Introduction
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are a class of glucose-lowering agents that are approved by the US Food and Drug Administration for the treatment of type 2 diabetes mellitus (T2DM) and obesity, lowering the risk of major adverse cardiovascular events, as well as lowering the risk of worsening kidney disease, kidney failure, and death due to cardiovascular disease in adults with type 2 diabetes and chronic kidney disease.Reference Collins and Costello1 GLP-1 RAs mimic the effects of endogenous GLP-1, which is an endocrine hormone produced in the L-cells of the intestine.Reference Zheng, Zong and Ma2 Various metabolic effects have been associated with GLP-1, including stimulation of insulin secretion, inhibition of glucagon secretion, slowing gastric emptying, and increasing satiety.Reference Zheng, Zong and Ma2 In addition to peripheral metabolic effects associated with GLP-1 RAs, extant literature reports GLP-1 RAs have effects within the central nervous system including improvements in cognitive and reward function, reducing the severity of the disparate domains of suicidality as well as protecting against neurodegeneration.Reference Au, Zheng and Le3–Reference McIntyre, Rasgon and Goldberg7 While the putative mechanism of action of GLP-1 RAs mainly focuses on agonism of the GLP-1 receptor, the downstream effects of GLP-1 receptor activation as well as potential off-target effects of GLP-1 RAs remain incompletely understood. Moreover, while GLP-1 RAs’ anti-obesity mechanism of action mainly focuses on effects in the periphery, GLP-1 receptors have been reported to be present in multiple brain regions including, but not limited to, the hypothalamus, amygdala and the hippocampus.Reference López-Ojeda and Hurley8 Research of GLP-1 RAs has also extended toward indications in mental disorders (ie, major depressive disorder [MDD], substance use disorder), which instantiates a need to further evaluate the effects of GLP-1 in the brain.Reference McIntyre, Powell and Kaidanovich-Beilin9
Notably, glutamate is the most widely expressed excitatory neurotransmitter in the brain with glutamatergic neurons being expressed throughout the brain and spinal cord.Reference Pal10 Glutamate signaling and its regulation is crucial for neurodevelopment, learning and memory, and overall brain function.Reference Pal10, Reference Fairless, Bading and Diem11 In addition, the glutamatergic system has been implicated in a number of diseases, including endocrine, neurodegenerative, and mood disorders.Reference Murrough, Abdallah and Mathew12 Furthermore, the glutamate system also regulates food intake and appetite regulation.Reference Delgado13, Reference Smith, Ogunseye, DeBenedictis, Peris, Kasper and Hommel14
Replicated study results indicate GLP-1 receptors are present on glutamatergic neurons.Reference McIntyre, Rasgon and Goldberg7, Reference Kabahizi, Wallace and Lieu15, Reference Cork, Richards, Holt, Gribble, Reimann and Trapp16 Specifically, preliminary evidence reports that activation of GLP-1 receptors on the glutamatergic neurons increases glutamate release to regulate autonomic and metabolic functions including food intake suppression.Reference Cork, Richards, Holt, Gribble, Reimann and Trapp16 In addition to overlapping physiological effects of the glutamatergic and GLP-1 systems, the foregoing observations suggest the presence of functional connectivity between the two signaling systems. Herein, we sought to determine whether GLP-1 RA signaling involves activity at the canonical glutamatergic signaling pathway. Secondarily, we aimed to evaluate whether GLP-1 RAs have effects on feeding behavior or weight change that are mediated by effects on the glutamatergic system.
Methods
Databases and search strategy
This systematic review was conducted in accordance with the 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses.Reference Page, McKenzie and Bossuyt17 To evaluate the effects of GLP-1 RAs on glutamatergic signaling, we performed a systematic search on PubMed, Ovid (ie, Medline, Embase, APA PsychInfo, AMED, JBI EBP), and Scopus databases from inception to March 5, 2025. An additional manual search was conducted on Google Scholar and through citation searching to ensure all possible studies were captured. For details on the search strings used for each database, see Supplementary Table S1.
Screening and eligibility criteria
Studies that were identified in the database search were screened independently by two reviewers (S.W. and G.H.L.) using the Covidence platform. Studies were first screened by title and abstract. For studies to proceed to the full-text screening stage, individual studies must have been considered relevant by at least one of the reviewers. Studies that were screened by their full text were screened against the eligibility criteria. Studies to be included must (1) be a primary study (ie, randomized controlled trial, open-label trial, intervention trial, animal study, in vitro study), (2) investigate the effects of a GLP-1 receptor agonist on glutamatergic and/or NMDA/AMPA receptor activity in vitro and/or in vivo, (3) for human participants, they must be 18–64 years of age, inclusive, (4) be published or translatable to English, and (5) have full-text availability. Studies were excluded if they met at least one of the following exclusion criteria: (1) non-primary research (e.g., review articles, commentaries, letters to the editor, dissertations, protocols, published abstracts), (2) case reports or case series, (3) does not use a GLP-1 receptor agonist as an intervention, and (4) does not report on glutamatergic or NMDA/AMPA receptor activity. Studies were not excluded based on medical diagnosis of human participants or for the type of animal model being employed. Studies that were included must be unanimously agreed upon by both reviewers. Any discrepancies were resolved through discussion.
Data extraction
Data was extracted from the included studies by one reviewer (S.W.). The data extraction stage was conducted using a piloted data extraction table. Data to be extracted were established a priori and included the following: (1) Author(s) and publication date, (2) study design (ie, in vitro, animal, clinical), (3) sample size, (4) sample characteristics (eg, animal model, genotype, species), (5) GLP-1 RA investigated and dosing regimen, (6) outcome measures, and (7) main results.
