Introduction
Goat farming constitutes a critical component of the livestock sector in India, offering a reliable source of livelihood, nutritional security and economic resilience to smallholder and marginal farmers. In rural and semi-arid regions, goats contribute significantly to household income and food availability, particularly under conditions of climatic uncertainty and land scarcity (Kumar et al., Reference Kumar, Singh and Bangar2013; Magotra et al., Reference Magotra, Gupta, Verma, Alex, Mr and Ahmad2019 ; Magotra et al., Reference Magotra, Bangar, Chauhan, Malik and Malik2021a, Reference Magotra, Bangar and Yadav2022). The growing demand for goat-derived meat and dairy products, driven by rising urbanisation, population growth and evolving dietary preferences, has placed renewed emphasis on improving the productivity and reproductive efficiency of goat populations.
Among the biological determinants of productivity in goats, reproductive traits such as litter size, kidding interval and kid survivability are of paramount importance (Bangar et al., Reference Bangar, Magotra, Yadav and Chauhan2022; Gaur et al., Reference Gaur, Malik, Bangar, Magotra, Chauhan and Yadav2021). Enhanced litter size, particularly through twinning or multiple births, can substantially increase production per breeding female, thereby improving overall flock efficiency (Magotra et al., Reference Magotra, Bangar, Kumar and Yadav2023). However, these traits are governed by complex interactions among genetic, environmental, nutritional and immunological factors (Boujenane and Hazzab, Reference Boujenane and Hazzab2008; Lalit et al., Reference Lalit, Dalal, Dahiya, Magotra and Patil2016; Selvan et al., Reference Selvan, Gupta, Verma, Chaudhari and Magotra2016; Bangar et al., Reference Bangar, Magotra and Yadav2020). Moreover, traits like twinning and neonatal survival often exhibit low to moderate heritability (Mellado et al., Reference Mellado, Mellado, García and López2005), making genetic gains through traditional selection relatively slow (Bangar et al., Reference Bangar, Magotra and Yadav2022). As a result, there is growing interest in molecular and biomarker-based approaches to complement phenotypic selection for enhanced reproductive performance.
One of the primary challenges faced by goat farmers and researchers alike is the lack of affordable and accessible tools for early pregnancy detection and litter size prediction. Ultrasonography, although accurate, is often economically and logistically impractical in smallholder and low-input systems due to its dependence on specialised equipment and trained personnel (Mellado et al., Reference Mellado, Garcia, Ledezma and Mellado2004; Pan et al., Reference Pan, Biswas, Majumdar, SenGupta, Patra, Ghosh and Haldar2015). Other proposed alternatives, such as endocrine profiling, blood metabolite assays and external morphometric measurements, have yet to demonstrate sufficient accuracy or cost-efficiency for widespread field adoption (Magotra et al., Reference Magotra, Bangar and Yadav2021b). These limitations underscore the need for reliable, early-stage candidate markers that are both biologically informative and practically applicable under farm conditions.
Molecular biomarkers, particularly gene expression-based, are valuable tools in reproductive biotechnology. Immune-related genes are crucial due to their role in maternal–fetal immune interactions (Magotra et al., Reference Magotra, Naskar, Das and Ahmad2015, Reference Magotra, Gupta, Ahmad and Alex2020; Jeet et al., Reference Jeet, Magotra, Bangar, Kumar, Garg, Yadav and Bahurupi2022; Pushpa et al., Reference Magotra, Bangar, Patil, Sindhu, Malik, Chaudhary, Garg and Kumar2023). Early embryogenesis depends on proper epigenetic regulation (Inbar-Feigenberg et al., Reference Inbar-Feigenberg, Choufani, Butcher, Roifman and Weksberg2013; Nelissen et al., Reference Nelissen, van Montfoort, Dumoulin and Evers2011), while placental development requires tightly controlled gene expression (Roifman et al., Reference Roifman, Choufani, Turinsky, Drewlo, Keating, Brudno and Weksberg2016). The innate immune system is vital during gestation for pathogen defense and successful placental implantation (Loke and King, Reference Loke and King2000). The maternal immune system must balance fetal tolerance with immune protection, especially in multifetal pregnancies. Toll-like receptor 2 (TLR2) is notable for its dual role in pathogen defense and reproductive modulation (Mansouri et al., Reference Mansouri, Akthar and Miyamoto2025). It detects PAMPs from Gram-positive bacteria (Akira et al., Reference Akira, Uematsu and Takeuchi2006; Oliveira-Nascimento et al., Reference Oliveira-Nascimento, Massari and Wetzler2012), mediating inflammation and supporting reproductive homeostasis.
