A κ-casein glycomacropeptide (GMP) is a fascinating compound found in sweet whey (or cheese whey, hereafter referred to as whey), which is a by-product obtained during cheese manufacturing. Approximately 90 kg of liquid whey and 10 kg of cheese are produced from 100 kg of milk. Whey containing lactose, proteins, peptides, fat and mineral salts causes a significant problem for the environment if discarded without proper treatment. About 40% of whey is discarded globally leading to a substantial loss of precious nutrients (Soumati et al., Reference Soumati, Atmani, Benabderrahmane and Benjelloun2023). Therefore, investigation of beneficial ways to use whey for human consumption is important for the dairy processing industry.
Glycomacropeptide, which is also known as caseinomacropeptide, is a biologically active compound and thought to be a potential ingredient for functional foods and pharmaceuticals (Córdova-Dávalos et al., Reference Córdova-Dávalos, Jiménez and Salinas2019; Feeney et al., Reference Feeney, Joshi, Hickey and Hayes2019; Embrahimi et al., Reference Embrahimi, Andishmand, Huo, Amjadi, Khezri, Hamishehker, Mahmoudzadeh and Kim2024). Glycomacropeptide from cow's milk is a sialylated phosphorylated peptide (or glycan-phosphopeptide complex) released from κ-casein by the action of chymosin during cheese making (Eigel et al., Reference Eigel, Butler, Ernstrom, Farrel, Harwalker, Jeness and Whitney1984). Chymosin is a proteinase that catalyses the cleavage of the peptide bond between Phe-105 and Met-106 of κ-casein, yielding insoluble para-κ-casein (residues 1–105), which remains in the curd, and soluble GMP (residues 106–169), which is released into whey. Glycomacropeptide can also be prepared from sodium caseinate by hydrolysis with chymosin (Chu et al., Reference Chu, MaCleod and Ozimek1996). Bovine GMP consists of a 64-amino-acid peptide to which oligosaccharide chains – including N-acetylneuraminic acid (sialic acid), galactose and N-acetylgalactosamine – are O-glycosidically linked through threonine residues (e.g., Thr-131, Thr133 and Thr-135) (Eigel et al., Reference Eigel, Butler, Ernstrom, Farrel, Harwalker, Jeness and Whitney1984). Sialic acid is a nine-carbon, carboxylated sugar found as a terminal monosaccharide in oligosaccharides. Phosphorylation of GMP occurs at serine residues, including Ser-149 (Rasmussen et al., Reference Rasmussen, Sorensen, Petersen, Nielsen and Thompson1997).
There are 11 known genetic variants of bovine κ-casein or GMP among which variants A and B are dominant (Farrell et al., Reference Farrell, Jimenez-Flores, Bleck, Brown, Butler, Creamer, Hicks, Hollar, Ng-Kwai-Hang and Swaisgood2004). The average molecular mass of peptides of bovine GMP (for genetic variants A and B) is 6771 Da and that of intact GMP is ∼7.5 kDa (Córdova-Dávalos et al., Reference Córdova-Dávalos, Jiménez and Salinas2019). The latter varies depending on the extent of glycosylation and phosphorylation of GMP. Nakano and Ozimek (Reference Nakano and Ozimek1998) studied GMP by gel filtration chromatography and found a GMP sialic acid peak corresponding to apparent molecular weight of 36–39 kDa. From this result, they suggested that GMP forms aggregates comprising approximately three monomers. Glycomacropeptide is a hydrophilic heat stable acidic compound (Córdova-Dávalos et al., Reference Córdova-Dávalos, Jiménez and Salinas2019) with an isoelectric point (pI) < 3.8 (Nakano and Ozimek, Reference Nakano and Ozimek2000a), which is lower than that (>4.1) of major whey proteins such as β-lactoglobulin and α-lactalbumin.
Because GMP lacks aromatic amino acids such as phenylalanine (Phe) and tyrosine (Tyr), it is considered suitable as a dietary amino acid source for patients with phenylketonuria (PKU; Pena et al., Reference Pena, Pinto, Daly, MacDonald, Azebedo, Rocha and Borges2018) and hereditary tyrosinaemia type I (HTI; Daly et al., Reference Daly, Evans, Pinto, Ashmore and MacDonald2021). Phenylketonuria is a rare genetic disorder of Phe metabolism (LaClair et al., Reference LaClair, Ney, Macleod and Etzel2009) caused by a deficiency in the enzyme that catalyses the conversion of Phe to Tyr. This condition can lead to intellectual disability if not treated with a Phe-restricted diet. Phenylketonuria occurs in approximately 1 in 24,000 births, with an estimated 450,000 individuals affected worldwide (Elhawary et al., Reference Elhawary, Aljahdali, Abmansour, Elhawary, Gaboon, Dandini, Madkhali, Alosaimi, Alzahrani, Aljohani and Melibary2022). Hereditary tyrosinaemia type I is also a rare disorder of Tyr metabolism that can cause liver and kidney failure (Daly et al., Reference Daly, Evans, Pinto, Ashmore and MacDonald2021). Patients with HTI require a diet limiting both Phe and Tyr. The global incidence of HTI is much lower than that of PKU, but it is more prevalent in certain areas, particularly in Northern Europe and Canada (Äärelä et al., Reference Äärelä, Hiltunen, Soini, Vuorla, Huhtala, Nevalainen, Heikinheimo, Kivelä and Kurppa2020).
