Introduction
Light exposure at high latitudes is characterised by extreme seasonal variation. During winter, the sun may not rise for extended periods, while in summer, it may scarcely set. This results in a profoundly altered day–night structure, where natural cues that typically stabilise human physiology, particularly the 24-h light–dark cycle, become unreliable or may disappear entirely (Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). From a circadian perspective, this disruption is significant. Human sleep–wake timing, alertness, core body temperature rhythms, hormone secretion (notably melatonin), mood regulation and cognitive performance are regulated by an endogenous circadian clock that anticipates bright light during the day and darkness at night (Aschoff, Reference Aschoff1965, Reference Aschoff1971; Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Roenneberg, Kumar, & Merrow, Reference Roenneberg, Kumar and Merrow2007). As such, light serves as the primary “zeitgeber” for this system, with its phase-shifting and melatonin-suppressing effects dependent on timing, intensity and spectrum, particularly short wavelengths detected by melanopsin-expressing retinal ganglion cells (Berson et al., Reference Berson, Dunn and Takao2002; Brainard et al., Reference Brainard, Hanifin, Greeson, Byrne, Glickman, Gerner and Rollag2001; Duffy & Czeisler, Reference Duffy and Czeisler2009; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011; Khalsa, Jewett, Cajochen, & Czeisler, Reference Khalsa, Jewett, Cajochen and Czeisler2003; Lewy, Wehr, Goodwin, Newsome, & Markey, Reference Lewy, Wehr, Goodwin, Newsome and Markey1980; Thapan, Arendt, & Skene, Reference Thapan, Arendt and Skene2001). When daylength and spectral composition are abnormal, or when light exposure is mistimed relative to social obligations, circadian phase may drift or become misaligned, resulting in chronic social jetlag that impairs sleep and mood (Ehlers, Frank, & Kupfer, Reference Ehlers, Frank and Kupfer1988; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Wittmann, Dinich, Merrow, & Roenneberg, Reference Wittmann, Dinich, Merrow and Roenneberg2006).
Polar regions serve as a natural stress test for human temporal biology. Research involving Antarctic winter-over crews and other isolated, confined and extreme (ICE) environments demonstrates that prolonged darkness, restricted or mistimed light exposure and inflexible duty schedules can induce circadian phase delays, reduced and lower-quality sleep, increased fatigue, diminished cognitive performance, irritability and interpersonal tension (Palinkas, Reference Palinkas1992; Pattyn, Van Puyvelde, Fernández-Téllez, Roelands, & Mairesse, Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). When circadian phase shifts later, but operational schedules remain fixed, individuals are required to wake and work during their biological night, resulting in truncated sleep, impaired alertness and reduced mood (Barger et al., Reference Barger, Flynn-Evans, Kubey, Walsh, Ronda, Wang, Wright, Czeisler and Klerman2014; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022). These effects have operational implications in settings such as Antarctic bases, long-duration confinement studies and maritime watchkeeping, where fatigue and vigilance deficits pose safety risks (Basner et al., Reference Basner, Dinges, Mollicone, Ecker, Jones, Hyder, Kan, Morukov and Sutton2014; Erez, Lieberman, Rafael, Ketko, & Moran, Reference Erez, Lieberman, Rafael, Ketko and Moran2025; Shattuck, Lawrence-Sidebottom, Matsangas, Arendt, & Belenky, Reference Shattuck, Lawrence-Sidebottom, Matsangas, Arendt and Belenky2024). Clinically, circadian misalignment and sleep restriction are associated with adverse cardiometabolic outcomes and impaired glucose regulation in controlled studies, as well as with real-world risk patterns linked to social jetlag (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Morris, Purvis, Hu, & Scheer, Reference Morris, Purvis, Hu and Scheer2016; Scheer, Hilton, Mantzoros, & Shea, Reference Scheer, Hilton, Mantzoros and Shea2009; Wittmann et al., Reference Wittmann, Dinich, Merrow and Roenneberg2006).
However, most of this evidence comes from specialist or expeditionary populations (e.g. overwintering crews, research staff or simulation participants) living in deliberately isolated, high-demand conditions. Much less is known about what it means to live long-term in those environments as an ordinary civilian resident: to hold a job, raise a family and maintain wellbeing when there is effectively no “normal” light–dark cycle for months. Population-based studies from sub-Arctic Tromsø (69°N) suggest that, at the group level, residents may adapt better than expected: seasonal or monthly shifts in sleep timing and insomnia complaints are often small, and average sleep duration may show little systematic change across the year once age, employment and related covariates are considered (Johnsen, Wynn, Allebrandt, & Bratlid, Reference Johnsen, Wynn, Allebrandt and Bratlid2013; Sivertsen, Friborg, Pallesen, Vedaa, & Hopstock, Reference Sivertsen, Friborg, Pallesen, Vedaa and Hopstock2021). Yet these same datasets also emphasise inter-individual differences: winter brings more insomnia symptoms for some people, and later chronotypes tend towards delayed sleep timing and greater difficulty under winter darkness (Friborg, Bjorvatn, Amponsah, & Pallesen, Reference Friborg, Bjorvatn, Amponsah and Pallesen2012, Friborg, Rosenvinge, Wynn, & Gradisar, Reference Friborg, Rosenvinge, Wynn and Gradisar2014; Johnsen, Wynn, & Bratlid, Reference Johnsen, Wynn and Bratlid2012). In other words, stable population averages can mask divergent seasonal experiences, particularly for evening-type individuals who, even in general-population samples, show elevated risk for mood problems (Merikanto et al., Reference Merikanto, Kronholm, Peltonen, Laatikainen, Vartiainen and Partonen2015).
