I. INTRODUCTION
Indacaterol maleate (as the 1:1 salt, marketed under the trade names Arcapta and Onbrez, among others) is used for the treatment of chronic obstructive pulmonary disease (COPD) as a long acting β-adrenoceptor agonist. Indacaterol is included in the Top 200 Small Molecule Drugs by Retail Sales in 2022 (McGrath et al., Reference McGrath, Brichacek and Njardarson2010). The systematic name of indacaterol maleate (CAS Registry Number 753498-25-8) is 5-[(1R)-2-[(5,6-diethyl-2,3-dihydro-1H-inden-2-yl)amino]-1-hydroxyethyl]-8-hydroxy-1H-quinolin-2-one (Z)-but-2-enedioic acid. A two-dimensional molecular diagram of indacaterol maleate is shown in Figure 1.
The two-dimensional structure of indacaterol hydrogen maleate.

The crystal structure of indacaterol maleate has been reported by Baur et al. (Reference Baur, Beattie, Beer, Bentley, Bradley, Bruce, Charlton, Cuenroud, Ernst, Fairhurst, Faller, Farr, Keller, Fozard, Fullerton, Garman, Hatto, Hayden, He, Howes, Janus, Jiang, Lewis, Loeuillet-Ritzler, Moser, Reilly, Steward, Tedaldi, Trifileff, Tweed, Watson, Wissler and Wyss2010; Novartis; CSD Refcode YIBRAG) in space group P1, with Z = 2. There are thus two independent cations and two independent anions in the structure, but the crystal structure is not discussed in any detail. There is currently no powder pattern calculated from this structure in the Powder Diffraction File (Gates-Rector and Blanton, Reference Gates-Rector and Blanton2019). Both PLATON (Spek, Reference Spek2009) and checkCIF (Spek, Reference Spek2020) suggest the presence of a center of symmetry, and thus that the true space group is P-1.
This work was carried out as part of a project (Kaduk et al., Reference Kaduk, Crowder, Zhong, Fawcett and Suchomel2014) to determine the crystal structures of large-volume commercial pharmaceuticals, and include high-quality powder diffraction data for them in the Powder Diffraction File.
II. EXPERIMENTAL
Indacaterol maleate was a commercial reagent, purchased from TargetMol (Batch #144181), and was used as-received. The white powder was packed into a 1.5-mm diameter Kapton capillary, and rotated during the measurement at ~50 Hz. The powder pattern was measured at 295 K at a beam line of 11-BM (Antao et al., Reference Antao, Hassan, Wang, Lee and Toby2008; Lee et al., Reference Lee, Shu, Ramanathan, Preissner, Wang, Beno, Von Dreele, Ribaud, Kurtz, Antao, Jiao and Toby2008; Wang et al., Reference Wang, Toby, Lee, Ribaud, Antao, Kurtz, Ramanathan, Von Dreele and Beno2008) of the Advanced Photon Source at Argonne National Laboratory using a wavelength of 0.459744(2) Å from 0.5° to 40° 2θ with a step size of 0.001° and a counting time of 0.1 s/step. The high-resolution powder diffraction data were collected using twelve silicon crystal analyzers that allow for high angular resolution, high precision, and accurate peak positions. A mixture of silicon (NIST SRM 640c) and alumina (NIST SRM 676a) standards (ratio Al2O3:Si = 2:1 by weight) was used to calibrate the instrument and refine the monochromatic wavelength used in the experiment.
