Introduction
The transmissible blood-borne viruses (BBVs) hepatitis C (HCV), hepatitis B (HBV) and HIV, share commonalities such as long periods of asymptomatic illness and susceptibility to treatments that can prevent onward transmission [Reference Hope1, Reference Hatzakis2]. Late diagnoses may result in significant morbidity and mortality [3]. Offering routine testing is an opportunity to identify new cases. It also provides a valuable opportunity to relink already-diagnosed individuals who have disengaged from care. Global targets to achieve a 30% reduction in new cases of chronic HBV and HCV have been introduced by the World Health Organisation's Global Health Sector Strategy on viral hepatitis [4]. Therefore, testing and linking newly diagnosed patients to treatment is a key part of the global strategy to manage viral hepatitis. For HIV, UNAIDS has set a target for 90% of all people with HIV to know their status by 2020 [5].
Highly effective, but costly, direct-acting antiviral (DAA) treatments for HCV are in use in many countries worldwide. However, during the pre-DAA era, many HCV patients tolerated interferon-based treatments very poorly and some disengaged from services [Reference Thomas6]. Finding ways of identifying and re-engaging these individuals is important in addressing the HCV epidemic. When considering HBV, in high prevalence HBV areas, each new diagnosis affords not only the chance of treatment and screening for complications of HBV but also the preventive opportunities for vaccination of household and sexual contacts.
Widespread HIV testing is recommended in all medical settings where ⩾2 in every 1000 people have HIV [7] and already occurs in many clinical settings such as emergency departments (ED) and primary care [7, 8]. Successful treatment reduces infectivity with no onward transmissions observed in observational cohorts and randomised controlled trials [Reference Cohen9, Reference Rodger10].
In England, unlike for HIV, testing for HBV and HCV is targeted to at-risk groups including people who inject drugs, prisoners, those held in immigration centres and family members of those with HBV [11, 12]. Opt-out testing for HBV and HIV in antenatal services is highly successful with >97% tested in 2015 [13, 14]. However, data on prospective testing in the ED setting for viral hepatitis are extremely limited [15, 16].
With almost a quarter of the population in England using the ED annually [17], it has proved an excellent place to introduce opportunistic testing for HIV [17–Reference Rayment20]. The ED is disproportionately used by marginalised and underserved groups (e.g. the homeless and IVDUs) in whom BBVs are known to be more prevalent, particularly HCV [16, Reference McGeary and French21–Reference Thakarar23].
In England, while sentinel surveillance figures in 2015 revealed that only 2.2% of HCV tests taken were derived from the ED, the HCV prevalence in those tested was 2.0%, five times greater than the national average (~0.4%) [16, 24]. A BBV testing week in 2014 in nine National Health Service (NHS) hospital EDs found an overall BBV prevalence of 3.34%, with a particularly high HCV antibody-positive prevalence of 1.83%. Prevalence peaked in white men aged 25–55 years (HCV 2.46%, HBV 1.09% and HIV 1.36%) [Reference Orkin25]. A Dublin ED testing study demonstrated BBV seroprevalence of 5.05% (HCV), 0.5% (HBV), 1.1% (HIV) [Reference O'Connell26]. Linkage to care (LTC) rates have varied: Dublin study 79–98%; BBV testing week 59.4% (new) and 20% (disengaged) patients.
In this implementation roll-out phase of BBV testing (named the ‘Going Viral’ campaign), we tested prospectively for the BBVs HCV, HBV and HIV in a London ED over a 9-month period. Our aims were: (1) to describe the seroprevalence of these three viral infections (all positives, new positives and previously diagnosed but disengaged from care), (2) assess the uptake of opt-out testing for HCV, HBV and HIV in a busy urban ED, (3) to determine LTC rates for new and disengaged patients for each BBV and (4) to quantify the number of attempts required to contact patients and estimate the time spent contacting patients for each BBV. Detailed estimates of seroprevalence and demographic risk factors for infection are described in a separate paper.
Methods
Design and data collection
This was a real-world implementation of a BBV testing programme at a large urban Emergency Department (ED) in London with high background prevalence of HIV (~6.25/1000). The overall prevalence of HIV is 1.9/1000 in England [3] and 5.4/1000 in London [27]. Opt-out testing occurred in all patients over the age of 18 who had blood taken as part of routine clinical care, with exclusions around incapacity to consent and language barrier.
ED staff had been trained the previous year during the aforementioned BBV testing week and received refresher training. Information materials were placed in the waiting areas explaining the result-giving policy, which was that ED attendees undergoing testing would be contacted within 14 days for repeat testing if needed. Consent for opt-out testing was taken verbally according to good clinical practice.