These individual differences are not trivial. Chronotype, broadly, whether someone is more morning-type or evening-type, is partly biological and partly determined by light exposure and societal routine. Evening types often experience recurrent social jetlag, a chronic mismatch between when the body tends to sleep and when work or school requires wakefulness (Roenneberg, Allebrandt, Merrow, & Vetter, Reference Roenneberg, Allebrandt, Merrow and Vetter2012; Wittmann et al., Reference Wittmann, Dinich, Merrow and Roenneberg2006). In high-latitude winters, once dawn arrives very late (or not at all), that misalignment can deepen: getting out of bed in the dark may feel physiologically harder, motivation can drop and people may report low energy, irritability, carbohydrate craving and withdrawal-like features that echo classic descriptions of seasonal affective symptoms (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984). In summer, by contrast, near-continuous ambient light can produce the opposite difficulty: people may feel wired, overstimulated and unable to downshift into sleep, leading to curtailed or fragmented rest and, over time, emotional volatility and exhaustion (Friborg et al., Reference Friborg, Rosenvinge, Wynn and Gradisar2014; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sivertsen et al., Reference Sivertsen, Friborg, Pallesen, Vedaa and Hopstock2021; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022). Qualitative and mixed-methods accounts from Antarctic personnel note both poles of this problem: depressive tone, lethargy and interpersonal friction during the long dark, as well as irritability, restlessness and insomnia under relentless daylight (Palinkas, Reference Palinkas1992; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). Comparable accounts from ordinary northern residents are much rarer, and population averages may conceal vulnerabilities among evening-type individuals who carry higher mood risk even in general samples (Merikanto et al., Reference Merikanto, Kronholm, Peltonen, Laatikainen, Vartiainen and Partonen2015).
These seasonal mood effects have traditionally been discussed under the label seasonal affective disorder (SAD), first described as a recurrent depressive pattern with winter onset and remission in spring/summer (Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984). Morning-timed bright-light therapy is effective for many individuals with winter SAD, with the strongest benefits when light is scheduled to advance circadian phase and stabilise sleep–wake timing (Blehar & Rosenthal, Reference Blehar and Rosenthal1989; Golden et al., Reference Golden, Gaynes, Ekstrom, Hamer, Jacobsen, Suppes, Wisner and Nemeroff2005; Khalsa et al., Reference Khalsa, Jewett, Cajochen and Czeisler2003; Terman et al., Reference Terman, Terman, Quitkin, McGrath, Stewart and Rafferty2005). More recent work broadens this framing substantially: in a cohort of more than 85,000 adults with objectively measured light exposure, higher daytime light was associated with reduced risk of major depressive disorder, PTSD, psychosis and self-harm, whereas higher night-time light was associated with increased risk of depression, anxiety, bipolar disorder and self-harm, independent of age, physical activity and photoperiod (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023). Convergent evidence also shows that evening/electronic light suppresses melatonin, delays circadian phase and degrades sleep (Chang, Aeschbach, Duffy and Czeisler Reference Chang, Aeschbach, Duffy and Czeisler2015; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011; LeGates, Fernandez, & Hattar, Reference LeGates, Fernandez and Hattar2014). In other words, bright days and dark nights appear broadly protective for mental health, whereas a broken light–dark structure is a risk, which is precisely the structure that high-latitude environments destabilise.
Beyond biology, there is also a social layer. Social zeitgeber theory proposes that regular social routines, such as waking, mealtimes, work and social contact at predictable times, help stabilise internal circadian rhythms, and that disruptions to those routines can precipitate mood disturbance in vulnerable individuals (Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Grandin, Alloy, & Abramson, Reference Grandin, Alloy and Abramson2006). Daily routine regularity can be quantified (e.g. the Social Rhythm Metric), and therapies that deliberately stabilise social and sleep–wake rhythms improve mood outcomes, especially in bipolar disorder (Frank et al., Reference Frank, Kupfer, Thase, Mallinger, Swartz, Fagiolini, Grochocinski, Houck, Scott, Thompson and Monk2005; Monk, Flaherty, Frank, Hoskinson, & Kupfer, Reference Monk, Flaherty, Frank, Hoskinson and Kupfer1990). In polar winter, social structure may erode: people describe “hibernation mode,” fewer social contacts, less outdoor activity and a later active day. In polar summer, by contrast, social and occupational demands may remain “normal,” but the environment rarely signals “night,” making disengagement and emotional decompression harder. Both scenarios challenge the maintenance of stable rhythms. From a mental health perspective, coping at high latitude is therefore not only about access to light but also about the deliberate imposition of routine, for example, fixed wake times, blackout blinds, morning light exposure, scheduled outdoor time, vitamin D supplementation, and, in some cases, temporary travel south during the darkest period (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022).
One further dimension, and the one that motivated the present work, is the question of time itself. Extreme photoperiods not only strain sleep and mood but may also destabilise how time is experienced, segmented and given meaning. In the absence of sunrise and sunset as natural anchors, people in high-latitude environments often describe time as something that must be actively imposed rather than intuitively felt; “day” becomes what the clock or workplace says it is, not what the sky is doing (Adam, Reference Adam1990; Nowotny, Reference Nowotny1994; Palinkas, Reference Palinkas1992; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022; Zerubavel, Reference Zerubavel1981). Accounts from Arctic and Antarctic settings suggest that under long darkness or unending daylight, people can feel temporally disoriented, lose a reliable sense of progression through the day and report that weeks “blur” together, while others develop deliberate temporal scaffolds (e.g. fixed mealtimes, shared routines and seasonal rituals) to retain structure (Monk et al., Reference Monk, Flaherty, Frank, Hoskinson and Kupfer1990; Palinkas, Reference Palinkas1992; Stuster, Reference Stuster2010; Suedfeld & Weiss, Reference Suedfeld and Weiss2000; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). This sits at the core of a wider programme in environmental temporal cognition: how humans construct, perceive and negotiate time when environmental time cues are atypical or absent (Wearden, Reference Wearden2016). Yet these temporal experiences, feeling out of sync with the day, dreading the long dark ahead and counting down to the return of light, are largely missing from the quantitative circadian literature, which tends to reduce “time” to clock-based phase markers. Capturing subjective temporal narratives is therefore essential if we are to understand not just how people sleep and feel in extreme light environments but how they inhabit time under those conditions.
