Introduction
Lead silicates form a diverse category of compounds, occurring in both natural mineral assemblages (skarns and hydrothermal veins) and in anthropogenic formations (e.g. ceramics and metallurgical slags). The diversity is underpinned by the behaviour of the Pb2⁺ ion, with a stereo-active lone pair of electrons, and its influence on the structural arrangement of the tetrahedral SiO4 units (e.g. Siidra et al., Reference Siidra, Zenko and Krivovichev2014). The inclusion of other metals in the crystal structures further expands the possibilities for diversification. As an environmental pollutant, Pb can be immobilised in silicate phases (Li et al., Reference Li, Zheng, Fu, Wu, Yang and Shen2012), but the element may also be released during hydrothermal or supergene alteration of those (e.g. Davis et al., Reference Davis, Drexler, Ruby and Nicholson1993). This is a motivation to study these compounds further.
About 50 different Pb silicates are known as minerals, most of them restricted to a few prolific ore localities, like the Långban Fe–Mn mine and its satellite deposits in the Filipstad district, Värmland, Sweden (Moore, Reference Moore1970; Holtstam and Langhof, Reference Holtstam and Langhof1999), Franklin-Sterling Hill, New Jersey (Dunn, Reference Dunn1985) and the Kombat mine, Namibia (Dunn, Reference Dunn1991). We have reinvestigated jagoite, a very rare mineral with the formula Pb18Fe3+4[Si4(Si,Fe3+)6][Pb4Si16(Si,Fe)4]O82Cl6. It was described briefly by Blix et al. (Reference Blix, Gabrielson and Wickman1957) from Långban and the crystal structure was later solved by Mellini and Merlino (Reference Mellini and Merlino1981). For this study, we used the type material, and additional samples not studied in detail before, from Långban and the Pajsberg Mn–Fe deposit (both Filipstad, Värmland).
Geological setting
The carbonate-hosted Långban-type Fe–Mn–(Ba–Pb–As–Sb) deposits are associated with Palaeoproterozoic (Orosirian period), mainly rhyolitic volcanic and volcanoclastic country rocks in the Bergslagen ore province (formally: the Bergslagen lithotectonic unit; Stephens and Jansson, Reference Stephens and Jansson2020) of south-central Sweden. This unique kind of mineralisation, known for its exceptional wealth of rare minerals, is attributed to a combination of exhalative–sedimentary, high-grade metamorphic and hydrothermal processes (Boström et al., Reference Boström, Rydell and Joensuu1979; Holtstam and Mansfeld, Reference Holtstam and Mansfeld2001). Metal-enriched sediments were deposited at ∼1.9 Ga from exhalative-volcanogenic solutions in a shallow-submarine, oxidising environment. During subsequent tectonic and metamorphic events, culminating during the Svecofennian orogeny (∼1.85 Ga), the primary ores recrystallised and intense skarn formation occurred. (Note that the term ‘skarn’ here is used in its original, non-genetic sense referring to silicate mineral assemblages associated with ore.) Peak conditions in the Långban area are estimated at 600 ± 50°C and 0.3–0.4 GPa (Christy and Gatedal, Reference Christy and Gatedal2005; Skelton et al., Reference Skelton, Mansfeld, Ahlin, Lundqvist, Linde and Nilsson2018). Later brittle deformation and hydrothermal activity, possibly related to the Sveconorwegian (1.2–0.9 Ga) orogeny, have played a crucial role in the remobilisation of key elements and further evolution of the deposits and led to the formation of various late-stage minerals (mostly in fissures at temperatures < 200°C and low lithostatic pressure; Jonsson and Broman, Reference Jonsson and Broman2002), contributing significantly to the species diversity here.
Methods
Selected representative samples from the collections of the Swedish Museum of Natural History, Department of Geosciences, have been investigated. For single-crystal work and spectroscopy, crystals from the type specimen #19410178 were used. All samples were examined under a FEI Quanta 650 field-emission-gun scanning electron microscope (SEM), fitted with a back-scattered electron (BSE) detector and an 80 mm2 X-MaxN Oxford Instruments energy-dispersion (EDS) micro-analyser, on polished sections in carbon-coated epoxy mounts. Powder X-ray diffraction (PXRD) using a PANalytical X’Pert PRO diffractometer (CuKα radiation) was used for complementary mineral identification.
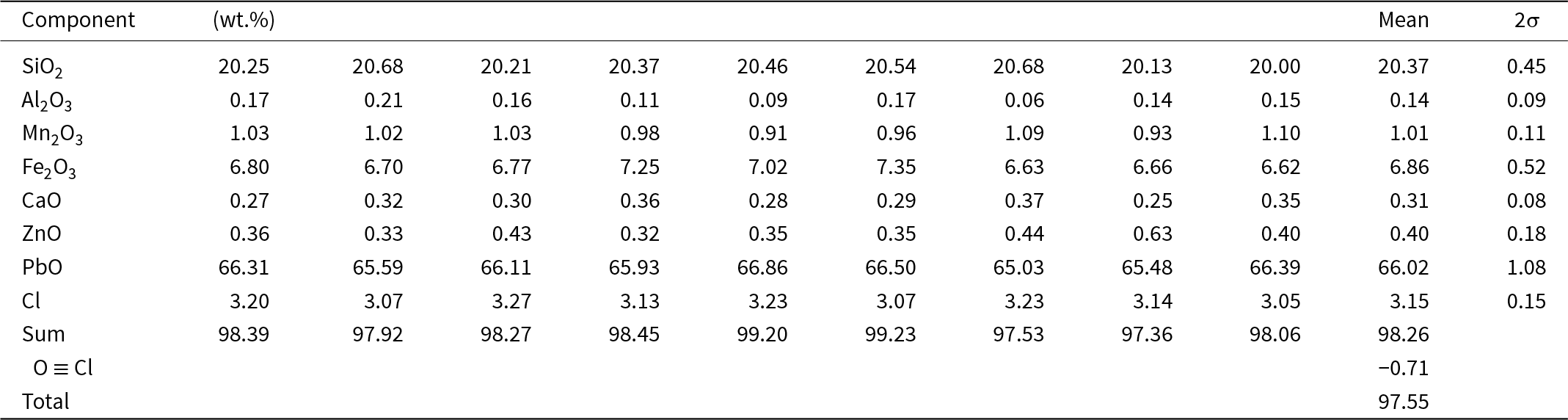
Electron-microprobe analyses (EMPA) of type jagoite (Table 1) were collected with an AZtecWave wavelength-dispersion (WDS) system attached to the SEM as above, at 20 kV and 14 nA, with a working distance of 10 mm. Mineral reference materials were used as follows: jadeite (NaKα), almandine (Al, Si, CaKα), rhodonite (MnKα), hematite (FeKα), willemite (ZnKα), crocoite (PbLα) and tugtupite (ClKα). Sodium, Mg and K were close to or below the detection limit with this method. Low analytical results were observed occasionally and may be related to inadequate conduction on detached mineral grains embedded in epoxy.
Electron-microprobe analyses of type jagoite, GEO-NRM #19410178

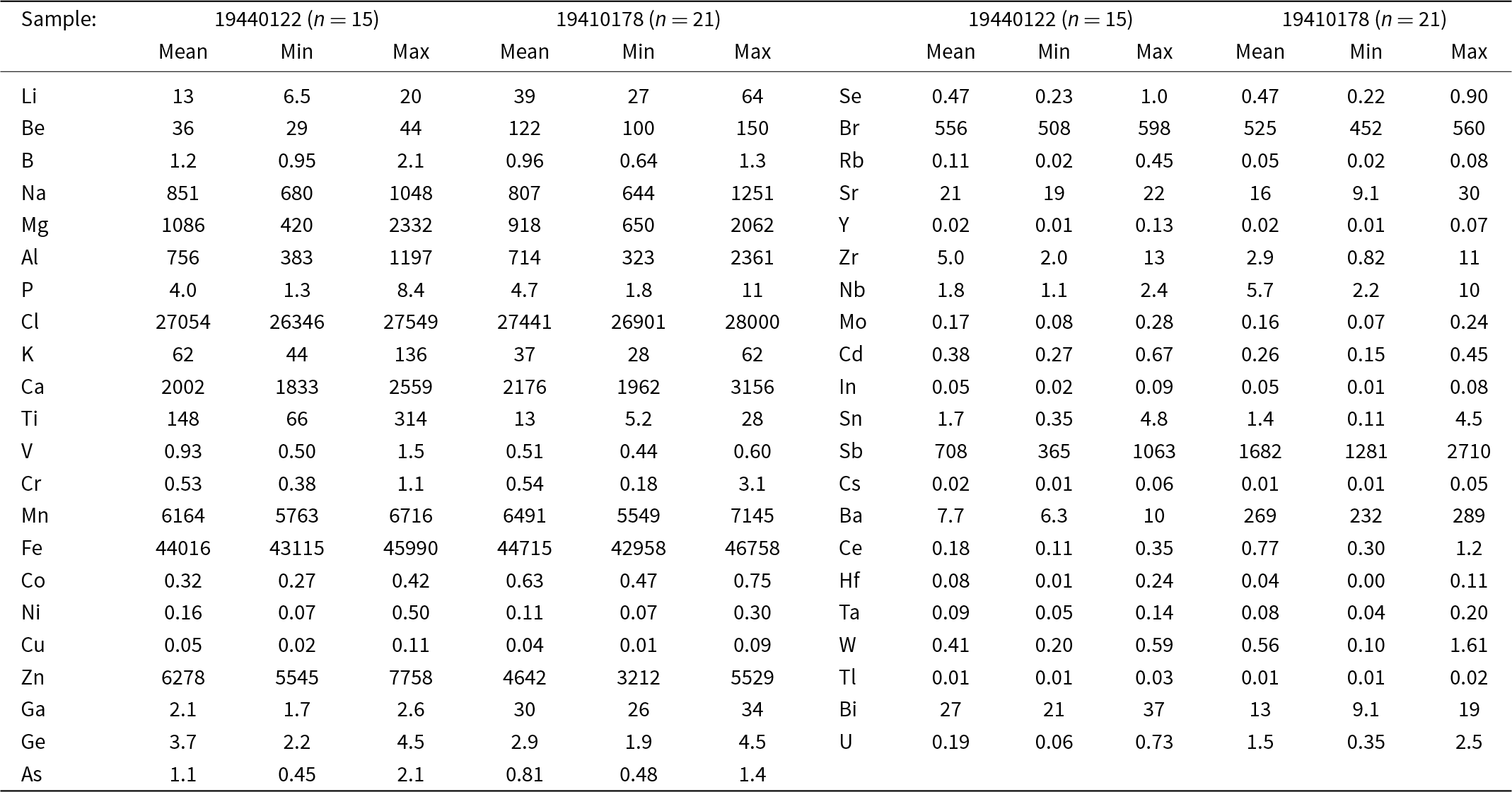
In situ chemical analyses by laser-ablation inductively coupled plasma mass spectrometry analyses (LA-ICP-MS) on jagoite (Table 2) were carried out with a New Wave Research 213 nm laser coupled to an Agilent 8800 MS/MS at the University of Gothenburg. The data were retrieved from two analytical sessions, one with a 70 µm circular spot and a laser repetition rate of 10 Hz, corresponding to a fluency of 5 J/cm2. For the second analytical session a laser spot of 50 µm was used, with all other settings kept the same. We used NIST SRM610 as the primary standard for minor and trace element quantification, with exceptions for Cl (sodalite, ILM127; Zahoransky et al., Reference Zahoransky, Friis and Marks2016) and Br (doped basaltic glass, GSE-1g, Marks et al., Reference Marks, Kendrick, Eby, Zack and Wenzel2017; Caulfield et al., Reference Caulfield, Tomlinson, Chew, Marks, McKenna, Ubide and Smith2020). Silica was used as an internal standard, which in turn was derived from electron-microprobe data. The accuracy and precision of the concentrations of minor and trace elements were evaluated with data on BCR2-g and GSE1-g. The elements S, Te, I and Th were sought but found to be close to or below the level of detection. Values obtained for Fe2O3 and CaO (wt.%), respectively, are 6.86±0.52, 0.31±0.08 (WDS) vs. 6.30±0.27, 0.30±0.07 (LA-ICP-MS) for the type specimen, confirming a good agreement between the methods for metals.
Element concentrations in jagoite from LA-ICP-MS analyses (µg/g)
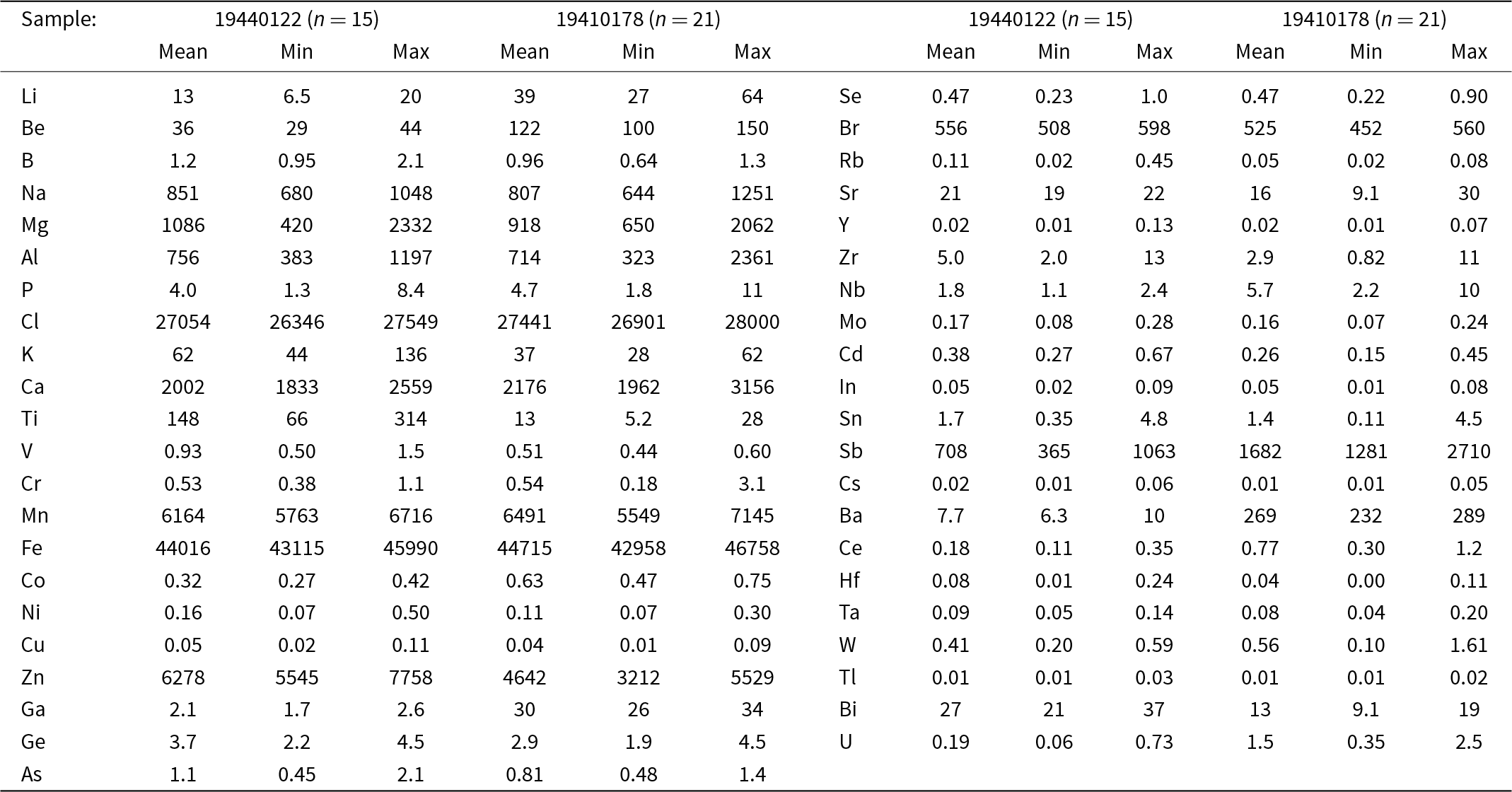
n = number of analyses.
