Introduction
Dantopaite, ideally Ag5Bi13S22, is the 6P homologue of the pavonite series (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010). In addition to its type locality, Erzwies, Austria, it is currently reported only from the Rozália mine, Slovakia (Jeleň et al., Reference Jeleň, Pršek, Kovalenker, Topa, Sejkora, Števko and Ozdín2012), the Clara mine, Germany (Kolitsch et al., Reference Kolitsch, Bayerl and Topa2019), the Yugo-Konevo deposit, Urals, Russia (Kasatkin et al., Reference Kasatkin, Belogub, Kuznetsov, Novoselov, Škoda, Nestola and D.A2023) and the Mohawk mine, Goldfield, USA (Musetti et al., Reference Musetti, Sejkora, Biagioni and Dolníček2024).
The selenium contents in homologues of the pavonite series are usually low. Topa and Paar (Reference Topa and Paar2008), for instance, reported 0.25 Se atoms per formula unit (apfu) in cupromakovickyite, and up to 0.10 Se apfu were found by Žák et al. (Reference Žák, Frýda, Mumme and Paar1994) in makovickyite. Higher Se contents are very rare. Kolitsch et al. (Reference Kolitsch, Bayerl and Topa2019) described the possible existence of the Se-analogue of dantopaite from the Clara mine in Germany; possible Se-dominant members of the pavonite series are also mentioned from the Ozernovskoe deposit, Kamchatka, Russia by Kovalenker et al. (Reference Kovalenker, Nekrasov, Sandomirskaya, Nekrasova, Malov, Danchenko and Dmitriyeva1989) and Kovalenker and Plotinskaya (Reference Kovalenker and O.Yu2005). The crystal structure of dantopaite with exceptionally high Se content (7.91 apfu) from the Mohawk mine, Goldfield, USA were published by Musetti et al. (Reference Musetti, Sejkora, Biagioni and Dolníček2024).
In 2017, an unnamed “Se-analogue of pavonite” was described from the Princ Evžen deposit, Potůčky, Czech Republic (Sejkora et al., Reference Sejkora, Šrein, Šreinová and Dolníček2017). New crystal-chemical investigation of this sample was undertaken, which allowed for the solution of its crystal structure and the proposal of the new mineral species selenodantopaite. This new mineral and its name were approved by the Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (IMA 2023-092, Sejkora et al., Reference Sejkora, Biagioni, Makovicky and Šrein2024). Selenodantopaite is named in accord with its composition and its relationship with dantopaite. Its mineral symbol, in accord with Warr (Reference L.N2021), is Sdnt. The holotype material (polished section) is deposited in the mineralogical collection of the Department of Mineralogy and Petrology of the National Museum, Prague, Czech Republic (catalogue number P1P 22/2023) and the crystal used for the single-crystal X-ray diffraction study is kept in the mineralogical collection of the Museo di Storia Naturale of the Università di Pisa, Via Roma 79, Calci (PI), Italy, under catalogue number 20068.
Occurrence and mineral description
Occurrence
