Introduction
With the fast development of scale and intensification of sheep farm in subtropical area, there were many problems appeared, such as low feed intake, susceptibility to disease and severe stress in high humidity and temperature environments, which lead to reduced growth performance and loss of economic benefits. Antibiotics have been widely used in animal husbandry and were once regarded as the “panacea” for animal health care under stress by prevention of bacterial infections; however, prolonged employment of antibiotics can lead to the development of resistance to these drugs, resulting in a decline or disappearance of the antimicrobial capacity of the drug. Therefore, the safe, non-residual antibiotic alternative has become a research hotspot in the animal husbandry. Plant essential oils are secondary metabolites extracted from plants with strong volatility and aroma. Among them, cinnamaldehyde (CA), which is derived from cinnamon, belongs to the aromatic compounds (Rota et al. Reference Rota, Carramiñana and Burillo2004). CA has the biological properties of strong aromatic, antibacterial, antiviral, anti-inflammatory, antioxidant, and immune-enhancing (Benchaar et al. Reference Benchaar, Petit and Berthiaume2006), which result in nutrient metabolism regulation and growth improvement in livestock. The molecular structure of CA has the aldehyde group and the double bond, which determines that CA has strong antioxidant properties. Research has established that CA possesses antimicrobial effects against various strains of both gram-positive and gram-negative bacteria, including Escherichia coli, Bacillus subtilis, Staphylococcus spp., and others (Al-Mijalli et al. Reference Al-Mijalli, Mrabti and El Hachlafi2023; Baskaran et al. Reference Baskaran, Amalaradjou and Hoagland2010; Gill and Holley Reference Gill and Holley2006; Vasconcelos et al. Reference Vasconcelos, Croda and Simionatto2018). Therefore, there were studies reported addition of CA to rations can manipulate ruminal fermentation by inhibiting specific microbes such as gram-positive bacteria and methanogen, which led to a decrease of ruminal acetate/propionate (A/P) ratio, ammonia, and methane production. Besides, some studies have determined the in vitro antioxidant capacity of CA and found that the scavenging rate of CA on free radicals was above 90% (Beniaich et al. Reference Beniaich, Zouirech and Allali2023). It has also been reported supplementation of ewe feed with 80 mg/kg CA, eugenol and chili oil resin mixture was effective in improving serum immunity and antioxidant capacity (An et al. Reference An, Wang and Wang2020). Based on these functions, numerous studies have shown that the addition of proper amounts of CA (100–500 mg/kg, dry matter [DM]) to ruminant diets can help improve production performance (Favaretto et al. Reference Favaretto, Alba and Marchiori2020). However, appropriate content of CA addition is also extremely important for ruminants, and if excessive CA is added to the ration (≥1600 mg/kg, DM), it may lead to a decrease in animal feed intake and affect production performance (Ornaghi et al. Reference Ornaghi, Passetti and Torrecilhas2017). Therefore, it is particularly important to add the right dose of CA to ruminant production.
Although many studies have been carried out on CA as a feed additive, there are relatively few studies on silica adsorbed CA. In addition, it was found that SiO2-EO (essential oil) nanocapsules exhibited more significant inhibitory activity against both gram-negative and gram-positive bacterial biofilms compared to free EO (Wang et al. Reference Wang, Wang and Wang2025). Therefore, this study was the first to determine the optimal supplemental strategy of adsorbed SiO2 CA in Hu sheep production by adding different levels of adsorbed SiO2 CA to the basal diet, and to systematically evaluate the effects of different levels of adsorbed SiO2 CA on growth performance, serum indexes, nutrient apparent digestibility, ruminal fermentation parameters, and rumen microbiota of Hu sheep.
Materials and methods
Animals, feed, and experimental design
This study was approved by the Experimental Animal Management Committee of Guangxi University (No. GXU-2023-0266). Sixty healthy male lambs (22.7 ± 0.3 kg) at the age of 4 months were randomly divided into three bars (20 lambs per bar) based on body weight (average weight). And then they were randomly assigned to one of the three treatments, the three treatments were as follows: (1) basal diet (CON); (2) basal diet + 400 mg/kg CA additive (total mixed ration [TMR] DM basis, CA400); (3) basal diet + 800 mg/kg CA additive (TMR DM basis, CA800). The feed volume of each group was weighed accurately daily and CA was allocated according to the feed volume, which was fully mixed with the diet of each group before feeding. The CA for the yellow powder, used silicon dioxide as diluent, calcium carbonate as the carrier, and the CA content was 25% (m/m) (purchased from Guangzhou Xinnong Biotechnology Co., Ltd).
