Key messages
Chapter 3.10 evaluates innovative financing for neglected diseases. Neglected diseases account for about a fifth of the global burden of disease and affect over 1 billion people. They are neglected because the pharmaceutical sector does not consider it profitable to develop treatments for them. This reflects the fact that neglected diseases are most prevalent in low- and middle-income countries (LMICs) with relatively low purchasing potential. Key learning includes that:
Global pharmaceutical R&D invests a disproportionate share of innovation and activity in diseases that affect high income countries, something which fosters significant inequities.
A range of push and pull incentive mechanisms have been developed to delink the cost of research from market profitability and promote innovation in areas of need. These include measures to:
○ reduce the upfront costs by subsidizing R&D pre-discovery (push incentives); and
○ offer a reward post-discovery (pull incentives).
The evidence on the effectiveness and reach of incentive schemes is scant and more needs to be done to understand the relative cost–effectiveness of the different incentive mechanisms and the extent to which they mitigate inequalities in innovation and access to new medicines.
A global, unified governance framework for needs assessment and resource allocation could usefully:
○ carry out systematic comparisons of the relative needs associated with neglected diseases globally;
○ assess the costs and benefits of addressing these;
○ set priorities for the coordinated global allocation of funding and targeted incentive mechanisms; and
○ consider payment mechanisms that will translate research into market launches.
Introduction
Neglected diseases (NDs) comprise a diverse set of around 20 diseases and disease groups, depending on the classification used. Typically considered to offer little scope for profitability to the pharmaceutical industry, these diseases are known as neglected diseases either because their prevalence is proportionally small (rare diseases) or because they are endemic in LMICs that cannot afford to buy either treatment or prevention therapies at market prices. As a result, market incentives for R&D to develop therapies for these conditions are weak (Maurer, 2005).
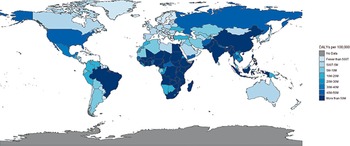
Neglected diseases include several parasitic, viral and bacterial diseases that cause substantial illness globally (Table 3.10.1). Neglected disease products were only 4% of all therapeutic products registered between 2000 and 2011 (Belen et al., Reference Belen2013). Despite these diseases affecting more than 1 billion people, causing around 19% of global burden of disease, with devastating health, social and economic consequences (Fitzpatrick et al., Reference Fitzpatrick and Holmes2017; IHME, 2019), there is substantial inertia in the development of treatments and prevention targeting these diseases. Fig. 3.10.1 illustrates the wide geographical distribution of disability-adjusted life years (DALYs) from neglected diseases between 1990 and 2010 for both sexes and all ages.
Disease categories
1. HIV/AIDS
2. TB
3. Malaria
4. Diarrhoeal disease (rotavirus, cholera, shigella, cryptosporidiosis, enterotoxigenic Escherichia coli, enteroaggregative E. coli and giardiasis)
5. Kinetoplastid disease (sleeping sickness or HAT, Leishmaniasis, Chagas disease (American trypanosomiasis))
6. Bacterial pneumonia and meningitis (Streptococcus pneumoniae, Neisseria meningitidis)
7. Salmonella infections (typhoid and paratyphoid, non-typhoidal Salmonella enterica)
8. Helminths infections (schistosomiasis, onchocerciasis, lymphatic filariasis, tapeworm, hookworm, whipworm, roundworm, strongloidiasis)
9. Dengue
10. Hepatitis B
11. Hepatitis C
12. Leprosy
13. Cryptococcal meningitis
14. Snakebite envenoming
15. Buruli ulcer
16. Trachoma
17. Leptospirosis
18. Rheumatic fever
19. Mycetoma
20. Other diseases (rare diseases, chikungunya, dracunculiasis, rabies, scabies, yaws)
Neglected diseases disproportionately affect LMICs
DALYs: disability-adjusted life-years.

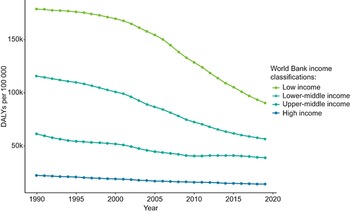
While over time there has been a decline in the global burden of disease associated with neglected diseases, they still disproportionately affect LMICs (Fig. 3.10.2).
Neglected diseases have declined in richer countries but remain constant in LMICs
B: billion; DALYs: disability-adjusted life-years; M: million.

Fig. 3.10.2 Long description
The y-axis represents the D A L Ys per 100,000 from 0 to 150k, while the x-axis represents the Year from 1990 to 2020. There are 4 lines representing the World Bank income classifications, as follows. Low-income: from (1990, 175k) to (2020, 100k). Lower-middle income: from (1990, 115k) to (2020, 65k). Upper-middle income: from (1990, 60k) to (2020, 50k). High income: (1990, 15k) to (2020, 10k).
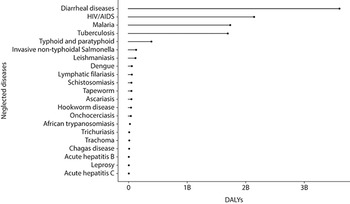
Diarrhoeal diseases, HIV/AIDS, malaria, tuberculosis (TB) and typhoid and paratyphoid are the top five diseases that contribute the highest burden of DALYs (Fig. 3.10.3). The bottom five diseases that contribute the lowest burden of DALYs are trachoma, Chagas disease, acute hepatitis B, leprosy and acute hepatitis C (Fig. 3.10.3).
Top five neglected diseases: diarrhoeal diseases, HIV/AIDS, malaria, TB, and typhoid and paratyphoid
B: billion; DALYs: disability-adjusted life-years.

Fig. 3.10.3 Long description
The x-axis represents D A L Ys from 0 to 3 billion, while the y-axis represents the diseases. Between 0 and 1 billion D A L Ys: Typhoid and paratyphoid, Invasive non-typhoidal, Salmonella, Leishmaniasis, Dengue, Lymphatic filariasis, Schistosomiasis, Tapeworm, Ascariasis, Hookworm disease, Onchocerciasis, African trypanosomiasis, Trichuriasis, Trachoma, Chagas disease, Acute hepatitis B, Leprosy, and Acute hepatitis C. Between 1 and 2 billion: Malaria and Tuberculosis. Between 2 and 3 billion: H I V/A I Ds. Above 3 billion: Diarrheal diseases.
World Health Organization’s (WHO’s) neglected diseases roadmap, 2021–2030
The WHO Road Map for Neglected Tropical Diseases 2021–2030 sets out global targets and milestones for 2030, including 90% reduction in cases requiring interventions, 75% reduction in DALYs lost to neglected diseases, working to eliminate at least one neglected disease in 100 countries, and eradicating two neglected diseases globally. The roadmap highlights research, development and innovation as “fundamental enablers of programmatic progress for all neglected diseases” (WHO, 2021a). To achieve this endeavour, the WHO Council highlights the need for a systematic approach to innovation and the elimination of unacceptable inequities in access to treatment and prevention (WHO, 2021b).
Delinking the costs of R&D from the cost of new therapies: financing incentive mechanisms
The eradication of neglected diseases or the mitigation of their impacts requires greater pharmaceutical innovation. The report on financing and coordination by WHO’s consultative expert working group on R&D (WHO, 2012) highlights that novel financing incentive mechanisms are required to address these challenges. The World Health Assembly passed a resolution in 2006 that highlighted the importance of “incentive mechanisms … addressing the linkage between the cost of R&D and the price of medicines, vaccines, diagnostic kits and other health care products” in addressing unmet needs for neglected diseases (WHO, 2007). The design of financing incentive mechanisms is important to ensure not only sustained investment in R&D but also an efficient targeting of effort and resources towards key areas of need.
In this chapter, we quantify some of the systemic gaps with regards to the distribution of pharmaceutical R&D activity and funding across neglected diseases, and describe some of the underlying reasons for these gaps. We then discuss the financial incentive mechanisms in use since 2007 in terms of pre-discovery push (public–private partnerships, in particular product development partnerships, R&D grants and subsidies, fiscal measures) and post-discovery pull (transferable patent extensions, patent buyouts, priority review vouchers (PRVs), advance market commitments (AMCs), prizes). We examine the evidence for their effectiveness levels, noting their relative advantages and disadvantages. We conclude with insights into the overall state of R&D funding for neglected diseases.Footnote 1
Trends in funding, 2007–2019
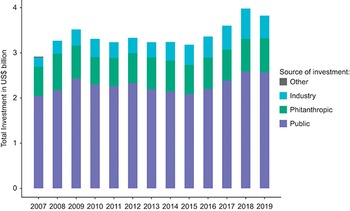
Fig. 3.10.4 illustrates total spending in US dollars by source of spending on R&D for new products and technologies to address challenges in public health caused by neglected diseases between 2007 and 2019. Total spending is around US$ 3.25 billion per year and was highest in 2009, 2017, 2018 and 2019 and lowest in 2007 and 2015. Between 2015 and 2018 spending on R&D for neglected diseases amounted to US$ 17.9 billion, 2.7% of global pharmaceutical R&D spending (US$ 660 billion) (Statista, 2021), signalling underinvestment in neglected disease R&D, considering that neglected diseases account for 19% of the total global burden of disease (IHME, 2019).
Global pharmaceutical companies are underinvesting in neglected disease R&D, 2019
B: billion.

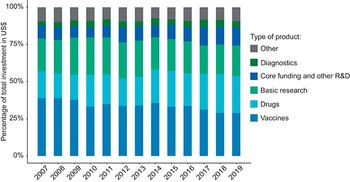
Between 2007 and 2019, on average, the three areas that accounted for the highest proportion of spending, in descending order, were vaccines, drugs and basic research (Fig. 3.10.5). The year 2014 saw the highest spending, with vaccines, drugs and basic research accounting for about 37.5%, 22.5% and 20% of total spending, respectively, when compared to other products such as diagnostics and core funding and other R&D products. Cumulatively, between 2015 and 2019, there was a gradual decline in total spending across all the different types of products.
Most money is spent on vaccines, drugs and basic research
R&D: research and development.