Selenodantopaite has been discovered in samples collected on the remnants of mine dumps (GPS coordinates: N 50°25.56855’, E 12°45.04677’) of the abandoned small uranium deposit Princ Evžen near Potůčky in Krušné hory Mountains, Czech Republic. This deposit, mined in the years 1956–1959, is represented by the main hydrothermal vein E1 (direction NNW–SSE, thickness 3–15 cm, explored over 1.5 km length to 150 m depth under the surface) with several less significant pennate veins (e.g. E6, E7, E8). The Princ Evžen deposit is localised in the Ordovician phyllite rocks of the Krušné hory crystalline complex in exocontact of the Horní Blatná granite body. Prevailing chlorite–sericite phyllites contain some intercalations of quartzites and amphibolites; the amphibolites especially, with impregnations of pyrite and pyrrhotite, were suitable for formation of uranium mineralization (Veselý, Reference Veselý1982; Sejkora et al., Reference Sejkora, Šrein, Šreinová and Dolníček2017).
Selenodantopaite occurs in quartz gangue with abundant strongly coffinitized uraninite, chalcopyrite and pyrite. Bohdanowiczite, an unnamed selenide with composition close to ideal formula (Bi,Ag)3(Se,S,Te)4, minerals of the galena–clausthalite solid solution, sphalerite and tennantite-(Fe) were found in the association (Sejkora et al., Reference Sejkora, Šrein, Šreinová and Dolníček2017). The crystallization of selenodantopaite is related to a low-T hydrothermal event.
Physical and optical properties
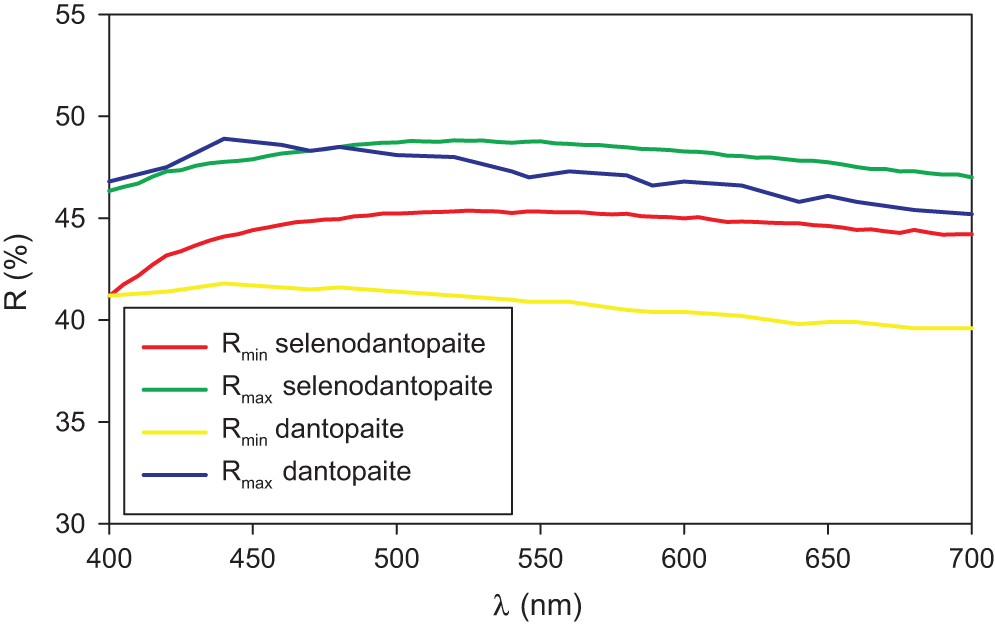
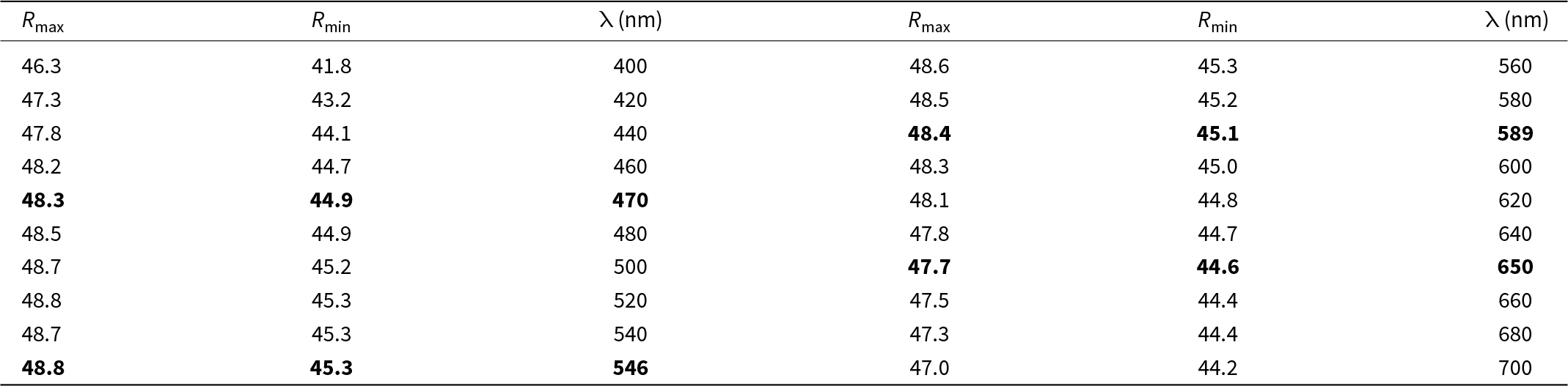
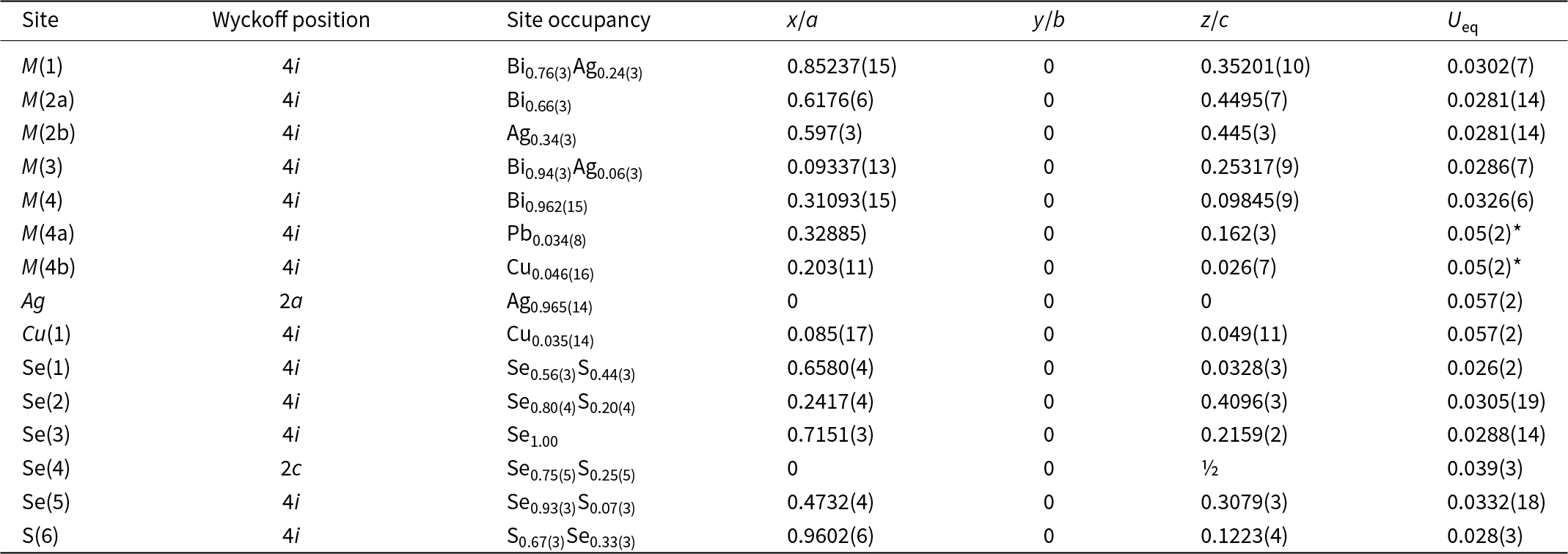
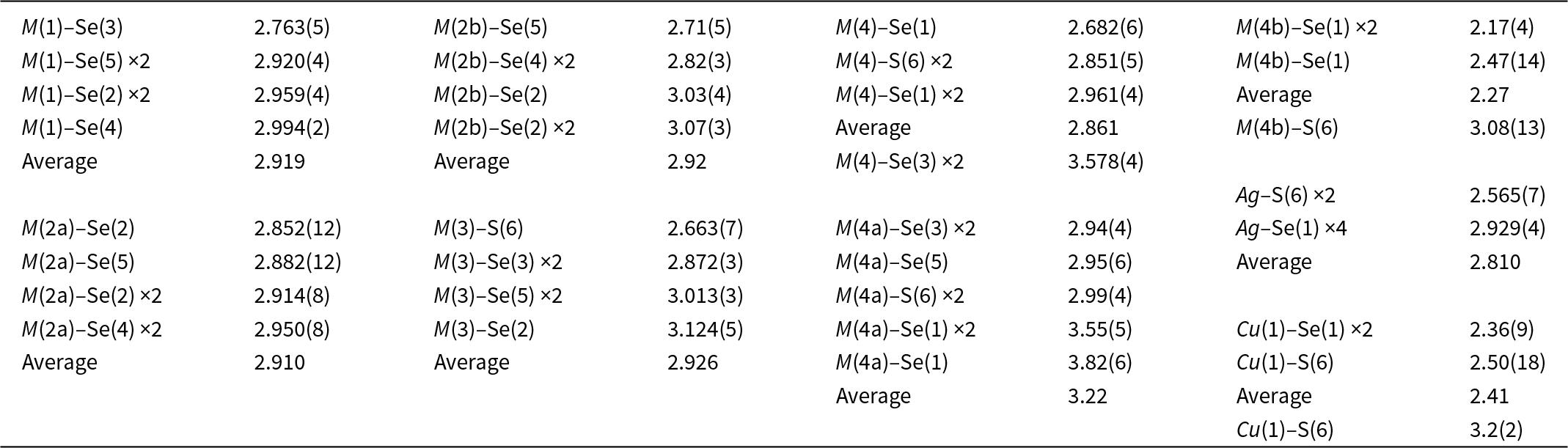
Selenodantopaite forms anhedral grains up to 100 μm but usually only 10–30 μm in size (Fig. 1). The mineral is dark grey in colour and opaque in transmitted light; it has a metallic lustre. Cleavage was not observed; it is brittle with a uneven fracture. Calculated density for the empirical formula (Z = 1) is 7.403 g cm–3 for Cu-bearing and 7.428 g cm–3 for Cu-poor selenodantopaite (based on the empirical formulae and single-crystal unit-cell parameters). Mohs hardness is probably ∼3½ by analogy with dantopaite. In reflected light, selenodantopaite is white to light grey; bireflectance and pleochroism are weak, anisotropy is distinct with light bluish white – light purplish brown rotation tints. Internal reflections were not observed. Reflectance spectra were measured in air with a TIDAS MSP400 spectrophotometer attached to a Leica microscope (50× objective) using a WTiC (Zeiss no. 370) standard, with a square sample measurement field of ca. 8 × 8 μm. The results for the 400–700 nm range are given in Table 1 and in Fig. 2 they are compared with published data for dantopaite from Erzwies, Austria (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010).
(a) Reflected light image of selenodantopaite from Potůčky (white) associated with chalcopyrite (yellow) and minor pyrite (light yellow) in quartz gangue with abundant strongly coffinitized uraninite. (b) Back-scattered electron image of the same area, selenodantopaite is white, coffinitized uraninite grey. Holotype sample (catalogue number P1P 22/2023).

Reflectance curves for selenodantopaite from Potůčky (this work) in comparison with the curves for dantopaite from Erzwies, Austria (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010).

Reflectance values (%) for selenodantopaite*

* The reference wavelengths required by the Commission on Ore Mineralogy (COM) are given in bold.