Based diet composition is shown in Table 1. The TMR was provided twice daily at 8:00 and 16:00 to all groups, maintain about 5% leftovers to ensure free-feeding. Animals were provided with ad libitum access to water, free movement. The 75-day study period included a 15-day prefeeding period, succeeded by a 60-day experimental phase.
Table 1. Composition and nutrient level of experimental diet (dry matter basis)

1 The premix provided the following per kg of diets: Fe 2000 mg, Cu 200 mg, Mn 500 mg, Zn 150 mg, Se 5.55 mg, I 1.5 mg, Co 2.5 mg, VA 60 000 IU, VD 5000 IU, VE 70 IU.
2 Calcium and phosphorus are calculated values, and the rest of the dietary nutrient levels are measured.
Feed intake, average daily gain, nutrient digestibility sampling, and measurement
During the experiment, the feed amount of each group was recorded every day, and the remaining feed of each group was weighed and the feed intake was calculated the next morning. The Hu sheep were weighed on days 0, 31, and 60, and the weighing was completed before morning feeding. These data were used to calculate average daily gain (ADG), and feed conversion efficiency was calculated as the ratio of dry matter intake (DMI) to ADG. Basal diets were collected on 1 day of weeks 1–6 and stored in a refrigerator at −20℃ for subsequent determination of dietary routine nutrients.
To test the apparent digestibility of the diets, six lambs were randomly selected from each group and fed in a single pen to each group on days 55–59 of the trial. Fecal samples were collected by rectal sampling twice a day, half an hour after each feeding, and feed samples were collected at the same time. The 3-day feed samples of each group were mixed with the fecal samples of each lamb. The fecal samples of each lamb were divided into two parts, one part was added with 10 mL of 5% concentrated sulfuric acid for the determination of crude protein (CP), and the other part was used for the determination of conventional nutrients. All samples were subsequently analyzed for DM content (Feldsine et al. Reference Feldsine, Abeyta and Andrews2002), and the dried samples were pulverized through a 1-mm screen using a Wiley mill (Arthur H. Thomas, Philadelphia, PA). The contents of organic matter (OM) and CP of feed and fecal samples were determined (Feldsine et al. Reference Feldsine, Abeyta and Andrews2002). Acid digestible fiber (ADF) and neutral detergent fibre (NDF) contents were determined according to the method described by Van Soest et al. (Reference Van Soest, Robertson and Lewis1991). Acid-insoluble ash (AIA), used as an internal marker to assess apparent nutrient digestibility, was processed in the diet and fecal samples according to the method prescribed by Van Keulen and Young (Mc Geough et al. Reference Mc Geough, O’Kiely and Kenny2010). Apparent nutrient digestibility is as follows:
where A is a nutrient composition (%) in the feed, B is a nutrient component in feces (%), C is feed indicator (%), D is indicator in feces (%), and the indicator is AIA.
Blood sampling and biochemistry, antioxidant, and immunity parameters measurement
Before morning feeding on the 60th experimental day, blood samples were drawn from eight sheep in each group one by one via the jugular vein by syringe. The samples were introduced into tubes designed for blood collection, which were pre-filled with sodium heparin. Subsequently, these samples underwent centrifugation at a rate of 3500 revolutions per minute (r/min) for a duration of 10 minutes to effectuate plasma separation. Subsequently, all plasma samples were stored at −20℃ for subsequent analysis of total protein (TP), alkaline phosphatase (ALP), albuminous aminotransferase (ALT), glutamine transaminase (AST), glucose (Glu-G), and lactate dehydrogenase (LDH) using an automatic biochemistry analyzer (model: Myriad BS-240); catalase (CAT), malondialdehyde (MDA), total antioxidant capacity (T-AOC), glutathione peroxidase (GSH-px), total superoxide dismutase (T-SOD), immunoglobulin G (IgG), immunoglobulin A (IgA), immunoglobulin M (IgM) using an enzyme labeling analyzer (model: Huawei Delang DR-3000) (Hu et al. Reference Hu, Bai and Xu2021).
Bacterial community composition analysis
Genomic DNA of the microbial community within ruminal fluid was successfully extracted employing the E.Z.N.A.® Soil DNA Kit (Omega Bio-tek, Norcross, GA, U.S.), following the guidelines provided by the manufacturer. Post-extraction validation of the DNA was conducted on a 1% agarose gel. Additionally, the quantification and purity assessment of the extracted DNA were carried out utilizing a NanoDrop 2000 UV–vis spectrophotometer (Thermo Scientific, Wilmington, USA). The V3-V4 hypervariable segment of the bacterial 16S rRNA gene was targeted for amplification using the primer pair 338F (5′-ACTCCTACGGGAGGCAG-3′) and 806R (5′-GGACTACHVGGGTWTCTAAT-3′), performed on an ABI GeneAmp® 9700 PCR thermocycler (ABI, CA, USA). The amplification of the 16S rRNA gene was executed with the following PCR protocol: an initial step of denaturation at 95°C for 3 minutes, succeeded by 27 cycles comprising denaturation at 95°C for 30 seconds, annealing at 55°C for 30 seconds, and extension at 72°C for 45 seconds, complemented by a final extension at 72°C for 10 minutes, and concluding with a hold at 4°C. The PCR was conducted in triplicate reactions. The 16S rRNA gene amplification product underwent gel extraction from a 2% agarose gel matrix and subsequent purification utilizing the AxyPrep DNA Gel Extraction Kit by Axygen Biosciences, based in Union City, CA, USA.
