Introduction
Hematopoietic stem cell transplantation (HSCT) or intensive chemotherapy for the treatment of malignant disease is a highly distressing, potentially traumatic, and life-threatening experience – not only physically but also psychologically – that can have dramatic impacts on a patient’s individual concept of life. This especially applies to pediatric, adolescent, or young adult patients who, instead of following their natural maturation and growing experiences and developing their individual characteristics and life goals, have to face severe sickness and an acute and persistent threat to life. This not only affects their mental health but also their personal relationships, the identity of their family, and their family members, all of whom experience severe distress (Guilcher Reference Guilcher2016; Linder and Hooke Reference Linder and Hooke2019; Packman et al. Reference Packman, Weber and Wallace2010; West et al. Reference West, Dusome and Winsor2020). Pediatric alloHSCT patients are at high risk for anxiety (16–37% of patients) and depression (7–25% higher than age-matched population) (Di Giuseppe et al. Reference Di Giuseppe, Thacker and Schechter2020) and are likely to develop symptoms of post-traumatic stress (Kronenberger et al. Reference Kronenberger, Carter and Stewart1996; Stuber et al. Reference Stuber, Nader and Yasuda1991). The psychological effects of oncological diseases and their treatment can occur before, during, or a long time after the situation and, depending on the underlying disease, its prognosis, or the patient’s age and gender, may significantly impair the patients’ overall mental health and quality of life (Bergerot et al. Reference Bergerot, Clark and Nonino2015; Chan et al. Reference Chan, Ng and Taib2018; Linden et al. Reference Linden, Vodermaier and Mackenzie2012; Luo et al. Reference Luo, Yang and Yang2019; Rodin et al. Reference Rodin, Lo and Mikulincer2009). Especially, the first year after HSCT in children and adolescents presents significant challenges for the survivors, including difficulties with social competence, self-esteem, and emotional well-being (Luo et al. Reference Luo, Yang and Yang2019; Phipps et al. Reference Phipps, Brenner and Heslop1995).
The affected person’s resilience, which can be defined as their ability to use personal resources to maintain their psychological and physical stability and ability to function while being exposed to major stress or threat (Sisto et al. Reference Sisto, Vicinanza and Campanozzi2019), is crucial to coping with such a challenging journey. Previous research has shown that personality-related variables such as optimism, hope, self-esteem, positive emotions, and personal control are important factors to build resilience, increase the quality of life, and reduce distress in patients with cancer. Resilience interventions that include stress management, redirection of perceptions, focus on growth and adjustment, or meaning-making can reduce distress, increase the quality of life, and improve optimism and self-efficacy in pediatric, adolescent, and adult cancer patients (Chung et al. Reference Chung, Li and Cheung2021; Greup et al. Reference Greup, Kaal and Jansen2018; Kazak et al. Reference Kazak, Cant and Jensen2003; Molina et al. Reference Molina, Yi and Martinez-Gutierrez2014; Rosenberg et al. Reference Rosenberg, Bradford and McCauley2018; Seiler and Jenewein Reference Seiler and Jenewein2019). The German national S3 Guideline “Psychosocial care in childhood and adolescent oncology” recommends basic psychosocial care in pediatric hemato-oncology patients that can be complemented with intensified psychosocial care if indicated. (Leiss et al. Reference Leiss, Griessmeier and Minetzke-Gruner2012; Schröder et al. Reference Schröder, Lilienthal and Schreiber-Gollwitzer2019).
The hypothalamus–pituitary–thyroid axis and the hypothalamus–pituitary–adrenal (HPA) axis are important neuroendocrine axes that are key regulators of the endocrine, neurological, neuroendocrine, psychological, and immune systems (Ortiga-Carvalho et al. Reference Ortiga-Carvalho, Chiamolera and Pazos-Moura2016). These axes can be significantly altered due to stress disorders, cancer, or inflammation (De Luca et al. Reference De Luca, Davis and Lin2021; Mazzoccoli et al. Reference Mazzoccoli, Pazienza and Piepoli2012; Olff et al. Reference Olff, Güzelcan and de Vries2006; Wong et al. Reference Wong, Dong and Maestre-Mesa2008). Psychoneuroimmunology research has clearly shown that the stress-dependent constitutive activation of the HPA axis in cancer patients is likely to undermine therapy success. In particular, several mechanisms of the impact of psychological distress and upregulated glucocorticoid secretion in cancer patients have been proposed, potentially subverting therapeutic tumor control via blocking of type I interferon responses in dendritic cells and interferon gamma (IFNγ) T cell activation (Ma et al. Reference Ma, Yang and Kroemer2020; Yang et al. Reference Yang, Xia and Chen2019).
