Introduction
The availability of long-acting injectable antipsychotics (LAIs) has transformed care for many individuals with schizophrenia, schizoaffective disorder, and bipolar disorder. The “guaranteed delivery” of a medication in a predictable and steady way can decrease the risk of relapse, rehospitalization, as well as reduce mortality.Reference Kishimoto, Hagi and Kurokawa 1 , Reference Correll, Solmi and Croatto 2 The revised American Psychiatric Association guidelines for the care of people with schizophrenia call out that LAIs should be considered in instances of poor or uncertain adherence, as well as for patients who would prefer this modality.Reference Keepers, Fochtmann and Anzia 3 The guidelines further add that LAIs can be useful in the transition from inpatient to outpatient care. Research has also demonstrated better outcomes when LAIs are used early in the disease course.Reference Subotnik, Casaus and Ventura 4 –Reference Tiihonen, Haukka and Taylor 6 An important benefit for providers of care is that LAI antipsychotics can eliminate the guesswork about adherence status and allow the clinician to focus on other reasons why symptoms may be exacerbated, such as psychosocial stressors or substance use. In the end, “preventing a relapse today can make a difference for a lifetime.”Reference Citrome 7
Of pragmatic concern is the limited array of molecules available as an LAI formulation. However, the formulations themselves differ, including several that contain the same molecule (Tables 1 and 2). This article reviews the evidence regarding the agents available as LAIs and considerations when selecting one over the other.
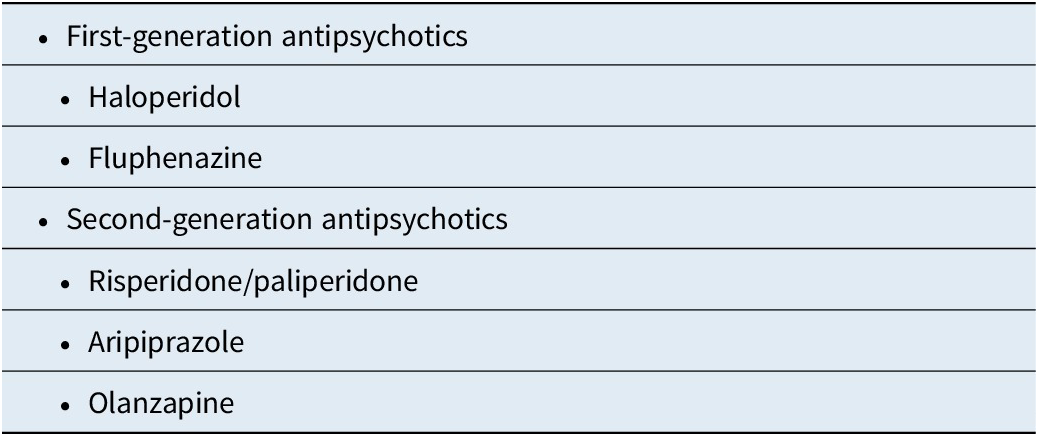
Antipsychotic Molecules with Long-Acting Formulations Available/Approved in the United States as of April 2025

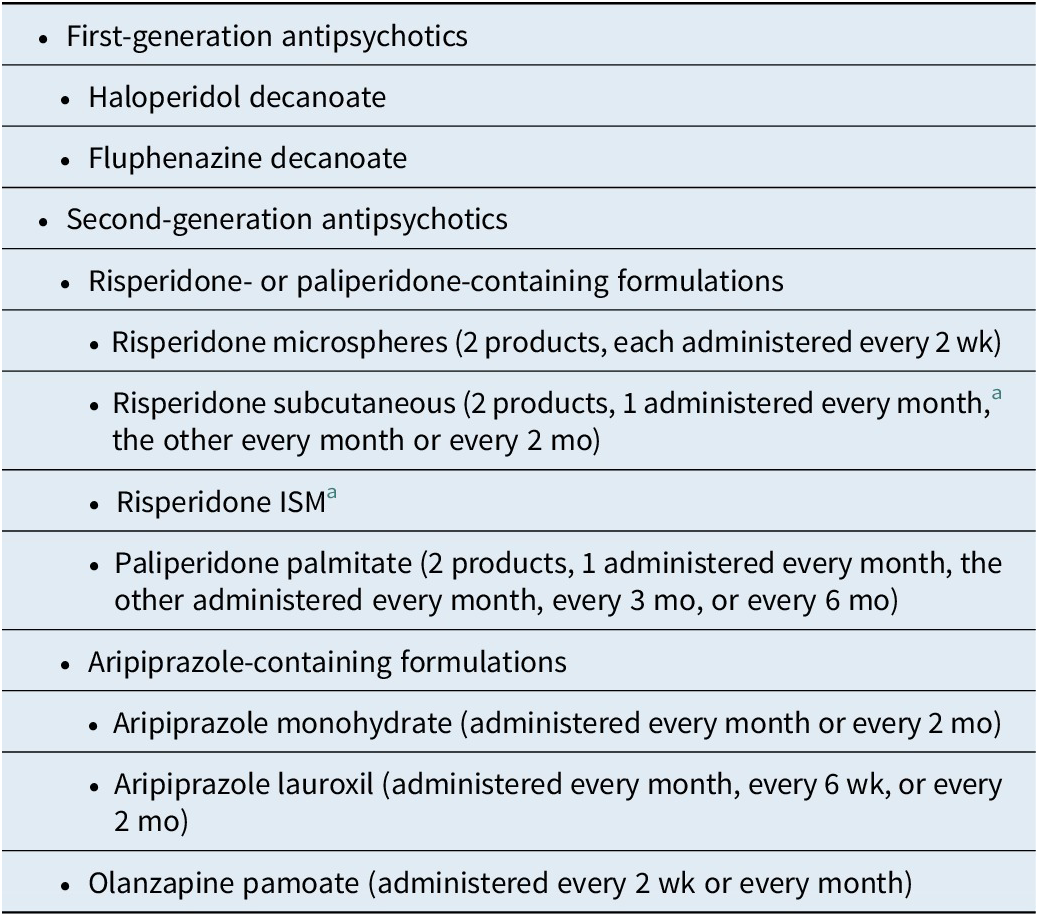
Formulations of Long-Acting Injectable Antipsychotics Approved in the United States as of April 2025

a Discontinued or never commercialized in the United States.
What are the molecules available as long-acting injectable antipsychotics?
Both first- (typical) and second- (atypical) generation antipsychotics are available as LAI formulations. However, the choice among molecules is currently limited to very few when compared to the plethora of oral treatments available for schizophrenia and bipolar disorder. In the United States, the clinician can choose among haloperidol, fluphenazine, risperidone/paliperidone, aripiprazole, and olanzapine.Reference Citrome 7 Haloperidol and fluphenazine are first-generation antipsychotics and are often co-administered with anticholinergic agents in order to prophylactically address drug-induced parkinsonism and acute dystonia, as well as actively treat these adverse effects when they occur. Not only does this strategy increase pill burden, but anticholinergic load is also associated with cognitive impairment.Reference Vanegas-Arroyave, Caroff and Citrome 8 –Reference Vinogradov, Fisher and Warm 10 Moreover, risk for developing tardive dyskinesia is greater for first-generation than for second-generation antipsychotics.Reference Carbon, Kane and Leucht 11 , Reference Carbon, Hsieh and Kane 12 Other first-generation LAI antipsychotics have been available outside the United States (eg, flupentixol, perphenazine, pipotiazine, and zuclopenthixol).Reference Taylor 13
Drug-induced parkinsonism and the need for anticholinergic medications can be minimized by using second-generation LAI antipsychotics. Among the second-generation antipsychotics, the molecules available in the United States include risperidone/paliperidone, aripiprazole, and olanzapine. These second-generation antipsychotics appear similar in terms of efficacy, with the possible exception of olanzapine, which may be more efficacious.Reference Meftah, Deckler and Citrome 14 , Reference Citrome, McEvoy and Todtenkopf 15 Of note is that paliperidone is the principal active metabolite of risperidone and shares with it many of its characteristics.Reference Citrome 16 There is more heterogeneity regarding tolerability than with efficacy,Reference Huhn, Nikolakopoulou and Schneider-Thoma 17 with liability for weight gain and metabolic adverse effects being more prominent for olanzapine, followed by risperidone, and aripiprazole; prolactin elevation with risperidone/paliperidone, followed by olanzapine, and aripiprazole; and somnolence with olanzapine, followed by risperidone, and aripiprazole.
A major treatment conundrum is the prescribing of an oral medication that appears efficacious and tolerable, but that does not have a corresponding long-acting formulation. Considerations when switching to an LAI antipsychotic will include the uncertainty of adequate response to a different molecule delivered by injection, balanced by the issues around poor or uncertain adherence with an oral agent. A not uncommon scenario is the patient who appears not to respond to oral antipsychotics and is thus considered potentially “treatment-resistant”.Reference Faden and Citrome 18 Care should be taken to rule out pseudo-resistance, which can confound the assessment of a patient who is covertly nonadherent, in which case a trial of a LAI antipsychotic is recommended to determine if the person is truly treatment resistant.Reference Howes, McCutcheon and Agid 19
A complete discussion of the similarities and differences among the oral formulations of risperidone/paliperidone, aripiprazole, and olanzapine (and others) is beyond the scope of this review. The reader is referred to a number of meta-analyses and other reviews that serve to compare and contrast,Reference Huhn, Nikolakopoulou and Schneider-Thoma 17 , Reference Volavka and Citrome 20 as well as indirect comparisons using the metrics of number needed to treat and number needed to harm (Ref.,Reference Citrome and Ketter 21 and also see Figure 1 and Table 6 as published in Ref. Reference Citrome, Neugebauer, Meli and Kando 22).
What are the formulations available?
Older first-generation antipsychotics such as fluphenazine decanoate (administered generally every 2 weeks) and haloperidol decanoate (administered generally every 4 weeks) remain available.Reference Citrome 23 They are relatively inexpensive, and haloperidol decanoate remains frequently prescribed. Both fluphenazine decanoate and haloperidol decanoate can be injected in either the deltoid or gluteal muscle, and fluphenazine decanoate can also be injected subcutaneously, although this is not commonly done.Reference Eugia 24 The first-generation antipsychotic LAIs are dissolved in sesame seed oil and can be more challenging to inject than the second-generation intramuscular LAI formulations, which are all suspended in water.
