Introduction
Over 721,000 healthcare-associated infections (HAIs) occur annually in the United States, with approximately 75,000 associated patient deaths. Reference Magill, Edwards and Bamberg1,Reference Burnham, Olsen and Kollef2 Pathogens responsible for HAIs are frequently discovered on various healthcare surfaces and equipment. Reference Warren, Barrett, Graves, King, Turner and Anderson3,Reference Weber, Anderson and Rutala4
Mobile devices have become an essential component of modern clinical care. Reference Ventola5,Reference Lee, Bin Mahmood, Lee, Smith and Tudor Car6 Inpatient clinician-assigned phones (ICAPs), distributed at the start of clinical shifts and used continuously throughout patient care, are handled frequently between patient encounters and within patient rooms but are not consistently incorporated into infection prevention workflows. Prior studies have demonstrated that healthcare worker mobile phones frequently harbor microbial contamination, including clinically important pathogens (CIPs) suggesting ICAPs may represent an overlooked reservoir. Reference Tannhäuser, Nickel and Lindner7,Reference Dhayhi, Kameli and Salawi8
Enhanced disinfection technologies have been proposed to address challenges associated with manual disinfection, including variability in technique, time constraints, and adherence. The SONOPRO disinfection cabinet delivers automated aerosolized hydrogen peroxide and peracetic acid within an enclosed chamber; however, its performance in real-world clinical settings relative to routine disinfection remains unclear.
To address these gaps, we conducted a prospective, randomized controlled study to evaluate microbial contamination of ICAPs and to compare the efficacy of enhanced disinfection using the SONOPRO cabinet with routine disinfection practices in a pediatric surgery unit.
Methods
Objectives
The primary objective of this randomized controlled trial was to determine the efficacy of an enhanced disinfection strategy, the SONOPRO disinfection cabinet (RESTEC Solutions, LLC; Glenshaw, PA), for reducing microbial bioburden on ICAPs in a real-world hospital environment compared with routine disinfection. Secondary objectives included characterizing baseline contamination and evaluating changes in bioburden and recovery of CIPs.
Study setting
The study was conducted in a pediatric surgery unit in the Duke University Hospital, a 1,062 bed tertiary-care academic medical center in Durham, North Carolina.
Study protocol
We conducted a prospective, randomized controlled study consisting of two sequential phases. Phase 1 (baseline disinfection measurements) was designed to 1) validate a split-surface sampling methodology for ICAPs and 2) characterize residual contamination following routine disinfection practices under real-world conditions. Phase 2 (randomized experimental comparison) was designed to compare the efficacy of enhanced disinfection using the SONOPRO disinfection cabinet with routine disinfection. Eligible ICAPs were clinician-assigned mobile phones used by clinicians providing inpatient care within the pediatric surgery unit and used during a full inpatient clinical shift.
Enhanced disinfection, the intervention arm, was performed using the SONOPRO disinfection cabinet according to the manufacturer’s standard operating cycle. The SONOPRO utilizes ultrasonic atomization to vaporized peracetic acid within an enclosed chamber. Routine disinfection, the control arm, consisted of wiping ICAPs with a quaternary ammonium-based disinfectant wipe (Sani-Cloth; PDI, Woodcliff Lake, NJ), consistent with institutional infection prevention practices. For the purposes of this study, the study team performed the disinfection strategy for both study arms.
All ICAPs were split into left and right surfaces for sampling, including both back and front of the phone, resulting in two samples per phone. In Phase 1, sampling occurred after completion of a full clinical shift and following routine disinfection by clinical staff. Clinicians were aware that phones were being sampled and adherence to routine disinfection practices was not measured. In Phase 2, ICAPs were randomized 1:1 to enhanced disinfection (SONOPRO) or routine disinfection. To perform within-device comparisons while controlling for inter-device variability, sides were further randomized 1:1 to pre or postdisinfection samples. Predisinfection samples were taken prior to any disinfection, while postdisinfection surfaces were sampled following completion of the assigned disinfection protocol (Supplemental Figure 1).
Microbiological methods
Environmental samples were collected from May 19, 2025, to October 20, 2025, using premoistened Eswabs and processed using standard microbiological techniques. Specimens were plated onto routine and selective media to quantify microbial bioburden as colony-forming units (CFUs) and to identify CIPs, defined as Staphylococcus aureus, Enterococcus spp., and gram-negative species. Reference Warren, Barrett, Graves, King, Turner and Anderson3,Reference Warren, Barrett and Graves9,Reference Warren, Fils-Aime, Graves, Barrett, Turner and Anderson10
Outcomes
The primary outcome was total microbial bioburden measured as CFUs per sample. Secondary outcomes included changes in CFUs and the proportion of samples positive for CIPs before and after disinfection.
Data analysis
CFU counts were compared using Wilcoxon rank-sum tests. Proportions were compared using χ2 or two-proportion Z tests. All tests were two-tailed, with P < .05 considered statistically significant.
Results
A total of 203 ICAPs were sampled across both study phases, yielding 406 total samples. During Phase 1, 50 ICAPs were sampled following routine disinfection by clinical staff, resulting in 100 samples. During Phase 2, 153 ICAPs were enrolled, including 73 randomized to enhanced disinfection and 80 randomized to routine disinfection, yielding 306 samples.