In this prospective randomized controlled single-center study, a total of 30 adolescent and young adult patients aged between 12 and 22 years who underwent allogeneic HSCT or high-dose chemotherapy for the treatment of a malignant or nonmalignant oncologic or hematologic disease, received either the standard psychosocial care during and after their treatment (control group [CG]) or additionally underwent a novel and specifically developed mental training program consisting of 14 one-hour training sessions that include elements of sports psychology, resource activation, motivational training, and resilience enhancement (therapy group [TG]). The study was created following the CONSORT 2010 guidelines for randomized controlled trials (Schulz et al. Reference Schulz, Altman and Moher2010). The major objectives of this study were to develop and evaluate an effective psycho-oncological training program that complements the standard psychosocial care and that focuses on the major strategies, influencing anxiety, especially reducing recurrence anxiety, increasing motivation, clearing goal orientation by stopping negative thoughts, increasing self-confidence, and making one’s own decisions, to prepare the patients and facilitate their transformation from a sick and fighting patient to a survivor who is strong and motivated to create their own, self-determined future.
We hypothesize that this training will therefore lessen anxiety and negative affect, as well as increase positive affect to a significantly greater extent than the standard care protocol in the CG. As a secondary hypothesis, we predict that through the training, heightened achievement motivation, goal orientation, as well as volition will be induced in participants, while there will be no enhancement of these traits following standard protocol in the CG. As an exploratory analysis, we will moreover analyze if these psychological changes further translate into biological changes in thyroid and immune function parameters. Due to the small sample size, no additional exploratory subgroup analyses will be computed.
Material and methods
Study design and patients
In this prospective randomized controlled intervention study (CentraXX study registration number UKT-2018-KI3-2685), adolescent and young adult patients of the University Children’s Hospital Tübingen between the ages of 12 and 22 who underwent an allogeneic HSCT or intensive chemotherapy between 2018 and 2020 were consecutively offered study participation and were subsequently recruited at the time of inpatient admission before the commencement of HSCT/chemotherapy. The exclusion criteria were psychoactive substance abuse and diagnosis of a mental disorder according to the International Classification of Disease-11 (Khoury et al. Reference Khoury, Kogan, Daouk, Zeigler-Hill and Shackelford2017). In total, 30 patients were recruited and randomized to the CG or the TG. The urn randomization was done by hand via random draw from a physical urn without replacement. Due to the limited capacity for training sessions in the TG, participants were recruited successively over the study period so that a maximum of 6 to 7 patients were simultaneously in the training phase. Specifically, this means that initially suitable subjects were invited to participate in the study consecutively at the beginning of the study period and, after consent, were randomized via urn randomization to the study groups until capacity in the TG was reached. Recruitment continued as soon as at least 3 places in the therapy group could be filled. Patients of the CG received the standard psychosocial care and patients of the TG additionally received a mental training program (see below). The observation period started on the day of inpatient admission and ended 8 months after study enrollment. The check-up days on which the patients were screened for laboratory markers and answered questionnaires were the day of study enrollment before starting chemotherapy (baseline/at study enrollment), 3 months (after session 6), 6 months (after session 14), and 8 months (6–8 weeks after session 14) after enrollment. All 30 patients completed all assessments, and every patient from the TG completed the full 14-session program. The study was carried out as an open-label study.
Psycho-oncological care and novel mental training
The patients of both the TG and the CG were offered the standard psycho-oncological care that includes social or psychological counseling with social workers, systemic therapists, or other professionals (e.g., art therapists). In addition, the patients of the TG received a novel and specifically developed mental training program consisting of 14 one-hour training sessions that include elements of sports psychology, resource activation, motivational training, and resilience enhancement. The training program focuses on the major strategies: influencing anxiety, especially reducing recurrence anxiety, increasing motivation, a clear goal orientation by stopping negative thoughts, increasing self-confidence, and making one’s own decisions. In the first session, medical history and personal goals of the patients are assessed. In the following sessions, patients learn several techniques to strengthen their concentration, attentiveness, stress management, and emotional control and regulation. Negative and anxious thoughts are analyzed and reframed, and personal abilities and strengths are explored. Then, the comprehensive concept of mental strength is introduced and linked to previously learned techniques using a currently existing problem of the patients, for which an individual mental training cycle is established. The last sessions focus on motivation for further development and reflection on the progress the patients made during the program. The description of the concept with a further summary of each session is provided in Supplementary data 1.
Applied questionnaires
On the observation days at baseline (at study enrollment), 3 months (after session 6), 6 months (after session 14), and 8 months (6–8 weeks after session 14), the patients of both cohorts were screened for generalized anxiety (generalized anxiety questionnaire [GAD-7]) (Spitzer et al. Reference Spitzer, Kroenke and Williams2006), affectivity (Positive and Negative Affect Schedule [PANAS]) (Krohne et al. Reference Krohne, Egloff and Kohlmann1996), sports orientation (Sports Orientation Questionnaire [SOQ]) (Elbe et al. Reference Elbe, Wenhold and Beckmann2008), and volition (volitional components in sports [VKS]) (Wenhold et al. Reference Wenhold, Elbe and Beckmann2008) using standardized questionnaires. The data sets were analyzed between the observation days (baseline, 3 months, 6 months, and 8 months) within the study groups.
