Introduction
Human activities contribute to roughly two-thirds of the world’s methane emissions (Saunois et al. Reference Saunois, Jackson and Bousquet2016), with agriculture being responsible for 41% of these emissions. This proportion includes processes such as rumen fermentation in ruminant livestock, manure management, and rice cultivation. Specifically, ruminant animals are responsible for 16% of global methane emissions. Due to population growth and the increasing demand for animal products, there is an urgent need to reduce methane emissions from ruminants and mitigate the effects of global warming. Methane emissions from enteric fermentation primarily occur in the forestomach of ruminants. This process involves a specific type of archaea, known as methanogens, which convert hydrogen into methane by reducing carbon dioxide. Various factors influence the production of methane and the growth of methanogens, including levels of hydrogen (both free and dissolved), pH balance, and the rate at which contents pass through the rumen (Janssen Peter and Kirs Reference Janssen Peter and Kirs2008). Methane significantly contributes to global radiative forcing, ranking as the second most impactful after carbon dioxide (Zhao et al. Reference Zhao, Yu and Zhao2023). Consequently, there is a critical need to mitigate methane emissions from ruminants while ensuring that animal productivity as well as the quality and safety of food are not compromised.
Although selective breeding and nutritional management strategies have been implemented to reduce methane emissions, it is crucial to deeply uncover the rumen micro-ecosystem and its functions, as well as the interactions with the host, in order to develop sustainable methane mitigation strategies (Wanapat et al. Reference Wanapat, Dagaew and Sommai2024). The advent of high-throughput technologies, collectively termed “meta-omics,” has revolutionized microbiology by enabling comprehensive studies of entire microbial communities (Valles-Colomer et al. Reference Valles-Colomer, Darzi and Vieira-Silva2016). Metagenomics involves sequencing DNA from microbial communities to identify the microbes present and infer their potential functions. Metatranscriptomics sequences the community’s RNA to monitor gene expression, providing insights into microbial activity at any given time. Metaproteomics, through proteomic profiling, sheds light on the proteins synthesized by these microbes, further elucidating their functional roles (Zhang et al. Reference Zhang, Li and Butcher2019).
These meta-omics techniques integrate to offer a multifaceted view of microbial communities, holding great promise for understanding the intricate role of rumen microbial ecosystems in methane production. With the widespread availability of meta-omics technologies to research laboratories globally and the standardization of bioinformatics analyses, the exploration of microbial dynamics and their environmental impacts has accelerated. This progress is crucial because understanding the microbial ecology of the rumen can guide the modulation of microbial populations and functions through dietary interventions, ultimately achieving more effective and sustainable ways to decrease ruminal methane production while ensuring ruminant productivity and product quality are not compromised. Technical specifics about these meta-omics methods and the corresponding bioinformatics tools for data processing have been extensively discussed in other literature (Andersen et al. Reference Andersen, Kunath and Hagen2021; Knight et al. Reference Knight, Vrbanac and Taylor2018; Mallick et al. Reference Mallick, Ma and Franzosa2017; Segata et al. Reference Segata, Boernigen and Tickle2013). This review aims to summary the current state of meta-omics research in methane emission mitigation, emphasizing the importance of holistic strategies to address the environmental impact of livestock farming.
Omic techniques in rumen microbiome studies
In the past, microbial diversity and function were primarily studied using traditional cultivation techniques (Lagier et al. Reference Lagier, Edouard and Pagnier2015). However, it has become increasingly clear that over 99% of microorganisms in natural environments cannot be cultivated under standard laboratory conditions, limiting our understanding of these complex communities (Riesenfeld et al. Reference Riesenfeld, Schloss and Handelsman2004). Furthermore, biological functions are often the result of intricate interactions within the entire microbial community rather than the activities of individual species alone (Lagier et al. Reference Lagier, Edouard and Pagnier2015; Riesenfeld et al. Reference Riesenfeld, Schloss and Handelsman2004). This recognition has driven the development and adoption of culture-independent methods, which are essential for a more complete characterization of uncultured microbes, the microbiome, and the interactions between microbes and their hosts.
The advent of high-throughput omics technologies has dramatically transformed microbiome research, allowing for comprehensive and large-scale investigations into the intricate structure and function of microbial communities. These advanced tools have opened up new possibilities for analyzing microbial ecosystems with a level of detail and complexity that was once beyond reach, significantly enhancing our understanding of microbial interactions and their roles in various environments (Di Bella et al. Reference Di Bella, Bao and Gloor2013). These technologies, which include metagenomics and metatranscriptomics, metaproteomics and metabolomics, provide a comprehensive view of the genetic, transcriptional, protein, and metabolic profiles of microbial communities. Unlike traditional methods, meta-omics approaches allow researchers to study microbial communities in their natural environments without the need for prior cultivation, thereby offering a more accurate and holistic understanding of microbial ecology and function (Helbling et al. Reference Helbling, Ackermann and Fenner2012).
In the context of rumen microbiome studies, these advanced omics techniques are particularly valuable. They enable researchers to explore the vast diversity of rumen microbes, including the identification of novel species and the elucidation of metabolic pathways involved in crucial processes like fiber digestion and methane production (Fig. 1). By integrating data from different omics layers, scientists can gain insights into the functional roles of microbial communities, how they interact with each other and with the host, and how these interactions can be manipulated to enhance animal health and reduce methane emissions. This multi-dimensional approach represents a significant advancement in our ability to study and understand complex microbial ecosystems like those found in the rumen.
Overview of host–microbiome interactions in the rumen, illustrating the use of multi-omics approaches – 16S rRNA sequencing, metagenomics, metatranscriptomics, metaproteomics, and metabolomics – to study microbial composition, gene expression, protein production, and metabolic activity, all contributing to methane emissions.