In reproductive contexts, TLR2 has been implicated in processes critical to pregnancy establishment and maintenance, including embryo implantation, placental development, maternal immune tolerance and cytokine signalling (Xie et al., Reference Xie, Turvey, Williams, Mor, Renaud and Olson2010; Abrahams et al., Reference Abrahams, Bole-Aldo, Kim, Straszewski-Chavez, Chaiworapongsa, Romero and Mor2004; Jaiswal et al., Reference Jaiswal, Agrawal, Mallers, Gilman-Sachs, Hirsch and Beaman2020). Notably, emerging evidence from studies in humans and livestock species suggests that TLR2 expression is modulated by reproductive status, with elevated expression observed in multifetal pregnancies compared to singletons or non-pregnant states (Pioli et al., Reference Pioli, Amiel, Schaefer, Connolly, Wira and Guyre2004; Mor and Cardenas, Reference Mor and Cardenas2010; Meglia et al., Reference Meglia, Johannisson, Petersson and Waller2001; Nayak et al., Reference Nayak, Rajawat, Jain, Sharma, Gondro, Tarafdar, Dutt and Panigrahi2024; Mansouri et al., Reference Mansouri, Akthar and Miyamoto2025). These observations point toward a potential role for TLR2 as a transcriptional marker of immune adaptation in response to increased fetal load.
TLR2 is predominantly expressed on innate immune cells in peripheral blood, where it functions as a sentinel of systemic immune status (Kumar et al., Reference Kumar, Kawai and Akira2009; Good et al., Reference Good, George and Watts2012). In ruminants, neutrophils and monocytes represent the principal cellular reservoirs of TLR2, although appreciable expression has also been reported in certain lymphocyte subsets, including B cells and specific T-cell populations (Kwong et al., Reference Kwong, Parsons, Patterson, Coffey, Thonur, Chang, Russell, Haig, Werling and Hope2011; Duan et al., Reference Duan, Du, Xing, Wang and Wang2022). Consequently, quantification of TLR2 mRNA levels in whole blood provides an integrated measure of transcriptional activity across major immune cell lineages, making it a practical and informative matrix for evaluating systemic immunological adaptations. This is particularly relevant during physiological states such as pregnancy, which involve dynamic alterations in immune cell function, distribution and homeostasis.
Despite this potential relevance, studies exploring TLR2 expression dynamics in goats remain limited, particularly in indigenous breeds adapted to tropical and subtropical agro-climatic conditions. These populations often possess distinct genetic and physiological attributes shaped by long-term natural selection and traditional management systems. Understanding TLR2 expression patterns in such genetically adapted breeds could therefore provide valuable insights into the immune–reproductive interface and help inform region-specific breeding strategies. Furthermore, incorporation of TLR2 into mRNA-based biomarker panels alongside candidate genes such as interferon-tau (IFNT), prolactin-related proteins and key interleukins may support the development of early, non-invasive tools for pregnancy diagnosis and identification of high-prolificacy females (Colleselli et al., Reference Colleselli, Stierschneider and Wiesner2023).
In this context, the present study aimed to evaluate differential expression of the TLR2 gene in goats across distinct reproductive states, namely non-pregnant, single-bearing and twin-bearing individuals.
Materials and methods
Animal selection and reproductive records
The study was conducted on a total of 30 clinically healthy does belonging to two indigenous Indian goat breeds: Beetal (n = 22) and Jakhrana (n = 8). All animals were reared under uniform feeding and managemental practices at the Goat Breeding Farm, Department of Animal Genetics and Breeding, Lala Lajpat Rai University of Veterinary and Animal Sciences (LUVAS), Hisar, India. To assess the reproductive status, transabdominal ultrasonography was performed using a 7.5 MHz linear-array transducer. Based on the number of detected fetuses, the animals were divided into three groups: Group I (Twin pregnancy) – does carrying twin fetuses (n = 12), Group II (Single pregnancy) – does carrying a single fetus (n = 10) and Group III (Non-pregnant) – does without detectable pregnancy (n = 8). Representative ultrasonographic images for fetal classification are provided in Figure 1. Preliminary statistical analyses revealed no significant differences (p > 0.05) between the two breeds for the parameters under study; therefore, the data from Beetal and Jakhrana were pooled to ensure sufficient statistical power for downstream gene expression analysis.
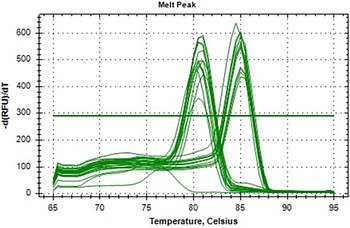
Melt curve analysis confirming specificity of qRT-PCR amplification for TLR2 gene expression in goat blood samples.