The GAD-7 is a screening tool to detect generalized anxiety disorders (Löwe et al. Reference Löwe, Decker and Müller2008; Spitzer et al. Reference Spitzer, Kroenke and Williams2006). Since anxiety may represent a normal reaction to a malignant disease, we chose a questionnaire that has been used in this context to assess pathological levels of anxiety in adult cancer patients (Esser et al. Reference Esser, Hartung and Friedrich2018). The recommended cutoff score for cancer patients is ≥7 (Esser et al. Reference Esser, Hartung and Friedrich2018).
The German version (Krohne et al. Reference Krohne, Egloff and Kohlmann1996) of the PANAS (Watson et al. Reference Watson, Clark and Tellegen1988) was applied to assess the positive and negative affect of the patients. The German PANAS is a 20-item questionnaire. Higher scores represent higher affect.
The SOQ was developed as an instrument designed to measure individual achievement motivation in sport situations and was validated for high school and university students. For this study, a validated German version was applied (Elbe et al. Reference Elbe, Wenhold and Beckmann2008). For young athletes (teens to adolescents; performance level 1 [participation in national or international competitions]), standard scores (ranges) for mean competitiveness, win orientation, and goal orientation were defined as 54 (range 48–59), 24 (range 20–27), and 26 (range 24–30), respectively.
The questionnaire for the assessment of VKS was designed to assess skills and deficits in self-direction or volition specifically for sport (Wenhold et al. Reference Wenhold, Elbe and Beckmann2008).
Laboratory analyses
Blood levels of cortisol and thyroid function parameters thyroid-stimulating hormone (TSH), free triiodothyronine (fT3), and free thyroxine (fT4) of the immune cells CD3+ T cells, CD4+ T cells, CD8+ T cells, and the CD4+/CD8+ ratio were assessed starting at baseline as well as 3 and 6 months thereafter. Blood collection was performed between 7.00 and 9.00 a.m. Cortisol measurements were not included during corticosteroid treatment. Likewise, thyroid function parameters (TSH, fT3, and fT4) were not included in the analyses during thyroxine treatment.
Statistical analysis
Questionnaire results were calculated in accordance with the individual analysis manuals and then averaged across participants for each time and group combination. Laboratory measurements were determined as mean (M) and standard deviation (SD) of the results of all patients of the respective observation day and study cohort. There was neither missing data from the laboratory analyses nor from the questionnaires. Due to frequent non-normality of data sets as tested by the Shapiro–Wilk normality test, as well as a generally small sample size, robust, nonparametric statistical tests were applied (Feys Reference Feys2016; Mair and Wilcox Reference Mair and Wilcox2020). Distributions of patient characteristics were compared between both groups using a Fisher’s exact test. To assess the primary psychological outcomes, as well as the secondary explorative laboratory analyses, robust mixed 2 × 4 analysis of variances (ANOVAs) with between-subject factor group (control and therapy) and within-subject factor time (baseline, 3, 6, and 8 months after HSCT) as independent variables were conducted using the WRS2 R Package. The robust mixed ANOVA uses the 20% trimmed means and returns the test statistic Q, which is approximately F-distributed; however, it does not return an effect size. Significant interactions were therefore followed up by carrying out simple effects analyses with robust t-tests, which compute a robust explanatory measure of effect size ξ (Wilcox and Tian Reference Wilcox and Tian2011). 0.10, 0.30, and 0.50 correspond to small, medium, and large effect sizes, respectively.
Graphs and statistical tests were created with GraphPad Prism version 8.4.3 (686) for Windows (GraphPad Software. Inc., La Jolla, CA, USA) and R version 4.1.1 (2021-08-10, Copyright 2021, The R Foundation for Statistical Computing). All statistical tests were 2-sided. p-Values <0.05 (*), <0.01 (**), and <0.001 (***) were defined as statistically significant.