The pattern was indexed using N-TREOR (Altomare et al., Reference Altomare, Cuocci, Giacovazzo, Moliterni, Rizzi, Corriero and Falcicchio2013) on a primitive triclinic unit cell with a = 8.86565, b = 9.76041, c = 16.67825 Å, α = 102.640, β = 94.169, γ = 113.266°, V = 1273.2 Å3, and Z = 2. Since indacaterol is not a chiral molecule, we assumed the space group to be P-1, which was confirmed by successful solution and refinement of the structure. A reduced cell search in the Cambridge Structural Database (Groom et al., Reference Groom, Bruno, Lightfoot and Ward2016) yielded three hits, including Refcode YIBRAG (Baur et al., Reference Baur, Beattie, Beer, Bentley, Bradley, Bruce, Charlton, Cuenroud, Ernst, Fairhurst, Faller, Farr, Keller, Fozard, Fullerton, Garman, Hatto, Hayden, He, Howes, Janus, Jiang, Lewis, Loeuillet-Ritzler, Moser, Reilly, Steward, Tedaldi, Trifileff, Tweed, Watson, Wissler and Wyss2010) for indacaterol maleate.
The indacaterol molecule was downloaded from PubChem (Kim et al., Reference Kim, Chen, Cheng, Gindulyte, He, He, Li, Shoemaker, Thiessen, Yu, Zaslavsky, Zhang and Bolton2023) as Conformer3D_CID_6918554.sdf, and was converted into a .mol2 file using Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020). The structure of the hydrogen maleate anion was extracted from the crystal structure of rosiglitazone hydrogen maleate hydrate (Cuffini et al., Reference Cuffini, Faudone, Ferro, Garland and Baggio2008), and saved as a .mol2 file using Materials Studio (Dassault, 2022). The structure was solved by Monte Carlo-simulated annealing techniques as implemented in EXPO2014 (Altomare et al., Reference Altomare, Cuocci, Giacovazzo, Moliterni, Rizzi, Corriero and Falcicchio2013). A neutral indacaterol molecule and a hydrogen maleate anion were used as fragments. All four torsion angles in the anion were fixed, to make it a rigid body. Analysis of potential hydrogen bond interactions made it clear that N4 was protonated, and H69 was added to that atom using Materials Studio.
Rietveld refinement was carried out with GSAS-II (Toby and Von Dreele, Reference Toby and Von Dreele2013). Only the 1.5°–25.0° portion of the pattern was included in the refinements (d min = 1.062 Å). All non-H bond distances and angles were subjected to restraints, based on a Mercury/Mogul Geometry Check (Bruno et al., Reference Bruno, Cole, Kessler, Luo, Motherwell, Purkis, Smith, Taylor, Cooper, Harris and Orpen2004; Sykes et al., Reference Sykes, McCabe, Allen, Battle, Bruno and Wood2011). The Mogul average and standard deviation for each quantity were used as the restraint parameters. The quinoline and phenyl rings were restrained to be planar. The restraints contributed 10.1% to the final χ 2. The hydrogen atoms were included in calculated positions, which were recalculated during the refinement using Materials Studio (Dassault, 2022). The Uiso of the C, N, and O atoms were grouped by chemical similarity. The Uiso for the H atoms were fixed at 1.3× the Uiso of the heavy atoms to which they are attached. The peak profiles were described using a uniaxial microstrain model, with 001 as the unique axis.
