Introduction
Decision-making is a complex cognitive function highly dependent on context (Gigerenzer, Reference Gigerenzer2021; Simon, Reference Simon1956). In simple, information-rich environments, logical processes can be employed to calculate the optimal choice (Starcke & Brand, Reference Starcke and Brand2016). Conversely, in more ambiguous contexts, individuals often rely on intuitive judgments grounded in past experiences (episodic memory), as well as on adaptive adjustment through feedback and reinforcement learning processes (Starcke & Brand, Reference Starcke and Brand2016; Volz & Gigerenzer, Reference Volz and Gigerenzer2012). Besides uncertainty levels, other factors, such as the individual versus social nature of the context, will determine which processes are more suitable when making decisions.
Social decision-making is a multifaceted process, often requiring the decision-maker to infer others’ mental states, integrate their personality and intentions, as well as to consider broader societal implications beyond individual gain (Báez-Mendoza et al., Reference Báez-Mendoza, Vázquez, Mastrobattista and Williams2021; Lee & Harris, Reference Lee and Harris2013). Therefore, social decisions are rooted in a rich tapestry of psychological, neurobiological, and sociological factors, indicating a deep interconnection between individual choices and societal outcomes (Bruch & Feinberg, Reference Bruch and Feinberg2017). This complexity is due to the intrinsic nature of social environments, where decisions impact not only the individual but also the larger group, influencing and being influenced by collective societal norms and behaviors (Tremblay et al., Reference Tremblay, Sharika and Platt2017). So, in social contexts, generosity, empathy, and justice may outweigh economic maximization strategies, fostering social relationships, as evidenced by research in social decision-making (Camerer, Reference Camerer2003; Frith & Singer, Reference Frith and Singer2008; Hinterbuchinger et al., Reference Hinterbuchinger, Kaltenboeck, Baumgartner, Mossaheb and Friedrich2018).
One of the most widespread paradigms for evaluating social decisions is the Ultimatum Game (UG). In this task, a “proposer” receives a sum of money to split with a “responder,” with possible splits ranging from equitable offers to extremely unbalanced offers. The responder must decide whether to accept or reject the proposal; if it accepts, the money is split as proposed, but if it rejects, both proposer and responder receive nothing (Hintze & Hertwig, Reference Hintze and Hertwig2016). This scenario, although simple, captures the complex interplay between economic self-interest and social justice (Gaertig et al., Reference Gaertig, Moser, Alguacil and Ruz2012). According to the classical economic model (Camerer, Reference Camerer2003), proposers should aim for the smallest offers, and responders should accept anything above zero for maximum benefit. However, social decisions combine rationality and emotions (Hinterbuchinger et al., Reference Hinterbuchinger, Kaltenboeck, Baumgartner, Mossaheb and Friedrich2018). Numerous studies indicate that typical proposers often display generosity by consistently offering 40%–45% in the UG (Hartley & Fisher, Reference Hartley and Fisher2018). Similarly, responders tend to reject offers below a third due to aversion to inequity, opting to punish selfish behavior to encourage fairer future offers (Hinterbuchinger et al., Reference Hinterbuchinger, Kaltenboeck, Baumgartner, Mossaheb and Friedrich2018). This decision-making approach is seen as adaptive, reducing self-interest impact while fostering cooperation and social cohesion (Hoffman et al., Reference Hoffman, McCabe and Smith2008).
However, the choice of a specific decision-making process extends beyond the nature of the decision environment and is also shaped by a spectrum of internal and external influences (Gigerenzer, Reference Gigerenzer2021; Simon, Reference Simon1955, Reference Simon1956). Given the marked increase in psychosocial stress in modern societies and the inherent complexity of social contexts, which amplify decision-making under stress (Steinbeis et al., Reference Steinbeis, Engert, Linz and Singer2015), understanding the influence of acute stress on social decisions becomes highly necessary.
Stress is thought to affect decision-making through two complementary neurobiological mechanisms. First, it reduces prefrontal-dependent control and impairs feedback-based learning, leading to less flexible adjustments in response to changing conditions (Fellows & Farah, Reference Fellows and Farah2007; Gu et al., Reference Gu, Wang, Hula, Wang, Xu, Lohrenz, Knight, Gao, Dayan and Montague2015; Pabst et al., Reference Pabst, Brand and Wolf2013; Starcke & Brand, Reference Starcke and Brand2012). Second, stress enhances dopaminergic activity in the striatum, which, according to the STARS model, biases decision-making toward reward salience and immediate reinforcement (Mather & Lighthall, Reference Mather and Lighthall2012; Scott et al., Reference Scott, Heitzeg, Koeppe, Stohler and Zubieta2006; Wood et al., Reference Wood, Schweinhardt, Jaeger, Dagher, Hakyemez, Rabiner, Bushnell and Chizh2007). Meta-analytic evidence supports that under stress, individuals often show riskier and more reward-oriented choices (Starcke et al., Reference Starcke and Brand2016). While these findings mainly come from individual decision-making paradigms, they provide a useful framework for understanding how stress may also shape behavior in social contexts, where both learning from interaction and sensitivity to reward and fairness cues are essential.