The present study addresses that gap. We conducted an online qualitative study with adults living at high northern latitudes who routinely experience very long winter darkness and very long summer light. We asked participants to describe, in their own words, how seasonal light affects their sleep, energy, motivation, mood and sense of time; how those effects differ between winter and summer; and what they actually do to cope. We analysed these accounts using reflexive thematic analysis to identify common patterns in perceived circadian disruption, seasonal mood change, chronotype-linked vulnerability, temporal disorientation and self-directed behavioural regulation (Braun & Clarke, Reference Braun and Clarke2006, Reference Braun and Clarke2019, Reference Braun and Clarke2021). Our aim was to articulate the lived ecology of circadian and temporal strain in high-latitude civilians: how people understand the links between light, time, sleep and mental health, and how they actively attempt to protect wellbeing in an environment where the external zeitgeber is no longer reliable.
Methods
Study design and epistemological position
To examine how people experience and manage extreme seasonal light and time in everyday civilian life, we used a qualitative design and analysed responses using reflexive thematic analysis. Reflexive thematic analysis is an interpretive approach in which meaning is generated through the researcher’s active engagement with the data, rather than being treated as something simply “discovered” as if it were fully objective (Braun & Clarke, Reference Braun and Clarke2006, Reference Braun and Clarke2019). This approach is well suited to questions of lived experience because it treats participants as narrators of their own realities while making the researcher’s analytic role explicit.
Our aim was not to test a predefined hypothesis (e.g. that winter produces a specific increase in fatigue) but to map how participants themselves understood seasonal changes in sleep, mood, energy, coping and identity in their own communities. This emphasis on meaning-making is also consistent with calls in extreme-environment research to examine not only physiological load (e.g. circadian delay and insomnia-like disruption) but also how such strain is interpreted, normalised and managed in context (Palinkas, Reference Palinkas1992; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022).
Participants and inclusion criteria
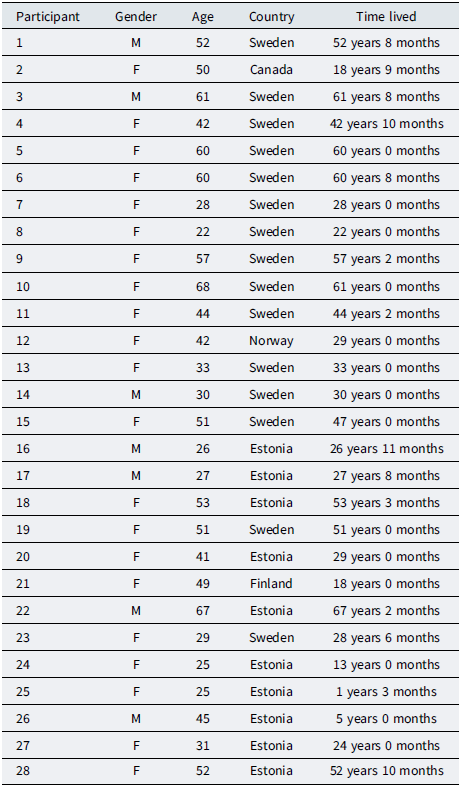
Twenty-eight adults (aged 18+ years) took part. All were living in high-latitude regions characterised by pronounced seasonal light variation (e.g. short winter photoperiods and very early sunrise/late sunset or near-continuous light in summer). Most participants were residents in Scandinavia (Norway, Sweden and Finland), with additional participants in Canada and Estonia. These settings routinely expose residents to long winter darkness and prolonged summer light, unlike expeditionary environments such as Antarctic winter-over stations (Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013; Palinkas, Reference Palinkas1992; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022).
Eligibility criteria were (a) age 18 years or older, (b) self-reported residence in a high-latitude or strong seasonal-light-variation location (broadly ≥55–60°N, or elsewhere with comparable seasonal light extremes), (c) ability to read and respond in English and (d) willingness to describe sleep, mood, time and coping across the year. We did not exclude participants on the basis of self-reported mental health history, sleep medication use or prior diagnoses (including SAD). Such disclosures were treated as part of participants lived contexts rather than as clinical classifications, consistent with a reflexive thematic approach (Braun & Clarke, Reference Braun and Clarke2019; Ehlers et al., Reference Ehlers, Frank and Kupfer1988). Participant characteristics are summarised in Table 1.
Participation information and demographics

Recruitment and procedure
Participants were recruited through informal circulation of the study link via personal and academic networks in Scandinavia and Canada and social media. This produced an opportunity (convenience) sample appropriate for exploratory qualitative work prioritising depth of account over demographic representativeness (Braun & Clarke, Reference Braun and Clarke2006, Reference Braun and Clarke2019). The study was presented as an exploration of lived experiences of seasonal light, sleep, mood, energy and coping, rather than as a clinical screen for SAD, to capture adaptation and self-regulation as well as distress (Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022).
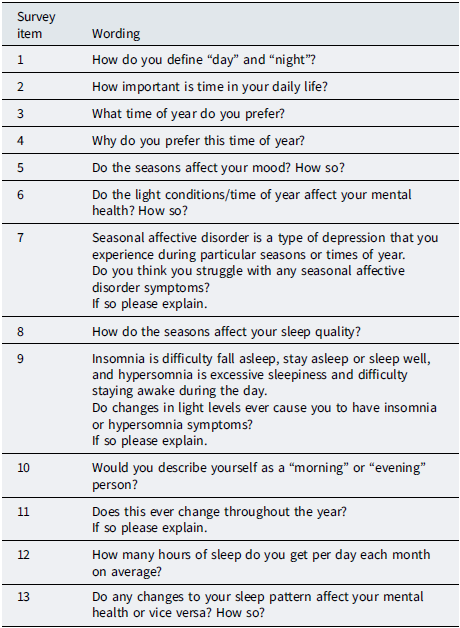
Data were collected via an online survey platform. After reading an information sheet and providing informed consent, participants created a unique code (e.g. three letters plus four numbers) to allow withdrawal within a defined time window without storing names alongside responses. Participants then completed brief structured items (e.g. age, country and approximate duration in location) followed by open-ended questions (Table 2).
