Introduction
Developmental programming is the concept that factors affecting development, primarily during the fetal and neonatal periods (although gametes can also be affected), do so by altering gene expression and therefore development and function of various organ systems in the short- and long-term. Reference Sinclair, Rutherford and Wallace1–Reference Caton, Crouse and Dahlen5 In addition, such effects on organ development and function can affect the health and well-being of the offspring during adulthood and even among subsequent generations. Reference Barker6,Reference Godfrey, Gluckman and Hanson7 Based on initial epidemiological studies in humans, parental nutrition was hypothesized to have a major influence on developmental programming, and subsequent epidemiological studies in humans as well as controlled studies in various animal models have confirmed this hypothesis Reference Dahlen, Ramírez-Zamudio and Bochantin-Winders4,Reference Caton, Crouse and Dahlen5,Reference Reynolds and Vonnahme8–Reference Caton, Crouse and Reynolds11 (see summary Table 1 where we have highlighted the repeatability of findings and effects within each topic covered herein).
Some livestock models of maternal nutrition and developmental programming

Much of the focus in developmental programming research has been on its negative effects on health, productivity, and even longevity of the offspring. However, it has been argued that developmental programming has remained in the genome because it is ultimately adaptive. Reference Reynolds, Diniz and Crouse9,Reference Dahlen, Borowicz and Ward12,Reference Reynolds, McLean, McCarthy and Wu13 In the long run, the effects of developmental programming must be positive overall. This is an important concept if we are to understand and perhaps even develop strategies to manage developmental programming for potential benefits to animal agriculture, as well as human health and wellbeing.
Our focus in this manuscript is to: 1) briefly review maternal nutrition-driven, and to some extent paternal nutrition-driven, livestock models of developmental programming with an understanding that these models are dual purpose with dual benefit for both livestock productivity and human health, 2) address placental development and its potential effects on nutrient supply to the conceptus, 3) describe the programming of the genetic and epigenetic landscape, 4) discuss programming of key tissues, including viscera and skeletal muscle, and the immune system, 5) discuss the programming of parturition, and 6) suggest future research directions and needs.
Livestock models of developmental programming: dual purpose with dual benefit for both livestock and humans
Livestock models have and will continue to contribute significantly to our understanding of the fundamental biological mechanisms associated with growth, development, and well-being. Accordingly, they will continue to provide a dual purpose with a dual-benefit approach to addressing critical questions in agricultural and biomedical sciences. Reference Reynolds, Ireland, Caton, Bauman and Davis14,Reference Ireland, Roberts, Palmer, Bauman and Bazer15 The journal “Animal Frontiers” has dedicated two entire issues on developmental programming and farm animals as biomedical models (“Developmental programming: What mom eats matters!,” Volume 7, Issue 3, July 2017 Reference Zinn, Govoni and Vonnahme16 ; and “Farm animals are important biomedical models, Volume 9, Issue 3, July 2019” which presents a collection of reviews on that very topic Reference Hamernik17 ). Specifically, the use of livestock models of maternal nutrition in developmental programming paradigms has contributed significantly to the field by leveraging insights into both agricultural production practices and human biomedicine.
A detailed presentation of all aspects of nutrition and developmental programming in livestock is beyond the scope of this review; however, several excellent reviews Reference Dahlen, Ramírez-Zamudio and Bochantin-Winders4,Reference Caton, Crouse and Dahlen5,Reference Reynolds and Vonnahme8,Reference Reynolds, Diniz and Crouse9,Reference Caton, Crouse and McLean18–Reference Caton, Crouse and Dahlen20 and several journal article collections (Animal Frontiers, “Developmental programming: What Mom eats Matters,” Reference Zinn, Govoni and Vonnahme16 ; Veterinary Clinics of North America, Food Animal Practice, Developmental Programming in Livestock Production,” Reference Funston and Mulliniks21 ; and Frontiers in Genetics, “Genomic Basis of Developmental Programming in Livestock, Insights into Nutrition, Health, and Production” Reference Diniz, Ward and Du22 ) have been published recently, and readers are referred to those publications for additional information and discussion.
Livestock models of developmental programming include global nutrition (i.e., over- or under-nutrition) and specific macronutrient and micronutrient excess or deficiency. In addition, strategic supplementation of nutrients has been successful in mitigating these negative consequences of developmental programming in both humans Reference Wilson and O’Connor23 and livestock. Reference Syring, Crouse and Entzie24–Reference Safain, Crouse and Syring27 These models have also focused on differing periods, or “windows,” of fetal/offspring development, which is logical as tissues and systems develop at different rates and times during gestation and postnatally. Reference Caton, Crouse and McLean18,Reference Hammer, Caton, Dahlen, Ward, Borowicz and Reynolds19,Reference Fowden, Giussani and Forhead28–Reference Dahlen, Amat, Caton, Crouse, Diniz and Reynolds30 Most studies have investigated the