Introduction
Lithium remains a first-line treatment for acute mania and for preventing relapse in bipolar disorder.Reference McIntyre, Berk and Brietzke 1 , Reference Yatham, Chakrabarty and Bond 2 However, long-term use carries a significant risk of kidney injury, with lithium-induced nephrotoxicity estimated to affect up to 20% of patients receiving chronic therapy.Reference Qian, Sud and Lee 3 –Reference Davis, Desmond and Berk 5 This complication is often progressive with continued lithium exposure and a not infrequent reason for treatment discontinuation. Pathology results typically show chronic tubulointerstitial nephropathy with evidence of tubular atrophy, interstitial nephritis and fibrosis as well as cortical microcysts.Reference Gong, Wang and Dworkin 6 , Reference Markowitz, Radhakrishnan, Kambham, Valeri, Hines and D’Agati 7 The mechanisms implicated are hypothesized to include inflammatory and oxidative stress pathways.Reference Ossani, Uceda and Acosta 8 Although treatments exist for lithium-induced nephrogenic diabetes insipidus, no therapy has yet been established to meaningfully prevent or slow the disease trajectory of lithium-induced nephrotoxicity.
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are FDA approved in the treatment of multiple medical conditions including, but not limited to, type 2 diabetes mellitus and obesity with acute- and long-term effects that have exceeded traditional agents. In addition to their metabolic effects, GLP-1 RAs have received U.S. Food and Drug Administration (FDA) approval for metabolic-dysfunction-associated steatohepatitis, obstructive sleep apnea, and reducing the progression of chronic kidney disease (CKD), end-stage kidney disease, and cardiovascular mortality in adults with type 2 diabetes and CKD.Reference Drucker 9 Mechanisms implicated in the renal protective effects of GLP-1 RAs include suppression of activated inflammatory effectors; decreased renal immune cell infiltration; regulation of innate immune activity; decreased receptor expression for advanced glycation end product (RAGE); inhibition of nuclear factor κB (NF-κB) signaling; and reductions in fibrosis, oxidative stress, and autophagy. Beneficial effects in microvasculature function and neural signaling are also implicated.Reference J, Me and Mt 10
The nephroprotective mechanisms of GLP-1 RAs introduce a testable and viable hypothesis that we preliminarily address in this study, ie, whether semaglutide, a GLP-1 RA currently approved by the FDA for reducing the risk of worsening kidney disease and kidney failure in adults with type 2 diabetes, may also exert protective and/or illness-modifying effects against lithium-induced nephrotoxicity. The aforementioned hypothesis derives from overlapping molecular mechanisms implicated in diabetic nephropathy and lithium-induced nephrotoxicity. A separate and related hypothesis is whether protective effects may be direct cytoprotective actions on molecular and cellular renal systems and/or indirectly by improvements in microvascular and metabolic function.Reference J, Me and Mt 10
Methods
Data source and extraction
Adverse event (AE) data were systematically retrieved from the U.S. FDA Adverse Event Reporting System (FAERS), a global post-marketing surveillance database that aggregates voluntary reports from healthcare professionals, manufacturers, and consumers. Reports submitted between December 2003 and December 2024 were analyzed using OpenVigil 2.1, a validated pharmacovigilance platform that interfaces with the openFDA API to provide structured access to FAERS. 11 –Reference Dores, Bryant-Genevier and Perez-Vilar 20 This system enables standardized data retrieval, filtering, deduplication, and preprocessing of large-scale safety datasets in alignment with international pharmacovigilance guidelines.
Data cleaning and curation
Drug names were standardized according to the United States Adopted Name (USAN) convention, with cross-references to DrugBank and Drugs@FDA to resolve brand and generic discrepancies as well as abbreviations. Automated mapping and error-correction algorithms were used to harmonize terminology across entries. Reports that were incomplete, structurally invalid, or internally inconsistent were excluded. Potential duplicate cases were identified through matching of demographic and case-level variables and verified against the FDA DEMO table before removal.