For fluphenazine decanoate, the initial dose is 12.5–25 mg and can be increased in increments of 12.5 mg, but the total amount administered at one time should not exceed 100 mg.Reference Citrome 23 , Reference Eugia 24 The initial dose for haloperidol decanoate should be 10–20 times the previous daily dose in oral haloperidol equivalents, but the initial injection is limited to 100 mg, followed by the balance 3–7 days later. The usual maintenance range is 10–15 times the previous daily dose in oral haloperidol equivalents, depending on clinical response.Reference Citrome 23 , 25 Clinical experience with haloperidol decanoate at doses greater than 450 mg/month is limited. 25 In order to reduce injection volume, a more concentrated formulation (100 mg/ml versus 50 mg/ml) is available. 25
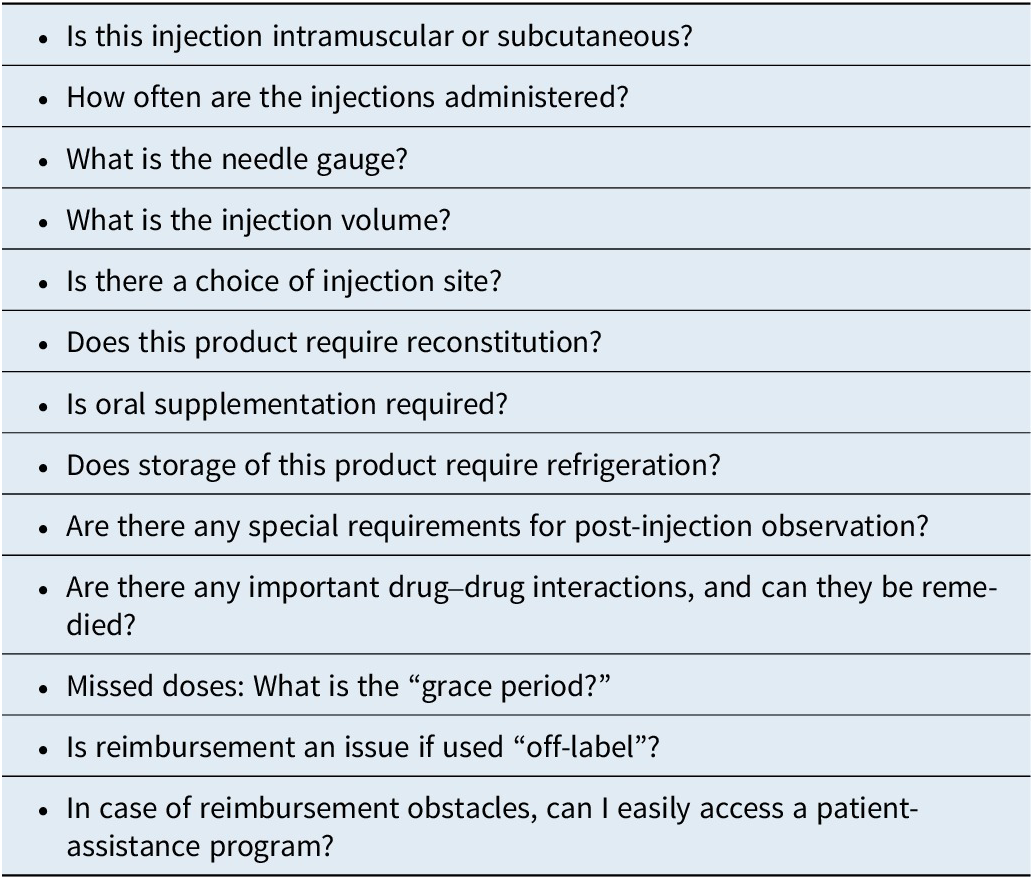
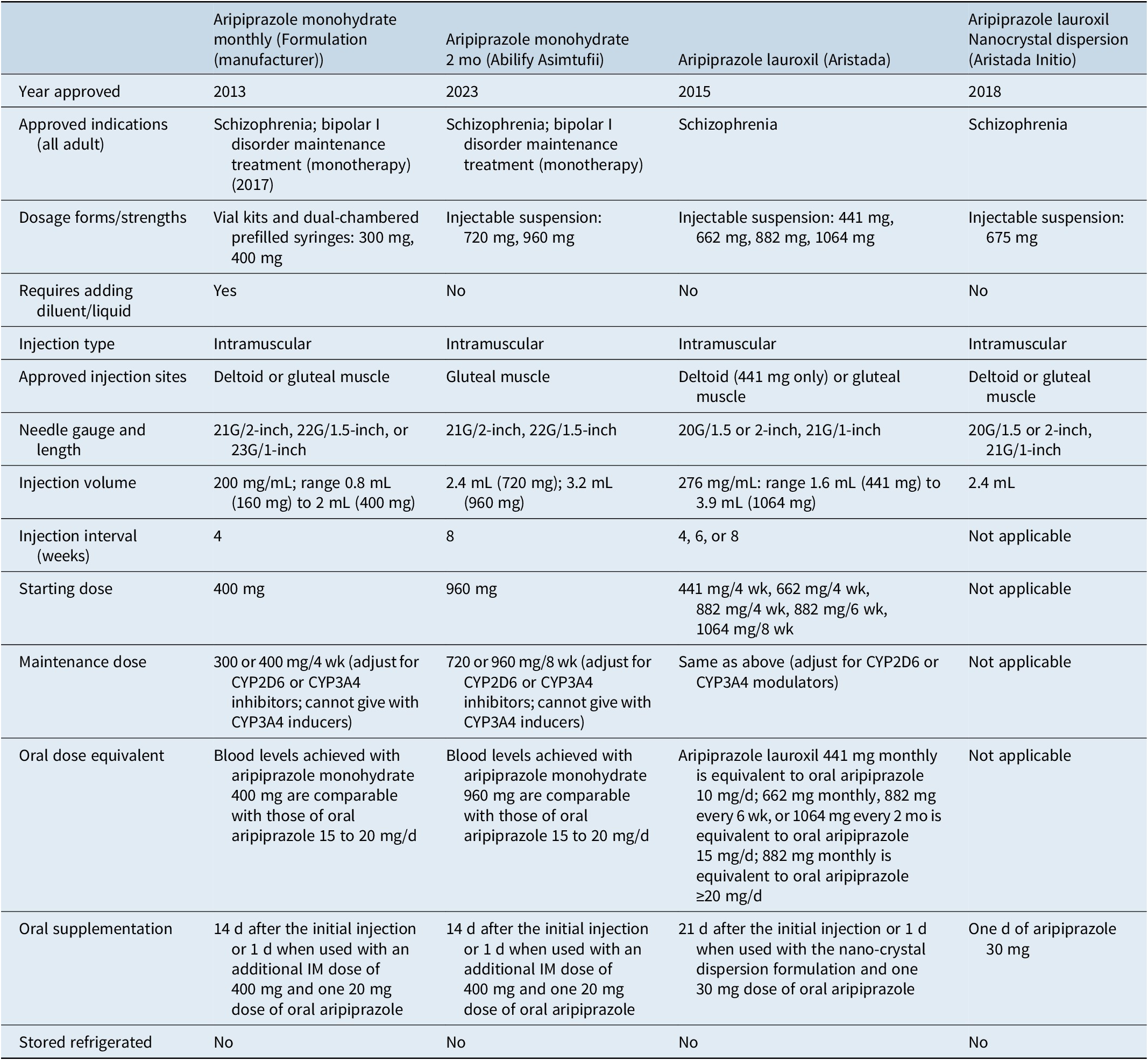
Among the 3 different second-generation antipsychotics currently available in LAI formulations, risperidone/paliperidone, aripiprazole, and olanzapine, several choices are currently available for risperidone/paliperidone and aripiprazole, and currently only 1 for olanzapine, although a subcutaneous formulation is in the late stage of development.Reference Correll, Ahn and Bar-Nur 26 The different formulations containing the same active molecule can be differentiated in terms of their “amenities of care” such as dosing intervals (from every 2 weeks to every 6 months), availability of different dose strengths, choice of injection site (deltoid muscle, gluteal muscle, subcutaneous), gauge and length of the needle, injection volume, storage and reconstitution requirements, need for oral supplementation, guidance regarding early or late dosing, approved indication, and requirement for observation post injection.Reference Citrome 7 A list of these pragmatic considerations is contained in Table 3.
