Introduction
Surveying nocturnal arboreal mammals presents a challenge for wildlife researchers. In the tropics, these species are often small and cryptic, spending the majority of their time high in the forest canopy or concealed within dense foliage (Kays et al. Reference Kays, Sheppard, Mclean, Welch, Paunescu, Wang, Kravit and Crofoot2019; Moore et al. Reference Moore, Pine, Mulindahabi, Niyigaba, Gatorano, Masozera and Beaudrot2020). Their nocturnal activity patterns further complicate observation, making them one of the most elusive and under-studied mammal groups in tropical ecosystems (Haysom et al. Reference Haysom, Deere, Wearn, Mahyudin, Jami, Reynolds and Struebig2021). Many are currently classified as data deficient by the International Union for Conservation of Nature (IUCN 2025), reflecting the limited knowledge available on their ecology and behaviour, yet they are known to play important functional roles within tropical forest systems (Whitworth et al. Reference Whitworth, Beirne, Huarcaya, Whittaker, Rojas, Tobler and MacLeod2019). Given current rates of deforestation (Global Forest Review 2025), understanding their ecological needs becomes increasingly urgent for effective conservation planning.
The majority of studies concerning tropical nocturnal arboreal mammals have focused on documenting species occurrence, abundance, and distribution (Bowler et al. Reference Bowler, Tobler, Endress, Gilmore and Anderson2017; Moore et al. Reference Moore, Pine, Mulindahabi, Niyigaba, Gatorano, Masozera and Beaudrot2020; Rojas-Sanchez et al. Reference Rojas-Sánchez, Coates, Sánchez-Cordero, Lavariega and Flores-Martínez2025). Fundamental information, such as diet, breeding behaviour, reproductive timing, habitat selection, and species interactions, is often absent for these species due in large part to the difficulties in observing within the tropical forest environment. A recent review by Astiazarán-Azcárraga et al. (Reference Astiazarán-Azcárraga, Delfín-Alfonso, Lira-Noriega, Prieto-Torres and Gallina-Tessaro2025) into the state of canopy-dwelling mammals found that only 18% of 1,800 studies were based on sampling in the canopy itself; the rest sampled from ground level only. They highlight that understanding how these mammals use the canopy is crucial to our understanding of the tropical environment. The current knowledge gap constrains the ability to formulate robust, species-specific conservation strategies.
Historically, visual surveys from line transects walked by observers using handheld spotlights were the primary method for surveying this group. Although line transects are effective for larger bodied diurnal species such as primates (Moore et al. Reference Moore, Pine, Mulindahabi, Niyigaba, Gatorano, Masozera and Beaudrot2020), both Haysom et al. (Reference Haysom, Deere, Wearn, Mahyudin, Jami, Reynolds and Struebig2021) and Whitworth et al. (Reference Whitworth, Braunholtz, Pillco, Macleod and Beirne2016) report that for small, nocturnal species active high in the canopy, accurate species identification from the ground can be challenging. Moreover, the use of bright lights can disturb animals, causing flight reactions or avoidance (Duckworth Reference Duckworth1998), reducing the likelihood of observing natural behaviours.
More recently, camera traps have been deployed in arboreal settings (Agostini et al. Reference Agostini, Pizzio, Varela, Iezzi, Cruz, Paviolo and Di Bitetti2022, Gracanin and Mikac Reference Gracanin and Mikac2022; Whitworth et al. Reference Whitworth, Braunholtz, Pillco, Macleod and Beirne2016), and reports suggest they are an improvement over transects for nocturnal arboreal mammal surveys (Bowler et al. Reference Bowler, Tobler, Endress, Gilmore and Anderson2017; Whitworth et al. Reference Whitworth, Braunholtz, Pillco, Macleod and Beirne2016). They have also been used successfully for targeted sites such as canopy bridges (Gregory et al. Reference Gregory, Carrasco-Rueda, Balbuena and Kolowski2022) and in fruiting trees (Zhu et al. Reference Zhu, Li, Gregory, Wang, Ren, Zeng, Kang, Ding and Si2022). Although arboreal camera traps are less intrusive than transects, their installation comes with drawbacks; difficulty in selecting camera positions and the need for trained staff/equipment for tree access can make them time/cost heavy. Arboreal camera traps are predominantly installed using rope climbing techniques; however, recently, non-climbing methods have been described that could reduce cost/time restraints depending on site conditions and research goals (Goebel et al. Reference Goebel, Bogoni, de Oliveira, de Souza-Ferreira and dos Santos-Filho2025). Arboreal camera traps are particularly suited to studies of occurrence and distribution because they can be deployed in large numbers over a wide area. While they have the potential to record a broad range of activity, their narrow, static field of view limits their ability to capture dynamic behaviours because most models are not currently able to track and follow movement. It should be noted, however, that arboreal camera traps have been used successfully to record behaviour in a number of studies, as reviewed by Moore et al. (Reference Moore, Soanes, Balbuena, Beirne, Bowler, Carrasco- Rueda, Cheyne, Coutant, Forget, Haysom, Houlihan, Olson, Lindshield, Martin, Tobler, Whitworth and Gregory2021). To move beyond occupancy and distribution studies towards species-specific behaviour studies, a complementary approach to arboreal camera traps that allows researchers to visually follow animals over a longer period as they move through the three-dimensional canopy structure could be useful. Visual surveys have been used for studying the behaviour of diurnal arboreal primates (e.g., Bolt et al. Reference Bolt, Schreier, Russell, Jacobson, Merrigan-Johnson, Barton and Coggeshall2019), where observers track individuals and groups, watching for interactions. For nocturnal species, enabling a similar capability requires (i) technology that allows observers to see in the dark without disturbance to mammals, and (ii) a way to reduce the observation distance by getting closer to the canopy where arboreal species are active.
