Introduction
Methane (CH4) is the second most important anthropogenic greenhouse gas with a global warming potential 80 times that of carbon dioxide over 20 years (IPCC 2021). It is formed by biological and geological processes and major sources include fossil fuels, agriculture and wetlands (Nisbet et al. Reference Nisbet, Manning, Dlugokencky, Michel, Lan, Röckmann, Denier van der Gon, Schmitt, Palmer, Dyonisius, Oh, Fisher, Lowry, France, White, Brailsford and Bromley2023; Saunois et al. Reference Saunois, Stavert, Poulter, Bousquet, Canadell, Jackson, Raymond, Dlugokencky, Houweling, Patra, Ciais, Arora, Bastviken, Bergamaschi, Blake, Brailsford, Bruhwiler, Carlson, Carrol, Castaldi, Chandra, Crevoisier, Crill, Covey, Curry, Etiope, Frankenberg, Gedney, Hegglin, Höglund-Isaksson, Hugelius, Ishizawa, Ito, Janssens-Maenhout, Jensen, Joos, Kleinen, Krummel, Langenfelds, Laruelle, Liu, Machida, Maksyutov, McDonald, McNorton, Miller, Melton, Morino, Müller, Murguia-Flores, Naik, Niwa, Noce, O’Doherty, Parker, Peng, Peng, Peters, Prigent, Prinn, Ramonet, Regnier, Riley, Rosentreter, Segers, Simpson, Shi, Smith, Steele, Thornton, Tian, Tohjima, Tubiello, Tsuruta, Viovy, Voulgarakis, Weber, van Weele, van der Werf, Weiss, Worthy, Wunch, Yin, Yoshida, Zhang, Zhang, Zhao, Zheng, Zhu, Zhu and Zhuang2020). Atmospheric methane concentrations have risen more than 150% since 1750, and so quantifying the sources, fates and processes involved in the global methane cycle is critical to understanding and mitigating climate change caused by rising methane concentrations (Nisbet et al. Reference Nisbet, Manning, Dlugokencky, Michel, Lan, Röckmann, Denier van der Gon, Schmitt, Palmer, Dyonisius, Oh, Fisher, Lowry, France, White, Brailsford and Bromley2023; Saunois et al. Reference Saunois, Jackson, Bousquet, Poulter and Canadell2016).
Radiocarbon (14C) can be used for source confirmation and apportionment of methane emissions in global systems (Graven et al. Reference Graven, Hocking and Zazzeri2019; Steinbach et al. Reference Steinbach, Holmstrand, Shcherbakova, Kosmach, Brüchert, Shakhova, Salyuk, Sapart, Chernykh, Noormets, Semiletov and Gustafsson2021). 14C is the long-lived radioactive isotope of carbon that naturally occurs together with the stable isotopes 12C and 13C which comprise 98.9% and 1.1% respectively. 14C is produced in the atmosphere from the action of cosmic rays on 14N which then decays and reacts to produce 14CO2. This is taken up by organic and inorganic materials through processes such as photosynthesis in plants and the precipitation of carbonates. 14C can then be released back into the atmosphere as 14CH4 through processes such as methanogenesis and thermogenesis.
Atmospheric CH4 and CO2 have a nominal 14C isotopic abundance of 1 × 10–12 relative to carbon 12C and 1 × 10–10 relative to 13C. 14C has a half-life of 5730 yr and it decays away to 0.1% after 60 kyr leaving predominantly the stable isotopes behind. This characteristic is particularly useful for distinguishing sources of modern biological carbon from old geological carbon and thus the impact of humans during the Holocene and Anthropocene. For example, 14C abundance can distinguish 14C free methane sources such as natural gas from contemporary methane sources such as biogas. However, due to its sub-ppt levels, the methods and instrumentation required to determine natural 14C are typically complex, expensive, and time-consuming (Hajdas et al. Reference Hajdas, Ascough, Garnett, Fallon, Pearson, Quarta, Spalding, Yamaguchi and Yoneda2021).
Analysis of 14C is conventionally performed using accelerator mass spectrometry (AMS) (Fahrni et al. Reference Fahrni, Wacker, Synal and Szidat2013; Litherland et al. Reference Litherland, Zhao and Kieser2011; Synal et al. Reference Synal, Stocker and Suter2007). AMS produces negative carbon ions in a Cs sputter source and uses an acceleration stage for removal of isobaric interferences such as 13CH ions at high kinetic energies before analysis of positive 12C, 13C and 14C ions. Samples are converted to CO2 gas and then reduced to solid graphite prior to AMS analysis. Alternatively, the CO2 gas can be directly analyzed using a hybrid Cs sputter source with titanium gas cathodes, albeit at lower measurement efficiency and precision.
