Introduction
Cytomegalovirus (CMV) is a ubiquitous, double-stranded DNA virus of the Herpesviridae family with significant public health implications, as it poses a risk to certain vulnerable populations [Reference Griffiths, Baraniak and Reeves1, Reference Boppana2]. Primary CMV infection is commonly acquired during early childhood, typically resulting in mild or asymptomatic infection in immunocompetent individuals [Reference Griffiths and Reeves3]. However, in immunocompromised individuals, such as solid organ and haematopoietic stem cell transplant recipients, CMV infection can lead to severe complications including graft rejection and organ damage [Reference Kotton4, Reference Ljungman5]. Furthermore, congenital CMV infection (cCMV) is a leading cause of sensorineural hearing loss and neurodevelopmental impairment globally [Reference Shoham6, Reference Rawlinson7].
Understanding the epidemiology of CMV is essential to assess population-level risk and to guide prevention strategies such as antenatal or newborn screening, the clinical management of transplant patients, and vaccination development [Reference Boppana2, Reference Fowler8]. Substantial variation in CMV prevalence has been reported globally, ranging from 40% in some developed countries to 90% in developing countries [Reference Fowler8, Reference Zuhair9]. Seroprevalence rates are influenced by demographic and socioeconomic factors, including age, sex, household crowding, and migration background [Reference Fowler8–Reference Hoehl10].
In Ireland, national CMV seroprevalence data are limited. Previous studies have reported low seroprevalence rates in Irish populations, including 33.4% in solid organ donors [Reference Hassan11] and 30.4% among pregnant Irish women compared to 89.7% in non-Irish women [Reference Knowles12]. These findings indicate substantially lower rates than reported in other European countries, with estimates of 41.9% and 45.6% in France and the Netherlands, respectively, [Reference Korndewal13, Reference Antona14] and 83.2% in Sweden [Reference Olsson15]. Collectively, these findings suggest that Ireland has one of the lowest CMV prevalence rates in Europe, which has implications for assessing population susceptibility to primary CMV infection.
CMV is transmitted through exposure to infected bodily fluids, including breast milk, saliva, genital secretions, urine, and blood [Reference Ziemann and Thiele16]. Following primary infection, the virus establishes lifelong latency, predominantly within mononuclear white blood cells, with potential for reactivation and reinfection [Reference Jückstock17]. Monitoring seroconversion rates can help identify groups with ongoing transmission risk and provide insights into age- or exposure-related trends [Reference Hyde, Schmid and Cannon18].
The present study was undertaken to estimate CMV seroprevalence and seroconversion among Irish blood donors, a large cohort that can serve as a surrogate population for infectious disease surveillance. This provides baseline data of CMV epidemiology to inform public health interventions, including cCMV prevention, future vaccination policy, and transfusion safety strategies.
Methods
Study population
The study population comprised 74,822 blood donors who provided 133,229 donations or samples to the Irish Blood Transfusion Service (IBTS) during 2020.
All donations from previously CMV seronegative donors, first-time donors, and sample-only new donors (SONDs), defined as first-time donors born outside of Ireland or the United Kingdom (UK), were screened for CMV IgG. Donors with a positive CMV IgG result on one occasion are permanently recorded as seropositive in the donor management system, and subsequent donations are not retested. Ethical approval for this study was not sought as testing and analyses were carried out as part of the IBTS routine service. Consent for infectious disease screening, including CMV, was collected at the time of donation or SOND.
CMV serological screening
CMV antibody screening was performed at the IBTS using a commercial chemiluminescent microparticle immunoassay (Alinity s CMV IgG Qualitative Assay, Abbott Laboratories), on the Alinity s serological testing platform according to the manufacturer’s instructions.
Investigation of recent CMV infection
To confirm recent CMV infection, a subset of donors who had seroconverted within an interval of 1 year from their last CMV-negative donation was selected for further analyses. These samples underwent CMV IgM testing using the Abbott CMV IgM assay on the Architect i2000 immunoassay analyser (Abbott Laboratories).