In addition to ultrasonographic evaluation, historical breeding records maintained at the farm were used to document the age at first service and body weight at service for each animal. These reproductive parameters served as physiological indicators and were considered during interpretation of gene expression profiles across the different reproductive states.
Blood collection and sample handling
A volume of 3 mL of whole blood was aseptically collected from each doe via jugular venipuncture into EDTA-coated vacutainer tubes after ultrasonographic evaluation. The animals selected for sampling were in their second parity and belonged to the age group of approximately 3–5 years. Immediately after collection, the blood samples were transferred to the Animal Genomics Laboratory, Department of Animal Genetics and Breeding, LUVAS, Hisar, Haryana, India, after proper labelling and documentation. To ensure RNA integrity, all samples were kept on ice and processed within 2 hours of collection, thereby minimising the risk of RNA degradation and ensuring high-quality samples for subsequent molecular and gene expression analyses.
Haematological analysis
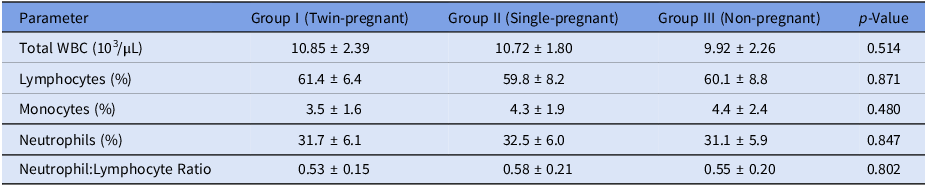
To determine if variations in leukocyte profiles could influence whole-blood gene expression data, a complete blood count (CBC) was performed on all EDTA blood samples immediately after collection using an automated veterinary haematology analyser. Parameters recorded included total leukocyte count (WBC), erythrocyte indices and differential leukocyte counts (lymphocytes L, monocytes M, neutrophils N, eosinophils E, basophils B). These haematological parameters were statistically compared across Group I (twin-pregnant), Group II (single-pregnant) and Group III (non-pregnant) to ensure that observed gene expression differences were not secondary to shifts in major leukocyte populations.
RNA isolation, cDNA synthesis and quantitative real-time PCR analysis
Total RNA was isolated from whole blood samples using the Maxwell® RSC simply RNA Blood Kit (Promega, USA) on the automated Maxwell® RSC Instrument, following the manufacturer’s instructions. Briefly, approximately 400 µL of EDTA blood was lysed with the provided lysis buffer and Proteinase K. The lysate was then loaded into pre-filled cartridges in the instrument, which performed magnetic bead-based RNA purification encompassing binding, washing and elution steps. RNA was eluted in 50 µL of nuclease-free water. The concentration and purity of the extracted RNA were assessed using a NanoDrop™ 2000 Spectrophotometer (Thermo Fisher Scientific, USA) and samples with an A260/A280 ratio greater than 1.8 were considered suitable for downstream analysis. RNA integrity was verified by electrophoresis on a 1.5% agarose gel, where the presence of sharp 28S and 18S rRNA bands confirmed intact and high-quality RNA. To eliminate potential genomic DNA contamination, all RNA samples were treated with RNase-free DNase I (Sigma-Aldrich, USA) prior to complementary DNA (cDNA) synthesis.
First-strand cDNA was synthesised from 1 µg of total RNA using the iScript™ cDNA Synthesis Kit (Bio-Rad, USA), following the kit protocol. The synthesised cDNA was stored at −20°C until use.
Quantitative real-time PCR (qPCR) was performed to evaluate the relative expression of the Toll-like receptor 2 (TLR2) gene using the CFX96 Touch™ Real-Time PCR Detection System (Bio-Rad, USA) with SYBR Green Master Mix (Bio-Rad, USA). Specific primers for the target gene and the reference housekeeping gene β-Actin were employed as follows:
TLR2 (target gene):
Forward:5′-TGCTGTGCCCTCTTCCTGTT-3′
Reverse: 5′-GGGACGAAGTCTCGCTTATGAA-3′
β-Actin (reference gene):
Forward:5′-CGGGAAATCGTCCGTGAC-3′
Reverse: 5′-CCGTGTTGGCGTAGAGGT-3′
Each 20 µL PCR reaction contained 10 µL of 2X SYBR Green Master Mix, 1 µL each of forward and reverse primers (10 µM), 2 µL of template cDNA and 6 µL of nuclease-free water. All samples were run in triplicates and each run included no-template controls (NTC) and no-reverse-transcriptase controls (NRT) to detect any contamination or genomic DNA amplification. The qPCR was performed under standard cycling conditions and melt curve analysis was conducted to confirm amplification specificity.