Long-Acting Injectable Antipsychotics: “Amenities of Care” (Adapted from Ref.Reference Citrome 7)

Long-acting injectable risperidone and paliperidone formulations, as approved by the US Food and Drug Administration
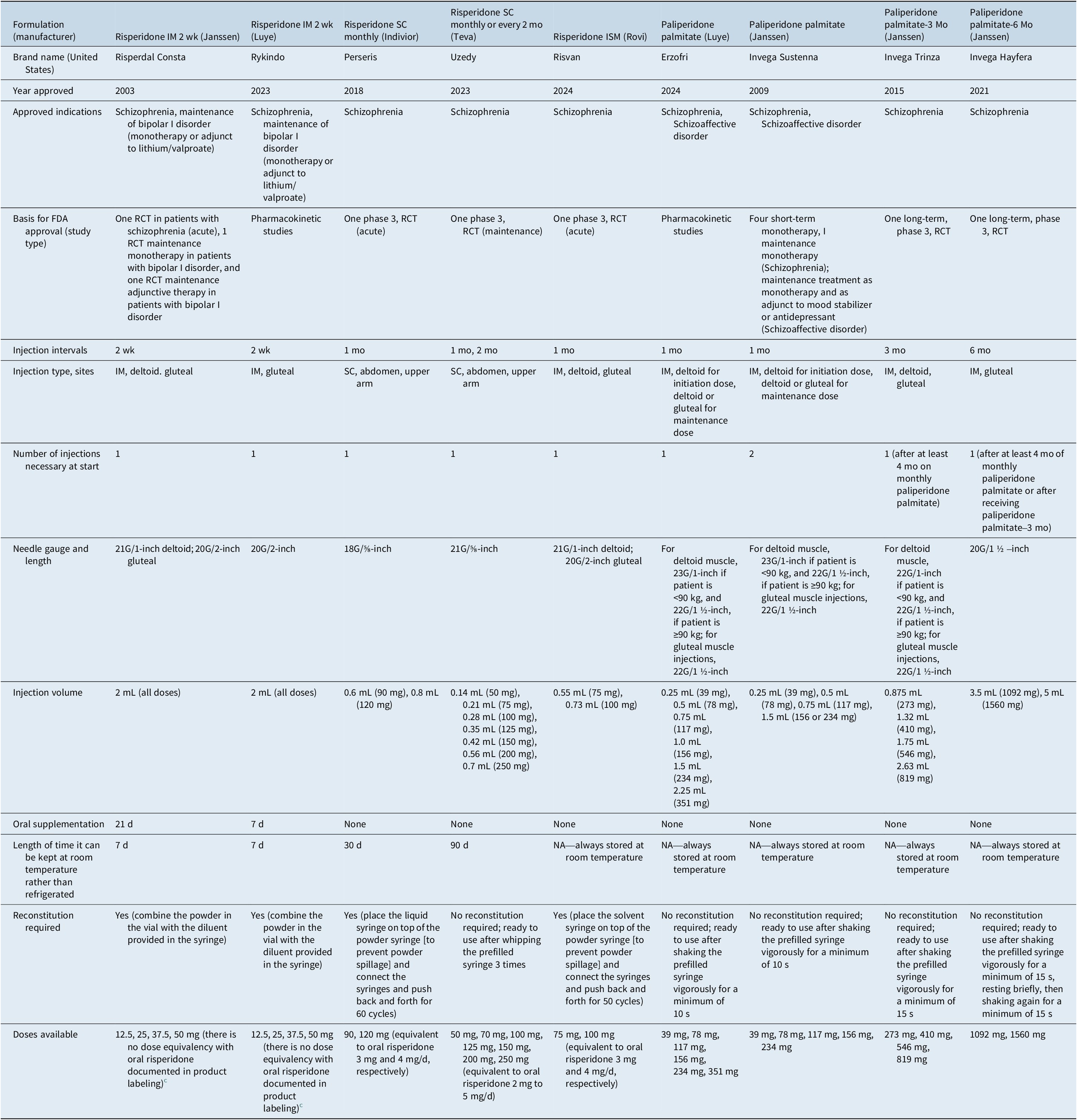
Table 4 outlines the 9 different formulations approved by the US Food and Drug Administration (FDA) that contain risperidone or paliperidone (2 are either discontinued or never commercialized in the United States). They differ broadly in approved indications, dosage forms/strengths, reconstitution requirements, injection sites and method of injection (intramuscular versus subcutaneous), needle gauge/length, injection volume, injection interval, requirement for oral supplementation, need for refrigeration when stored, and instructions for early or late dosing.
Long-Acting Injectable Risperidone and Paliperidone Formulations, as Approved by the US Food and Drug Administration (Adapted from Table 2 in Faden J, Citrome L. A new paliperidone palmitate formulation: how is it different and where does it fit in our array of choices for long-acting formulations of risperidone and paliperidone? Curr Med Res Opin. 2025;41(4):663–666. doi:10.1080/03007995.2025.2482654. Open Access distributed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/)

Abbreviations. IM, intramuscular; SC, subcutaneous; RCT, randomized controlled trial.
a On July 9, 2024, the manufacturer announced the cessation of all sales and marketing activities regarding this product, but will continue to supply it for the foreseeable future to avoid disruption to patient care (see https://otp.tools.investis.com/clients/uk/indivior2/ms/regulatory-story.aspx?newsid=1 840 448).
b On November 7, 2024, the manufacturer announced that the product will not be marketed in the United States and will focus on European development (https://www.rovi.es/en/content/first-nine-months-2024-results-press-release).
c Product labeling states that the recommended dose for the treatment of schizophrenia is 25 mg IM Q2W; patients not responding to 25 mg may benefit from 37.5 mg or 50 mg.
Source: Adapted from Faden et al.Reference Faden and Citrome [44]
In 2003, risperidone microspheres (Risperdal Consta, Janssen) became the first second-generation antipsychotic to be available in a LAI formulation at doses of 12.5, 25, 37.5, or 50 mg, administered every 2 weeks.Reference Ehret and Fuller 27 , Reference Harrison and Goa 28 In addition to the indication for schizophrenia, in 2009, risperidone microspheres was also approved as monotherapy or as adjunctive therapy to lithium or valproate for the maintenance treatment of bipolar I disorder.Reference Citrome 29 , 30 Of note, storage of the product requires refrigeration, but it can remain at room temperature for up to 7 days. 30 Because the main release of the drug does not begin until 3 weeks after administration, supplemental oral risperidone is required for 21 days after the first injection (hence overlapping the second injection) and after any dose increase. 30 After mixing the risperidone microspheres powder with the supplied aqueous diluent, it can be administered in the deltoid or gluteal muscle. The recommended starting dose is 25 mg/2 weeks, and the maximum recommended dose is 50 mg/2 weeks. With respect to total exposure, injections of 25, 50, or 75 mg every 2 weeks were found to be equivalent to daily oral doses of 2, 4, or 6 mg of risperidone, respectively.Reference Eerdekens, Van Hove and Remmerie 31
In 2023, another formulation of risperidone microspheres (Rykindo, Luye) from a different pharmaceutical company was approved by the FDA for the same indications as the original. 32 Approval was based on a pharmacokinetic study that demonstrated similar plasma levels as the original product.Reference Faden, Ramirez and Martinez 33 Although both formulations of risperidone microspheres share many common characteristics, they do differ in their pharmacokinetic profile. This newer version requires only 1 week of oral supplementation and is currently only approved for gluteal injection. The dosing recommendations for the 2 risperidone microsphere products are otherwise identical, save for the original formulation, where upward dose adjustment of risperidone microspheres (Janssen) should not be made more frequently than every 4 weeks and that the clinical effects of this dose adjustment should not be anticipated earlier than 3 weeks after the first injection with the higher dose. 30 When switching to the newer formulation (Luye), the dose should be the same as that of the previous treatment, with the first injection given 4 weeks (no later than 5 weeks) after the last injection of the previous treatment. 32
Three more novel risperidone formulations have been approved by the FDA for the treatment of schizophrenia, and importantly, do not require oral supplementation upon initiation: a subcutaneous preparation 34 that can be administered monthly but requires reconstitution (Perseris, Indivior; approved in 2018 and now discontinued 35 and thus not described here in detail), a subcutaneous preparation (Uzedy, Teva, referred to later as 1-month/2-month risperidone subcutaneous) that comes in a prefilled syringe and that can be administered on Day 1 either every month or every 2 months (approved in 2023, 36 and described below), and an intramuscular formulation that requires reconstitution and can be administered monthly (Risvan, Rovi); although FDA approved in 2024, this formulation is currently not anticipated to be available commercially in the United States and thus not described here in detail.Reference Faden, Ramirez and Martinez 33 , 37
One-month/2-month risperidone subcutaneous is available to match oral risperidone doses of 2, 3, 4, and 5 mg/d, with once monthly 50, 75, 100, and 125 mg, and 2-month 100, 150, 200, and 250 mg, respectively. 36 Oral supplementation or a loading dose is not required upon initiation at any dose. This formulation can be stored at room temperature for up to 90 days; otherwise, it must be refrigerated. 36
The product labels for the risperidone formulations contain similar advice that a dose adjustment may be necessary in the presence of drug–drug interaction(s) with CYP2D6 and/or CYP3A4 inhibitors and/or if the patient is a CYP2D6 poor metabolizer, or if the patient is taking a CYP3A4 inducer. 30 , 32 , 34 , 36 , 37
Paliperidone (9-OH risperidone) is the main active metabolite of risperidone, and a once-monthly injectable formulation (Invega Sustenna, Janssen) became available in the United States in 2009. 38 –Reference Greenberg and Citrome 40 In contrast to risperidone microspheres, which must be reconstituted as well as stored in a refrigerator, paliperidone palmitate is an aqueous suspension that comes in prefilled syringes and does not require refrigeration. Paliperidone palmitate has relatively small needle bores to select from. Instead of using oral supplementation, the initiating doses are all by injection: 234 mg on treatment day 1 and 156 mg 1 week later (± 4 days), both administered in the deltoid muscle. Although the recommended monthly maintenance dose is 117 mg for the treatment of schizophrenia, the maintenance dose can be within the range of 39–234 mg, equivalent to the dose range of 3–12 mg/d for oral paliperidone. 38 When converting from oral risperidone to paliperidone palmitate, oral risperidone doses of 1, 2, 3, 4, and 6 mg/d result in similar exposures as 39, 78, 117, 156, and 234 mg of paliperidone palmitate, respectively.Reference Russu, Kern Sliwa and Ravenstijn 41 The regular monthly maintenance doses can be administered in either the deltoid or the gluteal muscle. The product label recommends avoiding the use of a strong inducer of CYP3A4 and/or P-glycoprotein, such as carbamazepine, during a dosing interval. In addition to the indication for treatment of schizophrenia, paliperidone palmitate once monthly received approval in 2014 for use in schizoaffective disorder as monotherapy or as an adjunct to mood stabilizers or antidepressants.Reference Citrome 29 , Reference Greenberg and Citrome 40 , Reference Chue and Chue 42
In 2024, a new formulation of paliperidone palmitate (Erzofri, Luye) was approved in the United States for the treatment of schizophrenia and schizoaffective disorder 43 , Reference Faden and Citrome 44 and differs from the original in that it can be initiated with a single 351 mg dose administered in the deltoid muscle. Approval was based on a pharmacokinetic study,Reference Faden and Citrome 44 and this new formulation carries the identical dosing recommendations after initiation as the original. 38 , 43
An extension to the monthly injection interval became available in 2015 in the form of a 3-month formulation of paliperidone palmitate (Invega Trinza, Janssen). The indication is restricted to schizophrenia and for individuals who have been treated with the once-monthly formulation of paliperidone palmitate for ≥4 months.Reference Citrome 29 , 45 The 3-month formulation is packaged in water-based prefilled syringes; however, the product is denser than the once-monthly formulation and has a larger particle size.Reference Ravenstijn, Remmerie and Savitz 46 The doses that are available remain sufficiently small in volume so that they can be administered in the deltoid muscle, although gluteal injection remains an option. Dose for the 3-month formulation is calculated by multiplying the once-monthly dose by 3.5, and is available for the dose equivalent of 78, 117, 156, and 234 mg of once-monthly paliperidone palmitate. The 3-month formulation requires the use of special-purpose thin-walled needles that come packaged with the product, and these needles cannot be interchanged with those supplied with the once-monthly formulation or with other regular commercially available needles. A 6-month version of paliperidone palmitate (Invega Hafyera, Janssen) was approved in 2021 47 , Reference Faden and Citrome 48 with doses of 1092 and 1560 mg, equivalent to 156 and 234 mg of the once-monthly formulation, and 546 and 819 mg of the 3-month formulation, respectively. Similar to the 3-month formulation, the 6-month formulation is restricted to schizophrenia and for individuals who have been treated with the once-monthly formulation of paliperidone palmitate for ≥4 months, although it is possible to go from the 3-month to the 6-month formulation at the time of the next scheduled injection. 47
Long-acting injectable aripiprazole formulations, as approved by the US Food and Drug Administration
Table 5 outlines the 4 different formulations available that contain aripiprazole, with one of them (aripiprazole lauroxil nanocrystal dispersion) reserved for the initiation of aripiprazole lauroxil.Reference Citrome 29 , Reference Citrome 49 –Reference Hard, Wehr and Du 56 Principal differences between the formulations include approved indications, dosage forms/strengths, reconstitution requirements, injection sites, needle gauge/length, injection volume, injection interval, requirement for oral supplementation, concomitant use instructions with CYP3A4 inducers, and instructions for early or late dosing.