Non-protein products, typically an amino acid mixture free of Phe for PKU (van Calcar and Ney, Reference van Calcar and Ney2012) or free of both Phe and Tyr for HTI (Daly et al., Reference Daly, Evans, Pinto, Ashmore and MacDonald2021), have been used in the dietary treatment of these disorders. However, such products are often less palatable (van Calcar and Ney, Reference van Calcar and Ney2012) and more expensive than natural protein foods. Incorporating GMP into the amino acid mixture can improve palatability (van Calcar and Ney, Reference van Calcar and Ney2012; Pena et al., Reference Pena, Pinto, Daly, MacDonald, Azebedo, Rocha and Borges2018) and potentially reduce cost if GMP can be produced using inexpensive methods. Therefore, developing low-cost techniques to produce high-purity GMP with undetectable levels of Phe or both Phe and Tyr has been a long-standing challenge in the dairy food industry.
Glycomacropeptide is the third most abundant component (after β-lactoglobulin and α-lactalbumin) in whey protein/peptide mixtures, accounting for approximately 12–28% of whey products such as whey protein concentrate (WPC) and whey protein isolate (WPI) (Manso and López-Fandinõ, Reference Manso and Lépez-Fandinõ2004). Methods of GMP purification have been reviewed extensively (Abd El-Salam et al., Reference Abd El-Salam, El-Silbiny and Buchheim1996; Dziuba and Minkiewicz, Reference Dziuba and Minkiewicz1996; Brody, Reference Brody2000; Manso and López-Fandinõ, Reference Manso and Lépez-Fandinõ2004; Thomä-Worringer et al., Reference Thomä-Worringer, Sørensen and Løpez-Fandiño2006; Abd El-Salam, Reference Abd El-Salam2006; Neelima et al., Reference Neelima, Rajput and Mann2013; Feeney et al., Reference Feeney, Joshi, Hickey and Hayes2019; Karimidastjerd and Gulsunoglu-Konuskan, Reference Karimidastjerd and Gulsunoglu-Konuskan2021; Embrahimi et al., Reference Embrahimi, Andishmand, Huo, Amjadi, Khezri, Hamishehker, Mahmoudzadeh and Kim2024, and references therein). However, there are limited number of studies examining amino acid profiles of purified GMP, which is a critical parameter for determining its safety as a food ingredient for PKU or HTI patients. This review focuses on methods for preparing GMP with low to undetectable levels of Phe and Tyr.
Methods of GMP analysis
Monitoring the GMP content during whey fractionation is essential for its purification. Glycomacropeptide can be tracked by measuring both sialic acid and peptide contents. Sialic acid – a carbohydrate moiety of GMP that accounts for over 90% of the total sialic acid in whey (Nakano and Ozimek, Reference Nakano and Ozimek1999a) and occurs only in κ-casein (Eigel et al., Reference Eigel, Butler, Ernstrom, Farrel, Harwalker, Jeness and Whitney1984) in sodium caseinate – is considered a specific marker for GMP.
The content of sialic acid can be calorimetrically determined by the modified thiobarbituric acid reaction (Nakano et al., Reference Nakano, Nakano, Ahn and Sim1994; Nakano and Ozimek, Reference Nakano and Ozimek2014), in that cyclohexanone (a hazardous chemical) used as a chromophore extracting agent in the original method (Warren, Reference Warren1959) is replaced by 1-propanol to improve safety and analytical efficiency. This is a one-step, inexpensive and convenient method in which both GMP hydrolysis and chemical reaction occur in a single test tube. The method has been used to monitor sialic acid in whey samples and their chromatographic fractions (Nakano and Ozimek, Reference Nakano and Ozimek1999a). However, with the exception of our own studies cited in this review, there are very few reports in the literature that present a clear elution pattern–i.e., a distinct peak–of GMP-associated sialic acid during chromatographic separation of GMP from whey. Since a considerable number of biological activities of GMP are attributed to its sialic acid content (Dziuba and Minkiewicz, Reference Dziuba and Minkiewicz1996, Neelima et al., 2013), quantifying this sugar during fractionation of whey or caseinate hydrolysates is important.
Peptide contents of GMP can be monitored by measuring ultraviolet (UV) absorbance at 205–230 nm (Peterson, Reference Peterson1983, Nakano and Ozimek, Reference Nakano and Ozimek2015), which detects peptide bond of non-GMP peptide and protein as well as GMP, but does not distinguish GMP from non-GMP impurities. Glycomacropeptide which lacks aromatic amino acids does not show absorbance at 280 nm (Léonil and Mollé, Reference Léonil and Mollé1991), meaning that any absorbance at 280 nm likely indicates the presence of contaminating proteins and/or peptides in a GMP fraction.
Saito et al. (Reference Saito, Yamaji and Ito1991) and Thomä et al. (Reference Thomä, Krause and Kulozik2006) used reversed phase HPLC, while Doultani et al. (Reference Doultani, Turhan and Etzel2003) used size-exclusion HPLC to track GMP during fractionation of whey. Nakano and Ozimek (Reference Nakano and Ozimek2015), in refining commercial preparation of GMP by anion exchange chromatography, showed elution patterns of phosphorus, hexose and hexosamine as well as those of UV absorbance and sialic acid to confirm that the purified product is sialylated phosphorylated GMP.