effects of maternal nutrition on developmental programming during mid and late gestation rather than early gestation; however, the periconceptional period and very early gestation are likely critical windows of susceptibility to long-term developmental programming events. Reference Caton, Crouse and McLean18 Taken together, the preponderance of data indicates that developmental programming affects multiple organ systems, transcription of many genes, epigenetic landscape, metabolic outcomes, production and performance measures, and health and longevity of the offspring. Reference Reynolds, Diniz and Crouse9,Reference Diniz, Reynolds, Ward and Vaschetto10,Reference Reynolds, McLean, McCarthy and Wu13,Reference Hammer, Caton, Dahlen, Ward, Borowicz and Reynolds19,Reference Dahlen, Amat, Caton, Crouse, Diniz and Reynolds30,Reference Reynolds, Dahlen and Ward31 Emerging data with livestock models have confirmed that some effects of developmental programming can be multigenerational. Reference Dahlen, Ramírez-Zamudio and Bochantin-Winders4,Reference Caton, Crouse and Dahlen5,Reference Reynolds, Diniz and Crouse9,Reference Reynolds, McLean, McCarthy and Wu13,Reference Caton, Crouse and McLean18–Reference Caton, Crouse and Dahlen20,Reference Baumgaertner, Ramírez-Zamudio and Menezes32
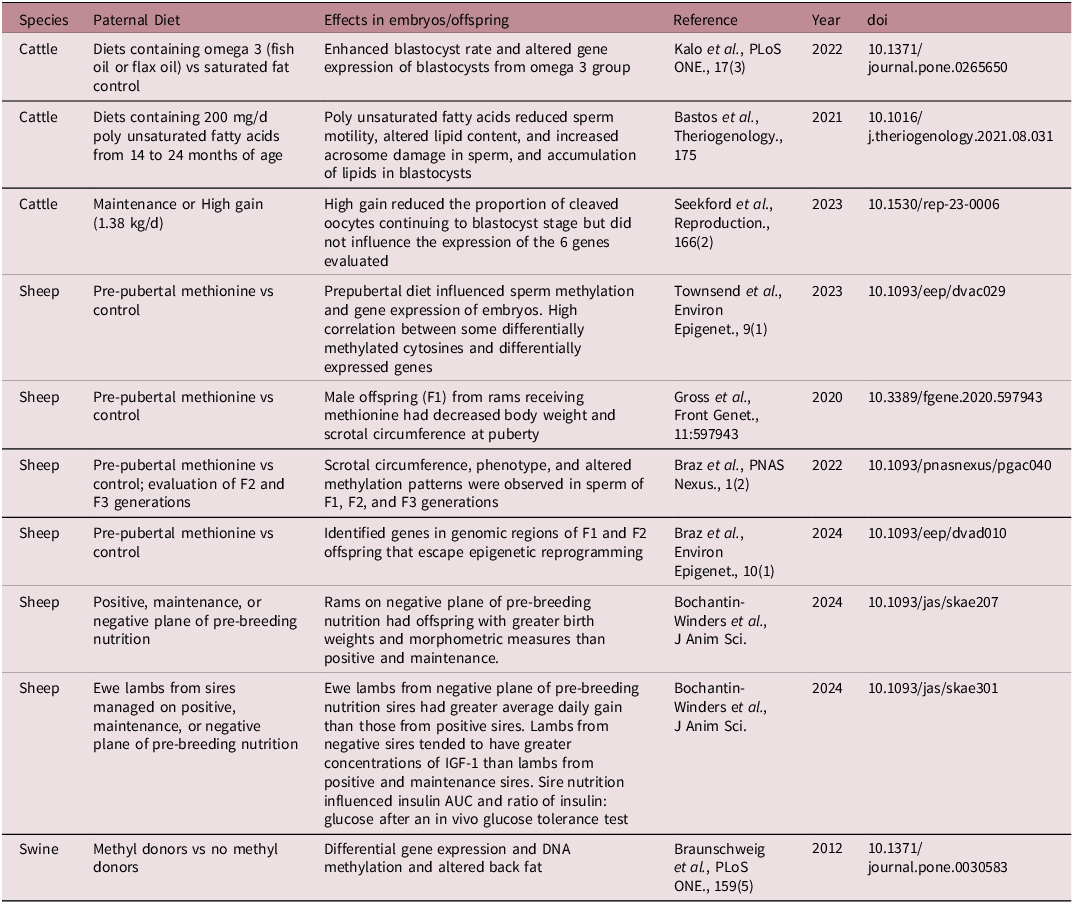
Though there are extensive data regarding the contributions of maternal nutrition on the developing offspring, the body of evidence regarding the contributions of sire nutrition during spermatogenesis is also growing (see summary in Table 2). The mode of action of paternal programming is likely through alterations in sperm miRNA, DNA methylation, histone modifications, oxidative stress/DNA damage, cytokine response, and/or seminal microbiome that subsequently impacts the embryo directly at conception or indirectly via the female immune response. Reference Dahlen, Amat, Caton, Crouse, Diniz and Reynolds30,Reference Kilama, Dahlen, Reynolds and Amat33 Feedstuffs providing substrates for epigenetic processes (methionine, folic acid, and other methyl donors) have been shown to affect embryo transcript abundance, DNA methylation, embryo production and epigenetic properties, and offspring phenotypes in swine, sheep, and cattle models. Reference Toschi, Capra and Anzalone34–Reference Braz, Passamonti and Khatib38 In addition, models of dietary alteration in polyunsaturated fatty acids have resulted in differences in embryo lipid content and gene expression in cattle. Reference Kalo, Reches and Netta39,Reference Bastos, Rossi and da Silva Leão40 Managing bulls for an increased rate of gain also reduced the proportion of embryos that reached the blastocyst stage of development, Reference Seekford, Davis and Dickson41 and models providing a common diet to rams and managing for either weight loss, maintenance, or gain resulted in alterations in lamb birth weight, daily gain, and metabolic response to a glucose challenge. Reference Bochantin-Winders, Baumgaertner and Hurlbert42,Reference Bochantin-Winders, Slavick and Jurgens43
Summary of livestock paternal nutrition models demonstrating paternal programming effects on embryos and offspring 1

1 Adapted from Dahlen et al., 2023 and Dahlen et al., 2024.
IGF-1, insulin like growth factor 1.
Placental function and developmental programming
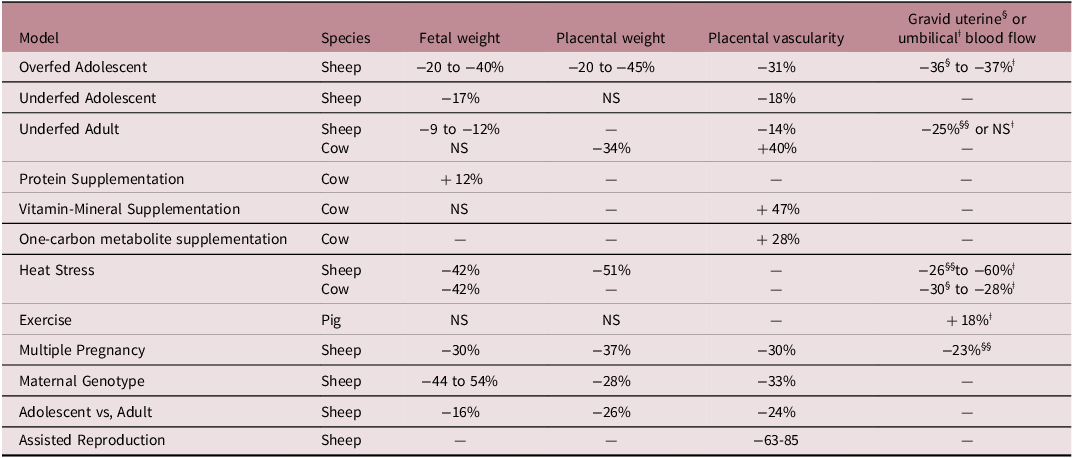
Several reviews have been published examining the role of the placenta in developmental programming, Reference Reynolds, Borowicz, Caton, Crouse, Dahlen and Ward2,Reference Reynolds, Dahlen and Ward31,Reference Reynolds, Caton and Redmer44–Reference Steinhauser, Askelson, Lambo, Hobbs, Bazer and Satterfield47 and the reader is referred to these for further details. Suffice it to say that there is strong evidence supporting placental development, especially vascular development and blood flow, which are major factors in developmental programming of fetuses, and therefore, in offspring development, health, productivity, and longevity. For example, in every model of pregnancy in which fetal and/or placental growth are compromised, as reflected by their weights, placental vascular development and blood flow are also affected. These models include overfed adolescents, underfed adolescents and adults, multiple fetuses, maternal genotype, maternal age, maternal heat stress, and assisted reproductive techniques (Table 3). In addition, in several models in which fetal or placental weights are unaffected (underfed adult cows, vitamin-mineral supplementation, one-carbon metabolite supplementation, and maternal exercise), placental vascular development or blood flows exhibit compensatory increases (Table 3). This latter observation implies not only that the placenta is capable of compensatory responses to maternal stressors, as we and others have argued before, Reference Reynolds, Diniz and Crouse9 but also that developmental programming responses can indeed be adaptive.
Fetal and placental weights (see footnote about stage of gestation 1 ), placental vascularity and placental (gravid uterine§ or umbilicalǂ) blood flows in various models of maternal stress in livestock

1 For sheep, samples were collected on day 130–135 (0.87–0.90) of gestation, except for Assisted Reproduction, which were collected on day 22 (0.15) of gestation; for cows, samples collected on day 63 (0.22; one-carbon metabolite supplementation) or 250 (0.89; undernutrition) of gestation or at parturition (vitamin-mineral supplementation); for pigs, samples collected on day 94 to 104 of gestation. Table adapted from Reynolds et al., 2023; https://doi.org/10.1016/j.anireprosci.2023.107322).
NS, not significant P > 0.05.
