Introduction
Invasive pest species are plant-eating pests that invade new areas with environmental conditions and food sources similar to those of their native regions, but they usually have few or no natural enemies, which helps them establish themselves and grow their populations (Wainhouse, Reference Wainhouse2005). However, the conditions in the regions where they have arrived and successfully established themselves are changing much faster than expected because of climate change (CC) and its various environmental impacts. In the Mediterranean region, CC causes rainfall regimes with altered frequency and intensity, followed by prolonged droughts, increased CO2 and greenhouse gases, and increases in both maximum and minimum temperatures (Cramer et al., Reference Cramer, Guiot, Marini, Azzopardi, Balzan, Cherif, Doblas-Miranda, Dos-Santos, Drobinski, Fader, Hassoun, Giupponi, Koubi, Lange, Lionello, Llasat, Moncada, Mrabet, Paz and Xoplaki2020).
The boxwood moth Cydalima perspectalis (Walker, 1859) (Lepidoptera: Crambidae), originally from East-Asia, was first detected in Europe in 2007 and later in the Mediterranean forests of northeastern Spain in 2018. This phytophagous insect has spread rapidly through the northeastern Pyrenees, moving from east to west and destroying and even killing centuries-old natural boxwood (Bassols and Oliveras, Reference Bassols and Oliveras2014; López and Eizaguirre, Reference López and Eizaguirre2019). From its initial detection to approximately 2021, the spread was exponential. However, since then, the rate of expansion has slowed (Fraile, Reference Fraile2024), which could be due to less favourable humidity and temperature conditions in recent years, likely linked to CC.
In phytophagous insects, CC can directly influence development, survival, or phenology, or indirectly through the plants they feed on or via natural enemies, which are among the most common factors (Björkman and Niemelä, Reference Björkman and Niemelä2015). In the Mediterranean region, the effect of CC on the boxwood moth C. perspectalis through natural enemies is unlikely to be significant, as few natural enemies capable of controlling it have been identified to date (Oltean et al., Reference Oltean, Ulujan, Hulujan, Varga, Totos and Florian2017; López et al., Reference López, Las Heras, Garrido-Jurado, Quesada-Moraga and Eizaguirre2022).
However, the effect of CC on plants can be significant because C. perspectalis is a monophagous species that lays eggs, feeds, and pupates exclusively on plants of the genus Buxus. In Europe and Iran, boxwood is a natural forest ecosystem that is threatened by various stresses related to CC (Alipour and Walas, Reference Alipour and Walas2023). de Jong et al. (Reference de Jong, Addink, Hoogenboom and Nijland2012) studied how the leaves of Buxus sempervirens L. (Buxales, Buxacea) responded to five different stress signals and showed the strongest response in plants subjected to drought and combined drought and heat, which caused leaf curling, shrinking, decolourisation, brittleness, canopy opening, and branch sagging. Other studies have aimed to predict how CC affects the present and future distribution and survival of boxwood by creating mathematical models (Alipour and Walas, Reference Alipour and Walas2023) or distribution models that include plants, pests, and fires (Canelles et al., Reference Canelles, Aquilué and Brotons2022).
The effects of rising temperatures and drought on boxwood influence C. perspectalis depending on how each insect stage interacts with the plant. Females are likely to use plant colour and canopy openness as signals to select optimal plants for laying eggs, with offspring survival possibly dependent on leaf humidity. Larvae, especially newly hatched larvae, had higher survival rates on turgid leaves than on curled or brittle leaves. The condition of the plant does not affect the pupae, which do not feed, or the adults, which are capable of strong flight and feeding on nectar from various plant genera. However, increased temperatures, whether the daily maximum or minimum, can affect the flight, survival, and mating abilities of adults. Additionally, feeding on poorly conditioned or senescent leaves may prompt earlier diapause induction or a greater proportion of diapausing larvae. An important factor in the insect-plant relationship is the location of boxwood plants, either in sunny or shady areas. In Mediterranean forests, during extreme summer temperatures over the past 3 or 4 years, the most significant C. perspectalis attacks occurred in shady areas, while sunny areas were less affected, despite boxwood being in worse condition. The complex interactions among various ecosystem factors require further research into individual effects and their interactions with other factors, as Canelles et al. (Reference Canelles, Aquilué and Brotons2022) have already begun to explore.