For the analysis of 14C in methane, a multi-step process with a specialized vacuum line is conventionally used to pre-concentrate, separate, purify and combust the samples CH4 to CO2 before graphitization and analysis (Ascough et al. Reference Ascough, Bompard, Garnett, Gulliver, Murray, Newton and Taylor2024; Espic et al. Reference Espic, Liechti, Battaglia, Paul, Röckmann and Szidat2019; Garnett et al. Reference Garnett, Murray, Gulliver and Ascough2019; Melchert et al. Reference Melchert, Gwozdz, Gierga, Wacker, Mücher and Rethemeyer2024; Zazzeri et al. Reference Zazzeri, Xu and Graven2021). Methane gas cannot be directly analyzed on conventional AMS instruments operating in the 1+ charge state with a hybrid Cs-sputter gas source as interfering molecules are not sufficiently destroyed under normal conditions and backgrounds are elevated (L. Wacker, personal communication, 2014). While the size and cost of AMS instruments has reduced considerably in recent years, the methods for sample preparation continue to require significant effort, expertise, and specialized preparation apparatus. Therefore, new analytical techniques for 14C analysis of methane have the potential for a significant impact in the field of climate research.
Positive ion mass spectrometry (PIMS) is an alternative to AMS for 14C analysis (Freeman et al. Reference Freeman, Shanks, Donzel and Gaubert2015; Freeman and Shanks Reference Freeman and Shanks2018; NEC 2024; Wilcken et al. Reference Wilcken, Freeman, Xu and Dougans2008). It uses an electron cyclotron resonance (ECR) plasma ion source to generate positive carbon ions from gases and then a gas charge exchange collision cell to simultaneously remove interfering isobaric species and produce negative carbon ions for mass analysis (Cui et al. Reference Cui, Peng, Li, Ma, Wu, Zhang, Dong, Guo and Chen2024; Li et al. Reference Li, Peng, Ma, Jiang, Cui, Wu, Zhang, Guo and Chen2024; Pantecknik 2024). Unlike AMS, the PIMS instrument does not use an accelerator stage or a Cs-sputter source and therefore it is a gas accepting mass spectrometer with a performance comparable to traditional graphite-based AMS systems. At SUERC, we operate the first prototype PIMS system and which is being developed to improve the 14C analysis of gas samples and, remove the need for graphitisation. Here, we report initial results for methane 14C analysis in which methane gas is injected directly into the ECR source.
Experimental
Instrumentation
Gas samples were analyzed at SUERC on a prototype PIMS system constructed by National Electrostatics Corp. (Madison, WI, USA) (NEC 2024). Briefly, the system has an injector consisting of a Microgan ECR ion source (Pantecknik 2024), a focus lens, a Wien filter and an Einzel lens, that injects 100 kV positive ions into a gas charge exchange collision cell. The source 10 GHz RF amplifier was operated at 43W power. The resultant negative ions are analyzed with 2 sequential double focusing mass spectrometers, a Faraday cup for 13C and a PIPS detector for 14C. Raw 14C/13C ratios are presented in this study they are indicative of the nominal isotopic abundance and demonstrate the system is generating, transporting and measuring an ion beam correctly in absolute terms. Also, we did not yet have a recognized normalising modern standard for methane and the tuning of the machine for CO2 and CH4 was sufficiently different that it did allow for normalization across gas species. A nominal 14C/13C ratio of 1 × 10–10 is used for our ∼100 pMC modern gases and <2 × 10–13 for our <0.2 pMC or ∼50 kyr old blank gases.
Gas samples were admitted to the source at a constant flow rate via a capillary leak from an open split (Merritt et al. Reference Merritt, Brand and Hayes1994). The open split was a 15 cm × 4 mm O.D. × 2mm I.D. glass tube closed at one end and purged with sample gas at 1 mL/min delivered via fused silica capillaries. The source leak capillary was 50 µm ID deactivated fused silica capillary with the length adjusted to maximize 13C– beam current. For our source with CO2, maximum current was obtained with a 1–1.5 m leak capillary, a source vacuum of 1.0 × 10–6 Torr and flowrate of 30–40 µL/min. The length of the capillary used for methane analysis is given in the respective experiments.
