Introduction
Clostridioides difficile infection (CDI) disproportionately harms hematology-oncology and hematopoietic cell transplant (HO/HCT) patients due to their immunosuppressed status, prolonged healthcare exposure, and receipt of antimicrobials. Reference Larrainzar-Coghen, Rodríguez-Pardo and Barba1 Admission screening for asymptomatic C. difficile colonization is a potential strategy to reduce rates of hospital-onset C. difficile infection (HO-CDI) because colonized patients, even if asymptomatic, can transmit C. difficile to others. Reference Sheth, Douchant and Uyanwune2 To reduce nosocomial transmission of C. difficile in our HO/HCT unit, we implemented C. difficile active surveillance for HO/HCT patients at time of admission, placing patients testing positive in isolation precautions. This study assesses the impact of C. difficile admission screening on unit-level HO-CDI rates and aims to identify possible unintended impacts of this strategy.
Methods
Rush University Medical Center (Chicago, IL) is a 678-bed hospital with a 32-bed HO/HCT unit. During the baseline period (May 1, 2020 to May 31, 2022), symptomatic patients with diarrhea underwent C. difficile testing of unformed stool by polymerase chain reaction (PCR) [Xpert® C. difficile/Epi; Cepheid, Sunnyvale, CA]; asymptomatic patients were not tested. During the intervention period (July 1, 2022 to July 31, 2024), C. difficile testing criteria for symptomatic patients remained unchanged, Reference Lin, Stein and Kothadia3 but asymptomatic patients underwent C. difficile colonization screening by PCR [Xpert® C. difficile] via perirectal swab at time of admission (testing decision flowchart, Supplemental Figure 1; test results with embedded provider education, Supplemental Figure 2). Patients transferred to the HO/HCT unit from other hospital units were not screened. Patients testing positive for C. difficile, whether asymptomatic or symptomatic, were placed in Contact Precautions with hand washing required at time of room exit. Prophylaxis (eg, oral vancomycin) to prevent CDI was not routinely recommended for asymptomatic patients. No other unit-level interventions for CDI prevention were introduced during the study period.
The primary outcome was unit-level HO-CDI incidence (CDI events per 10,000 patient-days); community-onset CDI (CO-CDI) was a secondary outcome. Both were defined as LabID events using criteria from the CDC’s National Healthcare Safety Network (NHSN). 4 Under NHSN rules, tests of asymptomatic patients are not considered LabID events; a patient who has a positive asymptomatic C. difficile test upon hospital admission remains eligible for a subsequent CO-CDI or HO-CDI event.
To assess unintended impacts of C. difficile screening, we analyzed unit-level enteral vancomycin days of therapy per 1,000 days present as a proxy for CDI treatment or prophylaxis and vancomycin-resistant Enterococcus (VRE) LabID events per 10,000 patient-days. 4
For all analyses involving monthly rate data, we used interrupted time series models adjusted for repeated measures to evaluate differences in intercept and slope between the baseline and intervention periods. Analytic models were appropriate to count data (ie, Poisson or negative binomial) and included offset variables to account for monthly variation in patient census.
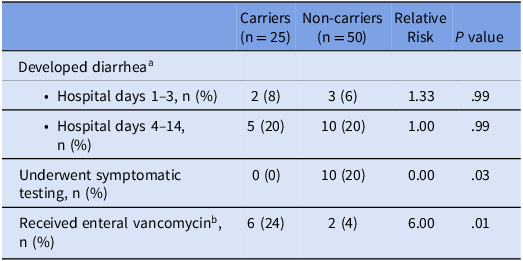
To assess patient-level outcomes, we created a retrospective cohort that included every asymptomatic C. difficile carrier (screen-positive) identified during the first year of intervention, plus non-carriers (screen-negative) selected in a 1:2 ratio based on hospital length of stay and admission date. Between carriers and non-carriers, we compared the following outcomes within the first fourteen days of hospitalization by calculating the relative risk using Fisher’s exact test: clinically significant diarrhea (ie, three or more unformed stools documented per day), symptomatic C. difficile testing, and receipt of enteral vancomycin therapy.
Statistical analyses were performed using SAS 9.4 software (SAS Institute, Cary, NC). This study was reviewed by the Rush University institutional review board and considered exempt, with informed consent waived.
Results
During the study period, there were 4,205 patient admissions to the HO/HCT unit (2,121 during baseline and 2,084 during intervention). During the baseline period, there were 525 symptomatic C. difficile PCR tests performed, of which 85 (16.2%) were positive. During the intervention period, 519 symptomatic and 1,286 asymptomatic C. difficile PCR tests were performed, of which 55 (10.6%) and 66 (5.1%) were positive, respectively.