Programming of gene expression and epigenetic landscape
The interplay between genetics and environment is central to a long-standing nature versus nurture debate. However, epigenetics has provided a new perspective to this debate, the mechanism to explain the origins of developmental programming or the DOHaD hypothesis, and new paradigms on genetic inheritance. Reference Beckett, Yates, Veysey, Duesing and Lucock48 Epigenetics has been defined as molecular factors and processes that can affect genome activity and gene expression without changes in the DNA sequence. Reference Thompson, Nilsson and Skinner49 Environmental factors, such as parental nutrition, can potentially leave epigenetic marks, or epimutations, that can be inherited through subsequent developmental stages and likely across generations. Epigenetic modulation of gene expression is likely the mechanism through which nutrient supply programs fetal development. Reference Diniz, Reynolds, Ward and Vaschetto10,Reference Aiken and Ozanne50–Reference Reynolds, Ward, Caton, Scanes and Hill52 However, the mechanisms, extent, and potential for transmitting epigenetic information across generations remain unclear. Based on our current knowledge, epigenetic marks are established during gametogenesis, including DNA methylation, non-coding RNAs (ncRNAs), and histone modifications. Reference Carrell and Hammoud53–Reference Watkins, Dias and Tsuro56 These mechanisms can regulate gene expression at the transcriptional level via DNA methylation and/or histone modifications and the translational level via ncRNAs. Reference Schagdarsurengin and Steger54 DNA methyltransferases (DNMTs) are key enzymes involved with maintenance and de novo DNA methylation. Reference Carrell and Hammoud53 It is important to note that during fertilization, germ cells deliver not only their haploid genomes but also their epigenomes, which contain codes for pre- and post-implantation reprogramming and embryonic development. Reference Schagdarsurengin and Steger54 This is of particular importance in the realm of developing programming, considering the parental epigenome will be transferred to their offspring. In fact, females’ oocytes will be developed and fixed in utero, therefore postnatal recovery or alterations are not possible. As such, it is likely negative alterations in the epigenome will likely be trans-generational.
Nutrients can interact directly with the genome through nutrient-sensing signaling pathways and nutrient-dependent genes or indirectly via epigenetic mechanisms. Epigenetic signatures in somatic tissues can be changed through enzymes that add or remove “epigenetic tags” to or from DNA and histones. Reference Carrell and Hammoud53,Reference Schagdarsurengin and Steger54 Our previous review of the molecular mechanisms and tissue adaptation in developmental programming discusses these interactions. Reference Diniz, Reynolds, Ward and Vaschetto10 In-depth reviews on the effects of specific nutrients, such as minerals and one-carbon metabolites, have also been published. Reference Braz, Taylor, Namous, Townsend, Crenshaw and Khatib37,Reference Wessels57–Reference Crouse, Freetly and Lindholm-Perry61 Mounting evidence has highlighted differential gene regulation and expression of the brain, liver, and muscle in the offspring in response to in utero nutrition. Reference Peñagaricano, Wang, Rosa, Radunz and Khatib62–Reference Palmer, Peñagaricano and Vedovatto65 We previously reported that nutrient restriction from breeding to day 50 of gestation affected the expression of fetal hepatic genes, including members of the histone H1, H2A, H2B, and H4 families. Reference Crouse, Caton and Cushman63 The 13 differentially expressed histone-modifying genes included those associated with acetylation and de-acetylation, methylation, phosphorylation, and ubiquitination. Interestingly, in skeletal muscle, we reported that the rewiring of major regulators likely modulates the expression of gene targets as an adaptive response to nutrient availability, affecting tissue development. For example, the myogenic regulatory factors MYOD1, MYF5, and MYOG were negatively and differentially correlated with ZBTB33 and/or ZNF131 transcription factors in nutrient-restricted fetuses, likely negatively affecting myogenesis. Reference Diniz, Crouse and Cushman64 These findings are relevant to human health and wellbeing because of the widespread malnutrition that still exists among the population, which is contributing to the programming of future generations as discussed previously. Furthermore, these nutritional impacts on skeletal muscle are of concern when considering both human and animal skeletal muscle in the landscape of overall health and wellbeing among the population, which is discussed in further detail within this review.
Most of the epigenomic studies related to fetal programming have focused on the F1 generation. Maternal nutrient restriction during an 84-d period of mid-gestation did not affect growth or development in beef cattle. Reference Gardner, Ineck and Quarnberg66 On the other hand, at weaning and in early adolescence, the expression of micro-RNAs (miRNAs) previously associated with the growth and/or development of skeletal muscle and/or adipose tissue was affected by the in utero environment. Reference Gardner, Ineck and Quarnberg66 Research in sheep has shown that under-nutrition 60 days before to 6 days after conception programs changes in insulin-signaling molecules and the expression of miRNAs in skeletal muscle and liver during late gestation (days 136–138). Reference Lie, Morrison and Williams-Wyss67,Reference Lie, Morrison and Williams-Wyss68 Transcriptional and post-transcriptional gene silencing is modulated by miRNAs through specific base pairing with target genes. Reference Wang Kevin and Chang Howard69 Developmental and physiological processes, including myogenesis, also are modulated by miRNAs. Reference Raza, Kaster and Khan70 Furthermore, miRNAs can modulate the function of components of the epigenetic machinery, interacting to form a complex, bi-directional regulatory circuit that regulates gene expression. Reference Beckett, Yates, Veysey, Duesing and Lucock48 Interestingly, in humans and mice, it has been shown that miRNAs can be trans-placentally transmitted from the mother to the fetus. Reference Li, Zhang and Li71 However, we still have a paucity of knowledge regarding mechanisms by which miRNA modulates gene expression in livestock models and its impacts on health and wellbeing.
DNA methylation is one of the most studied epigenetic mechanisms. Most of the evidence of programming through DNA methylation is related to exposure to toxicants and maternal nutritional changes. Reference Beckett, Yates, Veysey, Duesing and Lucock48,Reference Thompson, Nilsson and Skinner49,Reference Skinner, Haque, Nilsson, Bhandari and McCarrey72 Maternal supplementation with epigenetic modifiers (e.g., methionine, choline, folate, and vitamin B12) has been examined because of their roles as methyl donors or cofactors in methylation reactions. Reference Crouse, Caton and Claycombe-Larson73 Supplementation of choline to bovine embryos during in vitro fertilization resulted in a tendency for positive effects at birth and improved weaning weights. Reference Estrada-Cortés, Ortiz and Rabaglino74 These authors also reported differential methylation of genes involved with the mTOR signaling pathway, a key regulator of muscle development. Interestingly, maternal methionine supplementation during the periconceptual period through the completion of the first trimester of gestation led to persistent methylation differences over time in beef calves. Reference Amorín, Liu, Moriel, DiLorenzo, Lancaster and Peñagaricano58 Moreover, methionine supply during mid-gestation affected the expression and activity of placental nutrient transporters in a sex-dependent manner and upregulated placental mTOR and phosphorylated mTOR; however, changes in DNA methylation were not investigated. While these studies demonstrate DNA methylation is involved in developmental programming, its long term effects on growth, metabolism, and health and wellbeing are largely unexplored.