Thermal imaging offers a promising solution. Although it has been used in wildlife research for decades (Havens and Sharp Reference Havens and Sharp1998), the high cost previously limited widespread use (Cox et al. Reference Cox, Matthews, Halverson and Morris2021). Recent advances in consumer-grade thermal optics, largely driven by the hunting and security markets, have improved device quality while significantly reducing cost, making them more accessible for ecological research. Most current applications involve drone-mounted thermal camera systems for aerial surveys, particularly for group living terrestrial mammals such as deer (Larsen et al. Reference Larsen, Møller-Lassesen, Enevoldsen, Madsen, Obsen, Povlsen, Bruhn, Pertoldi and Pagh2023; Zabel et al. Reference Zabel, Findlay and White2023). Drones have been used to survey tropical arboreal mammals with success for larger-bodied mammals such as sloths (Santos et al. Reference Santos, Kaizer, Moreira, Melo and Mendes2023) and spider monkeys (Spaan et al. Reference Spaan, Burke, McAree, Aureli, Rangel-Rivera, Hutschenreiter, Longmore, McWhirter and Wich2019). However, Kays et al. (Reference Kays, Sheppard, Mclean, Welch, Paunescu, Wang, Kravit and Crofoot2019) found them to be of limited use due to the density of vegetation, an issue which could be exacerbated for small-bodied mammals found in the lower canopy. In contrast, handheld cameras and monoculars have been successfully used during transect surveys for detecting greater gliders (Petauroides volans) (Vinson et al. Reference Vinson, Johnson and Mikac2020) and other small arboreal mammals (Miard et al. Reference Miard, Xin, Hampshire, Rosely, Bernard and Ruppert2024; Underwood et al. Reference Underwood, Derhè and Jacups2022), but not all studies have found handheld thermal devices to be useful. Bakker et al. (Reference Bakker, Schoenefuss, Mifsud, Fuller and Baker2024) compared thermal binoculars with baited camera traps and live capture for surveying the rare Julia Creek dunnart (Sminthopsis douglasi) in Australia. They reported no detections with thermal binoculars. Although studies are beginning to be reported, the potential of handheld thermal optics for studying nocturnal arboreal mammals in the tropics remains largely underexplored, as does overcoming the problems of observer distance to canopy-living mammals.
Our aim was to evaluate direct visual observations using thermal optics from an elevated platform as a method for investigating nocturnal behaviour of arboreal mammals via a pilot study in a remote tropical forest. We did this by assessing the method against a range of a priori criteria, namely, that the method should (i) allow detection of a wide range of species, (ii) allow a wide field of view and overcome visual limitations of distance and foliage encountered during ground-based surveys, (iii) allow the observer to remain as discrete as possible so as not to impact behaviour, and (iv) allow rich enough data collection to be able to assess diel patterns and activity budgets. Furthermore, in order for the method to be readily adopted by practitioners, it should be mobile, cost-effective, and easily installed. Based on the extent to which it met these criteria, we discuss the method’s potential to enhance our understanding of this poorly known yet ecologically important group.
Materials and methods
Study site
The survey was conducted on Cocobolo Nature Reserve (CNR), Panama (9°17′45ʺN, 79°12′18ʺW) (Figure 1). Privately owned and run by conservation NGO Conservation through Research Education and Action (CREA), CNR is 410 ha of secondary lowland wet rainforest, grading to primary and cloud forest along a rising elevation gradient. It has no permanent human presence and is subject to mild poaching activity. CNR is situated on the Pacific slope of the continental divide at its narrowest point, bordered by Chagres National Park and the Nargana Wilderness Area to the north, connecting to the Caribbean slope.