Risk of bias analysis
Included studies were analyzed for risk of bias based on the study design utilized. Animal studies were evaluated for their methodological quality and risk of bias using the SYRCLE’s risk of bias tool.Reference Hooijmans, Rovers, de Vries, Leenaars, Ritskes-Hoitinga and Langendam18 As there is currently no validated risk of bias tool to analyze in vitro studies, we were unable to evaluate the included in vitro studies. Risk of bias assessments were conducted independently by two reviewers (S.W. and G.H.L.). Any differences in ratings were resolved through discussion.
Results
Search results and component study characteristics
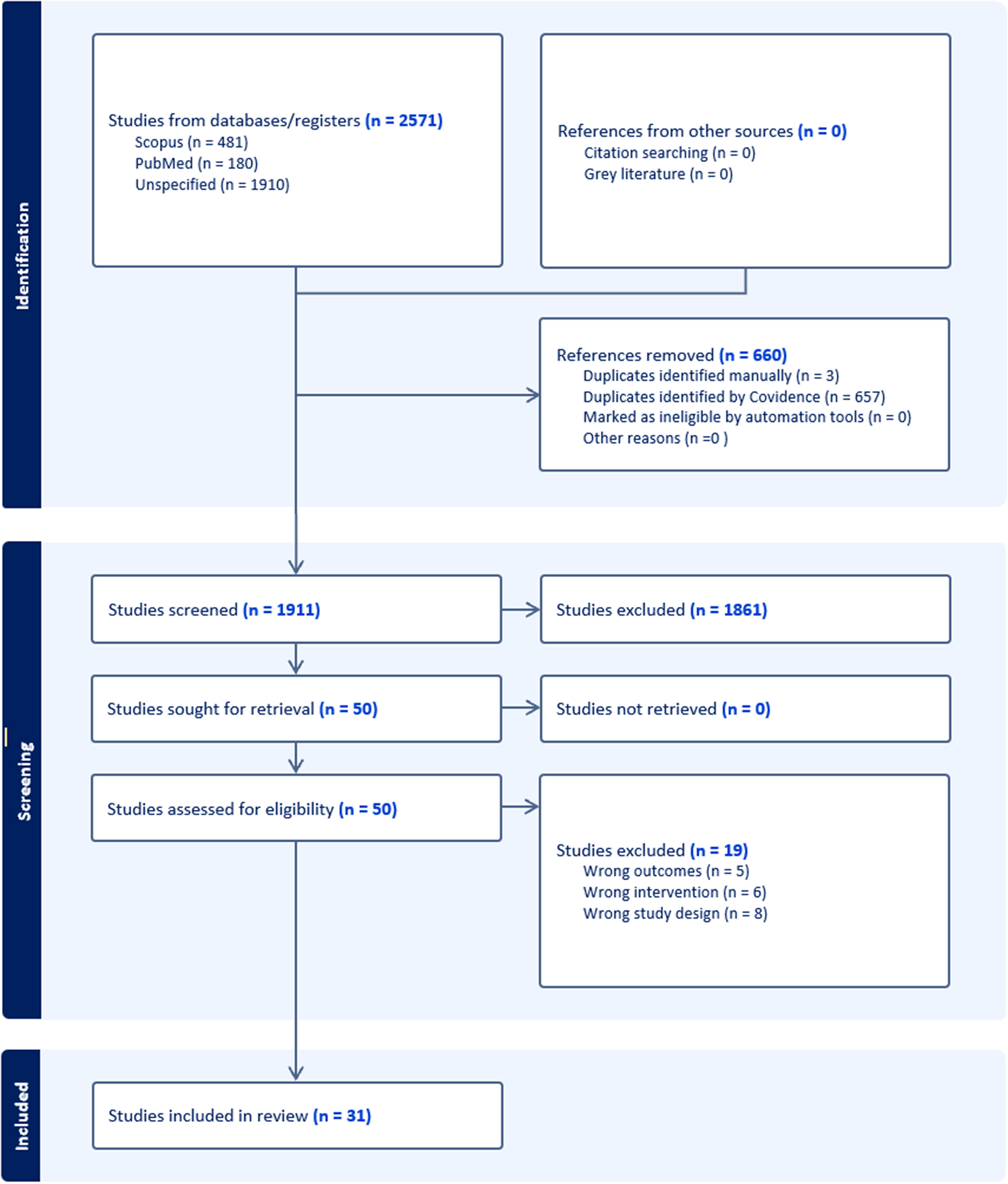
The search resulted in the identification of 2574 articles. Following the automatic removal of 660 duplicates, 1914 studies underwent title and abstract screening. A total of 50 studies were considered relevant and underwent full-text screening. Of the 50 studies, a total of 31 studies were considered eligible for inclusion in this review (n = 31) For full details on the study screening process, see Figure 1.

Figure 1. Flow Diagram of Study Screening and Inclusion Process.