The process adhered to the manufacturer’s guidelines and was concluded with quantification via the Quantus™ Fluorometer from Promega, a company located in the USA. Thereafter, the purified amplicons were combined in equal molar ratios and subjected to paired-end sequencing (2 × 300 bp) on the Illumina MiSeq platform, operated by Majorbio Bio-Pharm Technology Co., Ltd, a Shanghai, China-based entity, following their standard operating procedures. The raw 16S rRNA gene sequencing reads were processed to remove duplicates and underwent quality filtering with Trimmomatic. Subsequently, they were merged using FLASH (version 1.2.11). Taxonomic units, designated as operational taxonomic units (OTUs), were clustered with a 97% similarity threshold using UPARSE (version 7.1, http://drive5.com/uparse/), with the removal of chimeric sequences. The taxonomic classification of each OTU representative sequence was conducted using the RDP Classifier (http://rdp.cme.msu.edu/) against the 16S rRNA database, such as Silva SSU128, applying a confidence threshold of 0.7. The taxonomic abundance within each sample was quantified for both phylum and genus levels. Alpha diversity metrics, encompassing richness estimates (ACE and Chao1) and diversity indices (Shannon and Simpson), were computed with QIIME 2.
Ruminal fluid sampling, fermentation parameters measurement
Ruminal fluid was obtained from eight sheep in each group via an oral stomach tube around 3 hours post-morning feeding on the 60th experimental day. To mitigate potential saliva contamination, the initial 50 mL of rumen fluid was discarded. Following this, the remaining rumen fluid was passed through a filtration process involving four layers of cheesecloth. The pH of each sample was then promptly assessed using a portable pH meter (Lei Ci PHS-25, portable, Shanghai INESA Scientific Instrument Co., Ltd), and this measurement was conducted thrice for each sample to ensure accuracy. Ten milliliters of each ruminal fluid sample was preserved with the addition of 2 mL of 25% HPO3 for volatile fatty acid (VFA) analysis using gas chromatography (7820A, Agilent, USA). A further 10 mL of each ruminal fluid sample was subjected to analysis for ammonia nitrogen using a colorimetric method (Mc Geough et al. Reference Mc Geough, O’Kiely and Kenny2010). The reserved ruminal fluid samples were stored at −80°C for subsequent analysis of bacterial community composition.
Statistical analysis
The effects of the addition of CA into the diet of Hu sheep on growth performance, nutrients digestibility, ruminal fermentation parameters, and ruminal bacterial community composition at the genus and phylum levels. A one-way ANOVA was employed for statistical analysis using SPSS 18.0 for Windows, complemented by Duncan’s multiple range tests for further comparisons. The analysis focused on phyla and genera that were detected in over 50% of the samples and accounted for more than 0.5% of the total bacterial population. The statistical model is as follows: Yi = µ + αi + εi. Yij – dependent variable, αi – effect of CA (i = 1, 3), and εi is the residual error. Differences among means were tested using Duncan’s multiple-range tests. Statistical significance was defined as P < 0.05.
Results
Growth performance
As demonstrated in Table 2, the average daily weight gain (ADG) of the two treatments, CA400 and CA800, was found to be considerably higher than that of the CON group during both the 1–30-day and 1–60-day periods (P < 0.05). However, no significant differences were observed in the ADG of the 30–60-day period among the three groups. There were no differences in feed intake among the three groups (P > 0.05). The feed-to-gain (F/G) ratio was found to be significantly lower in groups of CA400 and CA800 compared to the control group in both 1–30 days and 1–60 days (P < 0.05), while there is no differences exist in F/G of 30–60 days among the three groups. The digestibility of OM in group CA400 was significantly higher than that in group control and CA800 (P < 0.05). Additionally, the digestibility of CP in groups CA400 and CA800 were significantly higher than that in Group A (P < 0.05). No significant differences were observed in the digestibility of DM, neutral detergent fiber, and acid detergent fiber across the three groups (P > 0.05).