An additional blood sample for BBV testing was drawn when undergoing routine blood tests. This sample was tested for Hepatitis C virus antibody (HCV Ab), Hepatitis B virus surface antigen (HBsAg) and HIV antigen/antibody (HIV Ag/Ab), using the ARCHITECT® fourth-generation combo assay, confirmed with two additional assay platforms (VIDAS® and Geenius™). Reactive HBV and HCV serum samples were automatically tested by our laboratory for e-antigen/antibody (eAg/eAb) and HCV RNA respectively (assay lower limit of detection <15 IU/ml defined as cleared virus).
The HIV team contacted and notified patients with reactive or inconclusive BBV results and arranged a follow-up clinic appointment for newly diagnosed patients and for known positive patients who had disengaged from care. Recall to clinic and confirmation of reactive test were not arranged for patients who confirmed that they were engaged with services at the time of notification. At clinic review, patients with a reactive HIV Ag/Ab test had a repeat sample sent to confirm this, along with an HIV RNA sample to measure HIV viral load (assay lower limit of detection <40 copies/ml defined as viral suppression).
Ethical approval
The Chair of East London and the City Ethics Committee approved this real-world testing roll-out as an extension of previous seroprevalence and testing work in the ED in 2014, without the need for a formal ethics application as the previously demonstrated clinical need and high prevalence of all three BBVs in previous projects supported its implementation.
Descriptive analysis
Data were extracted from the hospital IT system and multiple visits removed by retaining a patient's first BBV test or first visit to the ED if not tested. Uptake was defined as the proportion of patients being tested for at least two BBVs amongst the total population of patients requiring a full blood count (FBC) as part of routine care (FBC population). We described the rates of BBV diagnosis by age, sex, ethnicity and whether they were of no fixed abode (NFA) amongst all patients tested. We assessed the number of patients (i) with a reactive test result, (ii) with a new diagnosis, (iii) needing re-LTC, for BBVs overall and for each individual virus. Cases were defined as patients that were tested for at least two of the three BBVs with a reactive test result (recorded as antigen/antibody detected). New diagnoses were defined as cases where there was no record of the previous diagnosis and whose diagnosis was confirmed as new by the patient following notification of a positive test result. A proportion of patients with a reactive test were uncontactable and therefore we could not exclude the possibility that the uncontactable patients may have been newly diagnosed. Due to this uncertainty, they are defined separately as ‘uncontactable’ (unknown if new or known). Patients who required LTC were defined as living, viraemic patients whose diagnosis was classified as either new or previously known but disengaged from care, or those who were uncontactable.
LTC was defined in stages. ‘Notification’ was defined as the patient being informed of the positive test result. ‘Full LTC’ was defined as notification followed by two clinic visits (or one inpatient consultation plus one clinic visit). In the event that a patient was notified but only attended one clinic visit/inpatient consultation, we classified this as ‘partial LTC’. The total number of attempts at contacting each patient with a reactive test by means of a phone call, text message, letter, via GP, via local substance misuse team or via homeless teams, were recorded. We did not attempt to contact patients with a positive result where we could see evidence/documentation of engagement with a clinic. In addition, patients that were admitted to the hospital were notified during their admission, therefore, did not require contact attempts from the project team. The number of minutes spent on each of these contact types by clinic staff was recorded. The total number of attempts at contacting each patient were summed and multiplied by the average number of minutes spent on each type of contact. These totals were then divided by the number of positive patients to obtain the average number of attempts and time spent by each contact, per patient, per virus.
At the initial clinic review, data on markers of advanced disease were collected. For HBV and HCV infections, significant hepatic fibrosis and cirrhosis were assessed using AST-to-Platelet Ratio Index (APRI) scores, platelet count <150 (if AST not measured) and Fibroscan (where possible). Metavir scores of F3 and F4 were used as indicators of advanced disease and guided urgent referrals. For patients with HIV infection, CD4 count <350/mm3 and/or the presence of AIDS-defining illnesses were used as indicators of advanced disease. Data on cause of death were collected on those who died in order to determine if the BBV contributed directly to the death.
Results
Uptake of testing
After exclusions there were 65 136 unique attendances to the ED from 20 November 2015 to 7 August 2016, of whom 24 981 (38%) had blood taken for FBC assessment during their visit. 6211 of the FBC population met the Going Viral definition of being tested for two (280) or three (5931) BBVs, yielding an overall Going Viral testing uptake of 25% (Fig. 1).
A flowchart of testing uptake and seroprevalence.