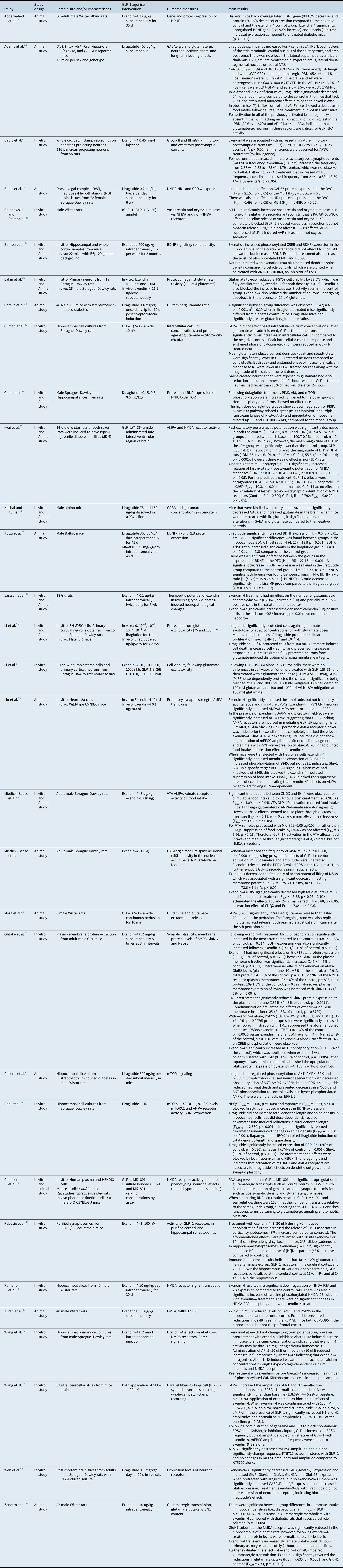
Of the 31 studies, 11 studies exclusively utilized in vitro methods, 11 studies exclusively utilized in vivo methods, and 9 studies utilized both in vitro and in vivo methods. We did not identify any human studies that evaluated the effects of GLP-1 RAs on glutamatergic signaling. From the included studies, the investigated GLP-1 RAs included liraglutide, dulaglutide, exendin-4/exenatide and GLP-1(7–36) amide. For details pertaining to the characteristics of each included study, refer to Table 1.
Table 1. Study Characteristics of Included Component Studies

Risk of bias results
From the risk of bias analysis, across all of the included studies, there are multiple sources of potential bias. Specifically, the most common sources of bias included an inadequate description of the randomization and blinding methods. As originally described by Hoojimans et al.Reference Hooijmans, Rovers, de Vries, Leenaars, Ritskes-Hoitinga and Langendam18 randomization and blinding of assessors is not standard practice for animal studies, which may explain why they are not commonly reported in the component studies. Inadequate blinding of study assessors and animal care staff may indirectly influence behavioral outcomes in the animals or may cause time differences when obtaining outcome measures. Differences in outcome measures, especially with metabolic and neuronal outcomes, may significantly interfere with the observed outcomes. Consequently, these and other potential sources of bias may weaken the certainty and strength of the results reported herein. Moreover, the aforementioned sources of bias may affect our inferences and interpretation of the results. Study-specific ratings for each domain are detailed in Supplementary Table S2. In addition, the overall strength of the body of evidence for each included study was assessed using the GRADE Approach for Preclinical Studies, which is detailed in Supplementary Table S3.
Effects of GLP-1 RAs on glutamate receptor activity and expression
Twelve of the included studies reported on the effects of GLP-1 RAs on glutamate receptor activity and expression. A study conducted by Adams et al. reports that 400 μg/kg liraglutide in vivo was associated with significant increases in Fos + cells across various brain regions including the central amygdala (CeA), lateral parabrachial nucleus (lPBN), bed nucleus of the stria terminalis (BNST), caudal nucleus of the solitary tract (cNTS), and the area postrema.Reference Gilman, Perry, Furukawa, Grieg, Egan and Mattson20 Notably, the increase in Fos + cells in the CeA and BNST were mostly GABAergic vGAT-GFP+ neurons while the lPBN primarily expressed glutamatergic vGlut2-GFP+ neurons.Reference Gilman, Perry, Furukawa, Grieg, Egan and Mattson20 The area postrema and the cNTS were heterogeneous with expression of both vGlut2I- and vGAT-GFP neurons. Furthermore, the aforementioned areas with Fos activation was absent in the vGlut deficient mice, indicating that liraglutide directly activates a population of glutamatergic GLP-1 receptor-expressing neurons with subsequent engagement of a neural network that is both glutamatergic and GABAergic to elicit physiological effects.Reference Gilman, Perry, Furukawa, Grieg, Egan and Mattson20
Administration of GLP-1 RAs in hippocampal cultures derived from Sprague–Dawley rats revealed that GLP-1 does not affect basal intracellular calcium concentrations; however, when neurons were pretreated with GLP-1 prior to glutamate administration, GLP-1 significantly attenuated increases in intracellular calcium compared to negative controls.Reference Adams, Pei and Sandoval27 Moreover, peak intracellular responses and sustained calcium elevation were reduced following GLP-1 treatment. The aforementioned observation was also accompanied by decreased mean glutamate-induced current densities.Reference Adams, Pei and Sandoval27
Similarly, GLP-1 RA administration inhibited Aβ1–42-induced increases in intracellular calcium concentrations in hippocampal slices of Sprague Dawley rats.Reference Wang, Wang, Jiang, Yuan, Yu and Li46 Administration of AP-5 and nifedipine, N-Methyl-D-Aspartate (NMDA) receptor antagonists, also inhibited Abeta1–42-mediated increases in intracellular calcium concentrations. Taken together, GLP-1 RAs may have cytoprotective effects through modulating intracellular calcium concentrations via L-type voltage-dependent calcium channels and NMDA receptors.