Data analysis and statistical methods
The relative quantification of TLR2 gene expression was performed using the 2^−ΔΔCt method as outlined by Livak and Schmittgen (Reference Livak and Schmittgen2001). In this method, the cycle threshold (Ct) values of the target gene (TLR2) were normalised against the Ct values of the internal reference gene, β-actin, to account for variations in cDNA input and amplification efficiency. The normalised expression levels were then compared across different experimental groups to determine relative changes in gene expression.
Statistical analyses were carried out using SPSS software version 16.0 (SPSS Inc., Chicago, IL, USA). The mean relative expression levels of TLR2 among the three groups twin pregnancy, single pregnancy and non-pregnant does were compared using one-way analysis of variance (ANOVA) to detect any statistically significant differences attributable to pregnancy status. Where applicable, post hoc tests were performed to identify specific group differences. All quantitative data are presented as mean ± standard error of the mean (SEM) to reflect variability and precision of the estimates.
A threshold of p < 0.05 was set to determine statistical significance. Values below this cut-off were considered indicative of meaningful differences in gene expression related to reproductive status, thus supporting biological interpretations of the data.
Results
Haematological parameters across reproductive states
Analysis of haematological profiles confirmed that the experimental groups were immunologically comparable at the cellular level. No significant differences (p > 0.05) were observed in total leukocyte count (WBC) or in the relative percentages of the major leukocyte subtypes – lymphocytes, monocytes and neutrophils – among twin-pregnant, single-pregnant and non-pregnant does (Table 1). Consequently, the calculated neutrophil-to-lymphocyte ratio (NLR), a systemic inflammatory index, also did not differ between groups. These results indicate that the differential expression of TLR2 mRNA is not an artifact of altered leukocyte counts but reflects transcriptional regulation within a stable circulating immune cell population.
Comparative analysis of haematological parameters in goats across different reproductive states

Values are presented as mean ± standard deviation. Group I: Twin-pregnant (n = 12), Group II: Single-pregnant (n = 12), Group III: Non-pregnant (n = 9). p-values were derived from one-way ANOVA; no significant differences (p > 0.05) were observed among groups for any parameter.
Descriptive statistics
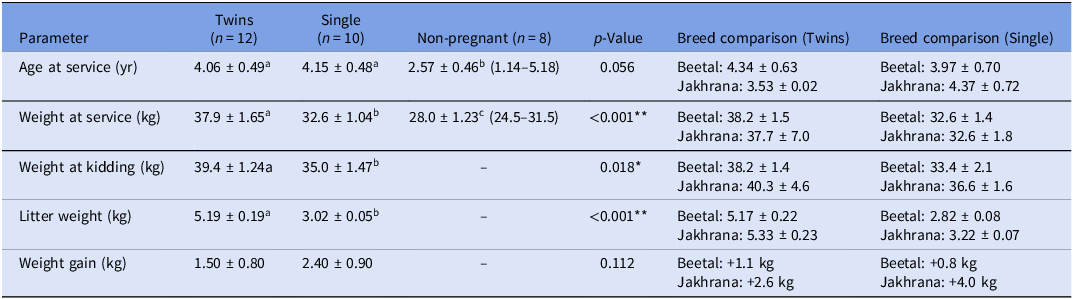
The details data structure and descriptive statistics for three groups of does which were sampled initially for the current study are given in Table 2. Descriptive statistics were analysed for the targeted goats prior to TLR2 gene expression profiling, with animals categorised as twin-bearing (n = 12), single-bearing (n = 10) and non-pregnant (n = 8) based on USG. Twin-bearing goats were significantly heavier at service (37.9 ± 1.65 kg) and at kidding (39.4 ± 1.24 kg) compared to single-bearing and non-pregnant goats (p < 0.05) and had higher litter weights (5.19 ± 0.19 kg vs. 3.02 ± 0.05 kg; p < 0.001). Age at service was comparable between twin- and single-bearing groups but lower in non-pregnant goats (2.57 ± 0.46 years; p = 0.056). Single-bearing goats showed a non-significantly higher weight gain during pregnancy (2.40 ± 0.90 kg) than twin-bearing does (1.50 ± 0.80 kg; p = 0.112), possibly reflecting different patterns of nutrient allocation. No significant differences (p > 0.05) were observed between Beetal and Jakhrana breeds across these parameters; hence, data from both breeds were pooled to increase statistical power for TLR2 expression profiling and subsequent association analyses.