Based on the described effects of stress on individual decision-making, it is expected that more economically rational decisions in social contexts, driven by self-benefit and with less influence from social context phenomena like generosity, equity, or aversion to injustice. However, the results are not straightforward. In a recent review by Faber and Häusser (Reference Faber and Häusser2022), while seven studies showed the anticipated decrease in prosocial behavior, another four found the opposite effect, and three found no differences between the stressed group and the control in their social decisions. This diversity could be influenced by various factors. First, the paradigms used vary, assessing different social decision contexts (such as proposer vs. responder), where stress might impact differently (Faber & Häusser, Reference Faber and Häusser2022). Additionally, considering potential moderating variables that could influence the relationship between decision-making and stress is crucial. For instance, in five other studies reflected in the same review, results depended on factors like gender or social distance (Faber & Häusser, Reference Faber and Häusser2022). Moreover, it is essential to acknowledge that a stressor may not impact all participants equally (Molins & Serrano, Reference Molins and Serrano2020), emphasizing the importance of addressing the degree of stress experienced, not just whether they experienced stress or not. Lastly, these studies often oversimplify decision-making as a single dimension, disregarding its multifaceted cognitive subprocesses (Alacreu-Crespo et al., Reference Alacreu-Crespo, Guillaume, Sénèque, Olié and Courtet2020). Computational advancements now allow delving into these processes to obtain more stress-sensitive results, leading to more specific conclusions (Ahn et al., Reference Ahn, Busemeyer, Wagenmakers and Stout2008, Reference Ahn, Haines and Zhang2017). Indeed, a computational model has been developed for the UG paradigm, specifically for the “responder” role (Gu et al., Reference Gu, Wang, Hula, Wang, Xu, Lohrenz, Knight, Gao, Dayan and Montague2015). This model assesses not only the acceptance or rejection of offers but also the degree of aversion to inequity displayed (α), the ability to learn during the task and adapt expectations about equity based on previously received offers (ε), and the flexibility or perseveration in the decision patterns (γ). However, as far as we know, there are no studies addressing how acute stress specifically affects these cognitive subprocesses during the UG paradigm.
Therefore, our study’s central aim is to test through computational modeling the stress effect on social decision making and cognitive subprocesses involved during the UG. Thus, based on the above, our specific hypotheses are as follows: given that stress would encourage the search for immediate rewards and more utilitarian decisions, (H1) a group exposed to the virtual version of the Trier Social Stress Test will accept a greater number of offers, regardless of whether some are unfair, and (H2) will display less aversion to inequity (α) than the control group. On the other hand, because of the negative effects that stress can exert, (H3) acute stress will hinder learning or adaptation during the task (ε), and (H4) will decrease flexibility in the decision patterns (γ). Finally, (H5) these differences between the control and stress groups will be moderated by changes in HRV, mood, and perceived stress levels, all of which are indicators of the greater or lesser impact of the stressor.
Method
Participants
Previous acute stress and UG studies involved 39 (Prasad et al., Reference Prasad, Narayanan, Lim, Koh, Koh and Mehta2017) to 118 participants (Youssef et al., Reference Youssef, Bachew, Bissessar, Crockett and Faber2018) without reported effect sizes. Seventy-six participants were initially recruited, but three were excluded due to registration issues, leaving a total of 73 participants in the study (age: M = 21.2, SD = 3.01; women: N = 79.5%). Although the sample was primarily one of convenience, a post hoc power analysis using G*Power indicated that our total of 76 participants (38 per group) was sufficient to achieve 80% power (α = .05) to detect medium effect sizes in our MANOVA design with three dependent variables. Inclusion criteria comprised no cardiovascular, endocrine, neurological, or psychiatric diseases; limited cigarette consumption (<5 per day); no habitual drug use; restricted exercise (<10 hours weekly); and no highly stressful events in the last month. Participants were also instructed to abstain from strenuous exercise, drugs, alcohol (24 h before the experiment), smoking, or stimulants (in the 2 h before the experiment). Participants were randomly assigned to two groups: stress (N = 35) and control (N = 38).