When considering the effects of GLP-1 RAs on ionotropic glutamatergic receptor activity, GLP-1(7–36) amide administered into the lateral ventricular brain region of rats with type 1 juvenile diabetes mellitus increased I-O relation of fast excitatory postsynaptic potentiation of NMDA responses (JDM alone: RReference Zheng, Zong and Ma2 = 0.820; JDM + GLP-1: RReference Zheng, Zong and Ma2 = 0.891; F1,107 = 5.17, p < 0.05).Reference Iwai, Suzuki, Kobayashi, Mori, Mogi and Oka29 Furthermore, ifenprodil co-treatment antagonized GLP-1(7–36) amide’s effects (JDM + GLP-1: RReference Zheng, Zong and Ma2 = 0.895; JDM + GLP-1 + ifenprodil: RReference Zheng, Zong and Ma2 = 0.959; F1,65 = 10.3, p < 0.01).Reference Iwai, Suzuki, Kobayashi, Mori, Mogi and Oka29
A separate study conducted by Mietlicki-Baase et al. reported that exendin-4 was associated with increased medium spiny neuron miniature excitatory postsynaptic current (mEPSC) frequency (t = 15.60, p < 0.0001) without affecting mEPSC kinetics and amplitude.Reference Gateva, Hristov and Ivanova26 In addition, exendin-4 decreased the paired-pulse ratio of evoked EPSCs (t = 4.31, p < 0.01) as well as decreased the frequency of action potential firing of medium spiny neurons, which was associated with a decreased resting membrane potential (aCSF = −75.3 ± 1.3 mV, aCSF + Ex-4 = −78.6 ± 1.1 mV; p < 0.02). Therefore, GLP-1 receptor activation also activates medium spiny neurons presynaptically through AMPA/kainate glutamatergic signaling.
When considering the effects of GLP-1 RAs on glutamatergic receptor activity, Babic et al. investigated the effects of GLP-1 RAs specifically on group II and III metabotropic glutamate receptor (mGluR) inhibitory and excitatory postsynaptic currents.Reference Babic, Sellers and Else22 GLP-1 was associated with increased miniature inhibitory postsynaptic currents (0.79 +/− 0.12 to 1.27 +/− 0.25 events s−1, p < 0.05), which was similar to what was observed with APDC, a group II mGluR agonist.Reference Babic, Sellers and Else22 With respect to miniature excitatory postsynaptic currents, GLP-1 RAs increased the current frequency from 2.83 +/− 0.82 to 4.88 +/− 1.79 events/s, which differed from L-AP4 treatment that increased mEPSC frequency. The aforementioned findings suggest that both group II and III mGluRs are involved in GLP-1 RAs activity.
Individual GLP-1 RAs differentially affect the activation and expression of NMDA receptor subunits. Specifically, Babic et al. reported that in vivo 0.2 mg/kg liraglutide in Sprague Dawley rats had no effects on GAD67 (F5,40 = 2.152, p > 0.05) and NR1 protein expression in the dorsal vagal complex (F5,42 = 0.493, p > 0.05) as well as the mediobasal hypothalamus (GAD67: F5,43 = 2.056, p > 0.5; NR1: F5,41 = 0.489, p > 0.05).Reference Babic, Sellers and Else22 Similarly, Ohtake et al. report that exendin-4 had no effect on NR1 expression (plasma membrane: 105 ± 6% of the control, p = 896; total protein: 109 ± 3% of the control, p = 0.779).Reference Ohtake, Saito, Eto and Seki39 Separately, GLP-1(7–36) amide enhanced the activity of the NR2B subunit of the NMDA receptor but did not alter NR2A or NR2B expression.Reference Iwai, Suzuki, Kobayashi, Mori, Mogi and Oka29 In contrast, intraperitoneal administration of exendin-4 10 μg/kg/day in the hippocampal slices of male Wistar rats was associated with significant downregulation of NR2A and -2B expression compared to control rats.Reference Romano, Villani, Sangineto, Cassano and Serviddio44 The downregulation observed was accompanied with a significant increase in tyrosine phosphorylated NR2B levels, but not NR2A.Reference Romano, Villani, Sangineto, Cassano and Serviddio44