Figure 1. Location of the study site within Panama.
Equipment
Hunter’s tree stands (Hawk Helium, Hawk, Frankenmuth, USA) were used as observation platforms. The stands, designed for medium-sized trees, are attached by two ratchet straps and provide a sturdy small platform to either sit on or stand on. Lightweight, they can be carried by a single person. Platforms were installed at 12 and 10 m above ground level, respectively, on tree trunks relatively free from understory vegetation to allow for a clear view but surrounded by trees with good interconnectivity through the canopy (see Supplementary Materials Figure S1). Platforms were installed and accessed using climbing equipment (ropes and harnesses) with the observer remaining attached throughout the session. The single rope technique (SRT) was used with the observer able to climb and descend without detaching any equipment. Some training is required to use climbing equipment, but no special training is required to install the platforms, and if set lower, they can be accessed by more conventional methods such as ladders or tree sticks.
Thermal optics
Hikmicro Habrok HQ35LN (Hangzhou Microimage Intelligent Technology, Hangzhou, China) binoculars were used to carry out nocturnal observations (see Supplementary Materials Figure S1). This model is multi-spectrum, combining both thermal and night vision capabilities in one unit, thus allowing the observer to scan using thermal mode and switch to night vision mode at the press of a button. Figure 2 shows how the observer scrolled through the different colour palettes of thermal mode and on to night vision mode whilst filming a kinkajou (Potus flavus). ‘Thermal mode’ shows a mammal’s heat signature, which is effective for finding mammals and following their movement. It does not require an additional light or emission source and is considered passive. ‘Night vision mode’ shows a mammal like a camera trap as a crisp black-and-white digital image and is useful for clarification on identification, as it shows definition such as coat pattern (illustrated in Figure 2d). When in night vision mode, an integral infrared (IR) illuminator (in this case at 940 nm) emits light in order to enhance the naturally available light.

Figure 2. Frames from a video of a kinkajou taken using the Hikmicro Habrok HQ35LN comparing thermal mode with (a) fusion palette, (b) black hot palette, and (c) white hot palette, plus the alternative (d) night vision mode which uses an infra-red illuminator.
The Hikmicro Habrok HQ35LN unit used had a video recording capability of 25 frames/s at a resolution of 1080p. The video recorded was often a combination of optical and thermal, which gave a variable bit rate of 7.5 Mb/s, indicating a potential recording capability of approximately 18 h to fill the 64 GB internal solid-state drive. The manufacturer states that the unit’s two replaceable, rechargeable lithium batteries should give a six-hour run time. However, during the study, the batteries were replaced at the start of each observer session regardless of the remaining power indicated. During each observer session (3 h), the unit was permanently on and recorded an average of 40 min of video without battery issues. The batteries are easy and fast to replace in the field.
Survey design
To maximise possible species encountered, we selected two sites 521 m apart: one was in secondary forest next to a stream at 285 m AMSL, and the second was in primary forest on a ridge at 365 m AMSL. Sites were surveyed between 18:00 h and 06:00 h EST for 9 nights each. A team of two alternated as the observer for four consecutive three-hour sessions. Whilst one surveyed, the other rested in a hammock setup 150 m away. Observer consistency was ensured as the two observers (LNH and TH) worked together to design the study method and detection protocol. They had equal experience in using the described technique and in identifying the mammals being observed. Potential bias in individual attention and fatigue was mitigated by swapping the order of shifts on each subsequent session so that if one observer surveyed shifts 1 and 3 on night A, they surveyed shifts 2 and 4 on night B. For these reasons, interobserver differences were not assessed in this study, but it is recognised that it could introduce bias. This aspect should be considered by researchers using this method. During a session, the observer, from a standing position, made constant slow scanning passes with the thermal unit, both in front of and behind the trunk supporting the platform, covering 360° from ground to canopy top. The maximum distance of effective observation due to limitations of vegetation was around 30 m, but this was entirely three-dimensional. The closest focal distance of the thermal optic was around 2 m. An ad libitum sampling strategy was followed. According to Bateson and Martin (Reference Bateson and Martin2021), this means that no constraints are placed on what is recorded and when and is useful during pilot studies such as ours. It was deemed the best strategy considering we were recording all non-volant mammal species encountered and did not have a species-specific behavioural question that we were investigating. Nevertheless, the method could readily be used for different behavioural sampling and recording rules. Once a mammal was seen via the thermal mode, the video was activated, and the mammal was followed until it passed out of view. If needed, the unit was switched to night vision mode to aid identification. Although our primary focus was on arboreal mammals, ground-dwelling mammals were also recorded.