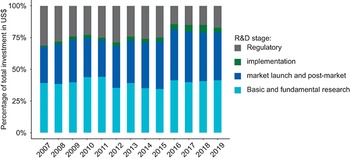
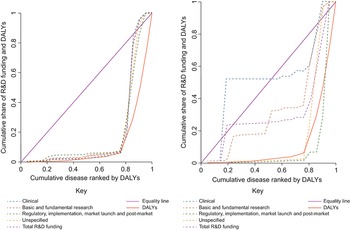
Between 2007 and 2019, although around 30% of total spending was unspecified, the majority (approximately 70%) of spending was shared between two stages, namely the clinical and the basic and fundamental research stages (Fig. 3.10.6). The regulatory, implementation, market launch and post-market stages received the smallest proportions of the total funding. Even though the proportion of total funding invested in clinical research has remained constant at around 35% during the period 2015 to 2019 (Fig. 3.10.6), the increased funding in clinical and preclinical R&D over the years could be translated into further innovation for neglected diseases in the years to come.
Most R&D spending to address neglected disease challenges goes on just two stages
R&D: research and development.

Sources of funding
The three most common sources of funding were industry, philanthropic and public sources. Between 2007 and 2019, public and philanthropic sources accounted for more than 70% and 20% of total expenditure, respectively, while industry sources accounted for about 10%.
The United States of America (USA) consistently ranks as the top funder, funding on average 65% of total spending between 2007 and 2019. Apart from the EU, which funds on average 3% of the total expenditure, other top funding countries include the United Kingdom and France, which fund on average 6% and 2%, respectively. However, it is important to note that over the same period, about 13% of the total spending came from countries that were not specified.
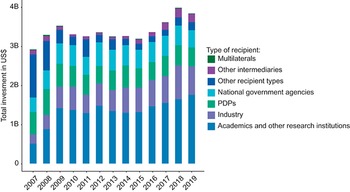
Fig. 3.10.7 shows total neglected disease R&D spending by type of recipient. On average academics and other research institutions receive the highest amount of investment followed by industry, whereas “other intermediaries”Footnote 2 receive the lowest amount of funding.
Academics and research institutions receive the biggest proportion of investment
PDP: product development partnership.

Fig. 3.10.7 Long description
The y-axis represents Total investment in U S dollars, from 0 to 4 billion, while the x-axis lists the years. The total values are as follows. 2007: 2.95 billion. 2008 to 2016: Between 3 and 3.5 billion. 2017: 3.6 billion. 2018: 4 billion. 2019: 3.9 billion.
Inequities and inequalities in R&D activity and funding
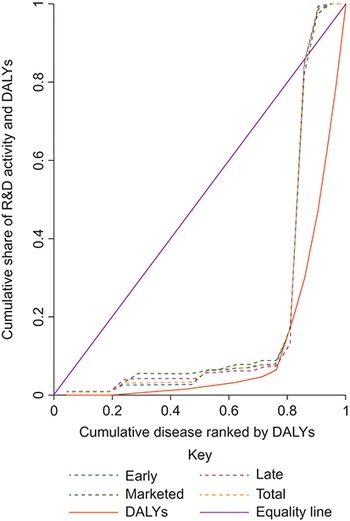
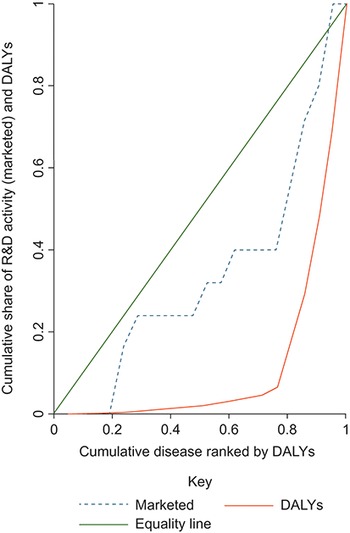
When we compare the distributions of pharmaceutical R&D activity and global DALYs across neglected diseases (Fig. 3.10.8), for the diseases at the top of the distribution of DALYs there is a disproportionate concentration of innovation in low-burden diseases, meaning that low-burden diseases receive more than the fair share of R&D activity when we consider their need relative to other diseases (inequity pro-low burden). This inequity is even more pronounced once we consider market launches in LMICs where we see low-burden diseases experience more market launches than their relative need would warrant when compared to high burden diseases (Fig. 3.10.9).
Low-burden diseases receive more than their fair share of R&D
DALYs: disability-adjusted life-years; R&D: research and development.
Note: Authors’ analyses.

LMICs receive few market launches for new pharmaceutical products
DALYs: disability-adjusted life-years; R&D: research and development.
Note: Authors’ analyses.

These inequities can be attributed to inequalities in the distribution of R&D funding across neglected diseases and/or the fact that most LMICs lack the resources and research infrastructure to develop their own medicines (Mrazek & Mossialos, Reference Mrazek and Mossialos2003).
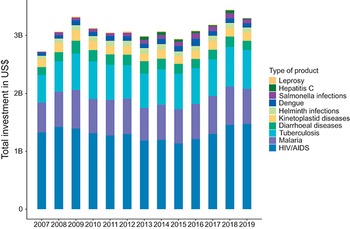
The 10 neglected diseases that received the highest R&D investment in US dollars between 2007 and 2019 for new products and technologies to address challenges in public health are shown in Fig. 3.10.10. HIV/AIDS received the highest investment by about US$ 16.9 billion, followed by malaria and TB, with US$ 7.9 billion and US$ 7.8 billion, respectively. Mycetoma, leptospirosis and hepatitis B received the lowest investment, which cumulatively in the last five years (2015 to 2019), was around US$ 1.5 million, US$ 9.3 million and US$ 16.5 million, respectively (Policy Cures Research, 2021a).
HIV/AIDS, malaria and TB receive most investment
B: billion.

Between 2015 and 2018 the cumulative spending has seen a steady increase, albeit remaining concentrated on HIV/AIDS, malaria and TB. Looking at global spending for R&D activity for neglected diseases, we observe a disproportionate concentration on low-burden neglected diseases.
Fig. 3.10.11 (the concentration curve of global R&D activity (1980–2019) and DALYs in LMICs (2018–2019)) and Fig. 3.10.12 (the concentration curve of market launches in LMICs (1980–2019) and DALYs (2018–2019) in LMICs (2018–2019)) show that low-burden diseases receive more than their fair share of funding given their relative disease burden. With the exception of spending on regulatory, implementation, market and post-market launch activities, this inequity is more pronounced when one considers funding allocated only to LMICs.
Low-burden diseases received more than their fair share of funding
DALYs: disability-adjusted life-years R&D; research and development.
Note: Authors’ analyses.