Survey questions

Open-ended questions asked about seasonal changes in sleep (timing, quality and quantity), energy/alertness, mood and mental wellbeing, perceived clinical significance (e.g. “seasonal depression”), sense of time and daily rhythm (including whether “morning” and “night” remained meaningful under midnight sun/polar night conditions) and coping strategies (e.g. light management, medication, vitamin D, routine changes, outdoor activity, travel and mindset shifts). Participants also provided rough month-by-month estimates of perceived sleep duration and indicated which part of the year they liked best or struggled with most. Those structured items were used as interpretive anchors for contextualising free-text responses and were not analysed statistically.
Some prompts explicitly referenced difficulties (e.g. winter dread; insomnia/hypersomnia-type experiences) because the study intended to capture seasonal strain as well as coping. As noted in the Limitations section, this may have increased the salience of problem-focused responses and influenced which aspects of experience participants foregrounded.
The survey concluded with a debrief sheet reiterating withdrawal rights, explaining the study purpose in plain language and providing mental health support resources relevant to northern Europe and Canada, consistent with ethical good practice for research addressing mood and sleep disturbance (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984).
Analytic approach
We analysed the data using reflexive thematic analysis (Braun & Clarke, Reference Braun and Clarke2006, Reference Braun and Clarke2019). Analysis proceeded iteratively through familiarisation with the dataset, initial coding, construction of candidate themes, theme review/refinement and final theme definition and writing. Responses were read repeatedly, coded for patterns related to sleep/circadian experience, mood and affect, chronotype/alignment, temporal experience, and coping and then organised into broader interpretive themes. During theme review, we also checked that the developing analysis captured variation across accounts, including narratives of resilience and enjoyment of seasonal change as well as strain (cf. Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013). The final analysis was organised around four themes: (i) circadian rhythm and sleep across seasons; (ii) mood and mental wellbeing across seasons; (iii) time, chronotype and alignment and (iv) adaptation, coping and seasonal resilience.
Reflexive thematic analysis treats the researcher as an active interpreter rather than a neutral extractor of “objective” themes (Braun & Clarke, Reference Braun and Clarke2019). We therefore made analytic positioning explicit. The primary analyst has training in circadian psychology and was aware, from prior literature, that extreme photoperiods are able to disrupt sleep and mood (Palinkas, Reference Palinkas1992; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022). To reduce over-privileging disruption-focused interpretations, the analysis deliberately attended to counterexamples and non-pathology-framed accounts (e.g. participants who described winter as enjoyable, restorative or meaningful), and these were incorporated into the final thematic structure (Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022).
Because the aim was ecological description and interpretive depth, we did not quantify code or theme prevalence. Numeric summaries (e.g. proportions using specific coping strategies) would imply a degree of statistical generalisability that this design does not claim and that is not required within this analytic tradition (Braun & Clarke, Reference Braun and Clarke2019). Instead, we use cautious comparative language (e.g. “many,” “several” and “a minority”) and support themes with direct quotations.
Data handling, anonymity and ethics
Responses were stored pseudo-anonymously using participant-generated codes; no names or direct identifiers were retained with the qualitative data. Data were stored on encrypted, password-protected institutional systems. Participants were informed that they could request data removal within a fixed withdrawal period by supplying their code.
The study received ethical approval from the School of Psychology Ethics Committee at Keele University. Procedures complied with the Declaration of Helsinki and institutional standards for human research. No data were collected from or about children. Participants were reminded that they could skip questions, withdraw without giving a reason, and that reporting low mood or seasonal tiredness in the survey would not trigger a clinical pathway. Instead, the debrief signposted appropriate support services and encouraged participants to seek professional advice if they recognised significant distress. This approach is consistent with ethical good practice in research addressing seasonal mood disturbance and sleep disruption in potentially vulnerable or high-strain populations (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Palinkas, Reference Palinkas1992; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984).
Results
Analysis generated four interlocking themes: (1) circadian rhythm and sleep across seasons; (2) mood and mental wellbeing across seasons; (3) time, chronotype and alignment; and (4) adaptation, coping and seasonal resilience.
Each theme is presented below with illustrative extracts. Quotations are reproduced verbatim (including spelling and grammar). We do not quantify response frequencies (e.g. “42% said X”), as reflexive thematic analysis prioritises conceptual richness over frequency claims (Braun & Clarke, Reference Braun and Clarke2006, Reference Braun and Clarke2019).
Theme 1. Circadian rhythm and sleep across seasons
Participants consistently described sleep as seasonally shaped by light and darkness. Winter was typically associated with longer sleep, difficulty waking, residual tiredness and a sense of “hibernation,” whereas summer was associated with shortened, fragmented or prematurely ended sleep linked to early light and difficulty “switching off.”
Several participants described winter in terms of extended sleep and marked sleepiness:
-
“Late autumn and winter is for hibernation … I sleep longer in the dark months.” (P20)
-
“I can sleep for more than 12 hours and still dont feel like i got enough sleep.” (P7)
-
“In the winter i am always tierd and does not feel like i get enogh sleep.” (P7)
-
“I can sometimes sleep better and longer in winter as it’s so dark (I can go to bed very early and I feel good after that).” (P25)
Others described winter sleep as heavy rather than restorative: “more sleepy in the dark season and it feels like you never get enough sleep … Perhaps the quality of sleep is not good.” (P12)
In contrast, summer was often described as lighter sleep with earlier waking, even when tiredness remained:
-
“The sun will wake me up earlier (for instance at 5am) and I won’t manage to fall back to sleep even if I’m tired.” (P25)
-
“I’m sensitive to the morning light starting in March. It wakes me earlier.” (P21)
-
“I tend to lean to insomnia during spring and summer time.” (P11)
Many participants explicitly framed this as a seasonal reversal: “hypersomnia is probably more the case in winter, insomnia more in summer.” (P26) Another participant described the same polarity: “I would say that I can have light insomnia in the light season and light hypersomnia in the dark season.” (P12)
Not everyone reported marked seasonal changes. A minority described sleep as broadly stable across the year (e.g. approximately 6–8 h), indicating that seasonal instability was common but not universal. This pattern is consistent with evidence that population averages can appear relatively stable even where substantial individual variation exists (Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013).