Table 2. Effect of cinnamaldehyde on growth performance and nutrient digestibility of Hu sheep

Notes: CON = control group; CA400 = 400 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis; CA800 = 800 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis (same as following tables).
Values are presented as means with standard error of the mean (SEM). Data were analyzed by one–way analysis of variance (ANOVA), followed by Duncan’s new multiple range test for post–hoc comparisons when the overall treatment effect was significant (P < 0.05). Within the same row, means with different lowercase superscript letters (a, b, c) differ significantly (P < 0.05).
Rumen fermentation parameters
As shown in Table 3, there were no significant differences were observed in the pH value and A/P ratio of rumen fluid among the three groups (P > 0.05), but the A/P ratio of the two experimental groups tended to lower than CON. The ruminal ammonia nitrogen of the two experimental groups (CA400 and CA800) were significantly lower than CON (P < 0.05). The concentration of total volatile fatty acids including acetate, propionate, butyrate, isobutyrate, valerate, and isovalerate was the highest in the CA400 group, followed by CON, while the lowest was in the CON group (P < 0.05).
Table 3. Effect of cinnamaldehyde on rumen fermentation parameters of Hu sheep

Note: CON = control group; CA400 = 400 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis; CA800 = 800 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis (same as following tables). Values are presented as means with standard error of the mean (SEM).. Within the same row, means with different lowercase superscript letters (a, b, c) differ significantly (P < 0.05).
Ruminal bacterial community diversity, abundance at phylum level and genus level
As shown in Table 4, there were no differences existed in α-diversity indexes of Ace, Chao1, Shannon, and Simpson among the three groups (P > 0.05). According to Fig. 1, the species composition of the CA400 and CA800 groups was close, while the species composition of the CON and CA400 groups differed significantly from that of the CA800 group, and the dispersion of the two experimental groups was small, indicating that there was little difference within the group. According to Fig. 2, there were 3024 OTUs in each test group, and the number of OTUs unique to test CON group was 822, the number of OTUs unique to test CA400 group was 585, and the number of OTUs unique to test CA800 group was 743. As shown in Table 5, The abundance of Bacteroidetes in CA800 group was significantly higher than that in CON group (P < 0.05), and the abundance of Firmicutes in CA400 and CA800 groups was significantly lower than that in CON group (P < 0.05). The abundance of Cyanobacteria in CA400 group was significantly higher than that in CON group (P < 0.05), and the abundance of Spirochaetota in CA800 group was significantly higher than that in CON group (P < 0.05). Fibrobacterota was significantly higher in CA400 and CA800 groups than in CON group (P < 0.05), while the abundance of other bacterial phyla was not different among the three groups (P > 0.05). As shown in Table 6, the abundance of Prevotella in CA400 and CA800 groups was significantly higher than that in CON group (P < 0.05), while the abundance of Rikenellaceae_RC9_gut_group was significantly lower than that in CON group (P < 0.05). There was no significant difference in the abundance of other bacterial genera among the three groups (P > 0.05).

Figure 1. PCoA analysis diagram.

Figure 2. Venn diagram of OTU distribution.
Table 4. Effect of cinnamaldehyde on rumen bacterial alpha diversity indexes of Hu sheep

Table 5. Effect of cinnamaldehyde on ruminal bacterial community at phylum level of Hu sheep

Note: CON = control group; CA400 = 400 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis; CA800 = 800 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis (same as following tables). Values are presented as means with standard error of the mean (SEM).. Within the same row, means with different lowercase superscript letters (a, b, c) differ significantly (P < 0.05).
Table 6. Effect of cinnamaldehyde on ruminal bacterial community at genus level of Hu sheep

Note: CON = control group; CA400 = 400 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis; CA800 = 800 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis (same as following tables). Values are presented as means with standard error of the mean (SEM).. Within the same row, means with different lowercase superscript letters (a, b, c) differ significantly (P < 0.05).