The direct impact of CC on insects has been examined in several reviews (Ward and Masters, Reference Ward and Masters2007; Harvey et al., Reference Harvey, Heinen, Gols and Thakur2020, Reference Harvey, Tougeron, Gols, Heinen, Abarca, Abram, Basset, Berg, Boggs, Brodeur and Cardoso2023). It is generally believed that CC negatively affects insect diversity (Harvey et al., Reference Harvey, Heinen, Gols and Thakur2020). However, in other insect pests, it is assumed that CC promotes their distribution, expansion, and colonisation of new niches (Björkman and Niemelä, Reference Björkman and Niemelä2015; Skendžić et al., Reference Skendžić, Zovko, Živković, Lešić and Lemić2021; Schneider et al., Reference Schneider, Rebetez and Rasmann2022; Subedi et al., Reference Subedi, Poudel and Aryal2023). Bale et al. (Reference Bale, Masters, Hodkinson, Awmack, Bezemer, Brown, Butterfield, Buse, Coulson, Farrar, Good, Harrington, Hartley, Jones, Lindroth, Press, Symrnioudis, Watt and Whittaker2022) showed the role of temperature in winter survival in temperate regions. However, the upper thermal limits of development, which could be crucial in Mediterranean areas, are less frequently evaluated (Buckley, Reference Buckley2022). Bale et al. (Reference Bale, Masters, Hodkinson, Awmack, Bezemer, Brown, Butterfield, Buse, Coulson, Farrar, Good, Harrington, Hartley, Jones, Lindroth, Press, Symrnioudis, Watt and Whittaker2022) also emphasised the importance of the photoperiod as a key signal in seasonal synchronisation, diapause induction, and voltinism. Other authors, as mentioned in the Harvey et al. (Reference Harvey, Heinen, Gols and Thakur2020) review, also suggested that the effect of rising temperatures varies depending on the developmental stage of the insect.
The increase in temperature due to CC in the study area could affect the number of generations the species experiences. This temperature increase may also affect the timing of the first adult flight from overwintering larvae, leading to earlier adult emergence (diapause completion and post-diapause development) during warmer years. Additionally, higher temperatures could alter the developmental rate of first- and second-generation larvae in the field (after wintering), potentially causing third-generation flights to occur before the critical photoperiod that induces diapause. Alternatively, it might affect second-generation larvae so that they no longer respond to the critical photoperiod when it occurs, meaning they would skip diapause. In this scenario, the third adult flight in autumn was significantly greater. Furthermore, increasing temperatures can alter how larvae respond to a critical photoperiod, delaying diapause induction in a larger proportion of larvae.
Therefore, this study aimed to predict the response of this pest to rising temperatures associated with CC in southern Europe, where boxwood is an endangered and ecologically important component of the Mediterranean forests. We examined how temperature affects the development and survival of C. perspectalis across multiple life stages under controlled laboratory conditions, while holding other variables, such as substrate humidity and light exposure, constant. We evaluated whether elevated temperatures influence diapause induction in autumnal larvae, potentially enabling additional generations in the region. We assessed the relative influence of thermal conditions and altitude on the timing of the first flight emergence in the field.
Material and methods
Insect rearing
To establish a laboratory population of C. perspectalis, larvae were collected on different dates in 2022, 2023, 2024, and 2025 from two different forest boxwood areas at Solsones, Catalonia, Spain (41°55′03.578″ N, 1°29′04.228″ E and 41°57′00.600″ N, 1°31′04.200″ E). All experiments were performed in climatic chambers with controlled temperature and photoperiod. Larvae were fed a semi-artificial diet (López et al., Reference López, Las Heras, Garrido-Jurado, Quesada-Moraga and Eizaguirre2022) and kept in a breeding climatic chamber at 25°C, a 16:8 (light:dark) photoperiod, and 70% humidity until pupation. The newly formed pupae were kept in a climatic chamber at 10°C and a 0:24 (light:dark) photoperiod to synchronise their development and prevent the adults of one sex from emerging before those of the other. When the desired number of males and females for each mating cage was reached, the pupae were separated by sex and placed in breeding chambers. Upon adult emergence, at least five pairs of same-age males and females were placed in each mating cage (50 cm3 methacrylate boxes, with three 20 cm2 windows covered in tulle) at 25°C under a 16:8 (light:dark) photoperiod and provided with a 5% sugar solution for feeding. Several B. sempervirens sprigs in small containers with water were added to the mating cages as laying substrates. Every day, sprigs were inspected for eggs, and leaves with eggs were removed for use in different experiments.