The majority of nocturnal arboreal mammals were either solitary or found in small family units. However, when two or more individuals were seen simultaneously, we tried to keep all in the field of view, but where this could not be maintained, the protocol was to follow the most easily visible individual in order to increase the chance of observing behaviour. This study’s focus was on arboreal mammals, so if both an arboreal and a ground-dwelling mammal appeared simultaneously, the protocol was to follow the arboreal mammal. By having a detailed protocol for various different scenarios and keeping observer numbers to a minimum, we hoped to minimise observer bias. The exact approach adopted by observers would need to be matched to the research question, as no single protocol will be adequate for every situation. It was assumed that there would be potential for missed detections as, despite the observer’s flexibility to scan a wide area, the entire space cannot be viewed simultaneously or continuously, and animals could thus be missed as they move through. However, in this study, we did not aim to estimate population density and so did not account for missed detections.
Sites were surveyed between 24 December 2024 and 24 February 2025. Survey nights alternated between the two sites in order to ensure that each site was surveyed under similar weather conditions and moon phases. Each site was surveyed for a minimum of 100 h.
Data analysis
During site setup a range of distances to prominent features were measured using the Habrok’s built-in laser rangefinder. These measurements were then used during data review to estimate mammal distance from and height relative to the observer (for arboreal mammals) and distance from the base of the tree (for ground-based mammals). For moving mammals, the height and distance occupied for the majority of the observation were taken.
Recordings were reviewed the following day by the same two observers independently and then jointly to confirm species identification, distance, height, and behaviour category. Potential observer detection by an animal was also recorded, defined as an animal pausing in an activity and looking at the observer or rapidly changing direction and leaving the area (see Supplementary Materials Table S1 for ethogram). When consensus could not be met, the final decision lay with the observer who recorded the event. Videos were analysed in real time and, where needed for greater clarity, frame by frame. Behaviours were coded manually into basic categories with no duration, as no species-specific questions were being investigated. However, raw footage was recorded following a continuous recording rule (Bateson and Martin Reference Bateson and Martin2021) enabling subsequent analysis to calculate activity budgets by categorising behaviour into states with start and end times. We selected six species which had a sufficient number of independent observations (>20) as examples. Where possible, mammals were classified to the species level following IUCN nomenclature. An observation was classed as a single recording, which stopped when an animal moved out of sight. However, in order not to miss possible behavioural activity, if it soon reappeared, a new recording would be made, potentially resulting in multiple clips of the same animal representing an event, something that happens frequently in camera trapping. Thus, in order to define an independent event, we chose to adopt a typically used camera trap protocol (Sollmann Reference Sollmann2018) of using a 30 min filter. This filter means that a new event was recognised only if 30 min or more had passed since the previous observation of the same species ended. The 30 min filter was applied using R statistical software (v4.4.1: R Core Team, 2024) to visualise independent events. To visualise diel activity, CamtrapR (Niedballa et al. Reference Niedballa, Sollmann, Courtiol and Wilting2016) was used in RStudio (v2024.1.764: R Studio Team 2024) to create radial plots. Four species were selected as examples which had a sufficient number of independent events (>10) to allow plotting.
Results
Results are given according to the four criteria we set for the method to be effective for investigating nocturnal behaviour of arboreal mammals.