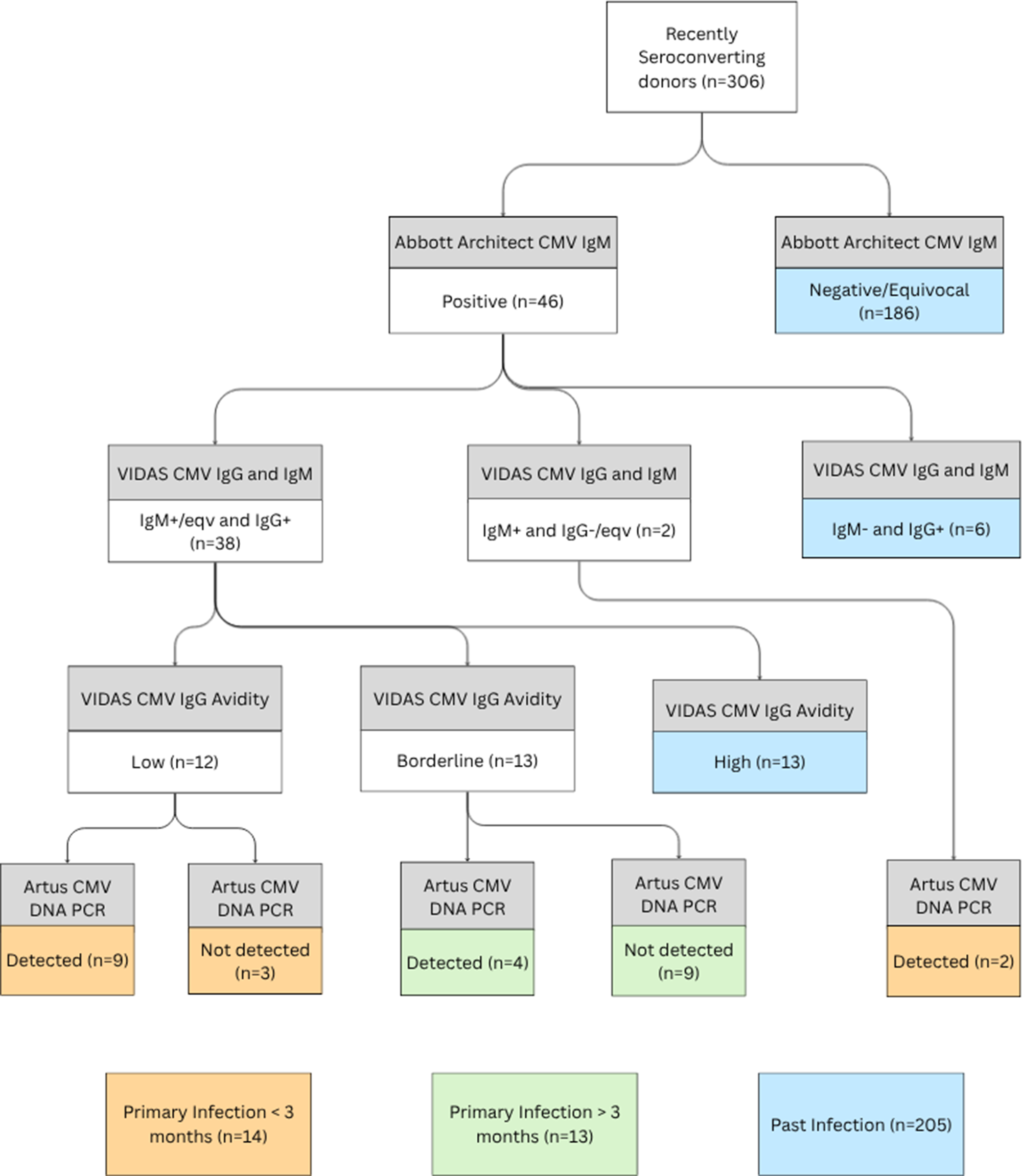
Samples that tested positive for CMV IgM were referred to the National Virus Reference Laboratory (NVRL), University College Dublin, for supplemental testing according to the study algorithm (Figure 1) as follows: The presence of CMV IgG and IgM was confirmed using the VIDAS CMV IgG and IgM assays (BioMérieux SA, RCS Lyon, France). Specimens that were reactive for CMV IgG were evaluated for avidity using the VIDAS CMV-IgG Avidity assay and were classified as either low, borderline, or high avidity, according to the manufacturer’s instructions. Samples with low and borderline avidity and those that were VIDAS CMV IgM only positive were tested for CMV DNA using the Artus CMV TM PCR (Qiagen, Valencia, CA).
Supplementary testing algorithm to confirm recency of CMV infection.

Data collection and analysis
Relevant data were extracted from the IBTS Blood Management System (eProgesa version 5.0.3) for all donors who presented in 2020. Variables included age, sex, donor type, geographical location of donation, country of birth, CMV serostatus at the end of 2020, and historical CMV results.