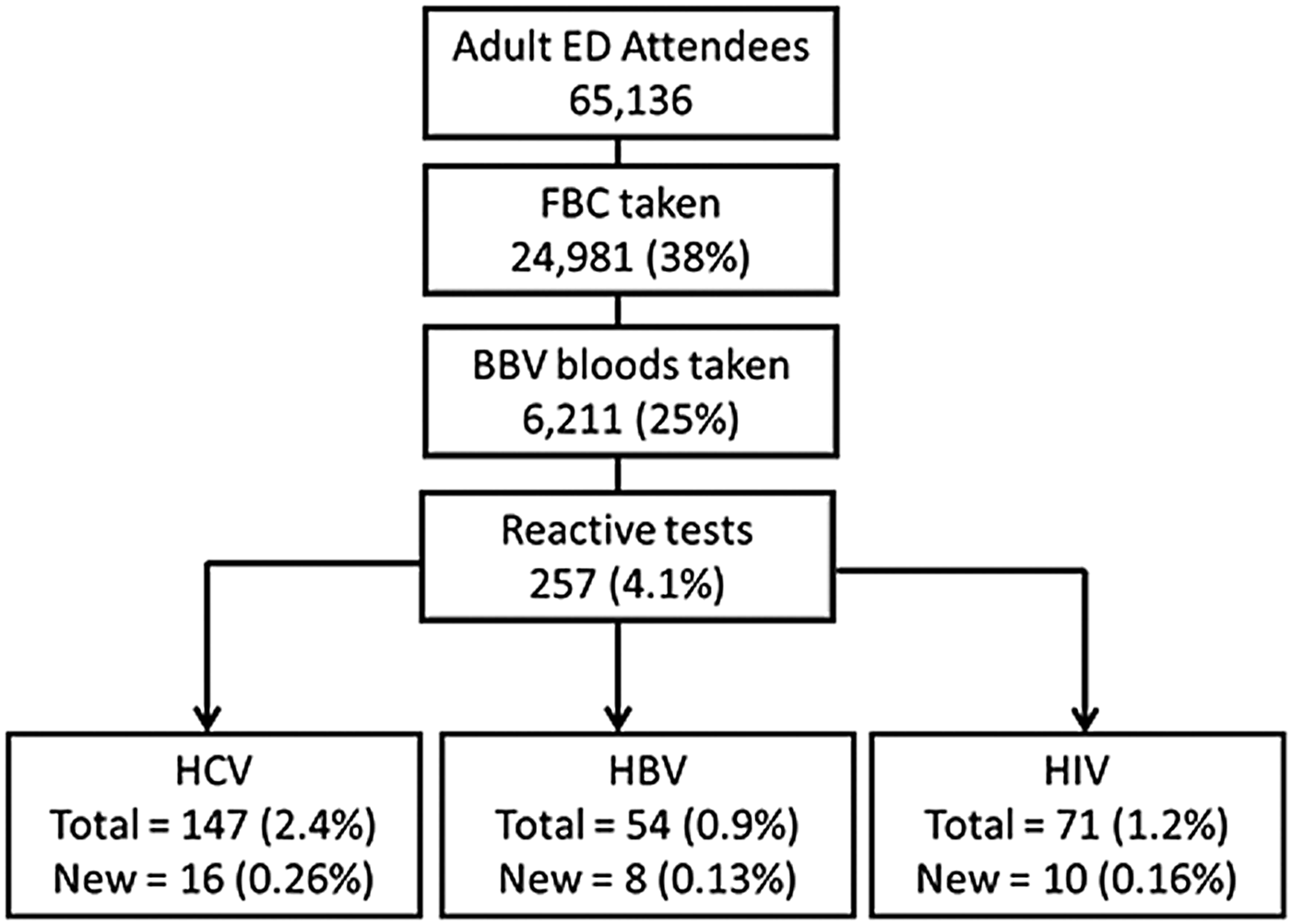
BBV diagnoses
Overall, BBVs were identified in 257 unique patients, resulting in a prevalence of 4.1%. There were 147 HCV diagnoses, 54 HBV diagnoses and 71 HIV diagnoses. Fifteen patients were co-infected; two HBV/HCV, three HBV/HIV, and 10 HCV/HIV. The prevalence for the three viruses varied: 2.4% for HCV (newly diagnosed 0.26%), 0.9% for HBV (newly diagnosed 0.13%) and 1.2% for HIV (newly diagnosed 0.16%). Due to the possibility that some uncontactable patients may have been new diagnoses, the new diagnosis prevalence's shown may represent slight underestimates. Overall, individuals aged 40–49 years had the highest BBV prevalence and 73% of cases were male (Table 1).
Numbers and distribution of patients tested at each stage of the pathway

a % given as a proportion of ED attendees with blood taken.
b % given as a proportion of those BBV tested.
c % given using number tested for that particular BBV as the denominator, per variable (total 6159 tested for HCV; 6023 tested for HBV; 6092 tested for HIV). U, unclassified; NFA, no fixed abode.