Descriptive statistics are presented as mean ± standard error
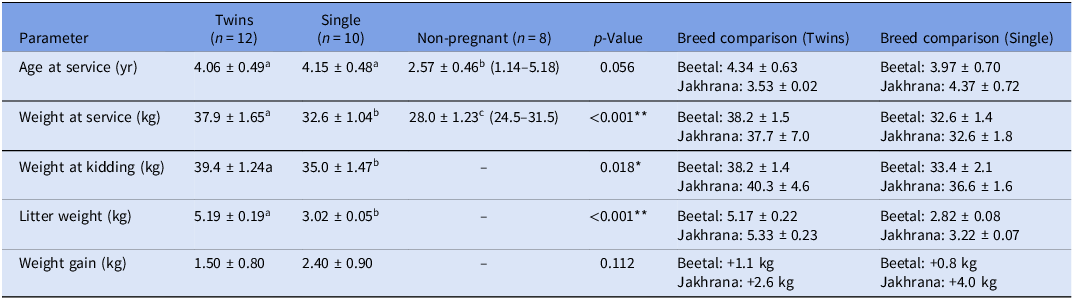
Values are mean ± standard error (range). Different superscript letters (a, b, c) within a row denote statistically significant differences (p < 0.05). Breed comparisons within the twin and single pregnancy groups are also shown. **p < 0.01, *p < 0.05.
Melt curve analysis graph
The melt curve analysis for TLR2 and the housekeeping gene Actin reveals key insights into primer specificity and amplification quality. The data shows temperature ranges from 65°C to 95°C, with distinct peaks expected for both genes. For TLR2, a single sharp peak around 80–85°C would indicate specific amplification, while Actin should display a consistent peak near 75–80°C, confirming its reliability as a stable reference (Figure 1).
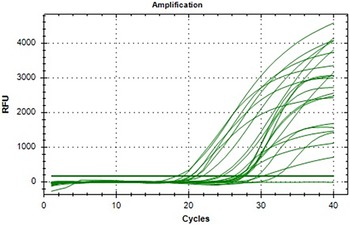
Amplification plot analysis
The real-time PCR amplification plot demonstrates clear and sigmoidal amplification curves across all tested samples, indicating efficient and exponential amplification of the TLR2 gene. The threshold cycle (Ct) values are well-separated among the groups, with earlier Ct values observed in the twin-bearing group, confirming higher transcript abundance. There is no evidence of primer-dimer formation or non-specific amplification in the early cycles, validating the specificity of the reaction. Consistent baseline fluorescence and sharp exponential rises support high reaction efficiency and good-quality RNA and cDNA synthesis (Figure 2).
Amplification plot of TLR2 gene expression in goat blood samples.

Expression analysis of TLR2 gene in goats across reproductive groups
The expression profile of the Toll-like receptor 2 (TLR2) gene was evaluated in goats from three distinct reproductive groups – twin-bearing, single-bearing and non-pregnant using quantitative real-time PCR (qRT-PCR).
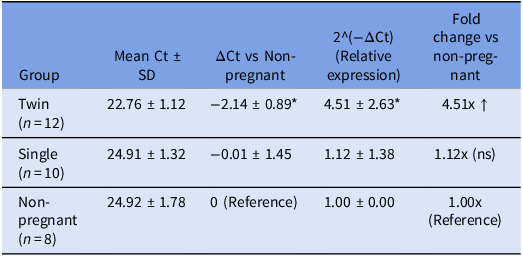
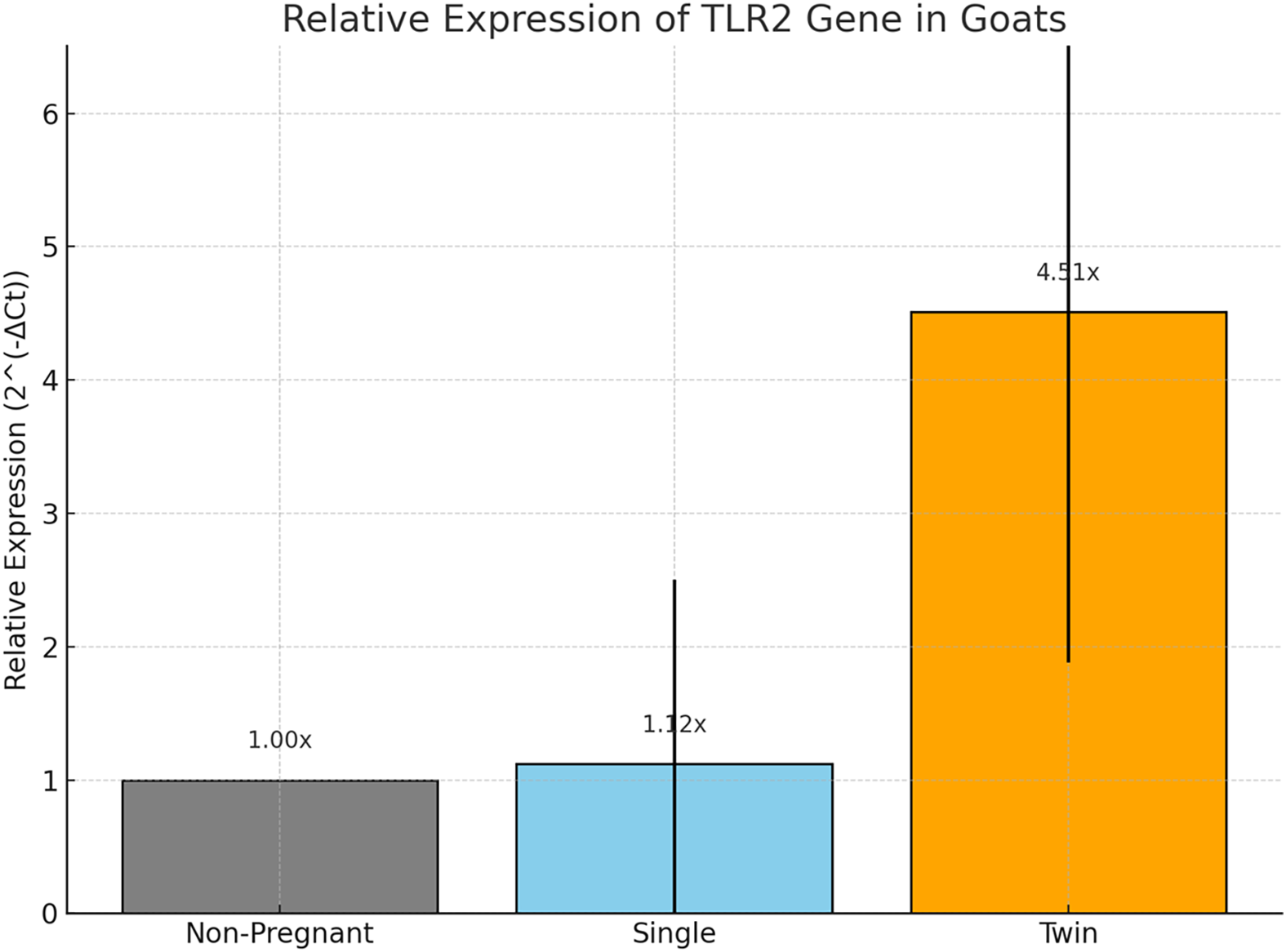
The non-pregnant group (n = 8) was designated as the reference for comparative analysis, with its mean Ct value set as the baseline for ΔCt calculation Table 3. In this group, the mean Ct value for TLR2 was 24.92 ± 1.78 and relative expression (2^(−ΔCt)) was set to 1.00 ± 0.00. Goats carrying twins (n = 12) exhibited a significantly lower mean Ct value of 22.76 ± 1.12, suggesting higher transcript abundance of TLR2 in this