Finally, participants also linked seasonal sleep disruption to daytime functioning and emotional regulation. As one participant put it, “If I don’t get enough sleep, I can feel more irritated and emotional.” (P10) Another stated, “changes in my sleep patterns affect my mental health negatively.” (P5) Across accounts, sleep was not experienced as a separate issue from wellbeing, but as part of a broader seasonal change in functioning. This subjective connection between circadian/sleep disruption and emotional reactivity echoes broader evidence that circadian misalignment and sleep curtailment are associated with mood dysregulation and increased psychiatric risk (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Ehlers et al., Reference Ehlers, Frank and Kupfer1988).
Theme 2. Mood and mental wellbeing across seasons
Participants described seasonal light as shaping not only sleep but also mood, motivation, sociability, anxiety and perceived psychological resilience. Winter was most often described as “low,” “flat,” “tense” or “anxious,” whereas high-light seasons were described as energising, optimistic and emotionally more robust.
Improved mood with light was often described directly and confidently:
“In the light season I have more energy, much better mood.” (P12)
-
“Light make you happy and it’s easy to handle bad stuff.” (P1)
-
“I feel more energized when there is more daylight.” (P4)
-
“Seeing the Sun is also very beneficial for my mood.” (P17)
By contrast, winter was frequently associated with irritability, anxiety, lethargy and psychological drag:
-
“If it’s dark and grey for longer time, it can feel depressive.” (P26)
-
“It’s difficult to be happy without seeing sun for days or weeks, which happens regularly in winter.” (P27)
-
“Shorter winter days cause excessive sleepiness, increase susceptibility to illness, depression, mood swings, fatigue, and difficulty concentrating.” (P22)
-
“I think the lack of light and sun in winter has a bad impact on my anxiety.” (P25)
Some participants also described what appeared to be anticipatory seasonal distress. One participant described autumn as psychologically difficult “because I’d know what was coming” and reported seeking support in advance because “the winter is hardest for me,” anticipating a decline in mood, energy and coping capacity as light disappeared. This forward-looking dread suggests that, for some participants, seasonal distress was experienced not only as reactive (“I feel bad now that it’s dark”) but also as cyclical and expected, consistent with the recurrent framing of seasonal mood disturbance in the SAD literature (Blehar & Rosenthal, Reference Blehar and Rosenthal1989; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984).
Participants also described close coupling between sleep disruption and affect. Poor or fragmented summer sleep (e.g. early waking and difficulty returning to sleep) was linked to irritability and emotional fragility, whereas winter tiredness and heavy sleep were linked to flatness, low motivation and social withdrawal (e.g. “In the winter I am always tired… I just want to stay in.” [P7]). These accounts are consistent with findings from polar and other ICE settings, where prolonged darkness, fatigue and circadian strain are associated with reduced positive affect, motivational decline and irritability (Palinkas, Reference Palinkas1992; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022) and with broader models linking sleep/circadian disruption to mood and anxiety vulnerability (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Ehlers et al., Reference Ehlers, Frank and Kupfer1988).
Crucially, however, not all participants framed winter darkness as negative. A minority described active appreciation of seasonal contrast:
-
“No, not really. I like long, light evenings in the summer, but also shorter days in the winter which enable me to rest more.” (P18)
-
“I’m a person who likes change so I’m always looking forward to the new season.” (P16)
-
“No, seasons do not affect my mood as I enjoy every season for different reasons.” (P5)
This pattern is consistent with evidence from high-latitude populations (e.g. Tromsø) suggesting that winter distress is not universal and that cultural meanings attached to winter may shape how seasonal change is experienced (Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013). Across accounts, mood and seasonal light were tightly linked, but not uniformly so: participants described both vulnerability and agency, including anticipatory preparation and more positive seasonal narratives.
Theme 3. Time, chronotype and alignment
This theme captures two linked ideas: first, that extreme light cycles can destabilise what “day” and “night” mean in everyday life; and second, that vulnerability to this destabilisation differs by chronotype.
When asked what “day” and “night” mean in their location, some participants defined them primarily by light (e.g. sunrise to sunset), whereas others defined them by clock time (e.g. “day is 7–20, night is 21–06”). Some blended both: “Day is bright and night is dark… or, timewise, 8-20 for day and 20-8 for night.” This tension suggests that in high-latitude environments, solar time and social time can diverge. Participants described having to impose socially meaningful temporal boundaries when the environment no longer reliably provides them:
-
“Time is a social construction I live in and live by … it’s making everyday life easier?” (P15)
-
“Important. I tend to eat, sleep, work, exercise based on the clock. My body is like a clock.” (P9)
-
“Very important … I have a lot of things that I want to do … I need to prioritize a lot.” (P4)
These accounts are consistent with the idea that, when light–dark cues are unreliable, people rely more heavily on structured social routines as surrogate zeitgebers (time-givers) that help stabilise daily rhythm (Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Grandin et al., Reference Grandin, Alloy and Abramson2006). Similar dynamics have been reported in Antarctic and other ICE settings, where participants describe temporal disorientation (e.g. days and weeks “blurring”) and deliberate reliance on shared schedules to maintain temporal progression and psychological normality (Palinkas, Reference Palinkas1992; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). Our participants described an analogous logic in civilian life.