Chemical composition
Chemical analyses were performed using a Cameca SX100 electron microprobe operating in wavelength-dispersive mode (25 kV, 20 nA and 0.7 μm beam size). The following standards and X-ray lines were used to minimise line overlaps: Ag (AgLα), Bi2Se3 (BiMβ), PbSe (SeLβ), chalcopyrite (CuKα, SKα), pyrite (FeKα) and PbS (PbMα). Peak counting times were 20 s for all elements, and 10 s for each background. Arsenic, Au, Cd, Co, Ga, Ge, Hg, Mo, Ni, Pd, Sn, Sb, Te, Tl and Zn were all found to be below the detection limits (0.02–0.05 wt.%). Raw intensities were converted to the concentrations of elements using the automatic ‘PAP’ (Pouchou and Pichoir, Reference Pouchou, Pichoir and Armstrong1985) matrix-correction procedure.
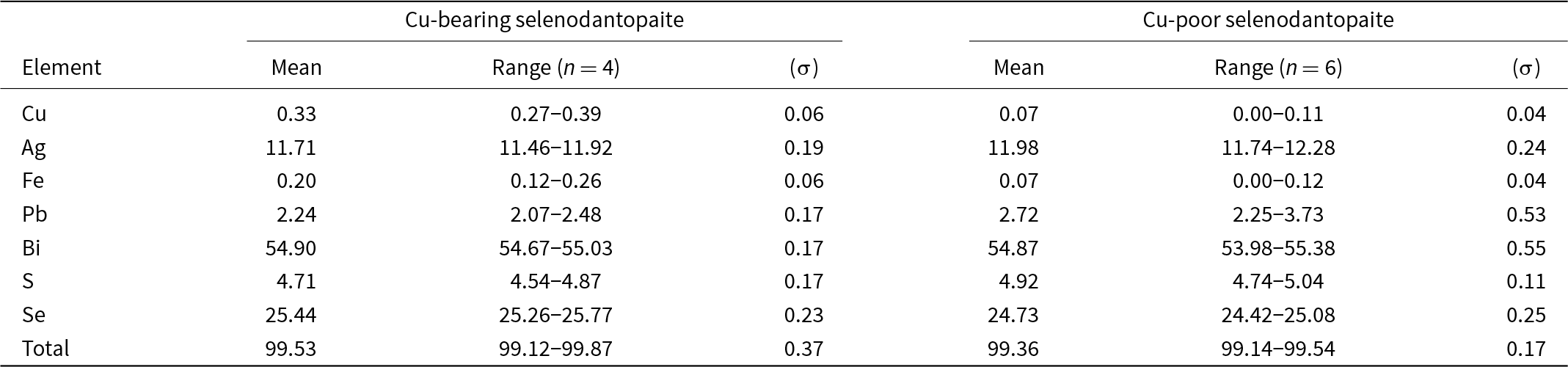
Analytical data for selenodantopaite sample used for X-ray single-crystal diffraction are given in Table 2. Two slightly different chemical compositions were observed: Cu-bearing (Cu content ranging between 0.20 and 0.29 apfu) and Cu-poor (up to 0.08 Cu apfu). The number of atoms per formula units (apfu) of selenodantopaite, calculated on the basis of (S + Se) = 22 apfu, is as follows: (1) Cu-bearing selenodantopaite: Cu0.24(4)Ag5.09(7)Fe0.17(5)Pb0.51(4)Bi12.32(21)Se15.11(21) S6.89(21); (2) Cu-poor selenodantopaite: Cu0.05(3)Ag5.23(11)Fe0.06(4)Pb0.62(12)Bi12.38(13)Se14.77(16)S7.23(16).
Chemical data (in wt.%) for selenodantopaite (crystal used for single crystal study)*

* (σ) = estimated standard deviation; n = number of spot analyses.
The simplified formula is (Ag,Cu)5(Bi,Pb,Fe)13(Se,S)22 and the ideal formula is Ag5Bi13Se22, which requires Ag 10.80, Bi 54.41, Se 34.79, a total of 100.00 wt.%.
X-ray diffraction data
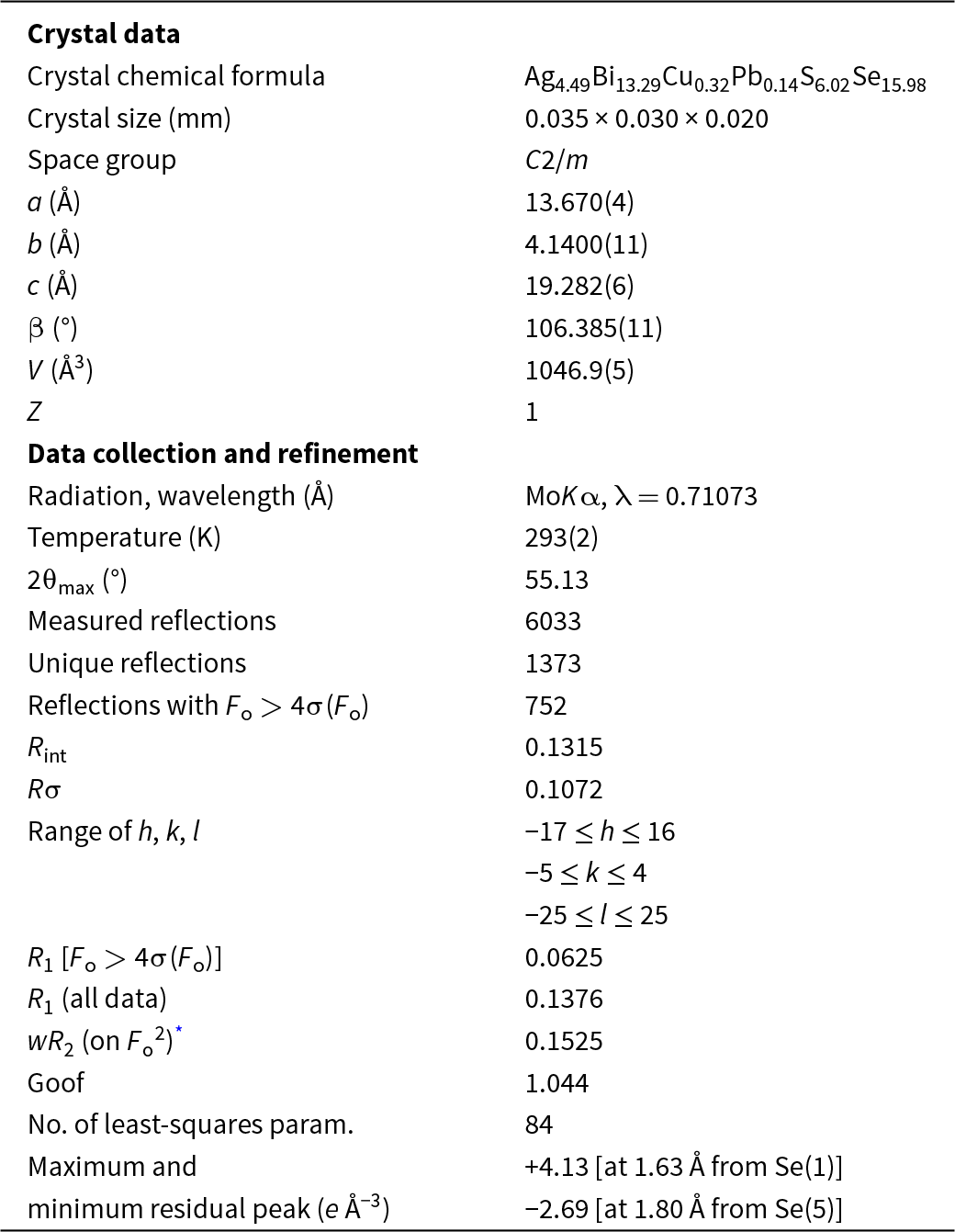
A short prismatic fragment of selenodantopaite, 35 × 30 × 20 μm in size, extracted from the polished section analysed using an electron microprobe, was mounted on a carbon fibre and examined with a Bruker D8 Venture single-crystal diffractometer equipped with an air-cooled Photon III area detector and microfocus MoKα radiation (Centro per l’Integrazione della Strumentazione Scientifica dell’Università di Pisa, University of Pisa). The detector-to-crystal distance was 38 mm. Data were collected using ω and φ scan modes, in 0.5° slices, with an exposure time of 120 s per frame. A total of 576 frames were collected and they were integrated with the Bruker SAINT software package using a narrow-frame algorithm. Data were corrected for Lorentz-polarization, absorption and background. Unit-cell parameters, for monoclinic space group C2/m, refined on the basis of the XYZ centroids of 1040 reflections above 20 σ(I) with 7.689 < 2θ < 47.60°, are a = 13.670(4), b = 4.1400(11), c = 19.282(6) Å, β = 106.385(11)° and V = 1046.9(5) Å3. The a:b:c ratio calculated from unit-cell parameters is 3.3019:1:4.6575.