Procedure
The study obtained approval from the Ethics Research Committee of the University of Valencia, following the ethical standards of the 1969 Declaration of Helsinki. Experimental sessions were carried out from 3:00 pm to 8:00 pm, lasting approximately 1 h. Participants were connected to an electrocardiogram (ECG) and rested for 10 min to acclimate, with the last 5 minutes serving as the baseline period. The stress group was submitted to the virtual Trier Social Stress Test (TSST-VR), while the control group engaged in a distractor task; specifically, the control group watched a neutral documentary video on marble manufacturing, matched in duration to the experimental task. This was designed as an active control condition, ensuring participant engagement without inducing stress, thereby paralleling the temporal structure of the stress task. Participants were assessed for positive and negative mood using the Positive and Negative Affect Schedule (Watson et al., Reference Watson, Clark and Tellegen1988) before and after the stressor/distractor and reported subjective stress levels. Subsequently, both groups participated in the UG.
Instruments
Virtual reality version of the Trier Social Stress Test (TSST-VR)
Following Montero-López et al. (Reference Montero-López, Santos-Ruiz, García-Ríos, Rodríguez-Blázquez, Pérez-García and Peralta-Ramírez2016), we employed a software enabling the creation of a 3D audience projected onto a 27″ screen placed 1 meter from the participant. This virtual-reality adaptation of the traditional TSST (Kirschbaum et al., Reference Kirschbaum, Pirke and Hellhammer1993) comprises four phases. Initially, participants faced a screen displaying a stage curtain and were instructed to deliver a speech convincing an audience about their suitability for their dream job. A microphone and camera were added to lend credibility, indicating to participants that both the content and formal aspects of their speech would be analyzed in real time and that the virtual audience would react accordingly. The second phase, a 5-min anticipatory stress period, required participants to prepare their speeches. They were given a sheet for notes, which was removed before the speech delivery. The third phase involved delivering the speech, with the virtual audience initially neutral but then showing signs of boredom after 2.5 min. Finally, the last phase introduced an arithmetic task, where participants had to repeatedly subtract a fixed amount from a given number as quickly as possible. To ensure the effectiveness of our stress induction, we evaluated HRV, negative and positive mood, and subjective stress perception.
Variables
Heart rate variability (HRV)
HRV refers to the variation in the time interval between heartbeats, which is influenced by both branches of the autonomic nervous system (sympathetic [SNS] and parasympathetic [PNS]) (Castaldo et al., Reference Castaldo, Melillo, Bracale, Caserta, Triassi and Pecchia2015; Grossmann et al., Reference Grossmann, Sahdra and Ciarrochi2016; Shaffer & Ginsberg, Reference Shaffer and Ginsberg2017). The ECG was conducted using Einthoven’s third derivation with three reusable electrodes, BIOPAC MP150, an ECG-100C transducer, and AcqKnowledge software, sampling at a frequency of 1000 Hz. Following (Task Force of The European Society of Cardiology and The North American Electrophysiology Society of Pacing and Electrophysiology, 1996), standardized 5-min recordings were taken. Subsequently, both the final five baseline minutes and the initial 5 min of the TSST-VR were exported to Kubios software to extract HRV markers. The initial 5 min of the TSST-VR were chosen because they coincide with the only phase of the stressor where participants do not have to speak. Thus, there was no risk of altering HRV due to changes in breathing while speaking (Shaffer & Ginsberg, Reference Shaffer and Ginsberg2017). Prior to analysis, ECG signals underwent visual inspection to identify ectopic beats and other artifacts. Three participants exhibited ectopic beats, corrected using the Heart Timing Signal method (Mateo & Laguna, Reference Mateo and Laguna2003), enabling their inclusion in subsequent analysis. In Kubios, each participant underwent individual analysis using the threshold-based artifact correction algorithm (Tarvainen et al., Reference Tarvainen, Niskanen, Lipponen, Ranta-aho and Karjalainen2014), which removes artifacts without distorting normal RR intervals. Following the recommendations of Tarvainen et al. (Reference Tarvainen, Niskanen, Lipponen, Ranta-aho and Karjalainen2014), the threshold value for artifact correction, varying from very low to very strong, was adjusted individually, considering HRV’s high individual variability. Finally, to control potential slow changes in mean HR during recording, the smooth prior’s filter (Tarvainen et al., Reference Tarvainen, Niskanen, Lipponen, Ranta-aho and Karjalainen2014) was employed as a detrending method.