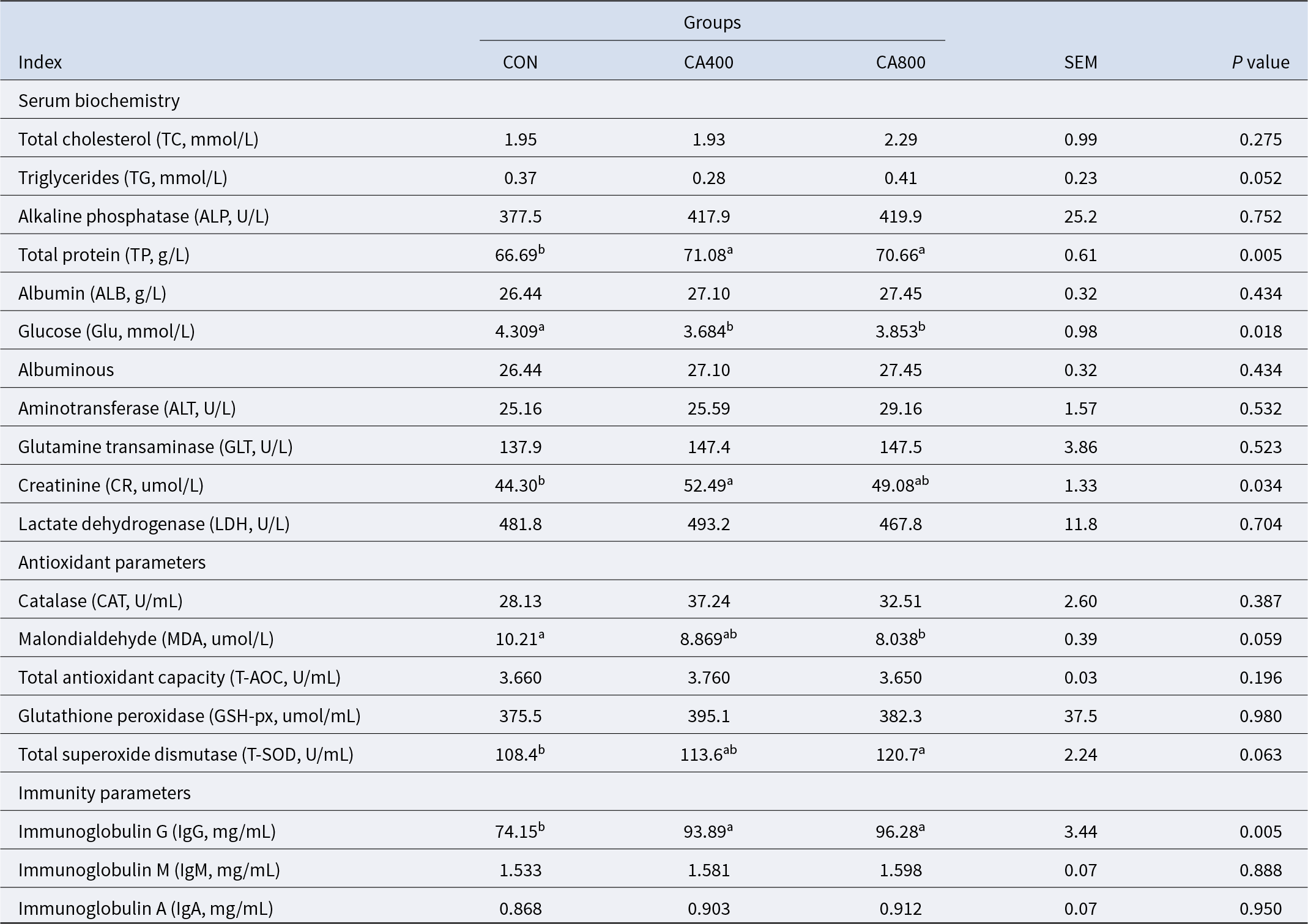
Serum indicators
As shown in Table 7, there were no differences exist in serum biochemistry parameters, including TC, TG, ALP, ALB, ALT, GLT, CR, and LDH among the three groups. The content of TP in the CA400 and CA800 groups was significantly higher than that in the CON group (P < 0.05), and glucose (Glu) was significantly lower than that in the CON group (P < 0.05). As for antioxidant parameters, the concentration of MDA in groups CA400 and CA800 was significantly lower than that of CON (P < 0.05), while there were no differences in parameters, including CAT, T-AOC, and GSH-px among the three groups (P > 0.05), the content of T-SOD in the CA800 group was significantly higher than in CON. As for immunity parameters, the concentration of IgG of group CA400 and CA800 was significantly higher than that of CON (P < 0.05), while there were no differences in parameters, including IgA and IgM (P > 0.05).
Table 7. Effects of cinnamaldehyde on serum biochemistry, antioxidant, and immunology parameters of Hu sheep

Note: CON = control group; CA400 = 400 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis; CA800 = 800 mg/kg SiO2 adsorbed CA additive on TMR dry matter basis (same as following tables). Values are presented as means with standard error of the mean (SEM).. Within the same row, means with different lowercase superscript letters (a, b, c) differ significantly (P < 0.05).