group. When ΔCt was calculated with respect to the non-pregnant group, twin-bearing goats demonstrated a mean ΔCt of −2.14 ± 0.89. This corresponded to a mean relative expression value of 4.51 ± 2.63, indicating a more than fourfold upregulation of TLR2 mRNA levels. The increase was statistically significant (p < 0.05), highlighting a potential association between twin pregnancies and heightened TLR2 expression.
Mean Ct, ΔCt, and relative gene expression (2^(−ΔCt)) across groups

Data are presented as mean ± standard deviation. ΔCt was calculated relative to the non-pregnant reference group. The relative expression (2^(−ΔCt)) and corresponding fold-change represent the effect size of gene expression difference. * indicates a significant difference from the non-pregnant group (p < 0.05, ANOVA with post-hoc Tukey test). ns: not significant.
In contrast, the single-bearing group (n = 10) showed a mean Ct value of 24.91 ± 1.32, which was nearly identical to that of the non-pregnant group. The ΔCt in this group was −0.01 ± 1.45, yielding a relative expression of 1.12 ± 1.38. This indicates only a marginal and statistically non-significant increase in TLR2 gene expression compared to the reference.
Statistical comparison of TLR2 gene expression among reproductive groups
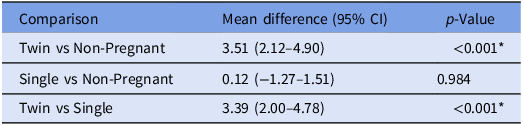
To evaluate the influence of reproductive status on TLR2 gene expression, relative expression levels (calculated as 2^(−ΔCt)) were statistically compared among three groups of goats: twin-bearing, single-bearing and non-pregnant. The effect size for the key comparison between twin-bearing and non-pregnant groups was substantial, with a mean difference of 3.51 (95% CI: 2.12–4.90). Most notably, the difference between twin- and single-bearing groups was also large and significant, with a mean difference of 3.39 (95% CI: 2.00–4.78; p < 0.001). However, no significant difference was observed between single-bearing and non-pregnant goats (mean difference: 0.12; 95% CI: –1.27 to 1.51; p = 0.984). These results confirm that the upregulation of TLR2 is both statistically significant and of considerable biological magnitude, specifically associated with twin pregnancy. The analysis demonstrated significant differences in gene expression between the groups, suggesting a biologically relevant modulation of TLR2 expression associated with reproductive status (Table 4).