Long-Acting Injectable Aripiprazole Formulations, as Approved by the US Food and Drug Administration (Adapted from Ref.Reference Citrome 7)

Aripiprazole monohydrate (Abilify Maintena, Otsuka/Lundbeck) was approved in the United States in 2013.Reference Citrome 23 , Reference Citrome 29 , Reference Citrome 49 , 52 In addition to being approved for the treatment of schizophrenia, aripiprazole monohydrate was approved in 2017 for maintenance monotherapy treatment of bipolar I disorder.Reference Citrome 29 , 52 Following reconstitution with water using either a 300 or 400 mg vial kit for doses as low as 160 mg, or prefilled dual-chambered syringes for the 300 and 400 mg dose strengths, the monthly injection can be administered in either the deltoid or gluteal muscle. The recommended initial and maintenance doses are 400 mg, although a reduction to 300 mg can be considered to manage tolerability concerns. Starting doses of 300, 200, and 160 mg are intended to be used in the presence of drug–drug interaction(s) with CYP2D6 and/or CYP3A4 inhibitors and/or if the patient is a CYP2D6 poor metabolizer. Use is to be avoided in the presence of a drug–drug interaction with a CYP3A4 inducer. There are 2 ways to initiate treatment with aripiprazole monohydrate: a 1-day initiation where 2 separate intramuscular injections of 400 mg (or 300 mg if there is a drug–drug interaction with a CYP2D6 or CYP3A4 inhibitor or if the patient is a CYP2D6 poor metabolizer) is administered along with a single oral dose of aripiprazole 20 mg; or a 14-day initiation where in conjunction with the first administration of aripiprazole monohydrate 400 mg (or 300 mg if there is a drug–drug interaction with a CYP2D6 or CYP3A4 inhibitor or if the patient is a CYP2D6 poor metabolizer), 14 consecutive days is required of either oral aripiprazole (10–20 mg) or the current oral antipsychotic. Blood levels achieved with aripiprazole monohydrate 400 mg are comparable with those of oral aripiprazole 15–20 mg/d.Reference Raoufinia, Peters-Strickland and Nylander 57 The 1-day initiation regimen was approved by the FDA in 2025, after having been available in Europe since 2020.Reference Fagiolini, Leopold and Pappa 58
In 2023, a 2-month formulation of aripiprazole monohydrate (Abilify Asimtufii, Otsuka/Lundbeck) was approved for the treatment of schizophrenia and for the maintenance treatment of bipolar I disorder. 53 Approval was based on a pharmacokinetic bridging study comparing the 2-month and once-monthly formulations,Reference Harlin, Yildirim and Such 59 –Reference Citrome, Such and Yildirim 61 and legacy data regarding the once-monthly formulation. 53 On average, 960 mg of the 2-monthly formulation provides sustained plasma concentrations comparable to 400 mg of the once-monthly formulation, but over a period of 2 months. In contrast to the once-monthly preparation, it is packaged in a ready-to-use prefilled syringe. The available doses of 960 and 720 mg are equivalent to the monthly doses of 400 and 300 mg, respectively. Lower dose strengths are not available. Injection is into the gluteal muscle only. 53 The 2-month formulation can be started on Day 1 by either administering an additional injection 400 mg (or 300 mg if there is a drug–drug interaction with a CYP2D6 or CYP3A4 inhibitor or if the patient is a CYP2D6 poor metabolizer) of the monthly formulation in a separate gluteal or deltoid muscle along with 1 dose of oral aripiprazole 20 mg; or a 14-day initiation can be used where in conjunction with the first administration of the 2-month formulation, 14 consecutive days is required of either oral aripiprazole (10–20 mg) or the current oral antipsychotic. For patients already receiving the monthly formulation, the 2-month formulation can be given at the time of the next scheduled injection. Of note, the 2-month formulation can be administered within a window of 2 weeks before to 2 weeks after the scheduled date. 53
Aripiprazole lauroxil (Aristada, Alkermes) is a different aripiprazole LAI formulation that was approved in the United States in 2015.Reference Citrome 29 , Reference Citrome 49 , 54 , Reference Citrome, Correll and Cutler 62 Aripiprazole lauroxil is supplied in prefilled syringes as an aqueous suspension. 54 Once injected into the deltoid muscle (approved for the 441 mg dose) or gluteal muscle (approved for any of the doses), the conversion of aripiprazole lauroxil to aripiprazole is governed by the slow dissolution of aripiprazole lauroxil and subsequent enzyme-mediated cleavage by esterases. When the product was launched, dose strengths of 441, 662, and 882 mg were initially available. These doses, when administered monthly, yield exposures to aripiprazole equivalent to oral aripiprazole 10, 15, and ≥ 20 mg/d, respectively. The dose of 882 mg administered every 6 weeks yields similar exposures as 662 mg administered monthly. In 2017, a dose strength of aripiprazole lauroxil 1064 mg administered every 2 months became available and yields equivalent exposures as 662 mg monthly or 882 mg every 6 weeks.Reference Citrome, Correll and Cutler 62 , Reference Sommi, Rege and Wehr 63 Instructions for the use of aripiprazole lauroxil suggest that any dose can be initiated on Day 1, including 1064 mg every 2 months. Selection of a dose is also dependent on the presence of drug–drug interaction(s) with CYP2D6 and/or CYP3A4 inhibitors and/or if the patient is a CYP2D6 poor metabolizer, or if the patient is taking a CYP3A4 inducer. 54 Initiation of aripiprazole lauroxil requires either 21 days of supplementation with oral aripiprazole or the use of the aripiprazole lauroxil nanocrystal dispersion (ALNCD) formulation (Aristada Initio, Alkermes), available since 2018.Reference Ehret, Davis and Luttrell 50 , Reference Hard, Wehr and von Moltke 51 , 54 –Reference Hard, Wehr and Du 56 ALNCD contains smaller particles than those used in standard aripiprazole lauroxil, and these particles have faster dissolution properties when injected into the muscle.Reference Hard, Wehr and von Moltke 51 An injection of the 675 mg ALNCD formulation into either the deltoid or gluteal muscle, plus administration of oral aripiprazole 30 mg that same day, can substitute for the 21 days of oral supplementation that would otherwise be required upon initiation.Reference Hard, Wehr and Du 56 The first injection of standard aripiprazole lauroxil may be administered on the same day as the ALNCD formulation or up to 10 days thereafter. 54 , 55 ALNCD is available in only 1 dose strength, thus its use is not possible in the presence of potential drug–drug interactions, such as with strong CYP2D6 or CYP3A4 inhibitors and strong CYP3A4 inducers, or if the patient is a CYP2D6 poor metabolizer. 54 , 55
Long-acting injectable olanzapine formulations, as approved by the US Food and Drug Administration
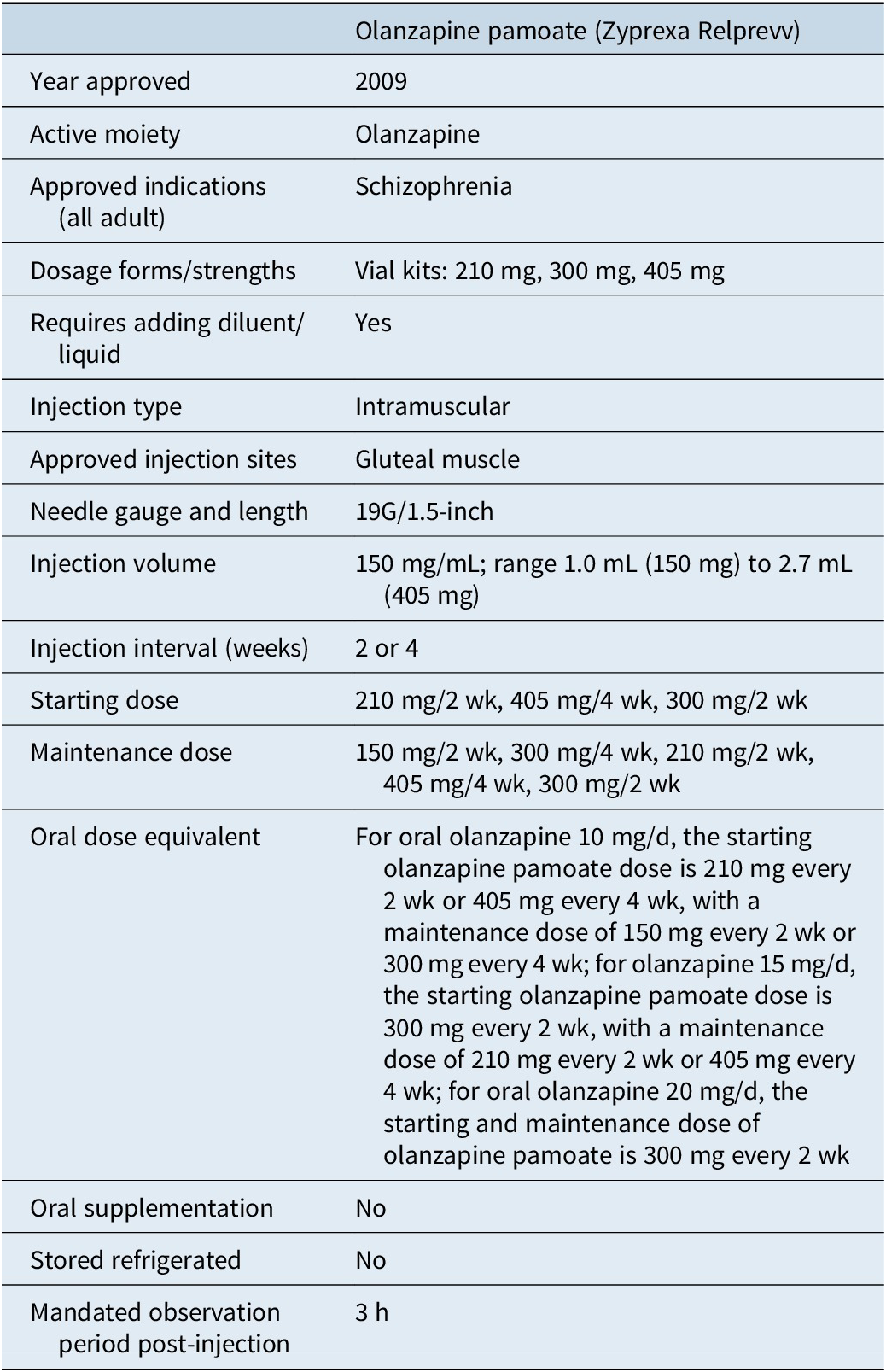