In sheep, Peterson et al. (2021) Reference Peterson, Gauvin, Pillai, Jones, McFadden, Cameron, Reed, Zinn and Govoni59 examined the effects of maternal under- and over-nutrition during gestation on offspring pancreas development and DNA methylation. They reported differential DNA methylation in fetal pancreas collected at days 90 or 135 of gestation or within 24 h of birth. Their findings suggest that DNA methylation patterns are diet- and sex-dependent. These results indicate that maternal nutrition during gestation significantly affects fetal epigenetic programming and gene expression. Furthermore, supplementation with key nutrients, such as choline and methionine, positively affects growth and performance Reference Estrada-Cortés, Ortiz and Rabaglino74 and can have long-term effects on growth and metabolic efficiency. Reference Amorín, Liu, Moriel, DiLorenzo, Lancaster and Peñagaricano58,Reference Haimon, Estrada-Cortés and Amaral75
There is a paucity of information related to the effects and mechanisms of intergenerational inheritance of epigenetic marks in livestock. This has been primarily influenced by limited data suggesting that parental nutrition, either prior to or during pregnancy, can affect growth and carcass traits in food-producing livestock. Reference Diniz, Reynolds, Ward and Vaschetto10 Additionally, the literature suggests there might be a lag time from “insult” to observable effects in offspring performance. Reference Dahlen, Amat, Caton, Crouse, Diniz and Reynolds30 Thus, as animals are slaughtered for human consumption, it is possible that long-term developmental programming effects may not have had enough time to manifest in livestock. However, some studies have shown transgenerational epigenetic inheritance in livestock. Braunschweig et al., Reference Braunschweig, Jagannathan, Gutzwiller and Bee35 investigated the effect of a paternal, methyl donor-enriched diet on subsequent generations. They reported significant changes in subcutaneous adipose, gene expression, and DNA methylation in the experimental compared with the control group. Reference Braunschweig, Jagannathan, Gutzwiller and Bee35
Similarly, Braz et al., Reference Braz, Taylor, Namous, Townsend, Crenshaw and Khatib37 reported 107 differentially methylated cytosines (DMCs) that were common between the F1 and F2 generations when F0 rams were supplemented with methionine from weaning to puberty. The authors reported that most long-term phenotypic negative effects were observed in the F2 generation. In F2 females, weaning and postweaning body weights were negatively affected by the paternal diet. Similar negative effects were observed in the F2 male’s loin muscle depth. Furthermore, the paternal diet reduced the testicular size in both F1 and F2 generations compared with the control group. Reference Braz, Taylor, Namous, Townsend, Crenshaw and Khatib37 In a follow-up study of the same experimental population, the authors reported that the DMCs were found in transposable elements, non-repetitive regions, and both imprinted and non-imprinted genes. They suggested that some genomic regions could escape reprogramming and serve as potential candidates for transgenerational epigenetic inheritance. New knowledge is needed to better understand long-term impacts for biomedicine, and transgenerational impacts for biomedicine and the production of animal products for human consumption.
In addition to the mechanisms described above, growing evidence suggests the role of the maternal microbiota in fetal developmental programming. However, the mechanisms underlying the regulation of the maternal and fetal microbiomes and their effects on fetal genome programming are yet to be determined. Data from beef cattle indicate that colonization with pioneer microbiota may occur during early gestation in bovine fetuses and that the maternal nutritional regime during gestation may influence the early fetal microbiota. Reference Amat, Holman and Schmidt77 Preclinical studies in mice have revealed the effects of the maternal microbiome on prenatal development through the transfer of microbial metabolites, Reference Radford-Smith and Anthony78 which affected fetal metabolism, immunity, and neurodevelopment. Reference Jašarević, Howerton, Howard and Bale79,Reference Macpherson, de Agüero and Ganal-Vonarburg80 Gustafson et al., Reference Gustafson, Busi and McAdams81 showed that the maternal gut microbiome affected the neurodevelopment, behavior, and body weight of the offspring in mice. They showed differences in hippocampal gene expression within differentially methylated regions and in genes underlying pathways associated with growth and feeding behavior. Further research, however, is still needed using livestock models to determine the effects and extent of maternal microbiomes on colonizing and programming the offspring.
With the development and refinement of methods and cost effectiveness to explore gene expression and the epigenetic landscape, it has become a rapidly evolving field of study. As such, emerging data suggests its role in developmental programming, particularly concerning trans-generational impacts. Nevertheless, we look to future findings to confirm these initial findings.
Programming of key metabolic tissues
Maintenance energy use is likely susceptible to developmental programming in utero. Reference Caton, Crouse and Reynolds11,Reference Carstens, Johnson, Holland and Odde82 In general, nutrient demand follows energy demand and energetic efficiency often reflects the overall efficiency of nutrient use. Tissue metabolic rate affects the rate of nutrient use. Consequently, more metabolically active tissues consume greater proportions of nutrients compared with less metabolically active tissues on a per unit of tissue basis. Specifically, in cattle, the portal drained viscera consume approximately 40% of maintenance energy demands but represent a comparatively small proportion of overall body weight. Within the viscera, the liver and intestinal tissues each consume approximately half of the 40%. Conversely, skeletal muscle represents a large portion of overall body weight and is metabolically active but consumes between 20 and 25% of maintenance energy. These concepts are relevant because small changes in nutrient energy use for maintenance can shift energy (and other nutrients) toward product formation and improve production efficiencies. In addition, metabolically active tissues appear to be responsive to nutritional planes, Reference Ferrell83,Reference Koong, Ferrell and Nienaber84 and consequently may be programmed by inappropriate maternal nutrition during gestation.
Programming of visceral tissues
Visceral tissues are responsive to nutritional plane. Reference Jin, Reynolds, Redmer, Caton and Crenshaw85–Reference Caton, Bauer and Hidari87 Likewise, in ruminant livestock, differing nutritional planes during gestation can have large impacts on maternal visceral tissues, including the liver and intestine. Reference Scheaffer, Caton, Redmer, Arnold and Reynolds88–Reference Meyer, Reed and Neville91 As discussed below, the data also indicate that maternal plane of nutrition during gestation can influence visceral tissues of the offspring both pre- and post-natal.
Offspring liver
Proper liver function is critical for efficient metabolic function, particularly concerning glucose, which is a key nutrient for skeletal muscle growth. Data from a sheep model of compromised maternal nutrition and developmental programming of offspring Reference Prezotto, Thorson and Borowicz92,Reference Prezotto, Lemley and Camacho93 demonstrated that liver oxygen consumption at both d 130 of gestation and d 54 postnatal is reduced in offspring from nutrient-restricted compared with control fed dams. In beef heifers subjected to maternal nutrient restriction during pregnancy, Safain et al., Reference Safain, Hirchert and Ward94 showed that mitochondrial respiration rates from d 161 fetuses were increased in response to dietary supplementation with methionine, choline, folate and vitamin B12 from d 0 to 63 of gestation. Their work agrees with earlier data Reference Crouse, Caton and Claycombe-Larson73 showing that mitochondrial respiration was responsive to both planes of nutrition and one-carbon metabolite supplementation in a bovine cell culture model.
Fetal hepatic transcript abundance was also influenced by maternal nutritional intake during early pregnancy, Reference Crouse, Caton and Cushman63 with pathway analysis demonstrating changes in gene expression of energy, amino acids, carbohydrates, and other metabolic pathways. Research Reference Syring, Crouse and Entzie24,Reference Safain, Crouse and Syring27,Reference Crouse, McCarthy and Menezes95 has investigated the influence of moderate nutrient restriction of beef heifers during early gestation on metabolomic profiles in fetal liver. Their work demonstrates that changes in maternal nutrition during early gestation can alter fetal hepatic gene expression and metabolic profiles within multiple pathways, including carbohydrate, amino acid, and energy metabolism. However, research is lacking regarding the postnatal implications of altered fetal liver metabolism on growth, efficiency and health outcomes in the offspring.