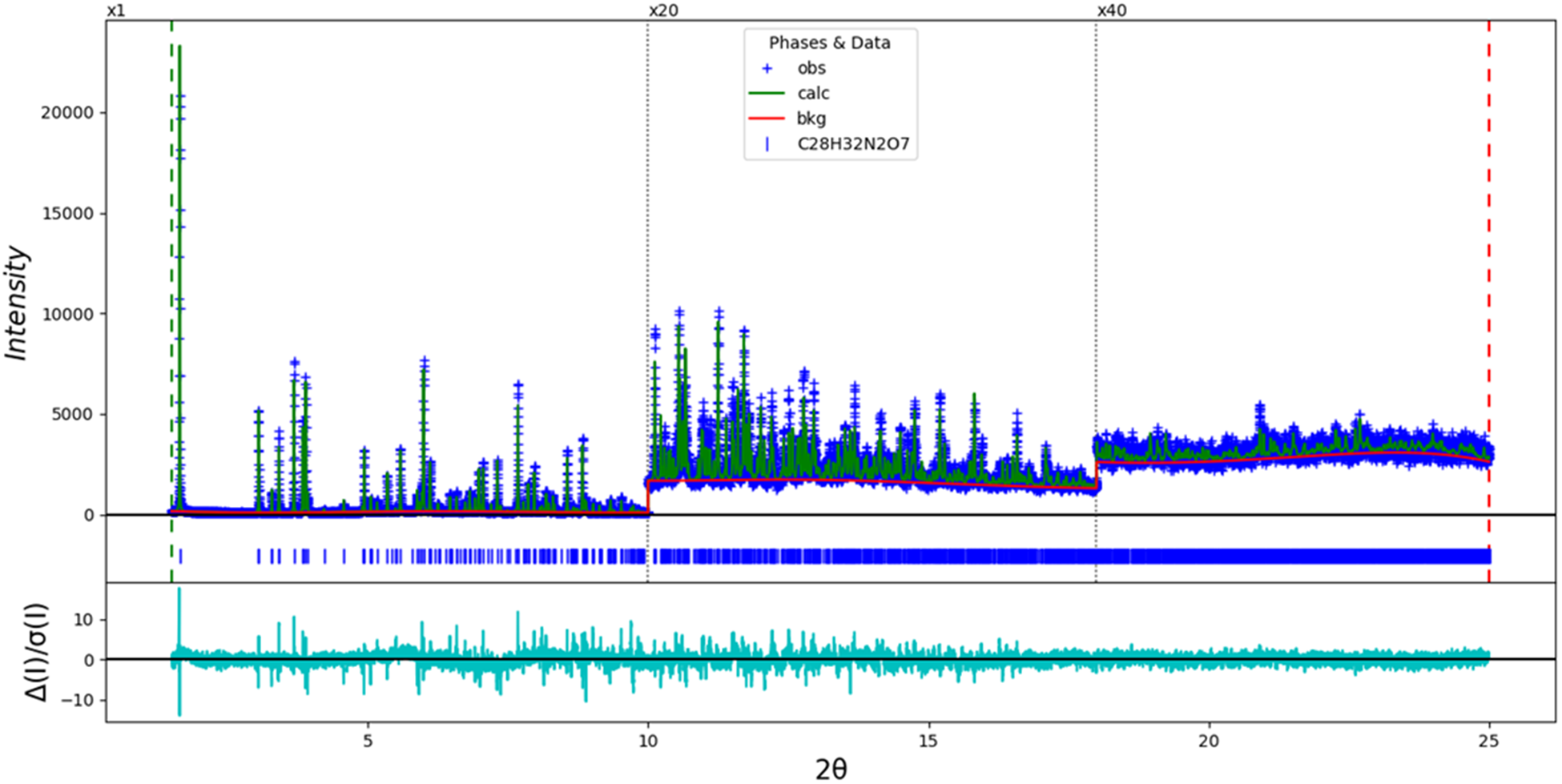
The final refinement of 137 variables using 23,538 observations and ninety-five restraints yielded the residuals Rwp = 0.13002 and GOF = 1.62. The largest peak (0.58 Å from N4) and hole (1.684 Å from C13) in the difference Fourier map were 0.75(10) and −0.47(10) eÅ−3, respectively. The final Rietveld plot is shown in Figure 2. The largest features in the normalized error plot represent subtle errors in peak positions, and probably indicate changes to the specimen during the measurement.
The Rietveld plot for the refinement of indacaterol hydrogen maleate. The x-axis is 2θ, and the y-axis is counts. The blue crosses represent the observed data points and the green line is the calculated pattern. The cyan curve is the normalized error plot and the red line is the background curve. The vertical scale has been multiplied by a factor of 20× for 2θ > 10.0° and by a factor of 40× for 2θ > 18.0°.