Statistical comparison of relative expression (2^(−ΔCt))

The table presents the mean difference (effect size) with 95% confidence intervals (CI) and associated p-values for comparisons between groups. The large, positive mean differences for comparisons involving the twin-pregnant group quantify the substantial magnitude of TLR2 upregulation. *p < 0.001.
Discussion
Reproductive and growth parameters in goats: influence of pregnancy type
This study compared key reproductive and growth parameters among twin-bearing (n = 12), single-bearing (n = 10) and non-pregnant goats (n = 8). Age at service did not differ significantly between twin-bearing (4.06 ± 0.49 years) and single-bearing goats (4.15 ± 0.48 years), but non-pregnant goats were younger (2.57 ± 0.46 years), suggesting that age may influence conception rates (p = 0.056). Body weight at service was significantly higher in twin-bearing goats (37.9 ± 1.65 kg) compared to single-bearing (32.6 ± 1.04 kg) and non-pregnant goats (28.0 ± 1.23 kg) (p < 0.001), indicating that heavier goats were more likely to conceive twins (Figure 3). This aligns with previous findings that higher pre-breeding body condition enhances ovulation rates and twin pregnancies in small ruminants (Mellado et al., Reference Mellado, Garcia, Ledezma and Mellado2004; Magotra et al., Reference Magotra, Bangar, Kumar and Yadav2023).
Bar graph showing the relative expression levels of the TLR2 gene across non-pregnant, single-bearing, and twin-bearing goat groups, with error bars indicating standard deviation.

At kidding, twin-bearing goats remained heavier (39.4 ± 1.24 kg) than single-bearing goats (35.0 ± 1.47 kg) (p = 0.018), further supporting the association between maternal body weight and reproductive success. Litter weight was significantly greater in twin pregnancies (5.19 ± 0.19 kg) than in single pregnancies (3.02 ± 0.05 kg) (p < 0.001), consistent with the presence of an additional kid. Interestingly, single-bearing goats exhibited numerically higher maternal weight gain during pregnancy (2.40 ± 0.90 kg) compared to twin-bearing goats (1.50 ± 0.80 kg), though this difference was not statistically significant (p = 0.112). This trend suggests that single pregnancies may allow for greater nutrient partitioning toward maternal reserves, whereas twin pregnancies prioritise fetal growth, resulting in higher litter weight but reduced maternal weight retention a phenomenon previously observed in sheep (Yisahak et al., Reference Yisahak, Hinkle, Mumford, Grantz, Zhang, Newman, Grobman, Albert, Sciscione, Wing, Owen, Chien, Buck Louis and Grewal2024). These findings highlight the influence of body weight and age on reproductive outcomes in goats, with heavier and older goats more likely to conceive twins. The differential weight gain patterns between single and twin pregnancies may reflect distinct metabolic adaptations, where twin-bearing goats allocate more resources to fetal development, while single-bearing goats retain additional maternal reserves (Magotra et al., Reference Magotra, Bangar, Chauhan, Malik and Malik2021a).
TLR2 expression is associated with twin pregnancy in goats
The present study reveals a marked and statistically significant upregulation of TLR2 mRNA expression in the peripheral blood of twin-bearing goats, with an approximately 4.5-fold increase compared to non-pregnant controls (p < 0.001), whereas no comparable change was observed in single-bearing goats. This pattern indicates that elevated systemic TLR2 transcription is specifically associated with twin pregnancy rather than pregnancy alone. Importantly, the absence of significant differences in total or differential leukocyte counts among groups suggests that the observed upregulation reflects a targeted transcriptional response linked to twin gestation rather than alterations in circulating leukocyte populations.
TLR2 expression within the twin-bearing group exhibited relatively high biological variability (4.51 ± 2.63), which, as reported in earlier studies, may be attributed to inter-individual differences in immune responsiveness, subtle variation in gestational stage at sampling and differences in placental efficiency or fetal developmental status. Similar heterogeneity in immune-related gene expression among individuals within the same pregnancy category has been documented previously and highlights the complex, multifactorial nature of maternal immune adaptation during gestation (Robinson and Klein, Reference Robinson and Klein2012). Consistent with this, studies in ruminants and humans indicate that multiple gestations are more frequently associated with distinct immune modifications than singleton pregnancies (Koga and Mor, Reference Koga and Mor2010; Mor and Cardenas, Reference Mor and Cardenas2010; Meglia et al., 2001).