Table 6 outlines the characteristics of olanzapine pamoate (Zyprexa Relprevv, Eli Lilly). 64 –Reference Citrome 66 There are currently no alternative LAI formulations of olanzapine commercially available, although a subcutaneous formulation is in late stage of development.Reference Correll, Ahn and Bar-Nur 26 Olanzapine pamoate was approved in the United States in 2009. It differs from the other LAI antipsychotics in that its use is governed by a Risk Evaluation and Mitigation Strategy program (REMS), requiring a 3-hour post-injection monitoring period after each injection.Reference Citrome 66 This is to better manage the potential risk of Post-injection Delirium Sedation Syndrome (PDSS), as described below, a risk that appears to be absent with the experimental subcutaneous formulation.Reference Krtalic, Juretic and Komlosi 67
Long-Acting Injectable Olanzapine Formulations, as Approved by the US Food and Drug Administration (Adapted from Ref.Reference Citrome 7)

Olanzapine pamoate is a crystalline salt formulation composed of olanzapine and pamoic acid. 64 , Reference Citrome 65 After reconstitution in water, it is injected into the gluteal muscle, and the salt slowly dissolves, releasing olanzapine over a period of weeks. However, when olanzapine pamoate comes into contact with a substantial amount of blood or plasma, the salt dissolves more quickly, resulting in the release of a larger amount of olanzapine, potentially leading to PDSS characterized by sedation, confusion, slurred speech, altered gait, or unconsciousness. PDSS can be expected to occur in approximately 0.07% of injections and is time-limited but may require symptomatic treatment.Reference Detke, McDonnell and Brunner 68 , Reference Luedecke, Schöttle and Karow 69 Because there are no clear identifiable risk factors, a REMS was instituted, and olanzapine pamoate can only be provided at registered healthcare facilities, and patients must be monitored by appropriately qualified staff for at least 3 hours after injection. 64 , Reference Citrome 66 In addition, patients must be accompanied to their next destination upon leaving the facility. Because the risk of PDSS is cumulative, patients receiving olanzapine pamoate every 2 weeks can decrease their risk of PDSS by 50% by switching to monthly injections. PDSS is not common; from a provider perspective, a clinic with 60 patients receiving an injection every 2 weeks might expect approximately 1 event per year.Reference Detke, McDonnell and Brunner 68 Although all the other LAI antipsychotics have hypersensitivity listed as a contraindication, no contraindications are noted in the product label for olanzapine pamoate. 64
Initiation of olanzapine pamoate does not require oral supplementation; however, a higher dose is administered for the first injection, with the exception of the highest dose available. 64 The starting and maintenance dose is dependent on the dose of oral olanzapine required for stabilization: for patients requiring olanzapine 10 mg/d, the starting olanzapine pamoate dose is 210 mg every 2 weeks or 405 mg every 4 weeks, and then if clinically indicated, patients can be evaluated 2 months later for a reduction to a maintenance dose of 150 mg every 2 weeks or 300 mg every 4 weeks; for patients requiring oral olanzapine 15 mg/d, the starting olanzapine pamoate dose is 300 mg every 2 weeks, and then if clinically indicated, patients can be evaluated 2 months later for a reduction to a maintenance dose of 210 mg every 2 weeks or 405 mg every 4 weeks; for patients requiring oral olanzapine 20 mg/d, the recommended starting and maintenance dose of olanzapine pamoate is 300 mg every 2 weeks. 64
Acute treatment with long-acting injectable antipsychotics: what is the evidence?
Among the approved second-generation antipsychotic LAI formulations in the United States, efficacy in acutely exacerbated patients with schizophrenia has been formally evaluated for once-monthly paliperidone palmitate,Reference Pandina, Lindenmayer and Lull 70 –Reference Kramer, Litman and Hough 73 olanzapine pamoate,Reference Lauriello, Lambert and Andersen 74 aripiprazole monohydrate once-monthly,Reference Kane, Peters-Strickland and Baker 75 aripiprazole lauroxil,Reference Meltzer, Risinger and Nasrallah 76 risperidone subcutaneous injection (Indivior),Reference Nasser, Henderson and Fava 77 and risperidone IM (Rovi).Reference Correll, Litman and Filts 78 Although the initiation procedures vary among the different products, starting an LAI antipsychotic while a patient is hospitalized with an acute exacerbation of schizophrenia has consistently demonstrated robust superiority over placebo in reducing psychotic symptoms.Reference Vita, Pollini and Canozzi 79 Of additional interest is the ability to reduce hostility and agitation.Reference Citrome, Du, Risinger and Stankovic 80 , Reference Citrome and Volavka 81 For LAI formulations not formally assessed in an acute trial, but for which supporting data exists for that molecule, it would be expected that they too would be suitable for acute use (for example, 2-month aripiprazole monohydrate and 1-month/2-month risperidone subcutaneous injection).
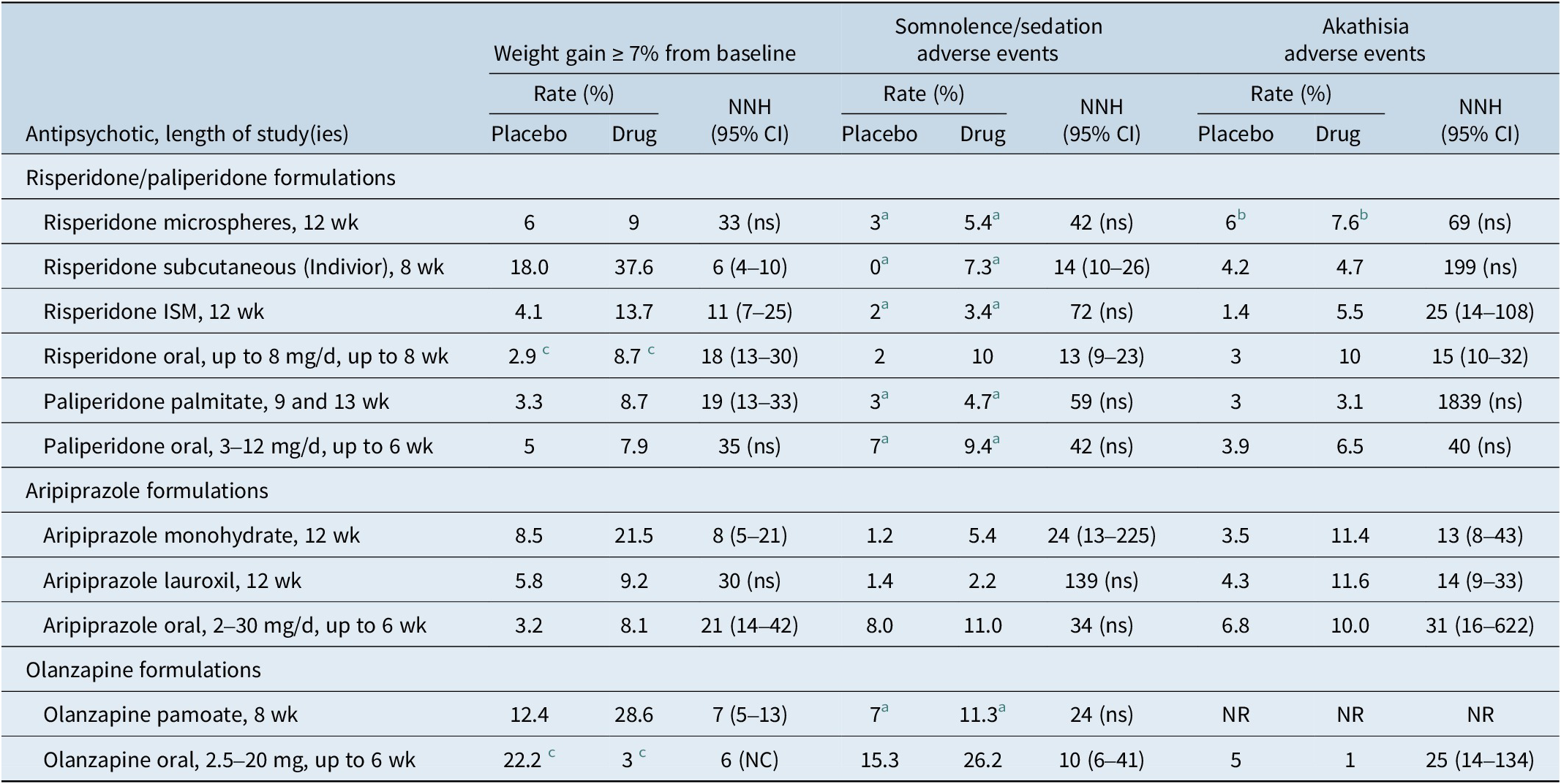
Aside from potential adverse effects related to the injection itself (such as pain, redness, induration, or nodule in case of subcutaneous injection), there are adverse reactions related to the molecule itself. Number needed to harm (NNH) versus placebo can be used to indirectly compare risk for weight gain, sedation, and akathisia (Table 7). 30 , 34 , 38 , 52 , 54 , 64 , Reference Correll, Litman and Filts 78 , Reference Citrome 82 – 87 NNH values less than 10 denote events that would be more commonly encountered; this would be the case for weight gain ≥7% from baseline for risperidone subcutaneous injection (Indivior), aripiprazole monohydrate, and olanzapine pamoate, as calculated from their short-term acute registration studies. The weight gain data is counter-intuitive for risperidone subcutaneous injection and aripiprazole monohydrate and appears to differ somewhat from what has been calculated from registration studies of the oral formulations of risperidone and aripiprazole, where the NNH versus placebo estimates for weight gain ≥7% were 18 and 21, respectively 84 , 86; this could be a reflection of study design, where patients remained hospitalized throughout the study and potential skewing of the characteristics of the study participants toward those more prone to weight gain.Reference Kane, Peters-Strickland and Baker 75 , Reference Nasser, Henderson and Fava 77 It has also been suggested that the acute use of LAI antipsychotics may be better tolerated than oral formulations.Reference Wang, Schneider-Thoma and Siafis 88
Rates and Number Needed to Harm versus Placebo for Weight Gain, Somnolence/Sedation, and Akathisia, for Approved Long-Acting Injectable Second-Generation Antipsychotics and Their Oral Counterparts in Adults as Observed in Acute Short-Term Studies of Long-Acting Injectable Antipsychotics for Schizophrenia (Doses Pooled) (Adapted from Ref.Reference Citrome 7)

a Pooled term of somnolence/sedation as reported in the product label.