The glutamatergic effects of GLP-1 RAs were further confirmed through RNA sequencing, which revealed that administration of disulfide-bonded GLP-1 and MK-801, a NMDA receptor antagonist, (GLP-1-MK-801) resulted in 1568 unique transcripts along with significant upregulation in glutamatergic transcripts such as Grin2a, Grin2b, Shisa6, and Slc17a7. Reference Petersen, Ludwig and Juozaityte42 Moreover, compared to semaglutide and MK-801 alone, GLP-1-MK-801 resulted in greater transcript upregulation and enrichment of functional terms, notably transcripts that participate in glutamatergic signaling and synaptic plasticity.Reference Petersen, Ludwig and Juozaityte42
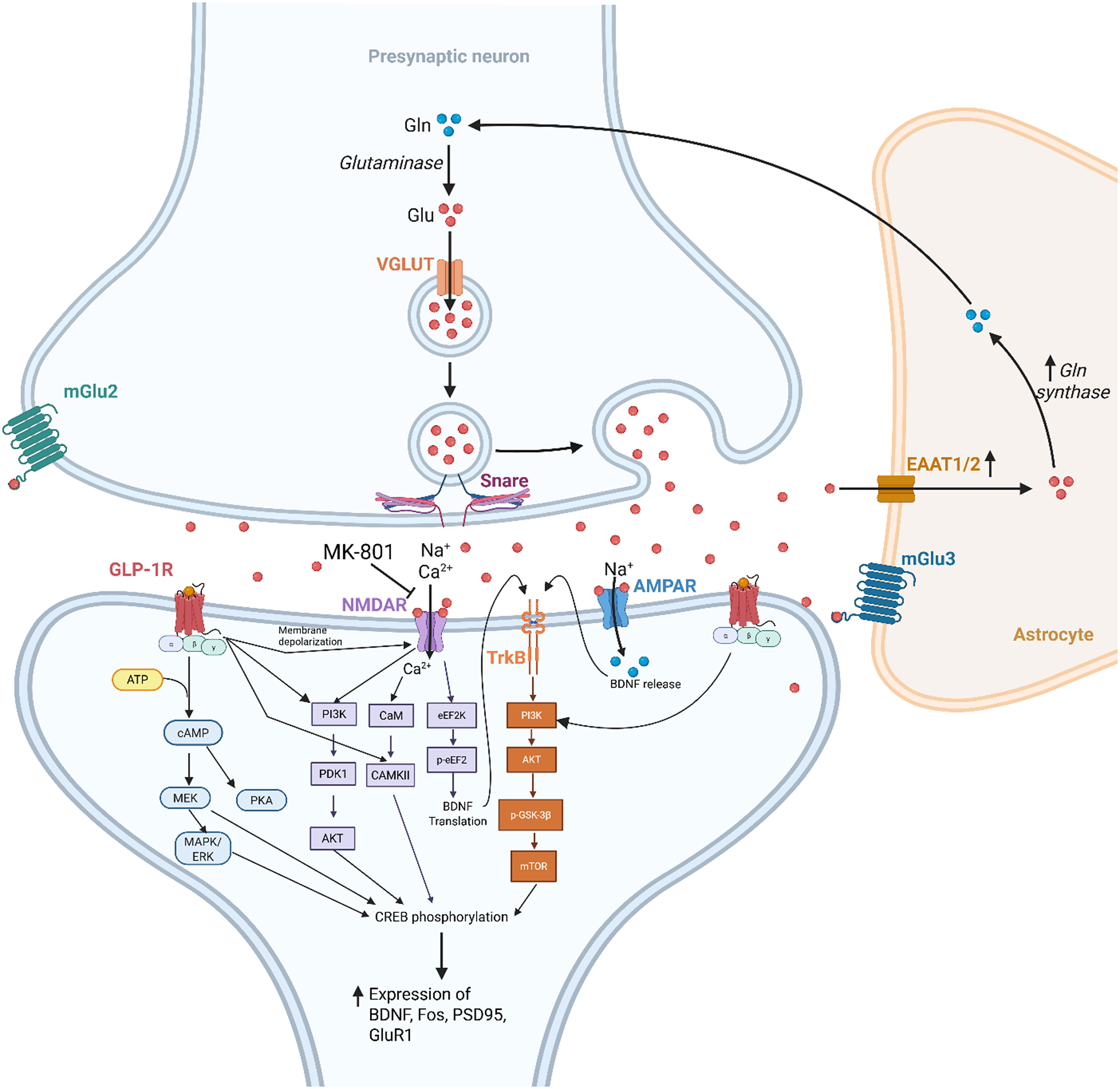
In addition, GLP-1 RAs were also associated with differential modulation of AMPA receptor expression. GLP-1 RAs differentially affected AMPA GluR expression wherein GluR1 expression in the plasma membrane fraction was significantly increased (145 +/− 6% of control, p < 0.001) but had no effects on GluR2 (plasma membrane: 101 ± 3% of the control, p = 0.913; total protein: 94 ± 7% of the control, p = 0.815).Reference Ohtake, Saito, Eto and Seki39 Plasma membrane expression of PSD95 increased with a commensurate increase in GluR1 expression (133 +/− 6%, p = 0.004) compared to the control group, which suggests that GLP-1 induces GluR1 and PSD95 insertion into the synaptic membrane.Reference Ohtake, Saito, Eto and Seki39 In contrast, Wen et al. reported that in hippocampal slices in seizure-induced Sprague–Dawley rats, exendin9–39, a GLP-1 receptor antagonist, significantly decreased GABAARbeta2/3 expression and increased GluR expression (ie, GluA1–4, GluN1, GluN2A, GluN2B).Reference Wen, Wu, Xie, Dan, Zhan and Shi48 When samples were treated with GLP-1 RA, there was increased GABAARβ2/3 expression and decreased GluR expression.Reference Wen, Wu, Xie, Dan, Zhan and Shi48 Overall, the included studies support that GLP-1 RAs may be associated with increased glutamatergic receptor activity via multiple cellular and molecular mechanisms (Figure 2).

Figure 2. Functional Connectivity Between GLP-1 and Glutamate Receptors.
Effects of GLP-1 RAs on glutamate release, uptake, and glutamate toxicity
We identified four studies that evaluated the effects of GLP-1 RAs on glutamate release and uptake. Specifically, Mora et al. reported that continuous perfusion of GLP-1 RA was significantly associated with increased extracellular glutamine and glutamic acid release.Reference Mora, Expösito, Sanz and Blázquez38 The aforementioned trend was replicated by Gateva et al. who reported a significantly greater glutamine/glutamate ratio in streptozotocin-induced diabetic mice treated with 0.4 mg/kg liraglutide.Reference Gateva, Hristov and Ivanova26 In addition, when mice had pentylenetetrazole-induced epilepsy, there were significant reductions in GABA and increased glutamate, which were prevented through pre-treatment with liraglutide post-mortem.Reference Koshal and Kumar30
Separately, Rebosio et al. investigated the effects of GLP-1 RAs on [H3]D-aspartate release in purified synaptosomes.Reference Rebosio, Balbi, Passalacqua, Ricciarelli and Fedele43 Notably, KCl-induced depolarization was potentiated with GLP-1 RA treatment wherein there was a 37% and 43% increase in [H3]D-aspartate release in the cortical and hippocampal synaptosomes, respectively.Reference Rebosio, Balbi, Passalacqua, Ricciarelli and Fedele43 Administration of exendin-3, a GLP-1 receptor antagonist, and 2′-5′-dideoxyadenosine both inhibited the observed increased in [H3]D-aspartate, suggesting that activation of GLP-1 receptors is associated with increased glutamatergic signaling.
