Introduction
Clozapine remains the only evidenced based pharmacotherapy for treatment resistant schizophrenia (TRS) (Mizuno et al. Reference Mizuno, McCutcheon, Brugger and Howes2020, Siskind et al. Reference Siskind, Siskind and Kisely2017, Lally and Gaughran Reference Lally and Gaughran2019, Wagner et al. Reference Wagner, Siafis, Fernando, Falkai, Honer, Röh, Siskind, Leucht and Hasan2021), but remains underused in the 30% of people with a diagnosis of schizophrenia who will meet criteria for treatment resistance (Lally et al. Reference Lally, Ajnakina, Di Forti, Trotta, Demjaha, Kolliakou, Mondelli, Reis Marques, Pariante, Dazzan, Shergil, Howes, David, MacCabe, Gaughran and Murray2016a). Clozapine was approved in the United States (US) and Europe for TRS in 1989 and introduced into clinical practice in Ireland and the UK with strict haematological monitoring programmes in 1990. The Summary of Product Characteristics (SmPC) for clozapine mandating regular FBC monitoring was developed at that time for use in UK and Ireland, and until 2025 remained unchanged in relation to key full blood count (FBC) monitoring requirements (EMA 2025).
There is global variation in haematological monitoring guidelines for clozapine use and application of these. In those countries with national guidelines for clozapine haematological monitoring (n = 102), 90% of countries (n = 92) recommend routine FBC monitoring, with it being mandatory in only 45% of countries (n = 42) (Oloyede et al. Reference Oloyede, Blackman, Whiskey, Bachmann, Dzahini, Shergill, Taylor, McGuire and MacCabe2022a). This suggests variable evidence is used to determine monitoring schedules, or non-evidenced based approaches to monitoring are applied. For example, in 2015, the US Food and Drug Administration (FDA) allowed for continued clozapine treatment with absolute neutrophil count (ANC) 1.0–1.5 × 109/L and treatment interruption when ANC <1.0 × 109/L, along with removal of white cell count (WCC) monitoring (FDA 2015), revisions which were not replicated in Europe at that time.
Europe had remained one the most stringent regions in relation to clozapine FBC monitoring requirements. The recent EMA regulatory revisions for clozapine haematological monitoring (EMA 2025) were prompted by recommendations from a European wide clozapine expert group (Verdoux et al. Reference Verdoux, Bittner, Hasan, Qubad, Wagner, Lepetit, Arrojo-Romero, Bachmann, Beex-Oosterhuis, Bogers, Celofiga, Cohen, de Berardis, de Hert, de Las Cuevas, Ebdrup, Fountoulakis, Guinart, Keating, Kopeček, Lally, Lazáry, Luykx, Maronas Amigo, Molden, Nielsen, O’Donoghue, Oswald, Radulescu, Rohde, Sagud, Sanz, Šimunović Filipčić, Sommer, Taipale, Tiihonen, Tuppurainen, Veerman, Wilkowska, Spina and Schulte2025) and followed regulatory changes in the USA in which FDA approved the removal of a requirement for ANC monitoring during clozapine treatment (FDA 2025).
Prior to the EMA revised recommendations, in Ireland clozapine dispensing required adequate white blood cell (WBC) counts and neutrophil counts and mandated lifelong FBC monitoring.The burden of FBC monitoring, alongside concern about the risk of agranulocytosis is frequently identified as a barrier to clozapine treatment (Farooq et al. Reference Farooq, Choudry, Cohen, Naeem and Ayub2019, Verdoux et al. Reference Verdoux, Quiles, Bachmann and Siskind2018). In an Irish setting, 73% of consultant psychiatrists reported concerns regarding adherence with phlebotomy as a barrier to clozapine initiation, with 57% reporting that less frequent blood monitoring would facilitate clozapine initiation and 31% agreeing that they would advocate for reduced haematological monitoring after 12 months of clozapine and 26% after 6 months (21% reported that they did not agree with reductions in monitoring frequency at any time) (Grant et al. Reference Grant, Mcmanus, Belay, Mahon, Murad, O’ Donoghue and Lally2024). International cohort studies identify that clozapine discontinuation due to non-adherence with blood monitoring occurs in 8–21% of clozapine discontinuation cases (Legge et al. Reference Legge, Hamshere, Hayes, Downs, O’Donovan, Owen, Walters and MacCabe2016, Krivoy et al. Reference Krivoy, Malka, Fischel, Weizman and Valevski2011). The burden of repeat lifelong blood tests is a barrier for clozapine use, thus limiting access to a medication that may have a transformative effect on patient outcomes in TRS.