AE coding and case selection
All AEs were standardized using Preferred Terms (PTs) from the Medical Dictionary for Regulatory Activities (MedDRA) to ensure consistent classification of renal outcomes. 21 Primary endpoints included the MedDRA terms renal impairment, renal failure, CKD, end-stage renal disease (ESRD), and acute kidney injury (AKI), selected for their documented association with lithium-induced nephrotoxicity. The final dataset underwent clinical review by experts from the FDA’s Center for Drug Evaluation and Research (CDER) and Center for Biologics Evaluation and Research (CBER) to confirm completeness, clinical plausibility, and coding accuracy. 22 , 23
Analyses were limited to reports identifying lithium or semaglutide as the primary suspect drug to ensure accurate attribution of AEs to the drug of interest. In contrast, co-reported cases involving both lithium and semaglutide were queried without suspect filters to maximize sensitivity for detecting potential interaction signals, regardless of reporter designation.
Signal detection and statistical analysis
Disproportionality analysis was performed using the reporting odds ratio (ROR), defined as the odds of a renal AE event occurring with a given drug relative to all other drugs in the FAERS database. To strengthen signal detection accuracy, a multitiered analytic framework was applied, consistent with validated pharmacovigilance methodologies from prior studies.Reference Lakhani, Kwan, Mihalache, Popovic, Hurley and Muni 13 , Reference Lakhani, Kwan, Nguyen, Popovic, McIntyre and Wu 17 Reference McIntyre, Mansur, Rosenblat