Discussion
Feed intake, growth performance, and nutrients digestibility
Feed intake is tightly related to the growth performance of animals, and therefore, whether feed intake can be improved or not is an important effective evaluation standard for feed additives. The absence of a significant increase in DMI observed in Hu sheep aligns with previous findings reported by Chaves et al. (Reference Chaves, Dugan and Stanford2011) and An et al. (Reference An, Wang and Wang2020). This phenomenon could be attributed to variations in the additive formulation and administration dosage employed in the experimental design. Notably, the microencapsulation of CA through silica adsorption effectively mitigated potential odor-related feed aversion in the present study. Furthermore, the maximum supplemented CA concentration of 800 mg/kg in TMR DM corresponded to 200 mg/kg of pure CA, a dosage comparable to that implemented in Chaves et al.’s investigation (Chaves et al. Reference Chaves, Dugan and Stanford2011). In contrast, there were studies reported that CA supplementation increased the feed intake of sheep, dairy cows, and beef cattle (Hu et al. Reference Hu, Bai and Xu2021), the addition level ranged from 43 to 400 mg/kg of dietary DM, which may be attributed to the supplementation of CA changed diet flavor and improved feed palatability. However, there were studies that showed the addition of CA mixed EO decreased the feed intake of dairy cows, reasons attributed to high dosage influenced feed palatability or feed energy utilization efficiency was improved (Chaney and Marbach Reference Chaney and Marbach1962; Hamada et al. Reference Hamada, Omori and Kameoka1968). Therefore, the effects of essential oil on DMI appeared to be dosage, ration composition, and animal physiology dependent.
In the present study, the addition of CA has been shown to improve the growth performance of Hu sheep in both two experimental groups. In a study conducted by Chaves et al. (Reference Chaves, Dugan and Stanford2011), the addition of 200 mg/kg CA did not affect DMI but significantly increased the ADG of lambs. Favaretto et al. (Reference Favaretto, Alba and Marchiori2020) also showed addition of low concentrations, 34 mg/kg and 68 mg/kg, of CA to sheep feeds resulted in a linear increase in ADG of sheep. The growth promotion ability of CA is mainly due to the reasons of CA not only can improve the palatability of the animal ration but also can improve dietary nutrient utilization efficiency and body health. In studies by Yang et al. (Reference Yang, Benchaar and Ametaj2007) and Cantet et al. (Reference Cantet, Yu and Tucker2023), it was found that the addition of CA in the feed of dairy cows significantly improved the digestibility of CP and ruminal propionate content, and increased feed energy conversion. In our study, not only apparent digestibility of OM and CP improve but also the digestibility of NDF and ADF were improved in both two experimental groups, which was consistent with the results of the above study. Most studies on essential oil demonstrated its positive influence on the production performance of ruminants, even though the DMI was inhibited or not influenced (Chaves et al. Reference Chaves, Stanford and Dugan2008), which is consistent with our study. Therefore, the addition of an appropriate amount of CA to the ration can promote the nutrient digestion of Hu sheep, which in turn results in a positive effect on growth performance.
Ruminal fermentation parameters
Ruminal pH serves as a key indicator of fermentation status, regulated by dietary composition, salivary buffer capacity, and VFA production (Busquet et al. Reference Busquet, Calsamiglia and Ferret2005b; Dijkstra et al. Reference Dijkstra, van Gastelen and Dieho2020). In the present study, all experimental groups maintained physiologically stable pH values (6.0–7.0), consistent with optimal microbial activity and acidosis prevention. Meanwhile, ruminal NH3-N concentration reflects microbial nitrogen metabolism efficiency, demonstrating the dynamic equilibrium between dietary protein degradation and microbial utilization essential for rumen microbial proliferation (Roman-Garcia et al. Reference Roman-Garcia, El-Haddad and Piantoni2022). In this study, ruminal NH3-N concentration was decreased by the addition of CA, and the lowest concentration was observed in the CA800 group, which is similar to results of other studies indicating the addition of EO or CA can reduce ruminal ammonia production, increased nitrogen retention, and finally improve protein digestibility (Benchaar et al. Reference Benchaar, Petit and Berthiaume2006; Dias Junior et al. Reference Dias Junior, Dos Santos and da Silva2023; Newbold et al. Reference Newbold, McIntosh and Williams2004). It has been reported (McIntosh et al. Reference McIntosh, Williams and Losa2003) that plant essential oils and CA can inhibit bacteria responsible for the deamination of amino acids, and rumen protozoa population, which explains decreased NH3-N concentration in both CA-added groups of this study. Besides, the abundance of ruminal Firmicutes bacteria, which is a group of gram-positive and ammonia-producing bacteria, was reduced, and apparent protein digestibility was improved by the addition of CA in our study further supporting CA has the