Amino acid composition of purified GMP has been determined using amino acid analysers (Léonil and Mollé, Reference Léonil and Mollé1991; Coolbear et al., Reference Coolbear, Elgar and Ayers1996; Nakano and Ozimek, Reference Nakano and Ozimek1999b), the Association of Official Analytical Chemists (AOAC, Reference Coolbear, Elgar and Ayers2015) method (Arunkumar and Etzel, Reference Arunkumar and Etzel2018) and the Waters AccQ-Tag HPLC method (Nakano et al., Reference Nakano, Ozimek and Betti2018).
Electrophoresis is another important technique for characterizing GMP. Nakano and Ozimek (Reference Nakano and Ozimek2000b, Reference Nakano and Ozimek2000c) used sodium dodecyl sulphate–polyacrylamide gel electrophoresis to identify a major Coomassie blue–stained broad band of GMP aggregates with mobilities greater than that of dimeric β-lactoglobulin (36.6 kDa). Nakano et al. (Reference Nakano, Watanabe, Ikawa, Silva-Hernandez and Ozimek2009) used cellulose acetate electrophoresis to separate GMP (pI < 3.8) from major whey proteins (pI > 4.1). Furthermore, Nakano et al. (Reference Nakano, Ikawa and Ozimek2007) developed a method to detect sialylated phosphorylated GMP electrophoresed on both polyacrylamide gels and cellulose acetate strips, using the modified thiobarbituric acid reaction to detect sialic acid and the malachite green dye reaction to detect phosphorus.
Removal of non-GMP impurities from whey
Ultrafiltration (UF), deproteinization and chromatography are the primary techniques used to remove non-GMP substances from whey during purification of GMP. Ultrafiltration or dialysis is essential for eliminating lactose, minerals and other low-molecular-weight impurities, thereby producing WPC or the non-dialysable fraction of whey. This preparation step is particularly important when applying anion exchange chromatography, where the presence of negatively charged small molecules in whey can strongly influence binding behaviour.
Skudder (Reference Skudder1985) examined the effect of whey demineralization by UF on the adsorption of whey proteins to an anion exchanger (Spherosil QMA) and reported a 19% increase in protein adsorption following a 53% reduction in mineral content. Nakano and Ozimek (Reference Nakano and Ozimek2000a) further investigated how dialysis of whey influences the elution behaviour of sialylated GMP on a diethylaminoethyl (DEAE)-Sephacel anion exchange column. They found that in dialysed whey, most sialylated GMP were adsorbed to the column, whereas in non-dialysed whey, only highly sialylated GMP were retained on the column. Total recovery of sialylated GMP was approximately 1.8-fold higher in dialysed than in non-dialysed samples. These findings indicate that removing small molecular weight impurities improves accessibility and adsorption efficiency of GMP on anion exchangers. The lower recovery of sialylated GMP from non-dialysed sample is likely due to competition between GMP and negatively charged compounds including chloride, inorganic phosphates and citrate present in whey (Walstra et al., Reference Walstra, Wouters and Geurts2006; Deshwal et al., Reference Deshwal, van der Meulen and Huppertz2024). These ions exist in much higher morality than GMP and can bind tightly to the anion exchanger, thereby occupying binding sites that would otherwise be available to GMP.
Deproteinization is another useful step that facilitates chromatographic separation of high purity GMP from whey. Saito et al. (Reference Saito, Yamaji and Ito1991) employed heat treatment followed by protein precipitation with 50% (v/v) ethanol, while Nakano et al. (Reference Nakano, Ozimek and Betti2018) used heat and acid treatments to deproteinize WPC and WPI (see below for discussion of the importance of deproteinization).
Chromatographic separation of GMP from whey proteins
Glycomacropeptide differs from whey proteins in size, hydrophobicity and pI. These differences provide the basis for its separation from whey using techniques such as UF, gel filtration (or size-exclusion) chromatography, hydrophobic interaction chromatography and ion exchange chromatography suitable for isolating GMP from whey proteins. Kawasaki et al. (Reference Kawasaki, Kawakami, Tanimoto, Dosako, Tomizawa, Kotake and Nakajima1993) observed in a preliminary experiment that a GMP solution, which was turbid at neutral pH, became clear at acidic pH. Based on this observation, they hypothesized that the molecular size of GMP is pH dependent. To test this, they examined the elution patterns of GMP on a Superose-6 (Pharmacia) gel filtration column at pH 7.0 and 3.5. At pH 7.0, GMP eluted as a single peak corresponding to an apparent molecular mass between 20 and 50 kDa. At pH 3.5, however, it eluted as five distinct peaks with apparent molecular masses ranging from 10 to 30 kDa. This led them to develop a GMP purification procedure from WPC: the WPC solution was first subjected to UF using a 50 kDa molecular weight cut-off (MWCO) membrane at pH 3.5 to obtain a permeate containing GMP. The permeate was then neutralized and re-ultrafiltered using the same membrane, yielding a retentate containing 61% of the total GMP. Amino acid analysis of the final GMP preparation (81% purity) revealed small amounts of Phe and Tyr, each representing less than 1% (w/w) of total amino acids.