Following recommendations for transparency and replicability in behavioral sciences (Asendorpf et al., Reference Asendorpf, Conner, De Fruyt, De Houwer, Denissen, Fiedler, Fiedler, Funder, Kliegl and Nosek2013; Simmons et al., Reference Simmons, Nelson and Simonsohn2011), we sought to avoid cherry-picking of HRV markers yielding desirable results by examining a range of HRV markers. Specifically, the HRV indicators previously used by Grossman et al. (Reference Grossmann, Sahdra and Ciarrochi2016), which are linked to PNS activity, were selected. These indicators are currently among the most recommended (Quintana et al., Reference Quintana, Alvares and Heathers2016; Shaffer & Ginsberg, Reference Shaffer and Ginsberg2017). Some of them capture time-domain variability using statistical parameters such as SDNN (standard deviation of the inter-beat interval), RMSSD (root mean squared successive differences in the inter-beat interval), and pnn50 (percentage of absolute differences between consecutive inter-beat intervals greater than 50 ms). Others assess total variability based on RR histogram geometry, including HRVTI (HRV triangular index obtained as the integral of the histogram divided by its height) and TINN (baseline width of the RR histogram evaluated through triangular interpolation). Additionally, certain metrics allocate frequency bands and count beat-to-beat intervals within each band, such as examining HFPOWFF (high-frequency band power spectrum density using fast Fourier transform).
Finally, following Grossman et al. (Reference Grossmann, Sahdra and Ciarrochi2016), to minimize the chance of α-inflation due to multiple testing, a principal component analysis (PCA) on HRV markers and primary statistical analyses on the retained factor scores were performed. PCA yielded a one-component solution for baseline HRV (variance explained: 83.72%; loadings: 0.84–0.98) and a one-component solution for TSST-VR HRV (variance explained: 84.03%; loadings: 0.85–0.97). A HRV reactivity index was obtained by subtracting baseline HRV to TSST-VR HRV.
Positive and Negative Affect registry (PANAS)
PANAS (Watson et al., Reference Watson, Clark and Tellegen1988) comprises a 20-item Likert-type scale (ranging from 1, “more than usual,” to 4, “much less than usual”) assessing positive and negative mood. Each dimension consists of 10 items, with scores indicating the intensity of positive or negative mood. PANAS was administered before and after the stressor/distractor. Moreover, the negative and positive affect reactivity was also computed by subtracting baseline PANAS from post-TSST-VR PANAS.
Perceived stress
Participants reported their perceived stress both before and after the stressor/distractor using an ad hoc question: “How much stress do you feel right now?” Responses were collected on a Likert scale from 0, “no stress” to 10, “a lot of stress.” A perceived stress reactivity index was calculated by subtracting baseline perceived stress from post-TSST-VR perceived stress.
Ultimatum Game (UG)
In the UG, a proposer decides how to split a sum of money with the responder, who then must accept for a shared payout or reject it, leaving both with nothing. In line with Gu et al. (Reference Gu, Wang, Hula, Wang, Xu, Lohrenz, Knight, Gao, Dayan and Montague2015), in our study, all participants assumed the responder role in the UG across 45 trials. Each trial began with a €20 split offer, followed by the choice to accept or reject the offer. The offers were predetermined: 6 × €1,6 × €2,6 × €3,6 × €4,6 × €5,3 × €6,3 × €7,3 × €8,3 × €9,3 × €10, presented in a randomized order. The total number of accepted offers was counted for each participant, allowing the average for each group to be extracted. In addition, the Rescorla–Wagner (Delta) computational model (Gu et al., Reference Gu, Wang, Hula, Wang, Xu, Lohrenz, Knight, Gao, Dayan and Montague2015) was applied to each participant to deepen in their underlying cognitive processes during the decisional task.
Rescorla–Wagner-based norm-adaptation model for the Ultimatum Game. This model assumes that participants playing the UG have an internal norm (f i) on what is the fair amount that should be distributed to them. In addition, this norm can be updated via the Rescorla–Wagner delta rule based on the context changes, that is, the norm evolves as a function of observed offers (Xiang et al., Reference Xiang, Lohrenz and Read Montague2013). Following Gu et al. (Reference Gu, Wang, Hula, Wang, Xu, Lohrenz, Knight, Gao, Dayan and Montague2015), the initial internal norm (f 0) was fitted individually to each participant’s data (f 0 ∈ [0,20]), and the Rescorla–Wagner rule (Rescorla & Wagner, Reference Rescorla, Wagner, Black and Prokasy1972) was applied for updating the internal norm. For a detailed math description of the model, see Gu et al. (Reference Gu, Wang, Hula, Wang, Xu, Lohrenz, Knight, Gao, Dayan and Montague2015). Specifically, the model allowed to extract three parameters. α or “aversion to inequity” that represents sensitivity to norm prediction error; that is, the individual’s aversion to unequal splits. The higher the α, the greater the unwillingness to accept an offer below the internal norm (f i). ε or the “norm adaptation rate” refers to how much the internal norm is modified according to the immediately preceding offer, that is, the learning or adaptation capacity based on previous experiences during the task. A lower ε would indicate that the internal norm is more persistent and represents a lower learning capacity. Finally, γ or the inverse temperature parameter refers to the variability or flexibility of the choices. The higher is the γ, the higher the perseveration during the choices. For clarity, ε corresponds to the learning rate and γ to the inverse temperature in standard reinforcement-learning terminology.