Epidemiology of clozapine induced agranulocytosis (CIA)
The proposal to revise monitoring is based on data from meta-analyses and epidemiological studies of CIA, highlighting the reduced incidence of CIA with longer duration of clozapine treatment and negligible rates at 1-2 years post clozapine initiation (Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018, Northwood et al. Reference Northwood, Myles, Clark, Every-Palmer, Myles, Kisely, Warren and Siskind2024).
CIA is a rare event. A meta-analysis of 108 studies identified that the maximum incidence of CIA was at one month of treatment (89% of cases with ANC <0.5 × 109 during first month of treatment), with significant declines in incidence thereafter (Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018). The incidence of clozapine associated neutropenia was 3.8% (95% CI: 2.7–5.2%) and severe neutropenia (ANC <0.5 × 109) 0.9% (95% CI: 0.7–1.1%) (Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018). More recent meta-analyses identified CIA rates of 0.47% (Magistri and Melini Reference Magistri and Mellini2023) and 0.5% (Li et al. Reference Li, Zhong, Lu, Zheng, Wang, Rao, Wang, Ng, Ungvari, Wang and Xiang2020), lower than earlier estimates (Alvir et al. Reference Alvir, Lieberman, Safferman, Schwimmer and Schaaf1993, Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018). The risk of CIA after 18 weeks is 0.11% and after 1 year is 0.077% (Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018), equivalent to the agranulocytosis risk associated with phenothiazines (Schulte et al. Reference Schulte, Veerman, Bakker, Bogers, Jongkind and Cohen2024). There is no evidence of change in agranulocytosis incidence before and after the introduction of mandatory FBC monitoring, indirect evidence that monitoring leading to clozapine discontinuation with ANCs 0.5–1.5 × 109/L does not lead to reductions in agranulocytosis (Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018).
These findings are corroborated by registry-based cohort studies. A retrospective cohort study of Australian and New Zealand clozapine patients (n = 26,630 cases with 2.6 million blood counts) identified that peak incidence of CIA was in the first 18 weeks of clozapine treatment (weekly incidence of 0.13%), with median of 9 weeks to time of peak clozapine cessation due to CIA (Northwood et al. Reference Northwood, Myles, Clark, Every-Palmer, Myles, Kisely, Warren and Siskind2024). This study further highlighted the low incidence of CIA at 2 years (0.001%) and the very low incidence of any neutropenic event when clozapine was reintroduced in people with more than two years of monitoring and no history of neutropenia (Northwood et al. Reference Northwood, Myles, Clark, Every-Palmer, Myles, Kisely, Warren and Siskind2024).
In the United Kingdom and Ireland an analysis of data from the national clozapine monitoring service found that the peak incidence of CIA was in the first 6–18 weeks of clozapine treatment (Atkin et al. Reference Atkin, Kendall, Gould, Freeman, Liberman and O’sullivan1996). Registry data from Chile in over 5,000 clozapine treated cases identified that 88% of severe neutropenia cases occurred in the first 18 weeks of clozapine use (Mena et al. Reference Mena, Nachar, Crossley and González-Valderrama2019) – this prompted regulatory change in Chile in 2021 to reduce ANC monitoring frequency after one year of clozapine use and no history of neutropenia.
A Finish nationwide and case control study that assessed comparative risk for CIA compared with non-clozapine antipsychotics, identified that CIA risk decreased over time from an adjusted Odds Ratio (aOR) of 36.01 (95% CI16.79–77.22) for less than 6 months on clozapine to 4.38 (1.86–10.34) for clozapine use of 54 months or more, with a small difference in the number of cases between CIA and non-clozapine antipsychotic agranulocytosis (this was a marginal increased risk of a rare outcome, similar to the risk of agranulocytosis seen with non-clozapine antipsychotic use in the first 6 months of use, for which no monitoring is mandated) (Rubio et al. Reference Rubio, Kane, Tanskanen, Tiihonen and Taipale2024). The absolute cumulative risk of CIA over 22 years follow up was 1.37% (95% CI 0.58–3.16), higher than that seen for non-clozapine antipsychotics (0.13% (95% CI 0.04–0.23). The highest risk for CIA was in the first 6 months, although a residual neutropenia risk persisted after 2 years and it was only after 3 years that the risk of CIA is comparable to that associated with non-clozapine antipsychotics during their first 6 months of use (Rubio et al. Reference Rubio, Kane, Tanskanen, Tiihonen and Taipale2024). In 398 agranulocytosis cases (231 CIA; 167 non-clozapine antipsychotics), case fatality was low, 2.81 per 10,000 clozapine cases, and 0.63 per 10,000 non-clozapine antipsychotic treated cases, and only one person of 3,559 people starting clozapine died because of clozapine-induced agranulocytosis (Rubio et al. Reference Rubio, Kane, Tanskanen, Tiihonen and Taipale2024).