CMV seroprevalence was defined as the proportion of donors with a positive CMV status at the end of 2020 relative to the total donor population. CMV seroconversion was estimated among donors who presented in 2020 and had a documented CMV-negative result within the preceding 12 months. CMV seroconversion was defined as a first CMV-positive donation in 2020, following a CMV-negative result within the previous 12 months.
Statistical analysis was performed using Microsoft Excel and IBM SPSS version 29. Descriptive statistics are presented as percentages and numbers, 95% confidence intervals were calculated using the standard error of a proportion. Associations between serostatus and demographic variables were assessed using the Chi-Square test. Crude odds ratios (ORs) were estimated using univariate binary logistic regression; adjusted odds ratios (aORs) were estimated using multivariable binary logistic regression controlling for sex, age group, region of donation, and region of birth. Donor type was excluded from the multivariable model due to collinearity with country of birth. A value of p < 0.05 was considered statistically significant.
Results
CMV seroprevalence
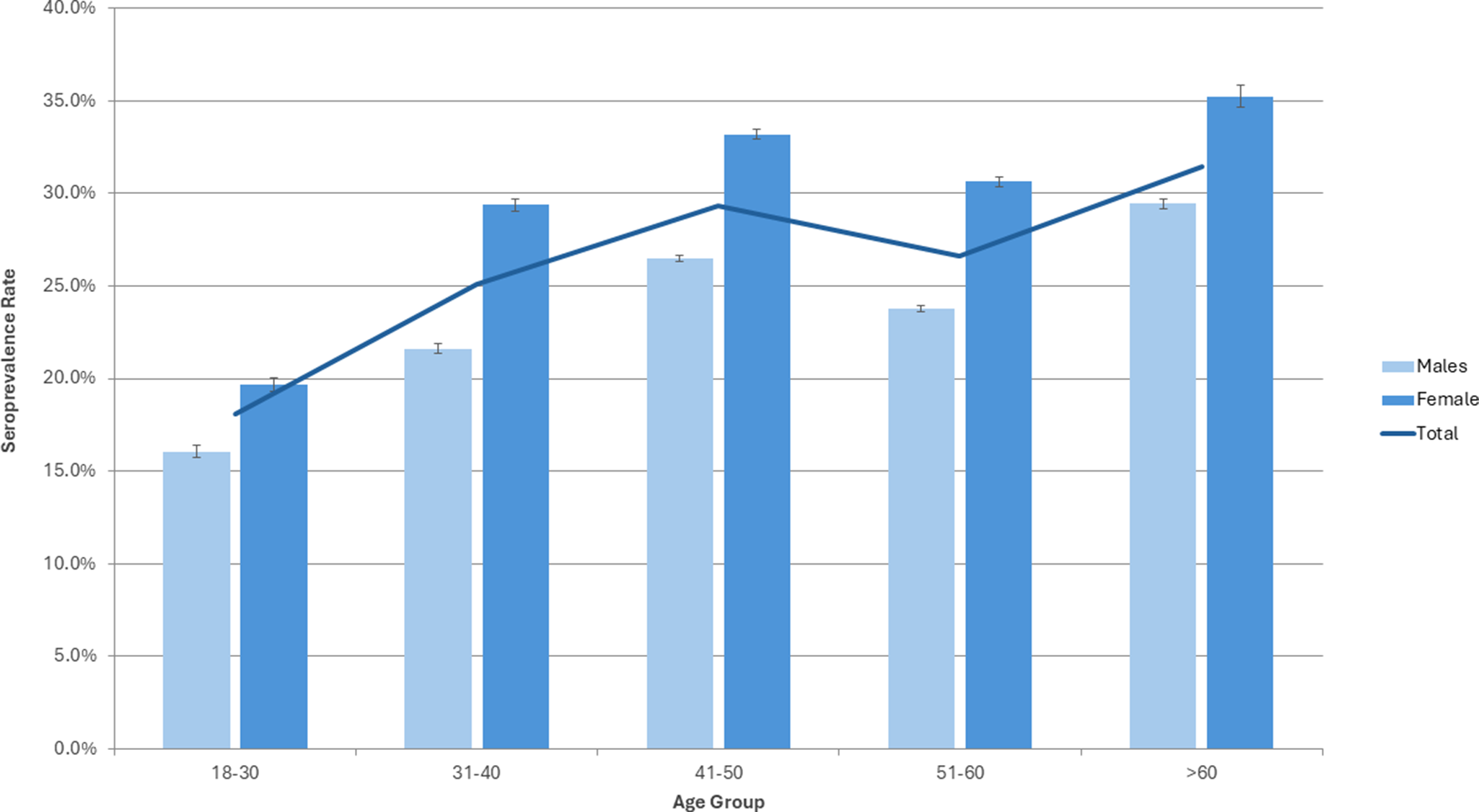
In 2020, CMV seroprevalence among Irish Blood Donors was 26.00% [95% CI: 25.69–26.32]. Seropositivity was significantly higher among females, at 28.88% [95% CI: 28.40–29.37] compared to 23.69% [95% CI: 23.28–24.10] in males. In multivariable logistic regression controlling for age group, geographical location, and region of birth, female donors had higher odds of seropositivity (aOR 1.38 [95% CI: 1.34–1.43]). This trend was consistent across all age groups (Figure 2).