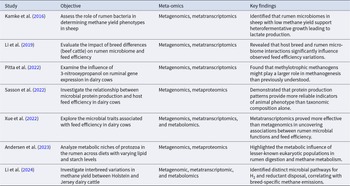
The information of meta-omics provided is presented in Table 1. Metagenomics, metatranscriptomics, and metaproteomics offer significant advantages in understanding the relationship between microbial function and methane emissions in ruminants, compared to metabolomics (Wallace et al. Reference Wallace, Snelling and McCartney2017). Therefore, we focus our review on the applications of metagenomics, metatranscriptomics, and metaproteomics in the study of rumen microbiota.
Summary of meta-omics approaches in rumen microbiome research, highlighting the molecules analyzed, knowledge gained, and limitations

Metagenomics
Metagenomics has emerged as a transformative tool in the study of microbial ecosystems, allowing scientists to explore the full genetic potential of microbial communities directly from environmental samples (Delgado et al. Reference Delgado, Bach and Guasch2019). Unlike traditional microbiological methods that rely on culturing organisms in the lab – a process that is often limited by the inability to grow many microbes outside their natural environment – metagenomics enables the direct sequencing of DNA from all organisms present in a sample (Escobar-Zepeda et al. Reference Escobar-Zepeda, Vera-Ponce and Sanchez-Flores2015). This approach has been particularly valuable in studying the rumen, a complex and dynamic ecosystem where diverse microbial populations drive crucial processes, including the production of methane through enteric fermentation (Sharpton Reference Sharpton2014). This has significantly advanced our understanding of the microbial underpinnings of methane emissions in ruminants and opened new pathways for developing targeted mitigation strategies.
Decoding rumen microbiota: Pathways in low methane emission and enhanced feed efficiency
Metagenomics has become a key tool in studying the microbial communities of ruminants such as cattle and sheep, enabling researchers to analyze microbial DNA sequences directly from these animals and investigate how variations in these communities are linked to animal phenotype (Denman and McSweeney Reference Denman and McSweeney2015; Huws et al. Reference Huws, Creevey and Oyama2018; Saborío-Montero et al. Reference Saborío-Montero, Gutiérrez-Rivas and García-Rodríguez2020; Shi et al. Reference Shi, Moon and Leahy2014; Wallace et al. Reference Wallace, Rooke and McKain2015). Data from these studies have highlighted the complex interactions between bacteria and archaea within the rumen, particularly under conditions where methane mitigation is effective. Henderson et al. (Reference Henderson, Cox and Ganesh2015) conducted an extensive global survey of rumen microbial communities, analyzing 742 samples from 32 ruminant species across 35 countries. The study found that while the dominant bacterial and archaeal species were consistent across different geographical regions and host species, the composition of these communities was predominantly influenced by the diet rather than the host. The core microbial groups, including Methanobrevibacter and other methanogens, were found to be highly conserved, suggesting that these microbes could be key targets for developing universal strategies to reduce methane emissions. The study also identified specific microbial interactions, such as the association between succinate producers and methanogens, which could play a crucial role in methanogenesis. Notably, succinate-producing bacteria from the Succinivibrionaceae family, succinate-consuming Dialister, and amino acid-utilizing Acidaminococcus were observed to interact closely with methanogens (Henderson et al. Reference Henderson, Cox and Ganesh2015).
Saborío-Montero et al. (Reference Saborío-Montero, Gutiérrez-Rivas and García-Rodríguez2020) use structural equation models to explore the complex relationships between the rumen microbiome, methane production, and host genetics in dairy cattle. The study revealed that specific microbial genera, particularly from the Ciliophora, Euryarchaeota, Chytridiomycota, and Fibrobacteres phyla, have strong positive genetic correlations with methane emissions. These findings suggest that both microbial composition and methane production are partially controlled by the host genotype. The study highlights the potential for integrating metagenomic and genomic data to improve the accuracy of methane emission predictions and support the development of breeding strategies aimed at reducing methane emissions in dairy cattle (Saborío-Montero et al. Reference Saborío-Montero, Gutiérrez-Rivas and García-Rodríguez2020). Recently, Saborío-Montero et al. (Reference Saborío-Montero, López-García and Gutiérrez-Rivas2021) and López-García et al. (Reference López-García, Saborío-Montero and González-Recio2022) demonstrated that specific eukaryotic microbes, particularly ciliates and fungi, are closely associated with high methane emissions, while certain bacterial groups are linked to lower emissions. Their study utilized whole metagenome sequencing to uncover the complex interactions between these microbes and their metabolic pathways.