CIA case fatality
Mortality due to CIA is very rare, with case fatality rates in meta-analysis of 2.1% (I in 7,700 clozapine treated cases) (Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018) and overall death of 0.05% in all clozapine treated patients (Li et al. Reference Li, Zhong, Lu, Zheng, Wang, Rao, Wang, Ng, Ungvari, Wang and Xiang2020). Case fatality rates of 2.7 to 3.1% in earlier cohort studies (Alvir et al. Reference Alvir, Lieberman, Safferman, Schwimmer and Schaaf1993, Honigfeld et al. Reference Honigfeld, Arellano, Sethi, Bianchini and Schein1998), are mirrored in analysis of the VigiBase pharmacovigilance worldwide database which identified that probable clozapine associated severe neutropenia was associated with 550 deaths, and a low case fatality rate of 1.6% (De Las Cuevas et al. Reference De Las Cuevas, Sanz and De Leon2024). It is apparent that the risk of CIA, and the risk due to CIA is less than previously thought, though the barrier to clozapine use due to intensive blood monitoring or non-adherence with monitoring continues to restrict access to the only evidenced based treatment in TRS (Lally and Gaughran Reference Lally and Gaughran2019), and to a treatment associated with reductions in all-cause mortality and suicide mortality compared to other antipsychotic use (Taipale et al. Reference Taipale, Tanskanen, Mehtälä, Vattulainen, Correll and Tiihonen2020, Vermeulen et al. Reference Vermeulen, van Rooijen, van de Kerkhof, Sutterland, Correll and de Haan2019).
Clozapine associated benign neutropenia
It may be largely unrecognised among psychiatrists, that transient neutropenia occurs in the general population, with natural background rates of neutropenia (ANC <1.5 × 109/L) of 4.5% in those of Black ethnicity and 0.8% in those of white ethnicity (Hsieh et al. Reference Hsieh, Everhart, Byrd-Holt, Tisdale and Rodgers2007). Observational study data identified neutropenia rates of 0.81% (n = 136) in the 30 days prior to clozapine use (de Leon et al. Reference de Leon, Baldessarini, Balon, Bilbily, Caroff, Citrome, Correll, Cotes, Davis, DeLisi, Faden, Freudenreich, Goldsmith, Gurrera, Josiassen, Kane, Kelly, Keshavan, Laitman, Lam, Leung, Love, McCollum, McGrane, Meyer, Nasrallah, Nucifora, Rothschild, Rubio, Sajatovic, Sarpal, Schoretsanitis, Shad, Shelton, Sher, Singh, Surya, Zarzar, Sanz and De las Cuevas2025). Many of the cases of neutropenia associated with clozapine use, are just that, coincidental associations with other causal mechanisms, and are not consistent with CIN or CIA. The stringent routine monitoring for clozapine increases the likelihood of detecting random, benign transient fluctuations in ANC, or ANC reductions unrelated to clozapine-this is of particular concern for those on longer term clozapine treatment, beyond the high risk period of the first 18 weeks, and certainly beyond one year of clozapine use (Oloyede et al. Reference Oloyede, Casetta, Dzahini, Segev, Gaughran, Shergill, Mijovic, Helthuis, Whiskey, MacCabe and Taylor2021, Taylor et al. Reference Taylor, Vallianatou, Whiskey, Dzahini and MacCabe2022). The use of neutrophil count thresholds to define CIN contributes to overdiagnosis of CIN, with the result that many patients have unnecessary clozapine discontinuation (Oloyede et al. Reference Oloyede, Whiskey, Casetta, Dzahini, Dunnett, Gandhi, Gaughran, Shergill, McGuire, MacCabe and Taylor2022b).