Each parameter of the model was estimated for each participant through Hierarchical Bayesian Analyses (HBA; see Ahn et al, Reference Ahn, Busemeyer, Wagenmakers and Stout2008 for more details), performed with the hBayesDM package (Ahn et al., Reference Ahn, Haines and Zhang2017) for the R software. The hBayesDM uses Stan 2.1.1 (Stan Development Team, 2017) with the Hamiltonian Monte Carlo (HMC) algorithm as MCMC for sampling the posterior distributions. Following Molins et al. (Reference Molins, Serrano and Alacreu-Crespo2021), we drawn 40.000 samples, after burn-in of 23.333 samples, in three different chains (in sum, a total of 120.000 samples and 70.000 burn-in). The Gelman–Rubin test (Gelman & Rubin, Reference Gelman and Rubin1992) was used to study whether the chains converged (Ȓ) to the target distribution. The maximum Ȓ across parameters was 1. Additionally, the MCMC chains were visually inspected, revealing no concerning trends or poor mixing. Therefore, we found no evidence of a lack of convergence.
Statistical analysis
Outliers were identified using the 2.5 standard deviations method and Mahalanobis distance for variables measured repeatedly (e.g., HRV), as a conservative quality check. No participant was excluded due to outliers, and results were qualitatively unchanged when including or excluding screened extreme observations. Assumption checks for normality relied primarily on visual diagnostics (Q–Q plots and residuals), complemented by the Kolmogorov–Smirnov test with Lilliefors correction; both indicated no material violations. Given our group sizes (>30 per group) and the robustness of parametric tests to mild non-normality, we retained the parametric approach. The analyses involved repeated-measures ANOVAs, with group (stress vs control) as a between-participants factor, to evaluate the effectiveness of stress induction, both physiologically (HRV) and subjectively (perceived stress, positive affect, negative affect), always controlling for sex. Additionally, accepted offers and participant-level parameters from the Rescorla–Wagner (Delta) model were compared between groups through MANOVA, using each participant’s posterior mean estimates from the hierarchical Bayesian fit as dependent variables, and controlling for sex, negative mood, perceived stress, and HRV reactivities. The significance level (α) was set at .05, and the partial eta square (η2 p) indicated the effect size. All analyses were conducted using IBM SPSS Statistics 25.
Results
Preliminary analyses
Stress and control groups were homogeneously distributed with no significant differences in age (stress: M = 21.85, SD = 3.19; control: M = 20.58, SD = 2.72), p = .078; in BMI (stress: M = 22.39, SD = 3.86; control: M = 21.85, SD = 4.06), p = .56; nor in socioeconomic status (stress: M = 6.33, SD = .69; control: M = 6.21, SD = 1.05), p = .94. Moreover, there were more women than men, but the chi-square test revealed that both women (stress: 74.3%; control: 84.2%) and men (stress: 25.7%; control: 15.8%), p = .29, maintained similar percentages in both groups. Sensitivity analyses comparing models with all observations versus excluding screened extreme values yielded the same pattern of statistical significance; for parsimony, we report the full-sample analyses.