Studies on low-methane-emitting ruminants have identified a possible connection between reduced methane emission and the presence of specific microbial taxa, particularly certain members of the Succinivibrionaceae and species of Prevotella noted in cattle (Danielsson et al. Reference Danielsson, Dicksved and Sun2017; Tapio et al. Reference Tapio, Snelling and Strozzi2017; Wallace et al. Reference Wallace, Rooke and McKain2015). The study of Pope et al. (Reference Pope, Smith and Denman2011) explored the unique microbial ecosystem within the Tammar wallaby’s foregut, focusing on a specific bacterial group within the Succinivibrionaceae family, designated as Wallaby Group 1 (WG-1). The researchers isolated this bacterium, which is associated with low methane emissions, a trait of interest due to the wallaby’s significantly lower methane production compared to ruminants like cattle. The Succinivibrionaceae family was found to be four times more abundant in low methane-emitting cattle (Wallace et al. Reference Wallace, Rooke and McKain2015). These bacteria are associated with the production of succinate rather than hydrogen, which reduces the availability of hydrogen for methanogenesis, thereby contributing to lower methane emissions (Wallace et al. Reference Wallace, Rooke and McKain2015). The results suggest that increasing the abundance of Succinivibrionaceae in the rumen could be a potential strategy for reducing methane emissions in ruminants, aligning with findings from other studies that show a similar role for these bacteria in other low-emitting animals, such as the Tammar wallaby (Pope et al. Reference Pope, Smith and Denman2011). The study by McCabe et al. (Reference McCabe, Cormican and Keogh2015) utilized Illumina MiSeq amplicon sequencing to analyze the effects of feed restriction on the rumen microbiome in cattle. They observed a significant reduction in the relative abundance of an uncharacterized Succinivibrionaceae species (OTU-S3004) and a corresponding increase in the Methanobrevibacter gottschalkii clade (OTU-M7) in feed-restricted cattle. The study also noted a strong negative correlation between these two OTUs, suggesting that Succinivibrionaceae may compete with methanogens for hydrogen, thereby influencing methane production (McCabe et al. Reference McCabe, Cormican and Keogh2015). Additionally, an increase in the acetate ratio was observed in feed-restricted animals, indicating a potential shift toward increased methane production (McCabe et al. Reference McCabe, Cormican and Keogh2015). Similarly, cows with lower methane emissions had distinct microbial communities in their rumen, particularly showing a higher abundance of Succinivibrionaceae and certain species within the Methanobrevibacter genus (Danielsson et al. Reference Danielsson, Dicksved and Sun2017).
Shabat et al. (Reference Shabat, Sasson and Doron-Faigenboim2016) assessed the association between the ruminal microbiome and feed efficiency in dairy cows. They found that cows with higher feed efficiency had lower microbial diversity but a higher dominance of specific functional groups, such as Megasphaera elsdenii, which plays an important role in the production of volatile fatty acid (VFA) like propionate and butyrate. These VFA are beneficial for the host’s energy requirements and are associated with reduced methane emissions. Additionally, Kamke et al. (Reference Kamke, Kittelmann and Soni2016) focused on the rumen microbiomes of sheep with varying methane emissions, utilizing metagenomic and metatranscriptomic analyses. It found that sheep with low methane yield had a microbiome enriched with Sharpea species and other lactate-producing bacteria. These microbes contribute to a metabolic pathway favoring lactate and butyrate production over hydrogen, thereby reducing methane production (Fig. 2). These studies highlight that animals with higher feed efficiency tend to have a less diverse but more functionally specialized rumen microbiome, which favors the production of VFA like propionate, thereby reducing hydrogen availability for methanogenesis.
Comparative pathways leading to low and high methane yield in ruminants. The left panel illustrates the metabolic pathway in low methane yield (LMY) animals, characterized by the dominance of Sharpea spp. and Megasphaera spp., leading to rapid fermentation with reduced hydrogen (H₂) production and increased butyrate absorption. The right panel shows the pathway in high methane yield (HMY) animals, where Lachnospiraceae and Ruminococcaceae dominate, resulting in higher hydrogen production and increased acetate and butyrate formation, contributing to greater methane (CH₄) emissions. Adapted from Kamke et al. (Reference Kamke, Kittelmann and Soni2016).

Unlocking hidden potentials: Metagenome-assembled genomes and microbial genomic references in rumen studies
The application of metagenome-assembled genomes (MAGs) and microbial genomic references in rumen studies has significantly advanced our understanding of the complex microbial ecosystems involved in methane production. By reconstructing genomes directly from metagenomic data, researchers have been able to identify previously unrecognized microbial species and functional pathways that contribute to the methanogenic processes in the rumen (Xie et al. Reference Xie, Jin and Si2021). MAGs offer a powerful tool for exploring the functional diversity within the rumen microbiome, providing insights into the metabolic capabilities of uncultured and often rare microorganisms. Xie et al. (Reference Xie, Jin and Si2021) constructed an extensive gastrointestinal tract (GIT) microbial gene catalog from seven ruminant species, including over 154 million nonredundant genes and 10,373 MAGs. The research uncovered 8,745 previously unidentified uncultured bacterial and archaeal species, broadening our knowledge of the microbial diversity within the ruminant GIT. These discoveries underscore the important contribution of lesser-known microbial communities to methane production and feed efficiency, providing fresh perspectives for enhancing ruminant livestock production. A new published study explored the functional potential of the rumen microbiome, emphasizing the contributions of novel microbial species Methanomassiliicoccales to hydrogen metabolism and methane production (Xie et al. Reference Xie, Zhao and Zhan2024).
Currently, the scope of metagenomic and microbial genomic sequencing is broadening to include a variety of sample types, such as the rumen microbiome (Kim et al. Reference Kim, Park and Yu2017; Martínez-Álvaro et al. Reference Martínez-Álvaro, Auffret and Stewart2020; Stewart et al. Reference Stewart, Auffret and Warr2018). This expansion has enabled the assembly of nearly complete microbial genomes and the identification of numerous microbial strains and species within the rumen (Stewart et al. Reference Stewart, Auffret and Warr2018). Key initiatives, such as the Rumen Microbial Genomics Network and the Hungate1000 project, are instrumental in expanding the repository of reference microbial genomes. These efforts are crucial in advancing our understanding of the rumen microbiome by providing a comprehensive genetic framework, which aids in the exploration of microbial diversity and functions within the rumen ecosystem (Seshadri et al. Reference Seshadri, Leahy and Attwood2018). Additionally, the study associated with the Global Rumen Census revealed that despite the vast diversity in diet, host species, and geographic locations, a core microbiome of dominant bacterial and archaeal groups exists across ruminants worldwide (Henderson et al. Reference Henderson, Cox and Ganesh2015). The research highlighted that dietary factors have a more significant impact than host species on the composition of the rumen microbiome (Henderson et al. Reference Henderson, Cox and Ganesh2015). This global survey provides valuable insights into the rumen microbial ecosystem, offering a foundation for future research aimed at understanding and potentially manipulating these microbial communities to enhance ruminant productivity and reduce methane emissions.