CMV seroprevalence rate in Irish Blood Donors by age and sex 2020. Error bars represent 95% confidence intervals.

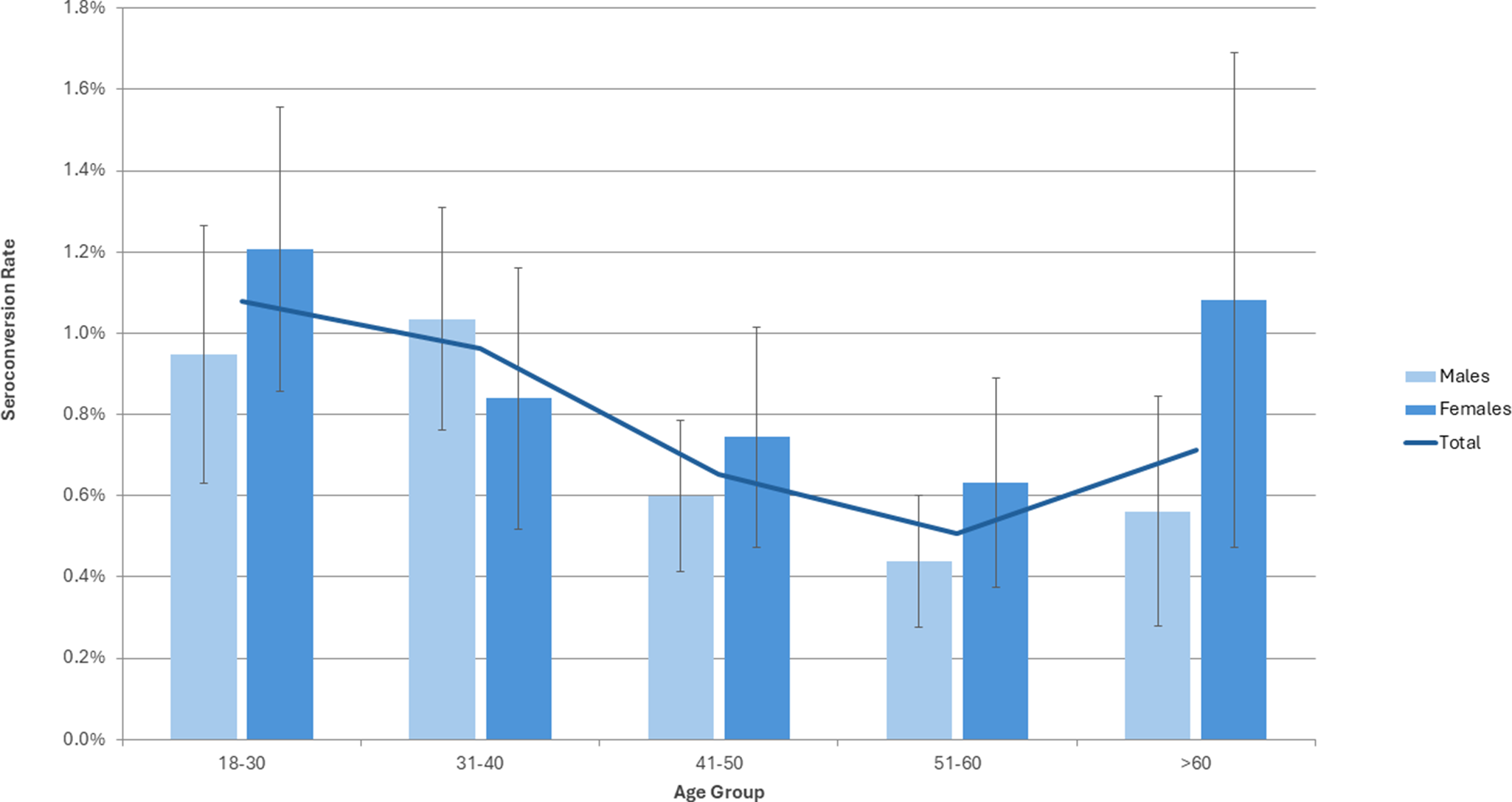
CMV seroconversion estimate in Irish Blood Donors by age and sex 2020. Error bars represent 95% confidence intervals.