The impact and safety of reduced frequency of FBC monitoring in those on clozapine for >1 year was highlighted in a UK mirror image study, which found no significant difference between rates of neutropenia between patients monitored every 12 weeks (n = 459) and those monitored monthly (n = 110) during the COVID-19 pandemic (Oloyede et al. Reference Oloyede, Dzahini, Abolou, Gee, Whiskey, Malhotra, Hussain, Osborne, Casetta, Mcguire, Maccabe and Taylor2023). A larger study from the same group demonstrated the safety of 3 monthly FBC monitoring in clozapine cases treated for longer than 1 year (n = 1025); there were no agranulocytosis cases or related deaths identified during 12 weekly FBC monitoring over a median follow up of 4 years (IQR = 1), while 9 cases had ANC <1.5 × 109/L with none progressing to agranulocytosis (Taylor et al. Reference Taylor, Gee, Helthuis and Oloyede2025).
CIA is a defined non-transient phenomenon
Despite this, for a generation or more of psychiatrists, it was assumed that a neutrophil count of <1.5 × 109/L, but >0.5 × 109/L was equivalent to, or a risk for agranulocytosis, leading to clozapine discontinuation and non-rechallenge. An ANC <0.5 × 109/L is consistent with severe neutropenia or agranulocytosis and may be associated with an increased risk of opportunistic infection, whereas a neutrophil count of 1–1.5 × 109/L in a non-immunocompromised patient is not (Lally and Flanagan Reference Lally, Flanagan, Manu, Flanagan and Ronaldson2016b).
CIA is associated with sustained neutropenia with a significantly higher risk of infection and mortality. However, the majority (75%) of mild neutropenia (1.0–1.5 × 109/L) cases do not progress to CIA (Myles et al. Reference Myles, Myles, Xia, Large, Kisely, Galletly, Bird and Siskind2018). This was corroborated by an Icelandic cohort study that found only 2.9% (1 out of 34 cases) who developed neutropenia progressed to CIA (Ingimarsson et al. Reference Ingimarsson, MacCabe, Haraldsson, Jónsdóttir and Sigurdsson2016).
It is important that clinicians recognise clozapine induced neutropenia to be a distinct phenomenon, one associated with agranulocytosis, that is a non-transient event and one that occurs most commonly in the first 18 weeks of treatment. It is associated with a sudden and precipitous drop within 14 (or 7) days of ANC to below 0.5 × 109/L (Taylor et al. Reference Taylor, Vallianatou, Whiskey, Dzahini and MacCabe2022), and with prolonged neutropenia (not resolving in 2–4 days) (Lally and Flanagan Reference Lally, Flanagan, Manu, Flanagan and Ronaldson2016b). In CIA, ANCs will decrease further even with clozapine cessation. The median duration to improvement of ANC >0.5 × 109/L is 4 days, and longer (10 days) to attain ANC >1.5 ×109/L (Matsui et al. Reference Matsui, Ishibashi, Kawano, Oshibuchi, Ishigooka, Nishimura and Inada2020). The speed of onset of CIA limits the usefulness of FBC monitoring at 2-or 4-week intervals in identifying CIA cases.