When considering glutamate uptake, Zanotto et al. observed significant between-group differences in hippocampal glutamate uptake between diabetic and control mice (F2,14 = 10.84, p = 0.0014) wherein diabetic mice had significantly lower glutamate uptake.Reference Zanotto, Hansen and Galland49 Following GLP-1 RA treatment, there was a significant reversal in glutamate uptake impairment wherein a 68.3% increase in glutamatergic metabolism was observed compared to diabetic rats that received negative control (p = 0.0005). Moreover, GLP-1 RAs transiently increased glutamate uptake up to 24 hours post-treatment in primary astrocytes as well as acutely (ie, 1 hour post-treatment) in hippocampal slices.
Four of the included studies investigated the protective effects of GLP-1 RAs on glutamate-mediated neuronal toxicity. Across all four studies, GLP-1 RAs were significantly associated with protection against glutamate-induced neuronal death. Specifically, at 60 μM glutamate, GLP-1 RA-treated primary hippocampal neurons were reported to have a 10% reduction in neuronal cell viability 24 hours post-treatment, which significantly differed from the 55% reduction observed in the saline-treated neurons.Reference Adams, Pei and Sandoval27 Similarly at 100 mM glutamate, there was a 37.5% reduction in SH-SY5Y cell viability, which was fully ameliorated by exendin-4 at 1 and 100 uM.Reference Eakin, Li and Chiang25 Protection against cell death in SH-SY5Y cells were further replicated with liraglutide and GLP-1(9–36) amide at 75–150 mM.Reference Li, Bader and Tamargo33, Reference Li, Glotfelty, Karlsson, Fortuno, Harvey and Greig34 The foregoing triangulation of evidence indicates that GLP-1 RAs may be associated with increased glutamate metabolism and uptake to protect cells against glutamate excitotoxicity.
Effects of GLP-1 RAs NMDA/AMPA signal transduction
Nine of the included studies reported on the effects of GLP-1 RAs on NMDA/AMPA signal transduction. Specifically, the expression and activity of secondary messengers including BDNF, mTOR, CREB, PSD95, and CaMKII were evaluated. In addition, one study reported on vasopressin and oxytocin release via NMDA and non-NDMA receptors following GLP-1 administration.Reference Bojanowska and Stempniak23
Notably, western blot analysis indicated that GLP-1 RA treatment was associated with increased phosphorylation of CREB, ERK5, PSD95, and BDNF in the hippocampus.Reference Bomba, Granzotto and Castelli24 The aforementioned results were also accompanied by increased activation of TrkB in the hippocampus. Furthermore, GLP-1 RA administration in diabetic mice was associated with significant BDNF gene (376.92%) and protein (115.13%) upregulation compared to untreated diabetic mice.Reference Abdelwahed, Tork, el Din, Rashed and Zickri19 BDNF upregulation was also replicated with liraglutide treatment.Reference Kutlu, Kose and Akillioglu31, Reference Park, Mansur and Lee41 Park et al. report that administration of NBQX, a AMPA receptor antagonist, with liraglutide completely ameliorated liraglutide’s effects on BDNF expression.Reference Park, Mansur and Lee41 When liraglutide was co-administered with MK-801, there were no significant differences in BDNF expression compared to liraglutide alone in the hippocampus (U = 10.0, p = 0.599, z = −0.5) and the prefrontal cortex (U = 0.0, p < 0.01, z = −2.8). The BDNF/TrkB ratio was significantly greater in the liraglutide-treated group compared to control in the hippocampus (U = 0.0, p < 0.01, z = −2.8), which was significantly ameliorated when co-treated with MK-801 (U = 0.0, p < 0.01, z = −2.7).Reference Park, Mansur and Lee41 Similarly in the prefrontal cortex, the liraglutide-treated group had significantly greater BDNF/TrkB ratio compared to the liraglutide and MK-801 co-treated group (U = 0.0, p < 0.01, z = −2.7).Reference Park, Mansur and Lee41 Taken together, GLP-1 RAs may modulate BDNF expression through interaction with the AMPA receptor (Figure 2).
In terms of the expression of second messengers in the PI3K cascade, GLP-1 RAs induced significantly increased phosphorylation of PI3K, Akt, and mTOR compared to the control group.Reference Guan, Xiao and Xie28, Reference Palleria, Leo and Andreozzi40 Notably, GLP-1 RA treatment significantly increased PSD-95 (156% of control, p = 0.020) and synapsin I (174% of control, p = 0.001) expression, which was ameliorated with rapamycin and NBQX.Reference Park, Mansur and Lee41 Furthermore, in Wistar rats that had 72 hours of rapid eye movement sleep deprivation, there were reduced levels of CaMKII and PSD95 in the hippocampus and prefrontal cortex, which was prevented with GLP-1 RA treatment.Reference Turan, Sayan Ozacmak, Ozacmak, Ergenc and Bayraktaroğlu45 Similar trends were observed in rats with Abeta1–42 treated with GLP-1 RAs wherein there was an increase in the number of phosphorylated CaMKIIalpha-positive cells in the hippocampus.Reference Wang, Wang, Jiang, Yuan, Yu and Li46 Taken together, GLP-1 RA’s effects on synaptic plasticity and neuroprotective effects may be mediated via mTOR subsequent to AMPA receptor activation (Figure 2).