The crystal structure of selenodantopaite was refined using Shelxl-2018 (Sheldrick, Reference Sheldrick2015) starting from the atomic coordinates of dantopaite (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010). Neutral scattering curves for Cu, Ag, Pb, Bi, S and Se were taken from the International Tables for Crystallography (Wilson, Reference Wilson1992). After several cycles of refinement, the R 1 converged to 0.083, thus confirming the correctness of the structural model. In dantopaite, the metal excess is due to the accumulation of Cu and Ag in the thin structural slab, owing to the occurrence of partially occupied Ag (at 0, 0, 0), Cu(1) (at x, 0, z), and Cu(2) (at 0, y, 0) sites (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010). During the refinement of the crystal structure of selenodantopaite, the Cu(1) and Cu(2) sites were initially removed, because their displacement parameters were negatively defined. In the final stages of the crystal structure refinement, an anisotropic structural model refined to R 1 = 0.0666, with the Ag site fully occupied. However, as both varieties of selenodantopaite observed in the studied material show a metal excess (0.33 and 0.34 apfu, for the Cu-bearing and Cu-poor variety, respectively), some additional metal positions, having low occupancy, should be located close to the Ag site. A scrutiny of the difference-Fourier map was performed, locating again two low-occupied positions close to Ag at (x, 0, z) and (0, y, 0). Unfortunately, any attempt to refine their fractional atom coordinates was unsuccessful, and consequently their positions were fixed, whereas their site occupancies were freely refined. Moreover, their position was very close to the Ag site, at ∼0.55–0.65 Å, and could be interpreted as due to the atomic displacement of Ag atoms. This configuration is not able to explain the metal excess shown by selenodantopaite, because the split positions are mutually exclusive. Moreover, the total site scattering at the cation sites is ∼1351 electrons per formula unit, whereas the anion site scattering is ∼645 electrons. These values have to be compared with those calculated from electron microprobe analyses: ∼1315 and ∼1327 for cations in Cu-bearing and Cu-poor selenodantopaite, and ∼624 and ∼618 for anion positions, respectively. Owing to these shortcomings and to the necessity to find interstitial sites justifying the metal excess, a different solution was proposed. The maximum residual in the difference-Fourier map was close to the M(4) site and, taking into account bond distances, it was interpreted as a partially occupied Pb site. Such a maximum was also observed in dantopaite, where it was interpreted as due to the possible presence of subsidiary 7P lamellae in 6P dantopaite (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010). Another two additional maxima were found: one of them still close to M(4), whereas the other is close to the Ag position. The displacement parameter of the latter was refined anisotropically, constraining it to be the same as that of the Ag site, whereas the displacement parameters of the two subsidiary sites of the M(4) position were refined isotropically. The structural model of selenodantopaite converged to R = 0.0625 for 752 reflections with F o > 4σ(F o) and 84 refined parameters. In this model, the total number of electrons refined at the cation sites is 1334.5, and those of anion positions is 639.6 electrons.
Details of data collection and refinement are given in Table 3. Fractional atomic coordinates and equivalent isotropic displacement parameters are reported in Table 4. Table 5 reports selected bond distances, whereas bond-valence sums (BVS), calculated according to Brese and O’Keeffe (Reference Brese and O’Keeffe1991), are shown in Table 6. Anisotropic displacement parameters are reported in the crystallographic information file (CIF) that has been deposited with the Principal Editor of Mineralogical Magazine and is available as Supplementary material.
Summary of data collection conditions and refinement parameters for selenodantopaite

* w = 1/[σ2(Fo2) +0.0534P 2 + 56.1525P], where P = (Fo2 + 2F c2)/3.
Site, Wyckoff position, site occupancy, fractional atom coordinates, and isotropic (*) or equivalent isotropic displacement parameters (Å2) for selenodantopaite

Selected bond distances (in Å) for selenodantopaite

Weighted bond valences* (in valence units) for selenodantopaite

* Obtained using the site occupancies from structural refinement.
Powder X-ray diffraction data could not be collected, due to the paucity of available material. Consequently, powder X-ray diffraction data, given in Table 7, were calculated using the software PowderCell 2.3 (Kraus and Nolze, Reference Kraus and Nolze1996) on the basis of the structural model given in Tables 3 and 4.
Calculated powder X-ray diffraction data (d in Å) for selenodantopaite*

* Intensity and d calc (in Å) were calculated using the software PowderCell2.3 (Kraus and Nolze, Reference Kraus and Nolze1996) on the basis of the structural data given in Tables 3 and 4. Only reflections with I rel. ≥ 5 are listed. The five strongest reflections are given in bold.