Single-crystal X-ray diffraction data were collected from a thin crystal fragment (dimensions 0.189 × 0.085 × 0.030 mm) and with a sensitive detector, HyPix, mounted on a XtaLAB Synergy by Rigaku, using MoKα radiation. A dataset of 46 runs of 0.5° wide images were collected at 15 and 40 seconds for a total of 6604 images to ensure full coverage of the reciprocal space and correct absorption correction.
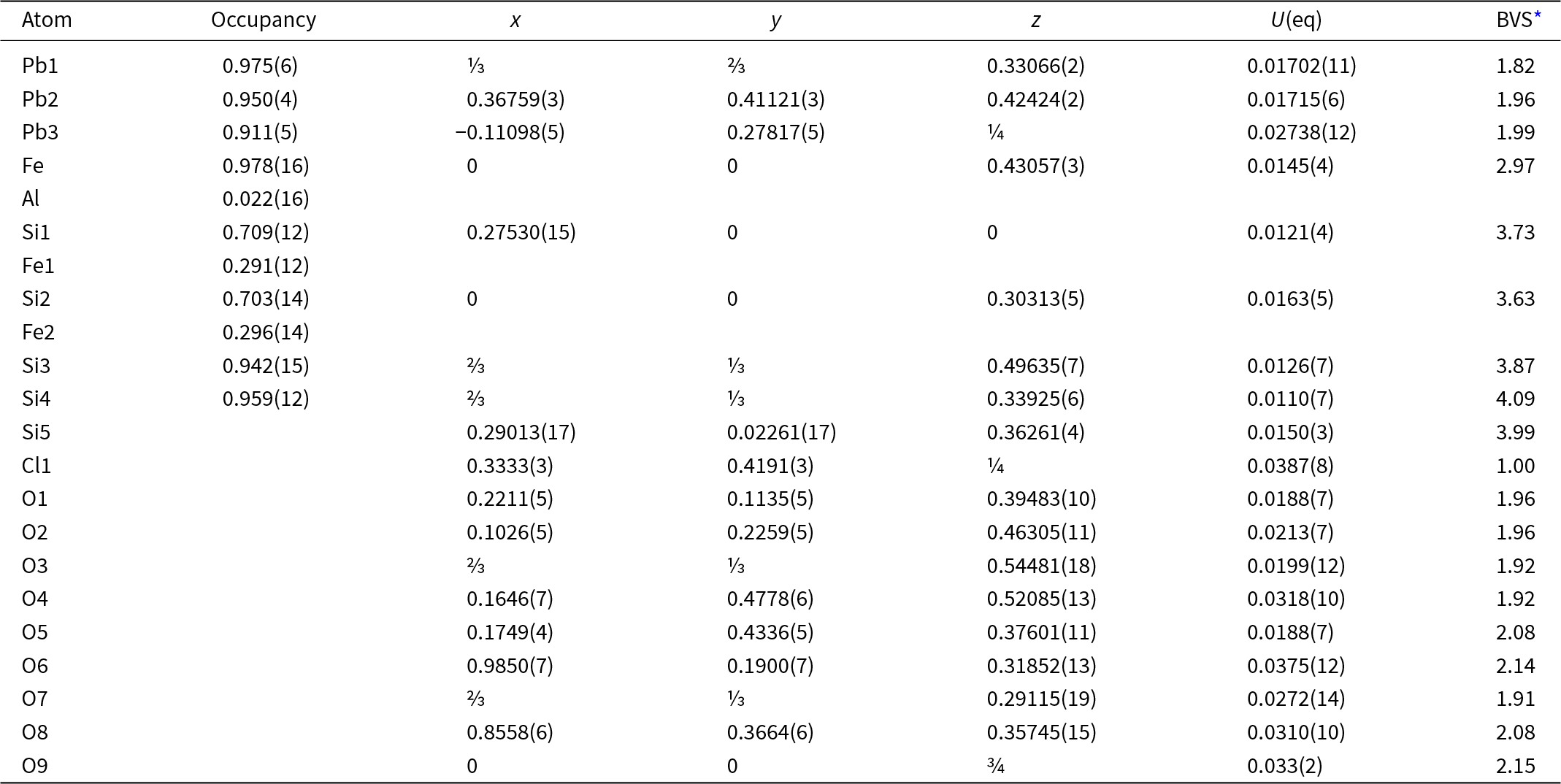
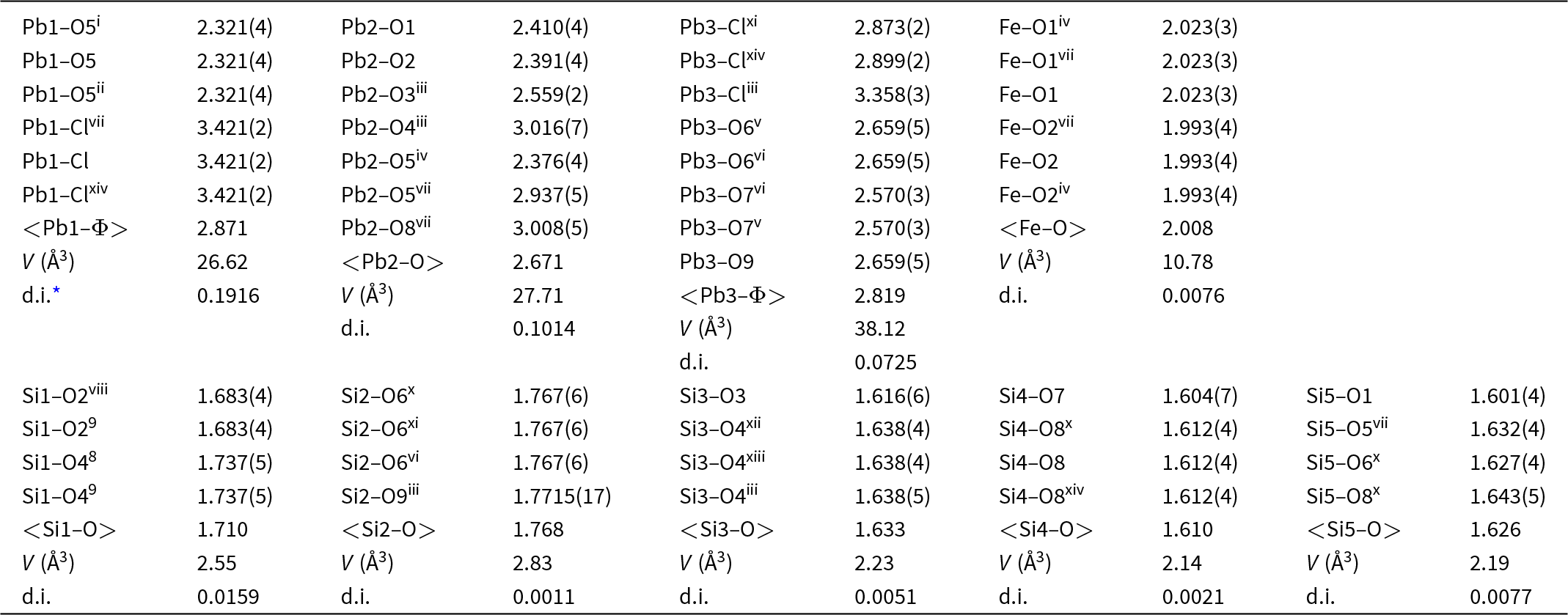
The crystal structure of jagoite was refined by using Olex 2 v. 1.3 (Dolomanov et al., Reference Dolomanov, Bourhis, Gildea, Howard and Puschmann2009) and SHELXL 2018/3 (Sheldrick, Reference Sheldrick2015) starting from the atom coordinates of Mellini and Merlino (Reference Mellini and Merlino1981) using neutral scattering curves and refining Fe versus Al in the Fe octahedron, Si versus Fe in the Si1 and Si2 tetrahedra, Si in the Si3, Si4 and Si5 tetrahedra, Pb at the Pb1, Pb2 and Pb3 sites, Cl at the Cl1 site and oxygen at the nine anion sites in the asymmetric unit. The experimental and crystal data are summarised in Table 3. Atomic coordinates, displacement parameters and calculated bond-valences are given in Table 4. Bond distances are reported in Table 5. The crystallographic information file of this study has been deposited with the journal and is available as Supplementary material (see below).
Crystal data and structure refinement for jagoite, sample GEO-NRM #19410178

Weight = 1/[σ2(F o2) + (0.0481 × P)2 + 34.76 × P] where P = (Max(F o2, 0) + 2 × F c2)/3.
Fractional atomic coordinates and equivalent isotropic displacement parameters (Å2) for jagoite. U eq is defined as ⅓ of the trace of the orthogonalised U ij tensor. Bond valence sums (BVS) are in valence units (vu)
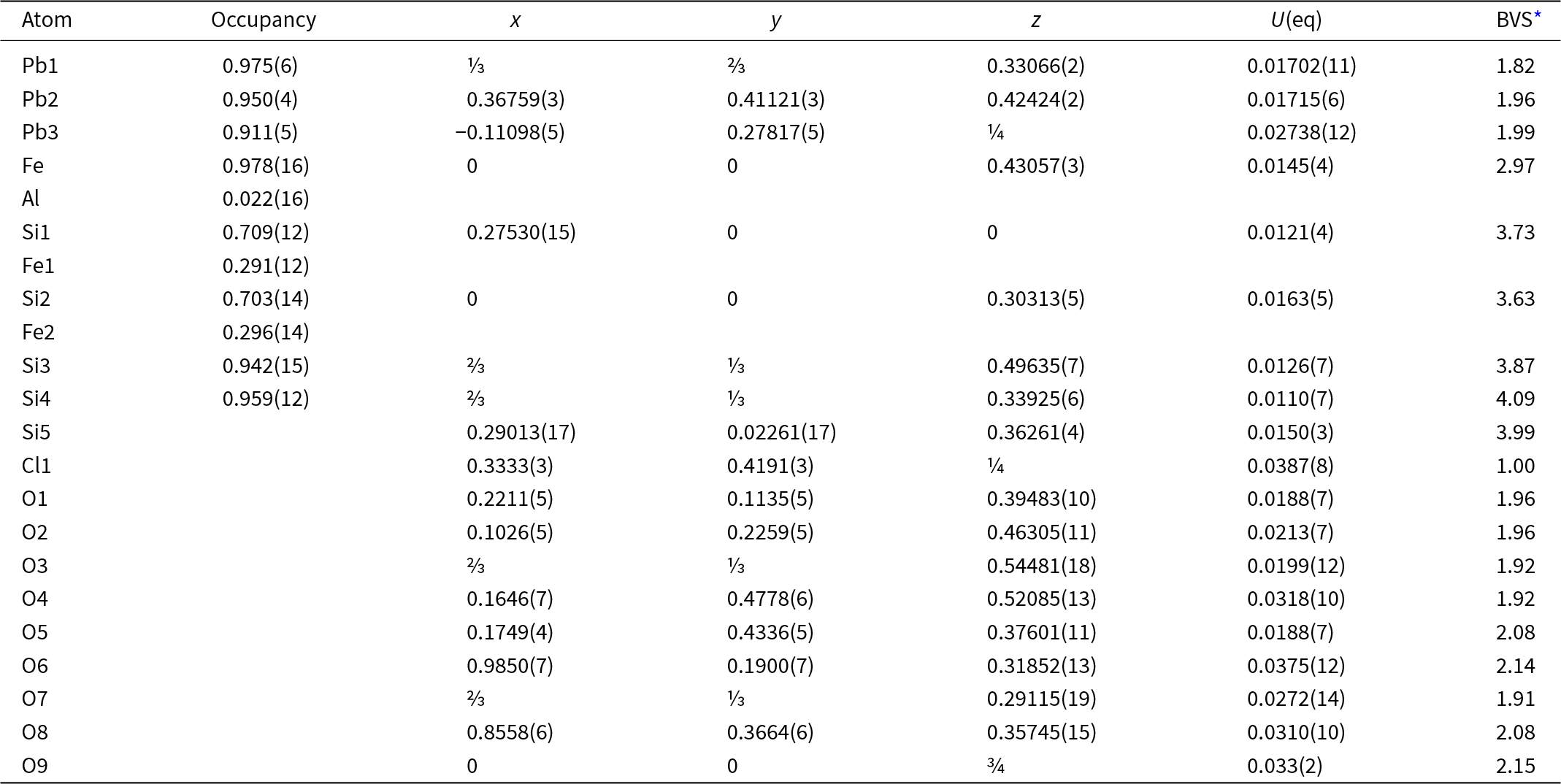
* Bond valence parameters for cation–oxygen bonds taken from Gagné and Hawthorne (Reference Gagné and Hawthorne2015) and Brese and O’Keeffe (Reference Brese and O’Keeffe1991) for cation–chlorine bonds.