Samples
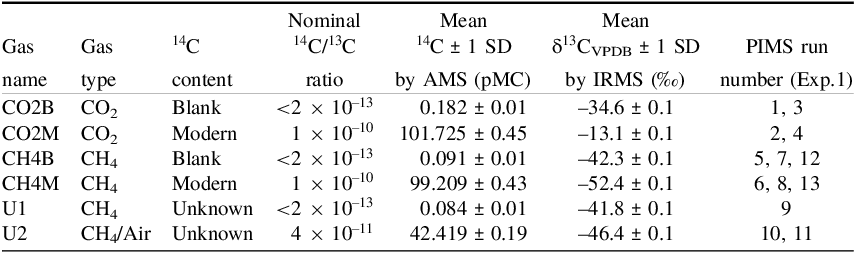
The gases used in this study where blank and modern CO2 and CH4, and 2 unknown gases. These gases were characterized by conventional IRMS and AMS for δ13C and 14C content at the NEIF Radiocarbon Laboratory and the SUERC AMS Laboratory respectively (Table 1) (Ascough et al. Reference Ascough, Bompard, Garnett, Gulliver, Murray, Newton and Taylor2024). Blank CO2 (CO2B; BOC, UK), modern CO2 (CO2M; BOC, UK) and blank CH4 (CH4B; Air Products, UK) were obtained from the NRCL or from commercial sources. Modern methane (CH4M; 97% purity) was obtained from a gas-to-grid biomethane plant (GENeco, UK). Two additional unknown gases were analyzed in this study. Unknown 1 was a cylinder of research grade methane derived from a North Sea fossil fuel source (U1, Table 1; Air products, UK) and Unknown 2 was an air/methane mixture prepared in-house (U2; Table 1). Briefly, a 150-cc lecture bottle containing air at STP was additionally pressurized to approximately 3.5 bar with modern CH4 and then further to 8 bar with 14C-dead CH4. This gave a mixture that was 89.5% CH4, 9.9% air and 0.6% CO2 by gas chromatographic analysis. The concentration of CO2 was <1% and its contribution considered negligible to the CH4 analysis. The δ13C value of the mixture by IRMS indicated it was 41:59 modern:dead methane by isotopic mass balance (Table 1).
Table 1. Summary of 14C analysis by AMS and δ13C analysis by IRMS for CO2 and CH4 analyzed by PIMS in Experiment 1 and 2. Full data is given in Table S1 and the PIMS run numbers for Experiment 1 are indicated. The nominal 14C/13C ratio of a 100 pMC modern gas is 1 × 10–10 and for a 0.2 pMC ∼50 ka old blank gas is 2 × 10–13

Experiments
Two PIMS experiments were performed: 1) discrimination of modern and blank CO2 and CH4, and two unknown CH4 gases, and 2) determining the flowrate of gas in charge exchange collision cell needed to attain a 14C/13C ratio <2 × 10–13 equivalent to a background greater than 50 kyr.
Experiment 1 setup
The gases in Table 1 were measured for 5 × 5 min each over a 24-hr period. CH4 was mixed in the open split with UHP oxygen (Air Products, UK) in a 1:2 v/v stoichiometric ratio. The source was tuned on CO2 for the first four CO2 analyses and then retuned on CH4/O2 for the last nine CH4 analysis runs. Modern and blank samples were run alternately, with duplicate measurements for CO2, triplicate for CH4 and a single measurement for the unknowns. The charge exchange collision cell gas was Isobutane at 6.5 sccm. A 1.25 m leak capillary was connected to the rear of the ion source with a 1/16” union and a Valcon capillary reducing ferrule (Vici Valco, USA). 0.6 m of the leak capillary was removed between the CO2 and CH4 analysis due to an increase in the gas viscosity of the CH4/O2 feed. Samples were changed by exchanging the sample delivered to the open split and data acquisition was commenced when a stable 14C/13C ratio had been reached. Unknown 2 was analyzed at two flow rates of supplementary oxygen of 1:2 and 1:1.5 v/v to bracket and compensate for the oxygen contained in the sample.
Experiment 2 setup
The source was operated with a quartz liner with the leak capillary directly attached to the liner. The leak capillary was 1 m for all measurements and charge exchange collision cell gas was argon. Satisfactory 14C/13C ratios and beam currents were first confirmed with modern and blank CO2 with a cell gas flowrate of 6.5 sccm argon (Table 1, CO2M and CO2B). Then, modern and blank CH4 was analyzed with argon cell gas flowrates from 6.5 to 9.0 sccm in 0.5 sccm steps (Table 1, CH4M and CH4B). The system was retuned after each change in gas and cell gas flowrate. Data was recorded to get < 2% counting statistics for modern CH4 and < 20% for blank CH4. The 14C/13C ratio at each flowrate for the blank gas was corrected by subtracting dark counts and normalizing the result to the ratio and AMS 14C content of the modern gas. Dark counts are counts measured in the PIPS detector due to electronic noise when it is blocked by a faraday cup and no real signal can register (Shanks and Freeman Reference Shanks and Freeman2015). We recorded dark counts for 1405 min to obtain counting statistics < 5%.