Linkage
Of the 257 patients, diagnosis status (new/known) was established for 234 patients. Eighty-four of the 257 patients tested (33%) required LTC (Table 2). Thirty of the cases confirmed as new required linkage (0.55%) and all were notified of their infection (Tables 2 and 3). A further 31 people required re-LTC as they were previously diagnosed but not currently engaged with services [HCV 17 cases (0.27%), HBV 10 (0.16%), HIV 5 (0.08%), one HIV/HCV co-infection]. Twenty-three patients were uncontactable; four of whom were confirmed known cases by case-review and 19 of whom were of undetermined status. Overall, notification was achieved in 59 (70%); 14 (17%) attended only one appointment, 40 (48%) attended two or more appointments and 30 (36%) remained unlinked. Of the 61 contactable patients, notification was achieved in 97%, 23% attended only one appointment, 66% attended two or more appointments and 11% remained unlinked. The breakdown of linkage per BBV is shown in Table 3.
Diagnosis status of HBV, HCV and HIV cases identified
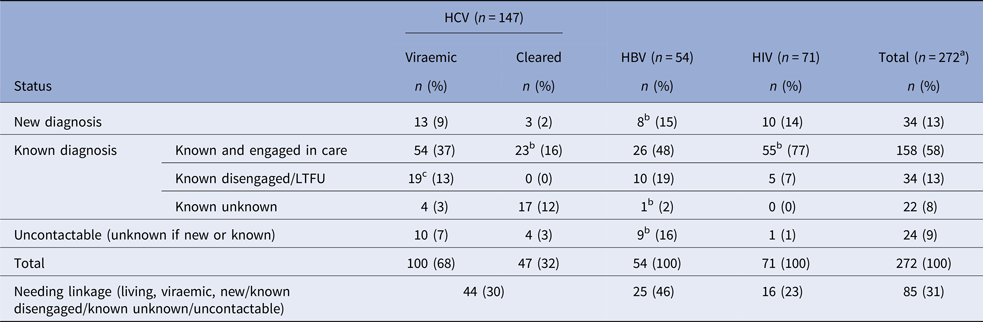
a Total n = 272 due to 15 co-infections.
b Includes one patient deceased during follow-up.
c Includes two patients deceased during follow-up.
Breakdown of linkage outcomes
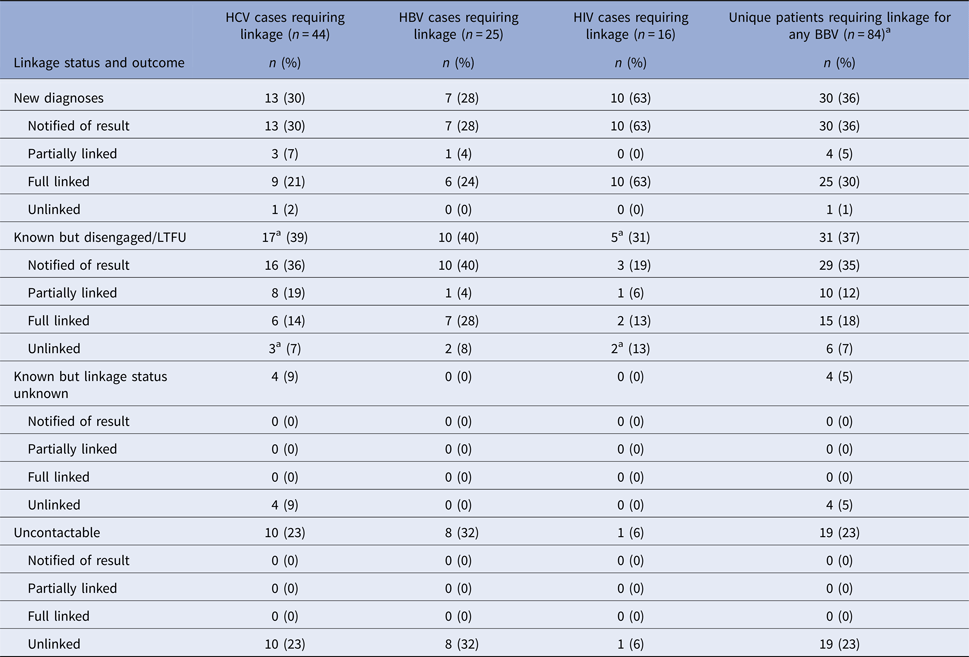
a 1 HIV/HCV co-infected patient.
Outcome of those with positive tests (Table 3)
Overall, 83% of patients were contactable. Of the contacted patients, 15% had new infections and 85% were known. Seventeen per cent of patients were uncontactable; of these, 48% were confirmed to have known infections by the case-note review, 52% were of undetermined status. The highest LTC rates (partial or full) were achieved for HIV (81%), followed by HBV (60%), then HCV (59%). Of the contactable patients, those with a newly diagnosed BBV were much more likely to be successfully linked to care (100% notified, 83% fully linked, 13% partially linked and 3% unlinked) than those with a previously known infection.