Nakano and Ozimek (Reference Nakano and Ozimek1998) examined the elution patterns of GMP and whey proteins by gel filtration chromatography on Sephacryl S-200 in 0.1 M sodium acetate at pH 7.0 and 3.5, and on Sephadex G-75 (Pharmacia) in 0.1 M sodium acetate with 6 M guanidine hydrochloride (dissociative condition). They found that the molecular size of GMP was not affected by pH, contradicting the report of Kawasaki et al. (Reference Kawasaki, Kawakami, Tanimoto, Dosako, Tomizawa, Kotake and Nakajima1993) but supporting that of Mikkelson et al. (Reference Mikkelson, Frøkiær, Topp, Bonomi, Lametti, Picalliello, Ferranti and Barkholt2005), who studied GMP using Superdex-75 (Pharmacia) at pH 8.0 and 3.4 and observed no pH dependent variation in the molecular size of GMP. The reason for the inconsistent results between the two studies is unclear. However, the following interpretation may be made: On Superose-6, as used by Kawasaki et al. (Reference Kawasaki, Kawakami, Tanimoto, Dosako, Tomizawa, Kotake and Nakajima1993), a solute (GMP)-stationary phase interaction occurred at pH 3.5, and thus elution volume of GMP was higher at pH 3.5 than at pH 7.0, although this possibility was not discussed by Kawasaki et al. (Reference Kawasaki, Kawakami, Tanimoto, Dosako, Tomizawa, Kotake and Nakajima1993). In contrast, there was no appreciable pH dependent interaction seen between GMP and Sephacryl S-200 or Superdex-75.
Nakano and Ozimek (Reference Nakano and Ozimek1998, Reference Nakano and Ozimek2015) also reported that (1) GMP forms aggregates in 0.1 M sodium acetate at pH 7.0 (associative condition), which dissociate into monomers in 6 M guanidine hydrochloride (dissociative condition); and (2) under associative condition, the elution position of GMP aggregates on Sephacryl column is close to that of dimeric β-lactoglobulin (36.6 kDa), while under dissociative condition, GMP monomers on Sephadex G-75 elute slightly later than does α-lactalbumin (14.2 kDa). These results suggest that high-purity GMP cannot be separated from whey proteins in a single step using gel filtration chromatography under associative condition, but that separation of GMP monomers from β-lactoglobulin may be possible under dissociative condition. The hypothesis of the latter possibility may be supported by the report of Nakano and Ozimek (Reference Nakano and Ozimek2015) who examined elution patterns of GMP and β-lactoglobulin on a Sephacryl column under dissociative condition and showed a GMP peak eluting later than dimeric β-lactoglobulin. However, the use of guanidine hydrochloride, a non-food grade chemical for preparing dissociative condition, is not recommended for food processing.
Nakano and Ozimek (Reference Nakano and Ozimek1998) further compared elution profiles of GMP and whey proteins on Sephacryl S-200 at pH 7.0 and 3.5 and found that the extent of solute-Sephacryl gel interaction occurring at pH 3.5 (but not at pH 7.0) was greater in whey proteins than in GMP. Based on this finding, Nakano and Ozimek (Reference Nakano and Ozimek2002b) developed a method of purification of GMP with low Phe and Tyr contents (each <1 mol%).
Nakano and Ozimek (Reference Nakano and Ozimek2000b) investigated isolation of GMP from whey proteins by hydrophobic interaction chromatography on a column of phenyl-agarose (Sigma). They found that most whey proteins (>95% of total) are adsorbed on the column at high concentration (5 M) of NaCl, while GMP with relatively high hydrophilicity is not, and emerged in flow-through fractions. Amino acid analysis of the GMP fraction obtained showed the presence of low contents (each <1 mol%) of Phe, Tyr, His and Arg (amino acids not found in GMP). The yield of GMP fraction was ∼1.6 g/l of whey. Phenyl-agarose chromatography has also been used for purification of goat GMP in our laboratory (Silva-Hernandez et al., Reference Silva-Hernandez, Nakano and Ozimek2002, Reference Silva-Hernandez, Nakano, Verdalet-Guzman and Ozimek2004). This method involving the desorption of GMP by retaining most whey proteins in the column is, however, less efficient compared to the anion exchange method involving selective binding of GMP to the column (see below).
Ion exchange chromatography is one of the most versatile methods used for GMP purification. The technique in general involves electrostatic interaction between fixed charge on ion exchanger and surface charge of the solute compound, the latter being influenced by pH (Ammersham Biosciences, 1999). Glycomacropeptide has a net negative charge at pH values above its pI and a net positive charge at pH values below it. Accordingly, it is important to know the pI of GMP, which differs among sources of whey due to variations in the content of sialic acid (pKa 2.6) or phosphate (pKa 2.0), both of which contribute to lowering the pI.
Since the calculated pI values for the 64-residue peptide of GMP are 4.04 and 4.14 for variants A and B, respectively (Nakano and Ozimek, Reference Nakano and Ozimek2000a), the pI of sialylated phosphorylated GMP must be lower than that of the peptide. Nakano and Ozimek (Reference Nakano and Ozimek2000a) used chromatofocusing (Pharmacia) to determine the pI of GMP but failed to see a GMP sialic acid peak eluting with a pH gradient of 6.0 to 3.8, and thus they suggested the pI < 3.8. Tran et al. (Reference Tran, Daali, Cherkaoui, Taverna, Neeser and Veuthey2001) later reported that the pI of GMP determined by two-step capillary isoelectric focusing ranged from 2.74 to 3.58. Kreuβ et al. (Reference Kreuβ, Strixner and Kulozik2009) found that the pI of glycosylated and non-glycosylated GMP determined by zeta potential measurement was 3.15 and 4.15, respectively.