To maintain overall rearing after hatching, neonate larvae were placed in small containers (5 cm diameter, 3 cm height) arranged in a five-by-five grid, with a piece of semi-artificial diet (López et al., Reference López, Las Heras, Garrido-Jurado, Quesada-Moraga and Eizaguirre2022) approximately 1 cm3 in size, and used in different experiments.
All experiments were conducted to isolate the effects of temperature by preventing drought stress through daily diet changes in small boxes or by adding hydroponic gardening sponges to the mating cages.
The experimental temperatures were chosen according to the usual temperatures from April to September in the study area. The highest temperatures correspond to multi-day heat waves that have occurred in recent years.
Developmental rate and mortality at different temperatures
Eggs
Sprigs in the breeding boxes were checked daily to confirm egg presence. When eggs were found, the leaves were removed from the sprigs and placed in a small container (5 cm in diameter, 3 cm high) containing an agar-agar solution (10:1), similar to those used for larval rearing. This solidified agar solution helped the leaves to retain humidity in a sterile environment, preventing fungal growth. Consequently, the leaves remained fresh enough for the eggs to survive and hatch, even at the highest temperatures. The leaves with egg masses were placed in climatic chambers set at different temperatures (15, 17, 19, 21, 23, 25, 27, 30, and 35°C) with a 16:8 (light:dark) photoperiod; the solidified agar ensured proper humidity in each container. More than nine egg masses, each containing at least 65 eggs, were placed in each temperature chamber. The eggs were checked daily for hatching, and developmental time and mortality rates were recorded. After hatching, the larvae were used for subsequent experiments.
Larvae
To determine the developmental duration of the larvae and mortality rates at different temperatures (15, 17, 19, 21, 23, 25, 27, 30, and 35°C) under a 16:8 (light:dark) photoperiod, newly hatched larvae (<24 h) were isolated with a piece of diet in small containers (5 cm in diameter, 3 cm high) for diet. The larvae were checked daily until pupation, and their diet was changed every 1–2 days to maintain proper humidity at higher temperatures. A minimum of 50 larvae was placed in each chamber. To improve pupation, when larvae reached the L5 instar, the semi-artificial diet was supplemented with small Buxus sprigs, and the diet was checked daily to prevent fungal contamination. When necessary, the rearing containers and diets were changed.
Pupae
When the pupae from the previous experiments emerged, the new pupae were sexed and placed in a clean rearing container with 1 cm3 of Sesamia’ diet (Eizaguirre and Albajes, Reference Eizaguirre and Albajes1992) as a humectant. This diet provided a suitable humidity for pupal development under sterile conditions. Similar to the larvae, at least 50 pupae, less than 24 h old, were placed in each temperature chamber.
Adults
Seven pairs of males and females were housed in each mating cage within chamber rooms maintained at 15, 20, 25, 30, or 33°C, with a 16:8 (light:dark) cycle. Three replicates were analysed at each temperature. Each cage was supplied with a 5% sugar solution for adult feeding, B. sempervirens branches as laying substrates, and garden sponges to help maintain proper humidity. Every day, the eggs were checked for sprigs and leaves containing eggs were removed. The number of days to the first egg, peak laying period, and total egg production were recorded. Additionally, adult lifespan, female mating status, and overall condition were documented.
Determination of the sensitive larval instar to diapause induction
To identify the larval stage at which diapause occurred, approximately 500 newly hatched larvae (<24 h old) were divided into two groups. One group was exposed to a diapause-inducing 12:12 h (light:dark) photoperiod, whereas the other group was exposed to a non-diapause-inducing 16:8 h (light:dark) photoperiod. Both groups were maintained at 25°C. Immediately after each ecdysis, when the cephalic capsule of the previous instar was still visible, 40 larvae from each group were transferred between the two photoperiods (table 1). At least 40 larvae were maintained under each photoperiod (inducing or non-inducing diapause) during development. The number of larvae that formed cocoons (diapausing larvae) was recorded for each condition.