HCV accounted for the highest number of diagnosed cases (147) and the highest number requiring LTC (44). However, the HCV cohort had the lowest rate of successful linkage, with 18/44 (41%) remaining unlinked; 14 of these were uncontactable (four confirmed known by case review, 10 undetermined status). Of the 30 contactable patients, all 13 new HCV diagnoses were notified of their infection, 12 (92%) attended at least one clinic appointment and 9 (69%) attended hepatology clinic follow-up. Of the 17 previously diagnosed disengaged patients, 16 (94%) were notified, 14 (82%) attended at least one clinic appointment and six (35%) attended hepatology clinic follow-up. Eight of the 25 (32%) HBV patients requiring linkage were uncontactable and of undetermined status. Of the 17 contactable patients, all seven new HBV diagnoses were notified and all attended at least one clinic appointment. Of the 10 disengaged patients, all were notified and eight (80%) attended at least one clinic appointment. Of the 16 patients requiring linkage for HIV, one was uncontactable and of undetermined status. Of the 15 contactable patients, all 10 newly diagnosed with HIV were notified and linked to HIV care; two have subsequently defaulted from follow-up and the remaining eight have started ART. Of the five disengaged patients, three (60%) attended at least one clinic appointment.
Patient recall
A total of 1041 contact attempts were made to contact the BBV cases (average 4.1 contact attempts per patient), with a total of 2816 min spent (average 11 min per patient). The 875 contact attempts to HCV cases accounted for 84% of the total contacts made for all BBVs and 2315 min accounted for 82% of the time spent, requiring 6.0 attempts and 15.7 min per case on average. HBV cases required average 2.0 contact attempts and 6.7 min per case. HIV cases required average 0.8 attempts and 1.9 min per case.
Uncomplicated disease vs. serious BBV-related outcomes
Of the 84 patients requiring LTC, 26 (31%) had advanced disease and/or serious BBV-related outcomes (Table 4). Disease status was not established for patients already engaged in care.
Disease status in those requiring linkage to care
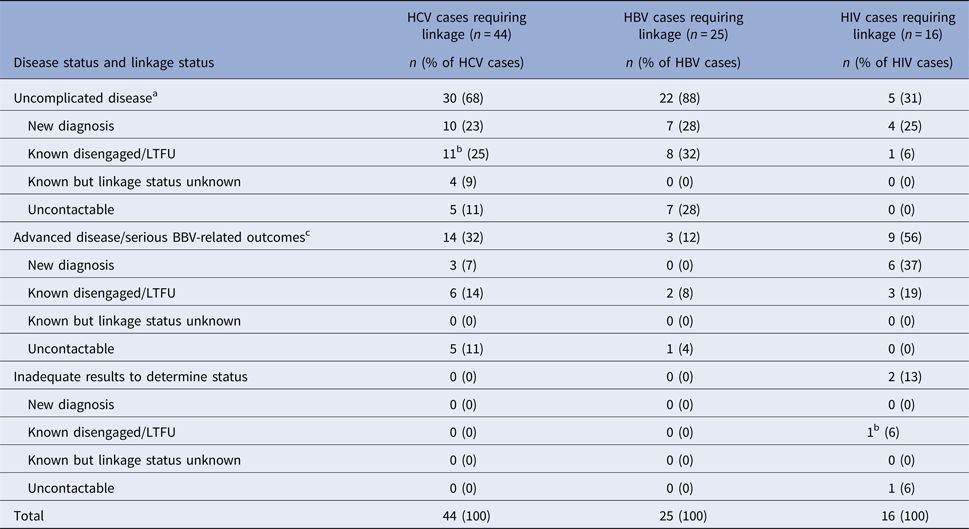
a CD4 >350 or no evidence of cirrhosis (platelets <150, APRI >1, FS score ⩾F3) or HCC.
b denotes co-infected patient.
c CD4 <350, AIDS-defining illness, evidence of cirrhosis or HCC.
Nine of the 16 (56%) who required linkage for HIV infection were found to have late HIV disease, of whom five also had HIV-indicator conditions. A third of the HCV cases requiring LTC had advanced disease, including one newly diagnosed patient who was diagnosed with metastatic hepatocellular carcinoma (HCC). Three (12%) people requiring LTC for HBV infection had serious BBV-related outcomes, including one patient with HCC.
Acute infection
Four patients (1.6%) were assessed as having possible acute infections. One person presented with evidence of HIV seroconversion, evidenced by an incident avidity test and a negative HIV test within the previous 6 months. Three presented with possible acute HBV infection, evidenced by very high HBV DNA, positive core antibody IgM and e-antigen positive status. Two became chronically infected; the third cleared the virus and reverted to an HBV surface antigen negative status.