Offspring small intestine
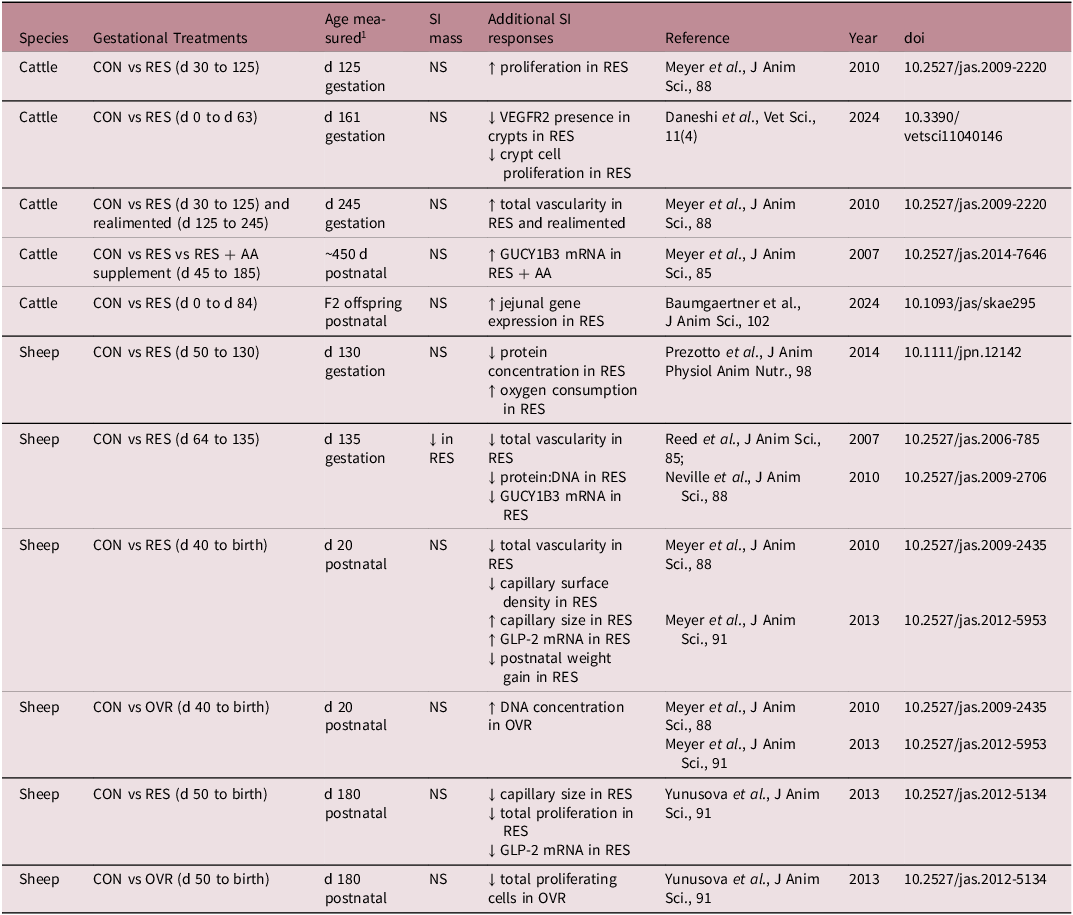
Healthy offspring intestinal tissues are essential to neonatal survival and growth. Consequently, understanding the development and potential programming of these tissues by compromised maternal nutrition should inform management decisions and improve offspring outcomes and growth efficiencies. As shown in Table 4, research over the past decade demonstrates that both pre- and post-natal intestinal tissues are responsive to maternal plane of nutrition and appear to be functionally programmed postnatally. Reference Meyer, Reed and Vonnahme96–Reference Yunusova, Neville and Vonnahme99 Specifically, fetuses from nutrient restricted sheep had reduced small intestine vasculature, protein concentration, protein to DNA ratio, and oxygen consumption. Reference Reed, Ward and Vonnahme89,Reference Prezotto, Lemley and Camacho93 Similarly, fetuses from nutrient restricted cows that were realimented had increased small intestine vascularity but decreased VEGFR2 in crypts, and decreased crypt cell proliferation. Reference Daneshi, Borowicz and Entzie26,Reference Meyer, Reed and Vonnahme96 Moreover, lambs born from nutrient restricted ewes had decreased small intestine capillary surface density and size, total vascularity, cell proliferation, and weight. Reference Meyer, Reed and Vonnahme96,Reference Meyer, Neville and Reed97,Reference Yunusova, Neville and Vonnahme99 Whereas lambs born from overnourished ewes had increased small intestine DNA content but reduced cell proliferation. Reference Meyer, Reed and Vonnahme96,Reference Yunusova, Neville and Vonnahme99 Interestingly, recent work in a heifer model of cross-generational developmental programming has demonstrated that F2 offspring exhibit altered gene expression and morphometrics in jejunal tissues. Reference Baumgaertner, Ramírez-Zamudio and Menezes32,Reference Craner, Dahlen and Hurlbert100,Reference Menezes, McCarthy and Kassetas101 Detailed investigation of intestinal function and health as affected by maternal stress, particularly nutritional stress, during critical windows of development will increase our understanding of both maternal and offspring management.
Summary of effects of maternal nutrition on ruminant offspring small intestine (SI) from selected studies

1 Approximate gestation lengths: cattle = 285 d, sheep = 150 d, CON = control, maintenance diet; GLP-2 = glucagon-like peptide 2; GUCY1B3 = soluble guanylate cyclase (NO receptor); NS = not significant (P > 0.10); OVR = over nutrition; RES = nutrient restriction; RES + AA = nutrient restriction with protein supplementation to meet essential AA of control; VEGFR2 = vascular endothelial growth factor receptor 2.
Offspring skeletal muscle
Proper skeletal muscle development in utero and subsequent growth postnatally is important for human development. In addition, livestock skeletal muscle is an important end-product of production agriculture, providing high-quality protein and essential micronutrients to the human diet. In humans, skeletal muscle mass is important to physical health, including weight management, insulin responsiveness, disease prevention, and longevity. Reference Srikanthan and Karlamangla102–Reference Lee, Kuk and Davidson104 Additionally, proper skeletal muscle development in utero is critical in livestock and humans because skeletal muscle fiber number in mammals is fixed at birth, and thus skeletal muscle growth postnatally can occur only through hypertrophy of the existing fibers. Reference Rehfeldt and Kuhn105,Reference Chaze, Meunier, Chambon, Jurie and Picard106 Consequently, if poor maternal nutrition during gestation, and even in the periconceptual period, negatively affects the development of skeletal muscle fibers, it will result in poor skeletal muscle metabolic efficiency in livestock and humans alike. This will ultimately reduce the economic and environmental sustainability of production agriculture and increase the incidence of nutritional, including non-communicable, diseases in humans. Reference Diniz, Crouse and Cushman64,Reference Rehfeldt and Kuhn105,Reference Zhu, Ford, Means, Hess, Nathanielsz and Du107–Reference Sun, Chen and Liao111 The negative effects of poor maternal nutrition on offspring skeletal muscle mass and function have been previously reviewed by Caton et al. (2024). Reference Caton, Crouse and Dahlen5
In livestock, maternal undernutrition occurs during seasonal changes in forage quality and quantity, extreme or abnormal weather events, and breeding young females that have yet to reach maturity. Reference Caton, Crouse and Reynolds11,Reference Guide112,113 In humans, food insecurity remains a critical issue globally (2.4 billion people in 2022), and within the United States (18 million households in 2023). 114,Reference Nations115 Maternal undernutrition throughout gestation has severe consequences for skeletal muscle development in utero and, thus for hypertrophy and metabolic function postnatally. Specifically, skeletal muscle satellite cells derived from offspring subjected to maternal undernutrition had increased myogenic regulatory factors, MyoD and myogenin, from birth through the juvenile stage, but reduced myoblast fusion in the juvenile stage. Reference Raja, Hoffman, Govoni, Zinn and Reed116 Offspring from undernourished pregnancies had decreased skeletal muscle fibers, primary to secondary fiber ratio, and increased transcript abundance of ATP binding and protein binding factors. Reference Gauvin, Pillai and Reed110
Additionally, mature offspring from nutrient-restricted pregnancies had reduced type IIa fibers and carnitine palmitoyltransferase-1 activity, increased type IIb fibers, increased intramuscular triglyceride content and altered protein content. Reference Zhu, Ford, Means, Hess, Nathanielsz and Du107 Furthermore, individual muscle weights and myosin heavy chain (MHC)-fast fibers were decreased but MHC-slow fibers were increased in offspring from nutrient-restricted pregnancies. Reference Fahey, Brameld, Parr and Buttery117 In addition, myofiber number, capillary density, and capillary to myofiber ratio were reduced, and insulin and insulin-like growth factor (IGF-1) receptors and glucose transporter type 4 (GLUT4) were increased in offspring from nutrient-restricted pregnancies. Reference Costello, Rowlerson and Astaman118 Maternal undernutrition was also associated with altered metabolites in the tricarboxylic acid cycle (TCA), synthesis of bioactive amines, and several other metabolic pathways in offspring skeletal muscle, which persisted even following maternal realimentation. Reference Smith, Vásquez-Hidalgo and Li119 Moreover, maternal undernutrition decreased metabolic markers, metabolites, and the function of mitochondria in offspring skeletal muscle. Reference Zou, Yu and Yu120 Finally, maternal nutrient restriction altered gene expression in offspring skeletal muscle, including upregulation of genes associated with contraction, intermediate filaments, actin, myosin, troponin, and calcium-binding, whereas genes associated with microtubules and ATP-binding were downregulated, ultimately altering biological pathways involved in hemostasis, and forebrain and skeletal muscle development. Reference Crouse, Caton and Cushman63,Reference Diniz, Crouse and Cushman64 These observations indicate that maternal nutrient restriction during gestation negatively affects skeletal muscle growth and development and skeletal muscle metabolic function, reducing metabolic efficiency and ultimately impairing muscle development in livestock as well as metabolic health in humans.