Table 1. Percentage of Cydalima perspectalis larvae that entered diapause according to the number of instars developed under either non-diapausing (16:8, light:dark photoperiod) or diapausing (12:12, light:dark photoperiod) conditions at 25°C

Note: Groups sharing the same letter are significantly similar in diapause percentage.
Diapause induction under high temperatures
To determine whether high temperatures reduce diapause induction, newly hatched larvae (<24 h old) were divided into five groups of at least 50 larvae each based on temperature. They were then exposed to a 12:12 (light:dark) photoperiod, which induces diapause, at five temperatures during development, 23, 25, 27, 30, and 33°C. The number of larvae that pupated (non-diapausing) and the final mortality rate at each temperature were recorded.
Emergence of first flight adults (termination and post-diapause period)
C. perspectalis was detected in the study area in 2014. By 2018, the Catalan Government had established a pest monitoring network of pheromone traps across multiple locations at various altitudes (ranging from 433 m to 1405 m) in Catalonia, where boxwood is an important part of the natural forest.
For this study, locations with consistent capture data from 2018 to 2023 were included. As a result, catches from 18 locations over 6 years, with very diverse local climates, were analysed.
For each location and year, it was assumed that the day the first adult was captured marked the end of the diapause-post-diapause period (calendar day). Additionally, the thermal time (degree days, DGDs) accumulated from January 1 to that day was calculated using a threshold temperature of 9.5°C (López et al., Reference López, Las Heras, Garrido-Jurado, Quesada-Moraga and Eizaguirre2022).
Therefore, the variables included in the statistical analysis for each location and year were location altitude, calendar day (from 1 January until the first capture of C. perspectalis in the pheromone trap), and the DGD accumulated on that day. Correlation indices and coefficients of variation were calculated using these variables.
Statistical evaluation of results
Statistical analyses were performed using JMP® Pro 18 (2025).
In the developmental experiments, in addition to recording the variables mentioned above, development rates were also calculated. One-way analysis of variance was used to assess the effect of temperature on developmental rates, and, if necessary, Tukey’s HSD test was employed to compare the means.
An approximation to the binomial (Zar, Reference Zar1999) was used to compare (a) the mortality of each life stage exposed to different temperatures and (b) the larval stage during which diapause occurred by comparing the cocoons and pupae of each photoperiodic condition.
To clarify the factors that influence post-diapause emergence under field conditions, correlation indices and coefficients of variation for the variables studied were computed.
Analysis of covariance was used to examine the relationships among response variables, calendar day (days from January 1st), and thermal time, measured on growing-degree days with a base temperature of 9.5°C. A heterogeneity-of-slopes model (with altitude as a continuous variable, year as a categorical variable, and their interaction) was constructed using JMP® 17 (2025).
Results
Developmental rates and mortality at various temperatures
The rate of egg development increased linearly with temperature, without reaching a maximum value. However, the larval development rate increases with temperature, peaking at 25°C, then declines at higher temperatures. The maximum pupal development rate occurred at 30°C (fig. 1A).

Figure 1. Development rate (A) and mortality (B) of eggs (A1 and B1), larvae (A2 and B2), and pupae (A3 and B3) of Cydalima perspectalis reared at different temperatures under a 16:8 (light: dark) photoperiod.
The effect of temperature on mortality varied across the three developmental stages (fig. 1B). Egg mortality seems to be relatively unaffected by temperature; it is minimal at 23°C but approaches 50% at 35°C. Larval mortality appears to be much more strongly influenced by temperature, both the lower temperatures tested (15 and 17°C) and the higher temperatures (30 and 35°C) result in high mortality, with 70–80% of larvae dying at the highest temperatures. Pupal mortality at the lowest temperatures (15, 17, and 19°C) and at 35°C approaches 50%, while temperatures between 21°C and 27°C cause very low pupal mortality.
Longevity and fertility of adults across different temperatures
Adult longevity decreased with increasing temperature, from a maximum of 21 days at 20°C, with males living longer, to a minimum of 9 days at 33°C for both males and females (fig. 2). Figures 2 and 3 do not show results for 15°C because, 30 days after the experiment began, some adults were still alive, but none of the tested females were mated, and those still alive did not lay eggs.

Figure 2. Longevity of adult males and females of C. perspectalis at temperatures of 20, 25, 30, and 33°C, with a 16:8 (light:dark) photoperiod.