Stress induction
Physiological stress
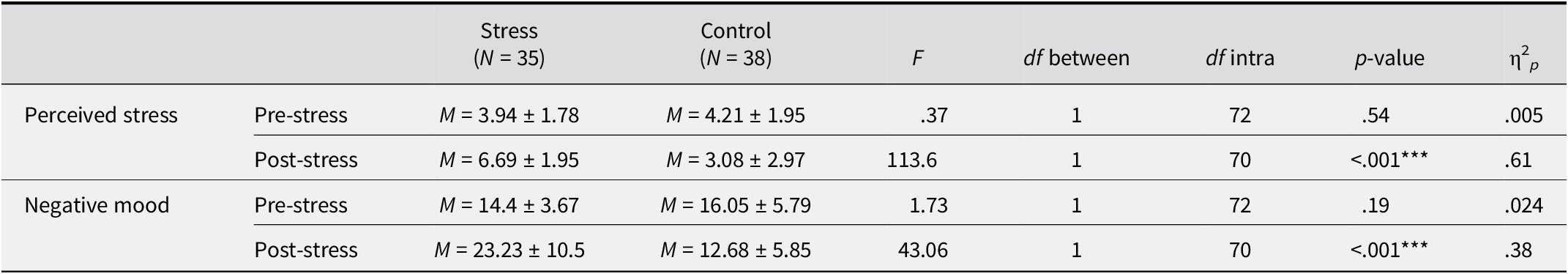
A repeated-measures ANOVA including group (stress vs. control) as a between-factor, and controlling for sex, was performed to test whether the stress induction was effective at the physiological level. Analyses revealed a significant group × moment interaction, F(1, 70) = 6.25, p = .01, η2 p = .08, which indicates that HRV evolution was different for both groups (see Figure 1). The intergroup analysis controlling for individual basal levels (see Table 1) revealed that, although both groups did not differ in their baseline HRV, the stress group showed significantly lower HRV than the control group during the TSST-VR/distractor.
Heart rate variability during baseline and TSST-VR/distractor by group. Stress and Control groups significantly differed in their HRV level during the TSST-VR/distractor. ** Significant contrast at the .01 level; M ± 95% confidence interval.

Intergroup differences in heart rate variability (HRV) during baseline and TSST-VR/distractor

Note: M = mean; ± SD; df = degrees of freedom; ** significant contrast at the .01 level.
Psychological stress
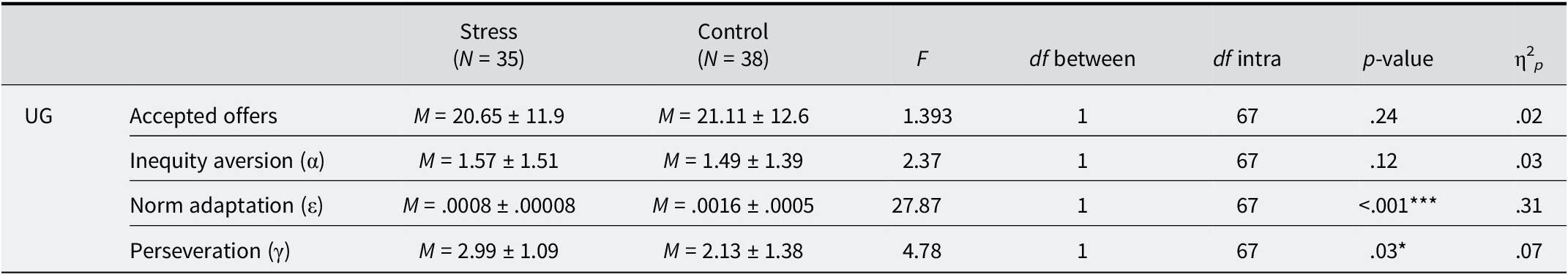
Regarding the psychological impact of the stress, repeated measures ANOVAs (including group as between-factor and controlling for sex) were carried out to study differences pre- and post-TSST-VR/distractor in the perceived stress and both the positive and the negative affect measured with PANAS. Focusing on the perceived stress, it was found a significant group × moment interaction, F(1, 70) = 113.96, p < .001, η2 p = .61. So, both groups did not differ at their basal level, but the perceived stress of the stress group was significantly higher than the level reported by the control group after the stressor/distractor (see Table 2). By the other side, regarding positive affect, no pre-post differences between groups were found (p’s > .05). However, regarding negative affect, analyses revealed a significant group × moment interaction, F(1, 70) = 49.48, p < .001, η2 p = .41. Stress and control groups did not present differences pre-stressor/distractor. Nevertheless, post-TSST-VR/distractor levels significantly differed between groups, with the stress group showing an increase in their negative mood (see Table 2).
Intergroup differences in perceived stress and negative mood pre- and post-stressor/distractor

Note: M, mean; ± SD; df, degrees of freedom; *** significant contrast at the .001 level.