Detection of species
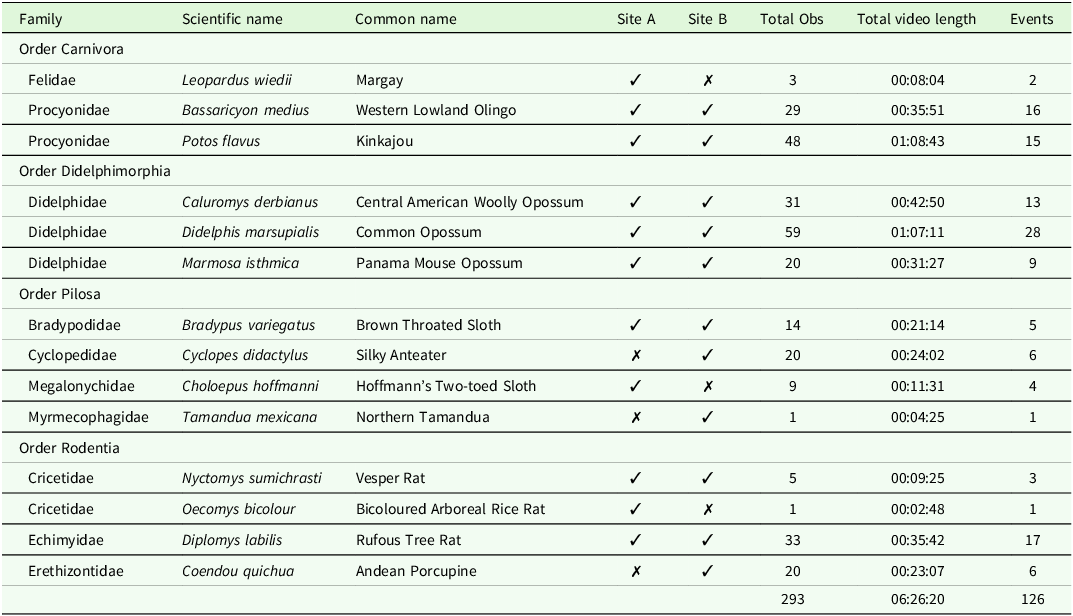
With a total survey effort of 205 h carried out equally across the two sites, 651 observations were recorded, of which 61% were nocturnal arboreal species, 20% were ground-dwelling species, and 19% were unknown or non-mammalian. When a 30 min filter was applied to the nocturnal arboreal group, there were 198 independent events, of which 126 were identifiable to species level, with 14 species recognised from four orders: Carnivora, Didelphimorphia, Pilosa, and Rodentia (Table 1). A further two arboreal species were recorded, but these were primarily diurnal species observed at dusk and so were not included in further analysis. For nocturnal arboreal mammals, a mean of 4.2 species (range 2–6, SD 1.1) and a mean of 8.2 (range 4–13, SD 2.7) independent species-level events were recorded per 12-hour session. The mean length of observation for this group was 1 m 15 s (range 4 s–7 m 21 s; SD 1 m 4 s). A breakdown of the number of observations and cumulative observation time of each species can be seen in Table 1. An additional 11 species were recorded on the ground from five orders: Carnivora, Didelphimorphia, Rodentia, Cingulata, and Lagomorpha. However, they are not considered further here (Supplementary Material Table S2 for full species data).
Table 1. Summary of nocturnal arboreal species observations during 205 h of surveying over 18 nights at two sites. For each species the number of observations, total recording time and number of events after a 30-minute filter had been applied are shown

Field of view
Arboreal events were recorded at a mean distance of 14 m (range 3–30 m, SD 5.7) from the observer and a mean height of 13 m (range 2–25 m, SD 5.5) from the ground. Ground events were recorded at a mean distance of 13 m (range 0–30 m, SD 4.5) from the base of the observer tree.
The ability to observe the full three-dimensional environment was highlighted when, on 14 February, a pair of Andean porcupines (Coendou quichua) were recorded interacting for 20 min, with the frequency of activity broken down to feeding near each other (61%), following each other (16%) and embracing each other (23%) (see video V1 https://youtu.be/VAbZa146K2k ). Eleven nights later at the same site, an adult pair were seen foraging about 2 m from each other, whilst the presence of a third was noted 30 m away in another tree. All three could be seen by the observer simultaneously, but the third individual was not close to, nor observed interacting with, the pair who remained the principal focus of the observer.
Impact of the observer on mammals being observed
Most observations were of mammals foraging, travelling, or resting (example video V2 https://youtu.be/TjkGG3ypF_0 ), but a number of specific behavioural observations suggested that it was highly probable the observer remained undetected or that the mammal was not impacted by the human’s presence. This assumption was based on a behavioural ecology theory that a mammal will not continue with activities that leave it vulnerable if they detect a possible threat (Lima and Bednekoff Reference Lima and Bednekoff1999). For example, we recorded mating behaviour in the Panama mouse opossum (Marmosa isthmica) and the Andean porcupine. We also observed intraspecific interactions between kinkajous and western lowland olingos (Bassaricyon medius). Nest material gathering was seen in the common opossum (Didelphis marsupialis), and multiple events of sloth species descending to the ground to defecate were observed. In one notable event, an ocelot (Leopardus pardalis) walked directly under the observer (10 m distance) without stopping or changing pace, showing no sign of detecting the observer (see video V3 https://youtu.be/cnh87KJTZPg ).
Occasionally, it was apparent that the observer was detected. However, in each case, there was no indication of fright. These detections accounted for 1.1% of all events and occurred with kinkajous, western lowland olingos, rufous tree rats (Diplomys labilis), and margays (Leopardus wiedii). For example, a western lowland olingo approached the observer from above, descending slowly before stopping about 1 m above the observer for several minutes. It then turned around and moved away at a steady, unhurried pace.