A study conducted by Bojanowska and Stempniak investigated the effects of GLP-1(7–36) amide (tGLP-1) on vasopressin and oxytocin release through glutamate receptors.Reference Bojanowska and Stempniak23 tGLP-1 alone significantly increased both vasopressin and oxytocin release; however, administration of glutamate receptor antagonists, kynurenic acid (KA), AP-5, and DNQX differentially affected vasopressin and oxytocin release. Specifically, KA and AP-5 blocked tGLP-1-induced vasopressin secretion, but not oxytocin, while DNQX did not affect tGLP-1’s effects. As AP-5 abolished tGLP-1’s effects on vasopressin, this suggests that GLP-1’s effects on arginine-vasopressin neurons may be mediated through NMDA receptors.
Effects of GLP-1 RAs neuron morphology
From the included studies, four investigated the effects of GLP-1 RAs on neuron morphology, specifically dendritic spine density and protection against diabetes-induced neuropathological changes. Notably, in primary hippocampal neurons, GLP-1 RA treatment was associated with increased dendritic spine density compared to vehicle controls.Reference Bomba, Granzotto and Castelli24 When an inhibitor of TrkB, 10 μM ANA-12, was administered, GLP-1 RAs’ effects were blunted, suggesting that exenatide may induce increased dendritic spine density through BDNF–TrkB signaling. In rat models for juvenile type 1 diabetes mellitus, GLP-1 RA treatment improved the magnitude of long-term depression (JDM: 85.1+/− 6.1%, n = 6; JDM + GLP-1, 55.5 +/− 4.8%, n = 5; p < 0.0001), which was associated with activation of the NR2B.Reference Iwai, Suzuki, Kobayashi, Mori, Mogi and Oka29
In Goto-Kakizaki rats, a model for type 2 diabetes mellitus, GLP-1 RA treatment was associated with a 90% increase in the density of calbindin-positive cells in the striatum.Reference Larsson, Lietzau and Nathanson32 However, GLP-1 RAs had no effect on the number of glutamic acid decarboxylase-67, calretinin, and parvalbumin-positive cells in the striatum and cortex. Separately, GLP-1 RA pretreatment in Abeta1–42-treated hippocampal primary cell cultures significantly increased phosphorylated CaMKIIalpha-positive cells indicating that GLP-1 RAs may have neuroprotective effects through glutamate receptor-mediated responses.Reference Wang, Wang, Jiang, Yuan, Yu and Li46 Taken together, GLP-1 RAs may have protective effects against neuropathological changes by promoting neurogenesis and synaptic plasticity.
Effects of functional connectivity between GLP-1 and NMDA/AMPA receptors on food intake
From the included studies, three studies directly evaluated the effects of GLP-1 RAs on food intake and feeding behaviors following glutamatergic modulation. Specifically, Adams et al. measured short- and long-term feeding behaviors in Glp1r-flox compared to vGAT-, and vGlut2-deficient mice following 400 ug/kg liraglutide treatment.Reference Gilman, Perry, Furukawa, Grieg, Egan and Mattson20 In terms of short-term feeding both in normal and obese mice, liraglutide significantly decreased food intake in the vGAT-deficient mice but not in the vGlut2-deficient mice. Long-term treatment with liraglutide resulted in a plateau in weight loss effects for all groups.
Separately, a significant interaction effect of GLP-1 RAs and CNQX, an AMPA/kainate receptor antagonist, co-treatment was observed on food intake 3, 6, and 24 hours post-treatment (all ANOVAs F1,21 ≥ 4.89, p ≤ 0.04).Reference Mietlicki-Baase, Ortinski and Rupprecht36, Reference Mietlicki-Baase, Ortinski and Reiner37 Moreover, the foregoing trend indicates that antagonism of AMPA/kainate receptors significantly attenuates food intake suppression effects associated with GLP-1 RA treatment.Reference Gateva, Hristov and Ivanova26, Reference Mietlicki-Baase, Ortinski and Reiner37 The food suppressive effects were attributed to decreased meal size (F1,21 ≥ 6.11, p ≤ 0.03) and minimally on meal frequency (F1,21 ≥ 4.46, p ≤ 0.05).Reference Mietlicki-Baase, Ortinski and Rupprecht36 When the rats were co-treated with GLP-1 RA and MK-801, GLP-1 RA-induced suppression of food intake was not affected. Co-treatment of GLP-1 RAs and AP-5 also had no effect on food intake or body weight.Reference Mietlicki-Baase, Ortinski and Reiner37Taken together, the aforementioned triangulation of evidence indicates that GLP-1 RA-induced suppression of food intake and effects on body weight may be mediated through AMPA/kainate receptor signaling but not NMDA receptors.
Discussion
Herein, the results of our systematic review suggest that GLP-1 RAs’ ignite a molecular cascade of intracellular events directly. In addition, they also exert their cellular effects via cross functional activity with glutamate neurons and their consequent signal transduction cascades. In general, the included studies suggest that GLP-1 RA administration modulated intracellular calcium concentrations as well as AMPA and NMDA receptor depolarization. Notably, across disparate GLP-1 RAs, AMPA, and NMDA receptor subunit expression was differentially affected. For example, GLP-1 RAs were associated with upregulation of GluR1 with mixed results observed for the effects of GLP-1 RAs on NR1, NR2A, and NR2B. Across the evaluated GLP-1 RAs, there was a consistent trend wherein GLP-1 RA administration was associated with increased AMPA receptor expression and activation as well as increased expression and activity of second messengers associated with neurodifferentiation, neurogenesis and synaptic plasticity. Moreover, GLP-1 RA’s physiological effects on food intake were dependent on AMPA receptor, but not NMDA receptor, activity. Therefore, converging lines of evidence suggest that there is potential functional connectivity between glutamatergic and GLP-1 systems.