In the past, a wide range of pH values (e.g., 3.0–9.0) were used for anion exchange techniques to purify GMP (Saito et al., Reference Saito, Yamaji and Ito1991; Outinen et al., Reference Outinen, Tossavainen, Syväoja and Korhonen1995; Ayers et al., Reference Ayers, Coolbear, Elgar and Pritchard1998; Etzel, Reference Etzel2001). An optimal pH may vary among the sources of whey and/or anion exchange resins. If so, a preliminary experiment to examine the yield and purity of GMP adsorbed on an anion exchanger may be useful. We (Nakano and Ozimek, Reference Nakano and Ozimek2000a; Nakano et al., Reference Nakano, Ikawa and Ozimek2004) investigated elution profiles of whey on anion exchanger at different pH values (2.0–4.5) and found that both the yield and the Phe content or absorbance at 280 nm (both reflecting amounts of impurity) were the lowest at pH 2.0 and increased with increasing pH value. Based on these results, we considered that an appropriate pH for our whey samples and anion exchangers may be ∼3.0.
Nakano and Ozimek (Reference Nakano and Ozimek1999b) applied a sample of WPC to anion exchange column of DEAE-Sephacel at pH 3.0 and found that most of whey proteins were desorbed from the column, and materials adsorbed on the column was eluted with 1 M NaCl to obtain GMP fraction with low Phe content (<1 mole%) and undetectable level of Tyr. Ayers et al. (Reference Ayers, Coolbear, Elgar and Pritchard1998) invented anion exchange and cation exchange techniques using QA GiboCel and SP GibcoCel (Life Technologies, New Zealand), respectively to purify GMP from WPC with Phe content <0.5% (w/w). Léonil and Mollé (Reference Léonil and Mollé1991) used cation exchange HPLC on a Mono S (Pharmacia) column at pH 2.0 to adsorb both GMP and whey proteins. Glycomacropeptide was then separated from whey proteins by elution with a 0–1.5 M NaCl gradient, in that the weakly bound GMP was first eluted as a single peak with 0.1 M NaCl before whey proteins were eluted with increased molarity of NaCl. Christensen and Holst (Reference Christensen and Holst2014) developed a method of large-scale production of GMP. They prepared GMP rich fraction (e.g. 79 g GMP/100 g protein) using UF. This was then chromatographed on SP Sepharose Big Beads Food Grade cation exchanger (GE Healthcare) at pH 3.5, in which GMP was eluted as a flow-throw (desorbed) material with low level of Phe (0.09–0.26% of protein).
The information discussed above suggests that both anion and cation exchangers can be used for separation of high purity GMP. However, we prefer the use of anion exchanger to selectively adsorb GMP (but not whey protein), which is more efficient compared to that of a cation exchanger (to adsorb both GMP and whey proteins or desorb GMP first and then adsorb whey proteins) for purification of GMP.
Separation of GMP from casein
Coolbear et al. (Reference Coolbear, Elgar and Ayers1996) hydrolysed bovine κ-casein with chymosin to release GMP, which was separated from non-GMP contaminants by gel filtration chromatography on Sephadex G-75 to recover GMP with undetectable levels of Phe and His. Chu et al. (Reference Chu, MaCleod and Ozimek1996) fractionated caseinate hydrolysate using UF to obtain crude GMP fraction, which they refined by cation exchange HPLC on HR5/5 mono S column (Pharmacia) to obtain GMP with undetectable level of Phe and Tyr. These are probably the first reports of GMP preparation without detectable Phe and/or Tyr. Nakano and Ozimek (Reference Nakano and Ozimek2000c) isolated GMP from caseinate hydrolysate by gel filtration chromatography on Sephacryl S-200 and acid treatment. The GMP fraction they obtained accounted for 5.3% and 0.7% of dry caseinate hydrolysate and sodium caseinate powder (used to prepare its hydrolysate), respectively. Amino acid analysis showed the presence of small amounts of Phe, Tyr, His and Arg (each <1 mol%).
Purification of GMP using charged UF membranes
It was mentioned above that separation of high purity GMP from whey without contaminating β-lactoglobulin is difficult using UF membrane only. However, Bhushan and Etzel (Reference Bhushan and Etzel2009) developed a positively charged UF membrane having higher selectivity of separation. Using this membrane, they separated GMP from the mixture of GMP and β-lactoglobulin. Arunkumar and Etzel (Reference Arunkumar and Etzel2018) further reported a new method to purify GMP from whey using a 1000 kDa MWCO positively charged UF membrane carrying polyhexamethylene biguanide (PHMB) as a ligand. These authors reported that (1) the membrane at pH 3.5 and 22°C had a high transmission of GMP (sieving coefficient = 0.61) with almost complete rejection of whey proteins (sieving coefficient = 0.029); and (2) the GMP fraction collected as permeate showed 90% purity for the one-stage system, corresponding to 2 mg Phe/g of dry protein, and 97% purity for the three-stage system, corresponding to 1.4 mg Phe/g of dry protein.
Polyhexamethylene biguanide is an antimicrobial agent used in a wide range of products, including cosmetics and sanitizers. To our knowledge, no information is available that supports its use as a functional component of food-contact UF membranes intended for human food production.