Phase 1: baseline contamination following routine disinfection
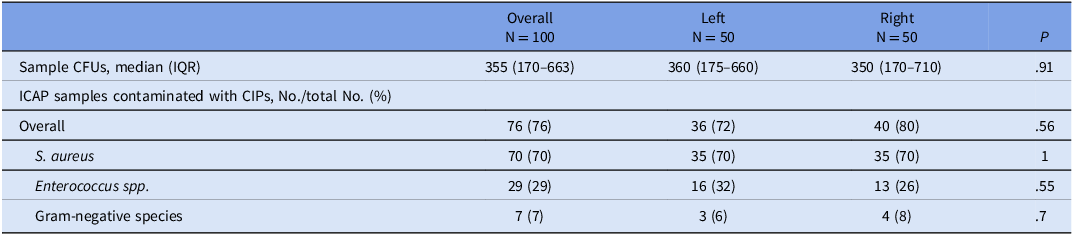
Following routine disinfection performed by clinical staff the median bioburden across all phase 1 samples was 355 CFUs (IQR,170–663), with no significant differences observed between left and right surfaces (P = .91).
At the sample level, 76 (76 %) samples were positive for at least one CIP. S. aureus was the most frequently recovered organism, detected in 70(70%) samples, followed by Enterococcus spp. 29 (29%) and gram-negative species 7 (7%). No significant differences in CIP recovery were observed between left and right surfaces (Table 1).
Bioburden in colony-forming units (CFU) and recovered clinically important pathogens (CIPs) after clinical use of inpatient clinician-assigned phones after standard Disinfection

Phase 2: routine versus enhanced disinfection
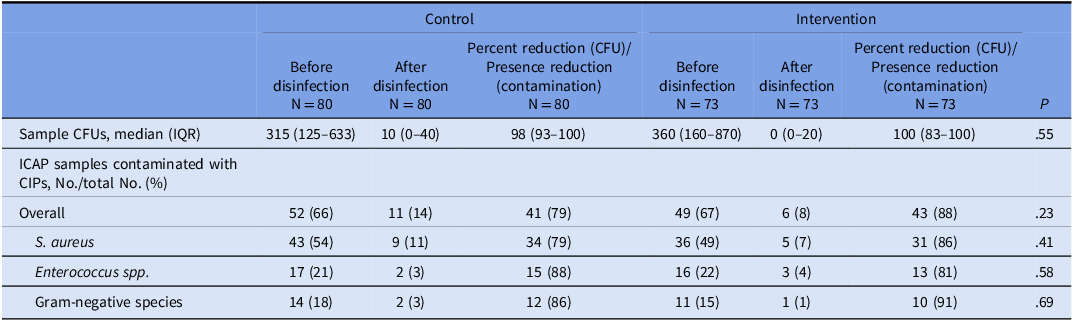
Among ICAPs randomized to enhanced disinfection, median CFUs decreased from 360 (IQR,160–870) before disinfection to 0 (IQR,0–20) after disinfection, representing a median 100% [IQR,83–100] reduction in bioburden. Among ICAPs randomized to routine disinfection, median CFUs decreased from 315 (IQR,125–633) before disinfection to 10 (IQR, 0–40) after disinfection, representing a median 98% [IQR,93–100] reduction in bioburden. The magnitude of CFU reduction did not differ significantly between enhanced and routine disinfection (P = .55).
Prior to disinfection, CIPs were recovered from 52 (66%) of control samples and 49 (67%) of intervention samples. Following disinfection, the proportion of samples positive for CIPs decreased to 11 (14%) in the routine disinfection arm and 6 (8%) in the enhanced disinfection arm. Reductions in CIP recovery were observed for S. aureus, Enterococcus spp., and gram-negative species in both study arms. The magnitude of reduction in CIP presence did not differ significantly between enhanced and routine disinfection for overall CIPs or for individual organism groups (all P > .05) (Table 2).
Bioburden in colony-forming units (CFU) and recovered clinically important pathogens (CIPs) after clinical use of inpatient clinician-assigned phones (ICAPs) before and after enhanced disinfection and before and after standard disinfection

Discussion
In this randomized controlled study of ICAPs in a pediatric surgery unit, both disinfection strategies resulted in marked reductions in microbial bioburden and recovery of CIPs. Enhanced disinfection using the SONOPRO cabinet was as highly efficacious as routine disinfection when both were applied by the study team.
In contrast, Phase 1 demonstrated substantially higher residual contamination following routine disinfection performed by clinicians in routine practice. This discrepancy suggests that adherence to established disinfection protocols, rather than intrinsic disinfectant efficacy, is a primary determinant of ICAP contamination. Although enhanced disinfection did not demonstrate microbiological superiority, automated systems may improve consistency by reducing variability in technique and time constraints. If implementation increases routine disinfection adherence, such systems may reduce contamination in practice despite equivalent laboratory performance.
This study has limitations. It was conducted in a single unit at a single institution, and patient-level outcomes were not assessed. Adherence to routine disinfection practices during Phase 1 was not directly measured, and, while the split-surface design enabled robust within-device comparisons, it may not fully represent whole-device contamination dynamics.
In summary, enhanced disinfection was equivalent to routine disinfection when standardized. However, high baseline contamination under routine practice highlights adherence as a critical driver of ICAP contamination. Future studies should evaluate whether automated systems improve real-world integration and sustained compliance.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/ice.2026.10460.
Acknowledgments
None.
Financial support
This study was funded by RESTEC Solutions, LLC, which manufactures the SONOPRO disinfection cabinet evaluated in this study.
Competing interests
None.