Bond lengths (Å) and geometrical parameters for jagoite
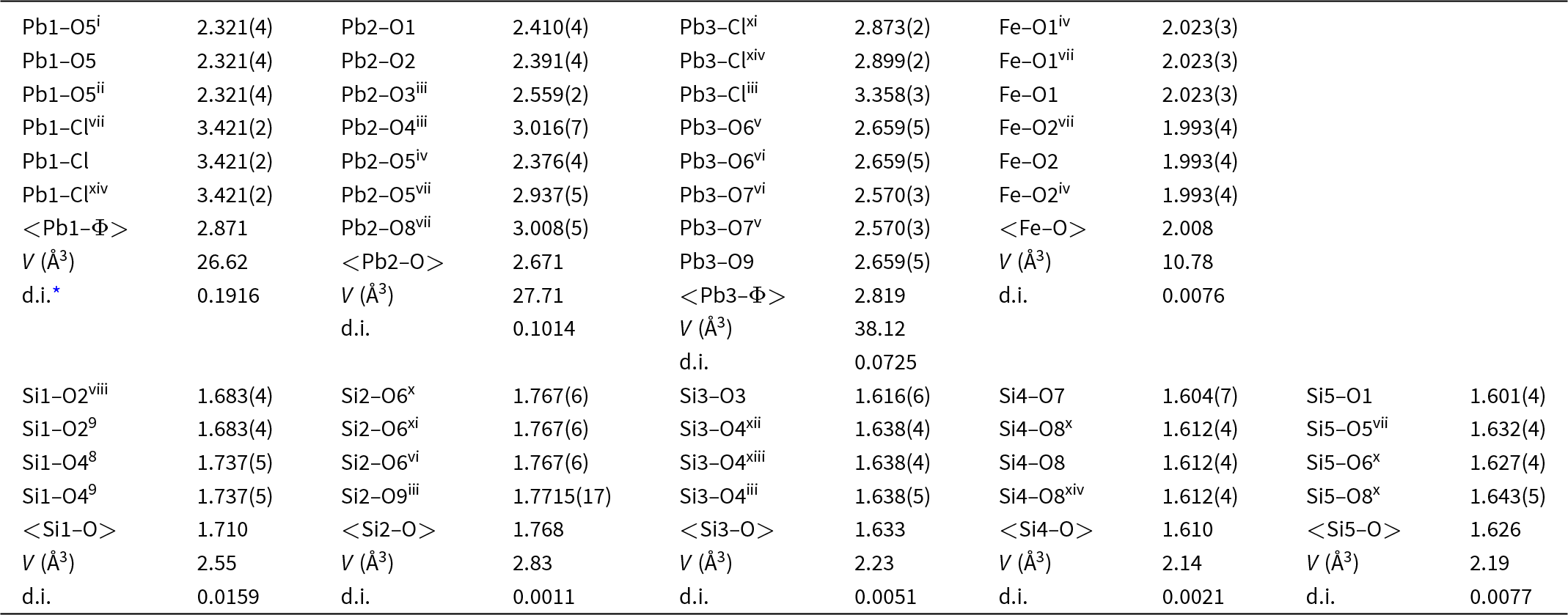
Notes: Symmetry codes: (i) +y–x, 1–x, +z; (ii) 1–y, 1+x–y, +z; (iii) +y, +x, 1–z; (iv) –y, +x–y, +z; (v) –1+x, +y, ½–z; (vi) –1+x, +y, +z; (vii) +y–x, –x, +z; (viii) +y–x, –x, ½–z; (ix) +y, +x, –½+z; (x) 1+y–x, 1–x, +z; (xi) –y, –1+x–y, +z; (xii) 1–x, –x+y, 1–z; (xiii) 1–y+x, 1–y, 1–z; (xiv) 1–y, +x–y, +z;
* distortion index (Baur, Reference Baur1974).
Transmission 57Fe Mössbauer spectra were obtained from a 30 mg jagoite powder absorber mixed with 100 mg acrylic resin and pressed into a circular tablet. To exclude contamination of the powder with alteration products and foreign inclusions, crystal fragments were hand-picked under the binocular microscope. A test run with PXRD showed only jagoite in the purified sample, with refined unit-cell parameters a = 8.5530(6) and c = 33.376(5) Å. For the experiments, 57Co γ-radiation (Rh matrix source, nominally 1.8 GBq) and a constant-acceleration spectrometer (WissEl) were used. Two-mirror image spectra (± 4.28 mm/s) were collected at 54.7° geometry to avoid preferred-orientation effects, at 298 K and 77 K (with a liquid-nitrogen cryostat) during 15 and 5 days, respectively, over 1024 channels. The raw data were calibrated against a 25 µm iron foil. The MossA software (Prescher et al., Reference Prescher, McCammon and Dubrovinsky2012) was employed for spectrum analysis, under the assumption of Lorentzian line shapes and a ‘thin’ absorber.
Raman spectra were obtained from randomly orientated polished grains using a LabRAM HR 800 micro-spectrometer. The analysis employed a 514 nm Ar-ion laser source with a power output of ∼1 mW, a Peltier-cooled (–70°C) 1024 × 256 pixel CCD detector (Synapse), and an Olympus M Plan N 100×/0.9 NA objective, providing a laser spot size of ∼3 μm. A 600 grooves/cm grating was used, achieving a resolution of about 1 cm⁻1. Peak positions were calibrated against the Raman band at 789 cm⁻1 of a silicon carbide crystal measured on {0001}. Data acquisition and instrument control were managed with LabSpec 5 software, covering a spectral range of 100–4000 cm⁻1, with 30 second exposure times repeated over five cycles.
Results
Mineral assemblages
The description by Blix et al. (Reference Blix, Gabrielson and Wickman1957) of the mineral association is skeletal; the mineral was found in hematite ore from the Canberra stope (at 220 m depth; Magnusson, Reference Magnusson1930) of the Långban mine in the 1940s, together with melanotekite, quartz and an unidentified mineral.
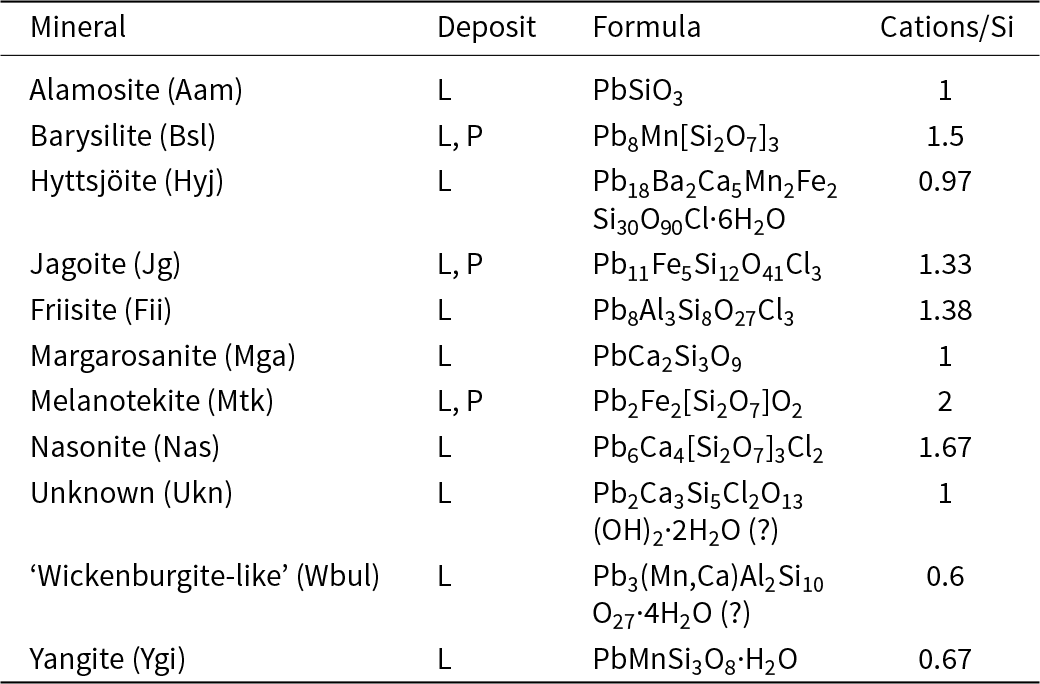
From the present study, it is clear that the mineral assemblage is a highly heterogeneous skarn, in contact with dolomitic marble, with ubiquitous andradite, pyroxene (diopside–aegirine), hematite and quartz, plus three or more of the lead silicates alamosite, barysilite, hyttsjöite, jagoite, margarosanite, melanotekite, nasonite and yangite, in each specimen (Figs 1–4). Most of these minerals approach their nominal chemical compositions (see Table 6).
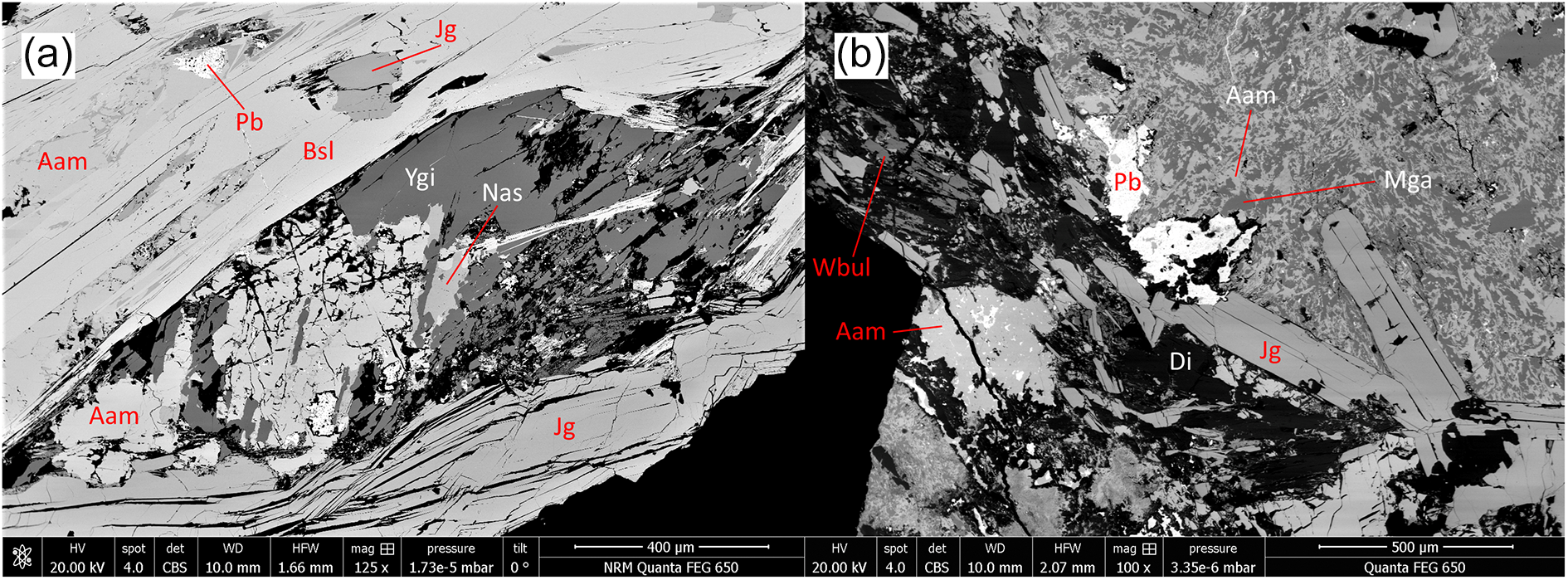
BSE images of polished sections, samples GEO-NRM #19440118 (a) and #g19348 (b). Mineral symbols from Table 6. Adr = andradite; Aeg = aegirine.

Lead silicates found in the present study (L = Långban, P = Pajsberg)
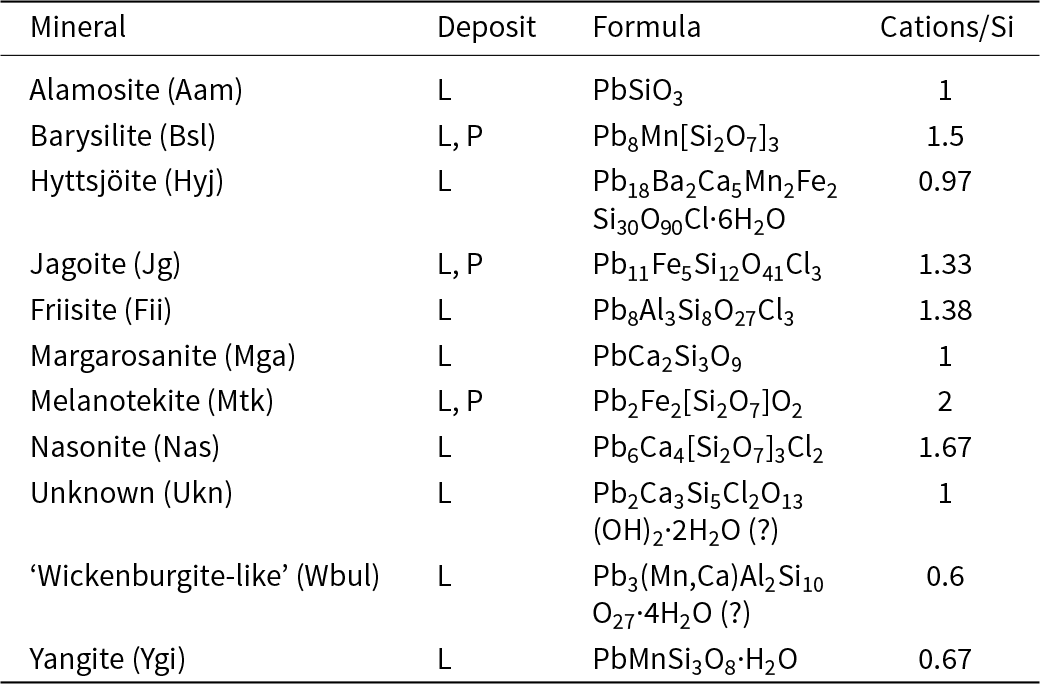
Notes: Mineral symbols are after Warr (Reference Warr2021).