Results and discussion
Early unpublished experiments on the PIMS system with carbon monoxide and 16O2 found that gases other than CO2 could produce a usable beam current, a low background and that the ECR source extensively disassociated molecules. Following this, we reasoned that methane may also be disassociated enough after ionization and charge exchange such that we could obtain a molecular free beam that was usable for 14C analysis. Subsequent tests with pure methane gas did not produce a stable beam current and we observed a significant decrease in beam current over several hours. This was due to the deposition of elemental carbon in the source which was observed during experiments studying quartz liners. To counteract the deposition of carbon, oxygen was added in a slight excess of the stoichiometric ratio 1:2 for CH4:O2 so that the equilibrium would favour oxygenated species and inhibit carbon deposition. With added oxygen, stable beam currents and ratios were obtained.
Experiment 1
Samples of blank CO2 (CO2B) and modern CO2 (CO2M) were analyzed in duplicate to confirm normal beam currents, isotope ratios and stability (Table 2, Runs 1–4). The instrument is typically used for CO2 analysis and is tuned to maximize 13C current by optimising the source DC bias, Wien filter, focus lens, Einzel lens, magnets and electrostatic analyzers. This gave repeatable negative 13C currents of 85–90 nA in the Faraday cup after the second analyzing magnet, a 14C/13C ratio of 1 × 10–10 for modern CO2 and 3 × 10–13 for blank CO2. These data are not corrected for dark counts from the detector which further reduces the blank gas value to less than 2 × 10–13 which is consistent with the AMS value of 0.182 pMC or greater than 50 kyr. The negative 13C current for CO2M gas was 2 nA higher than for CO2B which was consistent with CO2M being more enriched in 13C relative to CO2B (Table 1).
Table 2. PIMS analysis of carbon dioxide and methane gas. Data are the average and standard deviation of 5 × 5-min measurements. Gases were also analyzed by conventional AMS and IRMS for 14C and 13C content (Table 1). 13C currents were measured after the second analyzing magnet. Raw ratios are not dark count corrected. Unknown 1 is blank methane and Unknown 2 is a mixture of 10% air and 90% methane with a 14C content of 42.5 pMC. Runs 10 and 11 (Unknown 2) were analyzed with oxygen in a ratio of CH4:O2 of 1:2 and 1:1.5 respectively

After CO2, methane samples were run with oxygen added to the open split (Table 2, runs 5–13). This was to ensure that there was a stoichiometric amount of oxygen in source to prevent carbon deposition and degradation of the beam current. The length of the source leak capillary was shortened to compensate for an increase in gas viscosity and a corresponding decrease in flowrate of the gases flowing into the source. The system was briefly retuned to maximize the negative 13C current. Parameters such as the source gas flow rate and collision cell gas flow rate were not optimized further in this experiment.
Methane samples were alternated between blank and modern methane and the 14C/13C raw ratios were less than 8 × 10–13 for blank methane and 7.5 × 10–11 for modern methane. The ratios differ by 2 orders of magnitude and show that modern and blank methane could be clearly distinguished at natural abundance levels. The negative 13C current for methane dropped by approximately 50% which is consistent with the addition of oxygen to the methane. Lower currents lengthen the time required to acquire sufficient counts however, higher currents and shorter analysis times are possible when the ECR source is operated at higher RF power. Again, it was observed that the PIMS negative 13C currents differed by around 2 nA between the biogenic and fossil methane which was consistent with CH4B being enriched in 13C relative to CH4M (Table 1). The exact reason for the modern methane producing a ratio 75% of modern CO2 was not explored further here but reduced beam transport fidelity through the source Wien filter and beamline have been identified as contributing factors. The addition of oxygen to the methane changes the composition and characteristics of the beam emitted from the source such as the space-charge distribution.
Two additional samples were analyzed to demonstrate that unknown samples and gas mixtures could be analyzed by PIMS and give results that were consistent with AMS data (Samples U1 and U2, Table 2, runs 9–11). The PIMS analysis of Unknown 1 gave a 14C/13C ratio of 8 × 10–13 which matched that of our in-house blank methane (CH4B, Table 1). This was consistent with the origin of the gas being fossil and with the conventional AMS and IRMS analysis (Table 1). PIMS analysis of the second unknown sample with oxygen set at 2 ratios of 1:1.5 and 1:2 to bracket a stoichiometric ratio of 1:2, gave a mean 14C/13C ratio of 0.32 × 10–11. When corrected for the mean 14C/13C ratio of 0.75 × 10–11 for the modern methane (CH4M, Table 1), this gave a corrected 14C/13C ratio of 0.42 × 10–11 for Unknown 2 that is consistent with the nominal value of 4 × 10–11 obtained by AMS (Table 1). The PIMS 13C current for U2 was lower and consistent with the higher content of O2 and N2 in the source. Without more rigorous analysis we feel it is not justified drawing more detailed conclusions about the accuracy and precision of these samples. It was noted that the PIMS analysis allowed rapid screening of these samples within 30 min which would not have been readily possible by conventional AMS.