Reasons for inequities and inequalities
Several mechanisms may explain these inequities in innovation that impact differential access to treatment for neglected diseases to the detriment of health and well-being in LMICs. Concerns around pricing, affordability and the low potential for profit of those treatments in LMICs implies that firms have little incentive to develop innovations for these populations. This is aggravated by HICs giving priority to fund R&D for diseases that impact their own populations and the lack of international coordination in pooling and allocating resources to innovation taking account of health needs across all disease areas and geographies, as well as affordability.
These inequities are also intrinsically related to market failures and the effectiveness of mitigating strategies, including financing and incentive mechanisms.
Market failures in R&D: misalignment between private incentives and health need
There are several features of the R&D sector in the context of pharmaceutical innovation that are associated with market failures as a result of which the resource allocation for R&D efforts is not socially optimal (Arrow, Reference Arrow1962), thus justifying government intervention. Most notably there is imperfect appropriability, with significant knowledge spillovers implying that firms will not fully recoup the benefits of their investment in R&D (Henderson & Cockburn, Reference Henderson and Cockburn1996; Bloom, Schankerman & Van Reenen, Reference Bloom, Schankerman and Van Reenen2013). Research shows social rates of return to R&D (i.e. returns to the innovator, benefits to other firms and to society) to be up to five times higher than private rates of return (Salter et al., Reference Salter2000). These may lead to underinvestment (Romer, Reference Romer1990), in particular in risky areas where prospects of profitability are uncertain, such as neglected diseases.
There is also information asymmetry between pharmaceutical firms and funders because several aspects of innovation and its processes are difficult to describe contractually and there are often no incentives for private firms to disclose them. Asymmetry of information between firms and funders typically leads funders to underinvest in R&D in the light of risk and uncertainty (Czarnitzki & Toole, Reference Czarnitzki and Toole2007). This underinvestment may be more accentuated in markets with high levels of uncertainty such as those for neglected diseases. It also disproportionately impacts firms that may lack the financial resources to self-fund their activities; thus, decreasing prospects of breakthrough innovation by smaller but innovation-agile firms that play a crucial role in neglected disease markets (BVGH & BIO, 2012). Small and medium-sized enterprises (SMEs) play an important role in the development of innovation for neglected diseases being engaged in 41% of drug and vaccine R&D activity (BVGH & BIO, 2012), but they face severe funding constraints and often bear the risks and uncertainty associated with therapy development.
Infectious diseases do not have borders, thus eradication and to some extent herd immunity is a public good, which implies that some countries will free-ride on others’ efforts to invest in control and prevention measures. At an individual level there are externalities, implying that the social marginal benefits of infection control are higher than the marginal private benefits, leading to under provision of preventive and infectious control measures.
These factors, combined with the fact that pharmaceutical innovation is costly, lengthy and risky, result in firms focusing on markets that maximize the return on investment and therefore the level of innovation for neglected diseases is suboptimal.
These issues are often used as the rationale for intellectual property rights. Beyond the standard drawbacks of patent systems (e.g. costly patent races, incentives for incremental rather than breakthrough innovation, barriers to entry, pre-emptive patenting, etc.; see Gallini and Scotchmer (Reference Gallini, Scotchmer and Maurer2002) for a review), patent systems bring additional challenges in the context of neglected diseases. Patents are associated with lack of affordability in low-income settings where neglected diseases are endemic. And importantly, since markets for neglected diseases are therefore small, the incentives inherent in patent systems are reduced in the context of these diseases.
As a result, market incentives reinforced through a patent system alone are insufficient to stimulate innovation for neglected diseases and should, therefore, be coupled with other incentive mechanisms. In particular, policies related to public and donor financing have an important role to play. These financing policies can aim to build research capacity through the direct funding of innovation, or they can be designed to steer R&D and ensure equity and affordability through the implementation of a set of incentive mechanisms that directly or indirectly affect investment in R&D.
Using incentive mechanisms to stimulate innovation for neglected diseases: an overview
The underlying principle behind these mechanisms is the “delinkage” of the cost of research from market profitability so that the incentive to enter the market is not (or is less) contingent on the ability to charge high prices and recover costs through sales. Push mechanisms reduce upfront costs inherent to R&D activities by subsidizing R&D activities prior to product discoveries (i.e. ex ante). Pull mechanisms, on the contrary, offer a reward contingent on successful product discoveries (i.e. ex post). Hybrid mechanisms combine pull and push incentives.
Push mechanisms work better in settings where developers are capital constrained and risk averse. By subsidizing R&D costs push mechanisms also stimulate competition in innovation by enabling small companies and start-ups that lack capital to develop R&D activities. However, when effort is difficult to monitor and contracts are incomplete with research outcomes not contractable, push incentives lead to moral hazard, resulting in suboptimal levels and scope of innovation (Kremer, Reference Kremer2002).
Adverse selection may also arise. Payers have little insight into the relative chances of success of different R&D targets and the effort exerted in each project. Therefore, push mechanisms may incentivize projects with small chances of success (Kremer, Reference Kremer2002).
Under pull incentives, on the other hand, the risk lies with the innovator, with incentives accruing only when innovation occurs and targets key public health priorities. Innovators are better informed about the risk of investment but also, bearing the risk of development, have a strong incentive to prioritize projects with a higher chance of success and better prospects of social and financial impact. If public health needs are well defined, these pull mechanisms enable the funder to direct incentives to key priority areas as the reward is attributed after the innovation has occurred.
However, these mechanisms benefit larger companies that can leverage capital for R&D. Smaller firms (e.g. biotech companies) that are capital constrained and more risk averse are less likely to benefit from pull incentives. This is particularly relevant in markets where the prospects of profitability are weak or uncertain such as in the case of neglected diseases.
The extent to which push and pull incentives are effective also depends on the stage of the R&D process they target, given that the costs and risk vary considerably during the R&D process.
Pull incentives require the desired innovation to be specified in advance, so are less likely to encourage basic and fundamental research (Moran et al., Reference Moran2005; Grace & Kyle, Reference Grace and Kyle2009). Push mechanisms, on the contrary, incentivize R&D by decreasing the marginal cost of development and/or by ensuring access to key research inputs through collaboration with key stakeholders, thus favouring the invention phase of the R&D process when compared to pull. The facilitated collaboration with other stakeholders that is inherent to some forms of push mechanism (e.g. public–private partnerships) implies that they are also useful in supporting advanced stages of the R&D process, including regulatory hurdles and market launch.
The effectiveness of pull and push incentives is also likely to vary depending on the type of technology. Drug therapies that target treatment or management of chronic conditions offer a broader prospect for recouping R&D investments than prevention therapies or vaccines. Therapies and vaccines that target larger markets with high willingness and ability to pay for therapies (e.g. HIV) are less risky than smaller markets (e.g. schistosomiasis). Therapeutic areas also differ on scientific challenges and development times and costs. One could expect incentive mechanisms that enable collaboration among different stakeholders with different expertise and inputs to therefore perform better in areas where it is scientifically more challenging to innovate than those mechanisms that bring only R&D subsidization.
In what follows we present an overview of the main types of push and pull incentives. A full discussion of the advantages and disadvantages can be found in Table 3.10.2. Note that, in addition to push and pull mechanisms, pool mechanisms can be seen as indirect mechanisms to incentivize R&D. In pool mechanisms intellectual property and knowledge is “pooled” to enable collaboration in research, technology and knowledge sharing to fast-track R&D (Gold et al., Reference Gold2007). While these mechanisms relate to intellectual property and knowledge sharing rather than financing mechanisms, they have the potential to substantially decrease the costs and thus incentivize innovation. AMCs, one of the pull mechanisms discussed below, also involves pooling, but in terms of purchasing, rather than intellectual property and knowledge.