Results
Patient characteristics and observation period
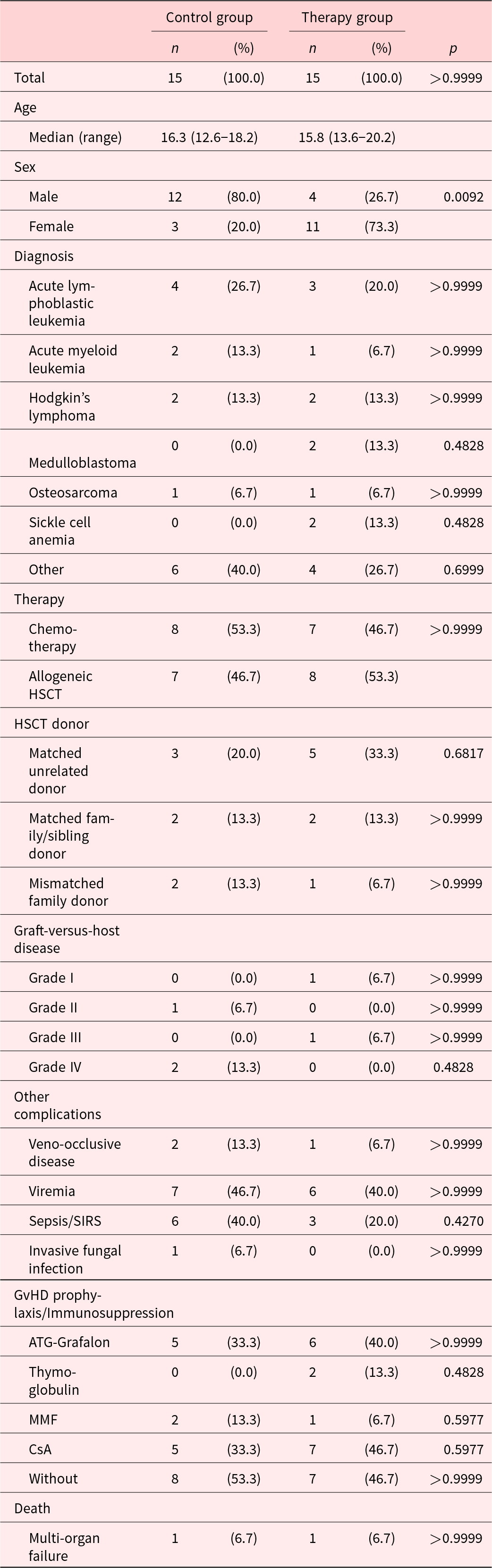
A total of 30 pediatric patients with a median age of 16.1 years (range 12.6–22.2 years) were enrolled in this analysis. The TG comprised a significantly higher percentage of female patients when compared to the CG (11 vs. 3 patients; p = 0.0092). Since the participants were randomized, this is considered to be coincidental. No other patient characteristic differed significantly between groups. In each group, one patient died of multi-organ failure (n = 2; 7%) after study completion. None of the patients were lost to follow-up. Detailed patient characteristics are summarized in Table 1.
Patient characteristics

CsA = cyclosporine A, HSCT = hematopoietic stem cell transplantation, MMF = mycophenolate mofetil, n = sample size, p = probability value.
Primary and secondary analyses of psychological measures
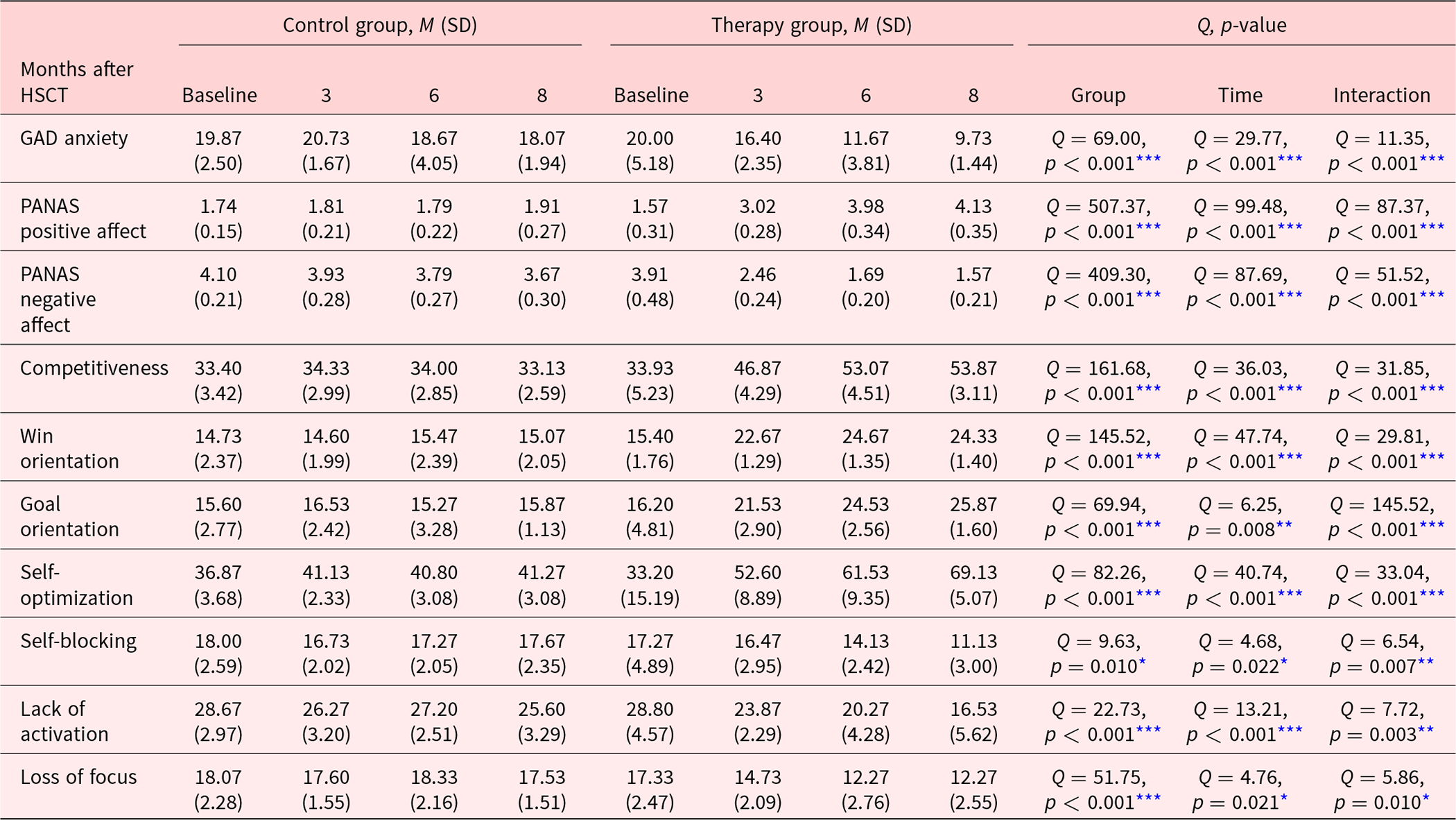
Means and standard deviations of the psychological measures for the respective observation day and study cohort, as well as the 2 main and the interaction effects examined by the robust 2-way mixed ANOVAs are summarized in Table 2. For the primary outcomes anxiety, positive affect and negative affect, the robust ANOVA revealed significant main effects for group, time, and importantly significant interaction effects (each p < 0.001), signifying that the change in the primary psychological measures differed between groups.
Questionnaire analyses

Means and standard deviations in the control and TG for psychological questionnaire measurements at baseline and 3, 6, and 8 months after hematopoietic stem cell transplantation (HSCT). The last 3 columns show the results of the statistical analysis, using a robust 2-way mixed ANOVA design with group and time/months after HSCT as factors.
* p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
To assess the interaction further, post hoc robust t-tests between groups were performed. For self-reported anxiety, the robust t-test showed no significant difference between groups at baseline measurement, t(11.05) = 0.58, p = 0.576, while there was a significant difference at 3, t(13.3) = 5.62, p < 0.001, ξ = 0.86; 6, t(13.26) = 7.88, p < 0.001, ξ = 0.91; and 8, t(16) = 9.72, p < 0.001, ξ = 0.94, months after HSCT, with the TG reporting significantly less anxiety than the CG. For positive affect, there was a significant difference