Maternal overnutrition also occurs in livestock but is probably more relevant to humans. In 2018, obesity was at an all-time high in the United States, with approximately 40% of adults of child-bearing age being obese. 121 Globally, the prevalence of overweight and obese adults has increased 4-fold since 1990, with more than 2.5 billion adults (43%) overweight and 890 million (16%) obese in 2022. 122 Maternal overnutrition and obesity affect fetal skeletal muscle growth and development and muscle function in the offspring.
Specifically, skeletal muscle protein content and adipogenic and fibrogenic markers were increased, and collagen deposition was decreased in offspring from overnourished pregnancies. Reference Gionbelli, Veloso and Rotta123,Reference Duarte, Gionbelli and Paulino124 Skeletal muscle lipid accumulation, follistatin gene expression, cross-sectional area neonatally, and type IIa fibers were increased, but type I fibers and cross-sectional area on juveniles were decreased among offspring from overnourished pregnancies. Reference Reed, Raja, Hoffman, Zinn and Govoni125 In addition, the ratio of type I to type II fibers, fiber area, and transcript abundance of insulin receptor isoform b receptors and IGF-1 receptors were increased but glucose transporters were decreased in skeletal muscle of offspring from overnourished pregnancies. Reference Bradbery, Coverdale, Hammer, Dunlap, Leatherwood and Satterfield126 Moreover, insulin receptors, IGF-1 receptors, and insulin signaling markers were decreased, but triglyceride content, fatty acid transporters, collagen content, and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling were increased in skeletal muscle of offspring from overnourished pregnancies. Reference Yan, Huang and Zhao127
Circulating glucose and insulin concentrations were also decreased in offspring from overnourished pregnancies. Reference Wallace, Bourke, Aitken, Leitch and Hay128 Similarly, plasma glucose, insulin, IGF-1, insulin receptor phosphorylation, tumor necrosis factor alpha (TNFα), and PPARγ were increased, but Akt and mTOR signaling and fiber density were reduced in skeletal muscle of offspring from overnourished pregnancies. Reference Zhu, Han and Tong129 Maternal overnutrition also alters metabolites in offspring skeletal muscle, including those associated with amino acid, peptide, carbohydrate, energy, lipid, and nucleotide metabolic pathways. Reference Martin, Jones and Pillai130 Furthermore, transcript abundance was altered in pathways associated with adipogenesis, regulation of skeletal muscle growth, differentiation, and atrophy, metabolic processes, and protein transcription factors in skeletal muscle of offspring from overnourished pregnancies. Reference Hoffman, Peck, Wegrzyn, Reed, Zinn and Govoni131 Undeniably, maternal overnutrition during gestation negatively affects offspring skeletal muscle growth and development and metabolic function, which has been demonstrated to span generations. Reference Pankey, Walton and Odhiambo132 These negative effects impair metabolic efficiency and, ultimately health and well-being of the offspring, which affect not only livestock production but also the incidence of non-communicable diseases in humans.
Dietary guidelines during pregnancy and lactation are well-defined in livestock. 113,133–136 However, nutritional requirements during pregnancy and lactation in women are alarmingly poorly defined. 137,138 Evidence suggests that adequate macronutrient consumption during pregnancy is vital for fetal and postnatal growth and development, and thus for offspring health, with protein being the most important maternal dietary nutrient for skeletal muscle development. Protein deficiency during pregnancy decreases skeletal muscle weight, myofiber cross-sectional area, circulating branched-chain amino acids, and markers of protein synthesis but increases myostatin pathway markers. Reference Liu, Pan, Li, Sun, Yang and Zhao139 Additionally, genes associated with oxidative phosphorylation, Kreb’s cycle, and fatty acid metabolism were upregulated, whereas genes associated with the cell cycle were downregulated, in skeletal muscle of offspring from pregnancies in which the maternal diet was protein deficient. Reference Oster, Murani, Metges, Ponsuksili and Wimmers140 Furthermore, skeletal muscle mass, myofiber cross-sectional area, primary, secondary, and total myofiber number, DNA, RNA, and protein contents, and creatinine kinase activity were decreased and adipocyte area was increased in skeletal muscle of offspring from protein deficient dams. Overall, protein deficiency during pregnancy negatively affects skeletal muscle growth and development, and function in the offspring. 141,Reference Dohlman, Hansen and Chambers142
In summary, poor maternal nutrition during pregnancy negatively affects fetal and postnatal skeletal muscle growth and development, and metabolic function. These poor outcomes significantly affect both production efficiency in livestock as well as human health.