The Hungate1000 project, as detailed by Seshadri et al. (Reference Seshadri, Leahy and Attwood2018), provided a comprehensive collection of 410 cultured bacterial and archaeal species, representing nearly all known cultivated families associated with the rumen microbiome. This extensive genomic resource has significantly enhanced our understanding of rumen microbial diversity, particularly in relation to polysaccharide degradation and methanogenesis. By combining these genomes with metagenomic datasets, the project demonstrated that around 75% of the genus-level taxa found in the rumen are represented in the Hungate collection. However, the study also pointed out several limitations. Despite the extensive coverage of the collection, a significant portion of the rumen microbiome remains uncultured and poorly characterized, especially within the lesser-known taxa. This highlights the ongoing need to culture and sequence new microbial species to fully understand the functional roles of the entire rumen microbiome. Future research should aim to expand the genomic catalog to include these uncultured species and develop methods to integrate these data with functional analyses. Such initiatives are essential for enhancing our ability to regulate the rumen microbiome to improve feed efficiency and reduce methane emissions.
While shotgun sequencing presents many benefits, it also faces significant challenges that require further refinement. One major challenge involves the steps needed for DNA preparation and subsequent data analysis. As noted by Zhang et al. (Reference Zhang, Li and Butcher2019), managing the enormous amount of data generated by this method requires a high degree of technical expertise and the use of costly equipment. The process is complicated further by reliance on reference databases to identify gene lists and match DNA reads, which can be limiting if the databases lack certain reference sequences, making it difficult to fully map out genomes and understand their functions (Tilocca et al. Reference Tilocca, Pieroni and Soggiu2020; Weinstock Reference Weinstock2012) Although predicting genes de novo can bypass these limitations, it significantly adds to both the time and cost of analysis. Another drawback of shotgun metagenomics is its inability to distinguish between live and dead cells, which can complicate the interpretation of microbial activity within a sample (Heintz-Buschart et al. Reference Heintz-Buschart, May and Laczny2016).
Metatranscriptomics
Metatranscriptomics is a valuable meta-omics approach that sheds light on the functional dynamics of the rumen microbiome by examining the collective RNA transcripts expressed by the microbial community at a specific moment (Pitta et al. Reference Pitta, Indugu and Baker2018). In contrast to metagenomics, which uncovers the genetic potential of the microbiome, metatranscriptomics identifies which genes are actively being transcribed, providing a real-time snapshot of microbial activity and functional roles within the rumen ecosystem (Shakya et al. Reference Shakya, Lo and Chain2019). In the context of rumen methane emissions, metatranscriptomics is invaluable for identifying key microbial pathways and enzymes involved in methanogenesis. By focusing on the active transcriptome, researchers can pinpoint which methanogenic archaea and associated microbial taxa are most actively contributing to methane production under different dietary and environmental conditions.
A growing body of research is increasingly leveraging metatranscriptomic techniques to explore the functional dynamics of the rumen microbiome, particularly in relation to host feed efficiency and methane output. Unlike metagenomics, which focuses on the genetic potential of microbial communities, metatranscriptomics offers a more nuanced view by capturing active gene expression, thereby providing clearer insights into how microbial functions influence host phenotype (Xue et al. Reference Xue, Xie and Zhong2022). For instance, Li and Guan Le (Reference Li and Guan Le2017) highlight the significant role of metatranscriptomics in uncovering the active microbial processes in the rumen, particularly in relation to feed efficiency in beef cattle. It identified that the Succinivibrionaceae family, which was the most abundant active bacterial group, plays a crucial role in propionate production – a key pathway that competes with methanogenesis by utilizing hydrogen. Additionally, the study revealed that inefficient cattle exhibited more diverse microbial activities and metabolic pathways, which could be linked to their higher intake levels. These findings suggest that targeting specific microbial functions identified through metatranscriptomic profiling could be a potential strategy for improving feed efficiency and reducing methane emissions in ruminants. Li et al. (Reference Li, Hitch and Chen2019) conducted a comparative analysis of the rumen microbiomes across different breeds of beef cattle with varying feed efficiencies using both metagenomic and metatranscriptomic approaches. It revealed that while the metagenomic profiles (DNA-based) showed more conservation across individuals, the metatranscriptomic profiles (RNA-based) exhibited higher variability, reflecting real-time microbial activities (Li et al. Reference Li, Hitch and Chen2019). The study highlighted that functional gene expression levels in the rumen microbiome are closely linked to host phenotypes, such as feed efficiency, suggesting that metatranscriptomics provides a more accurate reflection of the microbiome’s role in influencing these traits (Li et al. Reference Li, Hitch and Chen2019). These findings emphasize the importance of using metatranscriptomic analysis to better understand and manipulate the rumen microbiome for enhanced livestock productivity.
The study by Neves et al. (Reference Neves, Chen and Le Cao2020) conducted a study using metatranscriptomics to investigate the taxonomic and functional characteristics of the rumen microbiome in Black and Red Angus cattle. The research uncovered breed-specific microbial signatures, with Black Angus showing a higher abundance of fibrolytic species such as Chitinophaga pinensis and Clostridium stercorarium, which are linked to enhanced feed efficiency. In contrast, Red Angus cattle exhibited an enrichment of species like Oscillibacter valericigenes and pathways related to carbohydrate metabolism. This study demonstrates how metatranscriptomics can reveal subtle differences in microbial activity and function between closely related breeds, offering valuable insights into the microbial mechanisms that influence feed efficiency and methane production.