function of improving ruminant protein utilization. Ruminal volatile fatty acids are the main energy-supplying substances for ruminants. CA can change the structure of the microbial community in the rumen, which in turn affects the ruminal VFA concentration and composition. It has been reported that the addition of a high dosage of CA or EO inhibited ruminal fermentation and induced VFA concentration reduction in many in vitro experiments (Lin et al. Reference Lin, Lu and Salem2013), while in vivo studies on the effects of CA or EO on rumen VFA production have different voices, depend on feed, animal, and source of EO (Tomkins et al. Reference Tomkins, Denman and Pilajun2015). For example, the VFA concentration was increased by the addition of 750 mg/d blend EO when dairy cows were fed with alfalfa silage, while decreased when fed with corn silage (Ran et al. Reference Ran, Tang and Yu2021). In our study, total VFA and other VFA concentrations were increased by the addition of 400 mg/kg CA additive while decreased by 800 mg/kg CA additive. The reason was probably related to the addition of EO-promoted rumen motility at high dosages which increased VFA absorption but needs to be further researched. In this study, the A/P ratio was decreased by the addition of CA, which was consistent with most of the in vivo studies on EO (Busquet et al. Reference Busquet, Calsamiglia and Ferret2005a). The change of ruminal fermentation mode by increasing propionate proportion was tightly related to the effects of EO on rumen microbe community. Gram-positive bacterial cells lack a protective outer membrane around the cell wall, which increases their sensitivity to essential oil as compared with gram-negative bacteria (Zhou et al. Reference Zhou, Wu and Lang2020) and gram-positive bacteria, mainly phylum Firmicutes in the rumen response to acetate production (Wang et al. Reference Wang, Wang and Guo2023), and therefore, the ruminal A/P ratio was decreased in this study as ruminal Firmicutes abundance was decreased by CA.
Rumen microbiota
The ruminal bacterial community and composition are mainly affected by genetic, environmental, and dietary factors. In this study, ruminal bacterial composition was analyzed at the phylum and genus level, and the alpha-diversity indices, including Chao1, Ace, and Shannon index, tend to be higher in two CA-added groups as compared with control, indicating that the addition of CA to the diets of Hu sheep tends to increase species richness and diversity in the rumen. The increased bacterial diversity is helpful in improving ruminal nutrient digestibility (Wang et al. Reference Wang, Li and Zhang2022), as confirmed by the higher digestibility of OM, NDF, and CP in the two CA-added groups. In our study, an abundance of phylum Bacteroidetes was increased, while Firmicutes was decreased indicating ruminal bacterial community composition was modified by the addition of CA. The phylum Bacteroidetes is a group of gram-negative bacteria that mainly includes genus Bacteroides, Prevotella, Desulfovibrio, etc. Bacteroidetes play an important role in OM conversion, protein degradation, and non-fiber carbohydrate digestion (Valerio de Oliveira et al. Reference Valerio de Oliveira, Jewell and Freitas2013) and tend to produce propionate in rumen, and it has been reported beef cattle with a higher relative abundance of Bacteroidetes in rumen had a higher feed conversion ratio (Lopes et al. Reference Lopes, de Souza Duarte and La Reau2021), while phylum Firmicutes belongs to gram-positive bacteria, which is mainly involved in digestion of fibrous feedstuff and tends to produce acetate and butyrate. Therefore, the change in bacterial community explained the ruminal A/P ratio was decreased and OM digestibility was improved by the addition of CA in our study. At the genus level, the dominant genera were Prevotella and Rikenellaceae_RC9_gut_group, and there was a tendency that the relative abundance of Prevotella was increased with increasing concentrations of CA, suggesting the addition of CA in the diet probably promoted the Prevotella growth, which was consistent with the findings of Bekele (Bekele et al. Reference Bekele, Koike and Kobayashi2010). Prevotella is a group of gram-negative bacteria belonging to Bacteroidetes, and they are mainly involved in the metabolism of non-fiber carbohydrate proteins, etc. (Avgustin et al. Reference Avgustin, Ramsak and Peterka2001). Therefore, its low sensitivity to CA probably benefits for survival of Prevotella in the rumen, and the increased abundance of Prevotella probably contributes to the increase of OM and CP digestibility in our study. Rikenellaceae_RC9_gut_group belongs to the phylum Bacteroidetes, and its role is to decompose carbohydrates (Liu et al. Reference Liu, Wu and Chen2022), but Rikenellaceae_RC9_gut_group as the second largest bacterial genus is not common. Prevotellaceae_UCG-003 belongs to the family Prevotellaceae, which is one of the major genera of rumen fiber-degrading bacteria, and also participates in protein metabolism (Zhao et al. Reference Zhao, Chen and Zhou2017), and the addition of 400 mg/kg CA additive, which was equal to 100 mg/kg pure CA, in this experiment significantly increased the abundance of this genus and was a benefit for rumen fiber degradation ability enhancement.