Figure 3. Average number of mated females (A) and eggs per female (B) of C. perspectalis at 20, 25, 30, and 33°C temperatures under a 16:8 (light: dark) photoperiod.
There were no differences in the percentage of mated females between 20°C and 30°C. At 33°C, only one female out of three replicates was mated (fig. 3A). However, the number of eggs per female was higher in females exposed to 25°C than in females exposed to 20°C, 30°C, or 33°C (fig. 3B).
Sensitive larval instar for diapause induction
The first instars are essential for diapause induction (table 1). If the first instar, L1, is completed under non-diapause conditions and the later instars are completed under inducing conditions (Group 2), 65% of the larvae enter diapause. If both L1 and L2 were completed under non-inducing conditions and the remaining instars were completed under inducing conditions (Group 3), only 20% of the larvae entered diapause. When three or more instars were completed under non-inducing conditions (groups 4, 5, 6, and 7), diapause was typically not induced. However, the 5% diapause observed in Group 6 may reflect species plasticity in the diapause response. Conversely, if only the first instar occurred under inducing conditions and the rest occurred under non-inducing conditions (Group 8), diapause was entirely suppressed. However, if two (Group 9) or three (Group 10) instars were completed under inducing conditions and the rest were completed under non-inducing conditions, diapause was not prevented in 10% or 60% of the larvae, respectively. If larvae completed four or more instars under inducing conditions (groups 11, 12, and 13), they maintained diapause.
Diapause induction at short day lengths (12:12, light:dark) and high temperatures
During the inducing photoperiod (12:12, light:dark, photoperiod), high temperatures did not prevent diapause induction, as none of the larvae pupated at any of the temperatures tested, although some formed cocoons. At the highest temperatures (30 and 35°C), most larvae died, as had already occurred in the test prior to the long 16:8 light:dark photoperiod (fig. 4).

Figure 4. Percentage of C. perspectalis larvae that enter diapause or die under short photoperiod (12:12, light:dark) and at high temperatures.
Emergence of first flight adults (termination and post-diapause period)
The first independent variable fitted the data better than thermal time (R 2 = 65% vs. 21%, table 2).
Table 2. Results of the analysis of covariance for calendar day, days from the first of the year to the first moth of Cydalima perspectalis captured in the pheromone trap, at each location each year, and thermal time or DGD accumulated on that day

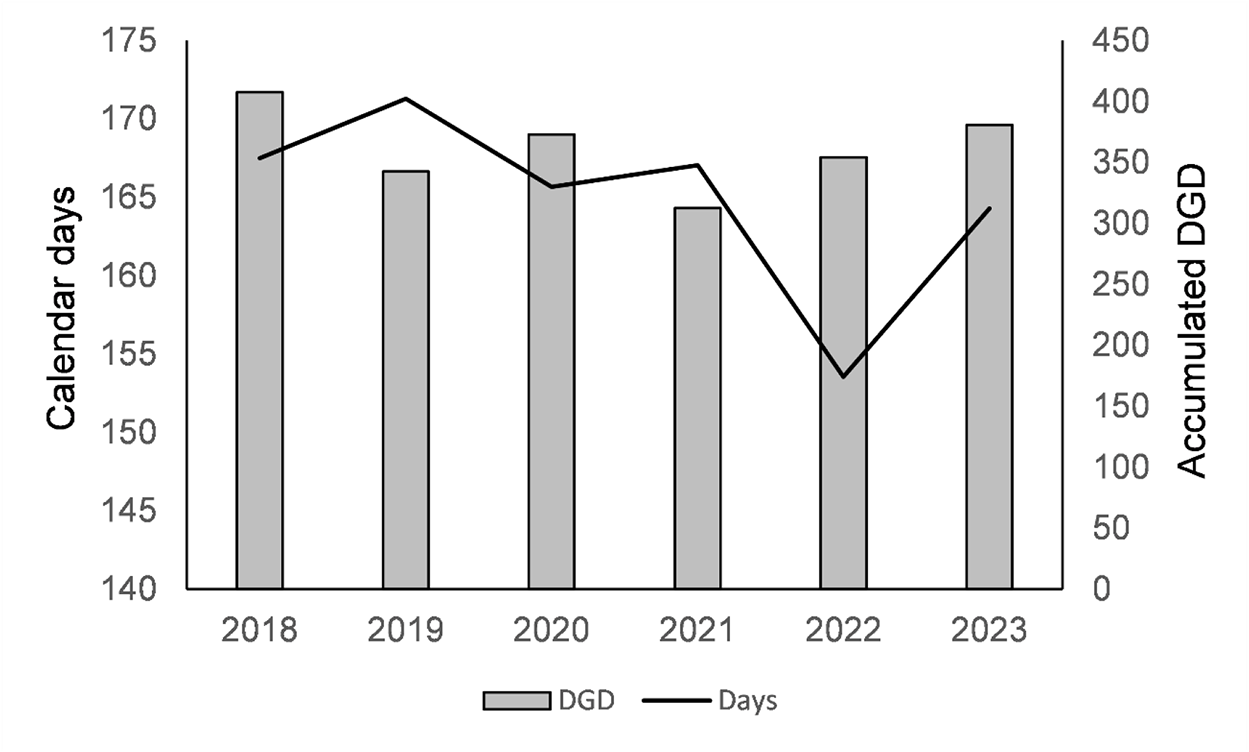