A metatranscriptomic approach offers vital insights into the active transcriptional processes of rumen microbes involved in methane production. Notably, genera such as Methanocaldococcus and Methanobrevibacter have been identified as highly active, with a substantial portion of their transcripts linked to both methane synthesis and carbohydrate metabolism (Mann et al. Reference Mann, Wetzels and Wagner2018). In the methanogenesis pathway, methyl-coenzyme M reductase (Mcr) plays a central role, catalyzing the final step of methanogenesis (Söllinger et al. Reference Söllinger, Tveit and Poulsen2018). The gene encoding the α-subunit of this enzyme, mcrA, is considered a key functional and phylogenetic marker for identifying methanogenic archaea (Lueders et al. Reference Lueders, Chin and Conrad2001; Paul et al. Reference Paul, Nonoh James and Mikulski2012). Söllinger et al. (Reference Söllinger, Tveit and Poulsen2018) employed quantitative metatranscriptomics to comprehensively analyze the dynamics of the ruminal microbiome during anaerobic feed digestion, offering valuable insights into microbial activity under these conditions. The research highlighted the significant contributions of bacteria, archaea, and eukaryotes to CH4 production and VFA synthesis, revealing a high degree of functional redundancy among microbial taxa. Notably, the study identified Methanomassiliicoccales and Methanosphaera as key methanogens responsible for CH4 emissions immediately following feed intake. The study also demonstrated that shifts in microbial activity, rather than changes in community composition, primarily drive the temporal dynamics of methane production.
Metatranscriptomics, like metagenomics, serves as a powerful approach for analyzing rumen microbiota, particularly in ruminants exposed to the same diet but exhibiting different levels of methane output. Shi et al. (Reference Shi, Moon and Leahy2014) employed metatranscriptomic analysis to investigate the expression levels of methanogenesis-related genes in the rumen microbiome of sheep with different methane emission levels. The key findings reveal that high methane-emitting sheep exhibit significantly elevated expression of genes involved in the hydrogenotrophic methanogenesis pathway, particularly those encoding the enzyme methyl-coenzyme M reductase (MCR), which catalyzes the final and rate-limiting step in methane production (Shi et al. Reference Shi, Moon and Leahy2014). This increase in gene expression was not matched by an increase in the abundance of these genes, indicating that transcriptional regulation plays a critical role in determining methane yield. Similarly, Zhang et al. (Reference Zhang, Xu and Wang2016) used metatranscriptomic analysis to explore the transcriptional activity within the rumen microbiomes of high-altitude mammals, specifically yaks and Tibetan sheep, which have adapted to produce low methane emissions and high levels of VFA. The key findings showed that there is significant upregulation of genes involved in VFA transport and absorption in these high-altitude ruminants compared to their lowland counterparts (Zhang et al. Reference Zhang, Xu and Wang2016). This upregulation supports a metabolic adaptation that enhances energy extraction from feed while simultaneously reducing methane production.
Metatranscriptomics is a powerful tool not only for analyzing microbial composition and functions linked to methane production within a single ruminant species but also for comparing function disparities in methane generation across various ruminant breeds. For instance, the metatranscriptomic analysis revealed that Holstein cows (low-CH4-emitter) exhibited higher expression levels of genes involved in alternative hydrogen disposal pathways, particularly those related to amino acid synthesis and propionate production (Li et al. Reference Li, Ma and Huo2024). This suggests a more efficient use of hydrogen that limits its availability for methanogenesis, thereby reducing methane emissions. Conversely, in Jersey cows (high-CH4-emitter), the metatranscriptomic data indicated an upregulation of genes associated with hydrogenotrophic methanogenesis, leading to higher methane output (Li et al. Reference Li, Ma and Huo2024). These findings underscore the value of metatranscriptomics in identifying key metabolic pathways and gene expression profiles that contribute to methane emission differences between breeds, offering potential targets for mitigation strategies.
Metaproteomics
Metaproteomics provides a unique lens through which the functional proteins of the rumen microbiota can be directly examined, offering insights into the biochemical activities that underpin methane production (Wilmes and Bond Reference Wilmes and Bond2004). Unlike metagenomics, which reveals the genetic blueprint of microbial communities, or metatranscriptomics, which captures gene expression, metaproteomics focuses on the proteins that are produced and active within the rumen environment. This approach allows researchers to study the enzymes and protein complexes in real-time, reflecting the immediate metabolic processes occurring within the rumen.
Metaproteomics analysis has revealed distinct variations in metabolic pathways across the major phyla within the rumen microbiota, highlighting the presence of diverse ecological niches (Hart et al. Reference Hart, Creevey and Hitch2018). Although the field of rumen metaproteomics is still in its early stages, research has gradually expanded to include various animals, such as adult sheep (Snelling and Wallace Reference Snelling and Wallace2017), pre-weaned lambs (Palomba et al. Reference Palomba, Tanca and Fraumene2017), cows (Deusch et al. Reference Deusch, Camarinha-Silva and Conrad2017; Deusch and Seifert Reference Deusch and Seifert2015; Hagen et al. Reference Hagen, Brooke and Shaw2021; Hart et al. Reference Hart, Creevey and Hitch2018; Honan and Greenwood Reference Honan and Greenwood2020; Mulakala et al. Reference Mulakala, Smith and Snider2023, Reference Mulakala, Smith and Snider2022; Sasson et al. Reference Sasson, Moraïs and Kokou2022), beef cattle (Li et al. Reference Li, Zang and Zhao2021), and moose (Solden et al. Reference Solden, Naas and Roux2018). These studies have begun to uncover the complexity of the rumen metaproteome, with a focus on how protein expression patterns differ between species and developmental stages. Honan and Greenwood (Honan and Greenwood Reference Honan and Greenwood2020) highlighted that the rumen microbiota’s protein expression, particularly translation-related proteins such as ribosomal subunits, fluctuated significantly after feeding, underscoring the importance of temporal sampling in understanding microbial activity and its impact on nutrient metabolism in the rumen.