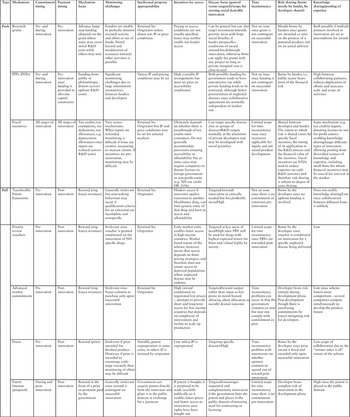
IP: intellectual property; ND: neglected disease; NTD: neglected tropical disease; PPP: public–public partnership; PRV: priority review voucher; R&D: research and development.
Table 3.10.2 Long description
The table has 11 main columns: Mechanism, Commitment timing, Payment timing, Mechanism focus, Monitoring challenges, Intellectual property appropriability, Incentives for access, Disease focus (general versus targeted) or scope for steering social desirable innovation, Time inconsistency, Risk sharing (borne mostly by funder, by developer, shared), and Knowledge sharing or pooling of expertise. It reads as follows.
Under Push: Row 1: Research grants. Commitment timing: Pre- and during innovation. Payment timing: Pre-innovation. Mechanism focus: Advance lump sum funding (depends on the grant where some may cover initial R and D costs while others may not). Monitoring challenges: Funders are unable to perfectly monitor research activity and effort or use of funds. Moral hazard and misdirection of resources towards other activities is possible. Intellectual property appropriability: Retained by: Originator unless donor sets IP or price conditions. Incentives for access: Pricing or access conditions are not usually specified, hence may neither enable nor hinder access. Disease focus: Can be general but can also target investment towards priority areas with large social benefits or High, if funder prespecifies conditions of award around breakthrough innovation, otherwise firms can apply for grants with any project so long as private marginal cost is close to zero. Time inconsistency: Not an issue since grant is not contingent on successful innovation. Risk sharing: Mostly borne by funders since grants are awarded ex ante on the premise of a potential product, not on an actual delivery. Knowledge sharing: Both possible if multiple partners involved in innovation are set as precondition for award.
Row 2: P P Ps. Commitment timing: Pre- and during innovation. Payment timing: Pre-innovation since funding is provided to alleviate capital constraints. Mechanism focus: Funding from public or philanthropic donors (covers upfront R and D costs). Monitoring challenges: Significant monitoring challenges due to large information asymmetries between funders and developers. Intellectual property appropriability: Varies; IP and pricing conditions may be set. Incentives for access: High (variable I P arrangements but most set price or accessibility conditions). Disease focus: Both possible: funding by government tends to have unrestrictive use while private funding tends to be restricted, although better prioritization of neglected diseases since collaborative agreements are normally independent of market size or High. Time inconsistency: Not an issue since funding is not contingent on successful innovation. Risk sharing: Borne by funder, i.e. public sector bears most of the financial risk. Knowledge sharing: High between collaborating partners, reduces duplication of efforts and increases scale and scope of activities.
Row 3: Fiscal incentives. Commitment timing: All stages of innovation. Payment timing: All stages of innovation. Mechanism focus: Tax credits, tax exemptions, tax deductions, tax allowances, for example, depreciation allowances once inputs are realized (covers R and D costs). Monitoring challenges: Vary across mechanisms. When inputs are rewarded monitoring is difficult if firms use creative accounting to increase claims. If incentives are pre-innovation, monitoring may be difficult. Intellectual property appropriability: Retained by: Originator but IP and price conditions may be set for selected markets. Incentives for access: Ultimately depends on whether there is passthrough of tax credits onto consumers. Do not generally accommodate provisions ensuring accessibility or affordability but in some cases may require companies to donate licenses to foreign government or non-profit entity (for example, N D tax credit H R 3156). Disease focus: Can target specific disease area or groups of diseases or R and D targets normally at the discretion of private developers and may be misaligned with social priorities. Time inconsistency: Limited scope for time inconsistency issue since incentives applicable for inputs and not actual product development. Risk sharing: Shared between developer and funder. The extent to which risk is shared varies by specific fiscal incentive, the timing of its application in the R and D process and the financial value of the incentive. Fiscal incentives on N T Ds tend to reduce expenses on early R and D activities and therefore risk sharing is subject to degree of costs sharing. Knowledge sharing: Some mechanism (e.g. tax credits) require donating licenses to not for-profit entities enabling knowledge sharing or target different types of innovators allowing pooling from diversified sources of knowledge and expertise, including small firms for whom financial incentives may be crucial for survival in the market.
Under Pull. Row 4: Transferable Patent Extensions. Commitment timing. Payment timing: Post innovation. Mechanism focus: Reward (ring fences revenues). Monitoring challenges: Generally irrelevant but rent-seeking behaviour may occur if qualification criteria for an extension are incomplete and nonspecific. Intellectual property appropriability: Retained by: Originator. Incentives for access: Hinders access if innovator applies extension to another blockbuster drug, can limit generic entry of that drug and limit its access and affordability. Disease focus: Targeted towards innovation in critically needed but less profitable areas or High. Time inconsistency: Not an issue since there is no commitment of extension pre-innovation. Risk sharing: Borne by the developer since no upfront funding is involved. Knowledge sharing: Does not enable knowledge sharing/Low since collaboration between different firms is unlikely.
Row 5: Priority review vouchers. Commitment timing: Pre-innovation. Payment timing: Post-innovation. Mechanism focus: Reward (ring fences revenues). Monitoring challenges: Irrelevant since voucher is granted conditional on the innovation of N D-specific drugs. Intellectual property appropriability: Retained by: Originator. Incentives for access: Early market entry enables faster access in high-income countries. Market based nature of the scheme, however, means that access depends on firms pricing strategies, and therefore does not ensure access to deprived populations where neglected disease may be endemic. Disease focus: Targeted at key areas of need/High since PRV will be used for drugs with highest expected return for firms and valued highly by society. Time inconsistency: Limited scope for time inconsistency since P R Vs are rewarded post innovation. Risk sharing: Borne by the developer since voucher is conditional on innovation for a specific neglected disease being delivered. Knowledge sharing: Low.
Row 6: Advanced market commitments. Commitment timing: Pre-innovation. Payment timing: Post-innovation. Mechanism focus: Reward (ring fences revenues). Monitoring challenges: Irrelevant since voucher is granted conditional on the innovation of N D specific drugs. Intellectual property appropriability: Retained by: Originator. Incentives for access: High (award conditional on negotiated low prices) - attempts to provide short and long-term access for low-income countries but depends on complexity of intervention and facility to scale up production. Disease focus: Targeted or reward output rather than input so less prone to moral hazard, allowing effort allocations to socially desired outcome. Time inconsistency: Time inconsistency problems can occur in that the government commits ex ante but may not comply with commitment ex post. Risk sharing: Developer bears risk entirely during development phase. Post development though there is purchasing commitments by payers mitigating risk for developers. Knowledge sharing: Low since scheme fosters more competition – several companies compete simultaneously to develop the product fastest.
Row 7: Prizes. Commitment timing: Pre-innovation. Payment timing: Post-innovation. Mechanism focus: Reward (prize). Monitoring challenges: Irrelevant if prize awarded for finished product. However, if prize is awarded to encourage early-stage research, then monitoring of effort may be difficult. Intellectual property appropriability: Variable; patent expropriation in some cases, in others I P is retained by originator. Incentives for access: Low unless I P is expropriated. Disease focus: Targeting specific diseases or High. Time inconsistency: Time inconsistency problem with uncertainty on whether sponsor commits to agreed size of reward post-innovation. Risk sharing: Borne by the developer since prize award is fixed and awarded only upon successful innovation. Knowledge sharing: Low scope of collaboration due to the winner-takes-it-all nature of the scheme.
Row 8: Patent buyouts (proposal). Commitment timing: During and post-innovation. Payment timing: Post-innovation. Mechanism focus: Reward in the form of a prize or premium paid by the government. Monitoring challenges: Generally irrelevant since reward is contingent on successful innovation. Intellectual property appropriability: Government can acquire patent directly from the innovator and place it in the public domain in exchange for a payment. Incentives for access: If patent is bought, it is proposed to be made available publicly so it enables lower prices and better access to innovation once rights have been bought out. Disease focus: Targeted or encourages sequential and complementary innovation if the government buys the patent and places in the public domain eliminating need for contracting or licensing. Time inconsistency: Limited scope for time inconsistency since there is no commitment pre-innovation. Risk sharing: Developer bears complete risk of innovation in the development phase. Knowledge sharing: High since the patent is placed in the public domain.
Push mechanisms
Product development partnerships (PDPs)
PDPs are the most common type of public–private partnerships for neglected diseases (Aerts et al., Reference Aerts2017; Widdus, Reference Widdus2005). PDPs may exist as voluntary collaborations between private companies and public funders, but may also involve contract-based stakeholders such as academic centres that provide scientific expertise in drug development (Moran, Reference Moran2005; Moran et al., Reference Moran2005; Widdus, Reference Widdus2005). More recently, along with public and private partners (e.g. academia, public funding organizations and pharmaceutical companies) PDPs, and public–private partnerships more broadly, include diverse stakeholders such as regulators, insurers and patient organizations (de Vruehm, de Vlieger & Crommelin, Reference de Vrueh, de Vlieger and Crommelin2019).
In 2017, there were around two dozen PDPs for neglected diseases, most created around 2000 (Borrás, Reference Borrás2017), with most focusing on malaria, HIV/AIDS and tuberculosis (TB). Notable examples are shown in Table 3.10.3 (Munoz et al., Reference Munoz2015; Borrás, Reference Borrás2017).
While focusing on all R&D stages, PDPs are particularly relevant in incentivizing early-stage innovation when firms may not have easy access to capital (Grace & Kyle, Reference Grace and Kyle2009) by financing the large upfront R&D cost through public or private funding, or a combination of both (Chapman et al., Reference Chapman2020; Sloan & Hsieh, Reference Sloan and Hsieh2017).
External financing (mainly through the public sector and multiple philanthropic organizations) and amounting to 16% of total “external” R&D funding for neglected diseases (Chapman et al., Reference Chapman2020) reduces the cost of failure for collaborating partners by sharing the risk of failure early on between them, thus incentivizing private R&D (Moran et al., Reference Moran2005; WHO, 2013). In exchange for funding and research inputs, the developer commits to invest in less profitable products at early R&D stages of basic research and to a reduction in eventual profits. The public sector contributes to the development and/or clinical trial stages that require most of the investment (Moran, Reference Moran2005). In most cases, PDPs channel funding through pharma partners, who own the rights to the product. In return for those rights, the partner commits to ensuring global access to the product at a reasonable price for those in developing countries (NBR, 2014). Notably in some product development schemes intellectual property rights (IPR) are owned by the partnership, so enabling some pricing control once the innovation is developed and thus promoting affordability and access (Buse & Walt, Reference Buse and Walt2000). Box 3.10.1 provides a case study of a PDP and Table 3.10.3 provides selected examples to show PDPs’ common features and differences with regard to IPR.
Who are Medicines for Malaria Venture (MMV)?
A collaborative non-profit product development partnership established in 1999 to fund and manage R&D and access to antimalarial medicines for 400 partners in 50 countries. This includes clinical centres in endemic countries across sub-Saharan Africa, South-East Asia and South America. The mission of MMV is to support specific, targeted interventions to reduce the burden of malaria in disease-endemic countries by discovering, developing and facilitating the delivery of new, effective and affordable antimalarial drugs.
Funding
US$ 350 million, funding source more than 60% private.
How does the PDP model work?
With financial support from donors including government agencies, private foundations, international organizations, corporations, corporate foundations and private individuals, MMV operates a virtual R&D model bringing together partners from academia and the public and private sectors and uses funds to establish a portfolio of R&D projects for developing antimalarial medicines.
The PDP does not itself have the capacity or infrastructure to undertake early-stage development projects in-house; instead, it relies on its partners for financing and other in-kind contributions (i.e. laboratories and expertise) and then allocates resources to the most promising projects, coordinates partner activities for various stages of the R&D process and manages the project portfolio. The PDP acts as a facilitator, bringing dedicated sources of funding and know-how to committed researchers so they can collaborate on the right projects to fulfil the objectives of the PDP’s mission.
IPR and access
When MMV enters into contractual relationships with partners, they encourage partners to work with them to guarantee that malaria drugs that they develop and launch will be accessible to those most in need in malaria endemic countries. This means ensuring first and foremost that agreements made within the PDP meet this public health mission. MMV negotiates for new medicines to be made available at an affordable price – typically with no profit and no loss – through public sector channels.
To achieve this, MMV must retain the rights to any intellectual property arising from the R&D undertaken by their partners. This is essential in allowing them to develop and launch drugs for the benefit of target patient groups. The following principles are applicable to all MMV contracts.
Exclusivity: If MMV does not own the necessary IPR outright, it insists on being granted an exclusive licence to use the “programme intellectual property rights” and any necessary “background intellectual property rights” to develop a drug for malaria and bring it to market. That licence must be worldwide, to ensure maximum flexibility for later-stage activities such as manufacturing and distribution.
Royalty-free: Any such licenses are preferably royalty-free, at least in malaria endemic countries, to help keep costs to a minimum and ensure that the drug will be sold at the lowest price possible in these countries.
Transferable: MMV does not conduct any R&D in-house or any manufacturing and, therefore, requires IPR that can be transferred to other partners – especially manufacturing partners – if necessary.
Portfolio
MMV and partners manage a portfolio of over 65 projects, the largest portfolio of antimalarial R&D and access projects ever assembled. The 2021 portfolio includes 11 compounds in clinical development addressing unmet medical needs in malaria, including medicines for children, pregnant women and relapsing malaria and drugs that could support the elimination/eradication agenda.
Notable successful products – Coartem Dispersible
Coartem is an antimalarial paediatric formulation that was developed by Novartis and gained approval by securing a United States Food & Drug Administration (FDA) priority review voucher (“fast-track voucher”) in 2009. It is approved in over 80 countries and by several regulatory agencies in Africa, Swissmedic and the EMA, and it is the only artemisinin-based combination therapy which has been approved by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use regulatory authorities. The Coartem combination of artemether and lumefantrine reduces the risk of resistance developing to either agent, and is therefore useful for the treatment of chloroquine-resistant Plasmodium falciparum cases.