at baseline level, with the CG reporting significantly more positive affect than the TG, t(8.94) = 2.35, p = 0.044, ξ = 0.61. For months 3, t(10.65) = 11.01, p < 0.001, ξ = 0.98; 6, t(15) = 23.49, p < 0.001, ξ = 0.96; and 8, t(14.1) = 16.16, p < 0.001, ξ = 0.93, the difference between groups was again significant; however, the direction of the effect changed. For the months after the therapy program was started and then completed, participants in the TG reported significantly more positive affect than CG participants. For negative affect, no significant difference between groups was found at baseline, t(13.17) = 0.79, p = 0.442. However, at 3, t(14.12) = 15.43, p < 0.001, ξ = 0.97; 6, t(15.98) = 25.84, p < 0.001, ξ = 0.95; and 8, t(12.07) = 17.21, p < 0.001, ξ = 0.94, months after HSCT, the robust t-test revealed that the CG reported significantly more negative affect than the TG.
For the secondary outcomes sports orientation and volition, the subscale competitiveness, win orientation, goal orientation, self-optimization, self-blocking, lack of activation, and loss of focus were assessed. Again, the robust 2-way mixed ANOVAs showed significant main effects of time and group for all subscales, as well as significant interaction effects (competitiveness, win orientation, goal orientation, and self-optimization with p < 0.001 self-blocking and lack of activation with p < 0.010 and loss of focus with p = 0.010). To specify the interaction, post hoc robust t-tests between groups for every level of the time factor were performed. For competitiveness, win orientation, goal orientation, self-optimization, and loss of focus, the robust t-test at baseline showed no significant difference, while the t-tests performed for month 3, 6, and 8 showed significant differences between groups (all p < 0.001, except loss of focus on month 3 comparison at p = 0.005, ξ-Range: 0.86–1). For each subscale, TG participants reported on average significantly higher scores after starting and completing the program than CG participants receiving standard care. For the subscales, self-blocking and lack of activation on both the baseline and the 3-month comparison between groups were nonsignificant. At months 6 and 8, a significant difference between groups was shown (all p < 0.010, ξ-Range: 0.79–0.94). Therapy group participants reported significantly lower self-blocking tendencies and lack of activation than control participants.