Sasson et al. (Reference Sasson, Moraïs and Kokou2022) used metaproteomics to explore the association between rumen microbial protein production and host feed efficiency. The researchers revealed that the rumen microbiome’s protein production profiles were more predictive of the host’s feed efficiency state than either metagenomic or taxonomic data. Specifically, the study highlighted the plasticity of the rumen metaproteome, revealing significant differences in protein production between high and low feed efficiency states. These differences were linked to distinct metabolic functions, particularly those related to carbohydrate metabolism and fermentation. The findings emphasize the potential of metaproteomics to provide insights into the functional roles of microbial communities in the rumen and their impact on methane emissions and overall animal productivity.
The study by Deusch et al. (Reference Deusch, Camarinha-Silva and Conrad2017) utilized metaproteomics to identify specific microbial groups that are directly involved in methane production within the rumen. Their research highlighted the significant roles of Methanobrevibacter and Methanosphaera in the methanogenic pathways, particularly under high-fiber dietary conditions. Additionally, the study uncovered that members of the Firmicutes phylum, especially those within the Clostridiales order, play a critical role in providing substrates like hydrogen that methanogens utilize for methane production. Trautmann et al. (Reference Trautmann, Schleicher and Koch2023) highlights the adaptive response of the ruminal microbiome to monensin, a common feed additive known to reduce methane production. Using metaproteomics, the study observed a significant shift toward succinate-producing Prevotella species within the rumen microbiota when exposed to monensin. This shift was accompanied by a decrease in methane production and a change in the fermentation metabolism from acetate to succinate. These findings illustrate how Prevotella species play a crucial role in stabilizing rumen functionality under monensin treatment by altering metabolic pathways that are less favorable for methane production.
Metaproteomic studies have shed light on the vital metabolic roles played by previously understudied ruminal eukaryotic populations, revealing their significant contributions to both fiber degradation and CH4 emission. Using a genome-centric metaproteomic approach, Hagen et al. (Reference Hagen, Brooke and Shaw2021) pointed to the significant contribution of fungi, especially from the genera Neocallimastix and Piromyces, in breaking down lignocellulosic material through the production of carbohydrate-active enzymes (CAZymes), such as those from the GH48 family. These enzymes are essential for the efficient degradation of crystalline cellulose. Furthermore, the study illustrates the complementary interaction between fungi and bacteria in the rumen, where fungi primarily degrade resistant fibers, while bacteria target non-cellulosic carbohydrates (Hagen et al. Reference Hagen, Brooke and Shaw2021). These insights contribute to a better understanding of microbial cooperation in fiber digestion and its potential impact on methane emissions in ruminants. The proteomic analysis revealed active metabolic processes in these protozoal species that are closely associated with methane emissions. Specifically, Dasytricha and Isotricha were found to contain hydrogenases, while Epidinium was rich in CAZymes, and both Entodinium and Epidinium were involved in VFA production (Andersen et al. Reference Andersen, Altshuler and Vera-Ponce2023). It was observed that the metabolic activity of these protozoa is influenced by diet, with higher detection of proteins involved in fiber and starch degradation in animals fed a control diet compared to those on a starch-rich diet (Andersen et al. Reference Andersen, Altshuler and Vera-Ponce2023). These findings highlight the importance of protozoal contributions to overall rumen function and their potential impact on methane emissions, providing valuable insights for developing targeted methane mitigation strategies in ruminants.
Although metaproteomic techniques have been slow to gain traction in rumen research, recent advancements are accelerating their use. Integrating metaproteomic data with multi-omic datasets or metadata, despite its complexity, is vital for understanding the intricate host–microbiome–environment interactions. This holistic approach, known as holo-omics, is key to enhancing animal health and performance, although it is still in its early stages. Promising strategies include the construction of co-expression networks (Heirendt et al. Reference Heirendt, Arreckx and Pfau2019) and the application of constraint-based models (Langfelder and Horvath Reference Langfelder and Horvath2008) that combine host and microbiome data, offering significant potential for future research and applications.
Integrative meta-omics for understanding ruminal methane emissions
Integrating data from multiple meta-omic approaches provides additional insights into rumen microbiome functions, particularly for studies focused on methane reduction. For instance, combining metagenomics with metatranscriptomics allows for the calculation of transcript-to-gene ratios, which can reveal whether genes are transcriptionally activated or repressed – an essential aspect for understanding microbial gene expression involved in methanogenesis (Li et al. Reference Li, Neves and Ghoshal2018). Additionally, metaproteomics is often integrated with metagenomics to facilitate protein identification through matched metagenome database searches or to assess protein expression levels within the microbiome that are related to methane production pathways (Andersen et al. Reference Andersen, Kunath and Hagen2021). Compared to metagenomics, metatranscriptomics, and metaproteomics, single metabolomics analysis cannot directly associate the composition and function of rumen microorganisms. One of the major challenges of metabolomics in ruminant livestock studies is distinguishing between metabolites derived from the host and those from the microbiome and associating specific metabolites with particular archaea taxa (Nicholson et al. Reference Nicholson, Holmes and Kinross2012). Nonetheless, there is a growing trend to integrate metabolomics with metagenomics in order to uncover co-variation patterns between metabolites and the composition or function of microbial communities. This approach also helps in identifying the phylogenetic contributions to metabolite production that impact methane emissions (Li et al. Reference Li, Ma and Huo2024).