Jagoite occurs in aggregates of greenish yellow subhedral crystals of a micaceous nature with a perfect {0001} cleavage, in portions of the skarn rich in melanotekite. It is often concentrated in strings that cross-cut the rock matrix but partly follow grain boundaries of pre-existing minerals. Jagoite is in most specimens partly surrounded by fine-grained alteration products, whitish and turbid under the binocular microscope, on fringes and along cleavage planes. In a single specimen (#19610234), a new Pb silicate mineral with an ideal formula Pb8Al3Si8O27Cl3 and similar in physical appearance to jagoite (but colourless), occurs as subhedral grains up to 300 µm in width. These occur in close association with melanotekite and jagoite; the full description of the new mineral, friisite (IMA2024-047; Holtstam et al., Reference Holtstam, Cámara and Karlsson2024), is in progress.
Långban is the second recorded locality of yangite (sample #19410179, Fig. 2a), which was previously only known from its type specimen (Kombat, Namibia; Downs et al., Reference Downs, Pinch, Thompson, Evans and Megaw2016). It occurs as elongated, colourless to slightly wine-yellow crystals in aggregates up to 1 mm. The refined triclinic unit-cell parameters of yangite (based on PXRD data) from Långban are a = 9.597(2) Å, b = 7.281(2) Å, c = 7.968(2) Å, α = 106.03(1)°, β = 118.14(1)°, γ = 109.85(1)° and V = 392.7(1) Å3, in good agreement with original data (Downs et al., Reference Downs, Pinch, Thompson, Evans and Megaw2016). A mineral similar in composition to wickenburgite (Pb3CaAl2Si10O27·4H2O, Williams, Reference Williams1968; possibly a Mn analogue) is found as thin flaky crystals associated with jagoite and alamosite (Fig. 2b).
BSE images of polished sections, samples GEO-NRM #19410179 (a) and #19610234 (b). Mineral symbols from Table 6. Di = diopside; Pb = native lead.
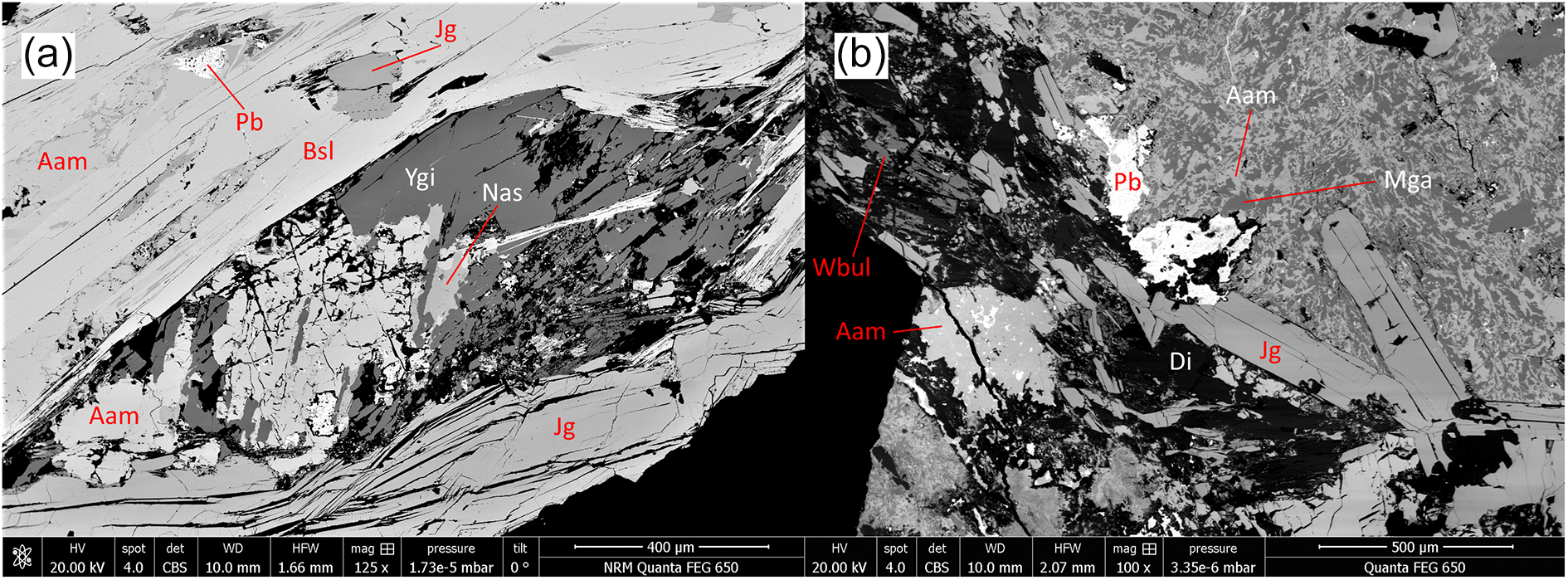
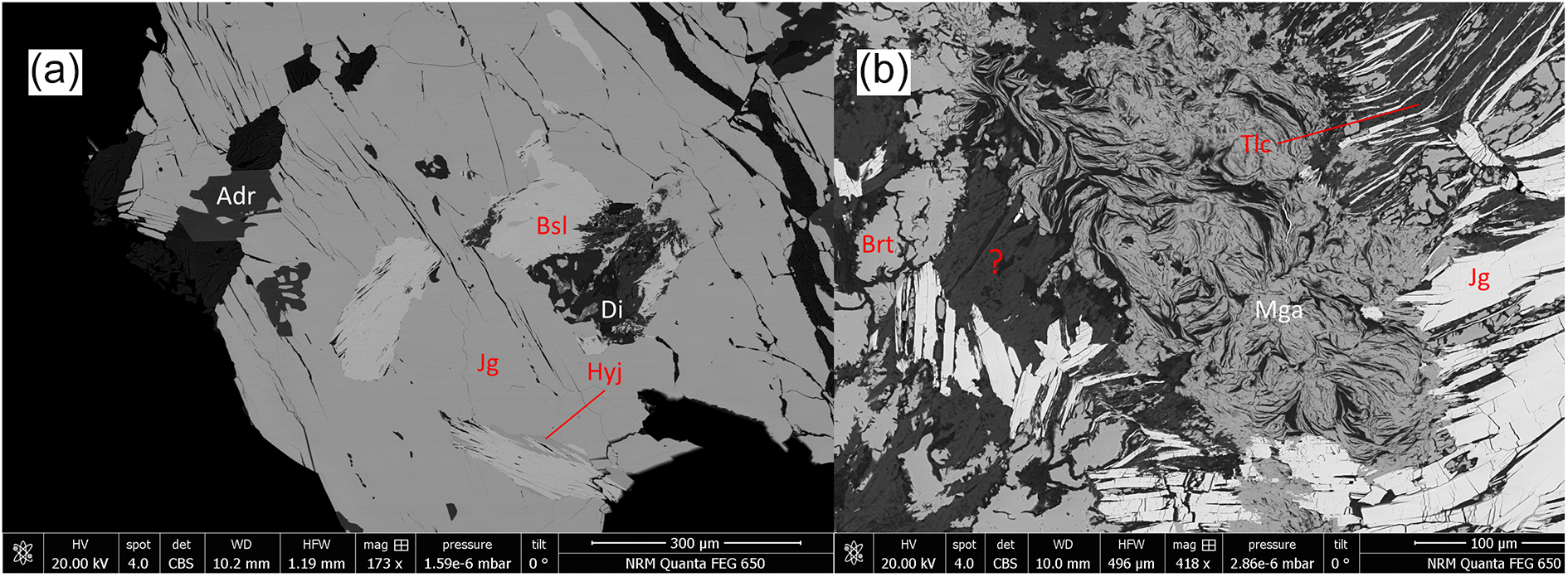
Margarosanite commonly occurs as a single phase in the alteration zones surrounding jagoite (Fig. 3a), but in some cases also in a eutectoid texture, as intergrowths with alamosite (Fig. 2b), possibly representing remnants of another altered lead silicate. An unknown Pb–Ca–Cl–O–H silicate (described in more detail below) formed during jagoite alteration, has also been observed in several samples (Fig. 3b).
BSE images of polished sections, samples GEO-NRM #19440121 (a) and #19440122 (b) Mineral symbols from Table 6. Adr = andradite; Di = diopside; Srp = ‘serpentine’.

BSE images of polished sections, samples GEO-NRM #19410178 (a) and #19440122 (b). Mineral symbols from Table 6. Adr = andradite; Brt = baryte; Di = diopside; Tlc = talc. The question mark represents an unknown phase with composition close to orlymanite.

Other minerals occurring in minor or trace amounts in the sample population are: baryte, calcite, native lead, fluorapophyllite-(K), talc, ‘serpentine’, albite, orlymanite (?) fluorapatite and phosphohedyphane. Minor amounts of additional late-stage alteration phases occur in some specimens, usually comprising very fine-grained material that contains Ca, Mg, Mn, Si ± Al, Pb, Fe and H2O, but so far are not possible to characterise properly.
On the label of specimen #g19348, it is written ‘Pajsberg’, which probably means the Stora Pajsberg mine (59°47.04′N; 14°19.03′E), belonging to the Pajsberg Fe–Mn ore field (Lee, Reference Lee1958; Holtstam et al., Reference Holtstam, Cámara, Skogby, Karlsson and Langhof2019). The sample comprises a dense skarn assemblage with diopside, andradite, melanotekite, barysilite, jagoite and minor calcite. In contrast to the Långban specimens, no alteration of jagoite was observed in this case (Fig. 1b).
The unknown Pb–Ca–Cl silicate
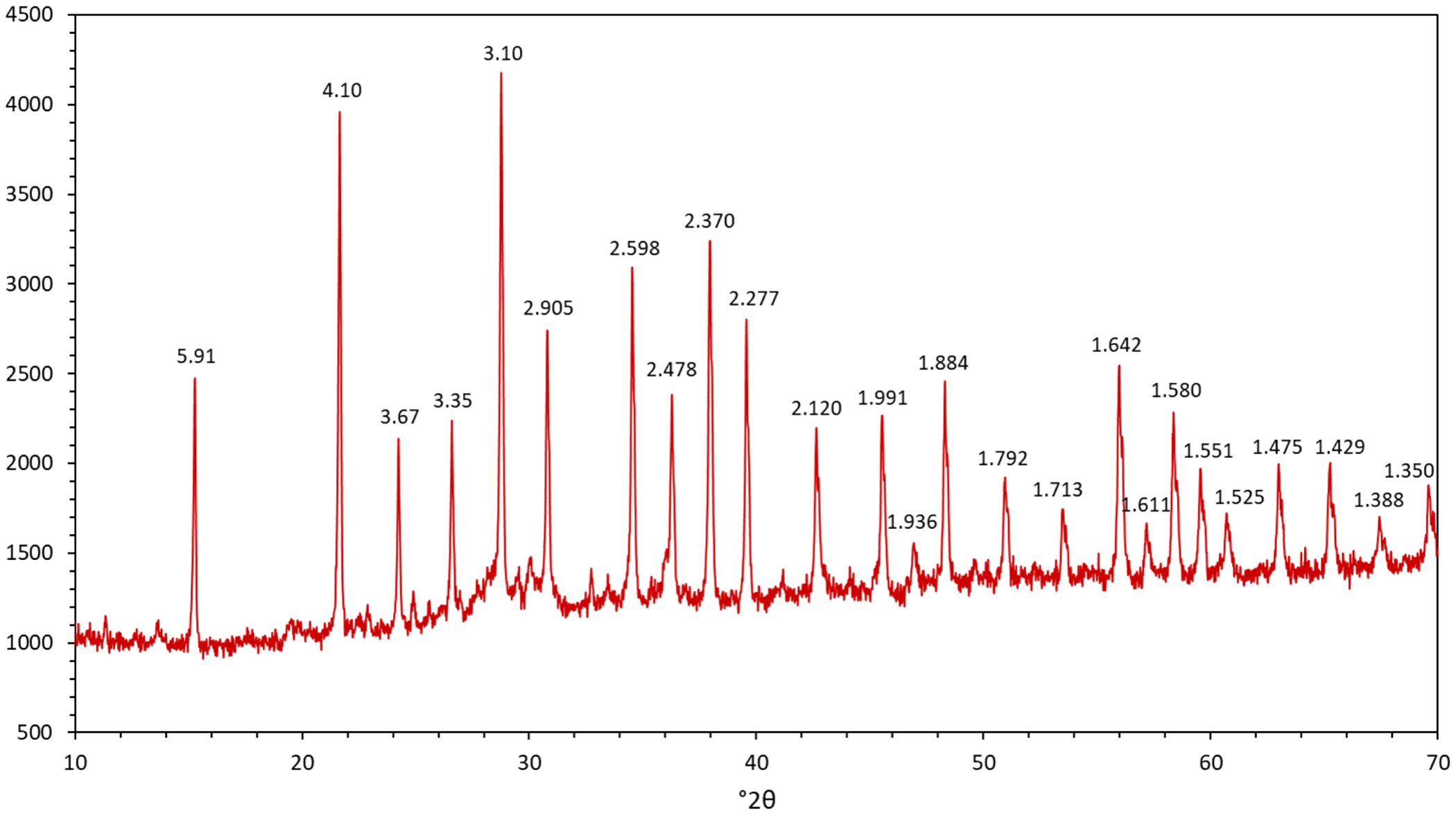
As mentioned, an unknown mineral substance is observed as an abundant alteration product of jagoite in some samples. It has so far resisted our attempts to determine its crystal structure, using both X-ray and electron diffraction methods, despite the fact that is has a unique and reproducible powder X-ray diffraction pattern (Fig. 5). A brief description, which may facilitate recognition of the phase and possibly lead to a full description in the future, is included here. It appears in aggregates with a felt-like, fine-grained texture. The colour is greyish white to beige. The mineral shows a distinct bright yellow fluorescence colour under SW UV light; associated margarosanite and calcite are light blue and orange, respectively, under the same conditions.
Powder-X-ray diffraction profile of the unknown Pb–Ca–Cl-silicate, sample GEO-NRM #19440122 with d values (in Å) of the Bragg peaks indicated. CuKα-radiation data.