Crystal structure of selenodantopaite
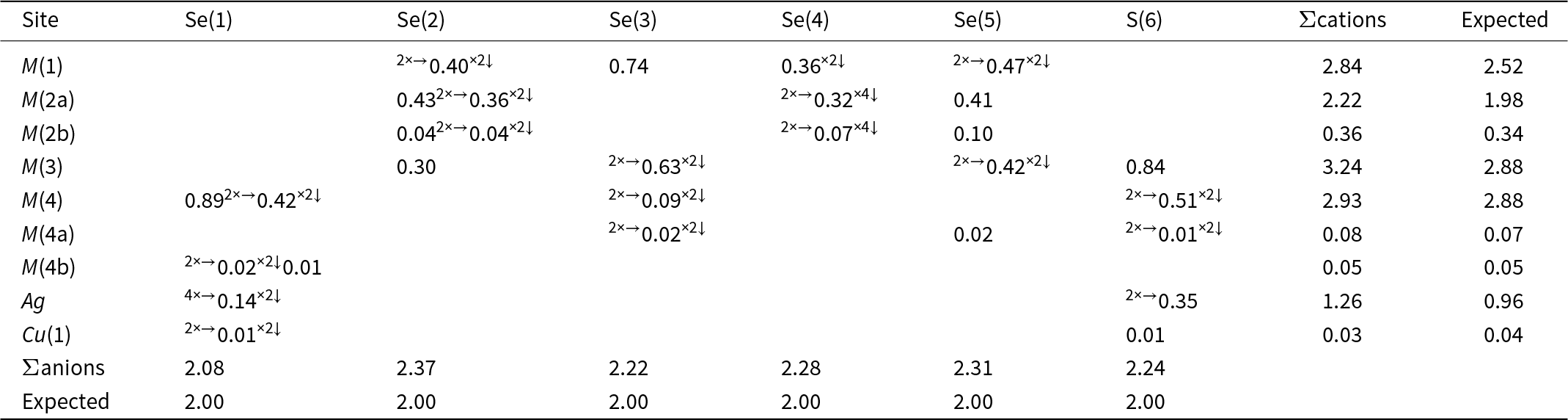
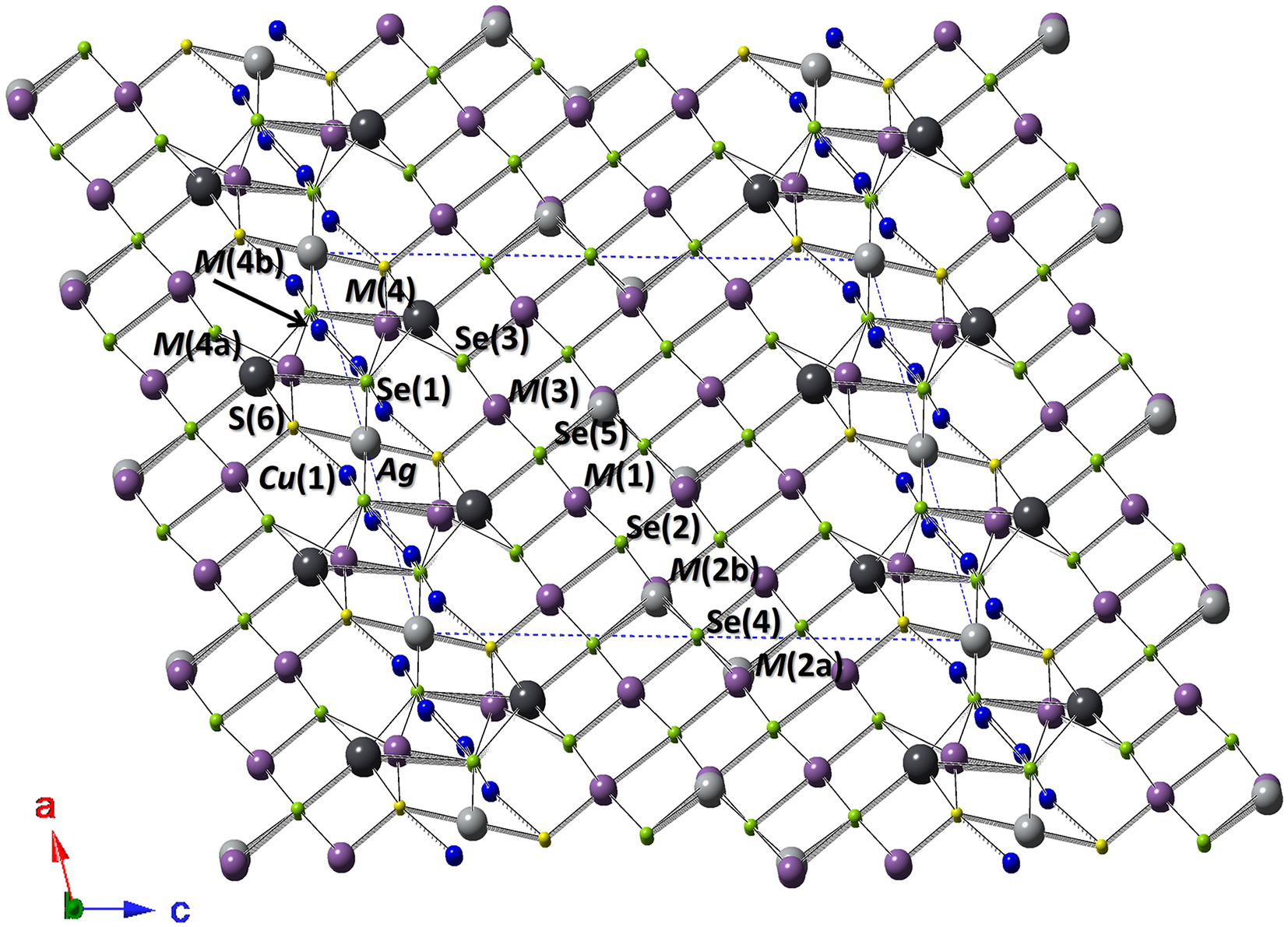
Ignoring low-occupancy split positions, the crystal structure of selenodantopaite is isotypic with that of dantopaite (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010) and it is composed by two kinds of slabs, parallel to (001), i.e., a PbS-like thick slab and a thin slab (Fig. 3), following the classical structural scheme of pavonite homologues (e.g. Makovicky et al., Reference Makovicky, Mumme and Watts1977).
Crystal structure of selenodantopaite as seen down b. Violet, grey, dark grey, and blue circles represent Bi, Ag, Pb and Cu-hosting sites, whereas yellow and green circles show S- and Se-dominant positions. Drawn with CrystalMaker (Palmer, Reference Palmer2014).

The thicker slab is formed by (Bi,Ag)-centred octahedra, namely the M(1)–M(3) sites. In agreement with the homologue order number N = 6, there are six octahedra in a diagonal row across the thicker slab, according to the sequence M(3)–M(1)–M(2)–M(2)–M(1)–M(3).
The M(1) site is a mixed (Bi,Ag) position, with a Bi/(Bi+Ag) ratio of 0.76. In type dantopaite, this site has a pure Bi occupancy (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010). The site is characterized by a short M(1)–Se(3) bond of 2.763 Å, with Se(3) located on the interslab surface; this arrangement is similar to the short M(1)–S(3) bond of 2.664 Å observed in dantopaite, the lengthening of the bond being due to the replacement of S by Se. The coordination of M(1) is completed by five additional bonds ranging between 2.920 and 2.994 Å. The average <M(1)–Se> distance is 2.919 Å, longer than that observed in dantopaite (2.832 Å) and agreeing with the replacement of S by the larger anion Se2-.
The M(2) site is located at the centre of the thick PbS-like slab. This site was split in two sub-positions, in agreement with the structural model of dantopaite (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010). In the sample studied by these authors, the ratio of the two sub-sites is 82:18. In selenodantopaite such a ratio is 66:34. The average <M(2a)–Se> and <M(2b)–Se> distances are 2.910 and 2.92 Å, respectively, longer than the values observed in dantopaite (∼2.85 Å). According to Makovicky et al. (Reference Makovicky, Paar, Putz and Zagler2010), the M(2) site could host Pb.
The M(3) site is located on the margins of the PbS-like slab and it shows a Bi/(Bi+Ag) ratio of 0.94. As for M(1), it shows a short M(3)–S(6) distance of 2.663 Å. Also in this case, the anion is located on the interslab surface. The observed distance can be compared with 2.628 Å given by Makovicky et al. (Reference Makovicky, Paar, Putz and Zagler2010) for dantopaite; the increase in the bond length is probably due to the partial Se-to-S substitution at this anion site, that is the only S-dominant position in the crystal structure of selenodantopaite. The M(3) has two additional relatively short bonds (2.872 Å, longer than those reported by Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010, i.e. 2.779 Å), two bonds at 3.013 Å and a long bond at 3.124 Å. The average <M(3)–ϕ> (where ϕ = Se, S) distance is 2.926 Å, longer than that observed in dantopaite (2.857 Å – Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010).
The thinner slab contains single columns of edge-sharing Ag-centred octahedra (Ag site), flanked by double columns of Bi-centred square pyramids [M(4) site].