Diel patterns and activity budgets
Diel activity patterns for independent events of all nocturnal arboreal species combined can be seen in Figure 3a. Two activity peaks are apparent, the first between 19:00 and 20:00 and the second around 01:00. Figure 3b–e show diel activity for four species, demonstrating that at a species scale there are likely to be different patterns, something that could be studied with longer monitoring. Kinkajou activity showed two peaks at 18:00 h and 05:00 h. The aim of this work was to assess the potential of the method as a means for conducting behavioural studies. As such, we recorded the general behaviour of all mammals. However, activity budgets for six species were calculated (Figure 4), with feeding taking up the largest proportion of all activities across all species (32–85%).

Figure 3. Example diel activity pattern radial plots for nocturnal observations based on number of events per hour for (a) all species-level events, (b) kinkajou, (c) Central American woolly opossum, (d) western lowland olingo and (e) rufous tree rat.

Figure 4. Activity budget for six nocturnal arboreal species showing frequency (%) of six key behaviours for kinkajou, Central American woolly opossum, western lowland olingo, rufous tree rat, Andean porcupine and Panama mouse opossum. Durations were summed across all observations for that species.
Discussion
Effectiveness of the method
Nocturnal arboreal mammals are amongst the most difficult mammals to study because their activity takes place high in the canopy after dark, two conditions that are difficult for human observers to overcome passively. This study demonstrates that using thermal optics from an elevated observation platform enables the detection of a wide variety of species, gives a substantial field of observation, allows the observer in most cases to remain undetected and allows detailed behavioural and activity data to be collected for individual species.
Surveying at night only, we recorded 24 species (body mass range 25 g to 15 kg), of which 14 were observed arboreally. These results are comparable to similar studies using arboreal camera traps from Peru (Bowler et al. Reference Bowler, Tobler, Endress, Gilmore and Anderson2017; Whitworth et al. Reference Whitworth, Braunholtz, Pillco, Macleod and Beirne2016) and Brazil (Kaizer et al. Reference Kaizer, Alvim, Novaes, McDevitt and Young2022), which report between 17 and 24 arboreal species, numbers that include both diurnal and nocturnal species, as camera traps were operable day and night. However, the potential for this novel method to uncover important details about little-known, often small, arboreal mammals lies in the ability of the observer to watch mammal activity unobtrusively over a broad area, in this instance a radius of roughly 30 m, and from the ground to the high canopy. For example, the Andean porcupine is listed as Data Deficient on the IUCN Red List (Delgado Reference Delgado2016), and little is known about their behaviour. Delgado (Reference Delgado2016) presumes they are solitary. Yet, as described in the results, we observed three individuals simultaneously but spread out across the area. This observation illustrates the advantage a human observer has over arboreal camera traps for behavioural studies in the ability to follow multiple individuals in the canopy, something a fixed camera could not do. However, limitations remain; in a rainforest environment, observable distance is still limited by vegetation, and this may vary between sites depending on canopy structure.
Platform positioning is central to survey outcomes and represents a key consideration in survey design. Watching from the tree trunk, the observer doesn’t disrupt the travelways of either ground-dwelling or canopy mammals. The height chosen was such that it significantly reduced the visual distance to the canopy and minimised obstructing foliage compared with surveying from the ground. As stated by Whitworth et al. (Reference Whitworth, Braunholtz, Pillco, Macleod and Beirne2016), observing from the ground through a dense understory into a 20–40 m high canopy is challenging. The mean distance of our observations was 14 m, making identification easier. Additionally, this distance represents a large three-dimensional volume visible to the observer compared with the narrow field of view and smaller detection distance of an arboreal camera trap. Thus, the potential of detecting activity is greatly increased. In a separate study in 2023 (unpublished data), we used arboreal camera traps in the same study area; across 13 sites and over 806 camera trap nights, we recorded eight nocturnal arboreal species compared with the 14 species recorded from two elevated observation sites in 18 nights in this study.