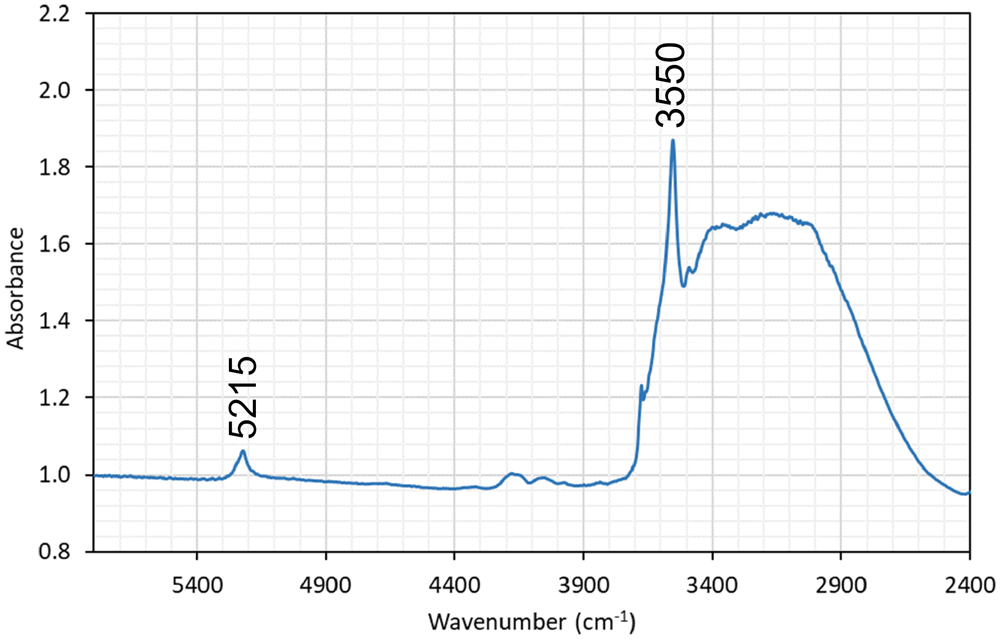
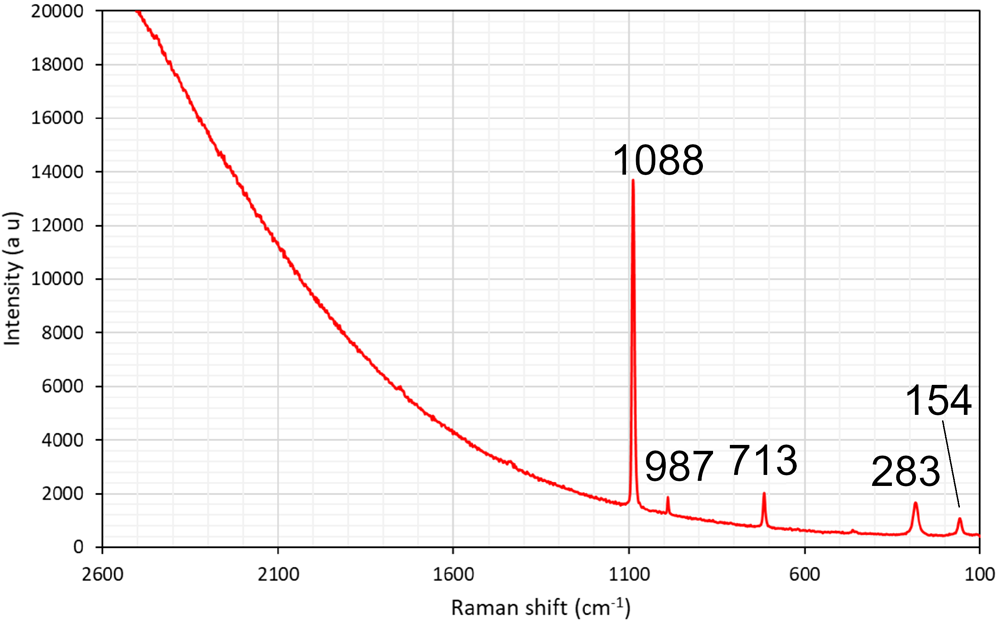
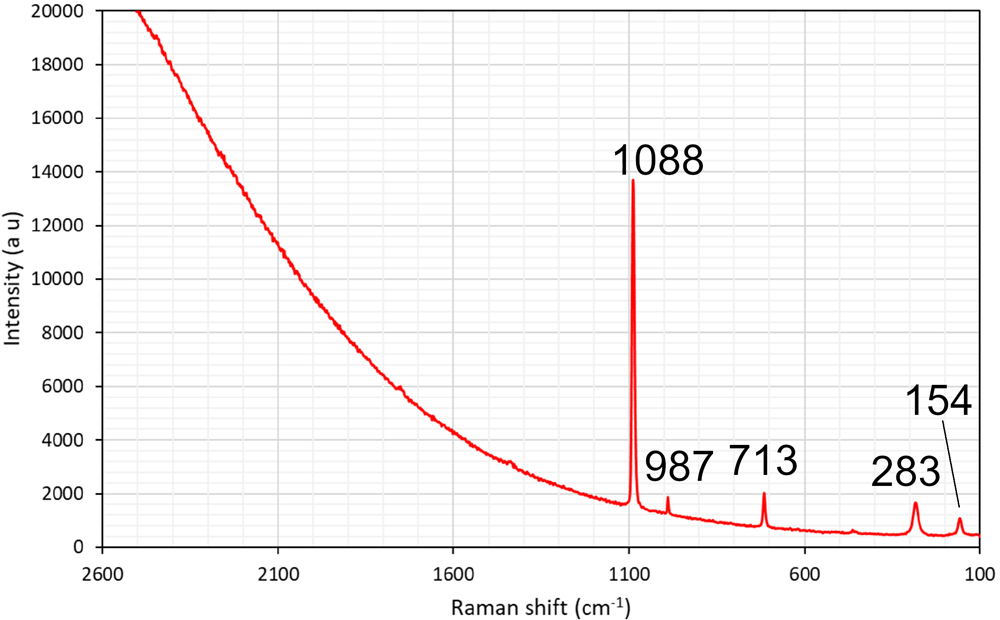
The formula can at this point only be approximately determined, to circa Pb2Ca3Si5Cl2O13(OH)2·nH2O. The composition is based on EDS point analyses, giving SiO2 28.0, MnO 2.3, CaO 14.8, PbO 44.4, Cl 6.7 (wt.%), and infrared (IR) spectroscopy. The presence of H2O and OH– is indicated in the IR spectrum (Fig. 6) from the combination mode at circa 5200 cm–1 and the narrow band at 3550 cm–1 related to O–H stretching vibrations, respectively. Laser-Raman spectra exhibit a very broad signal centred at ∼3600 cm–1 (possibly related to fluorescence) but also distinct bands at 1088, 987, 713, 283 and 154 cm–1 (Fig. 7).
Unpolarised IR spectrum of the unknown Pb–Ca–Cl-silicate obtained on an ∼100 µm thick aggregate. Sample GEO-NRM #19884399.

Laser-Raman spectrum of the unknown Pb–Ca–Cl silicate obtained with a 514 nm laser. Sample GEO-NRM #19440122.
Physical properties of jagoite
Micro-indentation hardness measurements on randomly orientated grains, obtained by means of a Shimadzu type-M tester for a 15 s indentation time, gave VHN 100 = 495 (=4.85 GPa) based on eleven measurements in a total range of 388–558. The shape of indentations is straight to slightly concave, accompanied by star radial fractures (in the terminology of Jambor and Vaughan, Reference Jambor and Vaughan1990). The microhardness obtained corresponds to a value intermediate to 4 and 5 on the Mohs scale (Broz et al., Reference Broz, Cook and Whitney2006), which is significantly higher than H = 3 reported by Blix et al. (Reference Blix, Gabrielson and Wickman1957). The calculated density, 5.73(1) g·cm–3, based on single-crystal unit-cell data and an empirical formula of the type specimen (see below), is also higher than the measured value given by Blix et al. (Reference Blix, Gabrielson and Wickman1957), 5.43 g·cm–3.
Crystal structure of jagoite
Refinement of the crystal parameters from single-crystal X-ray diffraction data of the jagoite type specimen [R 1 = 1.2% for 2089 reflections with F o > 4σ(F o)] gave a = 8.53926(5), c = 33.3399(2) Å with Z = 12 and systematic absences compatible with a P ![]() $\bar 6$2c unit cell, in good agreement with previous results. The structure model by Mellini and Merlino (Reference Mellini and Merlino1981) is corroborated, but our data are a high-resolution dataset obtained from a thin plate. This allows for adequate absorption correction, anisotropic atomic displacement factors and thus a considerable improvement in the accuracy of bond distances and bond angles.
$\bar 6$2c unit cell, in good agreement with previous results. The structure model by Mellini and Merlino (Reference Mellini and Merlino1981) is corroborated, but our data are a high-resolution dataset obtained from a thin plate. This allows for adequate absorption correction, anisotropic atomic displacement factors and thus a considerable improvement in the accuracy of bond distances and bond angles.
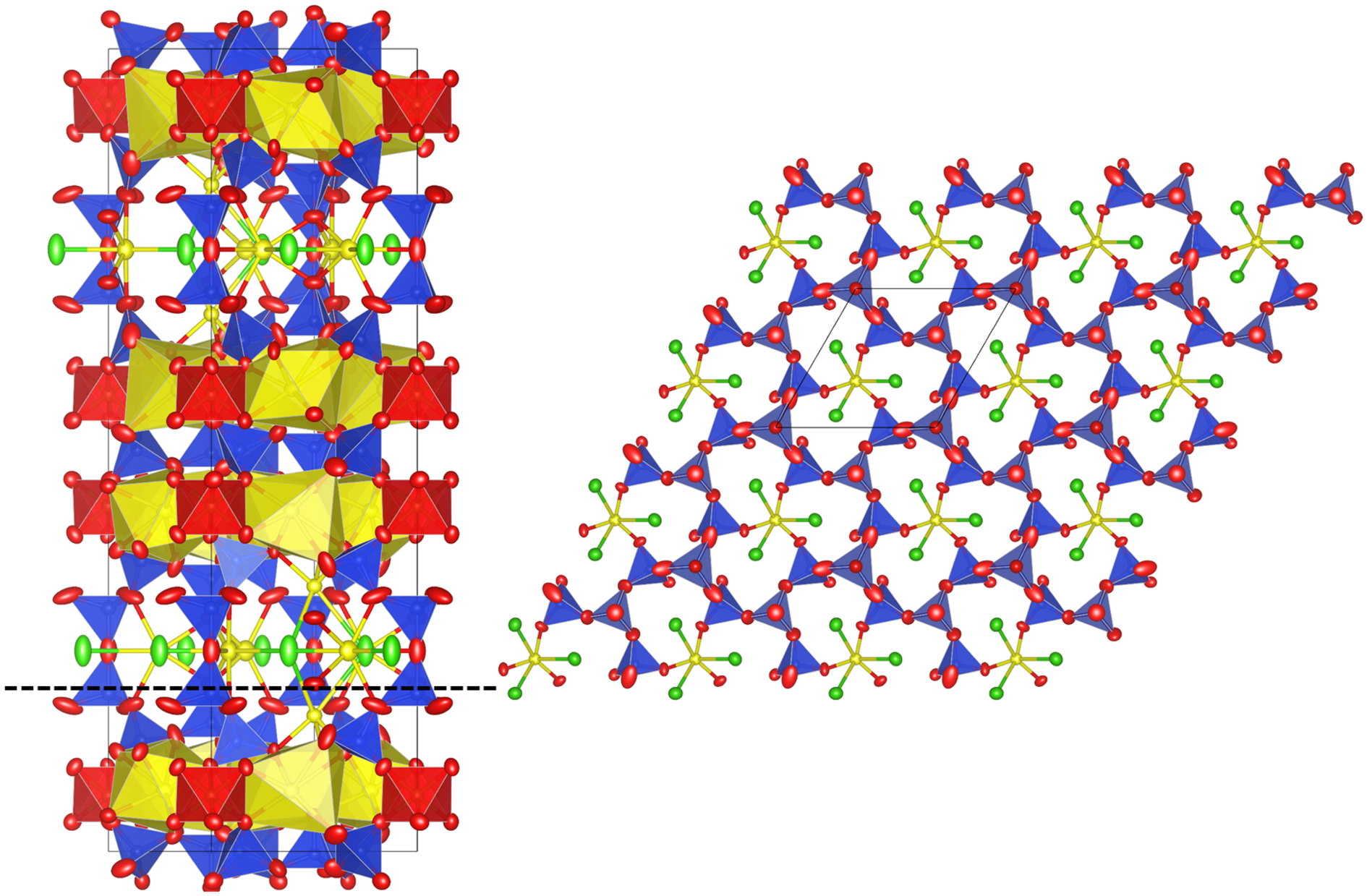
Jagoite has a single and a double layer of SiO4 tetrahedra (Fig. 8), like reyerite [(Na,K)2Ca14[Al2Si22O58](OH)8(H2O)6] (Merlino, Reference Merlino1988) and minehillite [K2Ca28Al4[Zn5□Si40O112](OH)16] (Dai et al., Reference Dai, Post and Appleman1995). The single-layer sheet (Fig. 9) is based on the (122)3(123)2 net (Hawthorne et al., Reference Hawthorne, Uvarova and Sokolova2019) that also occurs in zeophyllite (Merlino, Reference Merlino1972) and asbecasite (Sacerdoti et al., Reference Sacerdoti, Parodi, Mottana, Maras and Della Ventura1993). In jagoite, the single layer is linked on both sides to a (FeO6–PbO7)-layer. The FeO6 octahedra occur at the origin in the single layer. The double layer (Fig. 8, left side) hosts Pb (=Pb3) and Cl atoms that conform to a PbΦ8 layer (Φ = O, Cl) of edge-sharing distorted hexahedra. The Pb1 atom is 6-coordinated (3 × O, 3 × Cl), and Pb2 is 7-coordinated (7 × O). Jagoite is also related to wickenburgite [Pb3CaAl2Si10O27·4H2O], as in its double layer, one of 12 [(Al, Si)O4] tetrahedra is replaced by a [O3Ca(H2O)3] octahedron (Hesse et al., Reference Hesse, Liebau and Eulert2003).
The jagoite crystal structure seen along [010] (left) and [001] at z = 0.20 (right). Symbols: Si tetrahedra = blue. Fe octahedra = red. Pb atoms and polyhedra = yellow. Cl atoms = green. Oxygen atoms = red ellipsoids. Crystal-structure drawings made by VESTA 3 (Momma and Izumi, Reference Momma and Izumi2011).
