptoms consistent with oral and/or genital candidiasis, or by a microbiologically positive Candida sample from an oral or genital swab. A control was defined as the absence of clinical symptoms consistent with oral and/or genital candidiasis, or by a microbiologically negative Candida sample from an oral or genital swab.
Infection control measures
Following the identification of the initial Candida case, a comprehensive review and reinforcement of infection control measures was undertaken. The objective was to interrupt the chain of transmission and minimize the risk of further cases among newborns.
Hand hygiene protocols were strengthened for both mothers and healthcare professionals involved in neonatal and perinatal care. Staff used contact precautions, including individual protective devices such as gloves and gowns, during patient handling and during any procedures or any contact with biological fluids. The use of single-patient devices was enforced, including nipple shields and breast pumps. These were either sent to the sterilization unit upon patient discharge or disinfected between uses through immersion in appropriate solutions. After the detection of the first case, all cribs and neonatal care devices underwent extraordinary sanitation procedures. The availability of sanitary products for disinfection and cleaning was verified and improved. Disinfection of single-use or shared supplies, including pacifier and nipple guard, was carried out using sodium hypochlorite-based solutions. Practices related to breastfeeding and colostrum administration were also revised. Colostrum was collected by mothers using disposable spoons, and both the spoons and contact surfaces were immediately sanitized after use. Breastfeeding management protocols were updated to align with UNICEF guidelines, with reinforced hygiene of the maternal nipple and handwashing procedures for both staff and mothers. 10
To assess clinical progression and potential late-onset symptoms, all neonates were evaluated between 7 and 10 days after discharge. Confirmed cases of neonatal Candida infection were treated with specific antifungal therapy.
Additionally, a structured checklist was introduced to monitor healthcare staff compliance with the revised infection control measures.
Microbiological methods
Pharyngeal and genital swabs were collected from newborns at birth and at discharge or twice weekly during the hospitalization period.
Vaginal swabs were obtained from mothers at hospital admission and again at the time of discharge for fungal screening. All healthcare personnel, including physicians, nurses, healthcare assistants, and midwives, underwent interdigital and subungual swabs for fungal culture testing as part of staff management and surveillance procedures.
The samples were analyzed using specific culture media (chromogenic Candida agar and Sabouraud dextrose agar supplemented with chloramphenicol). Cultures were incubated, and results were considered negative after 72 hours of incubation in the absence of any growth.
Statistical analysis
Results were summarized as frequencies and percentages. Bivariate analyses were performed on categorical variables, comparing groups using Pearson’s χ2 test or Fisher’s exact test when expected cell counts were <5. Two-sided P-values were reported, with a significance threshold set at .05. Bayesian logistic regression models were fitted to estimate associations with case/control status. This approach was chosen due to the presence of separation in some predictors. Univariate models were adjusted for sex and month of birth. A multivariate model was then built, with variable selection based on clinical hypotheses. All variables included in the regression models had complete data; no imputation procedures were applied. Normal priors centered at zero with standard deviation 2.5 were specified for the regression coefficients to regularize estimates and address separation. Posterior estimates were summarized as adjusted odds ratios (AdjOR) with corresponding 95% credible intervals (CI).
All analyses were carried out using R version 4.4.1 (R Core Team, Vienna, Austria).
Results
A total of 125 neonates and mothers were included in the study. Among the neonates, 16 were identified as cases with confirmed Candida cases, while the remaining 109 served as controls without evidence of fungal infection. The complete features of variables are reported in Tables 1 and 2 for newborns and mothers, respectively. Figure 1 shows the temporal trend of outbreak control.

Figure 1. Trend of Candida cases from March to June. The number of reported cases remained at zero through March, began increasing in early April, and peaked in early May before declining again by June. A recognized outbreak was identified between April 1st–15th marked by the red dashed line. The Infections and Prevention Control (IPC) measures were applied from May 1st, 2024. The surveillance period lasted from April 1st to July 30th, 2024.