An age-related trend was observed, with CMV seropositivity generally increasing with age in both males and females (Figure 2). Compared to donors aged 18–30 (seroprevalence 18.10% [95% CI: 17.45–18.75]), the adjusted odds of seropositivity were significantly higher across all older age groups, peaking in the >60 age group (aOR: 2.42, [95% CI: 2.27–2.58]) (Table 1). Among women of childbearing age (18–49 years), CMV seroprevalence was 27.53% [95% CI: 26.95–28.11].
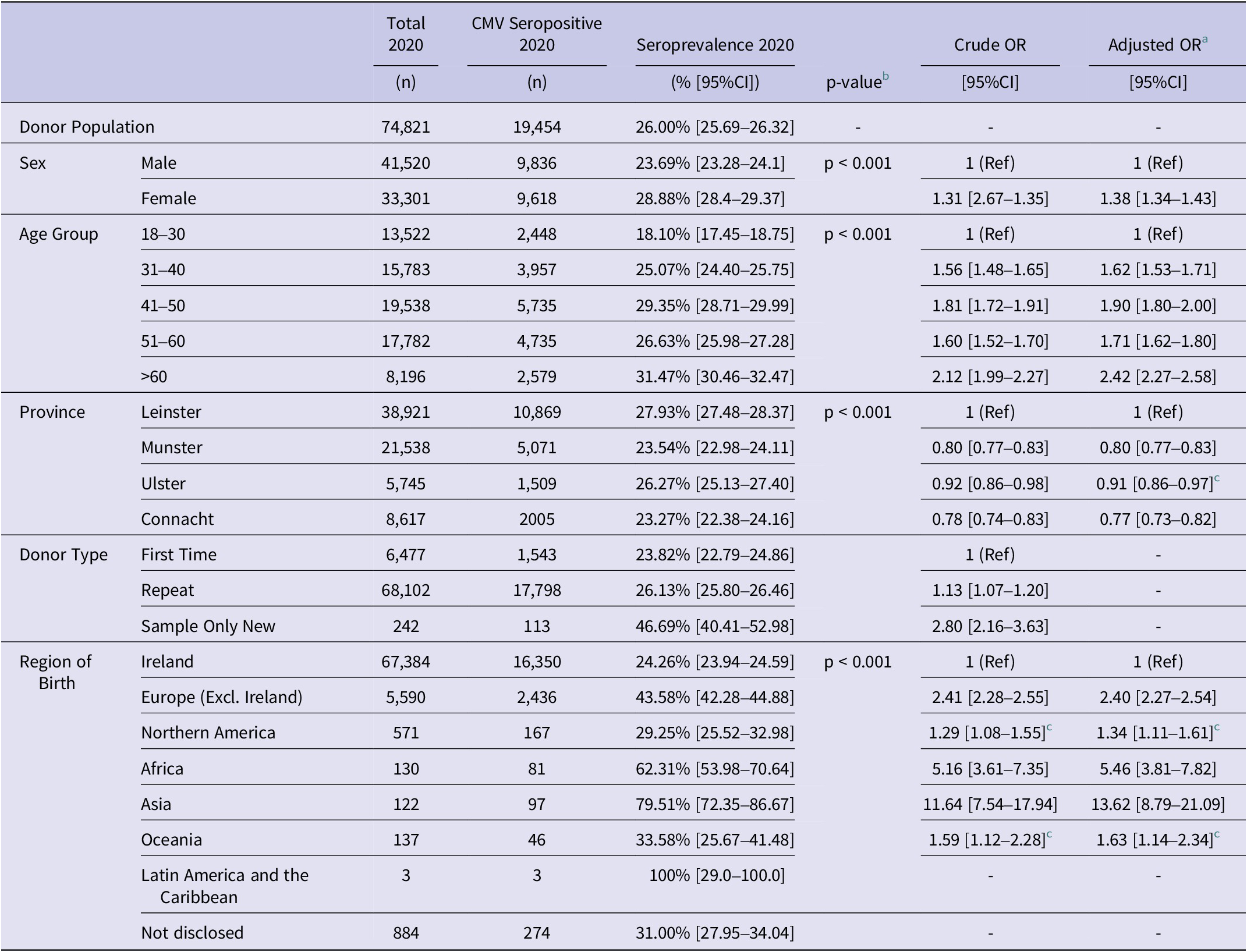
CMV Seroprevalence by demographic characteristics and year, with crude and adjusted odds ratios

a Adjusted Odds Ratios (aORs) derived from multivariable logistic regression controlling for sex, age group, region of birth, and geographical location.
b p-values calculated using Pearson’s Chi-Square Test of Independence.
c Not statistically significant (p > 0.05) in logistic regression.