Exploratory laboratory analyses
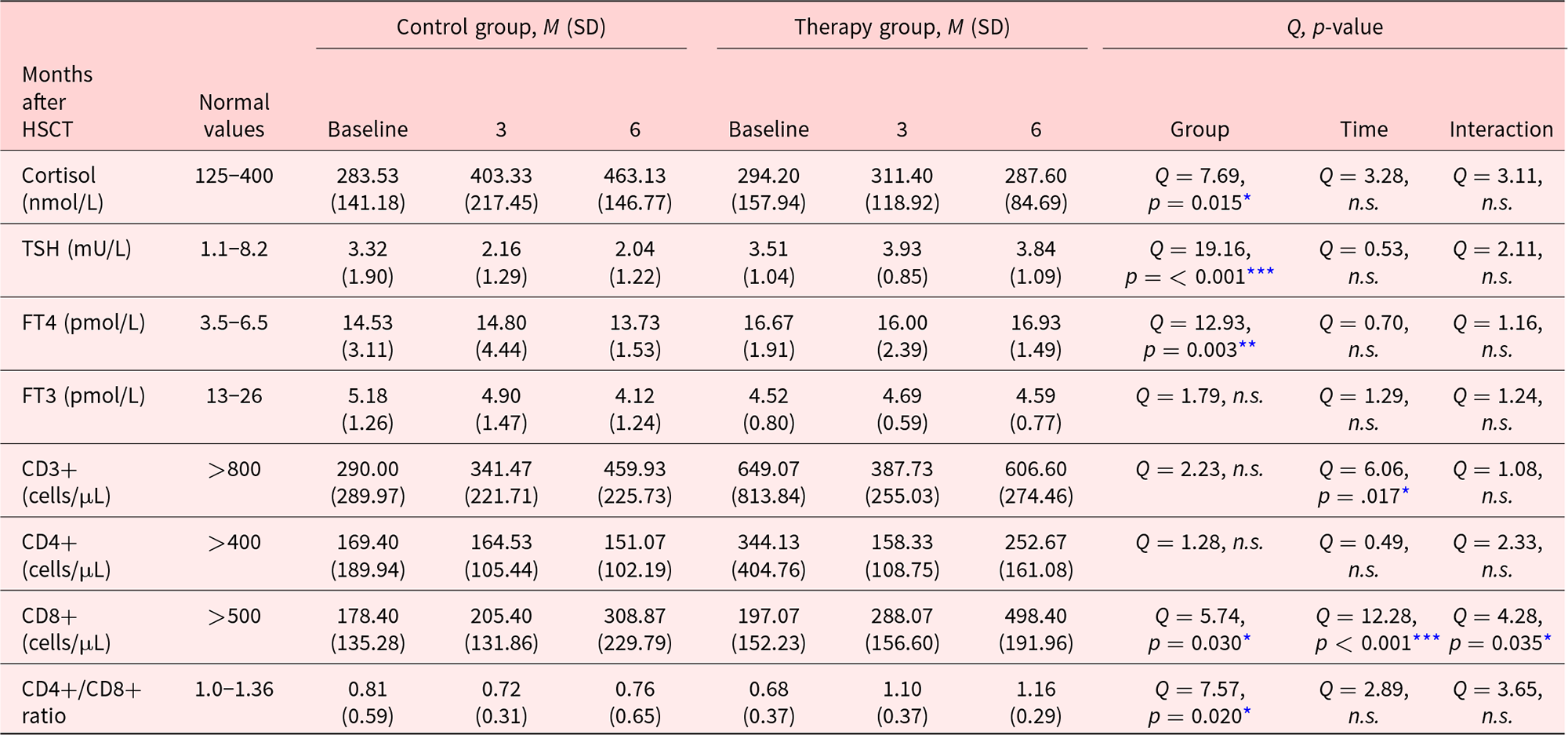
Normal values, means, and standard deviations of the laboratory measures for the respective observation day and study cohort, as well as the 2 main and the interaction effect examined by a robust 2-way mixed ANOVAs, are summarized in Table 3. We found significant main effects of group for TSH (p < 0.001), FT4 (p < 0.01), cortisol, CD8+ T cells, and the CD4+/CD8+ ratio (p < 0.05). A significant main effect of time was found in the analysis of CD3+ T cells (p < 0.05), and CD8+ T cells (p < 0.001). CD8+ T cells furthermore showed a significant interaction effect (p < 0.05). No other interaction effect was significant. Therefore, for cortisol, CD8+ cells, and the CD4+/CD8+ ratio, it can be said that there was a significant difference between groups, independent from the time of measurement. The significant main effects of time for CD3+ cells, on the other hand, signify that there is a difference between the different times of measurement, independent from group affiliation. For CD8+ T cells, the significant interaction shows that the change in cell concentration differed between groups. To explore this interaction further, post hoc robust t-tests were performed. Neither the difference in CD8+ T cell concentration at baseline, t(15.26) = 0.58, p = 0.867, nor at month 3, t(15.92) = 1.13, p = 0.276, was significant. However, at month 6, the TG had significantly more CD8+ T cells than the CG, t(15.91) = 3.74, p = 0.002, ξ = 0.74.