CIA risk remains and new monitoring recommendations
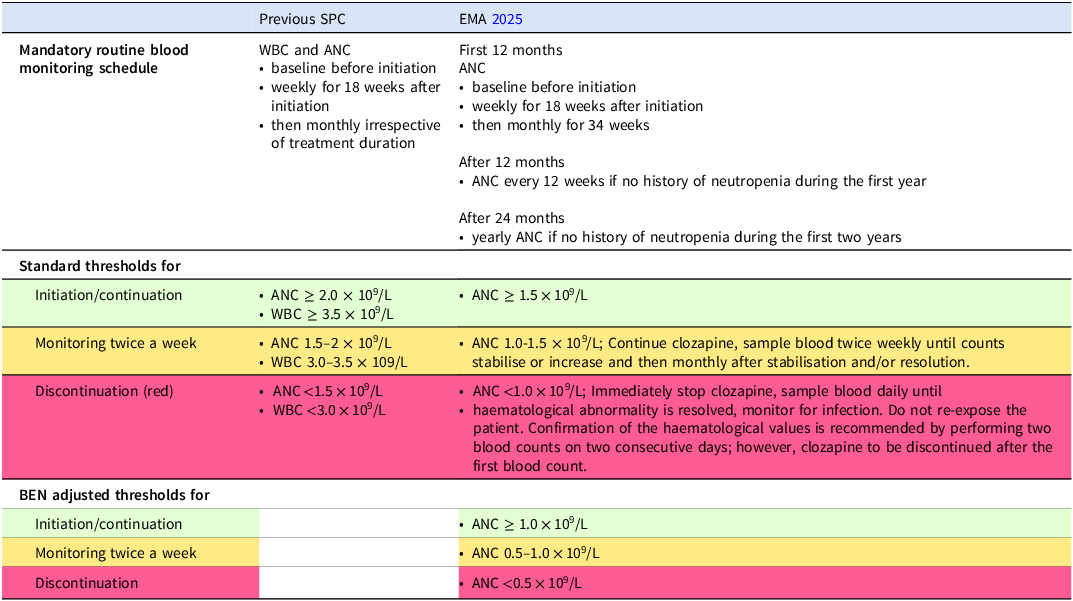
The risk of severe neutropenia with clozapine use remains, though this risk is less than previously thought, and primarily occurs in the early phase of clozapine treatment. There remains a requirement for vigilant weekly ANC monitoring in this high-risk period covering the first 18 weeks of clozapine use as recommended by the EMA (Table 1). Subsequent to this, and dependent on individual factors (such as history of neutropenia (<1.5 × 109/L)), monitoring can be reduced to monthly (rather than 2 weekly) for the next 34 weeks, and 3 monthly during year two, followed by annual monitoring thereafter in patients who have no prior history of neutropenia (ANC <1.5 × 109/L). The recommendation for annual monitoring after year 2 is less related to a risk for CIA but may provide a means to monitor for haematological malignancies in clozapine treated cases (although the absolute risk for this remains small) (Chrétien et al. Reference Chrétien, Lelong-Boulouard, Chantepie, Sassier, Bertho, Brazo, Humbert, Alexandre, Fedrizzi and Dolladille2021, Tiihonen et al., Reference Tiihonen, Tanskanen, Bell, Dawson, Kataja and Taipale2022). These recommendations are based on proportionate responses to the diminishing risk of CIA with longer treatment, while reducing the burden of unnecessary intensive blood monitoring and potentially increasing accessibility to, and adherence with clozapine.
EMA clozapine monitoring requirements

Further, monitoring is now based on ANC only, mirroring evidence that ANC is a more specific and clinically important marker for infection risk. There is no longer a need for WBC monitoring. Thresholds for ANC have changed also, with recommendations to continue with clozapine with twice weekly ANC monitoring for ANC 1.0–1.5 × 109/L, and discontinuation of clozapine with ANC below 1.0 × 109/L. In patients with benign ethnic neutropenia (BEN), clozapine initiation is recommended in cases with ANC >1.0 × 109/L. People with BEN meet criteria for clozapine discontinuation with ANC <0.5 × 109/L (rather than <1.0 × 109/L) – the reduced ANC threshold for BEN cases prevents unnecessary clozapine discontinuation and does not impact those patients’ safety. Recommendations in relation to monitoring after treatment interruptions are summarised in Table 2. The use of concurrent medications with neutropenic risk such as sodium valproate (Malik et al. Reference Malik, Lally, Ajnakina, Pritchard, Krivoy, Gaughran, Shetty, Flanagan and MacCabe2018) merit consideration for reinstating more regular ANC monitoring during the initiation period-though clearer recommendations on this remain to be made.