The calendar days for the first flight (fig. 5A) were strongly linked to altitude, accounting for 45% of the total variability, with most of the remaining differences explained by the model (semi-partial R 2 = 70%; Logworth (p-value) = 14.10), respectively. The association with year was weaker (semi-partial R 2 = 27%; Logworth (p-value) = 5.29), with 2022 being significantly earlier by more than 10 days than the other years (fig. 6). No significant interaction between altitude and year was found (p = 0.2451), indicating that the effect of altitude was similar across the years. In other words, the impact of altitude was similarly evident across sites for other environmental and meteorological factors. According to this model, there is a 0.03-day delay for every meter of increase in altitude.

Figure 5. Calendar days (A) and accumulated DGD (lower temperature threshold of 9.5°C) (B) from January 1 until the first capture of C. perspectalis moth in the pheromone traps in each of the 18 localities, arranged from the lowest (1:253 m) to the highest (18:1405 m) altitude, monitored over the 6 years.

Figure 6. Average of calendar days and accumulated DGD (lower temperature threshold 9.5°C), from January 1 until the first capture of C. perspectalis moth in the pheromone traps in the 18 localities over the 6 years.
The results for thermal time (DGD) to first flight (fig. 5B) differed significantly from those for the calendar days. The overall model was only marginally significant with a p-value of 0.0276. Altitude was the only significant factor in the model, accounting for 9.2% of the total variability. Its Logworth (p-value) of 2.42 was much lower than that of the model using calendar days as a response variable. No significance was found for the year (Logworth, p = 0.75) or their interaction (Logworth, p = 0.52), indicating that the use of thermal time accounted for other meteorological factors.
Discussion
Native boxwood forests across Europe are being destroyed by the arrival of the invasive box tree moth C. perspectalis. To date, climatic conditions and natural enemies have not been able to contain pests in the regions they reach. The rapid spread across all European countries, from the warmest to the coldest (López and Eizaguirre, Reference López and Eizaguirre2019; Poitou et al., Reference Poitou, Bras, Pineau, Lorme, Roques, Rousselet, Auger-Rozenberg and Laparie2020; Bereś et al., Reference Bereś, Ziętara, Nakonieczny, Kontowski, Michał and Augustyniak2021; Bras et al., Reference Bras, Lombaert and Kenis2022; Mascaró et al., Reference Mascaró, Guasch, Borrás, Lassnig, Gil and Pinya2023; Popov and Popova, Reference Popov and Popova2025; Schmera and Baur, Reference Schmera and Baur2025), indicates that the expansion of C. perspectalis is not currently limited by cold winters. The effect of high maximum temperatures on herbivorous insects has often been overlooked in CC research. In southern Europe and Turkey, an increase in maximum temperatures can affect not only the pest but also its host plant threatening, boxwood survival, and spread, as noted by Canelles et al. (Reference Canelles, Aquilué and Brotons2022) and Varol et al. (Reference Varol, Canturk, Cetin, Ozel, Sevik and Cetin2022) for B. sempervirens and Alipour and Wals (Reference Alipour and Walas2023) for Buxus hyrcana Pojark.