The intervention period was associated with a non-significant decline in adjusted HO-CDI incidence compared to baseline period (incident rate ratio [IRR] 0.45, P = .10, Figure 1a). CO-CDI incidence was significantly lower in the intervention period, compared to baseline (IRR 0.15, P = .049, Figure 1b). The IRRs for enteral vancomycin days of therapy and VRE events were 1.01 (P = .97) and 3.70 (P = .11), respectively.
Monthly hospital-onset [a] and community-onset [b] Clostridioides difficile infection (CDI) incidence per 10,000 patient-days. Baseline: symptomatic CDI testing only. Intervention: symptomatic CDI + admission C. difficile testing. Incidence rate ratio (IRR) refers to changes in intercept between baseline and intervention modeled rates. There were no significant changes in slope for either model presented.

In a cohort of patients stratified by admission asymptomatic screening result, C. difficile carriers were equally likely to develop diarrhea as non-carriers; however, carriers were less likely to receive C. difficile symptomatic testing and more likely to receive enteral vancomycin (Table 1).
Outcomes during hospitalization by Clostridioides difficile carriage status on admission, retrospective cohort

Note: aDocumentation of ≥3 unformed stools in 1 day.
bEnteral vancomycin could be administered for either the indication of prophylaxis or treatment.
Fidaxomicin was available as a restricted antibiotic for C. difficile infection treatment, requiring infectious diseases approval; no patient in this cohort analysis received fidaxomicin.
Discussion
Admission screening for asymptomatic C. difficile colonization among patients admitted to the HO/HCT unit was associated with a non-significant decrease in HO-CDI rate and a significant decrease in CO-CDI rate. During the intervention period, screen-positive patients (carriers) developed diarrhea at comparable rates to screen-negative patients (non-carriers). However, carriers were less likely to be tested for symptomatic CDI during hospitalization and more likely to receive enteral vancomycin therapy (as prophylaxis or treatment) than their non-carrier counterparts.
Several prior studies have reported a decrease in HO-CDI rates associated with asymptomatic admission screening. Cho et al. found an 80% decrease in the HO-CDI rate on an HCT unit after implementation of stool-based C. difficile admission screening and isolation of carriers. Reference Cho, Seville, Khanna, Pardi, Sampathkumar and Kashyap5 Collison et al. reported a 62% decrease in LabID events with hospitalwide admission C. difficile screening of inpatients, including patients admitted to the oncology unit. Reference Collison, Murillo and Marrs6
Although C. difficile admission screening may reduce HO-CDI rates through isolation precautions and judicious use of CDI-associated medications in screen-positive patients, Reference Gilboa, Meltzer and Barda7 our findings indicate it may also do so by altering provider testing practices. If asymptomatic screening primarily reduced patient-to-patient C. difficile transmission, we would expect a greater decrease in HO-CDI rates (reflecting nosocomial transmission) than in CO-CDI rates (which reflect acquisition prior to admission). Instead, we found that admission screening had a greater impact on reducing CO-CDI incidence than on HO-CDI incidence. Furthermore, we found that C. difficile carriers were much less likely to undergo CDI testing than non-carriers during hospital days 1−3 and 4−14 (eligibility periods for CO-CDI and HO-CDI, respectively), even though rates of clinically significant diarrhea were similar in both groups. Collectively, these findings indicate that both CO-CDI and HO-CDI metrics—LabID events defined exclusively by a positive C. difficile test result—are susceptible to variation in provider testing practices influenced by admission screening outcomes.
Although we detected a trend toward higher VRE incidence during the intervention period, we did not detect any increase in unit-level enteral vancomycin use and only a minority of confirmed carriers received enteral vancomycin in our cohort analysis.
Our study has several limitations. First, patients who were admitted to a non-HO/HCT unit (eg, intensive care) and later transferred to the HO/HCT unit were ineligible for screening. Second, perirectal swabbing may be less sensitive than stool-based testing for C. difficile detection. Reference Lee, Reske and Hink8 For both limitations, false-negative categorization of C. difficile colonized patients at time of admission could bias the intervention effect towards the null. Lastly, our study may not be generalizable to facilities that incorporate routine enzyme immunoassay C. difficile toxin testing.
In summary, admission screening for asymptomatic C. difficile colonization in HO/HCT patients influenced both HO-CDI and CO-CDI rates, partly by shaping subsequent provider testing decisions. These findings highlight that laboratory-defined quality metrics can be impacted by admission screening through both intended and unintended effects.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/ice.2026.10442.
Acknowledgements
None.
Financial support
This project was supported in part by cooperative agreement 6U54CK000607 from Centers for Disease Control and Prevention (CDC). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of CDC.
Competing interests
The authors report no conflicts of interest or competing interests.