Table 3.10.3 Long description
The table has 8 columns: Name of P D P, About or purpose of P D P, Funding amount, Stakeholders involved, Disease focus, Type of technology, Targeted R and D stage, and Intellectual property rights implications. It reads as follow. Row 1: Drugs for Neglected Disease Initiative (D N D i). About: Collaborative P D P. Non-profit R and D organization established in 2003 to fund and manage R and D development for medicines across over 200 partners. D N D i has already delivered eight field-adapted and affordable treatments for five deadly diseases, and aims to have delivered 25 new treatments by 2028. Funding amount: 615.5 million Euros (raised by end of 2019). Funding source: 57 percent public, 43 percent private. Stakeholders involved: Founding partners: W H O T D R, M S F, Malaysian Ministry of Health, Kenya Medical Research Institute, Indian Council of Medical Research, Oswaldo Cruz Foundation and Institute Pasteur. Donors: 1. Public: Australian Trade and Investment Commission, United Kingdom Department of Health and Social Care, Dutch Ministry of Foreign Affairs, French Development Agency, G I Z, European Commission, etc. 2. Private: B M G F, M S F, Takeda Pharmaceuticals, Wellcome Trust, etc. Disease focus: Visceral Leishmaniasis, Human African trypanosomiasis (sleeping sickness), Chagas, paediatric H I V, filarial infections, mycetoma and hepatitis C. Type of technology: Treatment (drugs). Targeted: R and D stage: Drug discovery, translational research, clinical trials and registration and access. Intellectual property rights implications: Variable per drug D N D i adheres to basic principles stated in its intellectual property rights policy. The overarching theme is that it will ensure that the results of the work carried out under its auspices are disseminated as widely as possible and its products made readily available and affordable in developing countries.
Row 2: Medicines for Malaria Venture (M M V). About: Collaborative P D P. Non-profit product development partnership established in 1999 to fund and manage R and D development and access to antimalarial medicines across around 400 partners in 50 countries (donors and clinical centres in endemic countries). MMV has validated more than 19 new malaria drug targets since 1999. Funding amount: 350 million U S D (raised by the end of 2018); Funding source: More than 60 percent private. Stakeholders involved: Founding partners: Launched with initial seed finance of 4 million U S D from the Government of Switzerland, United Kingdom Department for International Development, the Government of the Netherlands, the World Bank and the Rockefeller Foundation. Clinical centre partners: 50 clinical trial sites in sub-Saharan Africa, South East Asia and South America. Donors: 1. Public: United States government (U S A I D, N I H), Australian Government; Development of Foreign Affairs and Trade, United Kingdom Government (D F I D and D o H), Netherlands Ministry for Development Cooperation, etc. 2. Private: B M G F, Wellcome Trust, G S K, Sanofi, etc. Disease focus: Malaria. Type of technology: Treatment (drugs). Targeted R and D stage: Drug discovery, translational research, clinical trials and registration and access. Intellectual property rights implications: M M V retains rights to any Intellectual property rights that is essential in allowing them to develop and launch drugs for the benefit of M M V target group. Principles include: exclusivity, royalty-free, transferable Intellectual property rights.
Row 3: T B Alliance. About: Collaborative P D P. Non-profit product development partnership of around 38 partners established in 2000 to fund and manage R and D development and access to T B medicines. Funding amount: Around 184 million U S D (raised by end of 2019). Stakeholders involved: Founding partners: Launched with issuance of Cape Town Declaration by around 28 partners, including B M G F, European Commission, M S F, W H O, World Bank, Wellcome Trust, etc. Donors: 1. Public: Australian Aid, United Kingdom F C D O and M R C, U S A I D, Germany Federal Ministry of Education and Research, Netherlands Ministry of Foreign Affairs, Ministry of Foreign Affairs (Republic of Korea), etc. 2. Private: B M G F, The Rockefeller Foundation, E D C T P, Global Health Innovative Technology Fund (Japan), U N I T A I D, etc. Disease focus: Tuberculosis. Type of technology: Treatment (drugs). Targeted R and D stage: Drug discovery, translational research, clinical trials and registration and access. Intellectual property rights implications: T B Alliance’s mission includes an explicit commitment to A A A Mandate that all new products will be adopted, available and affordable to those with T B. No further information on Intellectual property rights implications.
BMGF: Bill & Melinda Gates Foundation; DFID: Department for International Development; DNDi: Drugs for Neglected Disease Initiative; DoH: Department of Health; EDCTP: European and Developing Countries Clinical Trials Partnership; FCDO: Foreign, Commonwealth and Development Office; GIZ: German International Cooperation Society; MMV: Medicines for Malaria Venture; MRC: Medical Research Council; MSF: Médecins sans Frontières; NIH: US National Institute of Health; PDP: product development partnership; TB: tuberculosis; TDR: Special Programme for Research and Training in Tropical Diseases; WHO:World Health Organization.
Despite the wide proliferation of public–private partnerships in recent years, in particular those focused on product development, a systematic review by Aerts et al. (Reference Aerts2017) did not find a single impact evaluation related to them, barring one analysis that compared the cost–effectiveness of the PDP model against AMCs and a hybrid scheme (PDP until phase II trials, followed by AMCs later) in the context of immunizations for malaria. While costs were lowest for the PDP scheme, DALYs averted were also the lowest, rendering PDPs a non-cost-effective approach. Most descriptive evidence, however, suggests that PDPs have led to a substantial increase in investment in R&D and innovation for neglected diseases. Between 2009 and 2013, 57% of new approvals in WHO’s list of essential medicines were developed through PDPs. PDPs have been particularly successful in vaccine development, with more than 400 PDP-led vaccines under development (Breitstein & Spigelman, Reference Breitstein and Spigelman2013); and over half of the vaccines for neglected diseases during the period 2009 to 2013 resulting from PDPs (Cohen, Sturgeon & Cohen, Reference Cohen, Sturgeon and Cohen2014). Despite these promising results, impact evaluations are seriously lacking and consistent data collection and availability of data to analyse cost–effectiveness of PDPs are necessary.
R&D grants and subsidies
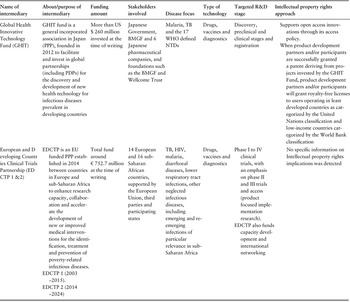
Grants and subsidies are normally in-advance lump sums awarded to fund R&D. They tend to focus on key priority areas identified by funders and cover the initiation of discovery or early stage research. Grants and subsidies constitute the main means to fund R&D adopted by public funders and third parties, contributing to 77% of total R&D funding for neglected diseases (Chapman et al., Reference Chapman2020). They specifically focus on early basic research, preclinical and clinical development stages, but tend to cover a wide range of neglected diseases (WIPO, 2014) and are primarily provided by public and philanthropic sources. This includes, for example, governments that allocate a proportion of their gross domestic product (GDP) such as the USA through the United States National Institute of Health and USAID, or the United Kingdom through the Department for International Development (DFID)Footnote 3 and Medical Research Council (MRC), as well as philanthropic organizations such as the Bill & Melinda Gates Foundation (BMGF) and the Wellcome Trust (Chapman et al., Reference Chapman2016). Grant funds are channelled either directly to researchers and developers or through a conduit (20% of research grants) such as public–private partnerships/PDPs with some private sector participation (Pugatch Consilium, 2017), or through non-PDP intermediaries such as the Global Health Innovative Technology Fund (GHIT) and the European and Developing Countries Clinical Trial Partnership. Non-PDP intermediaries generally have a broader funding base than PDPs, supported by science and technology organizations, aid agencies and philanthropic foundations, making them less vulnerable to changes from one donor funding stream (Table 3.10.4).

Table 3.10.4 Long description
The table has 8 columns: Name of intermediary, About or purpose of intermediary, Funding amount, Stakeholders involved, Disease focus, Type of technology, Targeted R and D stage, and Intellectual property rights approach. It reads as follows. Row 1: Global Health Innovative Technology Fund (G H I T). About: G H I T fund is a general incorporated association in Japan (P P P), founded in 2012 to facilitate and invest in global partnerships (including P D Ps) for the discovery and development of new health technology for infectious diseases prevalent in developing countries. Funding amount: More than 260 million U S D invested to date. Stakeholders involved: Japanese Government, B M G F and 6 Japanese pharmaceutical companies, and foundations such as the B M G F and Wellcome Trust. Disease focus: Malaria, T B and the 17 W H O defined N T Ds. Type of technology: Drugs, vaccines and diagnostics. Targeted R and D stage: Discovery, preclinical and clinical stages and registration. Intellectual property rights approach: Supports open access innovations through its access policy. When product development partners and/or participants are successfully granted a patent deriving from projects invested by the GHIT Fund, product development partners and/or participants will grant royalty-free licenses to users operating in least developed countries, as categorised by the United Nations classification and low-income countries, categorised by the World Bank classification.
Row 2: European and Developing Countries Clinical Trials Partnership (E D C T P 1 and 2). About: E D C T P is an E U-funded P P P established in 2014 between countries in Europe and sub-Saharan Africa to enhance research capacity, collaboration and accelerate the development of new or improved medical interventions for the identification, treatment and prevention of poverty-related infectious diseases. E D C T P 1 (2003 to 2015). E D C T P 2 (2014 to 2024). Funding amount: Total fund around 752.7 million Euros to date. Stakeholders involved: 14 European and 16 sub-Saharan African countries, supported by the European Union, third parties and participating states. Disease focus: T B, H I V, malaria, diarrhoeal diseases, lower respiratory tract infections, other neglected infectious diseases, including emerging and re-emerging infections of particular relevance in sub-Saharan Africa. Type of technology: Drugs, vaccines and diagnostics. Targeted R and D stage Phase 1 to 4 clinical trials, with an emphasis on phase 2 and 3 trials and access (product-focused implementation research). E D C T P also funds capacity development and international networking. Intellectual property rights approach: No specific information on Intellectual property rights implications was detected.
BMGF: Bill & Melinda Gates Foundation; EDCTP: European and Developing Countries Clinical Trials Partnership; EU: European Union; GHIT: Global Health Innovative Technology Fund; NTDs: neglected tropical disease; PDPs: product development partnership; PPP: public–public partnership; TB: tuberculosis; WHO: World Health Organization.
Evidence of the impact of grants and subsidies on neglected disease-specific R&D is lacking. However, general pharmaceutical industry research suggests that grants and subsidies have a positive effect on R&D investment, and the innovation of new molecules and patent applications (Toole, Reference Toole2012; Azoulay et al., Reference Azoulay2019; Kourouklis, Reference Kourouklis2021). These effects vary across different stages of drug development – in the case of orphan drugs, they are most pronounced in phase I and phase II of the drug development process (Kourouklis, Reference Kourouklis2021).
Fiscal incentives
Fiscal incentives focus on reducing the cost of R&D, raising the long-term rate of return on R&D investments, and include: tax exemptions, tax deferrals, tax allowances and tax credits (OECD, 2016).Footnote 4 Some apply to current expenditures, others to capital or revenues. Tax allowance, exemption and deduction consist of reducing the taxable amount by reducing the tax base before the tax liability is computed. With tax credits, in contrast, the tax liability is reduced by subtracting the credit after the liability has been determined. Tax deferrals do not reduce tax liability but defer tax payment. These incentives can be used to target single firms or foster collaboration across firms, with some schemes designed to support SMEs, and they can also be used to target a specific disease area or groups of diseases.
In practice, fiscal incentives for neglected diseases are mostly in the form of tax credits, exemptions and reliefs. Although the R&D phases targeted by fiscal incentives may vary across initiatives, tax incentives for neglected disease R&D have often been considered for encouraging basic and non-clinical research for vaccines or medicinal products (Rao, Reference Rao2011). By reducing the R&D cost, these incentives encourage innovation in areas of high risk and smaller returns on investment – such as neglected diseases. These particularly benefit SMEs that, while financially constrained, are often agile and innovative in neglected disease innovation (BVGH & BIO, 2012), thus increasing the prospects of breakthrough innovation.
There are two main tax incentive initiatives highlighted in literature (Monroe, Greenberg & Basey, 2015; Pugatch Consilium, 2017). These are the United States Neglected Disease Tax Credit Proposal (HR3156) which mandates a transfer of IPR, and the United Kingdom Vaccine Research Relief which is not necessarily limited to a neglected disease focus, but rather extends to orphan drugs and humanitarian driven innovations (Monroe, Greenberg & Basey, 2015; UK Government, 2016; US Congress, 2009a) (Table 3.10.5).