and Kwan 24 , Reference Kwan, Lakhani and Teopiz 25Specifically, the Evans criteria (n > 2, χ2 > 4, ROR > 2) were used alongside calculation of the lower bound of the 95% credibility interval (IC025).Reference Evans, Waller and Davis 26 A signal was considered statistically significant when IC025 > 0 and p < 0.05. In addition to ROR, proportional reporting ratio (PRR) and relative risk ratio (RRR) were calculated as secondary validation metrics.
To minimize reporting biases, we combined frequentist and Bayesian disproportionality methods with standardized signal thresholds to enhance the accuracy and stability of signal detection. The Bayesian IC025 metric helps reduce the impact of notoriety bias—where increased awareness temporarily boosts reporting—and the Weber effect, which reflects early post-marketing spikes in AE submissions. This stabilization occurs through Bayesian shrinkage, which adjusts observed-to-expected ratios toward the overall reporting mean, reducing the influence of random variation, small sample sizes, and short-term reporting fluctuations.Reference Goligher, Heath and Harhay 27 , Reference Röver and Friede 28 Visualization of RORs, including forest plots, was performed using RStudio (version 2023.06.1 + 524, “Desert Sunflower” release).
Ethical considerations
Institutional review board (IRB) approval was not required because FAERS is a publicly accessible de-identified database.
Results
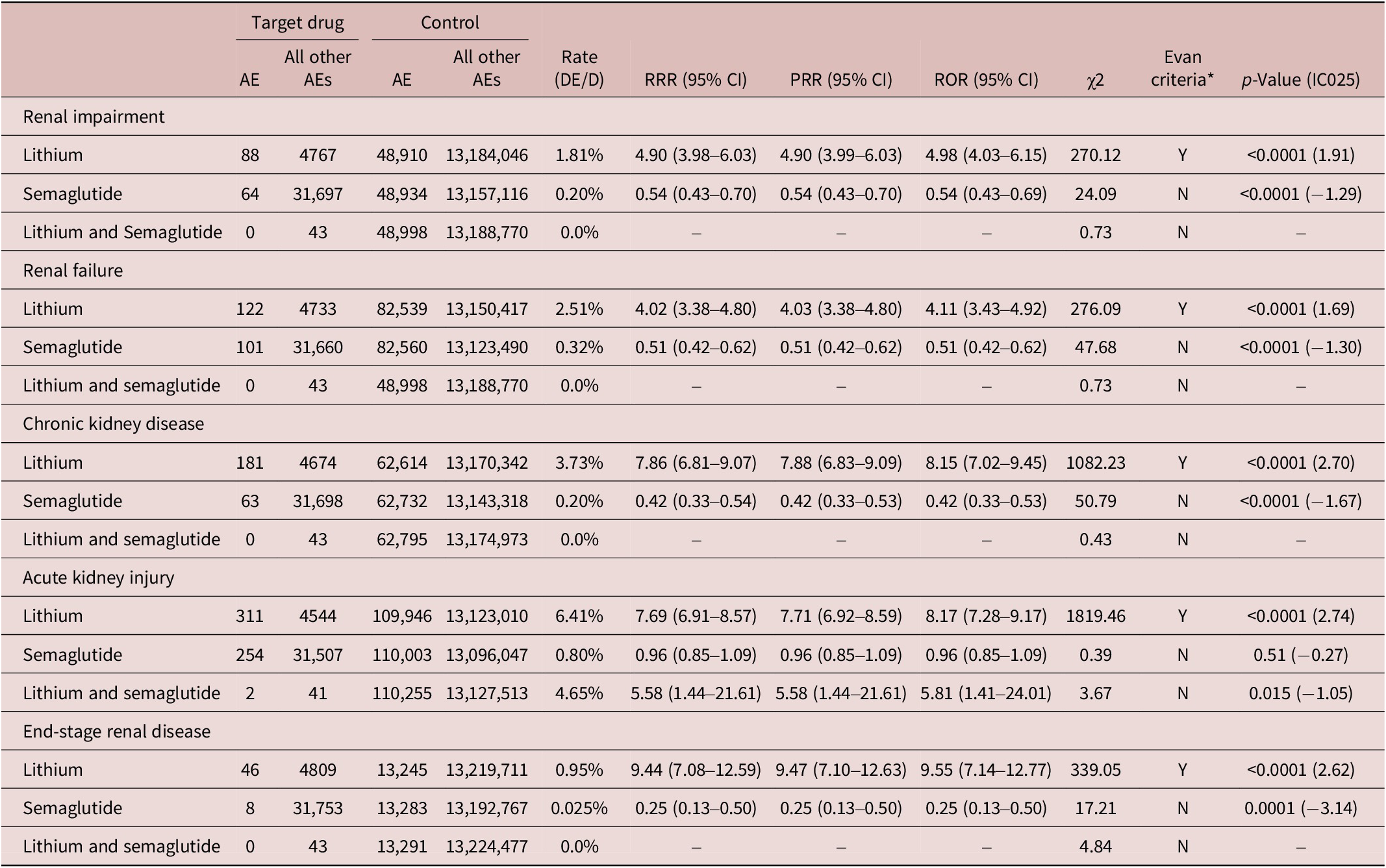
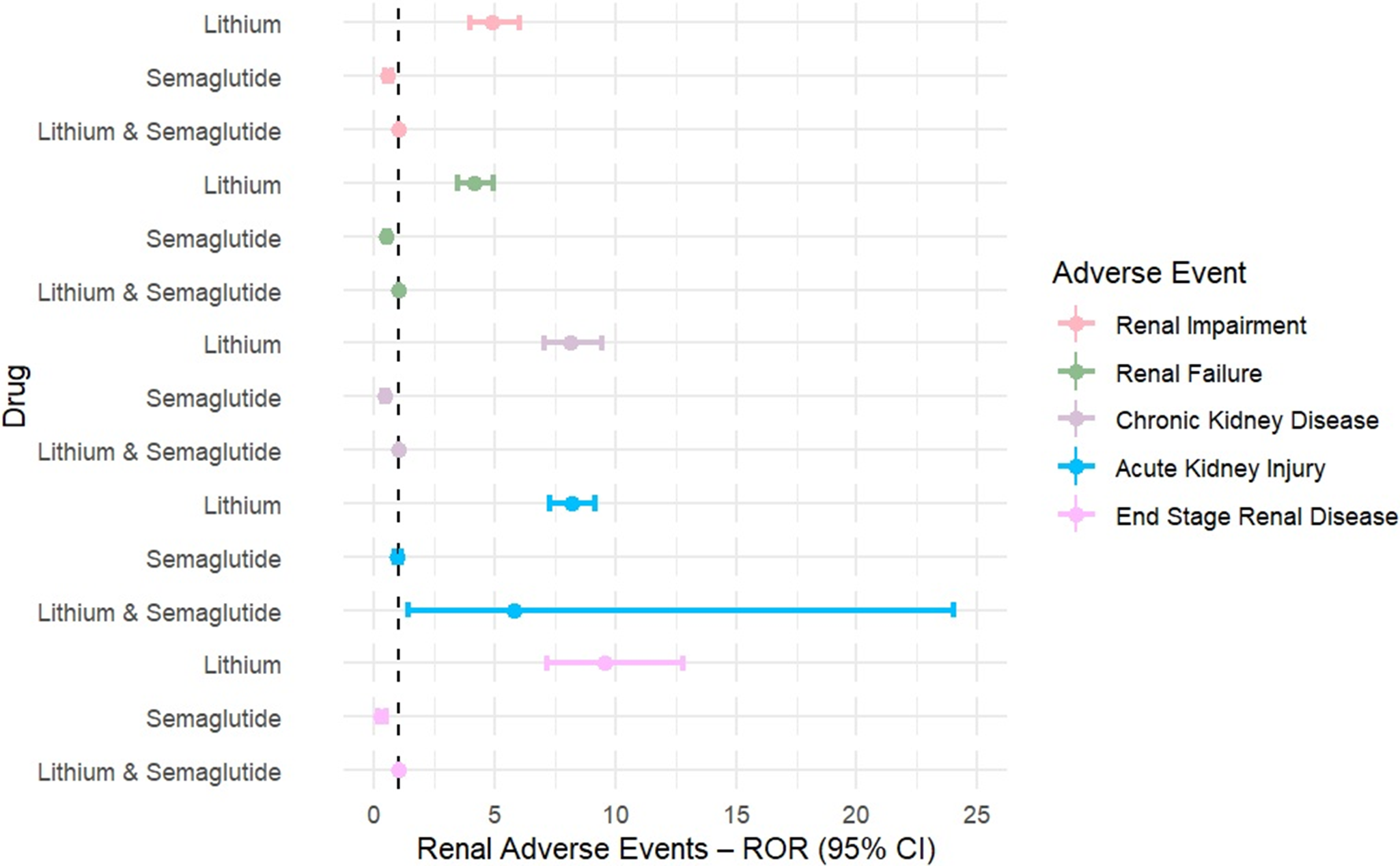