Deaths
Of the 257 patients that tested positive for a BBV during this study, seven died during the follow-up period (2.7%) (Table 5). There were five deaths (1.9%) directly attributable to the BBV; four were liver-related deaths.
Deaths during the follow-up period

Ke, Known engaged in care; Ku, known but engagement status unknown; Kd, Known disengaged/LTFU; HCC, hepatocellular carcinoma.
Hepatitis C
Median (IQR) age of cases was 46 (37–55) years and 105 patients (71%) were male. Prevalence was higher amongst males (3.2%) and highest amongst the White ethnic group (4.1%; 4.3% amongst White British ethnicity, 3.8% amongst White other) (Table 1). We were able to establish whether the diagnosis was new or known for 133 cases; of these, 16 (12%) were newly diagnosed and 117 (88%) were previously known, giving a new diagnosis prevalence of 0.26% in the tested population.
100 of the 147 (68%) HCV cases were viraemic (Table 2). The mean HCV RNA was 1 799 739 IU/ml (range 30–5 1 135 911 IU/ml). Sixteen of the 86 contactable patients were newly diagnosed, of whom 13 (9% of total positive) were viraemic (mean HCV RNA 1 718 790 IU/ml). Ten were male, median (IQR) age was 49 (36–63) years. Risk factor information was available for 11/13 newly diagnosed viraemic patients; six had a history of intravenous drug use (three current, three past), two reported a history of intranasal only drug use, one person had received a blood transfusion in the 1970s, one had an HCV-positive partner who was most probably infected from infected blood products pre-1991 and one had had surgery outside of the UK. Prevalence was highest amongst NFA patients (11%) (Table 1). However, of the contactable patients (in whom we could establish diagnosis status), no newly diagnosed HCV cases were homeless.
Nineteen patients were previously diagnosed and unlinked to care, of whom two died. All were viraemic (mean HCV RNA 3 227 776 IU/ml). Risk factor information was gathered for the 14 patients that attended at least one clinic appointment; seven had a history of intravenous drug use (two current, five past), three reported a history of intranasal only drug use, one person had received a blood transfusion pre-1991, two people reported unsterile tattoos/piercings and one had had surgery outside of the UK. Fourteen patients were uncontactable (at least nine of these were homeless/registered as NFA). All were viraemic (mean HCV RNA 1 432 774 IU/ml).
Of the 77 patients who self-reported a known diagnosis and were already engaged with HCV services, almost three quarters were viraemic (54 patients, 70%).
More than half (57%) of the HCV-positive patients had attended the ED at least once in the preceding 2 years (Table 6). The most frequent ED user group was patients with a known HCV diagnosis that self-reported being engaged in HCV care (mean 5.3 visits per patient in the preceding 2 years). Forty-six per cent of the newly diagnosed patients had attended ED in the preceding 2 years; therefore, if BBV testing was routine prior to the study time period, these patients may have been diagnosed earlier. 74% of the lost-to-follow-up group had attended before and 40% of the uncontactable group.
HCV patients use of ED in 2 years preceding study start
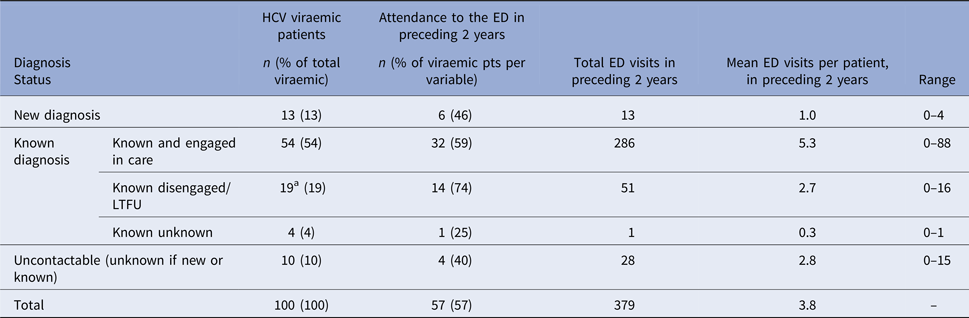
a Includes two patients deceased during follow-up.
Hepatitis B
A total of 54 patients from 6023 tested for HBV had a positive s-antigen blood test, an overall observed prevalence of 0.9% (Table 1). Median (IQR) age of cases was 43 (36–58) years and 36 cases (67%) were male. Prevalence was higher amongst the Black (2.4%) and White other (non-British) ethnicities (1.5%) (Table 1). Amongst the 45 contactable cases, eight (18%) were new diagnoses, giving a new diagnosis prevalence of 0.13%.