Chronotype layered onto this. Many participants spontaneously described themselves as “morning people” or “night owls” and then explained how that identity interacted with season. Several evening-type participants described winter as especially difficult:
-
“Never a morningperson in the winter.” (P7)
-
“Not sure that changes, but it’s certainly more easy to get up in summer than in winter.” (P26)
-
“It is easier to wake up and go to work during the summer when the sun is shining early in the morning.” (P3)
For these participants, winter was described as persistent misalignment: they were required to function on social time that felt biologically too early, at a time of year when dawn light was weak or absent. They described heavy tiredness, irritability and reduced motivation. Some also described summer as problematic in a different way (e.g. difficulty sleeping early enough, or very early waking due to light), producing a different form of seasonal misalignment. This pattern is consistent with work linking later chronotype to social jetlag and greater vulnerability to mood disturbance under misaligned schedules (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Wittmann et al., Reference Wittmann, Dinich, Merrow and Roenneberg2006).
Participants who reported strong seasonal struggle often framed that struggle in terms of timing and alignment (e.g. difficulty “getting going” in winter mornings or “shutting off” in summer), rather than describing winter or summer as abstractly “bad.” This suggests that temporal alignment (biological time relative to required social time) is a central lived issue in high-latitude life, rather than only a technical concept in circadian theory (Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Wittmann et al., Reference Wittmann, Dinich, Merrow and Roenneberg2006).
Theme 4. Adaptation, coping and seasonal resilience
Participants did not describe themselves as passively enduring seasonal light extremes. Instead, they described active, layered strategies to protect sleep, mood, functioning and family life. These adaptations fell into four broad classes: light management, pharmacological/nutritional support, behavioural structuring and cognitive reframing.
Light management
Many participants described modifying the light environment (or shielding themselves from it) to create a more manageable day–night cycle:
-
“We have blackout blinds if I need to go to sleep and it is still light out.” (P2)
-
“I use blinds in lighter months for sleeping longer.” (P20)
-
“I have bought blackout curtains for this.” (P17)
Others described compensating for winter darkness using bright light or seasonal relocation: “Friends use special daylight lamps or move to Spain/Portugal or Asia for the coldest and darkest months.” (P20) These accounts are consistent with clinical and operational recommendations that use light exposure and blackout control to support sleep and mood under extreme photoperiods (Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022).
Pharmacological and nutritional support
Several participants described using prescribed sleep medication or supplements as seasonal tools:
-
“I got prescribed zopiclone by my doctor … it made me able to cope.” (P2)
-
“I take sleeping pills.” (P19)
-
“Once I was vitamin d-deficient during a winter and that changed my mood immensenly.” (P13)
Vitamin D was also described as important for winter wellbeing. These accounts suggest that some participants treated seasonal strain as something to actively plan for, rather than simply endure (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023).
Behavioural structuring
Participants also described designing different routines for winter and summer:
-
“In the light season I have more energy… easier to find activities for the kids outside and be more spontaneous.” (P12)
-
“In winter more thought-through activities (movement, winter swimming, other self-care) are needed to support good mental health.” (P27)
-
“The winter means I’m either braving the cold or trapped indoors!” (P2)
-
“In the winter I skee and iceskate. In the summer I enjoy the sun and long evenings. In the fall I pick mushrooms in the forest. In the spring I enjoy the nature flourishing.” (P5)
These accounts suggest strategic seasonal planning to maintain mood, activity and social connection. They are also consistent with the idea that social and behavioural routines can function as stabilising time-givers when environmental cues become unreliable (Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Grandin et al., Reference Grandin, Alloy and Abramson2006; Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013).
Cognitive reframing and identity
Finally, several participants described a form of seasonal identity work: choosing to experience winter not only as a problem but also as meaningful or restorative:
-
“I like long, light evenings in the summer, but also shorter days in the winter which enable me to rest more.” (P18)
-
“I’m a person who likes change so I’m always looking forward to the new season.” (P16)
These accounts suggest that adaptation involved not only practical strategies (e.g. blackout blinds and light boxes) but also interpretive ones, including a seasonal worldview in which winter and summer were both expected and manageable. Across accounts, participants described constructing stability through a combination of environmental control, routine and meaning-making.
Discussion
This study set out to understand what it is like to live with long winter darkness and long summer light, not as an overwintering researcher in Antarctica, but instead as an ordinary resident trying to sleep, work, parent, stay emotionally steady and hold a sense of time together in an environment that does not respect a 24-h day. The accounts show that extreme photoperiods are experienced as a chronic physiological and psychological negotiation (Palinkas, Reference Palinkas1992; Palinkas & Suedfeld, Reference Palinkas and Suedfeld2008; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022). Participants described seasonal shifts in sleep, energy, mood and routine, alongside deliberate efforts to rebuild structure. Taken together, these accounts connect three levels often studied separately: light and the circadian system (Aschoff, Reference Aschoff1965; Cajochen, Reference Cajochen2007; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011; Khalsa et al., Reference Khalsa, Jewett, Cajochen and Czeisler2003; Wehr, Reference Wehr1991), mood and coping (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Ehlers, Frank, & Kupfer, Reference Ehlers, Frank and Kupfer1988; Frank et al., Reference Frank, Kupfer, Thase, Mallinger, Swartz, Fagiolini, Grochocinski, Houck, Scott, Thompson and Monk2005; Golden et al., Reference Golden, Gaynes, Ekstrom, Hamer, Jacobsen, Suppes, Wisner and Nemeroff2005; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984) and the experienced organisation of temporal order within environments that are, in important respects, temporally disordered (Palinkas, Reference Palinkas1992; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022; Wearden, Reference Wearden2016; Zerubavel, Reference Zerubavel1981).