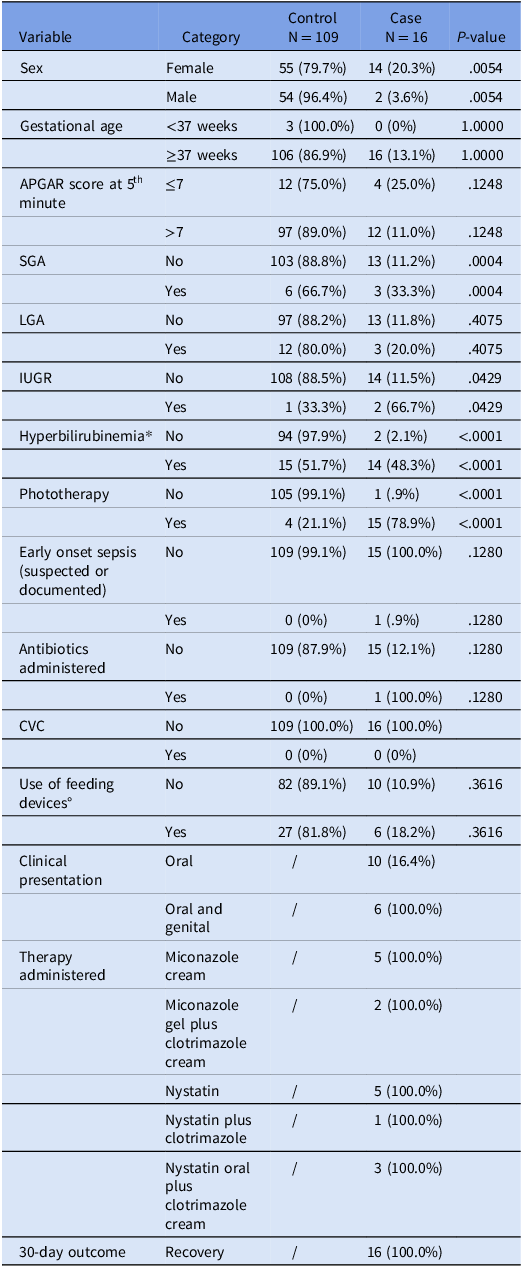
Table 1. Clinical and demographic features of newborns

SGA, Small for Gestational Age; LGA, Large for Gestational Age; IUGR, Intrauterine Growth Restriction. CVC, Central Venous Catheter. *considered >14 mg/dL. ° including pacifier, nipple shields, and other support devices.
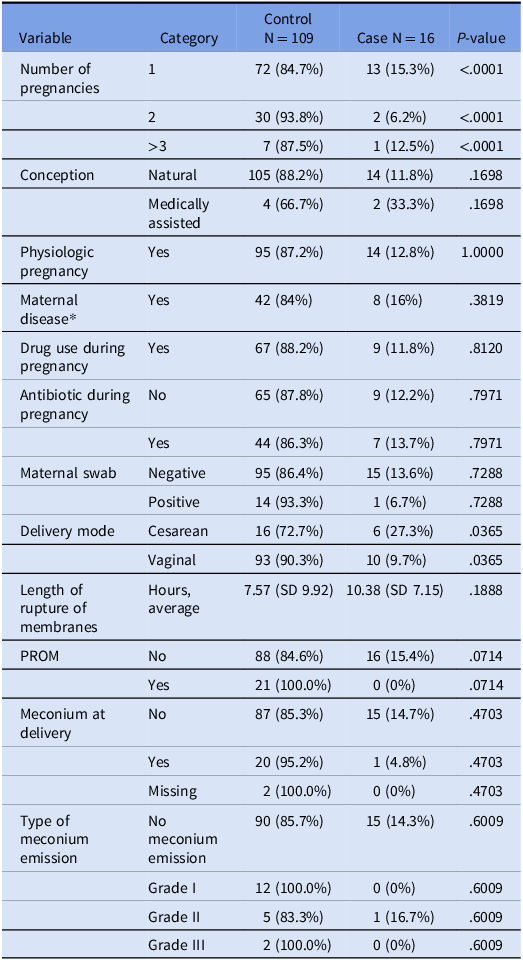
Table 2. Clinical and demographic features of mothers

SD, Standard deviation; PROM, Premature Rupture of Membranes.
* The definition maternal disease includes patients with gestational diabetes, thyroiditis, chronic hypertension, obesity, and other diseases. Gestational diabetes was not analyzed separately because its prevalence in our cohort was very low. None of the patients received steroids during pregnancy.
Female neonates were significantly more represented in the Candida case group, accounting for 87.5% (n = 14) of cases, compared to 50.5% (n = 55) among controls (P value: .0054). Only 3 neonates were preterm (<37 gestational weeks), and none of these presented a fungal infection. Most cases (n = 12) presented APGAR score at 5 minutes less than 7; Most of the patients were SGA (3/9, 33%) in cases with a significant distribution between cases and control (P value: .0004). Further, most of cases required phototherapy (15/16) for physiologic increase of bilirubin after birth (P value: .0001).