Table 3.10.5 Long description
The table has 6 columns: Fiscal incentive, About the scheme or incentive nature, Eligibility criteria or disease focus, Targeted companies, Benefits claimed, and Intellectual property rights implications. It reads as follows. Row 1: United Kingdom Vaccine Research Relief (V R R). About: Proposed and introduced in 2003, implemented and expired in 2017. In the United Kingdom R and D tax credits were introduced for S M Es in 2000 and extended to large companies from 2002. This is normally done through S M E scheme and Research and Development Expenditure Credits (R D E C) (non-neglected disease specific). V R R was introduced as an additional top-up relief for vaccines research. All companies may deduct a standard 100 percent of their R and D expenditure from their taxable income under S M E scheme and R D E C. While V R R allows 50 percent deduction on top for eligible R and D expenditure. Criteria: H I V/AIDS, T B and Malaria. Expenditures on activities ranging from identification of a chemical entity, or a potential vaccine through completion of Phase 3 clinical trials. Targeted companies: All companies. Large companies claiming VRR can only use the deduction option; they cannot claim payable credits for V R R. Benefits claimed: Number of claims under the V R R scheme remained at about 10 a year with support claimed of less than 5 million pounds each year between 2003 to 2004 and on average around 20 million pounds each year in the period up to 2012 to 2013. Intellectual property rights implications: No information detected on intellectual property rights implications.
Row 2: United States neglected disease tax credit proposal (H R 3156). About: Proposed in 2009, no information detected on introduction or implementation. This proposal was put forward by the biotechnology company (biotech) Genzyme with the sponsorship of Representative Donald Payne and others. 50 percent tax credit for non-clinical expenses for research on neglected disease treatments. Criteria: All N T Ds (see Table 3.10.1) and any infectious disease with unmet need or disproportionately affecting the poor. Qualified research: Only preclinical or basic research. Companies could not use the credit for expenses incurred from clinical trials or regulatory review. This includes expenses for in-house or contracted research activities undertaken for the purpose of non-clinical basic research only. With this specific criteria of discovering information - which is 1. technological in nature, and 2. the application of which is intended to be useful in the development of a new or improved business component of the taxpayer. Targeted companies: All companies. In-house or contracted research services. Benefits claimed: No information detected on implementation. Intellectual property rights implications: Mandates a transfer of intellectual property rights and could only apply for the credit if they have donated a royalty-free licence to a foreign government or a non-profit research organization for the claimed research in that year.
Given the reduced number of fiscal incentives for neglected diseases, their effectiveness in spurring research for neglected diseases is difficult to ascertain. Rao (Reference Rao2011) argues that tax credits in the United Kingdom did not attract new firms to invest in R&D for neglected diseases and alone are insufficient to increase R&D spending in global health. In contrast, the tax credits created in the Orphan Drug Act (P.L. 97–414) in 1983 have had a significant impact on orphan drug development, leading to 201 new orphan drugs in the market and this trend has been particularly strong in recent years; for example, 18 new drugs gained approval in 2014 (EY, 2015). Yin (Reference Yin2008) shows that the Orphan Drug Act-led income tax credit (equal to 50% of clinical trial expenses) increased the annual flow of clinical trials for drugs for rare diseases by 69%.
Pull mechanisms
Transferable patent extensions
Transferable patent extensions (TPEs) or wildcard patent extensions are a form of supply-side production response intended to encourage R&D for critically needed drugs in less attractive disease areas (Sonderholm, Reference Sonderholm2009). A successful innovator of a new drug is granted a one-time patent extension (ranging from six months to two years in the USA and five years in Europe) that can be applied to one of the innovator’s other drugs or sold to another firm. Such an extension is worthwhile for companies with a drug in their portfolio whose patent is near to expiry, and provide a source of return on investment (Moran, Reference Moran2005; Sonderholm, Reference Sonderholm2009; Seabury & Sood, Reference Seabury and Sood2017). These incentives can be particularly important for attracting companies to undertake R&D for neglected diseases because the return on investment in neglected disease markets remains insufficient due to limited market size and affordability (Seabury & Sood, Reference Seabury and Sood2017). Our review did not yield notable examples of TPEs granted for neglected diseases, but they have been proposed as a potential incentive in the context of developing new antimicrobials to tackle the problem of antimicrobial resistance (AMR) (Spellberg et al., Reference Spellberg2007; Spellberg et al., Reference Spellberg2008; Outterson, Samora & Keller-Cuda, Reference Outterson, Samora and Keller-Cuda2007). Within this context, Outterson and McDonnell (Reference Outterson and McDonnell2016) calculate the potential costs of a 12-month extension and conclude that such an extension would cost the USA’s health system more than US$ 4.8 billion, making such an extension extremely inefficient.
Patent buyouts
A patent buyout is an outcome-based pull mechanism aimed at delinking returns from price and volume-based sales by acquiring the patent directly from the innovator in exchange for a payment (Batista et al., Reference Batista2019). The government offers to buy the rights to the patent to encourage innovation and subsequently places the patent in the public domain for unrestricted access. The scheme may specify criteria ex ante to run a prize-like competition or may engage in ex post identification of a suitable patent candidate (Robinson, Ritchie & Kenny, Reference Robinson, Ritchie and Kenny2020). Patent buyouts add a markup to private market valuations and internalize the social returns to innovation, which in the context of limited market size for neglected diseases, assures innovators a satisfactory return on investment. By abolishing private patent rights, it also fosters access to innovation through lower prices.
Despite the theoretical applicability of buyouts, there is a lack of real-world application and empirical evidence on its effectiveness in the context of neglected diseases. In the context of neglected diseases, patent buyouts have only been proposed within the Medical Innovation Prize Fund (S.2210) as an end-product prize (US Congress, 2007). Theoretical evidence on welfare effects concludes that buyouts rewarding innovators based on observed market outcomes are an effective tool to improve social welfare (Galasso, Mitchell & Virag, Reference Galasso, Mitchell and Virag2016; Radhakrishnan, Reference Radhakrishnan2016). Outterson (Reference Outterson2006) argues that by recovering lost R&D costs, buyouts are a cost-effective method to improve access and health outcomes in LMICs. For example, the lost R&D cost recovery for GlaxoSmithKline’s Cervarix human papillomavirus vaccine is as little as US$ 29.2 million. Therefore, for a patent buyout that would cost as little as US$ 30 million, the medicine could be immediately available for millions in LMICs in its generic form.
Priority review vouchers (PRVs)
The PRV scheme enables firms with drugs approved for certain tropical diseases to apply for a voucher that can be used to obtain priority review for an unrelated drug application, so shortening the regulatory agency review time from a standard average of 10 months to a maximum of 6 months (Ridley, Grabowski & Moe, Reference Ridley, Grabowski and Moe2006; Gaffney, Mezher & Brennan, Reference Gaffney, Mezher and Brennan2019; Aerts et al., Reference Aerts2022). Alternatively, the firm can sell the voucher to another firm.
The innovator retains IPR and prices may be unaffordable if this mechanism is not complemented by differential (tier) pricing, donation or a drug access programme.
At the time of writing, PRV initiatives are primarily offered through the United States FDA’s scheme (FDA, 2020). To be eligible, products need to satisfy certain criteria including: approval by the FDA or the EMA; recognition as a product for serious conditions where there is unmet medical need; adequate and well-controlled trials have established that the drug influences a surrogate end-point and there is clinical superiority to other existing therapies. PRVs mostly focus on late-stage clinical development, registration and post-marketing and delivery stages of the R&D model and are commonly awarded to drugs. For a potential blockbuster drug, the value of PRV has been estimated by some experts to be worth anywhere between US$ 50 million to US$ 500 million (Robertson et al., Reference Robertson2012). As of late 2018, seven PRVs had been awarded for neglected/tropical diseases – in malaria, tuberculosis, leishmaniasis, cholera, onchocerciasis (river blindness) and Chagas disease (US FDA, 2017; Cohen, 2018). Since then, the PRV programmes have expanded substantially, with many more vouchers issued across tropical disease, rare paediatric disease and medical countermeasure indications).
Notable examples include: Coartem, which is now a first-line treatment for malaria across Africa and South-East Asia; benznidazole indicated for Chagas disease (in use as early as the 1970s, approved by FDA in 2017); Krintafel, the first new antimalarial in over 18 years (approved in 2018); and Moxidectin, a treatment for onchocerciasis (approved in 2018) (MDGH, 2018; Cohen, Reference Cohen2018) (Table 3.10.6).