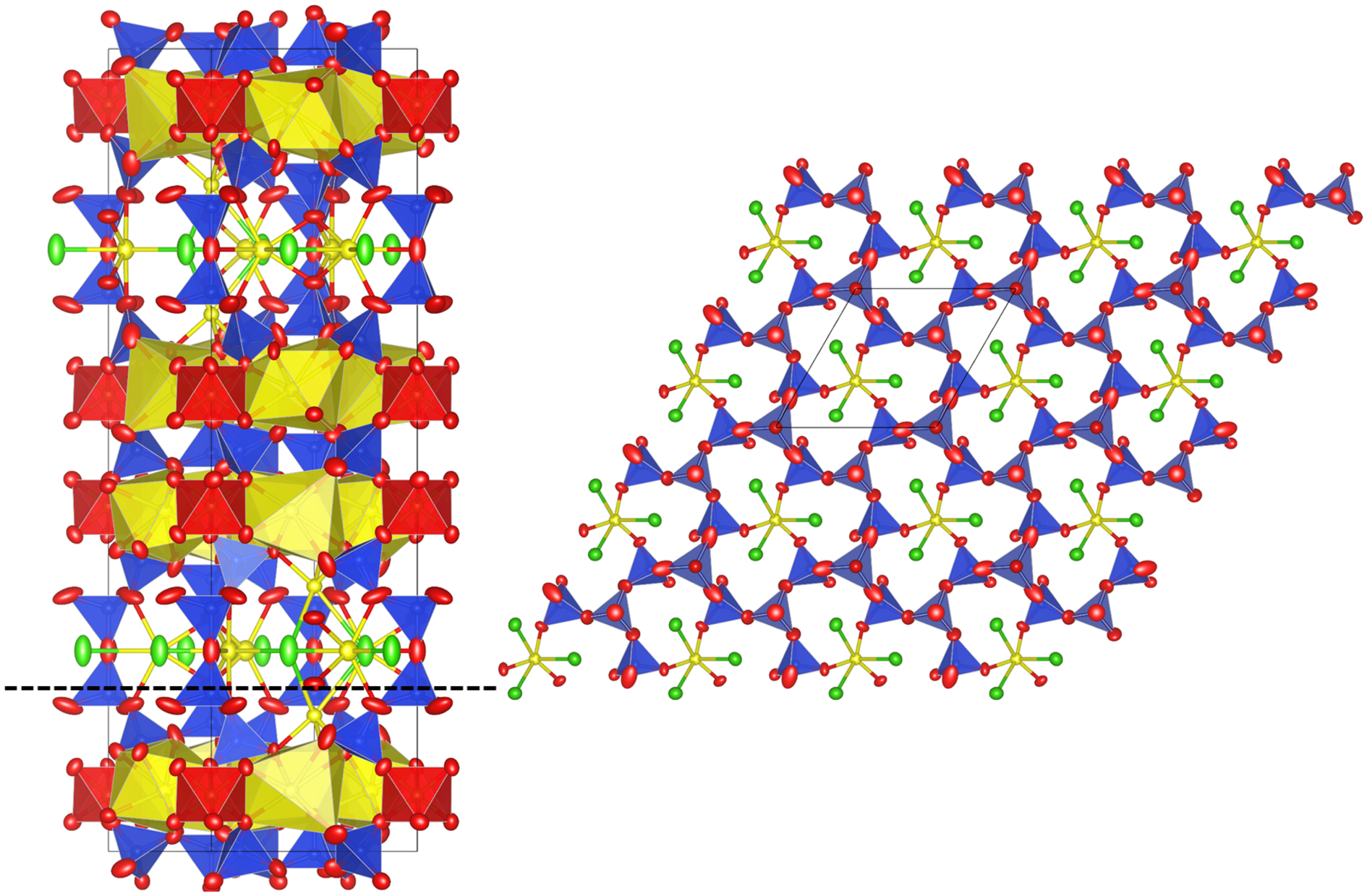
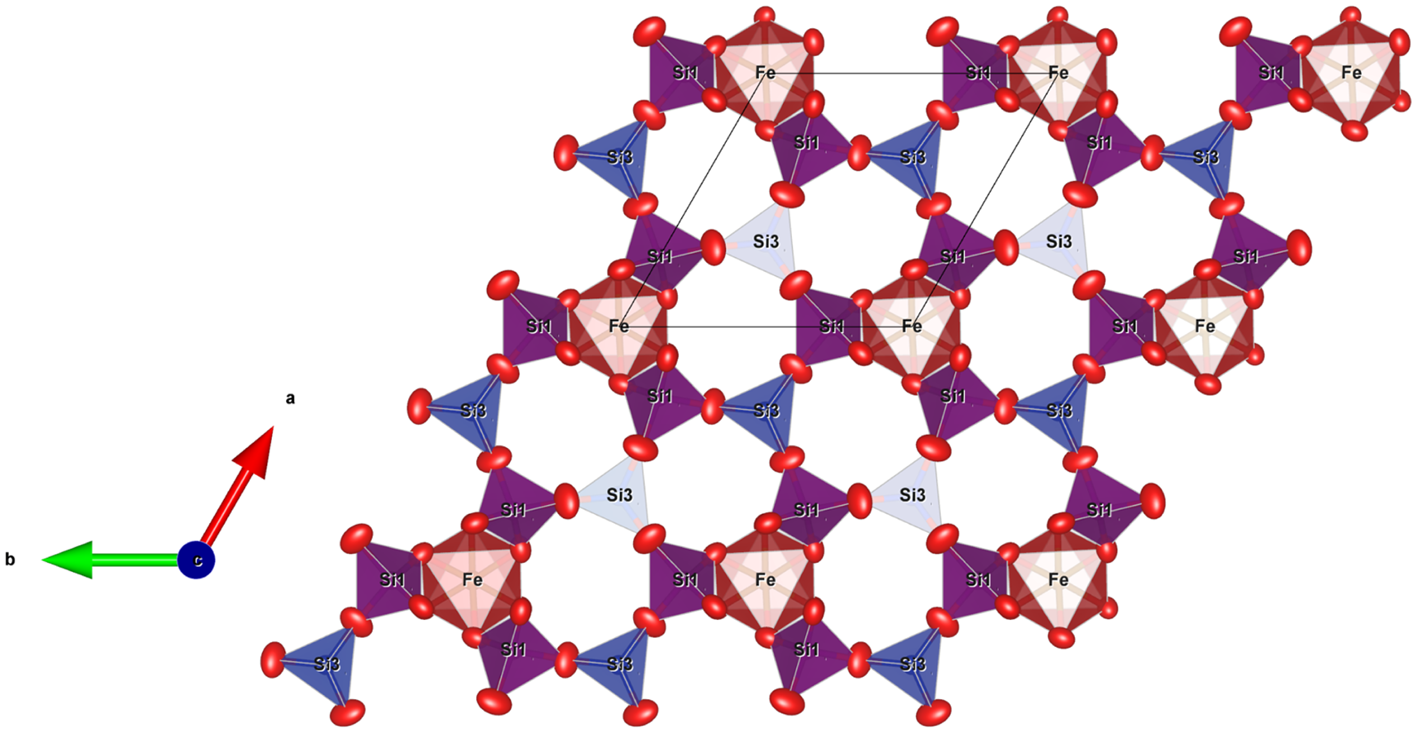
The jagoite crystal structure at z ≈ 0 seen along [001]. The Si1 tetrahedra with purple colour have a mixed Fe3+ + Si occupancy.

Two out of five of the tetrahedra in jagoite, Si1 and Si2, have mixed Si:Fe occupancies. Tetrahedron Si2 is the one connecting the two layers of tetrahedra of the double layer. Tetrahedron Si1 alternates with Si3 (occupied by only Si) in the single layer, with each Si3 surrounded by 3 Si1 tetrahedra. Each Si1O4 tetrahedron is vertex-sharing with two FeO6 octahedra.
Mineral compositions
The original publication by Blix et al. (Reference Blix, Gabrielson and Wickman1957) showed (in wt.%) SiO2 22.35, BeO 0.12, Al2O3 0.50, Fe2O3 7.00 TiO2 0.10, MgO 0.60, MnO 0.88, CaO 0.65, PbO 64.26, Na2O 0.61, K2O 0.37, Cl 3.25, H2O 0.36, O ≡ Cl –0.73, total 100.32, from a wet-chemical analysis. Grew et al. (Reference Grew, Peacor, Rouse, Yates, Su and Marquez1996) reported a single electron-microprobe analysis (WDS) of jagoite from Långban with (in wt.%) SiO2 22.11, Fe2O3 6.43, MnO 0.94, CaO 0.40, PbO 68.00, Cl 3.22, i.e. with similar values as presented here for the type specimen (Table 1).
The LA-ICP-MS data on two samples (Table 2) show that jagoite is poor in most trace elements. Compared to the continental-crust average, however, Be, Sb and Bi are abundant. The enrichment of these elements is characteristic of Långban-type deposits (Holtstam and Mansfeld, Reference Holtstam and Mansfeld2001; Hålenius et al., Reference Hålenius, Bosi and Gatedal2013). A few elements show larger variations in the two samples analysed, Al (383–2361 ppm), Mg (420–2332 ppm), Ti (5–314 ppm), Ba (6–289 ppm) and Ga (2–34 ppm). The mineral is poor in most first-row transition metals, except Fe, Mn and Zn. In addition to Ca and Na, jagoite is not particularly enriched in large-ion alkali or alkaline earth metals (K, Rb, Cs, Sr and Ba) that could substitute for Pb2+ in the crystal structure. Rare earth elements, as indicated by very low concentrations of Y and Ce, are not abundant either. The halogen content is relatively constant; sample 19440122 contains 2.63–2.75 wt.% Cl and 534–596 ppm Br, and the jagoite holotype specimen 19410178 yields similar results, 2.69–2.80 wt.% Cl and 447–560 ppm Br. Notably, Cl concentrations obtained by LA-ICP-MS are lower than those obtained by EMPA, but used for the formula calculation because they are considered more reliable (a slight destruction of the surface of the tugtupite reference was noted during the analytical session at the EMPA).
From the compositional information presented here, some shortcomings of the original wet-chemical data of the type material become clear. In particular, concentrations of BeO, MgO, Na2O, K2O and H2O as determined by Blix et al. (Reference Blix, Gabrielson and Wickman1957) were too high and should not affect the present empirical formula to the extent that has been suggested before (Mellini and Merlino, Reference Mellini and Merlino1981). For instance, the results of the present work show the absence of hydrogen (see Raman spectroscopy results) and therefore, OH groups have not been considered a defining element of the ideal formula.
From the combined electron-microprobe and LA-ICP-MS data, the following empirical formula based on 28 cations is obtained:
Pb10.94Na0.13Ca0.21Fe3⁺3.18Sb0.05Zn0.18Mn3+0.48Al0.10Mg0.14
Si12.54Be0.05O41.12Cl2.86Br0.02.
Mineral spectroscopy
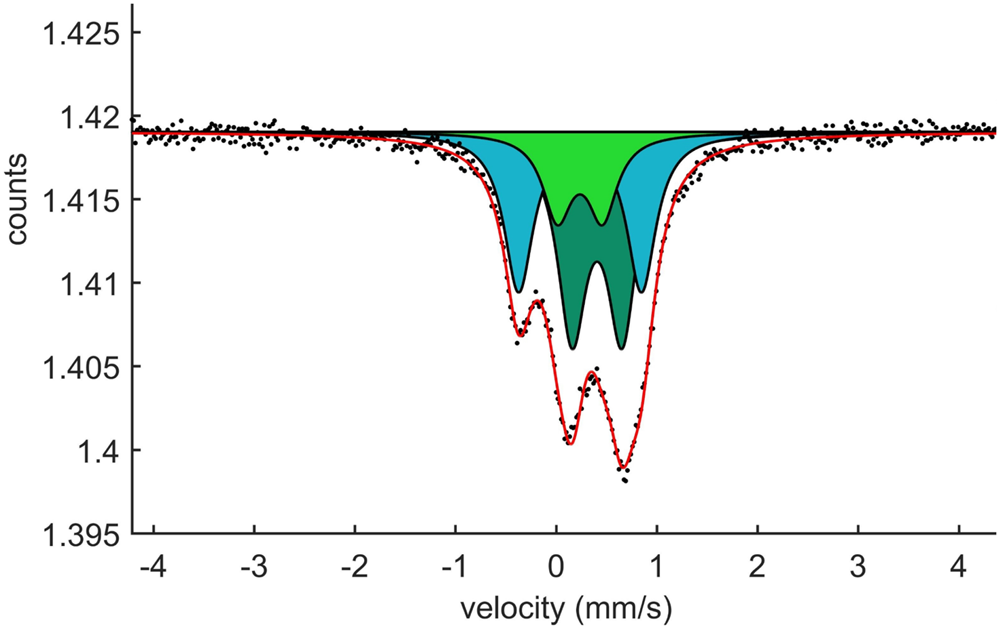
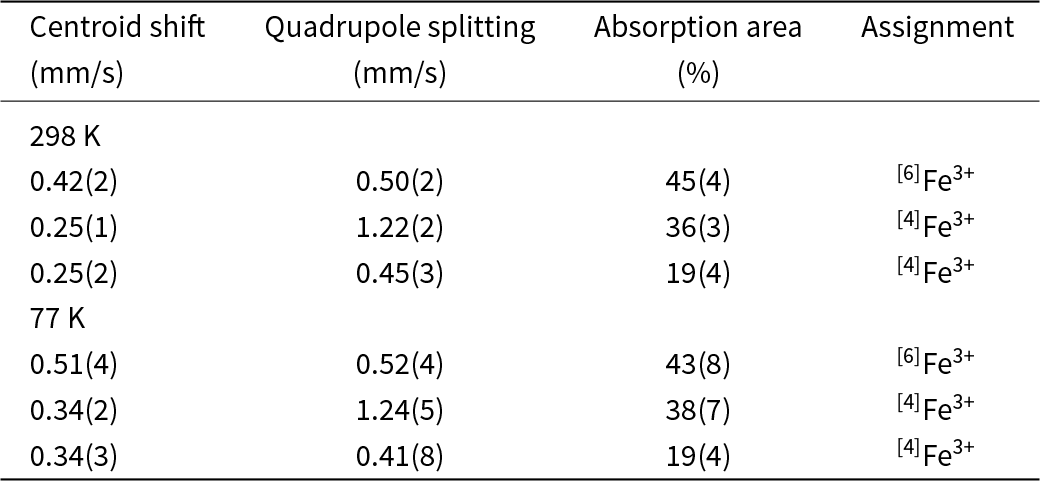
In the Mössbauer spectrum (Fig. 10) obtained at ambient temperature, the best fit (χ2 = 1.1 in the last cycle of the fitting procedure) with physically reasonable parameters is obtained with a three-doublet model. The hyperfine parameters determined are given in Table 7. Centroid shifts are consistent with Fe atoms being present only in the trivalent state (high spin), and distributed over both 6-coordinated and 4-coordinated sites. The higher quadrupole-splitting value associated with one of the [4]Fe doublets is related to the higher degree of distortion of the corresponding coordination polyhedron, thus probably Si1O4 (distortion index 0.0159; Table 5). The Mössbauer pattern obtained at 77 K is very similar to the room-temperature spectrum (except for the expected increase in the centroid shift with ∼0.1 mm/s, due to the second-order Doppler shift); no magnetic coupling or major phase transition thus occurs at low temperature.