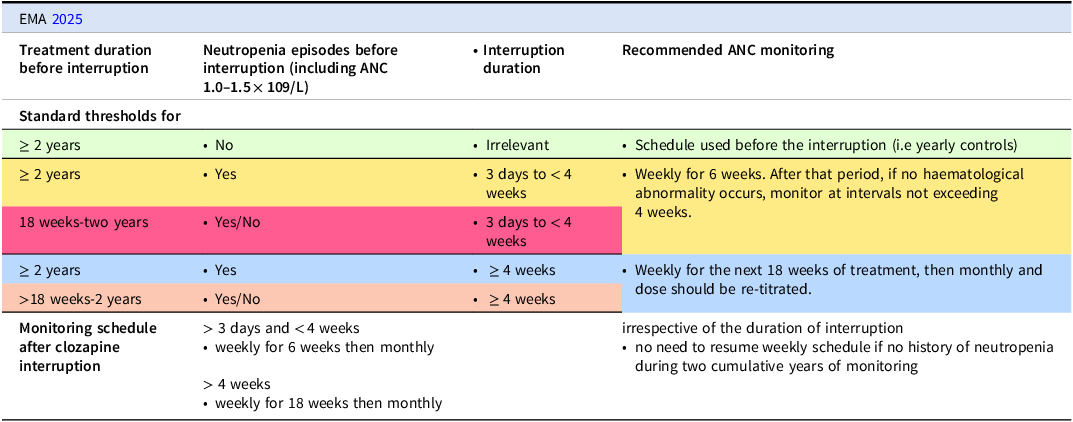
ANC monitoring upon resuming clozapine after treatment interruption for other reasons (not haematological)

Lived experience of clozapine use and blood monitoring (by Rita Bourke-An individual with lived experience, and named author of this paper).
I was put on Clozapine for TRS as all other medications had failed. When I started clozapine, it was a very difficult time for me and all the blood tests and monitoring of my vital signs caused me a lot of emotional turmoil. I think the changes to reduce monitoring to three monthly during year two and then yearly after that will be a great success as service users will be more compliant with less blood tests and monitoring. I think the reduced blood tests will make a big difference for people in employment, as they will need to get less time off work and also for those who live in remote areas, as they will need to travel less.
I didn’t mind the actual blood tests however when I was taking clozapine at the start, I worried a lot that I would develop neutropenia and would have to then stop my clozapine. I think these new rules will lead to more people deciding to take clozapine, as it won’t be as much of an inconvenience in the longer term.
It will be important to appreciate that some service users might be very anxious about blood monitoring changing from monthly to annually. They might question how accurate an annual blood test is compared to monthly test so a lot of reassurance and careful explanation needs to be given; this should be an understandable explanation, not one that is overly medical, about why this change is happening.There is a potential downside of not seeing the team as frequently, even for routine blood tests and having informal check ins with the nurse. There are ways around that issue, such as the person having a key worker or community nurse, who can check in with the service user taking clozapine at regular intervals. However, I believe that retaining the clozapine nurse and clinics is important-clinics may function differently, but ensuring that they continue will be crucial to my care. Even if the blood tests are now only annual, clozapine nurses are still needed to provide continuity of care which is so important to me as a clozapine user. Service users also might have a strong bond with their clozapine nurse and open up to them more than other mental health nurses or team members. Clozapine nurses and the clozapine clinic setting can provide ongoing support to encourage adherence. I think this is the most important as many service users don’t adhere to medication I did adhere to medication as I am a nurse but many do not. If possible, the clozapine nurse could continue to call or meet the service user monthly for a check in, as I know from experience I always discussed my problems at my monthly blood tests and if I was going through a hard time I had reassurance I could speak about these problems at my monthly check blood tests so this kept me well mentally if I was going through a hard time. The clozapine nurse and clinic can change to provide more holistic care and support.The following roles still need to be done by a clozapine clinic even if it is only annual blood tests that are now mandatory.Keeping clozapine nurses will allow for more mental health and physical health care checks and support. To monitor for side effects such as sedation, constipation, tachycardia, signs of infection or signs of psychosis. Support with adherence, and educating patients and their families about clozapine, this is ongoing, as service users can often ask the same question over and over again and need reassurance like I have in the past. Ensuring all patient data and monitoring results are accurately recorded and communicated. Liaising closely with patients, their carer’s, community teams, GPs, and psychiatrists for best care outcome. Follow local protocols for emergency management (e.g., stopping clozapine, arranging urgent medical review).
The service users care plan is very important for this transition to annual blood tests so needs to be updated regularly with service users input and best plan of care to keep the service user well and what needs to be put in place if service user does become unwell. If clozapine clinics are not kept in place due to no longer having monthly blood tests, the opportunity to talk to a clozapine nurse may be lost and you might go a long time without seeing a mental health professional.
This just my opinion. I really think the only downside to annual blood tests is the possible loss of the monthly check in with the nurses. As I have said before if I was having problems or going through a hard time, I always reassured myself I could talk to a nurse about it at my blood appointment and I used this as a skill to keep myself well mentally. I never saw the clozapine clinic as only a place to get my bloods done, it provides important support and continuity of care for me as a clozapine user and should continue to be available on a monthly or regular basis.