This study examined the effects of high temperatures, which can occur in southern Europe during spring and summer, on the growth of the invasive species C. perspectalis and its response to photoperiod. Under natural conditions, high temperatures are usually linked with low environmental humidity. In our experiments, analysing the effects of temperature alone was challenging. The methodologies applied to the eggs and larvae were highly effective in controlling extraneous variables. However, for adults, the impact of high temperatures is likely to be overestimated. In natural environments, adults can seek refuge in shaded microhabitats during the hottest periods of the day, an adaptive behaviour that could mitigate heat stress. Such behavioural thermoregulation was not possible within the rearing chambers, potentially amplifying the apparent effects of elevated temperature on adult performance and survival.
Although higher temperatures increased egg development rates, they clearly slowed larval development and increased the mortality of eggs, larvae, and pupae. Higher temperatures negatively affect adult longevity and fertility. The response of Paralobesia vietana (Clemens, 1860) (Lepidoptera, Tortricidae) to high temperatures (Tobin et al., Reference Tobin, Nagarkatti and Saunders2001) shows patterns similar to those observed in C. perspectalis, with decreased developmental rate and increased mortality. These data indicate that high temperatures do not shorten the duration of complete generation in summer; instead, they may increase it. These results suggest that in the Mediterranean Basin, spring-summer temperatures approach the upper temperature threshold for species development. Therefore, an increase in CC is unlikely to benefit pest species. According to the Meteorological Service of Catalonia (2026), the average annual temperature is expected to increase towards the middle of the 21st century, regardless of the scenario considered, with a statistically significant trend of +0.7 to +2.1°C over 45 years. Geographically, the largest projected increases are in the Pyrenees, and especially in the peaks of the western Pyrenees. The maximum temperature increases are projected for autumn, followed by summer. Therefore, as observed in Thaumetopoea pityocampa (Denis & Schiffermüller, 1775) (Lepidoptera, Thaumetopoeidae) in North Africa (Bourougaaoui et al., Reference Bourougaaoui, Ben Jamâa and Robinet2021) or in California and Italy with the olive fly Bactrocera oleae (Gutierrez et al., Reference Gutierrez, Ponti and Cossu2009), increasing the maximum temperature could lead to a decline in pest populations in the region.
The first two larval instars are sensitive to diapause induction. The diapause-inducing photoperiod occurred at this latitude in late August (López and Eizaguirre, Reference López and Eizaguirre2019). Therefore, larvae that hatch at that time will be susceptible to the inducing environmental conditions and will enter diapause. In contrast, those that are more developed by the end of August (L3–L6 instar larvae) will continue to grow and produce the third generation of adults, which will be partial in most years. The offspring of this third adult generation will be larvae hatched under the induced photoperiods, and all will enter diapause. The high temperatures tested did not decrease the percentage of larvae that entered diapause under an inducing photoperiod (100%) but did increase mortality. Therefore, hot springs and summers do not accelerate larval development or reduce the proportion of larvae entering diapause, making it unlikely that voltinism or the number of generations in a region will change. In contrast, high temperatures, especially early in summer, may cause higher mortality in the first field generation (adults, eggs, larvae, and pupae), leading to fewer second flights. During 2022–2023 (extremely dry and hot years), a decrease in C. perspectalis catches was observed in the study area (Fraile, Reference Fraile2024), suggesting that the spread of the species was slow. However, over the past 2 years (DARP, 2025), the species’ expansion increased again, likely because spring-summer drought conditions eased, improving boxwood growth conditions.
The catches from the pheromone traps used to track the flight activity of the species in our region indicated that altitude was the most significant factor influencing the emergence of the first adult flight (termination and post-diapause period) in natural populations. Data were obtained from 18 locations at different altitudes over 6 years. The days to flight start were adjusted according to Hopkins’ Bioclimatic Law (Hopkins, Reference Hopkins1919), showing an increase of approximately 3 days per 100 m of altitude on the day of the first adult capture. However, thermal time, measured as the sum of the accumulated DGDs until the first capture, was less closely linked to the start of flight. Additionally, the year, regardless of altitude, proved to be a significant factor for flight onset, indicating that factors beyond simple heat accumulation, such as plant effects, drought, humidity, precipitation, and atmospheric pressure, also influence the first adult flight from overwintering larvae. Studies such as those by Alipour and Walas (Reference Alipour and Walas2023) suggest that CC affects boxwood more through drought than through temperature, and that diapausing C. perspectalis larvae can detect this harmful state of the plant. It appears that, per se, temperature increases caused by CC will not shorten the termination and post-diapause periods. These findings differ from those of Poitou et al. (Reference Poitou, Bras, Pineau, Lorme, Roques, Rousselet, Auger-Rozenberg and Laparie2020), possibly because, as they noted, the larvae in their study were no longer in diapause or exposed to other natural conditions, so they responded to temperature but not to photoperiod. Altitudes, such as photoperiod and latitude, are fixed abiotic signals that are subject to unexpected fluctuations, both biotic and abiotic. These consistent signals can influence herbivore life cycles across different geographic regions.