Examples of integrating meta-omics approaches to reveal the methane emission or feed efficiency difference between individuals and mechanisms of methane mitigation strategies

Regrettably, only a limited number of studies have effectively utilized integrated multi-omics approaches to explore and understand methane emissions. While most of these studies have relied on well-established metagenomics techniques (Table 2), those that have incorporated metatranscriptomics and other advanced methods have demonstrated the unique advantages and added value of a multi-omics strategy. Specifically, researchers have employed a combination of metagenomics and metatranscriptomics to begin uncovering the intricate relationships among the ruminal microbiota, CH4 output, and feed efficiency of ruminants. Pitta et al. (Reference Pitta, Indugu and Melgar2022) utilized metagenomic and metatranscriptomic techniques to assess the effects of 3-nitrooxypropanol (3-NOP) on ruminal microbial structure. The key findings indicate that while metagenomic data revealed a high abundance of Methanobrevibacter species, metatranscriptomic analysis showed that Methanosphaera and other methylotrophic methanogens had a higher relative contribution to methane production than previously understood. This suggests that methylotrophic pathways, which were more active than indicated by gene abundance alone, play a significant role in ruminal methanogenesis. The study also found that 3-NOP led to a reduction in the expression of MCR, the enzyme critical for methane formation, highlighting the inhibitor’s effectiveness at the transcriptional level (Pitta et al. Reference Pitta, Indugu and Melgar2022). Xue et al. (Reference Xue, Xie and Zhong2022) employed an integrated meta-omics approach, combining metagenomics and metatranscriptomics, to investigate rumen microbial features associated with feed efficiency in dairy cattle. The key findings revealed that while metagenomic analysis identified the potential functional capabilities of the rumen microbiome, metatranscriptomic data provided a more accurate reflection of the active metabolic pathways. Specifically, the study found that high-efficiency cows exhibited significantly enriched metabolic pathways related to carbohydrate metabolism and energy production, such as glycolysis and the TCA cycle, in their rumen microbiomes. These pathways were more actively expressed in high-efficiency cows, suggesting that metatranscriptomics is crucial for uncovering the functional activities that directly influence feed efficiency in dairy cattle. The study of Li et al. (Reference Li, Ma and Huo2024) employed a multi-omics approach, integrating metagenomic and metatranscriptomic analyses, to investigate the microbial mechanisms underlying hydrogen and reductant disposal pathways in the rumen of Holstein and Jersey dairy cows, which have differing methane emission phenotypes. The findings revealed distinct differences between the breeds: the Holstein rumen microbiome exhibited an enhanced capacity for electron transfer associated with propionate and microbial biomass production, resulting in lower methane emissions. In contrast, the Jersey rumen microbiome showed a higher abundance of genes linked to fermentative hydrogen production and methanogenesis, leading to greater methane output.
Overview of the integrative use of meta-omics approaches – metagenomics, metatranscriptomics, metaproteomics, and metabolomics – in rumen microbiome research for methane (CH₄) mitigation. The figure illustrates how each meta-omics technique contributes to different levels of microbial analysis, including gene prediction, taxonomic profiling, functional annotation, and pathway analysis. The integration of these datasets enables the construction of various network models, such as genome-scale metabolic networks, co-occurrence networks, regulatory networks, protein–protein interaction (PPI) networks, and metabolomics-driven networks. These integrative networks provide comprehensive insights into the rumen microbiome’s structure and function, ultimately facilitating targeted microbiota manipulation strategies aimed at reducing methane emissions in ruminants.

Integrating multi-omics data poses considerable challenges, largely because of the growing complexity and variability in data structure, measurement depth, and the possibility of errors. Successfully navigating this integration demands the use of advanced bioinformatics tools and robust statistical approaches, such as multivariate analysis and machine learning techniques (for further details, readers can refer to relevant reviews (Franzosa et al. Reference Franzosa, Hsu and Sirota-Madi2015; Mallick et al. Reference Mallick, Ma and Franzosa2017; Valles-Colomer et al. Reference Valles-Colomer, Darzi and Vieira-Silva2016)). Integrating multi-omics data is a complex task due to the diversity and volume of data types involved. Approaches commonly used include correlation analyses like Pearson’s and Spearman’s rank correlations, which help identify relationships between datasets. In addition, methods such as correlation-based network analyses are frequently employed to reveal connections between variables across different omics layers. More advanced techniques, like multivariate statistical methods, play a crucial role in pinpointing key features that correlate across multiple datasets (Li et al. Reference Li, Ma and Huo2024; Xue et al. Reference Xue, Sun and Wu2020, Reference Xue, Xie and Zhong2022). These methods include partial least squares regression, orthogonal partial least squares, and nonmetric multidimensional scaling. Each of these approaches is tailored to extract meaningful patterns from the complex data, offering insights into how different omics layers interact with each other, which is essential for developing more comprehensive biological models. To assess the similarity or correlation between these datasets, techniques like Procrustes analysis and multiple co-inertia analysis are utilized (Blanco-Míguez et al. Reference Blanco-Míguez, Fdez-Riverola and Sánchez2019). While omics research has significantly expanded our understanding of the rumen microbiome, translating these findings into practical strategies remains a challenge. Much of the current research, particularly from metataxonomics and metagenomics, tends to be descriptive and often relies on basic correlation analyses. This approach can overlook the complex interactions within microbial communities. Moreover, the use of relative abundance for data normalization does not yield absolute quantifications, complicating the interpretation of results. Additionally, many identified microbial taxa and genes have unclear or unknown functions, limiting their utility in practical applications. To overcome these challenges, it is essential to enhance the integration of meta-omics results and refine analysis methods. This involves not only improving the accuracy of data interpretation but also focusing on uncovering the functional roles of under-characterized microbial species and genes. By advancing these areas, we can better leverage meta-omics to manipulate the rumen microbiome, ultimately leading to improved feed efficiency and reduced methane emissions in livestock.