Seasonal light as a driver of circadian strain
Participants repeatedly reported that winter darkness and summer brightness altered how, and how well, they slept. Winter was described as heavy, slow and difficult to wake from; summer as shallow, fractured and prematurely ended by early light. These descriptions closely mirror findings from high-latitude and Antarctic settings, where winter darkness is associated with delayed circadian phase and longer apparent sleep need, while extended summer light is associated with sleep fragmentation, melatonin suppression and insomnia-like complaints (Friborg et al., Reference Friborg, Rosenvinge, Wynn and Gradisar2014; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sivertsen et al., Reference Sivertsen, Friborg, Pallesen, Vedaa and Hopstock2021; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Wehr, Reference Wehr1991). Mechanistically, mistimed evening or night light can suppress melatonin, delay circadian phase and reduce sleep continuity (Chang et al., Reference Chang, Aeschbach, Duffy and Czeisler2015; Duffy & Czeisler, Reference Duffy and Czeisler2009; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011).
In winter darkness, the circadian system is missing a strong dawn cue. Without early bright light to advance phase, sleep timing can drift later and waking may occur in the “biological” night (Czeisler et al., Reference Czeisler, Allan, Strogatz, Ronda, Sánchez, Ríos, Freitag, Richardson and Kronauer1986; Khalsa et al., Reference Khalsa, Jewett, Cajochen and Czeisler2003; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022). Participants captured this viscerally (“Never a morningperson in the winter”). That pattern maps onto chronic social jetlag: being required to wake, commute, work and care for children while physiology remains in “night mode,” a mismatch associated with fatigue, impaired alertness and poorer mood (Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Van Dongen, Maislin, Mullington, & Dinges, Reference Van Dongen, Maislin, Mullington and Dinges2003; Wittmann et al., Reference Wittmann, Dinich, Merrow and Roenneberg2006). In summer, the problem often inverted as participants described early light entering bedrooms and curtailed sleep (“I can’t get back to sleep”), consistent with light-related sleep disruption and insomnia-like complaints under continuous daylight (Palinkas, Reference Palinkas1992; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Zeitzer, Dijk, Kronauer, Brown, & Czeisler, Reference Zeitzer, Dijk, Kronauer, Brown and Czeisler2000). Our data suggest these dynamics are not confined to expeditionary settings but are part of ordinary domestic life in the north.
A key contribution here is that participants did not describe these sleep changes as minor inconvenience. Seasonal disruption was linked directly to functioning, emotional regulation and mental health. This is consistent with evidence linking irregular light exposure, curtailed sleep and circadian misalignment to increased psychiatric risk, including depression, anxiety and self-harm (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Lyall et al., Reference Lyall, Wyse, Graham, Ferguson, Lyall, Cullen and Smith2018), as well as longitudinal and meta-analytic evidence linking insomnia and atypical sleep duration to later depression (Baglioni et al., Reference Baglioni, Battagliese, Feige, Spiegelhalder, Nissen, Voderholzer and Riemann2011; Zhai, Zhang & Zhang, Reference Zhai, Zhang and Zhang2015). At high latitude, disrupted light–dark structure is therefore better understood as a mental health risk context, not simply a seasonal nuisance.
Mood, motivation and anticipatory emotional load
Mood, motivation and emotional resilience were also described as seasonally contingent. Participants tended to frame high-light seasons as energising and dark seasons as psychologically heavier. This is consistent with classic descriptions of SAD and with evidence that morning-timed bright-light therapy is effective for many individuals (Blehar & Rosenthal, Reference Blehar and Rosenthal1989; Golden et al., Reference Golden, Gaynes, Ekstrom, Hamer, Jacobsen, Suppes, Wisner and Nemeroff2005; Lam & Levitan, Reference Lam and Levitan2000; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984). At the same time, the narratives here go beyond clinical language: participants described a lived seasonal cycle of contraction and expansion, with winter as withdrawal and low drive, and summer as sociability and spontaneity, but sometimes also overstimulation and insomnia. That pattern is consistent with wider measures of seasonality in energy, sleep and activity (Kasper, Wehr, Bartko, Gaist, & Rosenthal, Reference Kasper, Wehr, Bartko, Gaist and Rosenthal1989) and with high-latitude operational reports of sleep disruption during continuous daylight (Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022).
Two refinements are notable. First, several participants described anticipating winter as distressing, including dreading the dark months in advance and arranging support before “the worst” period. This frames seasonality as a predictable psychological threat and resonates with Antarctic winter-over reports in which personnel anticipate low mood, strain and cognitive slowing during the long dark (Palinkas, Reference Palinkas1992; Palinkas & Suedfeld, Reference Palinkas and Suedfeld2008; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). Clinically, this suggests a preventative window for mood check-ins, morning-light planning and routine-setting before the deepest winter, in line with social-rhythm models and proactive or pre-emptive coping frameworks (Aspinwall & Taylor, Reference Aspinwall and Taylor1997; Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Golden et al., Reference Golden, Gaynes, Ekstrom, Hamer, Jacobsen, Suppes, Wisner and Nemeroff2005; Terman et al., Reference Terman, Terman, Quitkin, McGrath, Stewart and Rafferty2005).
Second, winter darkness was not uniformly experienced as harmful. A meaningful minority described winter as restful, valued seasonal change or a welcome slowing of pace. This echoes work from Tromsø suggesting modest population-level seasonality in sleep/distress and underscoring cultural framing of winter (e.g. cosiness, communal activity and outdoor life) as a psychological buffer (Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013; Johnsen et al., Reference Johnsen, Wynn and Bratlid2012; Leibowitz & Vittersø, Reference Leibowitz and Vittersø2020). There is therefore no single “polar psychology.” For public-health messaging, this matters: support should be available for those who struggle without pathologising winter for those who experience it as meaningful or restorative (Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013; Leibowitz & Vittersø, Reference Leibowitz and Vittersø2020).
Time, alignment and the work of keeping a day
Perhaps the most distinctive conceptual contribution of this study is the explicit linking of seasonal light to temporal experience. Participants did not only report feeling tired or low; they also described feeling out of time, with day and night needing to be rebuilt through routine (Palinkas, Reference Palinkas1992; Wearden, Reference Wearden2016; Zerubavel, Reference Zerubavel1981).