Ultimatum Game
As can be seen in Table 3, both groups accepted on average 20.65 (stress group) and 21.11 (control group) offers, which constitute, respectively, 45.88% and 46.91% of the total of the 45 offers received. To test whether stress induced differences between groups not only in those accepting offers, but also in the underlying subprocesses during the UG (aversion to inequity [α], norm adaptation rate or learning [ε], and perseverance in decision-making patterns [γ]), we conducted a MANOVA, controlling for sex and including the effects of HRV, perceived stress, and negative mood reactivities. As seen in Table 3, differences between groups in the overall number of accepted offers were not statistically significant. And the same occurred regarding their level of aversion to inequity (α). However, the stressed group exhibited a lower capacity to adapt their internal norm regarding fairness (ε), indicating a poorer learning capacity. Furthermore, they also showed greater perseverance in their decision-making patterns (γ) compared to the control group. Neither sex nor any of the reactivities (HRV, perceived stress, and negative mood) accounted for a significant percentage of variance in any of the variables evaluated during the UG.
Intergroup differences in Ultimatum game measures

Note: M = mean; ± SD; df = degrees of freedom; *** significant contrast at the .001 level. * significant contrast at the .05 level.
Discussion
The aim of this study was to analyze whether acute stress can impair social decision-making in the UG by promoting economically rational but socially maladaptive decisions, while also hindering learning and flexibility during successive decisions. The results partially confirmed our hypotheses. On one hand, no differences were observed in the quantity of accepted offers or in the aversion displayed toward inequitable offers, indicating no less prosocial or more economically rational decisions. However, compared to the control group, the stress-exposed group exhibited a diminished ability to learn and adapt during the task, resulting in more rigid decision-making patterns. These findings will be further discussed in detail.
First, the effectiveness of the stress induction was demonstrated at both physiological and psychological levels, with the stress group showing significantly lower HRV, higher perceived stress, and increased negative affect after the TSST-VR compared to controls. These results confirm the effectiveness of stress manipulation and are consistent with previous literature linking acute stress to autonomic dysregulation and affective reactivity (Shaffer & Ginsberg, Reference Shaffer and Ginsberg2017). Importantly, since all HRV indicators included in this study were closely related to parasympathetic functioning (Grossmann et al., Reference Grossmann, Sahdra and Ciarrochi2016; Quintana et al., Reference Quintana, Alvares and Heathers2016; Shaffer & Ginsberg, Reference Shaffer and Ginsberg2017), the reduced HRV observed in the stress group aligns with the parasympathetic withdrawal associated with stress (Ciabattoni et al., Reference Ciabattoni, Ferracuti, Longhi, Pepa, Romeo and Verdini2017; Laborde et al., Reference Laborde, Mosley and Thayer2017; Michael et al., Reference Michael, Graham and Oam2017), further reinforcing the efficacy of our stress induction protocol.
However, despite robust stress responses, the overall acceptance rate of offers in the UG did not differ between groups. At first glance, this might suggest that stress has minimal impact on social decision-making; however, analysis of the latent computational parameters revealed a more nuanced picture. Specifically, the stress group exhibited a lower norm adaptation rate (ε) compared to controls, indicating that, across successive offers, they did not adequately adjust their internal standard of fairness, regardless of whether offers were equitable or inequitable (Gu et al., Reference Gu, Wang, Hula, Wang, Xu, Lohrenz, Knight, Gao, Dayan and Montague2015). This suggests a reduced capacity for learning from social experiences within the task. In addition, stressed participants demonstrated higher perseveration in their decision-making patterns (γ), reflecting less flexibility or variability in choices relative to the control group. While stable decisional patterns may be adaptive in environments with fixed rules (Delazer et al., Reference Delazer, Sinz, Zamarian and Benke2007; Gigerenzer, Reference Gigerenzer2021), in combination with reduced learning capacity, this pattern signals potential deficits in adaptive decision-making—critical for navigating uncertain social contexts (Feraco et al., Reference Feraco, Sella, Meneghetti and Cona2023; Laureiro-Martínez & Brusoni, Reference Laureiro-Martínez and Brusoni2018; Vuckovic et al., Reference Vuckovic, Kwantes and Neal2013).
These findings suggest that acute stress compromises cognitive flexibility and the integration of new social information, fostering more rigid, habitual strategies, while leaving the emotional salience of unfairness intact, as inequity aversion (α) remained unaffected. This dissociation aligns with evidence that stress impairs prefrontal-dependent control and shifts decision-making toward automatic or perseverative responding (Starcke & Brand, Reference Starcke and Brand2012), supporting the view that stress primarily disrupts higher-order adaptive processes such as learning and norm updating, while sparing affectively driven reactions (Hermans et al., Reference Hermans, Henckens, Joëls and Fernández2014). From a broader perspective, the observed reduction in learning and heightened perseveration may reflect a stress-induced shift away from “model-based” decision-making, in which individuals under neutral states typically employ calculated strategies, follow rules, and adapt to context (Otto et al., Reference Otto, Raio, Chiang, Phelps and Daw2013).