b Pooled term of akathisia/restlessness as reported in the product label.
c Pooled schizophrenia and bipolar as reported in the product label.
CI, confidence interval; NC, the 95% CI is not calculable as denominators were not provided in product labeling; NR, not reported (did not meet threshold for reporting); NNH, number needed to harm; ns, not significant at the p < 0.05 threshold and thus the 95% CI is not shown.
Prevention of relapse with long-acting injectable antipsychotics: what is the evidence?
Although LAI antipsychotics can be used acutely, LAI antipsychotics are more often considered as part of a long-term treatment strategy to minimize the risk of relapse. Real-world prospective and retrospective studies comparing LAI antipsychotics versus oral antipsychotics generally demonstrate decreases in relapse, hospitalization, and all-cause discontinuation for patients receiving LAI antipsychotics,Reference Kishimoto, Hagi and Kurokawa 1 , Reference Kirson, Weiden and Yermakov 89 as well as a decrease in all-cause mortality.Reference Aymerich, Salazar de Pablo and Pacho 90
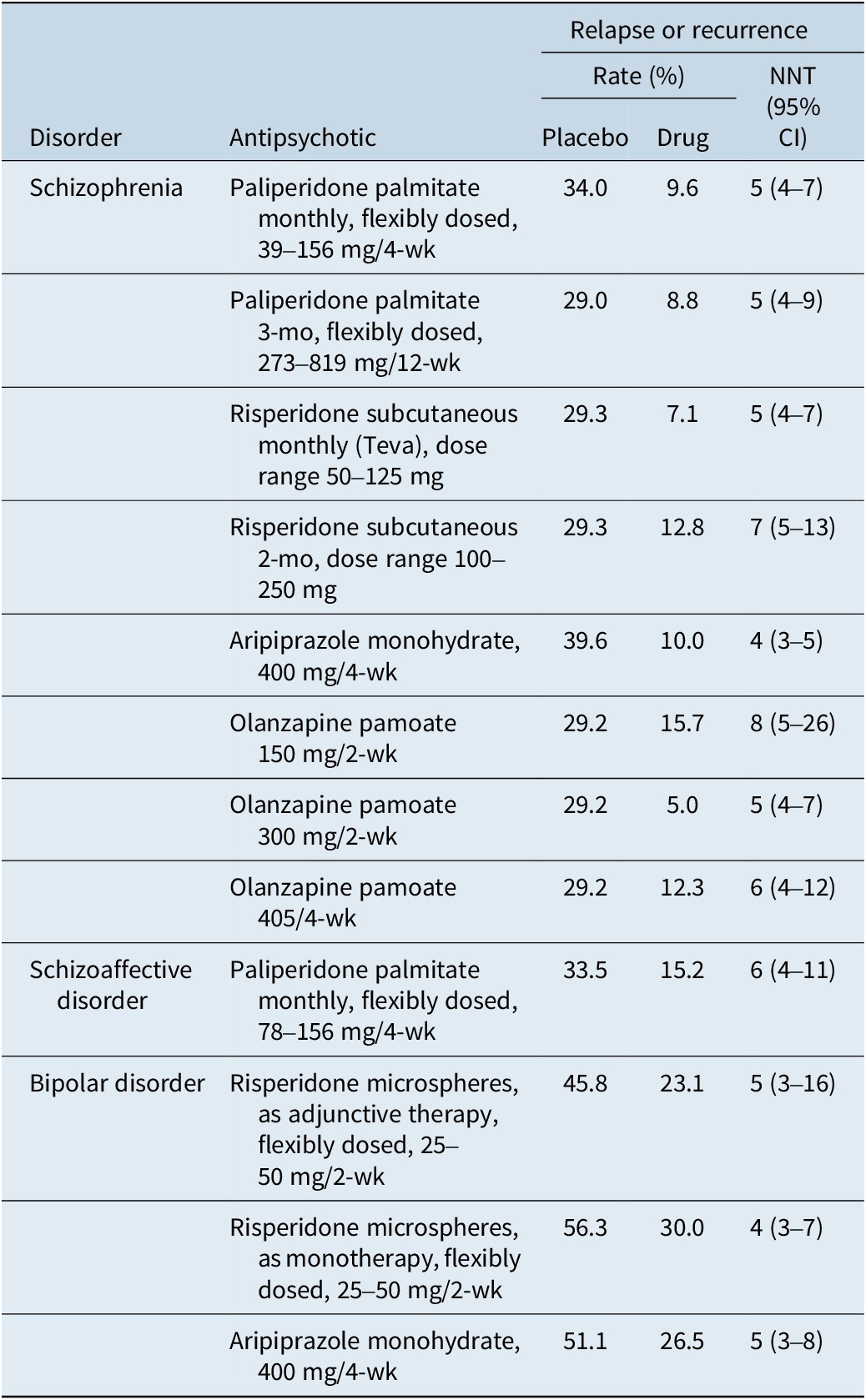
Although not without controversy,Reference Emsley, Fleischhacker and Galderisi 91 placebo-controlled randomized withdrawal study designs are often used to establish efficacy for the maintenance indication. The typical study design would be one where patients with the disease of interest are stabilized on the test medication and then subsequently randomized to either continue the test medication or receive a placebo. The primary outcome measure is usually time to relapse, impending relapse, or recurrence, depending on the disorder and the study. This has been formally assessed versus placebo in registration studies in individuals with schizophrenia for paliperidone palmitate administered monthlyReference Hough, Gopal and Vijapurkar 92 or every 3 months,Reference Berwaerts, Liu and Gopal 93 olanzapine pamoate,Reference Kane, Detke and Naber 94 aripiprazole monohydrate,Reference Kane, Sanchez and Perry 95 and 1-month/2-month risperidone subcutaneous.Reference Kane, Harary and Eshet 96 Registration studies using similar designs were also done in individuals with bipolar disorder for risperidone microspheres (monotherapy or adjunctive use)Reference Macfadden, Alphs and Haskins 97 , Reference Quiroz, Yatham and Palumbo 98 and aripiprazole monohydrate (monotherapy),Reference Calabrese, Sanchez and Jin 99 and in individuals with schizoaffective disorder for once-monthly paliperidone palmitate (monotherapy or adjunctive use).Reference Fu, Turkoz and Simonson 100 Number needed to treat versus placebo for prevention of relapse or recurrence for any of the tested medications for any of the indications range from 4 to 8, with overlap of the 95% confidence intervals (Table 8).Reference Citrome 29 , Reference Citrome 39 , Reference Greenberg and Citrome 40 , Reference Citrome 49 , Reference Citrome 65 , Reference Citrome 66 , Reference Kane, Harary and Eshet 96 , 101 , Reference Citrome, Tohami and Sharon 102 These effect sizes are consistent with the broader literature on maintenance treatment.Reference Ceraso, Lin and Schneider-Thoma 103 In general, there appears to be no clear differences between antipsychotics for relapse prevention, and thus, choice of antipsychotic for maintenance treatment can be guided mainly by tolerability.Reference Schneider-Thoma, Chalkou and Dörries 104
Prevention of Relapse or Recurrence as Quantified Using Number Needed to Treat versus Placebo (or versus 45 mg/4 wk for Olanzapine Pamoate), Data from US Registration Studies of Long-Acting Injectable Antipsychotics (Adapted from Ref.Reference Citrome 7)

CI, confidence interval; NNT, number needed to treat.
Selecting a long-acting injectable antipsychotic
The simplest scenario is if the patient is already receiving an antipsychotic that is available as an LAI formulation. Then it is a matter of educating the patient (and caregiver) about the availability of this different way of receiving medication. If there are competing LAI formulations for the same or related molecule, then a review of the “amenities of care” (Table 3) is in order. There may be a preference for a specific injection interval that is available with only some of the products. Patients and caregivers sometimes need to be reassured that a longer interval between injections does not necessarily mean that visits will be scheduled less often. If naïve to the molecule, it will be necessary to administer test doses of the oral formulation in order to rule out potential hypersensitivity as well as assess acute tolerability in general.