Lithium was consistently associated with significantly elevated reporting across all renal AEs. Disproportionality estimates were robust and exceeded signal thresholds for every endpoint: renal impairment (ROR = 4.98, 95%CI = 4.03–6.15, p < 0.0001, IC025 = 1.91), renal failure (ROR = 4.11, 95%CI = 3.43–4.92, p < 0.0001, IC025 = 1.69), CKD (ROR = 8.15, 95%CI = 7.02–9.45, p < 0.0001, IC025 = 2.70), ESRD (ROR = 9.55, 95%CI = 7.14–12.77, p < 0.0001, IC025 = 2.62), and AKI (ROR = 8.17, 95%CI = 7.28–9.17, p < 0.0001, IC025 = 2.74), as summarized in Table 1 and illustrated in Figure 1.
Renal Adverse Events Associated with Lithium, Semaglutide, and Their Combination Compared to All Other Drugs in FAERS

Forest plots of RORs were generated for lithium, semaglutide, and their combination across key renal adverse events.

In contrast, semaglutide showed inverse associations for renal impairment (ROR = 0.54, 95%CI = 0.43–0.69, p < 0.0001, IC025 = −1.29), renal failure (ROR = 0.51, 95%CI = 0.42–0.62, p < 0.0001, IC025 = −1.30), CKD (ROR = 0.42, 95%CI = 0.33–0.53, p < 0.0001, IC025 = −1.67), and ESRD (ROR = 0.25, 95%CI = 0.13–0.50, p = 0.0001, IC025 = −3.14), while the association with AKI was neutral (ROR = 0.96, 95%CI = 0.85–1.09, p = 0.51, IC025 = −0.27). None of the semaglutide outcomes met the disproportionality signal threshold (IC025 < 0).
Co-reporting of lithium and semaglutide produced no estimable RORs for renal impairment, renal failure, CKD, or ESRD because of zero reported cases. For AKI, 2 co-reported cases were identified (ROR = 5.81, 95%CI = 1.41–24.01, p = 0.015, IC025 = −1.05); however, this also did not meet the statistical signal threshold (IC025 < 0).