Other methods to separate Glycomacropeptide from whey for laboratory use
Nakano et al. (Reference Nakano, Silva-Hernandez, Ikawa and Ozimek2002a) treated whey powder with 9.3% (w/v) trichloroacetic acid to obtain GMP with low levels of Phe and Tyr (each <1 mol%). This preparation was further purified using a Sephacryl S-200 gel filtration column, yielding GMP with undetectable levels of Phe and Tyr. We have also developed methods of purifying GMP using epichlorohydrin-treated chitosan (Nakano et al., Reference Nakano, Ikawa and Ozimek2004, Reference Nakano, Ikawa and Ozimek2005) as anion exchangers to purify GMP with low Phe and Tyr contents (each <1 mol%). Cellulose acetate electrophoresis (see above) may be used to prepare high purity GMP for laboratory use (Nakano et al., Reference Nakano, Watanabe, Ikawa, Silva-Hernandez and Ozimek2009).
Preparation of GMP based on deproteinization followed by anion exchange methods
Producing GMP with undetectable level of Phe and Tyr by a single step procedure is challenging; therefore, combining multiple techniques is needed for preparing GMP suitable for PKU or HTI patients. Saito et al. (Reference Saito, Yamaji and Ito1991) deproteinized whey samples by heat treatment (98°C, 1 h) and precipitation with 50% (v/v) ethanol. After removal of protein precipitates, the supernatant obtained was adjusted to pH 9.0 and applied to anion exchange column, on which GMP was adsorbed and eluted with linear gradient of 0.2–1.0 M ammonium bicarbonate (pH 7.0). These authors did not explain why they performed anion exchange chromatography in alkaline pH instead of acidic pH close to, but above, the pI of GMP. We suggest the latter approach, as whey proteins (pI > 4.1) would remain unbound while GMP with pI < 3.8 would bind the anion exchanger. This may make separation of GMP more efficient compared to their method in that GMP and whey proteins are eluted with 0.3 M and 0.35 M ammonium bicarbonate, respectively (Saito et al., Reference Saito, Yamaji and Ito1991), indicating that clear separation of GMP from whey proteins is difficult. Another comment on the study of Saito et al. (Reference Saito, Yamaji and Ito1991) concerns the result of amino acid analysis of purified GMP, which was based on a total of 60 residues (calculated from their data) rather than the 64 residues expected, leaving the composition of four residues unknown.
We have deproteinized WPC and WPI by heat treatment (10 min in boiling water) followed by acid treatment (pH shift to 4.6) to obtain coagulated protein precipitates and soluble whey fraction (SWF) accounting for average 48% and 52% (w/w) of WPC, respectively (Nakano et al., Reference Nakano, Ozimek and Betti2018). The corresponding values for WPI were 70% and 30% (Nakano and Betti, Reference Nakano and Betti2020:, Reference Nakano and Betti2022).
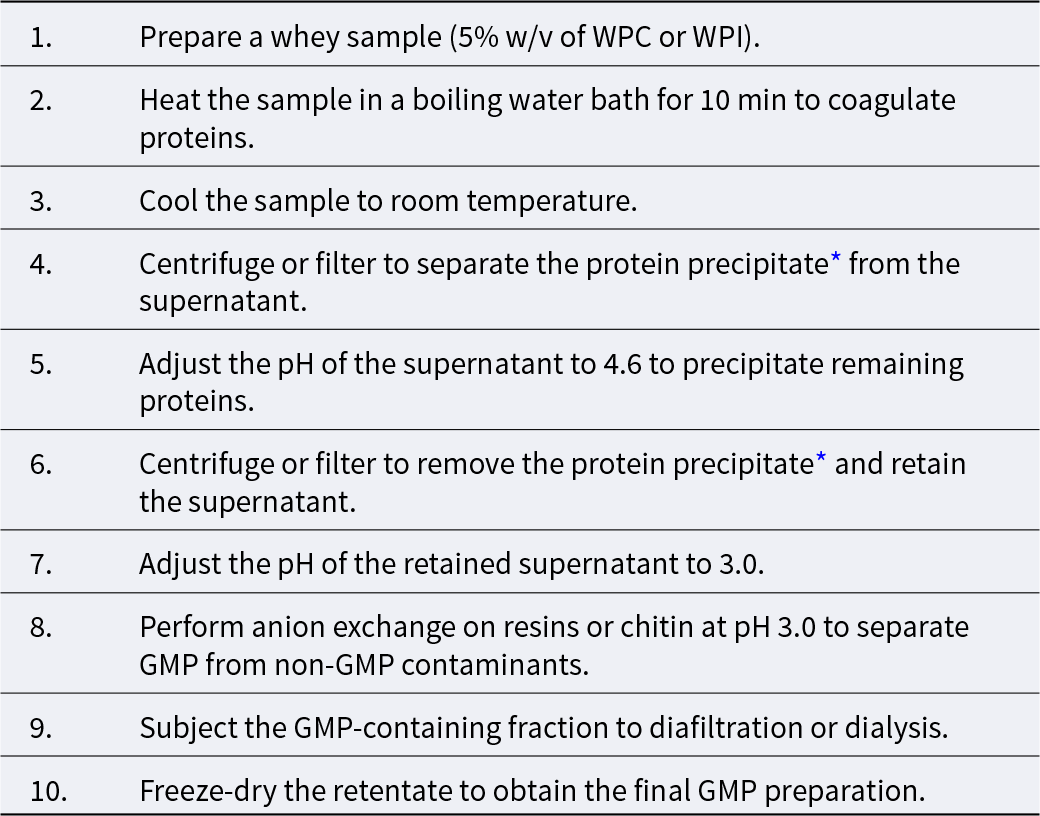
The SWF is a GMP rich fraction, in which most (average 86%) of GMP sialic acid from WPI is concentrated (Nakano and Betti, Reference Nakano and Betti2022). Nakano et al. (Reference Nakano, Ozimek and Betti2018) chromatographed SWF on DEAE-Sephacel to isolate GMP with undetectable level of contaminating amino acid including Phe Tyr, Arg and His (see Table 1 for the purification procedure).