To enhance our comprehensive understanding of the rumen ecosystem and translate genomic data into actionable predictive models for sustainable ruminant farming, it is crucial to employ modeling approaches that incorporate causality to clarify the mechanisms governing rumen metabolism. In this regard, genome-scale metabolic networks, co-occurrence networks, regulatory networks, protein–protein interaction networks, and metabolomics-driven networks have been widely utilized to dissect and interpret meta-omics data. In this context, genome-scale metabolic networks, co-occurrence networks, regulatory networks, protein–protein interaction networks, and metabolomics-driven networks have been extensively applied to analyze meta-omics data (as illustrated in Fig. 3). These modeling approaches have significantly contributed to our understanding of the dynamic behavior of methanogens, the thermodynamic limitations within the rumen, and the effects of various methane inhibitors on fermentation processes and methane output (Muñoz-Tamayo et al. Reference Muñoz-Tamayo, Giger-Reverdin and Sauvant2016; van Lingen et al. Reference van Lingen, Fadel and Moraes2019). Additionally, modeling techniques have been employed to investigate the ecological interactions within methanogen communities in the rumen (Muñoz-Tamayo et al. Reference Muñoz-Tamayo, Chagas and Ramin2021; van Lingen et al. Reference van Lingen, Fadel and Yáñez-Ruiz2021). However, despite continuous advancements, current rumen fermentation models have yet to fully integrate the genomic information available for the rumen microbiome. This gap highlights a significant disconnect between the metabolic functions represented in existing rumen models and the wealth of omics data that have been generated for rumen microorganisms. Bridging this gap is critical for enhancing our comprehensive understanding of rumen metabolism (Davoudkhani et al. Reference Davoudkhani, Rubino and Creevey2024; Muñoz-Tamayo et al. Reference Muñoz-Tamayo, Davoudkhani and Fakih2023).
Despite the significant challenges involved, there have been notable advancements in the field, including the development of genome-scale metabolic reconstructions and tools specifically designed for modeling and predicting microbiome metabolism (Fakih et al. Reference Fakih, Got and Robles-Rodriguez2023). Additionally, databases focusing on inter-species metabolic networks have been established (Jia et al. Reference Jia, Zhu and Xue2024), providing valuable resources for understanding microbial interactions within ecosystems like the rumen. Recent research has also begun to harness the power of advanced machine learning techniques, such as random forest algorithms, to distinguish between high and low methane-emitting ruminants and to pinpoint key factors that influence methane production (Peng et al. Reference Peng, May and Abeel2023; Zhang et al. Reference Zhang, Lin and Moraes2023). These machine learning approaches are set to transform our capacity to integrate and interpret complex multi-omics data. By leveraging these advanced analytical tools, researchers can more effectively identify strategies for reducing methane emissions in ruminant livestock, thereby contributing to more sustainable livestock production practices.
Perspectives
The development and combination of meta-omics have greatly deepened our knowledge of the intricate rumen microecosystems and their contributions to methane production. Meta-omics collectively provide comprehensive insights into the taxonomic composition, functional potential, active gene expression, protein dynamics, and metabolic profiles of rumen microorganisms. These detailed insights are crucial for unraveling the intricate biological processes and interactions that contribute to methane emissions in ruminants.
By elucidating the specific microbial pathways and key functional players involved in methanogenesis, meta-omics approaches facilitate the identification of novel targets for mitigation strategies. This deeper understanding enables the development of more precise and effective interventions, such as tailored dietary modifications, optimized use of feed additives, and the design of specific inhibitors that can disrupt methane production pathways without adversely affecting animal health or productivity. For instance, knowledge gained from meta-omics studies can inform the application and enhancement of methane inhibitors like plant extracts, 3-NOP, and bromoform by revealing their mechanisms of action and potential impacts on the rumen microbiome.
Furthermore, integrating multi-omics data through advanced computational and network modeling approaches offers the potential to predict and manipulate rumen microbial functions more accurately. Such integrative analyses can lead to the discovery of synergistic strategies that combine microbial modulation with conventional mitigation techniques, ultimately contributing to sustainable and environmentally friendly livestock production systems. Despite these promising developments, challenges remain in translating meta-omics findings into practical applications. Issues such as data complexity, standardization of methodologies, and the need for large-scale, longitudinal studies must be addressed to fully harness the potential of these technologies. To advance methane mitigation strategies in ruminant livestock, future research should prioritize the refinement of analytical tools and the enhancement of data integration techniques. Additionally, fostering interdisciplinary collaborations will be crucial in bridging the gap between fundamental microecology and practical applications. This approach will facilitate the development of more effective and sustainable solutions for reducing methane emissions in ruminants, leveraging insights from various fields to tackle this complex issue comprehensively.
In summary, while meta-omics technologies may not directly lower methane emissions in ruminants, they are crucial for unraveling the complex microbial interactions within the rumen that contribute to methane production. These insights are instrumental in shaping targeted, science-based mitigation strategies. As we continue to invest in and refine these advanced techniques, their application will be vital for achieving meaningful and sustainable reductions in methane emissions from livestock, aligning with global climate change mitigation efforts.
Acknowledgements
This study was supported by National Key Research and Development Program of China (2023YFD1301801) and Beijing Capital Agribusiness & Food Group Co., Ltd. “Integrated Innovation and Industrial Application of ‘Green Digital Intelligence’ Technology in the Whole Dairy Industry Chain” (SNSPKJ2022).
Author contributions
HYZ, SB, JT: writing-original draft, funding acquisition; ML: writing-review & editing; YCZ: conceptualization, methodology; LSJ: supervision, writing-review & editing. All authors approved the final manuscript.
Huiying Zhao and Sarula Bai authors are contributed equally to this work.
Conflict of interest
The authors declare no conflict of interest.