CI = confidence interval, Ref = reference category.
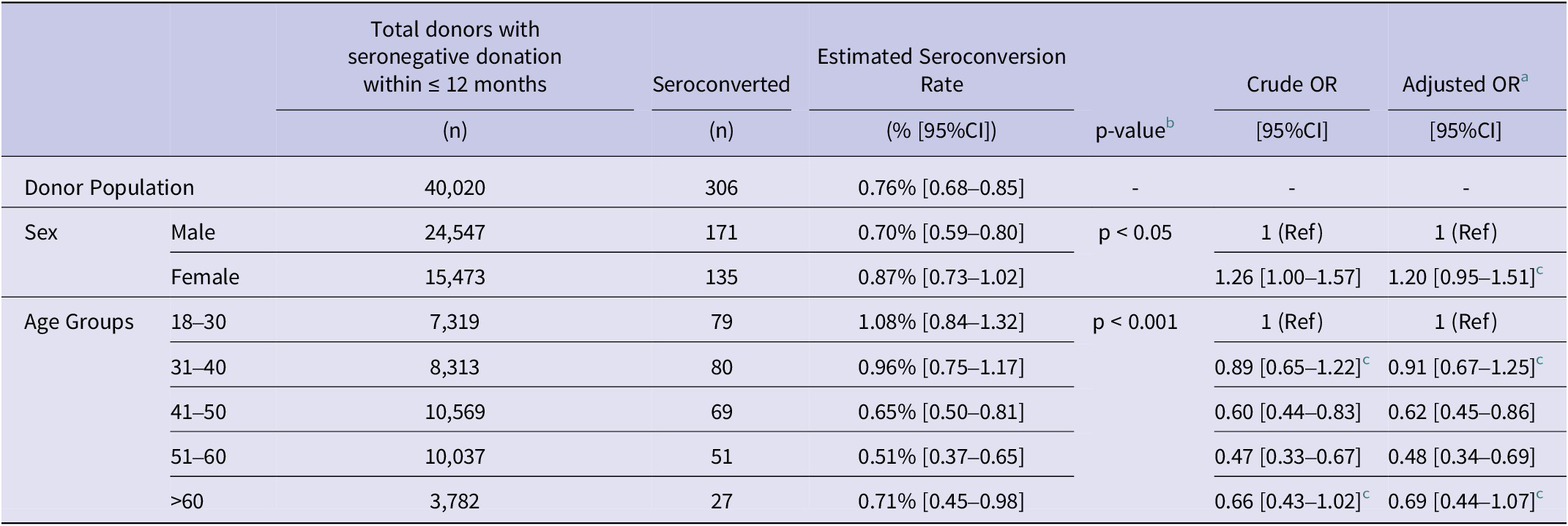
CMV Seroconversion Estimate by age and sex with crude and adjusted odds ratios

a Adjusted Odds Ratios (aORs) derived from multivariable logistic regression controlling for sex and age group.
b p-values calculated using Pearson’s Chi-Square Test of Independence.
c Not statistically significant (p > 0.05) in logistic regression.
CI = confidence interval, Ref = reference category.
Among first-time donors, CMV seroprevalence was 23.82% [95% CI: 22.79–24.86], whereas in first-time donors born outside of Ireland and the UK (SONDs), the seroprevalence was significantly higher, at 46.49% [95% CI: 40.41–52.98] (p < 0.001). In univariate analysis, SONDs had more than twice the odds of CMV seropositivity as first-time donors born in Ireland or the UK (OR: 2.80 [2.16–3.63]). Marked variation was observed by country of birth. Donors born in Ireland had a seroprevalence of 24.26% [95% CI: 23.94–24.59] compared to 43.58% [95% CI: 42.28–44.88] in those born in other European countries. The highest rates were seen in donors born in Africa (62.31% [95% CI: 53.98–70.64]) and Asia (79.51% [95% CI: 72.35–86.67]), though the number of donors from some regions was low (Table 1).