Procedure for purification of GMP with undetectable level of Phe or Tyr

Note:
* The protein precipitate will be stored frozen until it is used as an ingredient in whey cheese.
Nakano et al. (Reference Nakano, Ozimek and Betti2018) isolated GMP from deproteinized and non-deproteinized samples of WPI by chromatography on DEAE-Sephacel and compared the yield and amino acid composition of recovered GMP fraction. The yield was found to be 1.7 times higher in the deproteinized sample, which had no detectable amount of contaminating amino acid. However, in the non-deproteinized sample, small amounts of Phe and Tyr (each <1 mol%) were found. These findings suggest that deproteinization improves both the selectivity and accessibility of anion exchangers for GMP.
Because DEAE-Sephacel is a non-food-grade anion exchanger and therefore unsuitable for human food applications, Nakano and Betti (Reference Nakano and Betti2020) tested food-grade anion exchange resins (e.g., Dowex 1 × 2, Diaion HPA75) and shrimp shell chitin for GMP purification from SWF. They confirmed that amino acid composition of GMP eluted from chitin as well as from the resins was also similar to that purified by DEAE-Sephacel chromatography.
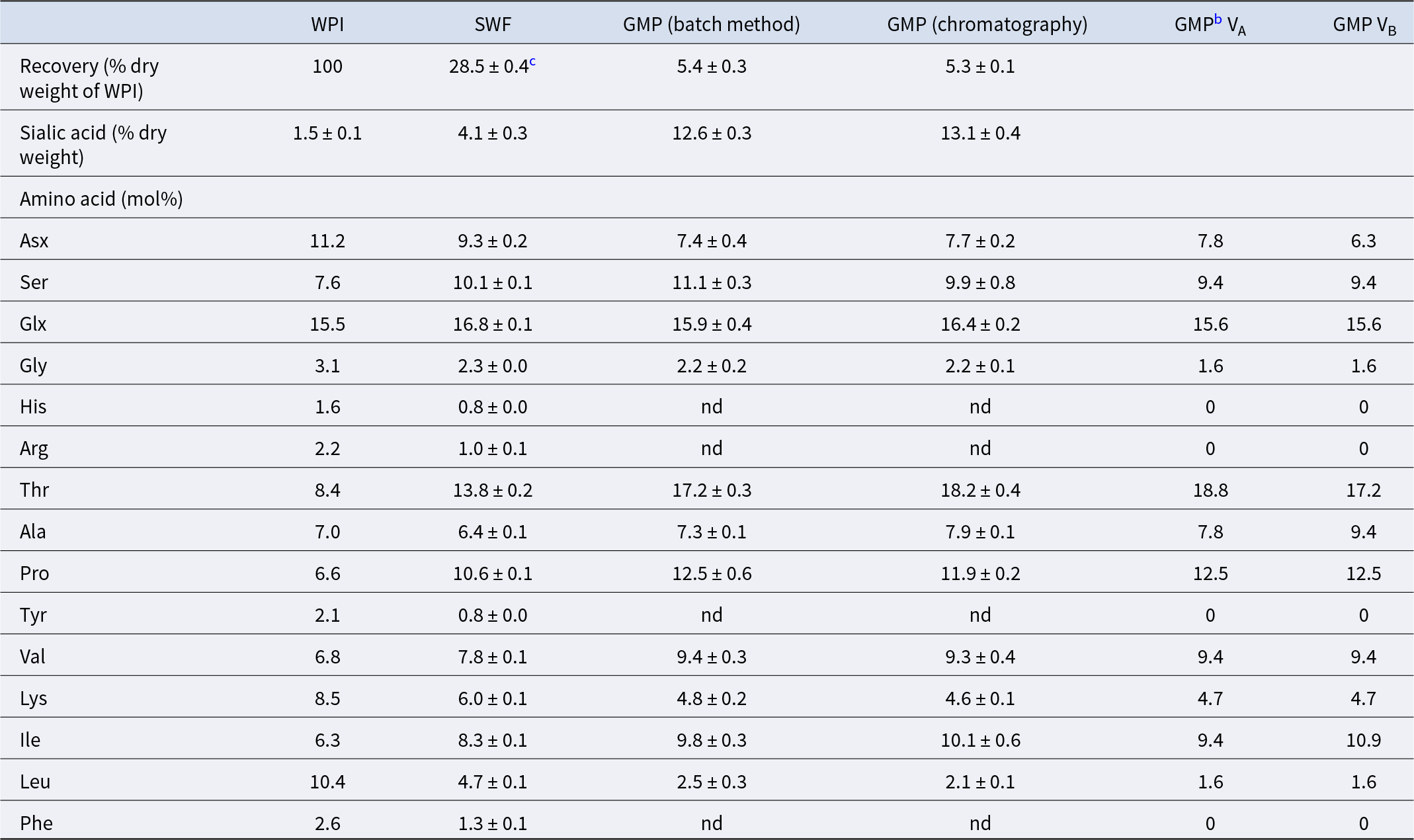
Nakano and Betti (Reference Nakano and Betti2022) then evaluated a batch method using shrimp shell chitin, which was considered more practical and economical than column chromatography. Results for GMP recovery, sialic acid concentration and amino acid composition were generally comparable between the chromatographic (Nakano et al., Reference Nakano, Ozimek and Betti2018; Nakano and Betti, Reference Nakano and Betti2020) and batch (Nakano and Betti, Reference Nakano and Betti2022) methods (Table 2). During WPI fractionation with the batch method, the sialic acid concentration increased three-fold in SWF and eight-fold in the GMP fraction compared with the starting WPI. This was consistent with amino acid composition changes: contaminating amino acids in SWF were reduced to ![]() $\leq \!50\%$ of those in WPI and were undetectable in the final GMP fraction. These findings indicate that preparing SWF by deproteinizing whey is a key step for efficient GMP separation via anion exchange.