Although this study establishes a strong association between twin pregnancy and elevated peripheral TLR2 expression, causal relationships and precise mechanistic roles in caprine pregnancy remain to be elucidated. Functional involvement of TLR2 in placentation and maternal–fetal immune tolerance is inferred from mechanistic evidence in other species (Akira et al., Reference Akira, Uematsu and Takeuchi2006; Basith et al., Reference Basith, Manavalan, Lee and Kim2011). The observed upregulation aligns with models proposing that twin pregnancy imposes greater immunological and metabolic demands, necessitating enhanced immune regulation and surveillance (Takeda et al., Reference Takeda, Kaisho and Akira2003; Abrahams et al., Reference Abrahams, Bole-Aldo, Kim, Straszewski-Chavez, Chaiworapongsa, Romero and Mor2004; Pioli et al., Reference Pioli, Amiel, Schaefer, Connolly, Wira and Guyre2004; Jaiswal et al., Reference Jaiswal, Agrawal, Mallers, Gilman-Sachs, Hirsch and Beaman2020; Meglia et al., 2001) and may represent a systemic adaptive response supporting placental development and fetal tolerance in multifetal gestation (Roifman et al., Reference Roifman, Choufani, Turinsky, Drewlo, Keating, Brudno and Weksberg2016; Mor and Cardenas, Reference Mor and Cardenas2010).
Interpretation of these findings should be made in the context of certain study limitations. Assessment of TLR2 expression in peripheral blood provides a practical indicator of systemic immune status but may not fully reflect localised immunological events at the maternal–fetal interface; therefore, future investigations should extend expression analysis to reproductive tissues such as the placenta and endometrium. Additionally, although animals were maintained under uniform management conditions with comparable parity, the lack of precise gestational age determination at the time of sampling may have contributed to expression variability, given that immune markers fluctuate across pregnancy stages. Longitudinal studies incorporating controlled breeding and timed sampling will be essential to disentangle the independent effects of fetal number and gestational progression. This study establishes a clear association between TLR2 gene expression and prenatal litter size in an Indian goat breed, highlighting its relevance as a marker linked to reproductive efficiency. However, validation on larger sample size needs to be warranted. Future investigations should focus on tissue-specific characterisation of TLR2 expression at the maternal fetal interface and on correlating transcript abundance with functional protein-level data to strengthen the biological significance and translational relevance of these observations.
Conclusion
The present study provides novel evidence of a strong association between twin pregnancy and the significant upregulation of the innate immune gene TLR2 in the peripheral blood of goats. The data reveal a distinct systemic immune gene expression profile linked specifically to multiple gestation, characterised by a more than fourfold increase in TLR2 mRNA in twin-bearing does compared to non-pregnant controls (4.5-fold, p < 0.001), with no significant change observed in single-bearing animals (1.12-fold, p = 0.984). While these findings highlight a clear correlation between fetal number and systemic TLR2 levels, it is important to emphasise that the functional role of TLR2 in mediating pregnancy adaptation in goats and whether this upregulation is a cause or consequence of twin gestation remains to be determined through targeted mechanistic studies. Our results, derived from a stable haematological background and uniform animal management, contribute meaningful insight into pregnancy-associated immune modulation in small ruminants. Moving forward, TLR2 emerges as a candidate biomarker worthy of further investigation. Future research should therefore focus on localising TLR2 expression at the maternal-fetal interface, validating its predictive value through longitudinal studies and integrating it with other biomarkers to assess its practical utility in goat breeding programmes.
Data availability statement
Data are available on request due to privacy/ethical restrictions.
Acknowledgements
We duly acknowledge the facilities provided by the Director of Research, LUVAS, Hisar, India and used to conduct this research. We also acknowledge funding obtained from the HSCSIT, Science and Technology Department (HSCSIT/R&D/2021/538), Haryana, India.
Author contributions
AM lead contributor, AM designed study, YCB performed statistical analysis, DKY, SK helped for data collection and wet lab work. AB and RK helped for manuscript writing.
Funding
Funding was received from the HSCSIT, Science and Technology Department, Haryana, India with grant no. HSCSIT/R&D/2021/538.
Competing interests
The authors declare that there is no conflict of interest regarding the publication of this paper.
Ethical standards
Ethical approval for conducting the study was granted by the Institutional Animal Ethics Committee (IAEC) of Lala Lajpat Rai University of Veterinary and Animal Sciences, Hisar, Haryana, India. The approval was issued under reference number VCC/IAEC/2022/1624-51.
Informed consent
All authors have read the manuscript and consented to publish.