Programming of immunological function
The health of neonates is established early in postnatal life, primarily through the transfer of maternal antibodies across the placenta or through ingestion of antibodies via maternal colostrum in the immediate postnatal period. Reference Costa, De Oliveira, Giachini, Lima and Bomfim143 A growing body of evidence suggests that in several species (e.g., primates, rodents, and livestock), insults and stressors to the female during pregnancy can impact several functions of immunity and inflammation in the offspring postnatally. Reference Costa, De Oliveira, Giachini, Lima and Bomfim143–Reference Hurlbert, Baumgaertner, Bochantin-Winders, Jurgens, Sedivec and Dahlen145 Maternal stressors that impose alterations to gene transcription patterns in the fetus during intrauterine development or postnatally during the suckling phase have been linked to an unhealthy phenotype in the offspring that appear later in life, including obesity, type II diabetes, asthma, cardiovascular disease/vascular dysfunction, hypertension, and neurological dysfunction. Reference Costa, De Oliveira, Giachini, Lima and Bomfim143,Reference Wallace, Bourke, Aitken and Cruickshank146–Reference Wang, Chen, Chan, Wang and Oliver150 Throughout intrauterine development, different windows of cell development promote continuous changes in gene expression and the components of the fetal immune system. Reference Veru, Laplante, Luheshi and King151 Furthermore, the level of immune system maturation in utero varies for different species, and the type and duration of maternal insult during gestation can create different responses in the innate and adaptive immune systems in the offspring. Reference Segerstrom and Miller152
In ruminant livestock species, the syndesmochorial placenta prohibits the transfer of large proteins like immunoglobulins (Ig) from the maternal to fetal blood supply. Reference Weaver, Tyler, VanMetre, Hostetler and Barrington153 Neonates ultimately rely on the transfer of Ig, immunologically active cells, leukocytes, lymphocytes, cytokines, and other nutritional components of colostrum postnatally to successfully obtain passive transfer of immunity from the maternal to neonatal systems. Reference Weaver, Tyler, VanMetre, Hostetler and Barrington153 Colostrum consumption for newborn ruminants paired with postnatal vaccine administration is imperative for both the innate and adaptive immune responses and subsequent long-term productivity and well-being. Because of the critical nature of colostrum for species in which most immune system maturation occurs postnatally, several studies have documented the influence of maternal nutrition on colostrogenesis, post-partum colostrum quality and yield, and health status of the offspring resulting from nutritional insults on the dam, Reference Redifer, Wichman, Rathert-Williams, Freetly and Meyer154–Reference Odde158 with late-gestation as opposed to early gestation nutrition providing the greatest influence on colostrum production. Reference Baumgaertner, Menezes and Diniz159 However, few reports have fully examined the role of maternal nutrition during pregnancy and its effects on the development of the innate and adaptive immune systems in the offspring.
Recent investigations in beef heifers have shown that prenatal vitamin and mineral supplementation from breeding to calving increased colostrum yield and micronutrient content; however, no differences in Ig concentrations in colostrum were detected. Reference Hurlbert, Baumgaertner and Menezes156 Furthermore, calves born to supplemented and non-supplemented dams had similar serum concentrations of Ig 24 h after consuming maternal colostrum. Despite the lack of differences in Ig in colostrum and serum, postnatal responses to vaccination, as reflected by antibody titers for several viral respiratory and diarrheal diseases, were greater at birth through weaning in calves born to vitamin and mineral supplemented dams. Reference Hurlbert, Baumgaertner and Menezes156 These data suggest a programmed response in immune function because of the maternal nutritional environment during gestation. We also investigated the effects of vitamin and mineral supplementation and maternal rate of gain in beef heifers during the first 12 weeks of gestation on antibody titer response to vaccination in the offspring but found no differences from birth through weaning. Reference Baumgaertner, Ramírez-Zamudio and Menezes32
Others have reported that dietary restriction to 70% of maintenance energy requirements in pregnant beef cows for 40 days before parturition reduced viral (BVD-1) antibody titers in the calves after weaning despite similar management, which suggests that calves born to energy-restricted dams may have a programmed increase in susceptibility to BVD-1 and perhaps other viral diseases during postnatal development. Reference Moriel, Piccolo, Artioli, Marques, Poore and Cooke160 Taken together, the mechanisms by which maternal nutrition “programs” offspring immune responses are unclear; however, late-gestation maternal nutrition, aligning with colostrogenesis (i.e., just before parturition), appears to have a greater influence on programming of the neonatal immune system compared with nutritional insults during early gestation. Despite the similar Ig content in colostrum and in the serum of the calf at 24 h of age, reported by Hurlbert et al., Reference Hurlbert, Baumgaertner, Bochantin-Winders, Jurgens, Sedivec and Dahlen145 the immune responses in calves through weaning was diminished, warranting future investigation. Perhaps epigenetic changes in utero are responsible for this “lag time” from the prenatal insult to the response(s) in the offspring postnatally. Reference Baumgaertner, Ramírez-Zamudio and Menezes32,Reference Hurlbert, Baumgaertner and Menezes156,Reference Baumgaertner, Menezes and Diniz159 Future studies may benefit from coupling the postnatal immune system and growth efficiency evaluations with investigations on the neonatal genome in order to untangle the complex web of maternal nutrition on programmed immune function in livestock species.
Programming of parturition
Parturition marks the culmination of the complex physiological interactions between the mother and the fetus, which involve a regulated sequence of events in which steroid hormones play a central role. Reference Jenkin and Young161 The timing and success of parturition are critical for maternal health and the viability and development of the offspring. The World Health Organization reports that preterm birth affects 11% of pregnancies globally, 162 and it accounts for over 75% of perinatal mortality. Reference Goldenberg, Culhane, Iams and Romero163 In humans, pre-term birth has been linked to developmental programming and long-term effects on cardiovascular, pulmonary, metabolic, and behavioral disease risk. Reference Markopoulou, Papanikolaou, Analytis, Zoumakis and Siahanidou164–Reference Schieve, Tian and Rankin168 Additionally, pre-term birth significantly increases the likelihood of low birth weight, which can result in immediate postnatal consequences and heightened risks of lifelong health challenges. Reference Reynolds, Vonnahme and Lemley169 Furthermore, among livestock species, pre-term birth is recognized as a significant contributing factor to annual neonatal losses of 10%-30% for lambs and 6%-16% for calves, representing a substantial loss in agricultural sustainability. Reference Wu, Bazer, Wallace and Spencer170,Reference Perry, Copping, Miguel-Pacheco and Hernandez- Medrano171
While the mechanisms initiating labor are still not fully understood, key factors have emerged, particularly those involved in the activation of the fetal hypothalamic-pituitary-adrenal axis (HPAA), which drives cortisol release, Reference Conley and Neto172 along with the roles of the steroid hormones estradiol (E2) and progesterone (P4). Reference Schuler, Fürbass and Klisch173,Reference Mesiano174 In ruminants, activation of the HPAA, resulting in a significant surge in fetal cortisol concentrations, determines the timing of parturition before birth. Reference Conley and Neto172,Reference Challis175,Reference Liggins176 In contrast, cortisol does not play a prominent role in initiating labor in horses Reference Conley and Ball177 or pigs Reference Ellendorff, Taverne and Elsaesser178 ; while cortisol levels increase toward the end of gestation due to the functional HPAA, labor in these species appears to be primarily driven by maternal factors like prostaglandin release or mechanical signals. Furthermore, a slower increase in cortisol concentration is characteristic of humans, Reference Liggins176,Reference Fowden, Li and Forhead179 where labor initiation involves an interplay of maternal, fetal, and placental signals. Reference Schwartz180,Reference Vannuccini, Bocchi, Severi, Challis and Petraglia181 Ultimately, regarding the initiating signal across species, there is a need to shift from progestogenic to estrogenic dominance, either systemically or at the functional level, and prostaglandin production to promote the myometrial contractions and cervical ripening that allows the expulsion of the fetus. Beyond the importance of fetal cortisol in the initiation of parturition, it also plays a critical role in fetal lung maturation by stimulating surfactant production and cellular differentiation, which is essential for extrauterine survival. Reference Kitterman, Liggins and Campos182–Reference Roberts184
The diverse mechanisms that initiate labor across species have made it challenging to reach a clear consensus on the exact triggers of parturition. However, recent studies have highlighted the influence of maternal nutrition before and during pregnancy on timing of delivery, Reference Wallace, Luther and Milne45,Reference Fowden and Silver185–Reference Bloomfield, Oliver and Hawkins188 and neonatal viability, which in most species depends primarily on cortisol-induced lung maturation. Reference Schwartz180,Reference Kitterman, Liggins and Campos182,Reference Arigliani, Spinelli, Liguoro and Cogo189 Furthermore, it’s important to recognize that maternal and fetal nutrition are distinct processes. Although maternal nutrition is a contributing factor, the fetus’s access to nutrients is also significantly affected by placental blood flow and metabolism, umbilical circulation, and the endocrine and hormonal environment. Nonetheless, research has demonstrated that maternal nutrition exerts a significant influence on all of these factors. Reference Reynolds, Vonnahme and Lemley169,Reference Barker190–Reference Harding and Johnston193
Research in humans has established a connection between maternal nutrition and the timing of delivery, which supports the concept of programming of parturition. It has been shown that inadequate maternal nutrition before and at various stages of pregnancy is linked to preterm birth. Reference Bloomfield194–Reference Martin, Sotres-Alvarez and Siega-Riz197 Although numerous studies have been conducted to understand the effects of maternal nutrition on preterm birth in humans, most have been epidemiological, relying on questionnaires to classify the types of foods mothers consume. For ethical reasons, it is not feasible to subject pregnant women to strictly controlled dietary regimens throughout pregnancy. In contrast, the ewe has emerged as the preferred animal model, Reference Barry and Anthony198 allowing for specific nutritional interventions that can be precisely administered.