Observer disturbance
Scientists strive to observe unobtrusively because disturbance of the subject animal can lead to potential negative impacts on the individual and introduce bias when behaviours seen are not representative. The combination of the elevated position and the thermal binoculars in this study likely masked the presence of the observer as a threat. Although for the most part the observer appeared to be ignored or unseen by mammals, it was noted that in 1.1% of events mammals did appear to show awareness of their presence. Whether this was through scent, sound or sight was difficult to ascertain. Movement of the observer, both accessing and on the platform, may generate noise, and it is difficult to mask our scent. The thermal equipment used is potentially a source of noise and light; studies have shown that the electrical and mechanical functions of camera traps are audible, and the IR light they emit is visible to mammals (Meek et al. Reference Meek, Ballard, Fleming, Schaefer, Williams and Falzon2014). For the binoculars used in this study, when in thermal mode, no light is emitted, and their use is invisible, but in night vision mode, an IR light at 940 nm is emitted. Although IR light is thought to be invisible to most mammals at this wavelength, little is known about spectral sensitivity in mammals and what they perceive (Meek et al. Reference Meek, Ballard, Fleming, Schaefer, Williams and Falzon2014), and Newbold and King (Reference Newbold and King2009) showed that wavelengths above 870 nm are visible to ferrets (Mustela furo). It is probable that nocturnally adapted mammals are able to see some light from the binoculars when operating in this mode. In fact, we observed the IR light source itself (illustrated in Supplementary Materials, Figure S2) when the integral IR illuminator was used in night-vision mode. Still, the IR emitted is minimal and far less intrusive than a white light (Miard et al. Reference Miard, Xin, Hampshire, Rosely, Bernard and Ruppert2024).
Despite these possible sources of disturbance, we found that observer discovery never elicited a flight reaction in any of the mammals we observed. When mammals did detect the observer, the reaction was to freeze for several minutes, watching the observer before resuming activity, seemingly unaffected by our presence, for example, a Central American woolly opossum (video V4 https://youtu.be/RlcEJ965woU) and a margay (video V5 https://youtu.be/yNn2fIGanCE). We speculate that a human is so unexpected at that height in the dark that these mammals’ usual flight response to humans is perhaps suppressed. Whilst the aim is to observe unobtrusively, it would be impossible for a human to be totally undetected by wildlife. Nevertheless, our study shows that the reaction to the observers was minimal and so should not impede behavioural observations where careful protocols are followed.
Activity patterns
In testing the method, we surveyed between 18:00 h and 06:00 h in order to collect data on as many nocturnal arboreal species as possible. Results show that most activity takes place between 19:00 h and 01:00 h. This is consistent with other studies investigating activity patterns of nocturnal arboreal mammals using camera trap data (Garcia-Casimiro and Santos-Moreno Reference García-Casimiro and Santos-Moreno2025; Gregory et al. Reference Gregory, Carrasco-Rueda, Balbuena and Kolowski2022). However, this period was for all species combined and can mask species-specific activity. For example, the kinkajou showed activity peaks at both 05:00 h and 18:00 h, which can be explained because one site encompassed a diurnal den, causing increased records as the kinkajou left or entered it. The den had not been noted when scouting the site and was only discovered by watching the kinkajou activity. This highlights that careful consideration should be given to choosing survey times and sites depending on questions posed. Although based on small sample sizes, for the Central American woolly opossum two peaks, one early (22:00 h) and one late (01:00 h), were shown. This is in contrast to Ferreira (Reference Ferreira, Cáceres and Dickman2023), who found that medium-sized marsupials tend to exhibit intense activity in the first part of the night. The pattern found here for the Central American woolly opossum could be an adaptation to climate conditions or food availability (Diete et al. Reference Diete, Meek, Dickman, Lisle and Leung2017). The western lowland olingo is closely related to the kinkajou and has a similar diet. The two peaks of activity shown (01:00 h and 04:00 h) do not overlap with the kinkajou. It is possible that this represents temporal partitioning of the two similar species (Kays Reference Kays2000). The diel activities presented for the Central American woolly opossum and rufous tree rat are, to our knowledge, the first for these species in Panama.
Activity budgets are an important way to understand an animal’s ecology (Garcia-Casimiro & Santos-Moreno Reference García-Casimiro and Santos-Moreno2025). Here, we recorded kinkajous spending a third of their time (32%) in feeding/foraging. This is in sharp contrast to Mensah et al. (Reference Mensah, Forget, Guilbert, Herrel, Ofori and Naas2024), who recorded feeding at fewer than 1% of events using arboreal camera traps. This disparity highlights the potential advantage of our novel method over camera traps to record certain behaviours. As Mensah et al. (Reference Mensah, Forget, Guilbert, Herrel, Ofori and Naas2024) point out, camera traps targeted on larger branches have limited coverage and potentially miss feeding activity on smaller outer branches. In another study which used both camera traps and observational sightings, Galvis et al. (Reference Galvis, Link, Mosquera, Vinueza-Hidalgo, Carrillo, Mopán-Chilito and Montilla2024) recorded feeding/foraging to be the second most frequent activity for kinkajous. The differing results of the three studies highlight the difficulty of recording behaviour in a mobile, nocturnal mammal and show that combining elevated thermal observations with arboreal camera traps could be useful. Although our elevated observation method revealed useful activity data, it should be noted that there are limitations. The small sample size for each species limited the analysis here because we collected data on all species seen. However, tailoring the method to a specific question or species should mitigate this. For example, locating the platform in a habitat known to be selected by the target species and focusing sampling time when it is most active could enhance the number of observations of that species per unit time. Logistical constraints such as poor weather conditions could bias the time spent or the seasons chosen to observe in. Questions that require sampling multiple sites over large areas simultaneously are likely to be better suited to arboreal camera traps. However fine-scale, targeted questions, such as observing interspecies use of a fruiting tree, could benefit from this method, and the two approaches could prove complementary.