57Fe Mössbauer spectrum of jagoite at room temperature. Counts given on the y axis are ×107. Sample GEO-NRM #19410178.

Mössbauer data for jagoite relative α-Fe
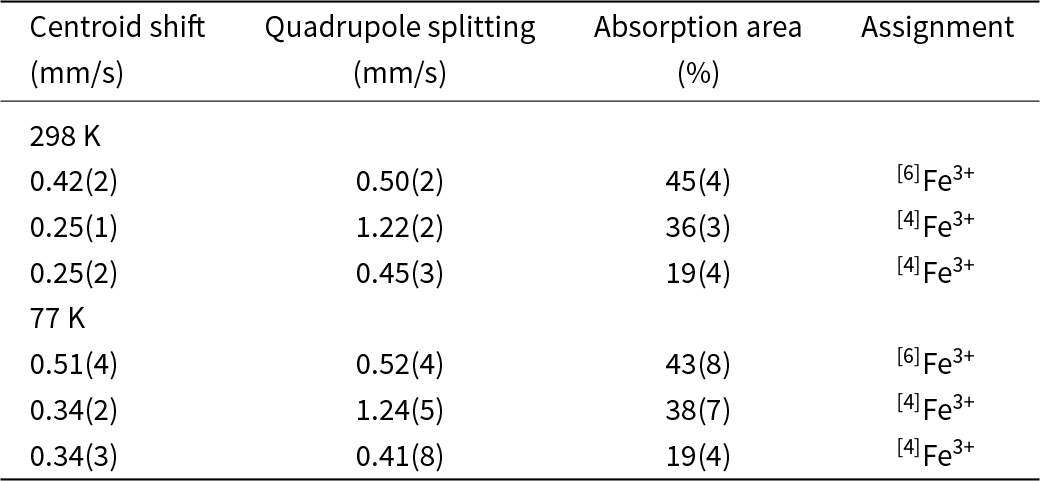
Notes: Errors given as 2σ.
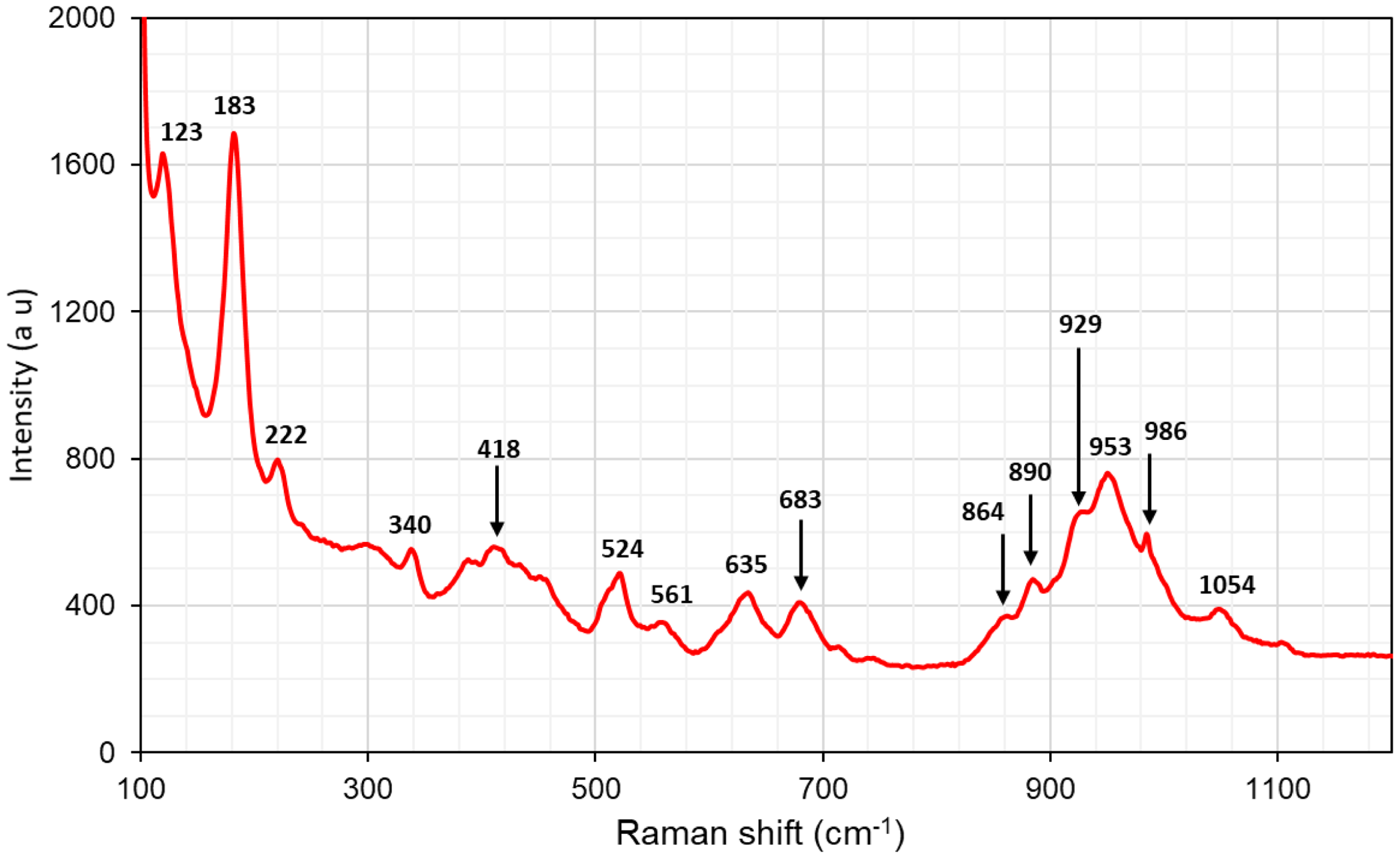
In the Raman spectrum (Fig. 11), a set of multiple bands in the region 860–1050 cm–1 are related to symmetric and antisymmetric stretching modes of Si–O bonds, whereas one at 680 cm–1 corresponds to the bending of Si–O–Si bridges. A distinct peak at 522 cm–1 is tentatively assigned to vibrations in the FeO6 octahedra, whereas tetrahedrally coordinated Fe3+ is recognisable by a band around 630 cm–1. The strongest band, in the low-wavenumber region at 185 cm–1, is possibly related to Cl–Pb–Cl bending modes (Frost and Williams, Reference Frost and Williams2004). The Raman spectrum is featureless from 1200 to 4000 cm–1, and thus no signals were detected in the OH-vibration region, indicating an essentially anhydrous composition of jagoite.
Raman spectrum of jagoite obtained with a 514 nm laser. Sample GEO-NRM #19410178.

Discussion
Paragenesis
From textural relations, the main mineral components of the skarn (melanotekite, barysilite, andradite, hematite, quartz and diopside) can be inferred to be primary (associated with peak metamorphism). At some point, chlorine has been mobilised in the system; Whether it was available already in the primary mineralisation (Magnusson, Reference Magnusson1930) or has been added in a later episode of geological evolution in the Långban area (Jonsson, Reference Jonsson2003) has not been settled. The formation of jagoite and nasonite occurred at the expense of the pre-existing Pb minerals (for symbols see list in Table 6). Jagoite may be obtained directly from melanotekite and quartz via:
11 Pb2Fe2Si2O9 + 2 SiO2 + 6 HCl(aq)
Mtk
→ 2 Pb11Fe5Si12O41Cl3 + 6 Fe2O3 + 3 H2O
Jg
Jagoite is a unique mineral in the PbO–Fe2O3–SiO2–Cl2 system. Hematophanite, Pb4Fe3O8Cl (without silica), probably forms under similar P–T conditions as jagoite but in a silica-undersaturated Pb–Fe-rich system (Holtstam et al., Reference Holtstam, Norrestam and Sjödin1995).
There is no sharp boundary between the metamorphic assemblage and the later recrystallisation products, as euhedral, poikiloblastic andradite may contain jagoite inclusions (Fig. 3a). This garnet growth could possibly be connected to a second temperature climax at Långban as inferred by Magnusson (Reference Magnusson1930). Grew et al. (Reference Grew, Yates, Belakovskiy, Rouse, Su and Marquez1994) also reported a second generation of andradite in Pb-bearing Mn-skarn from Långban.
Some barysilite has been converted to alamosite by the reaction:
Pb8Mn[Si2O7]3 + 4 SiO2 + H2O
Bsl→7 PbSiO3 + PbMnSi3O8·H2O,
Aam Ygi
which is supported by the fact that alamosite is more common than yangite in these assemblages.
The margarosanite–alamosite mixture noted in some samples could have formed by breakdown of ganomalite–wayneburnhamite (ganomalite is considered an early-formed skarn product in Långban; Magnusson, Reference Magnusson1930) with the addition of silica:
Pb9Ca6Si9O33 + 6 SiO2 → 3 PbCa2Si3O9 + 6 PbSiO3
Gnm Mga Aam
Hydrothermal alteration of the early lead silicates involving fluids bearing Ca2+ and SiO2 (aq) has probably produced late-stage minerals. Our observations suggest alteration of jagoite to margarosanite according to a reaction like (Fig. 3a):
Pb11Fe5Si12O41Cl3 + 22 Ca2+ + 21 SiO2 + 16 H2O
Jg
→ 11 PbCa2Si3O9 + 5 Fe3+ + 3 Cl– + 32 H+
Mga
The unknown Pb–Ca–Cl–O–H silicate may have formed by a similar kind of reaction with the addition of Cl (see Fig. 3b):
2 Pb11Fe5Si12O41Cl3 + 33 Ca2+ + 16 Cl– + 31 SiO2 (aq) + 43 H2O
Jg
→ 11 Pb2Ca3Si5Cl2(OH)2·2H2O + 10 Fe3+ + 20 H+
Ukn
The last two reactions are pH-dependent and rely on the fact that H+ is consumed in the carbonate-rich environment. Calcium and SiO2 have probably been released and made available for further reactions by the breakdown of diopside, forming serpentine or talc as additional products.
Hyttsjöite and the wickenburgite-like mineral may also be the result of hydrothermal alteration of jagoite (or the friisite mentioned). In the reactions suggested above, Pb is conserved in newly formed silicate minerals; the mechanisms of precipitation of native lead are not fully understood, (i.e. if it is related to breakdown of Pb silicate, or not).
During the transformation of the lead silicates, the molar ratio of metals (Pb, Ca, Mn, Fe and Al) to silicon in the products (Table 6) progressively diminish, with values ≤1 obtained for the latest alteration stage. Incorporation of structural H2O is also a characteristic feature of some of the alteration products, indicating lower temperatures of crystallisation, probably <300°C, at this stage.
Crystal chemistry
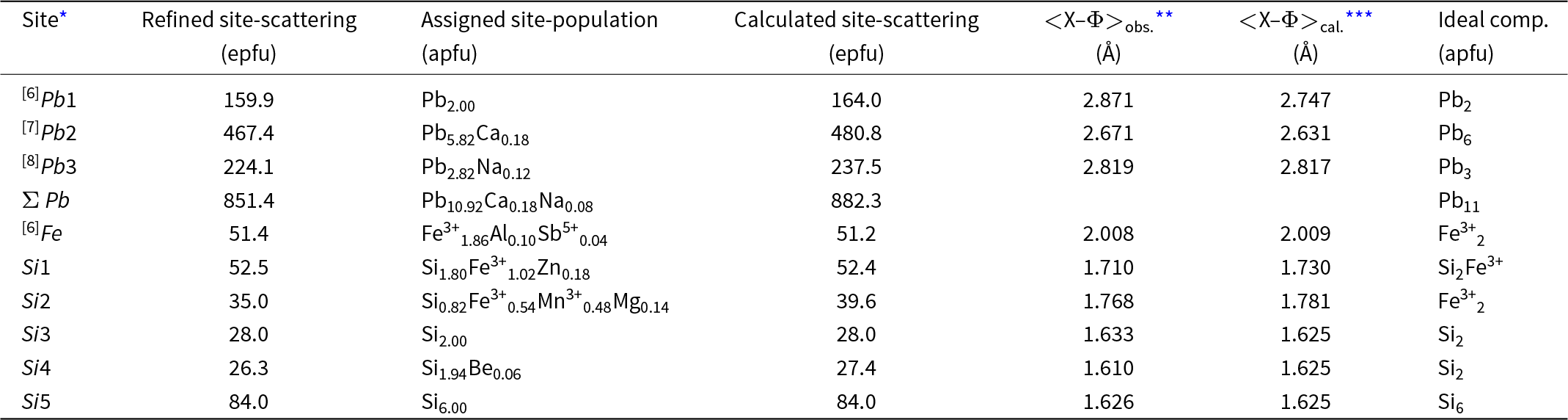
Taking into account the refined site-scattering values (Table 4) and the polyhedron dimensions (Table 5), the best agreement between site-occupancy and average bond distances is found with the following crystal-chemical formula for jagoite:
[6]Pb2[7](Pb5.82Ca0.18)Σ6[8](Pb2.88Na0.12)Σ3[6][Fe3+1.86Al0.10Sb5+0.04]Σ2[[4](Si3.80Fe3+1.02Zn0.18)O14][[4](Si8.78Fe3+0.54Mn3+0.48Mg0.14Be0.06)O27](Cl2.86Br0.02O0.12)
Differences between observed site-scattering values and the ones calculated from the above crystal-chemical formula are reported in Table 8.