Table 3.10.6 Long description
The table has 6 columns: Drug name, Supplier(s), Year P R V granted, Disease focus, How was the voucher redeemed or traded, and Approach to I P R transfer. It reads as follows. Row 1: Coartem (artemether or lumefantrine); Novartis; 2008; Malaria; First P R V so market value of P R V was unknown. In 2011, Novartis redeemed the voucher on a B L A for Ilaris (canakinumab) to treat gouty arthritis. F D A declined the application; Innovator retains I P R. Row 2: Benznidazole; Chemo Research; 2017; Chagas; blank; Innovator retains I P R. Row 3: Krintafel; G S K; 2018; P. vivax malaria; No information found or anonymously redeemed; Innovator retains I P R. Row 4: Moxidectin; Medicines Development for Global health; 2018; Onchocerciasis (river blindness); blank; Innovator retains I P R.
BLA: biologics licence application; FDA: Food & Drug Administration; IPR: intellectual property rights; PRV: priority review voucher.
Evidence on the impact of PRVs on R&D is hampered by a lack of data; sample sizes are small due to the low number of vouchers issued. PRVs have been found to not significantly impact clinical trial activity for neglected tropical disease (Aerts et al., Reference Aerts2022; Jain et al., Reference Jain2017) nor for rare paediatric conditions (Hwang et al., Reference Hwang2019) but have been found to increase the likelihood of initiation of large patient trials for paediatric rare disease (Hwang et al., Reference Hwang2019).
Advance market commitments (AMCs)
In AMCs the funder/donor commits to (fully or partially) subsidize the initial purchases of innovative medicines at an agreed price, for an established maximum threshold, to be provided to a specified set of countries. The subsidy is normally only triggered when countries purchase the innovation. The binding agreement is signed before the medicine is developed and contains a full specification of the desired product and use. They also often involve a commitment by the firm to sell at an affordable price (close to marginal cost and below the AMC price) all quantities beyond the agreed subsidization threshold in the agreed countries (Levine, Kremer & Albright, Reference Levine, Kremer and Albright2005).
By “pooling” purchase commitments across many countries the scheme creates a profitable and less risky market for the developer by establishing volume guarantees. By pooling funding from several donors and negotiating a low price it enables LMICs to access the medicine. The risk is shared by the developer and the funder: while in the development phase costs are born entirely by the developer who still bears fully the risks associated with the scientific challenges of R&D, when innovation occurs the funder commits to a level of demand in participating countries, thus minimizing for the developer the risk associated with low demand and reduced affordability in LMICs (including international advocacy for low prices of novel treatments) (Berndt et al., Reference Berndt2007). For the funder/sponsor the scheme is beneficial as the financial commitment only occurs if a medicine with social value targeting key areas of need is developed and launched in the market.
AMC is intended to focus on clinical development, registration and post-marketing and delivery stages of the R&D model (Pugatch Consilium, 2017). At the time of writing, only two AMCs have been implemented. In 2007, BMGF initiated the first AMC pilot programme targeting the development of pneumococcal conjugate vaccine (PCV) (Gavi-run pilot Pneumococcal AMC) and in June 2020, Gavi launched the second AMC, COVID-19 Vaccines AMC (COVAX AMC) to accelerate access to 1.8 billion COVID-19 vaccine doses for 92 lower-income countries (Gavi, 2022). Price details of the most recent Gavi supply agreement are provided in Table 3.10.7.

Table 3.10.7 Long description
The table has 9 columns and reads as follows. Name: Pneumo A M C 2009. About or purpose of A M C: It was launched by donors in 2009 to accelerate the development of vaccines that meet country needs, scale production and accelerate vaccine uptake, while testing the A M C concept for future applications. Funding amount: 1.5 billion U S D to the Pneumococcal A M C (as of January 2021). Stakeholders involved: A M C donors: B M G F, Governments of Canada, Italy, Norway, the Russian Federation and the United Kingdom. Vaccines rolled out in 45 countries across 3 continents by 2015. Manufacturers: G S K, Pfizer and S I I. Eligibility criteria and disease focus: Pneumococcal disease; Target product profile required to be prequalified by W H O. How is A M C awarded?: The A M C is delivered through supply agreements with manufacturers where donors make a financial commitment through Gavi alliance to fully or partially finance the purchase of treatments meeting pre-established criteria at a specified price to be distributed on Gavi countries. Specific supply agreements are available on the Gavi Pneumo A M C. A latest AMC was awarded in June 2020 by UNICEF in its capacity as Gavi’s procurement agency to SII for its recently W H O-prequalified P C V 10 product. S I I received an award of 10 million doses annually (Annual Supply Commitment) from 2020 for a period of 10 years, for a cumulative total of 100 million doses. 5 percent of the A M C funds are allocated to S I I according to the A M C terms and conditions, representing 75 million U S D. The Tail Price for this agreement is 2.00 U S D per dose (for the 5-dose vial presentation) from 2020 onwards, a reduction of over 30 percent from the previous lowest Tail Price of 2.90 U S D per dose. Type of technology: Vaccines. Targeted R and D stage: Focused mainly on late-stage R and D and launch. I P R implications Innovator retains I P R.
a Gavi: Supply agreements, https://www.gavi.org/investing-gavi/innovative-financing/pneumococcal-amc/manufacturers/supply-agreements
AMC: advance market commitment; BMGF: Bill & Melinda Gates Foundation; IPR: intellectual property rights; R&D: research and development; SII: Serum Institute of India. WHO: World Health Organization
With only two AMCs fully implemented, evidence is either descriptive due to a lack of appropriate counterfactuals or based on simulations. For the PCV AMC, Médecins sans Frontières (MSF) indicated that for over 12 years donors have granted more than US$ 1 billion in subsidies to two vaccine producers, GSK and Pfizer (main producers of the vaccine in the market) (MSF, 2020). Although in principle both manufacturers were obliged to cut the price after a predetermined level of doses was sold, or to license their technology to other manufacturers, prices were only marginally reduced and the transfer of technological know-how was not adequate to expand the manufacturer base into LMICs (MSF, 2020). COVAX AMC has also gathered more than US$ 6 billion in funds from governments, foundations and the private sector (WHO, 2021c). Further evidence suggests that the AMC for PCV has increased vaccination coverage across 60 countries immunizing over 49 million children and saving 14 to 17 million DALYs through to 2015 (Kremer, Levin & Snyder, Reference Kremer, Levin and Snyder2020; BCG, 2015). Both the PCV AMC as well as a vaccine AMC for malaria, TB and HIV/AIDS have been shown to be cost-effective in modelling studies (Berndt et al., Reference Berndt2007; Tasslimi et al., Reference Tasslimi2011).
Prizes
Prizes are rewards for innovations that may focus either on the technical specifications of a product or on the desired outcomes it delivers. Prizes may adopt different models but essentially are a pay for performance scheme where the developer receives a lump sum amount if a certain innovation with social impact is achieved (Hollis, Reference Hollis2005a; Reference Hollis2005b; Love & Hubbard, Reference Love and Hubbard2007), with the risk borne by the developer. Prizes thus incentivize innovations where social marginal benefits outweigh (or are equal to) marginal costs of development, thus promoting an efficient allocation of innovation effort. Some propose that this mechanism be voluntary, and complement the patent system by imposing structure on market rewards defined on the basis of incremental benefits (Hollis, Reference Hollis2005a; Pogge, Reference Pogge2005); for example, the Health Impact Fund prize scheme proposed by Hollis and Pogge (Reference Hollis and Pogge2008). Others recommend that such schemes be compulsory, with companies forgoing IPR so the developed drug could be made available globally, i.e. also in economically deprived populations (Love & Hubbard, Reference Love and Hubbard2009; Syed, Reference Syed2009; Shavell & Van Ypersele, Reference Shavell and Van Ypersele1999).
In the context of neglected diseases, prize schemes have mostly been used to reward innovation in relation to a specific disease; however, there have been some schemes that encompass any innovation for neglected diseases (Pugatch Consilium, 2017).
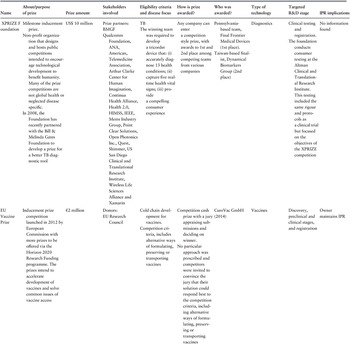
To date, only five of the 11 prize schemes in health targeted neglected diseases (Monroe, Greenberg & Basey, 2015; Pugatch Consilium, 2017), including the Qualcomm Tricorder XPRIZE (run by the XPRIZE Foundation and sponsored by the Qualcomm Foundation), the Health Impact Fund, the EU vaccine prize led by the European Research Council, the Longitude Prize led by Nesta (a United Kingdom innovation foundation) and a combined initiative proposed by Bangladesh, Barbados, Bolivia and Suriname. With a combined funding purse of roughly US$ 130 million, these prizes mostly focus on basic R&D research, preclinical development and post-marketing and delivery stages of the R&D model. Disease fields included treatments for TB, AMR and vaccine cold chain development, with some prizes allowing companies to maintain IPR and others replacing IPR rights with respective prize awards (Table 3.10.8).

Table 3.10.8a Long description
The table has 10 columns: Name, About or purpose of prize, Prize amount, Stakeholders involved, Eligibility criteria and disease focus, How is prize awarded, Who was awarded, Type of technology, Targeted R and D stage, and I P R implication. It reads as follows. Row 1: XPRIZE Foundation (10 million U S D). About: Milestone inducement prize. Non-profit organisation that designs and hosts public competitions intended to encourage technological development to benefit humanity. Many of the prize competitions are not global health or neglected disease-specific. In 2008, the Foundation has recently partnered with the Bill and Melinda Gates Foundation to develop a prize for a better T B diagnostic tool. Stakeholders involved: Prize partners: B M G F; Qualcomm Foundation, A N A, American, Telemedicine Association, Arthur Clarke Center for Human Imagination, Continua Health Alliance, Health 2.0, H I M S S, I E E E, MemsIndustry Group, Point Clear Solutions, Open Photonics Inc., Quest, Shimmer, U S San Diego Clinical and Translational Research Institute, Wireless Life Sciences Alliance and Xamarin. Eligibility criteria: T B; The winning team was required to develop a tricorder device that: 1. accurately diagnose 13 health conditions; 2. capture five real-time health vital signs; 3. provide a compelling consumer experience. How is prize awarded? Any company can enter a competition-style prize, with awards to first and second place among competing teams from various companies. Who was awarded? Pennsylvania-based team, Final Frontier Medical Devices (first place). Taiwan-based finalist, Dynamical Biomarkers Group (second place). Type of technology: Diagnostics. Targeted R&D stage: Clinical testing and registration. The foundation conducts consumer testing at the Altman Clinical and Translational Research Institute. This testing included the same rigour and protocols as a clinical trial but focused on the objectives of the XPRIZE competition. I P R implications: No information found.
Row 2: E U Vaccine Prize (2 million Euros). About: Inducement prize competition launched in 2012 by the European Commission, with more prizes to be offered via the Horizon 2020 Research Funding programme. The prizes intend to accelerate the development of vaccines and solve common issues of vaccine access. Stakeholders involved: Donors: E U Research Council. Eligibility criteria: Cold chain development for vaccines. Competition criteria, includes alternative ways of formulating, preserving or transporting vaccines. How is prize awarded? Competition cash prize with a jury appraising submissions and deciding on winner. No particular approach was prescribed and competitors were invited to convince the jury that their solution could respond best to the competition criteria, including alternative ways of formulating, preserving or transporting vaccines. Who was awarded? CureVac G m b H (2014). Type of technology: Vaccines. Targeted R&D stage: Discovery, preclinical and clinical stages, and registration. I P R implications: Owner maintains I P R.