Equipment performance, time, and monetary cost
The quality of optics used is likely to have an impact on the outcome of a study, particularly for identifying smaller mammals such as rodents and for recording finer behaviour. Our observations were carried out using a newly released multi-spectrum binocular, the Hikmicro Habrok HQ35LN. Although we used thermal mode for the majority of each session, it being easier to find and follow mammals in this mode in dense vegetation, the night vision mode was valuable for detail, such as recognising individuals and confident species identification. It is the multi-spectrum aspect that makes this tool of such high value for robust surveys, especially as the transition between optical and thermal was fast and simple, reducing loss of target during mode transitions. Our experience shows that the choice of optic should be carefully considered (Supplementary Materials Figure S3).
Even with high-quality optics, there are limitations to the method that should be recognised. High humidity interferes with thermal clarity (Supplementary Materials Figure S4), impacting the ability to identify mammals. Looking directly up into the canopy towards the skyline also obscures detail, and night vision does not perform well in close, dense vegetation. However, the elevated position we adopted mitigates these last two issues. In earlier exploratory work, we trialled a monocular (ATN OTS) but found it caused eye fatigue over the long survey times. We also trialled separate thermal (ATN Binox 4T) and night vision (ATN 4K) binoculars for scanning and identity clarification, respectively. While these optics caused less eye fatigue and provided good image quality, switching between them proved challenging due to differences in base magnification, which sometimes resulted in the subject being lost.
The Habrok HQ35LN cost c. $3,900 (2025), but a good optic can be bought for less, making them accessible to many researchers (see Supplementary Materials Table S3). The platforms used were highly portable and low cost (c. $250 in 2024). They can be carried across rugged terrain and setup easily by one person without specialist training, with installation taking less than 1 h. However, to position the platform at the heights required for the survey, climbing equipment was necessary for safety. This would require either prior climbing experience or formal training. We invested c. $500 in a harness, a 30 m rope, and accessories suitable for conducting the survey. This cost was lower than that of the climbing equipment we used to establish an arboreal camera trap array. As we had previous climbing experience, we did not undergo additional training, though relevant courses are available. A full risk assessment should be conducted prior to any climbing activities.
Conclusions
Using multi-spectrum thermal binoculars from an elevated platform was shown to be an effective method for observing nocturnal arboreal mammal behaviour in the tropics, meeting the criteria we set. Namely, the approach detected a wide range of species, and its large multi-directional field of view allowed us to capture detailed behavioural observations while remaining undetected or not causing any apparent disturbance. These features would make this method useful for targeting specific studies such as mammal/plant interactions and denning activity; tailoring variables such as height of platform and observation time to specific species could prove a strong tool for studying behaviour. Compared to traditional ground surveys, elevating the observer is likely to reduce the chance of disturbance and increase the detectability of mammals due to lessening observation distance and obstructing vegetation. Using the thermal imaging mode increased the ability for an observer to locate nocturnal arboreal mammals in dense vegetation and to watch without affecting their behaviour, something that transect surveys using white light are limited to. The wide field of view available to a human observer provides a greater opportunity to observe and follow mammal activity compared with the static narrow field of view from an arboreal camera trap. Cost and setup time are not overly prohibitive. There is potential for this elevated nocturnal observation (ENO) method to be applied to help fill current behavioural knowledge gaps in this little-known group of tropical mammals.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0266467426100509.
Data availability statement
Data are available from the corresponding author on reasonable request.
Acknowledgements
We would like to thank Michael Roy and Conservation Through Research Education and Action (CREA) for supporting us in this work and their management of the study area. We would also like to thank arborist Drew Bristow for his invaluable gift of time and expertise training us in canopy-access techniques. Thanks to two anonymous reviewers for comments on an earlier draft.
Financial support
This work was supported by Hikmicro, who provided the thermal equipment. Petzl Foundation, Yale Cordage, DMM Wales and Arb Sessions provided climbing equipment and ropes. Shaldon Wildlife Trust, Welsh Mountain Zoo and Boissiere Mervent Conservation provided funding.
Competing interests
The authors declare no competing interests.