The Ag site has a 2+4 coordination, with two linearly arranged Ag–S(6) bonds, with distance of 2.565 Å and a S(6)–Ag–S(6) angle of 180.0(3)°. Four additional bonds at 2.929 Å complete the bonding environment of this position. The site occupancy points to a partially occupied Ag position (Ag0.96). The average bond distance, 2.810 Å, is longer than that in the corresponding Ag1 site of the type sample of dantopaite, 2.764 Å. The Ag-centred octahedron is flanked by a partially occupied (Cu,Ag)-centred tetrahedra, namely Cu(1). Such a configuration is similar (but not identical) to those observed in makovickyite (Topa et al., Reference Topa, Makovicky and Balić-Žunić2008). The Cu(1) site has a quasi-planar triangular coordination, with two bonds at 2.36 Å with Se(1) and a longer bond at 2.50 Å with S(6). This site is too close to Ag [at ∼1.3 Å] whereas it is at ∼2.5 Å from a symmetry-related Cu(1) site, similar to the Cu–Cu distance in metallic Cu (e.g. 2.56 Å – Otte, Reference H.M1961).
The M(4) site has a square pyramidal coordination (average distance 2.861 Å), with two additional longer bonds at 3.578 Å. This site was refined as a virtually pure Bi position, with only a small amount of vacancy related to the presence of subsidiary low-occupied sites, i.e. M(4a) and M(4b). The M(4a) site was interpreted as a Pb-bearing position. This site is on the same row of polyhedra forming the thick slab and for this reason Makovicky et al. (Reference Makovicky, Paar, Putz and Zagler2010) interpreted it as due to the possible presence of minor 7P lamellae in 6P dantopaite. Moreover, it was usually assumed that Pb replaces Bi in the thick slab (e.g. Makovicky et al., Reference Makovicky, Mumme and Watts1977; Makovicky and Mumme, Reference Makovicky and Mumme1979). However, such a kind of Pb-to-Bi replacement in the thin slab is known, e.g. in kudriavite (Balić-Žunić and Makovicky, Reference Balić-Žunić and Makovicky2007). In cupromakovickyite, cupromakopavonite and cupropavonite, Pb occurs in a site of the thin slab (Topa et al., Reference Topa, Makovicky and Balić-Žunić2008; Topa et al., Reference Topa, Makovicky, Ilinca and Dittrich2012; Mumme et al., Reference Mumme, Gable and Wilson2015). In these latter ‘cupro’ phases, a doubling of the c parameter was observed; such a doubling was not detected in selenodantopaite. The M(4a) site is at 1.18 Å from the M(4) site, and consequently their occupancy is mutually exclusive. The M(4b) site was refined as a partially occupied Cu-bearing site, although it could host both Cu and Ag. It shows a planar triangular coordination, with two relatively short bonds of 2.17 Å and a longer one at 2.47 Å. This sub-site is at 1.73 Å from the M(4) site and consequently these two positions are mutually exclusive. Similarly, M(4b) is at only 1.8 Å from the Cu(1) site. M(4b) is at 2.71 Å from M(4a). This distance could be relatively short for a Pb–Cu contact; considering the experimental uncertainty (0.15 Å), it could be considered possible. Other relatively short (but probably physically possible) distances are M(4b)–Ag (2.67 Å) and M(4b)–M(4b) (2.77 Å). The possible occupancy of the M(4a) and M(4b) sites could give rise to the substitution mechanism M (4a)Pb2+ + M (4b)Cu+ = M (4)Bi3+ + M (4b)□. This could be another of the mechanisms favouring the accumulation of metal in the thin slab, allowing the metal excess observed in selenodantopaite.
Bond-valence sums at the cation positions are usually higher than those expected on the basis of the site occupancies. In particular, this could indicate the occurrence of admixed smaller cations (i.e. Ag) at the M(1) and M(3) site. Moreover, the actual location of minor Pb, having a similar scattering factor with Bi, is problematic and no clear indication of the positions occupied by Pb can be obtained.
Among the six independent anion sites, five are dominated by Se, i.e. Se(1)–Se(5). The only S-dominant site is S(6). The bond-valence sums at these sites range between 2.08 and 2.37 valence units. Such an overbonding could have several reasons: (1) difficulties in the correct assignments of Ag at the M(1)–M(3) sites; (2) poor accuracy of the bond parameters involving Se; and (3) increasing covalency of Me–Se bonds with respect to Me–S bonds.
The mineral formula obtained through single-crystal X-ray diffraction is t[Cu0.324Ag1.93Pb0.136Bi3.848(S4.44Se3.56)Σ8.00] T[Ag2.56Bi9.44(Se13.92S2.08)Σ14.00], where superscript t and T indicate thin and thick slabs, respectively. It is worth noting that the t and T slabs have Se/(S+Se) ratios of 0.445 and 0.994, respectively, indicating the preferential partitioning of Se in the PbS-like thick slab, whereas the thin slab preferentially host S. The sum of these two t and T structural modules gives the formula Cu0.324Ag4.49Pb0.136Bi13.288(Se15.98S6.02)Σ22.00 (Z = 1) that can be compared with that obtained through quantitative chemical analysis, i.e. Cu0.24(4)Ag5.09(7)Fe0.17(5)Pb0.51(4)Bi12.32(21)Se15.11(21)S6.89(21) and Cu0.05(3)Ag5.23(11)Fe0.06(4)Pb0.62(12)Bi12.38(13)Se14.77(16)S7.23(16) for the Cu-bearing bearing and Cu-poor varieties. As in dantopaite, there is an overestimation of heavy cations (Bi) with respect to Ag (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010).
Discussion
Shortcomings of the crystal structure refinement
The crystal structure of selenodantopaite is physically sound, but some shortcomings occur owing to the general low quality of the diffraction data, as testified by the high R int value, i.e. 0.1315, and the relatively high estimated standard deviations for some low-occupancy sites. According to Makovicky et al. (Reference Makovicky, Paar, Putz and Zagler2010), the high R int observed in dantopaite (0.1187) could be due to the intergrowths of two or more sulfosalts in every extracted grain, as for instance the occurrence of 7P lamellae in 6P dantopaite.
During the study of selenodantopaite we extracted two different grains. The first one was very small and intergrown with other not-identified phases and yielded an R int of 0.1643, allowing the solution and refinement of the crystal structure to R 1 = 0.0883 for 768 observed reflections and 72 least-square parameters. A larger second grain gave a sharper X-ray diffraction pattern and was free from any visible additional phase. This is the grain used for the data collection used for the refinement of the crystal structure here described. The occurrence of the residual maxima close to M(4) could be due to the intergrowth of 7P lamellae in selenodantopaite (as proposed by Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010); moreover, the combined Bi + Pb configuration in the thin slab is known in the cupropavonite series (e.g. Topa et al., Reference Topa, Makovicky and Balić-Žunić2008) and it cannot be excluded that the observed residuals could also be related to the intergrowth of lamellae of ‘cuproselenodantopaite’ (or ‘argentoselenodantopaite’, with Ag playing the same role of Cu as in dantopaite – see below). However, the proposed structural model of selenodantopaite shows these sub-positions, proposing an interpretation of their role in the crystal chemistry of this mineral and allowing the possible occurrence of a metal excess.
In our opinion, the shortcomings observed in the crystal structure refinement of selenodantopaite are similar to those encountered by previous authors dealing with pavonite and cupropavonite homologues (e.g. Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010) and do not prejudge the definition of selenodantopaite as the first selenide member of the pavonite homologous series.
Calculation of homologue order
The empirical formulae of the two varieties of selenodantopaite are Cu0.24(4)Ag5.09(7)Fe0.17(5)Pb0.51(4)Bi12.32(21)Se15.11(21)S6.89(21) and Cu0.05(3)Ag5.23(11)Fe0.06(4)Pb0.62(12)Bi12.38(13)Se14.77(16)S7.23(16). In both cases, a small excess of cations was observed, i.e. 0.33 and 0.34 apfu, respectively.