There may be some personal preferences regarding subcutaneous versus intramuscular injection on the part of both the patient and the provider.Reference Robinson, Suett and Wilhelm 105
Principally because of injection volume, not all formulations or doses can be given in the arm and must be administered in the gluteal muscle. Patients who are new to LAI antipsychotics may not be aware that injection is into the upper outer quadrant of the gluteal muscle, and that this location is relatively easy to access without embarrassment. The gauge and length of the needle may be of interest to the patient, who may be reassured when shown the different options available to select from.
In some situations, it may be unrealistic to expect adherence to oral supplementation, and alternatives should be considered. Some choices require an infrastructure (refrigeration for storage of almost all the formulations of risperidone, examination table for administering subcutaneous injections in the abdomen, ability to observe the patient for 3 hours after each injection of olanzapine pamoate). For patients receiving oral fluphenazine or haloperidol and a switch to a LAI antipsychotic is being considered, despite the relatively low cost of haloperidol decanoate or fluphenazine decanoate, the clinician needs to weigh the potential disadvantages of using concomitant oral anticholinergics as discussed earlier. On occasion, the supply chain for older generic medications sometimes gets interruptedReference Demers, Bilodeau and Laberge 106; the American Society of Health-System Pharmacists maintains a web resource that tracks drug shortages. 107
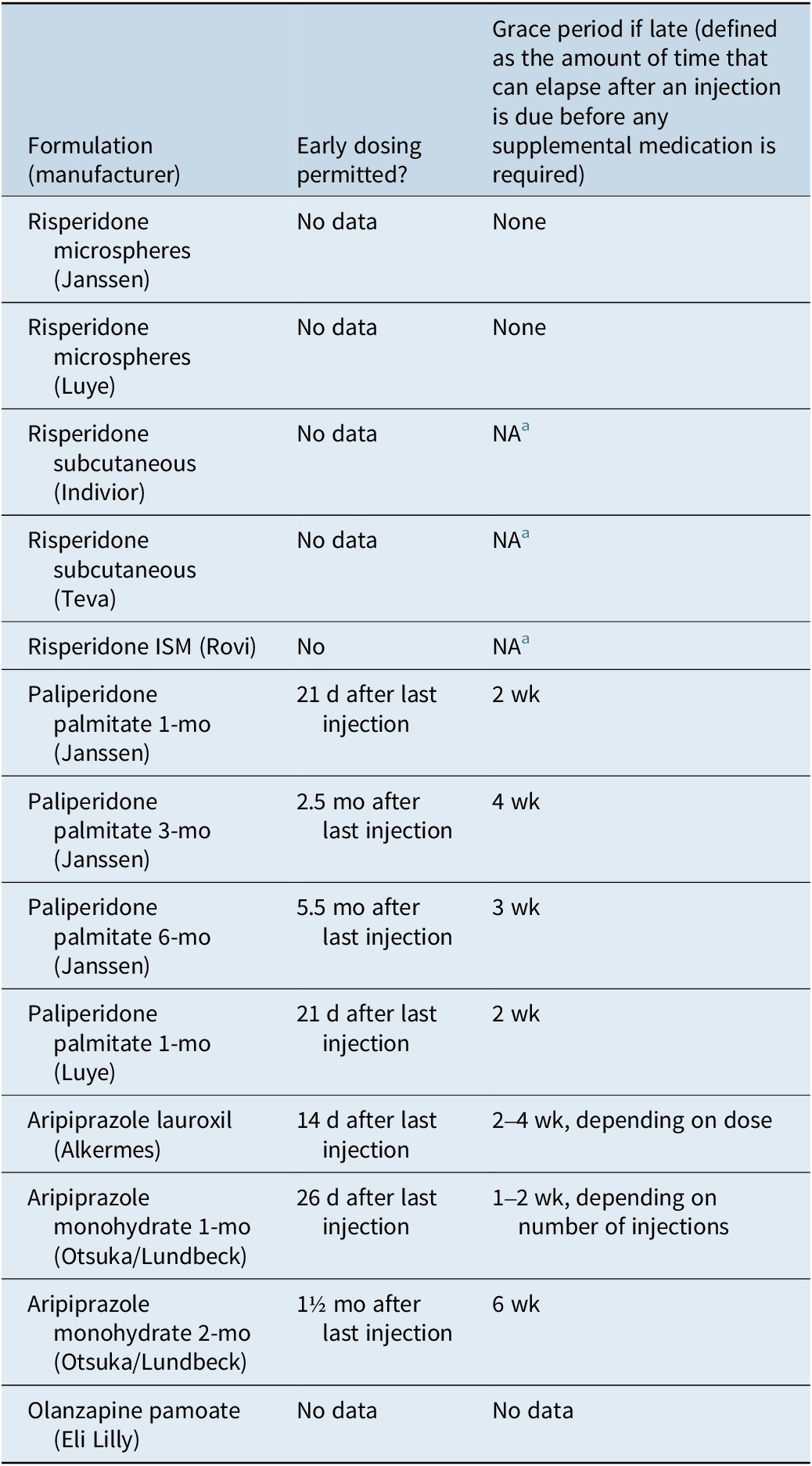
Additional pragmatic concerns for the provider are the presence of drug–drug interactions that cannot be easily remedied because of dosing constraints, as well as the lack of dosing flexibility when considering longer injection intervals. The requirements for reconstitution may be time-consuming and burdensome for some of the agents. Complex re-initiation requirements are present for some formulations; however, in general, the longer the injection interval, the greater the flexibility in the timing of an early or late injection (Table 9).
Formulations of Long-Acting Injectable Antipsychotics and Recommendations Regarding Early or Late Dosing as per US Product Labels

a NA, no oral supplementation or loading dose is required upon initiation with this formulation.
If the patient is receiving acute treatment, available options in the United States that do not require any oral supplementation are paliperidone palmitate, risperidone subcutaneous, and olanzapine pamoate; LAI antipsychotic formulations that can be initiated with the administration of only a single injection are paliperidone palmitate (Luye), risperidone subcutaneous, and olanzapine pamoate. Requiring only 1 dose of oral medication are the 1-day initiation options for aripiprazole monohydrate and aripiprazole lauroxil. Prior knowledge of tolerability and efficacy is important because once injected, the medication cannot be withdrawn. Oral or intramuscular short-acting antipsychotic medications are, in most situations, the most prudent way to initiate antipsychotic treatment in an individual who is treatment-naïve or if a medication history cannot be reliably obtained. A shortened initiation period is especially useful for hospitalized patients being transitioned to outpatient care, and in forensic settings such as jails.
Long-term considerations in maintenance treatment include weight gain and metabolic adverse effects, especially with olanzapine; first-generation LAI antipsychotics could possibly be considered under these circumstances and where a switch among the second-generation LAIs was not helpful. If prolactin-related adverse effects are a clinical concern, one of the aripiprazole LAI formulations would be the first choice; to be avoided under these circumstances would be paliperidone palmitate, risperidone microspheres, or the first-generation LAI antipsychotics.
Cost considerations are sometimes obstacles, and access to patient-assistance programs can be helpful. An important factor is whether reimbursement or enrollment in a patient-assistance program is possible with off-label use of a formulation, even when it would be logical to use it. For example, only risperidone microspheres and aripiprazole monohydrate are approved for maintenance treatment of bipolar I disorder, with risperidone microspheres the only LAI approved in combination with lithium or valproate for this indication, and only paliperidone once monthly is approved for the treatment of schizoaffective disorder. Reimbursement may prevent the use of risperidone subcutaneous, 3- or 6-month paliperidone palmitate, or aripiprazole lauroxil under these circumstances.
Conclusion
Only 3 second-generation antipsychotics are available as LAI preparations; however, there are multiple options in terms of formulation. Choosing among the different LAI antipsychotics is partly based on pragmatic concerns. For example, olanzapine pamoate would not be a practical option if the mandatory 3-hour post-injection observation period cannot be provided. For patients receiving oral risperidone, using risperidone microspheres can be inconvenient as that formulation is administered every 2 weeks, requires refrigeration and reconstitution, and must be accompanied by oral supplementation after the initial injection. Instead of risperidone microspheres, either 1-month/2-month risperidone subcutaneous or paliperidone palmitate can be considered as these choices do not require oral supplementation upon initiation, entail less frequent injections (either monthly or every other month for 1-month/2-month risperidone subcutaneous, or every 1, 3, or 6 months for paliperidone palmitate), are supplied in prefilled syringes, have a relatively small needle gauge, and is normally stored at room temperature (paliperidone palmitate) or can remain at room temperature for up to 90 days (1-month/2-month risperidone subcutaneous). Regarding aripiprazole LAI, there are 2 competing formulations available in the United States—aripiprazole monohydrate and aripiprazole lauroxil; although both can be initiated in 1 day and administered at the outset every month or every other month, there are differences in terms of dosage strengths and approved indications.
Author contribution
Conceptualization and execution: L.C.
Disclosures
Leslie Citrome has served as a consultant to AbbVie/Allergan, Acadia, Adamas, Adheretech, Alkermes, Alumis, Angelini, Astellas, Autobahn, Avanir, Axsome, Biogen, BioXcel, Bristol-Myers Squibb, Boehringer Ingelheim, Cadent Therapeutics, Cerevel, Clinilabs, COMPASS, Delpor, Draig Therapeutics, Eisai, Enteris BioPharma, HLS Therapeutics, Idorsia, INmune Bio, Impel, Intra-Cellular Therapies, Janssen, Karuna, Lundbeck, Luye, Lyndra, MapLight, Marvin, Medavante-ProPhase, Merck, Mitsubishi-Tanabe Pharma, Neumora, Neurocrine, Neurelis, Noema, Novartis, Noven, Otsuka, Ovid, Praxis, Recordati, Relmada, Reviva, Sage, Sumitomo/Sunovion, Supernus, Teva, University of Arizona, Vanda, Wells Fargo, and one-off ad hoc consulting for individuals/entities conducting marketing, commercial, or scientific scoping research; speaker for AbbVie/Allergan, Acadia, Alkermes, Angelini, Axsome, BioXcel, Bristol-Myers Squibb, Eisai, Idorsia, Intra-Cellular Therapies, Janssen, Lundbeck, Neurocrine, Neopharm, Noven, Otsuka, Recordati, Sage, Sunovion, Takeda, Teva, Vanda, and CME activities organized by medical education companies such as Medscape, NACCME, NEI, Vindico, and Universities and Professional Organizations/Societies; owns stocks (small number of shares of common stock) in Bristol-Myers Squibb, Eli Lilly, J & J, Merck, Pfizer purchased > 10 years ago, and stock options in Reviva; and has received royalties/publishing income from Taylor & Francis (Editor-in-Chief, Current Medical Research and Opinion, 2022-date), Wiley (Editor-in-Chief, International Journal of Clinical Practice, through end 2019), UpToDate (reviewer), Springer Healthcare (book), Elsevier (Topic Editor, Psychiatry, Clinical Therapeutics, through Spring 2025).