Regarding the use of supplies, 6 in cases and 27 in control required the use of supplies without a statistically significant (P value: .3616).
All cases required one or more topical antifungal treatments in detail topic miconazole (7/16), clotrimazole (6/16), and oral nystatin 8/16. The application surveillance measures were applied as previously described, and according to our results, only 2 cases were identified after the introduction of the infection control (P value: <.0001).
Regarding the mother’s cohort, primiparity was significantly associated with Candida cases (p value <.0001). Among mothers of infected neonates, 68.8% (n = 11) were primiparous, compared to only 27.5% (n = 30) in the control group. Cesarean section (C-section) was more frequent among cases (56.3%, n = 9) compared to controls (40.4%, n = 44), while vaginal delivery was observed in 43.7% of cases (n = 7) versus 59.6% (n = 65) of controls (P value: .2794). Meconium emission was reported in only one case and in 20 controls without a significant difference. The use of antibiotics predelivery was high in our cohort, with 44 in control and 7 patients in the case group, but this factor was not statistically significant (P value: .7971). Further, among the predelivery factors, the administration of drugs during pregnancy, the use of steroids, and the non-physiologic pregnancy were not statistically significant.
The microbiological surveillance screening in mothers showed a total of 11 positive swabs for Candida species, 10 Candida albicans, and one Candida glabrata. Particularly, only 3 of the newborns from positive mothers presented a clinical sign for fungal infection without a statistically significant result (P value: .6716).
The 16 cases of Candida infection were treated with topical azoles (7/16), clotrimazole (6/16) for patients presenting with vaginal or genital infections and oral nystatin (8/16) for oral Candida infections. All patients recovered fully without progression to invasive fungal infection, and no further diagnostic evaluations were required.
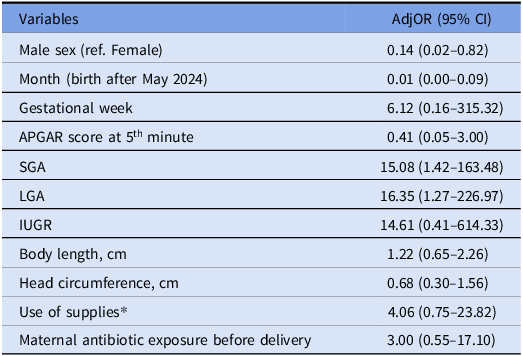
The multivariate analysis identified several factors associated with neonatal Candida infection (Figure 2 and Table 3). A strong temporal effect was observed: infants born after May 1st had a substantially lower risk of infection compared with those born earlier (Adj OR 0.01, 95% CI 0.001–0.09). Male sex and macrosomia also resulted a protective (Adj OR 0.14, 95% CI 0.02–0.82, and Adj OR 0.43, 95% CI 0.006–19.95, respectively).

Figure 2. Forest plot showing posterior median adjusted odds ratios and 95% credible intervals from a Bayesian logistic regression model for factors associated with infection. Reference categories: sex (female), weight status (normal), month (before May) and dichotomous variables (absence of condition).
Table 3. Multivariate Bayesian logistic regression analysis of factors associated with neonatal Candida infection

AdjOR, adjusted odds ratios; CI, credible intervals (CI); ref, reference class; SGA, Small for Gestational Age; LGA, Large for Gestational Age; IUGR, Intrauterine Growth Restriction.
* Including pacifier and nipple guard.
In contrast, increased gestational age resulted in a Candida infection risk factor (Adj OR 6.12, 95% CI 1.57–315.32). Small-for-gestational-age (SGA) infants showed markedly higher risk compared with those appropriate for gestational age (AdjOR 15.08, 95% CI 1.42–163.48).
Additional risk factors included low birth weight (AdjOR 5.44, 95% CI 0.12–209.27), use of supplies including pacifiers and nipple guards (AdjOR 4.06, 95% CI 0.75–23.82), and maternal antibiotic exposure before delivery (AdjOR 3.00, 95% CI 0.55–17.10).
The U-shaped relationship between fetal growth abnormalities and Candida risk suggests that both growth restriction and excessive growth may represent underlying conditions that predispose to fungal infection.