Discussion
In this large population-based pharmacovigilance study, and in accordance with lithium’s known nephrotoxic effects, lithium use was associated with significantly elevated reporting odds of renal impairment, acute renal failure, CKD, and ESRD. We also observed that concomitant administration of semaglutide with lithium did not yield a corresponding increase in nephrotoxicity signals. This observation potentially provides preliminary support for the hypothesis that GLP-1 RAs may exert a potentially protective effect on lithium-associated renal injury. Over the past 2 decades, longitudinal studies and registry analyses have revised prior estimates of lithium-induced nephropathy, indicating that while the absolute risk may be lower than historically reported, lithium exposure remains a cause and contributor to CKD progression and a not infrequent reason for treatment discontinuation.Reference Qian, Sud and Lee 3 , Reference Roxanas, Grace and George 29 –Reference McIntyre, Mancini, Parikh and Kennedy 31
GLP-1 RAs are increasingly prescribed for metabolic conditions that differentially affect individuals living with bipolar disorder, including obesity, type 2 diabetes mellitus, obstructive sleep apnea, and CKD.Reference McIntyre, Alda and Baldessarini 32 –Reference Liu, Ling and Lui 34 Beyond their metabolic benefits, GLP-1 RAs have demonstrated reductions in cardiovascular morbidity and mortality, which is the leading contributor to excess and premature mortality in bipolar disorder.Reference Goldstein, Baune and Bond 35 Moreover, GLP-1 RAs have been investigated for use in psychotropic-drug-related weight gain, binge eating, metabolic liver disease, and substance- or alcohol-use disorders, all of which are clinical presentations that are frequently encountered in the bipolar population.Reference McIntyre, Kwan, Rosenblat, Teopiz and Mansur 36 –Reference Zheng, Soegiharto and Au 40
The molecular and cellular dynamics affected by GLP-1 RAs have separately introduced a hypothesis that these agents may hold therapeutic potential in the treatment and prevention of mental disorders and progressive cognitive impairment.Reference McIntyre, Rasgon and Goldberg 41 For example, GLP-1 RAs are known to promote neurogenesis, neuroplasticity, and autophagy while reducing inflammation, oxidative stress, and neuronal apoptosis within critical neural circuits.Reference McIntyre, Rasgon and Goldberg 41 –Reference Au, Zheng and Le 43 The molecular, cellular, and circuit-level effects attributed to GLP1-RAs may confer neuroprotective effects against the progression of illness, particularly with respect to cognitive functioning.Reference Au, Lam, Lim and McIntyre 44
However, our findings must be carefully and cautiously interpreted, as they are regarded as highly exploratory and hypothesis-generating. The overarching reason for a measured interpretation of our findings is that analyzing data in a pharmacovigilance dataset (eg, FAERS) is not capable of establishing causality, and such databases are not intended to inform deliberation as to whether protective effects exist. Disproportionality analyses, including ROR and IC025, quantify reporting patterns relative to background rates and do not measure clinical risk or incidence.Reference Prieto-Merino, Quartey, Wang and Kim 45 Accordingly, an IC025 value below zero indicates an absence of disproportionality rather than a signal of protection.Reference Prieto-Merino, Quartey, Wang and Kim 45 These metrics are meant to detect safety signals, not infer benefit; an inverse IC025 reflects a reporting trend rather than therapeutic protection and requires validation in patient-level studies.Reference Böhm, Bulin, Waetzig, Cascorbi, Klein and Herdegen 46 These findings, therefore, cannot be interpreted as evidence that GLP-1 RAs confer a true renoprotective effect.
The limitations of our analysis, as well as any inference, interpretation, or implementation messaging derive from our methodology. Herein, we used the US FAERS, which relies on spontaneous reporting from the general population as well as industry sponsors (for whom reporting is mandatory). 47 Spontaneous reporting of any AE cannot be considered a comprehensive estimate of its occurrence. Moreover, FAERS does not include information related to variables that would be required before cause and effect can be established (ie, the Bradford Hill criteria).Reference Shimonovich, Pearce, Thomson, Keyes and Katikireddi 48 In addition, we are not able to know, in the general population, what percentage of persons prescribed lithium have also been prescribed a GLP-1 RA, including semaglutide. Notwithstanding this limitation, the prescription of GLP-1 RAs has increased approximately 700% in the US during the past 4 y, and approximately 12% of US adults have been prescribed GLP-1 RAs, with 6% of the population prescribed semaglutide.Reference Mahase 49 , Reference Harris 50 In addition, as stated earlier, GLP-1 RAs are indicated and/or prescribed off-label for many conditions known to differentially affect persons living with depressive and bipolar disorders.Reference McIntyre, Kwan, Rosenblat, Teopiz and Mansur 36 Consequently, it can be assumed that a percentage (albeit unknown percentage) of persons prescribed lithium may also have been co-prescribed a GLP-1 RA.