Karup-Møller and Makovicky (Reference Karup-Møller and Makovicky1979) distinguished two different kinds of Cu: (1) substitutional sCu, that is Cu fitting the ideal formula of pavonite homologues, and (2) interstitial iCu, corresponding to excess Cu. According to these authors, the calculation of the order number N of the pavonite homologue can be obtained normalizing the atomic proportions according to the relation Pb + Bi + Ag + sCu = 1 and then applying the equation N = [7 – 6Bi – 3(Pb + sCu)]/[2Bi + (Pb + sCu) – 1]. However, as pointed out by Karup-Møller and Makovicky (Reference Karup-Møller and Makovicky1979), it is not possible to distribute Cu a priori between ‘substitutional’ and ‘interstitial’. Consequently, they proposed to calculate three possible N numbers, N P1 (all Cu taken as iCu), N P2 (all Cu considered as sCu) and N P3 (Cu is equally distributed between substitutional and interstitial). Following Topa and Paar (Reference Topa and Paar2008), minor Fe was considered to replace for Pb. The three calculations lead to the meaningless values N P1 = 7.01, N P2 = 7.14 and N P3 = 7.07 for Cu-bearing selenodantopaite and N P1 = 7.23, N P2 = 7.26 and N P3 = 7.25 for the Cu-poor variety; indeed, the crystal structure solution clearly indicates that N P = 6. This discrepancy is due the fact in the calculation of the N value it is always assumed that Ag+ + Bi3+ = 2Pb2+ (e.g. Makovicky, Reference Makovicky2019). On the contrary, in selenodantopaite Ag seems to also play the role of an interstitial cation. This unusual role of Ag could be favoured by the unit-cell volume expansion of selenodantopaite (V ≈ 1047 Å3) with respect to dantopaite (V ≈ 976 Å3), ΔV = +7.3%. This volume increase may justify the insertion of larger Ag polyhedra which replace the Cu polyhedra in the same crystal-chemical role, as observed, for instance, in the two pavonite-related minerals watkinsonite, Cu2PbBi4Se8, and litochlebite, Ag2PbBi4Se8 (Topa et al., Reference Topa, Makovicky, Sejkora and Dittrich2010; Sejkora et al., Reference Sejkora, Makovicky, Topa, Putz, Zagler and Plášil2011).
Thus, in selenodantopaite, the two following substitution mechanisms involving cations take place:
(1) i(Ag,Cu)+ + (Pb,Fe)2+ = □ + Bi3+,
(2) 2Pb2+ = Ag+ + Bi3+,
as well as the replacement S2– = Se2–.
Applying these substitutions, one can obtain the formulae Ag5.17Bi12.83Se22 and Ag5.11Bi12.89Se22, close to the ideal one Ag5Bi13Se22.
Nomenclature issues
Following the results of the crystal structure analysis of selenodantopaite, its ‘substitution-free’ formula could be written as Ag5Bi13Se18S4 (Z = 1). Recently, we examined samples of Se-bearing dantopaite from the Mohawk mine, Nevada, USA, corresponding to the formula Ag5Bi13S12Se10, having three anion sites dominated by S and three by Se (Musetti et al., Reference Musetti, Sejkora, Biagioni and Dolníček2024). In both cases, Se is preferentially partitioned in the PbS-like thick slab, but whereas Se-bearing dantopaite has a Se/(S+Se) ratio of 0.135 in the thin slab, in selenodantopaite such a ratio is increased up to 0.445, with Se(1) being dominated by Se and S(6) showing the occupancy (S0.67Se0.33). The existence of Ag5Bi13Se22 is only possible if we admit that complete Se-to-S replacement can also occur in the thin slab.
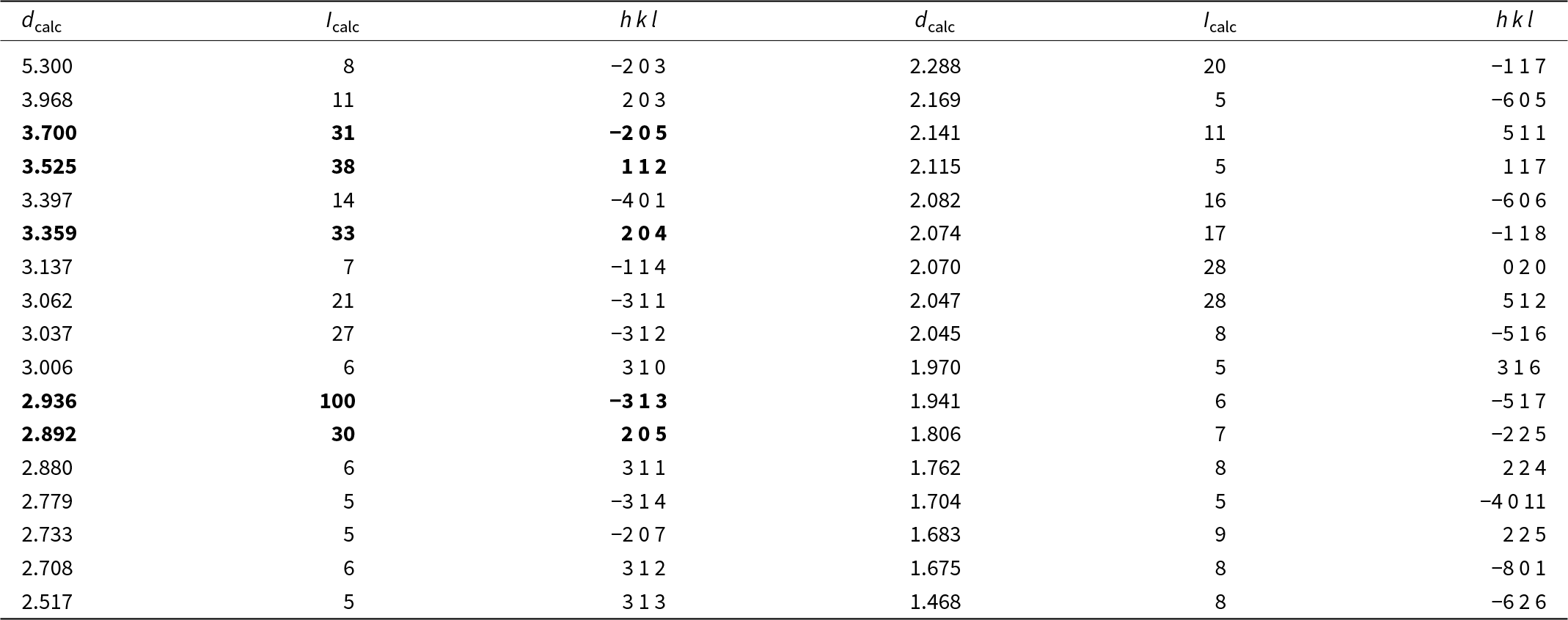
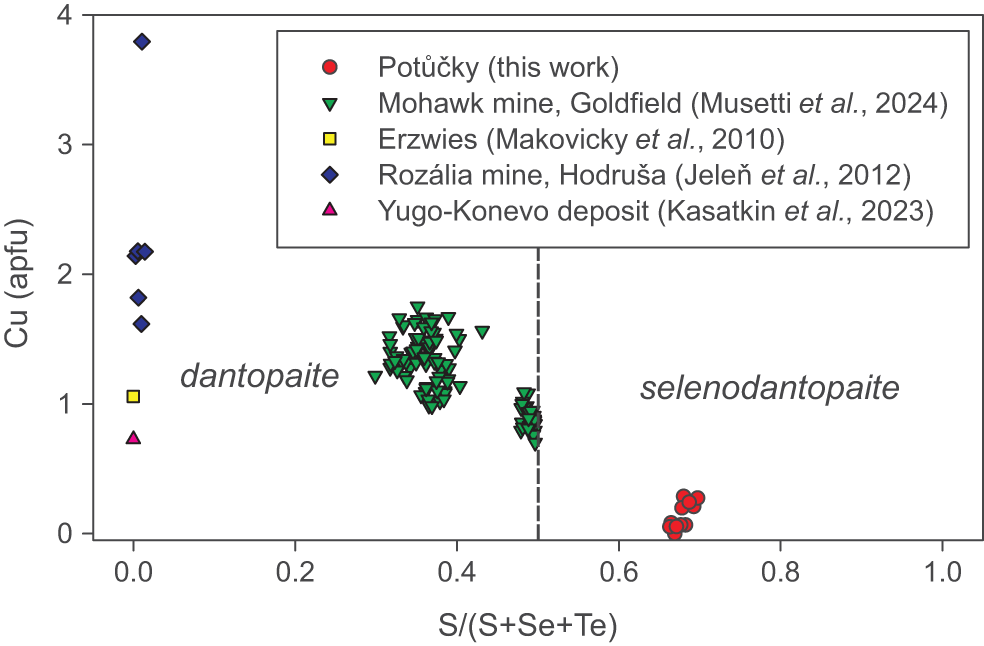
For the sake of simplicity, we think that, for nomenclature purposes, S(1)–S(6) sites should be considered as an aggregate site, avoiding the creation of different isotypes of dantopaite based on different S/Se ratios. On the contrary, species with S > Se should be classified as dantopaite, and those with Se > S could be described as selenodantopaite (Fig. 4).