The crystal structure of indacaterol hydrogen maleate was optimized (fixed experimental unit cell) with density functional techniques using VASP (Kresse and Furthmüller, Reference Kresse and Furthmüller1996) through the MedeA graphical interface (Materials Design, 2016). The calculation was carried out on 16 2.4-GHz processors (each with 4-Gb RAM) of a 64-processor HP Proliant DL580 Generation 7 Linux cluster at North Central College. The calculation used the GGA-PBE functional, a plane wave cutoff energy of 400.0 eV, and a k-point spacing of 0.5 Å−1 leading to a 2 × 2 × 1 mesh, and took ~9.9 h. Single-point density functional calculations (fixed experimental cell) and population analysis were carried out using CRYSTAL23 (Erba et al., Reference Erba, Desmarais, Casassa, Civalleri, Donà, Bush, Searle, Maschio, Daga, Cossard, Ribaldone, Ascrizzi, Marana, Flament and Kirtman2023). The basis sets for the H, C, N, and O atoms in the calculation were those of Gatti et al. (Reference Gatti, Saunders and Roetti1994). The calculations were run on a 3.5-GHz PC using 8 k-points and the B3LYP functional, and took ~3.9 h.
III. RESULTS AND DISCUSSION
The asymmetric unit contains one indacaterol cation and one hydrogen maleate anion (Figure 3). The compound is thus correctly described as indacaterol hydrogen maleate. The root-mean-square (rms) Cartesian displacement of the non-H atoms in the Rietveld-refined and VASP-optimized cation structures is 0.232 Å (Figure 4); the equivalent quantity for the anions is 0.221 Å. The agreement is within the normal range for correct structures (van de Streek and Neumann, Reference van de Streek and Neumann2014), and provides confirmation that the structure is correct. The remainder of this discussion will emphasize the VASP-optimized structure.
The asymmetric unit of indacaterol hydrogen maleate, with the atom numbering. The atoms are represented by 50% probability spheroids/ellipsoids. Image generated using Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020).

Comparison of the Rietveld-refined (red) and VASP-optimized (blue) structures of indacaterol hydrogen maleate. The rms Cartesian displacement for the cation is 0.232 Å, and for the anion is 0.221 Å. Image generated using Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020).

The P1 structure of YIBRAG and the P-1 structure determined here are essentially identical (Figure 5). When applied to YIBRAG, checkCIF (PLATON) yields a Level G alert, indicating the presence of center of symmetry, with a 97% fit. The centrosymmetric model is more chemically-reasonable, especially when fitting powder (even synchrotron) data, even though the P1 model yields lower residuals (more variables) and highly-correlated parameters. A positive second harmonic generation test could establish the absence of a center of symmetry.
Comparison of the P-1 structure of indacaterol hydrogen maleate determined here (red) to the P-1 structure of YIBRAG (green). Image generated using Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020).

Almost all of the bond distances and bond angles fall within the normal ranges indicated by a Mercury Mogul Geometry check (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020). Only the C13–C16–C19 angle of 115.5° (average = 109.1(20)°, Z-score = 3.2) is flagged as unusual. The torsion angles involving rotation about the C3–N4, C13–C16, and C16–C19 bonds are flagged as unusual. They lie on the tails of broad distributions, and reflect the orientation of the two parts of the cation, and the protonation at N4. The conformation of the cation is unusual.
Quantum chemical geometry optimization of the isolated cation (DFT/B3LYP/6-31G*/water) using Spartan ‘20 (Wavefunction, 2022) indicated that the solid-state conformation of the cation is 9.3 kcal/mol higher in energy than the local minimum, which is similar but has a different orientation of the quinoline ring system with respect to the rest of the cation. The global minimum-energy conformation of the cation is considerably more compact, with parallel ring systems. The solid-state conformation of the anion is 3.0 kcal/mol higher in energy than the local minimum, which is planar. The differences show that intermolecular interactions are important in determining the solid-state conformations.
The crystal structure (Figure 6) consists of layers of cations and anions parallel to the ab-plane. Traditional N–H⋯O and O–H⋯O hydrogen bonds link the cations and anions into chains along the a-axis (Figure 7). The mean plane of the quinoline ring system is approximately 1,6,−20, and that of the indene ring system is 7,3,7. The mean plane of the anion is approximately 3,6,−14, but it is significantly non-planar; the O61–C68–C66–C64 torsion angle is −34.0°, and the O59–C63–C64–C66 torsion is 20.5°. The Mercury Aromatic Analyser indicates only weak interactions, with the strongest between quinoline rings at a distance of 4.4 Å.