CMV seroprevalence varied by the geographical location of donation. The highest seroprevalence was observed among donors attending clinics in the East province of Leinster at 27.93% [95% CI: 27.48–28.37], lower rates were observed in the Western Province, Connacht (23.27% [95% CI: 22.38–24.16]), and Southern Province, Munster (23.54% [95% CI: 22.98–24.11]). After adjustment for age, sex, and region of birth, donors in Munster and Connacht had lower odds of CMV seropositivity compared to those in Leinster (aOR: 0.80 [95% CI: 0.77–0.83] and 0.78 [95% CI: 0.73–0.82] respectively).
CMV seroconversion
A total of 306 of 40,020 donors seroconverted in 2020, resulting in an estimated seroconversion rate of 0.76% [95% CI: 0.68–0.85] among those with a CMV-negative result in the preceding 12 months. The seroconversion rate was higher among females (0.87% [95% CI: 0.73–1.02]) compared to males (0.70% [95% CI: 0.59–0.80]); however, this association was not significant when the odds of seroconversion were adjusted for age group (aOR: 1.20 [95% CI: 0.95–1.51, p = 0.12]). Seroconversion occurred across all age groups, with the highest rate observed in 18–30 year olds (1.08% [95% CI: 0.84–1.32]), though differences between age groups were modest. The mean interdonation interval for seroconverters was 188 days.
To further investigate recency of infection, a subset of 232 seroconverting donors underwent additional testing according to the study algorithm (Figure 1). Of these, 46 (19.8%) were CMV IgM positive on the Abbott Architect and were referred for supplementary testing. Samples positive by the VIDAS CMV IgG with reactivity on the VIDAS IgM assay (n = 38) underwent testing with the VIDAS CMV-IgG Avidity assay. A low avidity index was interpreted as indicative of primary infection within the preceding 3 months, and a high avidity suggested infection more than 3 months prior. A total of 25 samples with low or borderline avidity, and two VIDAS IgM-only positive samples, underwent CMV DNA testing by PCR (n = 27). CMV DNA was detected in 15 of the 27 samples.
Overall, 6.47% (15/232) seroconverted donors were confirmed CMV DNA-positive, and a further 1.29% (3/232) had low avidity antibodies, indicating recent infection. Viral loads were generally low, with only two samples exceeding 1,000 copies/mL. The interdonation interval for CMV DNA-positive cases ranged from 92 to 353 days; eight donors had intervals of less than 120 days from their last CMV-negative donation.
Discussion
This study provides recent data on CMV epidemiology in Ireland based on a large cohort of blood donors. We report a CMV seroprevalence of 26.0% in 2020, which is among the lowest rates reported globally [Reference Fowler8, Reference Zuhair9]. Our estimate is lower than previously reported rates in other Irish subpopulations, such as pregnant women (30.4%) and solid organ donors (33.4%) [Reference Hassan11, Reference Knowles12] and also falls below seroprevalence estimates in blood donors in other high-income countries, including Canada (42.2%) [Reference Mabilangan19] and Germany (37.5%) [Reference Kowalzik20].
Consistent with patterns observed internationally, CMV seroprevalence in Ireland is age and sex dependent. The age-related increase likely reflects cumulative exposure to CMV over time, while the higher prevalence in females may relate to maternal exposure to younger children when there is a high level of viral shedding [Reference Griffiths, Baraniak and Reeves1]. These demographic associations align with previous studies in both high- and low-prevalence countries [Reference Griffiths, Baraniak and Reeves1, Reference Korndewal13, Reference Mabilangan19, Reference Lachmann21, Reference Lancini22]. In this study, 72.5% of women of childbearing age were CMV seronegative and therefore susceptible to primary infection during pregnancy. cCMV can be caused by primary infection, reactivation, or reinfection [Reference Rawlinson7]; however, in a low-prevalence setting, primary infection is the highest risk [Reference Kenneson and Cannon23]. The incidence of cCMV in Ireland is estimated at 0.23% [Reference Waters24], incidence rates reported globally vary widely, with a pooled estimate in high-income countries of 0.48%, based on a recent meta-analysis [Reference Ssentongo25].
The most significant demographic factor associated with CMV seropositivity in this study was donor type. Among first-time donors, those born outside of Ireland and the UK, termed SONDs, had significantly higher seropositivity (46.7%) compared to those born in Ireland or the UK (23.82%). This disparity reflects how the country of origin and migration influence the epidemiology of CMV [Reference Pembrey26]. CMV seroprevalence varied by geographical region of donation, with a significantly higher rate observed in Leinster compared to Munster and Connacht. This difference remained significant after adjustment for age group, sex, and region of birth, suggesting that the higher prevalence in Leinster may be attributable to other factors such as population density or socioeconomic factors.