Eight of the 44 contactable patients with HBV were newly diagnosed; six were male and median (IQR) age was 36.5 (35.5–42.5) years. Three patients were Black African, two South Asian and three White Other (Italian, Polish and Russian). Mode of acquisition included MSM (n = 2), country of high prevalence (n = 3) and heterosexual sex with an HBV-infected partner (n = 1). Two patients had no clear risk factor. All eight patients had detectable HBV DNA (mean 4.2 million IU/ml, range 92–28,192,236 IU/ml). Three of the patients presented with possible acute HBV infection and were found to be e-antigen positive. All three were symptomatic of jaundice and/or abdominal pain, had markedly elevated transaminases, high HBV DNA levels and were s-antigen positive and core antibody IgM positive. One of these went on to clear the virus; the other two seroconverted to e-antibody and have developed chronic infection.
HIV
There were 71 cases of HIV identified from 6092 HIV tests, resulting in a prevalence of 1.2%. Median (IQR) age of cases was 43 (30–50) years and 57 cases (80%) were male. Prevalence was higher amongst males (1.8%) and highest amongst the Black ethnic group (4.4%) (Table 1).
Ten of the seventy contactable patients with HIV were new diagnoses. Mode of acquisition included five MSM, four heterosexual patients from a country of high prevalence and one heterosexual patient whose partner subsequently tested HIV positive due to contact tracing offered by the programme. For the new diagnoses, CD4 count at diagnosis ranged from 1 to 1122 cells/mm3 and the mean HIV viral load was 145 588 cps/ml. Six patients presented with late disease, of whom four (each with CD4 <100 cells/mm3) had HIV-indicator conditions. Three patients presented with early disease and no clinical indicators for HIV infection. There was one diagnosed HIV seroconversion, based on a positive HIV antibody result with a documented negative test 6 months earlier.
Five patients presented with AIDS-defining illnesses (cerebral toxoplasmosis, pneumocystis pneumonia (PCP), chronic diarrhoea and wasting, cytomegalovirus (CMV) retinitis and recurrent bacterial pneumonia); four of these were new diagnoses (mean CD4 count 28 cells/mm3, range 1–53 cells/mm3), the fifth was known but lost to follow-up. All five are now retained in care and have started anti-retrovirals.
Discussion
Routine testing in an inner London ED identified a high combined BBV seroprevalence (4.1%), similar to the 6.6% BBV prevalence determined in ED patients tested in Dublin [Reference O'Connell26], and to the 3.4% in the previously described BBV testing week in nine UK EDs [Reference Orkin25]. Of those who needed LTC, 56% of patients with HIV and 32% with HCV presented in late disease. Seven of 257 individuals (2.7%) with reactive BBV tests died. Five of the deaths were BBV-related.
Our work in comparison with other studies focused on describing all those who needed LTC and we showed that even amongst known diagnoses there was a large proportion of viraemic individuals not retained in care. Our contention is that routine testing is a means not only of identifying new infections but crucially also an opportunity to link disengaged patients to care.
Our discussion focuses on HCV for the following reasons:
(1) High estimated burden of undiagnosed BBV infection (an estimated 50% of 160 000 cases in England [16] compared with 13% of 110 000 HIV cases in the UK [3]). Unfortunately, there is no routine information on the number of people treated for HBV, or prevalence surveys of the general population available.
(2) Recent roll-out of curative treatment for HCV.
(3) An opportunity to strive towards eradicating HCV with better testing and access to treatment.
HCV
The overall prevalence of HCV in our population was 2.4% with a peak prevalence of 4.9% in men aged 40–49 years. Although injecting drug use is thought to account for approximately 90% of UK infections [16, 28], amongst the 25 new or disengaged HCV-positive patients on whom risk factor data were collected, only half reported having ever injected drugs. Risk factors identified in our small group of ED patients included inhaled illicit drug use, surgery outside of the UK and receiving blood products prior to 1991. Homelessness was also a factor in patients with known HCV but not in our contactable newly diagnosed patients. We are unable to comment on risk factors for the fourteen uncontactable patients.
Sixty-eight per cent of the HCV Ab-positive patients were RNA positive and thus in need of assessment for HCV treatment. A third of these viraemic patients needed linkage to HCV clinics either as new patients or as people who had previously disengaged from care. It is also of interest that 70% of the patients who identified as engaged with hepatitis services were nonetheless viraemic and hence untreated. This reflects an as-yet-untreated pool of engaged patients attending HCV services but not yet treated with DAAs.
Hepatitis C is a curable infection thanks to highly efficacious modern treatment options. However, a large proportion of the 160 000 infected people in England have not yet been identified and similar low diagnosis rates have been reported in other European and North American countries. Identifying new cases and relinking disengaged patients to care is a very important way of increasing the pool of treatable patients and moving towards eradicating HCV. This is only ever achievable if a sufficient proportion of patients are diagnosed and treated.