Discussion
In this study, we describe a neonatal outbreak of Candida infection in a secondary hospital and detail the measures adopted for its management and control. Furthermore, in accordance with current IPC guidelines and within the IPC context, we analyzed the potential risk factors associated with non-invasive mucocutaneous fungal infections in this population. Reference Stone, Cooper and Kibbler11
Outbreak control in neonatal populations represents a major concern for healthcare systems, particularly in NICU settings. Reference Huang, Lin, Leu, Peng, Wu and Chang12 Our findings are aligned with those of other reports of neonatal Candida outbreaks, which emphasize both vertical and horizontal transmission pathways, though most existing literature focuses on invasive forms rather than non-invasive mucocutaneous infections. Reference Ferreras-Antolin, Chowdhary and Warris13
Infections of the oral and vaginal mucosa have been identified as significant risk factors for subsequent neonatal Candida colonization or infection, especially among low-birth-weight infants. Reference Sousa, Oliveira Diniz, Lapa Marinho, Rezende, Carellos and Maia de Castro Romanelli14,Reference Cinicola, Uva, Duse, Zicari and Buonsenso15 While we found a high proportion of Candida infections in terms of SGA neonates, interestingly, we did not find maternal vaginal fungal colonization to be a statistically significant risk factor in our cohort, a finding that contrasts with several prior studies, which have found vertical transmission and maternal colonization to be a significant contributor. Reference Zhang, Xie, Yuan, He, Dong and Lei16 However, the potential adverse outcomes associated with maternal Candida carriage remain a matter of debate. Reference Al-Rusan, Darwazeh and Lataifeh7,Reference Schuster, de Jonghe, Limpens, Budding and Painter17
Nevertheless, although our focus was on non-invasive mucocutaneous infections, the increasing incidence of Candida infections, recognized in the WHO priority pathogen list, and the emergence of antifungal-resistant strains such as Candida auris highlight the importance of rigorous IPC measures, even among healthy, full-term newborns. Reference Pyrpasopoulou, Zarras and Mouloudi18,Reference Casalini, Giacomelli and Antinori19
In neonates, infections at multiple sites has been shown to increase the risk of progression to invasive disease, underscoring that mucocutaneous involvement should not be dismissed as benign. Reference Sousa, Oliveira Diniz, Lapa Marinho, Rezende, Carellos and Maia de Castro Romanelli14
A key strength of our study lies in the effectiveness of the IPC strategies implemented: routine surveillance of patients and healthcare workers, strict environmental sanitation, and adherence to hand hygiene protocols, which significantly limited the spread of new infections within the ward. Most studies on IPC measures have been reported in the context of preventing antimicrobial resistance and the spread of multidrug-resistant pathogens. Reference Anello, Vianello and Baldo20
However, our findings confirm the important role of bundles of IPC measures even in controlling fungal outbreaks, consistent with evidence that prevention may be even more critical than prophylaxis in neonatal settings. 21
Our study has some limitations: the limited number and short temporal span of cases may not be representative of the overall newborn population, and we lacked data on in vitro susceptibility patterns, which limited our ability to interpret the characteristics of the pathogens identified.
In conclusion, our experience supports the concept that neonatal non-invasive Candida outbreaks, though less well described than invasive cases, merit the same level of vigilance. Even full-term infants may be at risk when mucocutaneous infections occur under outbreak conditions, and proactive IPC measures are essential to containment.
Data availability statement
Data are contained within the article.
Author contribution
Conceptualization: L.C. and F.G; methodology: L.C., F.G., and C.C; validation: S.M. and E.B.; formal analysis: M.B. and C.C.; investigation: L.C., C.C., M.B. and G.F.; resources: M.G., A.V, A.S., O.F., D.S., M.N., and I.P.; data curation: O.F., D.S., I.P, and M.N. writing—original draft preparation: L.C., G.F., M.B., and C.C.; writing—review and editing: V.B., S.M, and E.B.; supervision: S.M and E.B.
All authors have read and agreed to the published version of the manuscript.
Financial support
This study was carried out as part of our routine work.
Competing interests
The authors declare no conflict of interest.
Ethical standard
Privacy was guaranteed by assigning a specific alphanumeric code to each patient. The study was conducted according to the principles of Good Clinical Practice and the Helsinki Declaration. 22 This study was conducted in compliance with national and institutional guidelines and approved by the ethical committee. In the present study, all procedures were conducted in accordance with the ethical standards of the Helsinki Declaration. The study protocol was approved by the local Ethics Committee under approval number (CANDIFE cod. inter. CET ANV 2024-103), dated 04/11/2024. Informed consent was obtained from all participants or their legal guardians.