Table 3.10.8b Long description
The table has 10 columns: Name, About or purpose of prize, Prize amount, Stakeholders involved, Eligibility criteria and disease focus, How is prize awarded, Who was awarded, Type of technology, Targeted R and D stage, and I P R implication. It reads as follows. Row 3: Health Impact Fund (proposal; A reasonable minimum required is 6 billion U S D per year): About: A new proposal by Incentives for Global Health to provide a complementary or alternative to the patent system for development of pharmaceutical innovations intended for poor patients. Stakeholders involved: Founding partner: Incentives for Global Health. Potential funding partners: Governments, charitable foundations or international taxes. Eligibility criteria: Pharmaceutical innovations intended for poor patients. How is prize awarded? The product is supplied at a generic price with a potential mark up for increment health gains, and the developer is not rewarded for the R&D element until it can demonstrate that the resulting product has health value for the intended patients. A 10-year fixed payment contingent on development, making I P R available for competing manufacturers, and calculated according to health impact of developed product. Who was awarded? (blank). Type of technology: Drugs. Targeted R&D stage: Discovery, preclinical and clinical stages, and registration. I P R implications: Developer making I P R available for competing manufacturers.
Row 4: Longitude Prize (10 million-pound prize fund): About: An ex ante inducement prize launched in 2014, awarded to the submission considered most impactful and feasible, currently for a competitive A M R innovation. Stakeholders involved: Lead by: Nesta (United Kingdom). Funding partners: Innovate United Kingdom and Biotechnology Industry Research Assistance Council (B I R A C). Supported by: Amazon, B B C, Science Museum, and Marks and Clerk I P services. Eligibility criteria: A M R. Any team of innovators from anywhere in the world that can invent an affordable, accurate, fast and easy-to-use point-of-care diagnostic test for bacterial infections that will allow health professionals worldwide to administer the right antibiotics at the right time (prize rules). How is prize awarded? Competition style prize, with 8 million pounds pay-out to winner, judged by a Prize Advisory Panel. Who was awarded? The first challenge is ongoing with around 50 registered teams. Type of technology: Diagnostics. Targeted R&D stage: n/a. Testing will take place over the six months following the Prize Advisory Panel’s decision. If the Panel judges that the testing confirms that the entry meets the criteria, they will recommend the entry as a potential winner. I P R implications: Developer maintains I P R.
The table has 10 columns: Name, About or purpose of prize, Prize amount, Stakeholders involved, Eligibility criteria and disease focus, How is prize awarded, Who was awarded, Type of technology, Targeted R and D stage, and I P R implication. It reads as follows. Row 5: B B B S open-source dividend (100 million U S D for T B. 250 million U S D for Chagas. 10 percent of donor assistance for health (D A H) to enhance access): About: Proposal by Bangladesh, Barbados, Bolivia and Suriname. Introduced in 2009 and 2015, of an open-source dividend and milestone prizes fund to reward R&D open innovation and access for donor-supported markets. Some of the Bangladesh, Barbados, Bolivia and Suriname proposals include the obligation to grant open, reasonable and non-discriminatory licenses to patent pools (such as the UNITAID patent pool). Stakeholders involved: B B B S, W H O, and Knowledge Ecology International. Eligibility criteria: Chagas disease, T B, H I V/AIDS, and malaria. Information on technical specification information not found. How is prize awarded? B B B S developed a prize fund that is endowed through a committee placed in W H O, which then allocates prizes to neglected diseases in separate disease-specific proposals, each drafted separately. The main objective is to share a percentage of end-product prize money with those who openly, freely share knowledge materials and technology. Example: The 2015 Development of Low-Cost Rapid Diagnostic Test for Tuberculosis Proposal suggests endowing a 100 million U S D for an entrant T B diagnostic that satisfies criteria set by W H O committee. A licensing pool would manage the intellectual property and guarantee that the diagnostic would be made available at an accessible price. Who was awarded? No information found. Type of technology: Drugs, diagnostics and vaccines. Targeted R&D stage: Clinical trials. I P R implications: I P R to be transferred to patent pool.
AMR: antimicrobial resistance; ANA: All Nippon Airways; BBBS: Bangladesh, Barbados, Bolivia and Suriname; BBC: British Broadcasting Corporation; BMGF: Bill & Melinda Gates Foundation; EU: European Union; HIMSS: Healthcare Information and Management Systems Society; IEEE: Institute of Electrical and Electronics Engineers; IP: intellectual property; IPR: intellectual property rights; TB: tuberculosis; WHO: World Health Organization.
Different prize schemes focusing on specific disease areas and sometimes on unmet needs have spurred innovative activity in early stages of R&D. For instance, the Gotham Prize for Cancer research has led to the development of novel approaches to treat cancer (Bernstein, Reference Bernstein2008). Similarly, the Dr Paul Janssen Award, aimed at solving unmet medical needs and rewarding scientists for basic or clinical research, has advanced innovation in the treatment of rare diseases such as mosaicism (Johnson & Johnson, 2020). The Prize4Life organization has accelerated discovery of treatment of amyotrophic lateral sclerosis (ALS) through its ALS Prediction Prize scheme (Leone, Reference Leone2012). While such awards have increased early-stage discovery in critical areas as described above, systematic evidence on the impact of prizes on stimulating R&D for neglected diseases is virtually non-existent.
Policy relevance and conclusions
Since the early 2000s there has been a considerable amount of effort to tackle the global burden associated with neglected diseases by fostering innovation in safe, effective and affordable medicines and prevention. To stimulate R&D investment a complex range of innovative financing mechanisms have been proposed, with most of the funding provided by public, philanthropic and private industry sources. Over the course of the last decade total R&D funding grew by 14%, rising from US$ 3 410 billion in 2010 to US$ 3 876 billion in 2019 (Chapman et al., Reference Chapman2020). And although progress has been made, control of neglected diseases is still largely inadequate and extremely difficult to monitor or evaluate. There is little evidence on the effectiveness of the current mix of innovative financing mechanisms, either at the individual or the collective level.
At the disagregated level the prospect for effective delinkage of R&D from market considerations varies across schemes and specificities of initiatives (in particular, schemes that only partly fund R&D costs only enable partial delinkage; for example, in the case of prizes it can be difficult to measure the expected effects of a registered health product in order to estimate a suitable prize amount). The heterogeneity and lack of widespread implementation of these schemes means there is little evidence on whether they succeed in stimulating innovation in key areas of need and the extent to which they may lead to crowding-out of private investment.
From a policy perspective, it is important to assess the extent to which these different funding incentive mechanisms work, as well as their relative cost–effectiveness. This can be achieved by international collaboration in devising and implementing pilot schemes designed to allow an assessment of the factors that hinder or enable a particular mechanism’s effectiveness. Yet, to date, the rolling out of these schemes seems to be linked to political will and the strength of their proponents’ advocacy rather than the need to gather and examine evidence. For example, tax credits have been considered to have a low uptake as an incentivizing mechanism for R&D in the United Kingdom. The tax credit-based vaccine research relief initiative came to an end in 2017, with United Kingdom assessors having found evidence for minimal or negligible impact on incentivizing R&D uptake (UK Government, 2016). Meanwhile, in the USA, pharmaceutical companies can receive a 20% R&D tax credit (that can be increased to 50% if the neglected disease is low burden), reflecting a contrasting view of the potential effectiveness of this approach (Mueller-Langer, Reference Mueller-Langer2013).
Another issue that can be observed among innovative financing mechanisms is their primary focus on “innovation” as an objective rather than “access and affordability”. The ultimate desirability of each of these mechanisms relies on the extent to which they either embed or are complemented by provisions to ensure innovation is affordable and supplied in LMICs. Such provisions include knowledge and technology transfer, pricing and financing policies. In their absence, some of the incentives may further widen existing health inequalities, for example by enabling high prices through extending the effects of IPR arrangements. Schemes that combine incentives to foster innovation coupled with specific provisions with regard to weakening IPR and/or pricing arrangements (e.g. AMCs and the Health Impact Fund) are therefore more likely to contribute effectively to tackling neglected diseases in LMICs.
On the collective level, the current push and pull mechanisms remain fragmented and consistently lack a governing overarching framework. Most schemes implemented to date target one or few disease areas, with allocation of resources across neglected diseases largely depending on donors’ views of need or particular remit of action. There is no needs assessment that systematically compares the relative needs associated with neglected diseases globally, assesses the costs and benefits of addressing them, and defines guidance on priority-setting for the coordinated global allocation of funding and targeted incentive mechanisms. This state of affairs risks a suboptimal allocation of funding as well as negative spillover effects across initiatives. The relationship between push mechanisms (such as R&D tax credits or PDPs) and pull mechanisms (such as the prizes, patent system, patent pools, AMCs or wildcard extension) is a good example. The Medical Innovation Prize Act (US Congress, 2007) was proposed as a replacement for the patent system, while others such as the earlier United States Orphan Drug act (US Congress, 1983) aim to complement or augment existing patency jurisdictions.
A governance and resource allocation framework that embeds incentive schemes would need to assess their relative ability to generate valuable innovations across the different disease areas, how they interplay with intellectual property regulations, and consequently their implications for affordability. Given that the R&D landscape is not comprehensively mapped or supranationally governed, it is challenging to produce such a framework, hindering the ability to make informed decisions to allocate resources efficiently across key priority areas. As a result, considerable systemic gaps in funding exist across different R&D stages and disease areas. The Global Strategy and Plan of Action on Public Health, Innovation and Intellectual Property drawn up by WHO’s consultative expert working group (WHO, 2012) highlights the key elements desirable in such a framework as being: needs assessment and prioritization; promoting R&D and R&D capacity; transfer of technology; application and management of intellectual property to contribute to innovation and promote public health; improving delivery and access; promoting sustainable financing mechanisms; and establishing and monitoring reporting systems.
Finally, WHO recognizes that the way research is currently funded is integral to the perceived need for better coordination. There are many funders in both public and private sectors, as well as many research organizations in public and private sectors and in partnership between them. Coordination among these actors is strongly needed to identify and manage research priorities. Not only will coordination provide opportunities for pooling resources for donors to fund a multiplicity of research institutions in developing country neglected diseases, but it will also enable the strategic governance of initiatives and inputs made in priority-setting, monitoring and evaluation, coordination and advocacy, and impact assessment (WHO, 2012).Footnote 5