Refined site-scattering values and assigned site-populations for jagoite (apfu = atoms per formula unit; epfu = electrons per formula unit)
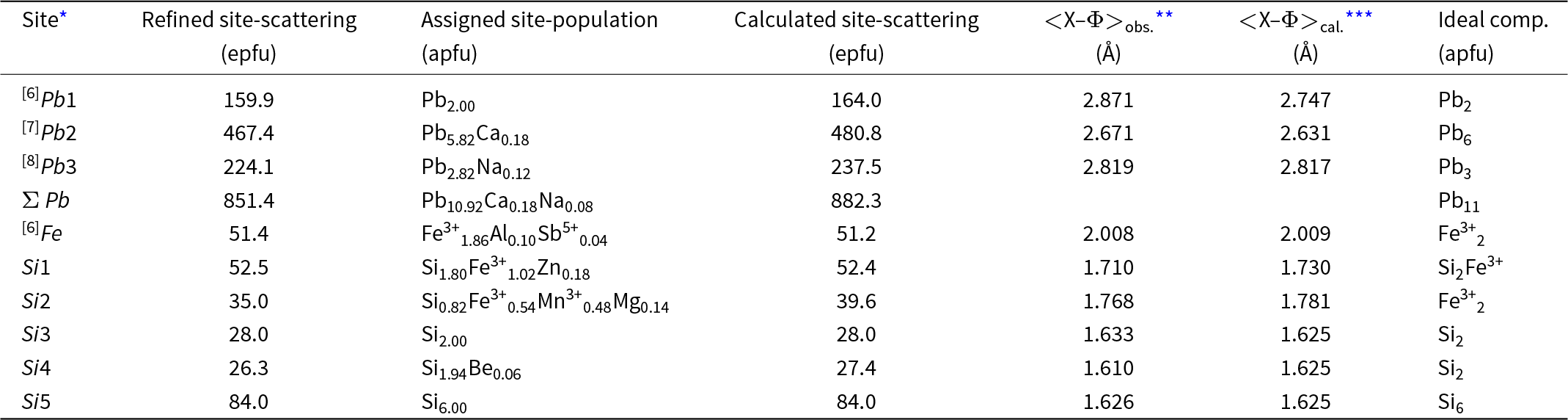
* Coordination numbers are given for non-[4]-coordinated sites;
** Φ = O, Cl;
*** Calculated using ionic radii taken from Hawthorne and Gagné (Reference Hawthorne and Gagné2024).
The assigned formula shows an 11% disagreement with the observed scattering at the Si2 site, which is rather high. However, the agreement is very good between observed and calculated mean bond distances, confirming the assignment of Mg to this site. The Si2 site is the largest among the fourfold coordinated sites and is the tetrahedron connecting the two layers of tetrahedra of the double layer. The distortion index is particularly low (0.0011, the lowest among all the tetrahedra in the structure; Table 5) and excludes the possibility of allotting Zn to this site, as this element prefers a more distorted four-fold coordinated environment (for example, in willemite, Klaska et al., Reference Klaska, Eck and Pohl1978). In addition, assigning Zn to this site would increase the disagreement between observed and refined site-scattering values. Conversely, the Si1 site shows the highest distortion parameter among tetrahedra (0.0159; Table 5) and it is the other tetrahedron hosting Fe3+. This site is also better suited to host Zn. Then the disagreement with observed site-scattering is very low (<1%), and the agreement is rather good between observed and calculated bond lengths, confirming the assignment of Zn and Fe3+ to this site. The higher polyhedron distortion is also in accord with the sharing of the bridging O4 anion site. This site is four-fold coordinated with one short and one long bond to the Pb2 site and two Si at the Si1 (Si2Fe3+) and Si3 (pure Si) sites, with a nominal charge incidence of 2 × 2/8+0.917+1 = 2.417+. Yet the bond-valence value at this site is acceptable (1.98 valence units, vu, Table 4) because the Pb–O bonds are rather long and the individual Si–O4 bond distance is the longest for these tetrahedra (Table 5). Such a relaxation mechanism of the structure imposes a strong deformation of the Si1 tetrahedron. The occurrence of a divalent cation at this site (Zn) also helps to match the bond-valence incidence at the O4 site. The Si4 sites probably host some Be (from LA-ICP-MS analyses, Table 2), but this does not account for its small size (it shows the smallest volume among all tetrahedra; Table 5). The lower volume among tetrahedra is related to the short Si–O7 bond, owing to the short O7–O7 distance along [0001], which is 2.74 Å, the edge shared by three Pb3 eight-fold polyhedra and the shortest among all the Pb3 polyhedron edges.
Another issue is the oxidation state of Mn and its partitioning among the sites. The best match between observed bond lengths and site scattering is obtained when considering all Mn as Mn3+, ordered at the Si2 sites, because it has a shorter ionic radius than Mn2+ ([4]Mn3+–O = 1.901 vs. [4]Mn2+–O = 2.046 Å, Hawthorne and Gagné, Reference Hawthorne and Gagné2024). The site is still dominantly occupied by trivalent atoms, but the Fe3+ is almost half of the amount present at the Si1 site. This is in better agreement with Mössbauer data, showing an ∼2:1 relation between the two tetrahedrally coordinated Fe3+ sites. The absence of a perfect agreement is probably related to the fact that the weakest absorption doublet is not well resolved (Fig. 10). We believe that this is the best match between structural data and the Mössbauer spectrum and therefore a reasonable site assignment.
Charge balance requires some Cl–1O exchange at the Cl site. This replacement has been considered when calculating the bond-valence sums (BVS) reported in Table 5. In addition, the Pb1 site is six-fold coordinated with three at the O5 site and three Cl at the Cl1 site. A substitution allowing for vacancy, thus with less Cl at the Cl site, would further lower the bond valance incident at the Pb1 site. The introduction of some oxygen or bromine at this site helps to compensate for changes and bond valence incidence (as Br has a larger ionic radius than Cl). Overall, the calculated BVS are very good with only two oxygen sites slightly overbonded (BVS ∼2.15 vu) which is still acceptable.
The polyhedron dimensions and site scattering suggest that Al-for-Fe3+ substitution occurs mainly at the Fe octahedra whereas Mn is ordered as Mn3+ at the less distorted Si2 tetrahedra. Antimony is probably pentavalent, occupying the Fe octahedra only. Coupled substitutions are inferred to incorporate divalent ions (Mg, Zn) in the jagoite structure, with excess Si4+ (above 12 atoms per formula unit): 2 Fe3+ = Si4+ + M 2+. From the large volume of the Si2O4 tetrahedra (Table 5), it is inferred that trivalent species are mostly enriched there. The Si3 and Si5 sites are essentially filled with Si.
To define the ideal formula, we must consider charge balance and charge arrangements. On the one hand, the structure has 44 anion sites with a formal charge of –85 (41 O2– + 3 Cl–). On the other hand, the cation count of the structure is 28. Most of the site compositions correspond to almost fixed charges. The structure has 11 sites hosted by divalent Pb and 2 sites hosted by trivalent cations (Fe3+) with six-fold coordination. These 13 cations sites account for 28+ charges. The other 15 cation sites left are in tetrahedral coordination. If we assume that all are occupied by Si4+ they would count for 60+ charges, with an excess of 3+ charges relative to the anion part. Some of the Si positions need to have less than 4+ average charges, and that can be achieved by substituting 3 Si4+ with 3 trivalent cations (in this case, Fe3+). That leads us to the following charge arrangement for the ideal formula:
Pb1(2+)2Pb2(2+)6Pb3(2+)3M(3+)2 T[(4+)12(3+)3]O(2−)41Cl(1−)3
There are only significant heterovalent substitutions at two tetrahedral sites, Si1 (symmetry 6g) and Si2 (symmetry 4e), that account for 5 out of 15 sites with tetrahedral coordination. We could simply allocate all 3 Fe3+ at the Si1 site as it corresponds to 3 atoms pfu, thus obtaining a charge-balanced ideal formula. But it will be against structural evidence (observed mean bond length, polyhedral volume and site scattering; see Table 5 and Table 4, respectively) and local bond-valence requirements (in terms of bond valence; see Table 4). If we choose to substitute Si4+ by Fe3+ at the Si2 sites (2 apfu), it is not sufficient, and a further Fe3+ should be placed at the Si1 site. To make a choice, we should follow the dominant-valency and dominant-constituent rules in agreement with the definition of an ideal formula proposed by Hawthorne (Reference Hawthorne2002), which implies that we may have mixed occupancy (and valence) at one site only. We could think of a situation with mixed occupancy at both Si1 (SiFe3+2) and Si2 (SiFe3+), but it would be against the current definition of an ideal formula. The fact is that we need to have a mixed charge site for charge balance requirements, either Si1 or Si2. From a formal charge point of view, it does not make any difference. But from a local charge-balance (bond-valence) and symmetry constraints, it does. The choice of Si 2Fe3+ and Si 1(Si2Fe3+) seems the best suited. Let us analyse the situation from the point of view of assigned site populations, symmetry restraints and local bond-valence requirements.
The double layer in the jagoite structure has 10 T sites (2 Si2, 2 Si4 and 6 Si5). In that layer, only 2 T sites (Si2) out of 10 are occupied by large cations (Fe3+, Mn3+ and Mg) along with Si (see Table 8), which are those connecting the two layers. The heterogeneous composition (both in element and in charge) is reflected by the atomic displacement parameters (adp) of all the oxygen atoms bonded to the Si2 site, i.e. O6 and O9, which have the highest adp values among all the oxygen sites (Table 4). From data reported in Table 8, the dominant valence at the Si2 site is 3+ (0.54 Fe3+ + 0.48 Mn3+ = 1.02 > 0.82 Si4+ ≫ 0.14 Mg2+), and thus the dominant species of the dominant charge is Fe3+ (Fe3+ > Mn3+). Therefore, in the ideal formula, we should write 2 Fe3+ apfu. That makes the ideal composition of the double layer to [Fe3+2Si8O27]. In the single layer, one site (Si3) is completely occupied by Si, whereas the other one (Si1) is occupied by 2 Si and one Fe3+ (plus 0.18 apfu of Zn), making the layer composition to [Si4Fe3+O14]. With this choice, we obtain 3 Fe3+ at the tetrahedral sites in the ideal formula, thus fulfilling the charge arrangement required.
One could argue that we have not applied the dominant-valency rule for the Si1 site as we did for the Si2 site. But that would lead to 2 Fe3+ at the tetrahedral sites, imposing an excess of charge of 1+ in the charge balance of the ideal formula. Both Si1 and Si2 sites are in special positions: the Si1 site is at 6e and can only shift along [1000], the Si2 site is at 6g and can only shift along [1000]. However, the oxygen atoms coordinating these sites have different degrees of freedom. Both O2 and O4 atoms (coordinating Si1) occupy general positions, whereas among the ones coordinating the Si2 site, O6 is in a general position, but O9 is in a special position, 2b. In addition, O9 gets bond a valence-contribution also from the Pb3 sites that are in one special position (6h), whereas O2 and O4 sites get bond valence contribution from Pb2 sites, which occupy a general position (12i). The Pb2–O2 distance is the shortest in the Pb2 polyhedron, and this notably increases the bond valence incidence from Pb at the Pb2 site on the O2 site (0.44 vu calculated from the refined structural model). This compensates the required balance at O2, allowing it to achieve 1.96 vu (Table 4). This dictates the way that the structure can adapt locally to the chemical strain introduced by the Si–1Fe3+ heterovalent substitution. We have obtained excellent diffraction data and a very accurate determination of geometry that allowed us to produce a site assignment in agreement with observed site scattering and bond geometry (Table 8). These results show that the largest tetrahedral polyhedron is Si2, thus supporting our choice of full Fe3+ occupancy in the ideal formula. We cannot exclude a different site population for Si1 having more Fe3+ occupancy, but that must be balanced by the substitution of the trivalent Fe3+ by a small divalent cation at the M sites, like Mg. The introduction of Sb5+ at this site operates in the opposite way.
Mellini and Merlino (Reference Mellini and Merlino1981) suggested a scheme of ordering among cations (Si, Fe, Mn, Mg etc.) in the crystal structure that could lead to a different space-group symmetry; such a pattern was not confirmed with our data. We also solved the structure in space groups P31c and P321, obtaining models with the same topology and with split sites that are equivalent, i.e. no cation ordering was observed from refined site-scattering values nor observed sizes of polyhedra. Therefore, a reduction in space group symmetry was not justified.
Thus, taking into account all the above considerations, a structural charge-balanced formula for jagoite with coordination numbers indicated by superscripts would ideally be [3+3]Pb2[7]Pb6[8]Pb3[6]Fe2{[4][Si2Fe3+][4][Si2]}single{[4][Fe3+2][4][Si2][4][Si6]}doubleO41Cl3 (curly brackets enclosing the two layers of tetrahedra, single and double refer to layer widths of the tetrahedral layers), simplified to Pb11Fe5Si12O41Cl3 for Z = 2.
Conclusions
Jagoite belongs to a far more diverse mineral association than suggested by the original description (Blix et al., Reference Blix, Gabrielson and Wickman1957). Primary lead silicates formed in the skarn during regional metamorphism, mainly melanotekite and barysilite, are susceptible to alteration, with Cl–, SiO2, Ca2+ and H2O as the principal modifying agents. In the process, the relative Si contents of the products increase and reflect a higher degree of polymerisation of the SiO4 units, along with hydration at lower temperatures under alkaline conditions. We conclude that most of the Pb is retained in silicate minerals during alteration overall; however, the mechanism of formation of sporadic occurrences of native Pb in some samples remains enigmatic.
The structural topology of jagoite, as described by Mellini and Merlino (Reference Mellini and Merlino1981), is confirmed with considerably improved data for the structural parameters. We have shown that minor elements are also hosted by jagoite, in particular Al at the octahedrally coordinated Fe site and Mn3+, Zn and Mg at four-coordinated mixed Fe–Si sites. Jagoite crystals are also enriched in Be, Sb, Bi and Br, but these elements play a minor crystal-chemical role.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/mgm.2025.10108.
Acknowledgements
The authors express their delight at the opportunity to contribute to this volume of the Mineralogical Magazine in honour of the eminent scientist Ed Grew, who has shown a long-standing interest in exotic mineral deposits, including Långban. We are most thankful to Guest Associate Editor Robert F. Martin and Sergey Krivovichev, Aaron Lussier and one anonymous journal referee for valuable reviews and recommendations.
Financial statement
F.C. acknowledges financial support from the Italian Ministry of Education (MUR) through the project “Dipartimenti di Eccellenza 2023–2027”.
Competing interests
The authors declare none.