The crystal structure of indacaterol hydrogen maleate, viewed down the b-axis. Image generated using Diamond (Crystal Impact, 2023).

The hydrogen bonded chains along the a-axis of cations and anions in indacaterol hydrogen maleate. Image generated using Mercury (Macrae et al., Reference Macrae, Sovago, Cottrell, Galek, McCabe, Pidcock, Platings, Shields, Stevens, Towler and Wood2020).

Analysis of the contributions to the total crystal energy of the structure using the Forcite module of Materials Studio (Dassault Systèmes, 2022) suggests that bond, angle, and torsion distortion terms contribute about equally to the intramolecular energy. The intermolecular energy is dominated by electrostatic attractions, which in this force field analysis include hydrogen bonds. The hydrogen bonds are better analyzed using the results of the density functional theory (DFT) calculation.
As expected, there is a strong intramolecular charge-assisted O–H⋯O hydrogen bond in the hydrogen maleate anion (Table I). There are also two C–H⋯O hydrogen bonds between the anion and cation. The cation makes a strong N4–H69⋯O62 hydrogen bond to the anion, but N4–H35 acts as a hydrogen bond donor to the aromatic C15. The amino group N5–H55 makes bifurcated N–H⋯O hydrogen bonds, one intramolecular to O2 and the other intermolecular to O1 in another cation. The hydroxyl group O1–H52 acts as a donor to O3 in another cation. Several C–H⋯O hydrogen bonds link cations and anions, as well as cations to other cations.
Hydrogen bonds (CRYSTAL23) in indacaterol hydrogen maleate.

a Intramolecular.
The volume enclosed by the Hirshfeld surface of indacaterol hydrogen maleate (Figure 8, Hirshfeld, Reference Hirshfeld1977; Spackman et al., Reference Spackman, Turner, McKinnon, Wolff, Grimwood, Jayatilaka and Spackman2021) is 626.48 Å3, 98.41% of the unit cell volume. The packing density is thus fairly typical. The only significant close contacts (red in Figure 8) involve the hydrogen bonds. The volume/non-hydrogen atom is smaller than usual, at 17.2 Å3, reflecting the strong hydrogen bonds.
The Hirshfeld surface of indacaterol hydrogen maleate. Intermolecular contacts longer than the sums of the van der Waals radii are colored blue, and contacts shorter than the sums of the radii are colored red. Contacts equal to the sums of radii are white. Image generated using CrystalExplorer (Spackman et al., Reference Spackman, Turner, McKinnon, Wolff, Grimwood, Jayatilaka and Spackman2021).

The Bravais–Friedel–Donnay–Harker (Bravais, Reference Bravais1866; Friedel, Reference Friedel1907; Donnay and Harker, Reference Donnay and Harker1937) morphology suggests that we might expect platy morphology for indacaterol hydrogen maleate, with {001} as the major faces. A second-order spherical harmonic model was included in the refinement. The texture index was 1.006(0), indicating that preferred orientation was not significant in this rotated capillary specimen.
IV. DEPOSITED DATA
The powder pattern of indacaterol maleate from this synchrotron data set has been submitted to ICDD for inclusion in the Powder Diffraction File. The Crystallographic Information Framework (CIF) files containing the results of the Rietveld refinement (including the raw data) and the DFT geometry optimization were deposited with the ICDD. The data can be requested at pdj@icdd.com.
ACKNOWLEDGEMENTS
Use of the Advanced Photon Source at Argonne National Laboratory was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, under Contract No. DE-AC02-06CH11357. This work was partially supported by the International Centre for Diffraction Data. We thank Saul Lapidus for his assistance in the data collection.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare.