Currently, boxwood is considered endangered in many natural areas; however, in 2012, Di Domenico et al. (Reference Di Domenico, Lucchese and Magri2012) noted that most boxwood populations in Europe’s temperate regions were vigorous. The CC and invasive C. perspectalis may have been responsible for this decline. Ledru et al. (Reference Ledru, Garnier, Gallet, Noûs and Ibanez2022) built a lattice model to determine the spatial structures of natural boxwood in the French Alps, which could promote the coexistence of plants and invasive pests. In the southern Pyrenees-Pre-Pyrenees, where boxwood is an abundant component of the forest along approximately 400 km of the mountain range (Anthos, Reference Anthosn.d., http://www.anthos.es/), coexistence may also depend on the orientation of the patches (sunny/shaded areas) of natural boxwood. Thus, in the southernmost parts of Europe, C. perspectalis will survive climate warming in shady areas or at high altitudes where maximum temperatures are lower than those in sunny areas. However, in these shady areas, the pest almost destroyed the natural boxwood. However, high temperatures can reduce pest populations in sunny areas. In these zones, the survival of the boxwood depends on its ability to adapt to these temperatures. According to Di Domenico et al. (Reference Di Domenico, Lucchese and Magri2012), aridification is the most likely cause of the decline in boxwood populations, reducing the presence of plants in moist areas. Furthermore, evidence shows that boxwood can disappear because of overconsumption by C. perspectalis, but the moth does not entirely disappear from devastated areas. If the plant regrows, the few leaves it produces are quickly colonised and destroyed by the remaining pests. In fact, in the study region, 7 years after the first devastating attack, the natural boxwood has not recovered, with sparse lower buds quickly consumed by the remaining C. perspectalis population. The survival of natural boxwood forests in Europe also depends on the adaptation of natural enemies to pests (Zamani et al., Reference Zamani, Farahani, Farashiani, Salehi and Samavat2017; Mostini, Reference Mostini2019; Hulujan et al., Reference Hulujan, Florian, Florian and Oltean2021; Can et al., Reference Can, Ercan and Ularlı2022; Ferracini et al., Reference Ferracini, Pogolotti, Mancardi, Miglio, Bonelli and Barbero2022; López et al., Reference López, Las Heras, Garrido-Jurado, Quesada-Moraga and Eizaguirre2022; Usta, Reference Usta2022; Bayramoğlu et al., Reference Bayramoğlu, Demir and Gencer2023; Bjeliš et al., Reference Bjeliš, Armanda, Nadel, Raper and Simmons2024; Brinquin et al., Reference Brinquin, Muller, Correard, Gili and Pénigot2024; Poloni et al., Reference Poloni, Dhennin, Mappes, Joron and Nokelainen2024), which are currently scarce, but their presence in the environment is gradually increasing.
Conclusion
Temperature increases associated with CC in southern Europe are unlikely to favour C. perspectalis populations, as the current spring–summer conditions are already approaching the upper thermal limits of the species. Elevated temperatures did not reduce diapause induction, and our results indicate that altitude, rather than accumulated thermal time, is the primary driver of first-flight emergence. Pest–host coexistence may further depend on boxwood orientation, with shaded habitats vulnerable to infestation. Future research should examine the potential for natural enemies to adapt to this invasive species, as increasing biocontrol pressure may be critical for the long-term persistence of native boxwood in Europe.
Acknowledgements
We thank Professor Ignacio Romagosa for his assistance with the statistical analyses. We are grateful to the technicians of the Xarxa de vigilància de la papallona del boix (Cydalima perspectalis) a Catalunya – especially Emili Bassols, Sandra Las Heras, and Miriam Sangerman – for providing log-term data on C. perspectalis flight activity in Catalonia.
Competing interests
The author(s) declare none.