The underlying cause of Ireland’s unusually low CMV seroprevalence remains unclear. Socio-behavioural factors such as small household size, breastfeeding practices, and hygiene factors may contribute to reduced early life exposure. In recent years, both breastfeeding rates and childcare use have increased in Ireland [Reference Pembrey26, 27], which could affect CMV epidemiology in younger cohorts as they reach adulthood. Ongoing surveillance will be important to detect such shifts. Some studies have suggested a role for host genetic factors, particularly HLA class I alleles, in CMV susceptibility. A 2016 study found that the most common inherited haplotype in the Irish population, HLA-A1, B8, was significantly associated with CMV seronegativity [Reference Hassan11]. In contrast, epidemiological studies have shown that children born in the UK to individuals from higher incidence countries have a lower risk of their adopted country [Reference Ssentongo25, 28], which may suggest a predominant role of environmental and social determinants in CMV transmission. Ireland’s low CMV seroprevalence is likely multifactorial, including interactions between social, behavioural, and genetic factors.
Among repeat donors, we estimate an annual seroconversion rate of 0.76%. Seroconversion was observed across all age groups, with the highest rate in the 18–30 age group. This supports previous evidence that primary CMV infection remains a persistent feature of CMV epidemiology [Reference Hyde, Schmid and Cannon18, Reference Kowalzik20, Reference Pembrey29, Reference Hecker30] even in a low-prevalence setting. Although our estimate may be subject to limitations, including reliance on donor return patterns, it provides evidence demonstrating that primary CMV infection occurs continuously in blood donors in all age groups.
To further investigate the recency of infection, we employed supplemental testing in a subset of seroconverted donors; among these, 6.5% had detectable CMV DNA. While most viral loads detected were low, the presence of CMV DNA in blood following seroconversion is consistent with previous reports [Reference Ziemann31]. This has important implications for transfusion safety. While leucodepletion is employed to remove white blood cells from blood products and is reported as being very effective at reducing the risk of transmitting cell-associated CMV, it does not remove cell-free virus, which may still pose a residual risk [Reference Kowalzik20, Reference Ziemann31].
The low seroprevalence in Ireland is relevant to multiple areas of public health. It suggests a high level of susceptibility to primary CMV infection in early adulthood and during reproductive years. This may support consideration of future national screening or vaccination programmes. The development of an effective CMV vaccine remains a global health priority [Reference Boppana2] and local seroprevalence data may help to identify higher-risk groups to inform vaccination strategies. These findings also reinforce the need to assess transfusion transmission risk, especially in a low-prevalence setting, where a greater proportion of the population is CMV naive.
A key limitation of this study is that donors with a single positive CMV result are permanently classified as seropositive; confirmatory testing and repeat serology are not routinely performed, which could lead to overestimation of seropositivity. Additionally, blood donors are not considered fully representative of the general population due to selection criteria that may exclude some groups [Reference O’Brien32], however they provide a readily accessible and easily monitored population, making them a valuable surrogate population for some infectious disease surveillance. Recent changes to donor deferral and screening policies in Ireland have expanded eligibility [33] and may improve the representativeness of blood donors to the general population over time. Continued monitoring of CMV epidemiology in this population may help detect shifts in transmission patterns and inform broader public health planning.
This study confirms that Ireland has one of the lowest CMV seroprevalence rates globally and that CMV seroconversion occurs consistently throughout all age groups in adults. This indicates that there is an ongoing risk of primary infection, particularly among high-risk groups. These data have important implications for transfusion safety, infectious disease surveillance, maternal healthcare, and future CMV vaccine implementation strategies. Analysis of blood donor CMV screening results is a useful epidemiological tool to assess population-level risk and possible future mitigation strategies.
Data availability statement
Aggregate data may be available upon request to research@ibts.ie and in accordance with the Irish Blood Transfusion Service data protection policy and the EU General Data Protection Regulations.
Acknowledgements
The authors acknowledge and thank our blood donors, whose continued contributions ensure the availability of blood for patients in Ireland.
Author contribution
Data curation: D.B.; Conceptualization: N.O., P.W., A.W.; Methodology: D.C., P.H., C.d.G.
Competing interests
The authors have no conflicts of interest to declare.