Chemical composition of members of the dantopaite–selenodantopaite solid solution in the plot S/(S+Se+Te) vs. Cu (apfu).

Conclusions
Selenodantopaite belongs to the pavonite homologous series (Table 8). It is the first Se-dominant member of this series and represents the Se-analogue of the 6P homologue dantopaite, Ag5Bi13S22 (Makovicky et al., Reference Makovicky, Paar, Putz and Zagler2010). Selenodantopaite does not correspond to any valid or invalid unnamed mineral (Smith and Nickel, Reference Smith and Nickel2007) and their updated lists (http://cnmnc.units.it/). It is identical with unnamed “Se-analogue of pavonite” from the Princ Evžen deposit, Potůčky (Sejkora et al., Reference Sejkora, Šrein, Šreinová and Dolníček2017). This species was also hypothetically reported by Kolitsch et al. (Reference Kolitsch, Bayerl and Topa2019) from the Clara mine, Germany. It may be also partly related to UM1989-28-Se:AgBiCuS from the Ozernovskoe deposit, Kamchatka, Russia (Kovalenker et al., Reference Kovalenker, Nekrasov, Sandomirskaya, Nekrasova, Malov, Danchenko and Dmitriyeva1989; Kovalenker and Plotinskaya, Reference Kovalenker and O.Yu2005).
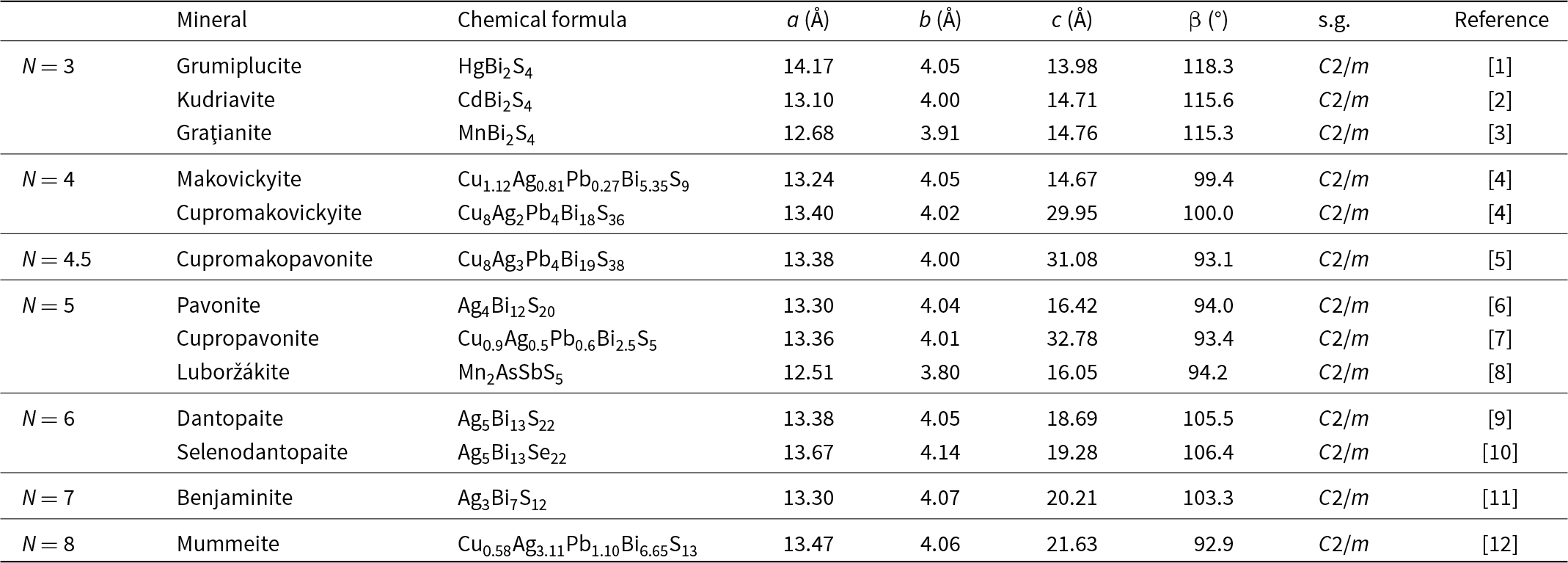
Members of the pavonite and cupropavonite homologous series

N = homologue order; s.g. = space group. [1] Merlino et al. (Reference Merlino, Biagioni and Orlandi2013); [2] Balić-Žunić and Makovicky (Reference Balić-Žunić and Makovicky2007); [3] Ciobanu et al. (Reference Ciobanu, Brugger, Cook, Mills, Elliott, Damian and Damian2014) [4] Topa et al. (Reference Topa, Makovicky and Balić-Žunić2008); [5] Topa et al. (Reference Topa, Makovicky, Ilinca and Dittrich2012); [6] Makovicky et al. (Reference Makovicky, Mumme and Watts1977); [7] Mumme et al. (Reference Mumme, Gable and Wilson2015); [8] Kasatkin et al. (Reference Kasatkin, Makovicky, Plášil, Škoda, Agakhanov, Stepanov and R.S2020); [9] Makovicky et al. (Reference Makovicky, Paar, Putz and Zagler2010); [10] this work; [11] Makovicky and Mumme (Reference Makovicky and Mumme1979); [12] Karup-Møller and Makovicky (Reference Karup-Møller and Makovicky1992).
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/mgm.2025.10111.
Acknowledgements
Special thanks go to Vladimír Šrein, who died suddenly in January 2025; he found the first sample of selenodantopaite and was a coauthor of its original proposal. The helpful comments of an anonymous reviewer, Peter Leverett, and Principal Editor Stuart Mills are greatly appreciated. The study was financially supported by the Ministry of Culture of the Czech Republic (long-term project DKRVO 2024-2028/1.II.c; National Museum, 00023272) for JS. The Centro per l’Integrazione della Strumentazione scientifica dell’Università di Pisa (C.I.S.U.P.) is thanked for the access to the single-crystal X-ray diffraction laboratory.
Competing interests
The authors declare none.