Our findings contribute to the broader debate on the impact of stress and anxiety on reinforcement learning and social decision-making. Consistent with previous research, Ting et al. (Reference Ting, Palminteri, Lebreton and Engelmann2022) reported that incidental anxiety produced inconsistent and elusive effects on reinforcement learning, highlighting the need to distinguish between observable behavioral outcomes and the underlying cognitive computations. Similarly, Laycock et al. (Reference Laycock, Schofield and McCall2024) demonstrated that threat selectively impaired complex decision-making in a virtual environment, without necessarily altering simpler behavioral measures. In line with these studies, our results indicate that while stress may not always manifest as changes in overt choice behavior (i.e., number of offers accepted), it does influence the cognitive mechanisms guiding decisions.
It is important to note that our study employed an incidental stress manipulation, with the decision-making task administered 15 min after the stressor. This design captures the influence of stress as a distractor, consistent with prior approaches, but does not encompass contexts in which stress and task demands occur simultaneously. Recent evidence emphasizes that both the timing and nature of stressors critically shape cognitive outcomes: Ting et al. (Reference Ting, Palminteri, Lebreton and Engelmann2022) showed that incidental anxiety does not always produce consistent reinforcement-learning effects, while Laycock et al. (Reference Laycock, Schofield and McCall2024) found that integral stress manipulations, where stress and task coincide, can affect complex decision-making differently than incidental stress. Therefore, our findings should be interpreted within the scope of incidental stress manipulations, and future work should systematically compare incidental versus integral stressors to clarify when stress primarily acts as a distractor and when it directly modulates task-related processing.
Taken together, the present study provides converging evidence that acute stress affects social decision-making at the computational rather than the behavioral level. The combination of reduced HRV, heightened perceived stress, and negative affect likely compromised prefrontal regulatory processes, leading to decreased norm adaptation and greater perseverance. This interpretation resonates with neurocognitive models positing that stress narrows the repertoire of adaptive responses, favoring rigidity over flexibility (Hermans et al., Reference Hermans, Henckens, Joëls and Fernández2014). Thus, the current findings highlight the importance of examining latent cognitive parameters rather than relying solely on behavioral outcomes, as the latter may obscure the more subtle yet functionally significant impact of stress on social decision-making.
Our study is not exempt from limitations. First, it is important to verify their generalizability by expanding the heterogeneity of the study sample. Our data were drawn from healthy university students with medium/high socioeconomic status, prompting the need to investigate if stress affects differently other populations with higher vulnerability. Moreover, while our analyses controlled for sex and revealed no differences, our sample was disproportionately skewed, with a significantly higher percentage of women than men. Therefore, we cannot confirm the absence of differences with certainty. Prior studies have indicated gender differences in decision-making (Duque et al., Reference Duque, Cano-López and Puig-Pérez2022), as well as in stress response (Pulópulos et al., Reference Pulópulos, Hidalgo, Puig and Salvador2018), so a thorough exploration would be necessary in future research. Similar considerations apply to the stressor type used; while we employed a gold-standard stressor, stressors of different nature elicit diverse psychophysiological responses and may differentially influence social decision-making (Hidalgo et al., Reference Hidalgo, Pulopulos and Salvador2019). Furthermore, exploring other paradigms of social decision-making beyond mere “responder” role could shed light on how stress influences the expression of other emotions pertinent to social contexts, such as generosity. Finally, the absence of direct neural and hormonal measurements limits the extent to which our findings can be integrated with broader theoretical frameworks, such as the STARS model (Mather & Lighthall, Reference Mather and Lighthall2012). Despite these limitations, this study represents the first examination of stress effects on the cognitive subprocesses underlying the prominent UG paradigm. Our findings, at least 15 min post-stressor onset, revealed that while there were no differences in the perception of equity or the number of accepted offers, there was indeed greater difficulty in learning and adapting the equity norm to the given context, leading to more persistent or rigid decision-making. While in a simple decisional task such as UG, this may not prove to be an overt problem, as seen in the absence of changes in immediate, overall prosocial behavior, these deficits may hinder the process and outcomes of decision making in more complex contexts outside of laboratory settings. These results affirm stress influence on social decision-making and highlight a significant target for preventing maladaptive decisions that could hinder social cohesion and integration.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgement
The authors declare that there are no acknowledgements to report.
Author contribution
F.M.: Conceptualization, Methodology, Formal analysis, Writing—original draft. M.P.: Investigation, Writing—review & editing. M.A.S.: Conceptualization, Methodology, Supervision, Writing—review & editing.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing interests
The authors declare no conflicts of interest.