$\leq \!50\%$ of those in WPI and were undetectable in the final GMP fraction. These findings indicate that preparing SWF by deproteinizing whey is a key step for efficient GMP separation via anion exchange.
Analysis of WPI, SWF and GMP fractions prepared by anion exchange with chitin as an adsorbenta

Notes: nd, not detected.
a Data are adopted from Nakano and Betti (Reference Nakano and Betti2022).
b Amino acid composition of genetic variant A (VA) or variant B (VB) is based on its primary structure (Eigel et al., Reference Eigel, Butler, Ernstrom, Farrel, Harwalker, Jeness and Whitney1984).
c All data except for amino acid analysis data for WPI are expressed as mean ± standard deviation (n = 3).
Comments on SWF
The process of deproteinizing whey with heat and acid treatment (Nakano et al., Reference Nakano, Ozimek and Betti2018) discussed above is comparable to that of Ricotta cheese production, in which the protein precipitate produced by heat and acid treatment (Modler and Emmons, Reference Modler and Emmons2001) is called Ricotta cheese (or whey cheese), and the liquid remaining after separation of the precipitate is called Scotta (or second cheese whey, SCW) (Pires et al., Reference Pires, Marnotes, Rubio, Garcia and Pereira2021; Fancello et al., Reference Fancello, Zara, Hamatti, Scano and Mannazzu2024). Our SWF (Nakano and Betti, Reference Nakano and Betti2022) is a type of SCW produced during WPI-based whey cheese making. Both SWF and SCW are deproteinized whey, although their chemical compositions may differ.
There has been limited information available concerning GMP in SCW. Monti et al. (Reference Monti, Donati, Zambrini and Contarini2018) studied the chemical composition of bovine Scotta and suggested the presence of GMP. However, these authors did not determine GMP concentrations in their samples. Further investigation is needed for GMP in Scotta or SCW from whey cheese manufacturers.
Conclusions
Purification of GMP with undetectable levels of Phe and Tyr requires removing non-GMP components – such as lactose, salts and proteins – from whey using non-chromatographic methods, including UF and protein precipitation. This step facilitates efficient GMP separation by chromatographic techniques, among which anion exchange is particularly effective for selectively adsorbing GMP from deproteinized whey (SWF).
Gel filtration chromatography, although unsuitable for large-scale GMP production due to its limited sample-handling capacity, remains valuable for small-scale purification in analytical purposes. Hydrophobic interaction and cation exchange chromatography may serve as secondary step techniques to remove minor contaminants from isolated GMP fractions.
High-purity GMP with undetectable Phe, Tyr, Arg and His can be obtained by deproteinizing WPC or WPI to produce SWF, followed by fractionation of SWF by anion exchange using resins and chitin, as has been described (Nakano and Betti, Reference Nakano and Betti2020, Reference Nakano and Betti2022). This laboratory scale method yields GMP which is a candidate as a food for PKU or HTI patients.
Future work requires (1) safety test of the purified GMP using animal models with PKU (Bobrova et al., Reference Bobrova, Lyukimova, Mishina, Lovanova, Valieva, Mitayaeva, Feoktistoba and Volchikov2025) or HTI (Neuckermas et al., Reference Neuckermas, Lequeue, Claes, Heymans, Hughes, Colemonts-Vroninks, Marcélis, Casimer, Goyens, Martens, Gallagher, Vankaeke, Bou-Gharios and Kock2023); (2) chemical and quantitative analysis of SCW – a by-product of whey cheese production that has not been seriously considered as a GMP source; and (3) development of cost-effective food-grade anion exchangers, such as chitin and chitosan for industrial production of GMP.
Acknowledgments
We would like to express our deep gratitude to the late Dr. Lech Ozimek, Director of the Alberta Dairy Association Research Unit (ADARU), for his long-standing support of our research on cheese whey and glycomacropeptide. His guidance and encouragement over many years made this review possible. The second author (E. S-H.) is currently employed at Eryck Silva Consulting, and works as a Senior Analyst Consultant at D4TA Data Analytics Solutions in Edmonton, Canada. He also wishes to acknowledge the support and encouragement of his colleagues throughout the preparation of this review article.