Laboratory analyses

n.s. = not significant. Normal values, means, and standard deviations in the control and TG for laboratory measurements at baseline and 3 and 6 months after hematopoietic stem cell transplantation (HSCT). The last 3 columns show the results of the statistical analysis, using a robust 2-way mixed ANOVA design with group and time/months after HSCT as factors.
* p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
Discussion
The incidence of anxiety and post-traumatic stress disorder is significantly and relevantly higher in survivors of pediatric alloHSCT than in the normal population (Sanders et al. Reference Sanders, Hoffmeister and Storer2010; Taskıran et al. Reference Taskıran, Sürer Adanır and Özatalay2016). During the acute and intensive treatment of their life-threatening disease, pediatric HSCT patients are requested to focus on the moment and to invest their whole energy and mind in every single day, one day at a time. In our own clinical experience, we noticed that as soon as the acute existential threat declines, that is, when the success of a disease treatment becomes increasingly evident, and the patients face the inpatient discharge after intensive chemotherapy or HSCT, they begin to focus on the journey that lies ahead of them. During this time, the patients, especially those aged 12 and older, frequently ask about how to find their way back to their past lives and how to create a future that they want to live in. This process is often accompanied by great anxiety and self-blockage. Based on this experience, we developed our mental training. The present study is the first study to investigate a psycho-oncological intervention program based on sport psychology elements and adapted to pediatric and young adult HSCT patients, focusing on methods to reduce anxiety, increase motivation and goal orientation, and to implement this in the form of mental training in 15 pediatric patients. The major objective of this intervention study was to show that our mental training over 14 sessions alleviates anxiety and its negative effects and promotes positive developments, compared to standard care. This could be confirmed. Thus, it was shown that the mental training led to decreased generalized anxiety and negative affect reported by patients significantly more than standard care. Furthermore, the increase of positive affect was significantly greater in the TG than in the CG.
There are only few comparable studies examining psycho-oncology intervention programs in children, adolescents, or young adults in the setting of HSCT. In a multicenter study of 171 patients and their parents, patients and parents in the TG received an intervention program with massage and humor therapy compared with the CG with standard care. The intervention began at hospital admission and lasted until week 3 after HSCT. Participants completed outcome measures at admission and 24 weeks after HSCT. Across the sample, significant improvements were seen in all outcomes from admission to week 24. Benefits were high at admission and increased at week +24. However, there were no statistically significant differences between intervention arms for any of the measures (Phipps et al. Reference Phipps, Peasant and Barrera2012). Another phase 2 randomized controlled trial assigned English-speaking adolescent and young adult patients (AYA; 12–25 years old) with cancer to the Promoting Resilience in Stress Management intervention or to usual care. The study suggests that brief, skills-based interventions could provide benefit for this patient population (Rosenberg et al. Reference Rosenberg, Bradford and McCauley2018).
In a study of 66 children and adolescents between 8 and 16 years of age with depressive or anxiety disorders, it was shown that patients who already had high positive and low negative affect prior to 8 weeks of cognitive behavioral therapy (with and without the use of serotonin reuptake inhibitors) responded significantly better to therapy (lower expression of symptoms of depression and anxiety) (Forbes et al. Reference Forbes, Stepp and Dahl2012). It has been demonstrated in 50 adult patients with generalized anxiety disorder that positive affect contributes relevantly to quality of life in these patients, and strategies to improve positive affect are a useful starting point in improving the quality of life in these patients (Das et al. Reference Das, Clerkin and Tolin2020). Positive affect was further described as a buffer against chronic stress by ameliorating symptoms of emotional disorders (Sewart et al. Reference Sewart, Zbozinek and Hammen2019). In this sense, the patients of the TG might have benefited both in terms of a reduction of anxiety and increased affect functioning, thereby potentially facilitating their overall treatment response to therapeutic interventions and alleviating stress levels. Research has demonstrated that the individual mental resources and resilience of cancer patients greatly influence their psychological health, post-traumatic growth (i.e., a positive change of life that results from major health or life crises), and quality of life during and after therapy (Rosenberg et al. Reference Rosenberg, Bradford and McCauley2018). In a systematic review, Seiler and Jenewein described personality-related variables such as optimism, hope, self-esteem, positive emotions, and personal control as important factors to build resilience and report an increased quality of life and less distress in patients with effective coping strategies (Seiler and Jenewein Reference Seiler and Jenewein2019). Symptoms of post-traumatic stress and anxiety decreased in patients and their families with the help of family group interventions that combines cognitive behavioral and family therapy approaches (Kazak et al. Reference Kazak, Alderfer and Streisand2004, Reference Kazak, Simms and Barakat1999).