Education regarding new monitoring
Clinician and patient education about the changes is an important component. The need for patient and carer education in relation to signs and symptoms of infection due to neutropenia remains unchanged. Specific education about, and clinical assessment for signs and symptoms including fever, flu-like symptoms, mouth ulcers, sore throat, gingivitis, sinusitis, respiratory, or gastrointestinal symptoms are important. Provisions for providing rapid ANC monitoring in those cases with symptoms concerning for infection are required. The development and validity of point-of-care-testing (POCT) for ANC/FBC monitoring in clozapine care is an important step in this respect (Taylor et al. Reference Taylor, Atkins, Harland, Baburina, MacCabe, Salamone and McGuire2021). A key component of adjustment to the new monitoring regimen will be maintaining the established regular clinical contact with clozapine patients, which previously occurred monthly for those on clozapine longer than 1 year. It would seem prudent for services to evolve to ensure that the supportive role of clozapine nurses and other team members through regular monthly contact is offered and sustained. Concerns exist that the regular clinical contact may be lost and that this could pose risks to patients-however, this can be managed on a case by case basis, allowing some to continue to attend monthly for prescriptions and clinical support, while others with established clinical stability may have a preference for less than monthly contact, and gain more benefit from more comprehensive review appointments on a 3 or 6 monthly basis.
In Ireland, the change to annual monitoring after 2 years may allow for clozapine to be changed from a hospital only medicine and provided with a GMS number to allow for it to be dispensed by community pharmacies after two years. This has the potential to increase capacity in the healthcare system for more clozapine to be initiated in community mental health teams, allowing it to be more widely prescribed and dispensed. Challenges for people in travelling to clozapine clinic sites may be a limiting factor in clozapine accessibility, and particularly in rural settings where people often have to travel long distances to clozapine clinics; community pharmacies are more accessible, thus supporting wider clozapine use.
The significant reduction in the blood monitoring requirements will save clinical services both time and money, however we advocate that the time saved should be focused on more important mental health, and physical health issues for people taking clozapine, more robust side effect monitoring, and specifically prevention or management of obesity, supporting increased uptake in vaccinations and proactive management of clozapine-induced gastrointestinal hypomotility. Addressing these three factors could improve the morbidity and mortality rates in this population, as ileus and pneumonia are associated with early mortality (Partanen et al. Reference Partanen, Häppölä, Kämpe, Ahola-Olli, Hellsten, Rask, Haaki, Hietala, Kampman, Tiihonen, Tanskanen, Ripatti, Palotie, Taipale, Lähteenvuo and Koskela2024) and newer medications for the management of obesity have been demonstrated to be effective, tolerable and safe in those taking clozapine (Siskind et al. Reference Siskind, Baker, Arnautovska, Warren, Russell, DeMonte, Halstead, Iyer, Korman, McKeon, Medland, Parker, Stedman and Trott2025). Providing increased mental health care, with increased time spent addressing suboptimal clozapine response, monitoring for and management of comorbid mental disorders, including depression, increased focus on managing negative and cognitive symptoms, and more time to identify recovery focused interventions such as educational, vocational training, employment, and social activities.
Conclusion
The accumulated scientific understanding of CIA indicate that the past monitoring regulations were overly stringent, were disproportionate to the risks associated with clozapine use and timing and incidence of agranulocytosis, and impeded clozapine use. Further, transient neutropenic episodes detected by intensive blood monitoring may be linked to other non-clozapine causes, as seen in the general population, leading to unnecessary clozapine discontinuation and non-rechallenge status. This led to the recent EMA recommendations for revised clozapine haematological monitoring-these are safe, evidenced based recommendations, allowing for less stringent long-term monitoring. This can reduce logistic burdens on patients and services and may increase uptake and continuation of clozapine for eligible patients – an important benefit given clozapine’s superior effectiveness for TRS.
Acknowledgements
The authors declare no conflict of interest.
Author contributions
JL developed the concept for this research. JL completed the first draft. OC, RB, DK, BOD participated in substantive reviews of the manuscript. The authors read and approved the final manuscript.
Funding statement
This work is supported by the Health Research Board (grant no. APRO-2023-005). The views of the funding body have not influenced the research or the content of systematic review.
Competing interests
The authors declare none.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committee on human experimentation with the Helsinki Declaration of 1975, as revised in 2008.