Serum biochemistry, immunology, and antioxidant indices
CA is a small molecule substance that can be absorbed into the animal body easily; therefore, its effect is not only exerted on the ruminal microbe but also on the animal body (Wells Reference Wells2024). The level of TP mainly reflects the synthesizing function of the liver and the body’s nutritional situation. The significant elevation in serum TP concentration observed in CA-supplemented groups (P < 0.05) suggests improved protein digestion in Hu sheep, consistent with the physiological effects of phytogenic compounds reported in ruminants. This finding aligns with Li et al. (Reference Li, Ru and Liu2012), where dietary supplementation with essential oils or CA increased circulating TP levels through improved hepatic protein synthesis and reduced amino acid catabolism, in addition, the abundance of Bacteroidetes, the dominant phylum in hydrolyzing proteins, was increased in all CA addition groups in this study, which may also account for the improved TP digestion in CA addition group. Glucose in the serum of ruminant animals mainly comes from body gluconeogenesis, and CA has hypoglycemic and lipotropic effects which can promote the conversion of glucose to fat and tend to decrease blood glucose (Chaves et al. Reference Chaves, Dugan and Stanford2011). In this study, the blood glucose levels of two CA-added groups were significantly lower than control, which was in line with the results of the studies conducted by Chaves et al. (Reference Chaves, Dugan and Stanford2011). The other serum biochemical indexes ALP, GLT, and LDH were not significantly affected by CA addition, indicating that the addition of two concentrations of CA did not affect the normal function of liver, kidney, and fat.
It has been reported that CA has an antioxidant effect on the animal body. In this study, the serum T-SOD was increased, which was in line with results of Valko et al. (Reference Valko, Leibfritz and Moncol2007), while MDA was decreased with the increase of CA additional level. T-SOD is the main member of the feed radical scavenging system in the body, which can protect cell membranes from oxidation and maintain metabolic homeostasis of oxygen radicals (Wang et al. Reference Wang, Wang and Xing2021). Reactive oxygen species (ROS) is the main cause of peroxidative damage in the organism, while MDA was the final product of lipid peroxidation caused by ROS. CA can eliminate ROS in the organism, which in turn can reduce the MDA content in the organism. The content of endogenous antioxidant enzymes including CAT, T-AOC, and GSH-px in the two CA-added groups tended to be higher than control though there were no significant differences, indicating the addition of CA is beneficial for sheep antioxidant ability improvement, which was in agreement with the findings of An et al. (Reference An, Wang and Wang2020).
Serum immunoglobulin content such as IgG, IgA, and IgM can reflect the healthy situation of immunology organs and the body’s ability to resist the invasion of pathogens such as bacteria and viruses. The immunology-modulatory effects of EO or CA were confirmed previously in ruminants, poultry, and swine (Barbour et al. Reference Barbour, Shaib and Azhar2013; El-Azrak et al. Reference El-Azrak, Morsy and Soltan2022; Zhang et al. Reference Zhang, Piao and Ma2015). In this study, the content of IgG in two CA-added groups was higher than that of the control group, and further supporting the conclusion dietary CA supplementation might influence the humoral immune responses of Hu sheep. The reason may be because the addition of CA into the sheep diet increased its nutrient digestibility and provided enough nutrition for natural killer cells and T cells, which promoted their activity.
Conclusions
Adding an appropriate amount of CA to the ration of Hu sheep significantly improved the growth performance of Hu sheep, promoted the digestibility and absorption of nutrients, improved body antioxidant and immunological capacity, and improved ruminal fermentation by increasing the richness and diversity of rumen bacteria. Among them, the addition of 400 mg/kg SiO2 adsorbed CA additive, which is equal to 100 mg/kg pure CA, to Hu sheep diet on a TMR DM basis had the best effect.
Author contributions
Bo Lin: Conceptualization, funding acquisition, reviewing, and editing. Deshuang Wei: Feeding experiment, original writing, participate in the measurement of indicators, data Analysis; Qi Yan: Conceive and revised the article; Qin Zhang: Revision of the paper. Xin Gao: Samples were collected and ammoniacal nitrogen, nutrient apparent digestion indices were determined; Cunyu Zhang: Participated in digestion and metabolism tests and sample collection; Ruizhanghui Wang: Determination of volatile fatty acids and sample collection.
Financial support
Scientific Breeding Post of Guangxi Beef Cattle and Sheep Innovation Team of National Modern Agricultural Industry Technology System (nycytxgxcxtd-2021-09-03); National Key Research and Development Program (2022YFD1300602); Guangxi Major Science and Technology Project (Guangxi AA22068099-5).
Conflict of interest
There was no conflict of interest.