In accordance with previously reported studies, GLP-1 RAs administration is associated with increased neurodifferentiation and synaptic plasticity, which potentially mediates GLP-1’s effects on neuroprotection and cognitive and reward function.Reference Au, Zheng and Le3, Reference Au, Zheng and Le50 Our search results build upon previous studies reporting that GLP-1 RAs are present throughout the glutamatergic system wherein GLP-1 RAs directly interact with ionotropic glutamate receptors to increase the release of BDNF and mTOR activation. The aforementioned triangulation of evidence further supports current research evaluating the implementation of GLP-1 RAs as augmentation treatments for disparate mood and neuropsychiatric disorders that are significantly subserved by metabolic mechanistic underpinnings (eg, MDD, Alzheimer’s disease).Reference Li, Wong and Fisher51 Across various mood and neurocognitive disorders, central insulin resistance and impaired insulin signaling are strongly implicated in the disease and treatment prognosis.Reference Zhang, Han and Lu52–Reference Maksyutynska, Stogios and Prasad54 For example, persons with MDD are at a greater risk of developing metabolic syndrome.Reference Penninx and Lange55 Moreover, the directionality of depressive symptom severity and insulin resistance has not been fully established; however, persons with insulin resistance are at greater risk of developing treatment resistant depression, are at increased risk of suicidality and have an overall decreased health-related quality of life.Reference McIntyre, Alsuwaidan and Baune56, Reference Wong and Mansur57 Therefore, GLP-1 RAs may have broader indications for mood and neuropsychiatric disorders. In addition, replicated evidence indicates that GLP-1 RAs may be effective in the treatment of pain disorders through modulation of inflammatory signaling pathways.Reference Halloum, Dughem, Beier and Pellesi58 As pain disorders are similarly observed to be subserved by robust glutamatergic disruptions, GLP-1 RAs’ clinical efficacy in the treatment of pain disorders may be partially mediated through the glutamate system. In efforts to explore the aforementioned research area, a research vista that can be conducted in the near future is to design and execute read out demonstrably showing central glutamatergic activity as evidenced by magnetic resonance spectroscopy and electroencephalography.Reference Mansur, Subramaniapillai and Lee59, Reference Le, Wong and Badulescu60
There are methodological limitations to our systematic review that may affect the inferences and interpretation so the results. Notably, across the included component studies, there were differences in the research methodologies (eg, cell and/or animal model, dosing and frequency of the evaluated GLP-1 RAs, individual GLP-1 RA investigated). Therefore, between-study heterogeneity may limit our ability to determine the degree to which GLP-1 RAs affect glutamatergic signaling across disparate brain regions and how the trends would extrapolate to human populations. Furthermore, due to the aforementioned differences in study methodologies, we could not conduct a quantitative analysis to determine the degree of association or evaluate for the presence of publication bias. Notably within the included studies, several studies utilized models of diabetes and Alzheimer’s disease. Replicated evidence indicates that both diabetes and Alzheimer’s disease are differentially affected by disruptions in the glutamate system, including impaired glutamate uptake, altered receptor expression, and increased excitotoxicity.Reference Hristov, Nankova and Andreeva-Gateva61–Reference Talantova, Sanz-Blasco and Zhang63 These pathological states may amplify or modify the observed effects of GLP-1 RAs, potentially enhancing neuroprotective outcomes. While these models offer valuable insights into disease-specific mechanisms, they may limit the generalizability of findings to other clinical populations.
Finally, we did not identify any human studies reporting on the effects of GLP-1 RAs on glutamatergic signaling. Therefore, our findings may not directly translate to human populations across disparate disease states. Notwithstanding, these findings provide the impetus for future translational research, including early-phase clinical trials or neuroimaging studies, to assess GLP-1 RA-induced glutamatergic modulation. Moreover, given the established clinical use of GLP-1 RAs in metabolic disorders and emerging evidence of neuroprotective and cognitive effects, this mechanistic insight could inform the discovery and development of novel treatments for neuropsychiatric or neurodegenerative conditions characterized by glutamatergic dysfunction.
Conclusion
Our results indicate that GLP-1 RAs may directly and/or indirectly interact with the glutamate system to mediate their metabolic effects and neuroprotective effects. In addition, GLP-1 RAs may have glutamatergic effects that aid in their therapeutic efficacy for the treatment of disorders associated with metabolic perturbations. Future research should aim to conduct target engagement studies in humans to determine the effects of GLP-1 RAs on glutamatergic signaling and the effects across disparate brain regions. Further investigation of the central metabolic effects of GLP-1 RAs may further inform the research and development of mechanistically informed metabolic and psychotropic agents. In addition, future clinical trials should evaluate whether GLP-1 RAs may work additively or synergistically with co-administered glutamatergic signaling modulators in the treatment and prevention of mental disorders.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S1092852926100844.
Data availability statement
This systematic review did not generate or analyze new datasets. All data supporting the findings of this study are derived from previously published studies, which are cited in the reference list.
Author contribution
Conceptualization and Methodology: S. W., R. S. M.; Investigation: S. W., G. H. L.; Writing - Original draft: S. W., R. S. M.; Writing - Review & Editing: All authors.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Disclosures
Dr. Roger S. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China (NSFC) and the Milken Institute; speaker/consultation fees from Lundbeck, Janssen, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Neurawell, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, Abbvie, and Atai Life Sciences.
Kayla M. Teopiz has received fees from Braxia Scientific Corp.
Sabrina Wong and Gia Han Le declare no competing interests.