In addition to the aforementioned points, multiple other limitations should be acknowledged. First, the absence of denominator data precludes estimation of event incidence or risk ratios, as the total number of exposed individuals (to lithium, GLP-1 RAs, or both) is unknown. Second, confounding by indication is likely, as GLP-1 RAs are prescribed primarily to individuals with type 2 diabetes or obesity, who differ from lithium-treated patients in metabolic risk, renal surveillance intensity, and access to nephrology care. Third, confounding by indication may introduce channeling bias, since patients with advanced renal disease are often excluded from GLP-1 RA therapy, potentially creating an artificial appearance of protection. Fourth, FAERS is inherently susceptible to multiple reporting biases—including notoriety bias, where regulatory or media attention increases reporting; the Weber effect, characterized by heightened reporting shortly after drug approval; and surveillance bias due to differential clinical monitoring. Fifth, co-reporting cases were extremely limited, yielding low statistical power and wide uncertainty intervals that preclude meaningful inference.
Furthermore, the broader limitations of spontaneous reporting systems must be recognized. FAERS data depend on voluntary submissions from clinicians, patients, and manufacturers, resulting in underreporting and variable data completeness. The database also lacks patient-level covariates such as baseline renal function, diabetes severity, medication dose, duration of exposure, and comorbid conditions—all of which are essential for causal inference. Temporal trends in drug utilization and awareness may further influence reporting frequency, adding uncertainty to observed associations. Consequently, these results should be regarded strictly within the descriptive and hypothesis-generating scope of pharmacovigilance rather than as confirmatory evidence.
The hypothesis that semaglutide and/or other GLP-1 RAs are protective against lithium-induced nephrotoxicity cannot be considered proven by our results. Instead, our results provide preliminary support for the viable and testable hypothesis of renal protection, and it is our hope that they provide impetus for comprehensive mechanistic, histopathologic, and clinical studies with adequate methodology. It also needs to be clearly stated that GLP-1 RAs have multiple AEs and safety concerns, which reduce acceptability and have been difficult to access for eligible persons due to acquisition cost barriers. In addition, early reports of suicidality associated with these agents resulted in regulatory investigations concluding that cause and effect cannot be established but should continue to be a variable for further evaluation.Reference McIntyre, Mansur, Rosenblat and Kwan 24 , Reference McIntyre, Mansur and Rosenblat 51 –Reference Wang, Volkow, Berger, Davis, Kaelber and Xu 53
In conclusion, our overarching aim is to encourage comprehensive assessments of whether semaglutide (and/or other incretin receptor agonists) is capable of exerting nephroprotective effects in persons exposed to lithium. Definitive evidence would require triangulation of histopathologic, clinical, and observational studies with adequate methodology to determine cause and effect. Analyses based on pharmacovigilance data are inadequate to address causality and can neither confirm nor refute potential benefits of incretin receptor agonists in this context. Nonetheless, the absence of significant co-reporting of lithium nephrotoxicity and semaglutide—though potentially unrelated—could be aligned with an as yetunconfirmed hypothesis that incretin receptor agonists may be renally protective against lithium-associated injury.
Data availability statement
All data relevant to the study are included in the article.
Author contribution
Both authors (R.S.M. and A.K.) conceptualized, designed, and drafted the manuscript, as well as provided critical review for important intellectual concepts and approved the final version to be published. R.S.M. and A.K. analyzed and interpreted the data. Both authors agree to be accountable for all aspects of the work.
Financial support
This study was not funded. The FAERS database is publicly available without restriction.
Disclosure
R.S.M. has received research grant support from CIHR/GACD/National Natural Science Foundation of China (NSFC). RSM has also received and the Milken Institute; speaker/consultation fees from Lundbeck, Janssen, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Neurawell, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, Abbvie, and Atai Life Sciences. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.