The hypothesis that the training induced an increased goal orientation and achievement motivation as well as an increased willpower in the participants, while there was no improvement in these characteristics in the CG according to the standard protocol, was confirmed. Analyses of the sports orientation and volitional components showed a significantly greater improvement of competitiveness, win orientation, goal orientation, and self-optimization scores in the TG than in the CG, as well as a significantly greater reduction in self-blocking and a significantly more reduced loss of focus and lack of activation than in patients receiving standard care. The role of goal orientation and competitiveness in achieving academic goals has been thoroughly investigated. Several studies indicated that goal setting in adults is associated with increased use of health activities that promote rehabilitation and recovery (Hurn et al. Reference Hurn, Kneebone and Cropley2006). The future orientation of adolescents is proposed to have a close relationship to their development outcomes and their individual physical and mental health (Johnson et al. Reference Johnson, Blum and Cheng2014). The determinations of the stress biomarker were only punctual measurements of a dynamic endocrine system that follows circadian variations; however, it is notable that the cortisol levels of the CG patients were significantly higher (p = 0.0030) at 6 months when compared to the baseline levels. Although a more dynamic development of thyroid function parameters was seen in the CG, monitoring for effects of the mental training on inflammatory signals and thyroid function should be a component in larger clinical trials, further investigating these observations. In our experiment, we observed significant main effects of group for cortisol, TSH, and FT4, signifying the levels of these measurements are significantly different between groups. However, there was neither a significant main effect of time nor a significant interaction effect, meaning the measurements remained relatively stable across time.
The CD4+/CD8+ ratio is decisive for a normal immune function (Robinson et al. Reference Robinson, O’Donohoe and Dadian1996). The CD8+ T cell levels increased significantly more in patients of the TG in contrast to patients of the CG. This effect might be based on several influencing factors such as the chemotherapy or immunosuppressive regime. In view of the diversity of the applied medication, we consider it likely that these were the major influencing effects on the T cell regeneration. These observations should be analyzed in larger cohorts.
The limitations of this analysis are the small cohort size and the unequal gender distribution, potentially biasing an inter-group comparison. Nevertheless, the results of this study indicate the importance and efficacy of a supportive and motivation-enhancing mental training. Taken together, the presented data indicate that the adolescent and young adult patients who underwent our mental training displayed less anxiety, better affect, and improved mental performance with less self-blocking. This resulted in improved goal orientation, competitiveness, self-optimization, and focus when compared to the CG patients. Moreover, the data suggest a favorable development of immune and stress function parameters in patients undergoing our mental training program. However, these effects and their interdependent relationships need to be further investigated to facilitate valid conclusions. Additional research questions to explore in this regard could be extensive subgroup analyses such as gender or age subgroups in bigger cohort studies. In this study, patients from the age of 12 to 22 were assessed. As this age range is rather large and spans from the beginning of puberty to early adulthood, it would be beneficial to examine whether certain age groups may benefit more from such a mental training. Similarly, gender is an important moderator for therapy success to explore. Furthermore, this study showed that the improvement of several psychological measurements remained stable at least up until 2 months after completion of the mental training. A long-term experiment could explore further how long the effects of this training last. Lastly, as the whole family is impacted by pediatric cancer, it could be beneficial to the family system to include siblings and parents in therapeutic interventions. A future study could examine whether this mental training program could be extended to contain sessions for other family members and whether this would further strengthen the perceived positive effects.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S1478951523000986.
Authors’ contributions
All authors made substantial contributions to the conception or design of the work or the acquisition, analysis, or interpretation of data. All authors drafted the work or revised it critically for important intellectual content. MD created the mental training program. MD, KMCS, and JX collected and analyzed the data and wrote the manuscript. DW, MS, ST, VS, and SM analyzed the data. MD and KMCS were head of the study and responsible for the study design. DW and JS contributed to the study conception and design. DW, SM, MS, JS, VS, and PL reviewed the manuscript. All authors read and approved the final manuscript and agreed to be accountable for all aspects of the work.
Funding
This work was supported by the Förderverein für krebskranke Kinder Tübingen e.V., Tübingen, Germany.
Competing interests
The authors declare that they have no conflict of interest.
Ethical approval
This analysis was performed in accordance with the Helsinki declaration adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964, and amended by the 64th WMA General Assembly, Fortaleza, Brazil, October 2013. The legal basis for the data processing is Art. 6, 7, 9, 89 of the general data protection regulation (EU) 2016/679 of the EU in combination with §§ 4, 5, 6, 8, 9, 12, 13 of the Landesdatenschutzgesetz Baden-Württemberg in its current form of May 25, 2018. Formal written consent for participation and publication of the data was obtained from the patients and/or their legal representatives. Approval for this analysis was granted by the Ethics Committee of the University Hospital Tübingen and the Eberhard Karls University of Tübingen (ID# 317/2018BO1).