CNS SPECTRUMS
CME Review Article
Choosing Amongst the Long-Acting Injectable Antipsychotics: An Evidence-Based Pragmatic Guide
This CME activity is provided by HMP Education and Neuroscience Education Institute (NEI).
CME/CE Information
Target Audience: This activity has been developed for the healthcare team or individual prescriber specializing in mental health. All other healthcare team members interested in psychopharmacology are welcome for advanced study.
Learning Objectives: After completing this educational activity, you should be better able to:
-
• Differentiate among the different antipsychotics and the formulations of the same antipsychotic in terms of clinical applications, including treatment of schizophrenia and maintenance therapy in bipolar I disorder
 Accreditation: In support of improving patient care, this activity has been planned and implemented by HMP Education and Neuroscience Education Institute (NEI). HMP Education is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Accreditation: In support of improving patient care, this activity has been planned and implemented by HMP Education and Neuroscience Education Institute (NEI). HMP Education is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Activity Overview: This activity is best supported via a computer or device with current versions of the following browsers: Mozilla Firefox, Google Chrome, or Safari. A PDF reader is required for print publications. A post-test score of 70% or higher is required to receive CME/CE credit.
Estimated Time to Complete: 1 hour.
Continuing Education credit will be available for three (3) years from the publication date of the associated article. Please visit https://nei.global/cnsspectrums2025 for additional information and to access the CE activity.
*NEI maintains a record of participation for six (6) years.
Instructions for Optional Posttest and CME Credit
-
1. Read the article
-
2. Successfully complete the posttest at https://nei.global/CNS/LAI-04
-
3. Print your certificate
Questions? Email customerservice@neiglobal.com.
Credit Designations: The following are being offered for this activity:
-
• Physician: ACCME AMA PRA Category 1 Credits™.
-
○ HMP Education designates this enduring material for a maximum of 1.00 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
-
-
• Nurse: ANCC contact hours.
-
○ This continuing nursing education activity awards 1.00 contact hour. Provider approved by the California Board of Registered Nursing, Provider #18006 for 1.00 contact hour.
-
-
• Nurse Practitioner: ACCME AMA PRA Category 1 Credit™.
-
○ American Academy of Nurse Practitioners National Certification Program accepts AMA PRA Category 1 Credits™ from organizations accredited by the ACCME.
-
○ The content in this activity pertaining to pharmacology is worth 1.00 continuing education hour of pharmacotherapeutics.
-
-
• Pharmacy: ACPE application-based contact hours.
-
○ This internet enduring, knowledge-based activity has been approved for a maximum of 1.00 contact hour (.10 CEU).
-
○ The official record of credit will be in the CPE Monitor system. Following ACPE Policy, NEI and HMP Education must transmit your claim to CPE Monitor within 60 days from the date you complete this CPE activity and are unable to report your claimed credit after this 60-day period. Ensure your profile includes your DOB and NABP ID.
-
-
• Physician Associate/Assistant: AAPA Category 1 CME credits.
-
○
-
 HMP Education has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credits for activities planned in accordance with the AAPA CME Criteria. This internet enduring activity is designated for 1.00 AAPA Category 1 credit. Approval is valid until February 11, 2028. PAs should only claim credit commensurate with the extent of their participation.
HMP Education has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credits for activities planned in accordance with the AAPA CME Criteria. This internet enduring activity is designated for 1.00 AAPA Category 1 credit. Approval is valid until February 11, 2028. PAs should only claim credit commensurate with the extent of their participation.
-
-
• Psychology: APA CE credits.
-
○
-
 Continuing Education (CE) credits for psychologists are provided through the co-sponsorship of the American Psychological Association (APA) Office of Continuing Education in Psychology (CEP). The APA CEP Office maintains responsibility for the content of the programs. This activity awards 1.00 CE Credit.
Continuing Education (CE) credits for psychologists are provided through the co-sponsorship of the American Psychological Association (APA) Office of Continuing Education in Psychology (CEP). The APA CEP Office maintains responsibility for the content of the programs. This activity awards 1.00 CE Credit.
-
-
• Social Work: ASWB-ACE CE credits.
-
○
-
As a Jointly Accredited Organization, HMP Education is approved to offer social work continuing education by the Association of Social Work Boards (ASWB) Approved Continuing Education (ACE) program. Organizations, not individual courses, are approved under this program. Regulatory boards are the final authority on courses accepted for continuing education credit. Social workers completing this internet enduring course receive general continuing education credit.
-
-
• Non-Physician Member of the Healthcare Team: Certificate of Participation.
-
○
-
HMP Education awards hours of participation (consistent with the designated number of AMA PRA Category 1 Credit™) to a participant who successfully completes this educational activity.
-
Peer Review: The content was peer-reviewed by an MD, MPH specializing in forensics, psychosis, schizophrenia, mood disorders, anxiety, and cognitive disorders — to ensure the scientific accuracy and medical relevance of information presented and its independence from commercial bias. NEI and HMP Education take responsibility for the content, quality, and scientific integrity of this CME/CE activity.
Disclosures: All individuals in a position to influence or control content are required to disclose any relevant financial relationships. Any relevant financial relationships were mitigated prior to the activity being planned, developed, or presented.
Faculty Author / Presenter
Leslie Citrome, MD, MPH
Clinical Professor of Psychiatry and Behavioral Sciences, New York Medical College, Valhalla, NY Consultant/Advisor: AbbVie/Allergan, Acadia, Adamas, Alkermes, Angelini, Astellas, Avanir, Axsome, Biogen, BioXcel, Bristol-Myers Squibb, Boehringer Ingelheim, Cadent Therapeutics, Cerevel, Clinilabs, COMPASS, Delpor, Eisai, Enteris BioPharma, HLS Therapeutics, Idorsia, INmune Bio, Impel, Intra-Cellular Therapies, Janssen, Karuna, Lundbeck, Luye, Lyndra, MapLight, Marvin, Medavante-ProPhase, Merck, Mitsubishi-Tanabe Pharma, Neumora, Neurocrine, Neurelis,
Noema, Novartis, Noven, Otsuka, Ovid, Praxis, Recordati, Relmada, Reviva, Sage, Sumitomo/Sunovion, Supernus, Teva, University of Arizona, Vanda, Wells Fargo, and ad hoc consulting for marketing, commercial, or scientific scoping research
Speakers Bureau: AbbVie/Allergan, Acadia, Alkermes, Angelini, Axsome, BioXcel, Bristol-Myers Squibb, Eisai, Idorsia, Intra-Cellular Therapies, Janssen, Lundbeck, Neurocrine,f Noven, Otsuka, Recordati, Sage, Sunovion, Takeda, Teva, Vanda, and CME activities organized by Medscape, NACCME, NEI, Vindico, and Universities/Professional Organizations
Stockholder: AbbVie/Allergan, Acadia, Alkermes, Angelini, Axsome, BioXcel, Bristol-Myers Squibb, Eisai, Idorsia, Intra-Cellular Therapies, Janssen, Lundbeck, Neurocrine,f Noven, Otsuka, Recordati, Sage, Sunovion, Takeda, Teva, Vanda, and CME activities organized by Medscape, NACCME, NEI, Vindico, and Universities/Professional Organizations
Stock Options: Reviva
Royalties/Publishing: Taylor & Francis (Editor-in-Chief, Current Medical Research and Opinion), Wiley (Editor-in-Chief, International Journal of Clinical Practice, through 2019), UpToDate (Reviewer), Springer Healthcare (Book), Elsevier (Topic Editor, Psychiatry, Clinical Therapeutics)
The remaining Planning Committee members, Content Editors, Peer Reviewer, and NEI planners/staff have no financial relationships to disclose. NEI and HMP Education planners and staff include Meghan Grady, Caroline O’Brien, MS, Ali Holladay, Moriah Carswell, Andrea Zimmerman, EdD, CHCP, Brielle Calleo, and Steven S. Simring, MD, MPH.
Disclosure of Off-Label Use: This educational activity may include discussion of unlabeled and/or investigational uses of agents that are not currently labeled for such use by the FDA. Please consult the product prescribing information for full disclosure of labeled uses.
Cultural Linguistic Competency and Implicit Bias: A variety of resources addressing cultural and linguistic competencies and strategies for understanding and reducing implicit bias can be found in this handout—download me.
Accessibility Statement
For questions regarding this educational activity, or to cancel your account, please email customerservice@neiglobal.com.
Support: This activity is supported by an unrestricted educational grant from Alkermes, Inc., Teva Pharmaceuticals, Johnson & Johnson Innovative Medicine and Otsuka America Pharmaceutical, Inc.

 HMP Education has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credits for activities planned in accordance with the AAPA CME Criteria. This internet enduring activity is designated for 1.00 AAPA Category 1 credit. Approval is valid until February 11, 2028. PAs should only claim credit commensurate with the extent of their participation.
HMP Education has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credits for activities planned in accordance with the AAPA CME Criteria. This internet enduring activity is designated for 1.00 AAPA Category 1 credit. Approval is valid until February 11, 2028. PAs should only claim credit commensurate with the extent of their participation. Continuing Education (CE) credits for psychologists are provided through the co-sponsorship of the American Psychological Association (APA) Office of Continuing Education in Psychology (CEP). The APA CEP Office maintains responsibility for the content of the programs. This activity awards 1.00 CE Credit.
Continuing Education (CE) credits for psychologists are provided through the co-sponsorship of the American Psychological Association (APA) Office of Continuing Education in Psychology (CEP). The APA CEP Office maintains responsibility for the content of the programs. This activity awards 1.00 CE Credit.