An additional point of note is that the analysis of methane and the unknown air mixture was possible with concentrations of oxygen greater than 50% v/v. It was found that as long as the gas composition is consistent and the instrument is tuned for maximum negative C current, the ion source was highly tolerant of a wide range of gases such as N2, O2, He and Ar. This is a clear advantage over hybrid Cs-sputter sources which suffer poisoning of the Titanium gas cathodes (Fahrni et al. Reference Fahrni, Wacker, Synal and Szidat2013). It is also an advantage in terms of preparation of atmospheric methane which may be able to be substantially reduced and simplified with and additional preconcentration step (Espic et al. Reference Espic, Liechti, Battaglia, Paul, Röckmann and Szidat2019; Garnett et al. Reference Garnett, Murray, Gulliver and Ascough2019). We believe that PIMS will be able to directly analyze a wide range of carbon containing gases and that this may substantially benefit research in climate change and renewable fuels.
Experiment 2
Obtaining backgrounds with 14C/13C ratio better than 2 × 10–13 or 50 kyr, is important for determining if a sample is sourced from fossil fuels. We then repeated the analysis of methane but varied the collision cell gas flowrate to study if the elevated backgrounds measured in experiment 1 could be reduced further. A system check with CO2 gas gave 55 nA of 13C– beam current, a modern 14C/13C ratio of 1 × 10–10 and blank 14C/13C ratio of < 2 × 10–13 which confirmed nominal ratios and acceptable currents. We then varied the argon cell gas from 6.5 to 9 sccm and observed a reduction in the corrected 14C/13C ratio to below 2 × 10–13 at 8 sccm (Table 3). 1276 dark counts were recorded over 1405 min which gave a mean dark count rate of 0.91 counts/min from the PIPS detector in our system. The ratio of the 14C/13C blank gas was corrected for the ratio on the modern gas and dark counts however the result is not equivalent to pMC or Fm as no 13C correction is applied here. These results confirmed that it was possible to obtain a background better than 50 kyr from the direct analysis of methane gas and confirms PIMS utility for 14C content in methane.
Table 3. Background 14C/13C ratio versus argon collision cell gas flowrate. Details of the blank methane (CH4B) and modern methane (CH4M) are given in Table 1. The blank 14C/13C ratio was corrected for 0.91 dark counts/min and the modern gas ratio

We operated the system with a quartz liner in the source, argon cell gas, a fixed length leak capillary and an open split. This improves system drift, sample crosstalk and beam transport but 13C– beam currents were reduced to 24–29 nA. Also, as the cell gas increases, the negative 13C beam current decreased 1 nA per 0.5 sccm due to losses from increased scattering. It was also observed that the 14C/13C ratio of the modern gas was higher than for experiment 1 due to the lower beam currents and increased beam transport fidelity. Again, we expect that increasing the RF power of the source will increase beam currents and lower analysis time however it is also likely to impact beam transport in our system.
Conclusions
We have shown that it is possible to directly determine the 14C content of methane gas using positive ion mass spectrometry. Samples can be analyzed as mixtures with high contents of air and oxygen which will simplify preparation and carbon containing gases other than CO2 can be analyzed. Methane from biological and fossil sources can be clearly differentiated such that the technique will be useful for screening of biogas where lower precision data is informative i.e. during production. Our next step will be to obtain a bulk sample of modern methane to characterize and use as a long-term normalizing standard.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/RDC.2025.10182
Acknowledgments
We would like to thank the following: Martyn Dolan at GENeco, Bristol, for their generous help with supply, site access and collection of the biomethane gas; Our partners at National Electrostatics Corp., USA and Pantechnik SA, France; Mark Garnett, Callum Murray, Chris Taylor at NEIF Radiocarbon Laboratory, East Kilbride for their expert assistance in preparation of methane and CO2 cylinder samples for AMS analysis; Terry Donnelly at SUERC for the analysis of the methane and air mixture. Funding for this project was supported by NERC Strategic Capital Call 2014 CC076.