Successful implementation of HIV testing in EDs and other hospital settings prevents late diagnoses and deaths; with this in mind, it is essential that we explore and evaluate where best to deliver HCV testing beyond the existing targeted-testing strategies in order to identify and notify new cases.
By quantifying and describing the number of contact attempts to achieve an LTC, we showed that amongst the contactable patients, we could successfully notify all the new cases; however, it was six times more difficult to engage individuals with HCV than those with HIV or HBV. Additionally, HCV patients who had previously been lost to follow up were less successfully linked to care compared to not only the other BBVs but also those with newly diagnosed HCV. We demonstrated a high proportion of homelessness in the contactable known, but not new, HCV cases which may account for the difficulties engaging these patients. Additionally, we must bear in mind that homelessness may have been more prevalent amongst the uncontactable patients, of whom new/known diagnosis status could not be determined. These time investments are to be borne in mind when considering costing such an intervention, especially the re-linkage of disengaged HCV patients.
HBV
Seroprevalence for HBV during the campaign was 0.9% (0.13% new diagnoses), 2.25 times higher than the national average in the UK (0.4%), and 3.2 times the national average in the 30–49-year age group. This may be due to the ethnic diversity of the ED population sampled, with only 4% of patients positive for HBV identifying as White British. We identified three acute cases, a high-risk period in terms of onward transmission, and three diagnosed patients subsequently died, two liver-related, highlighting the clinical importance of HBV testing.
HIV
Seroprevalence for HIV infection was 6.3 times higher than the UK average, comparable with prevalence rates in central London (0.6–1.4%) [3] and similar to previously described at this ED [Reference Orkin25]. The benefits of prompt HIV treatment both in terms of individual health benefits and negation of onward transmission risk are irrefutable, yet 13% of people with HIV in the UK remain undiagnosed and 39% are still diagnosed late which is associated with impaired health outcomes and mortality [3]. Numerous studies have demonstrated missed opportunities to test people who present late with HIV. Employing routine ED testing, as recommended by the National Institute for Health and Care Excellence (NICE) [7], is an important tool to utilise opportunities to test and improve outcomes for patients and their partners.
Limitations
An important limitation of this programme is that uptake averaged only 25%, which is comparable to previous UK testing programmes [Reference Palfreeman29] but introduces the possibility of selection bias which may falsely elevate prevalence if an ‘at-risk’ population was overrepresented in the cohort. More people in the higher risk ethnic groups and age groups were tested during our roll-out. Additionally, as per previous real-world work, we were unable to distinguish between tests not being offered and patients’ refusal to test. Another factor that limited interpretation of the data is that we did not establish risk factors for those who identified as being engaged with care when we contacted them to notify them of the reactive test, so we cannot compare these to that of the patients who attended follow-up. As a testing programme, rather than a formal trial, only people who required engagement in clinical care were called back in by our team. Patients were only classified as ‘new’ if we were able to contact them to clarify their testing history, so some were classified as ‘disease status unknown’ or ‘unclassified’; this may have resulted in an underestimate of new diagnoses. In addition, given the number of new diagnoses is small, and the proportion attending our centre for follow-up even smaller, we have not been able to comment on disease-modifying outcomes, as we do not feel we have enough data to make comments on this meaningful. We note that this high prevalence London area may not be transferable or relevant to other areas of the UK or abroad.
Conclusions
This high prevalence population conformed to an age-gender specific bracket of males between 40 and 49 years old across the BBV spectrum. One-third of those identified needed to be linked to care. One-third of those diagnosed presented with advanced disease or with serious BBV-related complications. A high percentage (2.7%) of the tested population died, predominantly of BBV-related disease. Testing was highly relevant to this population and ED setting. LTC was challenging, especially in individuals already known to be diagnosed with HCV, and more than a quarter of patients requiring linkage were completely uncontactable. Dedicated resources would be necessary to achieve effective engagement in this group.
Acknowledgements
CO is the guarantor of the article. CO conceptualised and directed the project. SP, KA and GRF saw the patients. SP and SU collected the data. CYT did the virology testing. NB and SB performed all statistical analyses. SP, NB and CO drafted the article. All authors revised the manuscript critically and approved the final version submitted for publication. This work was supported by the Gilead UK and Ireland Fellowship Fund. We thank Murad Ruf for advice regarding the grant submission, Bhavi Trivedi for her assistance with data management, Vince Lawlor for assisting with patient recall and notification, and all of the staff and patients of the Royal London Hospital Emergency Department. SP has received speaker fees and travel sponsorship from Janssen. GRF has received consultancy and speaker fees from AbbVie, Gilead, Merck and Roche. CO has received research grants, speaker fees, advisory board fees and travel sponsorship from all companies providing anti-retroviral medications: Janssen, MSD, Viiv and Gilead.
Declaration of interest
None.