In a study by Bloomfield et al., Reference Bloomfield, Oliver and Hawkins188 a 15% decrease in maternal weight during the period from 61 d before conception until 30 d after conception, with ad libitum feed intake thereafter, resulted in an early increase in fetal adrenocorticotropin hormone (ACTH) in all fetuses and elevated cortisol levels in half of the fetuses, ultimately leading to premature birth in all treated ewes. Despite this, fetuses of both undernourished and control groups were born with similar sizes, supporting the idea that maternal and fetal nutrition are distinct. Reference Harding and Johnston193 Shockingly, the study also found that although the preterm lambs were born at the normal size, they all died soon after birth, suggesting that their organs did not mature sufficiently to survive extrauterine life and that the rise in cortisol might not be enough for complete lung maturation. In a subsequent study, the same treatment was administered, and maternal blood samples were taken for cortisol and prostaglandin analysis, and to determine changes in the E2/P4 ratio from 126 d (approx. 0.85 of gestation) until delivery. Reference Kumarasamy, Mitchell and Bloomfield187 The results demonstrated that cortisol, prostaglandin, and the E2/P4 ratio increased before delivery in a similar pattern in both periconceptionally undernourished and control groups during the initiation of the labor. However, the hormonal changes occurred 7–10 days earlier in the undernourished group, leading to premature birth. Similarly, the provision of a maintenance control diet from the second week of pregnancy does not reverse the effect of maternal undernutrition 60 d prior to and during the first week of a twin pregnancy, as Edwards & McMillen Reference Edwards and McMillen199 showed. This periconceptional undernutrition stimulates the fetal HPAA and raises the concentration of ACTH in late gestation. Together, these studies have demonstrated that programming of parturition can occur in response to changes in maternal periconceptional nutrition, resulting in premature activation of the fetal HPA axis, ultimately leading to preterm birth.
Another study demonstrated a correlation between maternal nutritional status during late gestation and uterine prostaglandin F metabolite (PGFM) release and timing of delivery. Reference Fowden, Giussani and Forhead28 In this study, ewes were nutrient-restricted (but not deprived of water) for 48 h, which led to a significant increase in plasma PGFM levels. The ewes were divided into three groups based on the timing of food withdrawal: before 134 d of gestational age, between 134 and 137 d, and after 137 d. When food was reintroduced or glucose was infused, plasma PGFM levels rapidly returned to baseline. Notably, animals that fasted later in gestation (after 137 d of gestational age) exhibited the greatest plasma PGFM concentrations following fasting. These animals also experienced a decline in plasma P4 during the last 24 h of fasting, but uterine blood flow remained unchanged. Ewes in the late-fasting group gave birth within 12–24 h after the fast ended, a significantly shorter period compared with the other groups.
Similar to the study in ewes, researchers have examined the production of PGFM in pregnant mares with differing dietary states during late gestation. Reference Silver and Fowden186 As in sheep, there was an increase in PGFM concentration during fasting, and a rapid decrease in PGFM levels upon refeeding. Thus, nutritional status during late gestation can influence the timing of parturition in ewes and mares by increasing PGFM levels, thereby initiating the onset of labor.
In conclusion, the process of programming parturition and fetal organ maturation is shaped by the interplay between maternal, fetal, placental and environmental factors. Maternal nutrition, both before and during pregnancy, has emerged as an important influence on the timing of labor and neonatal outcomes, as demonstrated particularly in the ewe. Controlled studies in livestock models have revealed that maternal undernutrition can prematurely activate the fetal HPAA, while maternal overnutrition can increase prostaglandin concentrations, both of which can lead to premature delivery. These findings highlight the significance of maternal nutrition not only in determining the timing of parturition but also in shaping the long-term health and viability of the offspring.
Conclusions
Implications of livestock models for other species
Livestock species are particularly advantageous in biomedical research because the new knowledge can be applied to both human health and livestock production, the latter of which, as we have discussed, is critical for agricultural sustainability and food security (Figure 1). Beyond the dual purpose with dual benefit paradigm, livestock species, particularly ruminants, have some advantages to biomedicine compared with rodent models. Specifically, ruminants are not litter-bearing, have longer gestation lengths and in the case of cattle, timing of organ development is similar to that of humans, in vivo measurements are more easily obtained, and an abundance of tissue and bodily fluids are available.
Livestock models of developmental programming positively contribute to human health through translation of research and improving food security.

Importance of livestock in and of themselves
Livestock are of great importance in providing high-quality protein and micronutrients in the human diet. In fact, the availability of animal protein is linked to the rate of economic development in developing countries. Reference Randolph, Schelling and Grace200,Reference Otte, Costales and Dijkman201 Furthermore, as the world’s population continues to increase, so does the demand for animal protein because of its value as a nutrient-dense high-quality food. Reference Trostle and Seeley202–204 Additionally, skeletal muscle mass and function, which is dependent on adequate protein consumption, are key components in human health and well-being.
Future directions and recommendations
Very early during gestation, nutrients are supplied to the fetus primarily through the histotroph (uterine secretions), and as the placenta develops, it becomes the primary organ for nutrient delivery to the developing offspring (Reynolds et al., 2023a). Research utilizing livestock models to investigate the effects of nutritional supplementation during pregnancy on utero-placental nutrient delivery, specific nutrient demands for developing tissues, microbiome and epigenetic mechanisms responsible for developmental programming, cardiovascular and metabolic function in the fetus and offspring, and transgenerational impacts should improve our understanding of, and hence management and therapeutic strategies for fostering the positive and mitigating the negative aspects of, developmental programming.
Furthermore, we are convinced not only of the value of livestock models, as discussed already, but also of the value of epidemiological and transgenerational studies in humans and other species in furthering our understanding of the underlying mechanisms and consequences of developmental origins of health and disease.
Acknowledgments
None.
Financial support
This publication is a contribution of the South Dakota Agricultural Experiment Station. This material is based upon work that is supported in part by the National Institute of Food and Agriculture, U.S. Department of Agriculture, Hatch project under accession number 7,009,601 (R.M.S.).
Competing interests
The authors declare none. To the authors knowledge, all studies discussed in this invited review have followed ethical standards for animal and human research.
Ethical standard
This is a review paper. There is no human or animal research conducted in this paper. This paper has reviewed research papers, which each provide their own ethical standards statements.