Several participants described “day” and “night” as socially defined rather than environmentally cued, and others described wintertime as “blurring” unless anchored by clocks, mealtimes or children’s schedules. This closely parallels Antarctic winter-over accounts of temporal disorientation under continuous darkness/light, where routine and ritual are used to restore temporal flow and social normality (Palinkas, Reference Palinkas1992; Stuster, Reference Stuster2010; Suedfeld & Weiss, Reference Suedfeld and Weiss2000; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). Our data show an analogous civilian process: people use everyday schedules and social zeitgebers to rebuild day and night (Monk et al., Reference Monk, Flaherty, Frank, Hoskinson and Kupfer1990; Zerubavel, Reference Zerubavel1981).
This matters theoretically because much circadian research operationalises time as clock time or phase angle, whereas participants’ accounts show time also functioning as a cognitive and social achievement. Routines (“I tend to eat, sleep, work and exercise based on the clock”), social obligations (“when the kids are awake”) and culturally embedded seasonal scripts (e.g. winter skating/skiing and summer evening activity) acted as scaffolds for temporal order. This maps directly onto social zeitgeber theory, which proposes that regular social structure helps stabilise internal rhythms and that disruption to these structures can precipitate mood disturbance in vulnerable people (Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Grandin et al., Reference Grandin, Alloy and Abramson2006). The present findings are also consistent with sociological work on socially organised time and with clinical approaches that formalise and stabilise daily rhythms (Frank et al., Reference Frank, Kupfer, Thase, Mallinger, Swartz, Fagiolini, Grochocinski, Houck, Scott, Thompson and Monk2005; Monk et al., Reference Monk, Flaherty, Frank, Hoskinson and Kupfer1990; Zerubavel, Reference Zerubavel1981).
Chronotype sharpened these effects. Evening types in particular described winter as punishing and summer as more disruptive. This is consistent with evidence that evening chronotypes experience greater social jetlag, greater sleep debt and an elevated risk of mood disturbance under early social schedules (Adan et al., Reference Adan, Archer, Hidalgo, Di Milia, Natale and Randler2012; Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Merikanto et al., Reference Merikanto, Kronholm, Peltonen, Laatikainen, Vartiainen and Partonen2015; Roenneberg et al., Reference Roenneberg, Allebrandt, Merrow and Vetter2012; Wittmann et al., Reference Wittmann, Dinich, Merrow and Roenneberg2006). In our data, the same individuals also described summer sleep disruption due to early light (“the sun wakes me at 5am”). This suggests that, for some chronotypes, there may be no fully comfortable season: winter undermines alignment because mornings are biologically difficult, while summer undermines alignment because nights never properly begin. This “double exposure” may help explain why some participants described the year not as a gentle seasonal cycle but as a recurrent physiological and psychological assault (Kantermann, Juda, Merrow, & Roenneberg, Reference Kantermann, Juda, Merrow and Roenneberg2007).
Clinically and operationally, this supports the value of chronotype-aware anticipatory care. Brief self-report chronotype tools are feasible in primary care and occupational settings (Adan & Almirall, Reference Adan and Almirall1991; Horne & Östberg, Reference Horne and Östberg1976; Roenneberg, Wirz-Justice, & Merrow, Reference Roenneberg, Wirz-Justice and Merrow2003). Individuals who identify as evening-type and report seasonal mood drops or marked winter sleepiness may benefit from early intervention support (e.g. morning light exposure, protected sleep windows and workload adjustments during darker months) rather than delayed intervention after functional decline (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Wittmann et al., Reference Wittmann, Dinich, Merrow and Roenneberg2006). This resembles fatigue-risk approaches in Antarctic and other ICE environments, where circadian status is treated as a manageable operational variable (Dawson & McCulloch, Reference Dawson and McCulloch2005; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022).
Coping as infrastructure, not preference
Theme 4 shows that participants do not simply “cope” in an ad hoc sense; they build systems. They use blackout blinds and eye masks to recreate darkness in summer, bright-light boxes to manufacture morning in winter and routine-based activity/social contact to stabilise sleep and mood. These practices are consistent with evidence on light-related melatonin suppression and sleep disruption (Chang et al., Reference Chang, Aeschbach, Duffy and Czeisler2015; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011), the phase-advancing and mood benefits of morning bright-light treatment (Golden et al., Reference Golden, Gaynes, Ekstrom, Hamer, Jacobsen, Suppes, Wisner and Nemeroff2005; Terman et al., Reference Terman, Terman, Quitkin, McGrath, Stewart and Rafferty2005) and social-rhythm stabilisation (Frank et al., Reference Frank, Kupfer, Thase, Mallinger, Swartz, Fagiolini, Grochocinski, Houck, Scott, Thompson and Monk2005; Monk et al., Reference Monk, Flaherty, Frank, Hoskinson and Kupfer1990). Some participants also described vitamin D use in line with high-latitude public-health advice (Scientific Advisory Committee on Nutrition, 2016), while others described reframing winter as permitted slowness and recovery rather than pathology (Leibowitz & Vittersø, Reference Leibowitz and Vittersø2020). Taken together, these accounts suggest intentional regulation of physiology, mood and temporal structure.
Comparable strategies are well documented in Antarctic winter-over and other ICE settings, where strict routine, lighting control, communal meals and ritualised social events support morale, cognition and cohesion (Palinkas, Reference Palinkas1992; Palinkas & Suedfeld, Reference Palinkas and Suedfeld2008; Stuster, Reference Stuster2010; Suedfeld & Weiss, Reference Suedfeld and Weiss2000; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). In our civilian sample, analogous tools were embedded in ordinary domestic life: blackout curtains, morning light boxes, vitamin D, winter swimming, scheduling outdoor time during the brightest hours, brief seasonal relocation and deliberate mindset shifts. In both contexts, these practices function as temporal scaffolds that rebuild day and night to protect sleep, mood and social normality.
This also clarifies what resilience means in this context. Resilience was not simply denial of strain, but often an active reinterpretation of seasonal extremes as identity and rhythm (“this is our winter”). That pattern corresponds to evidence that winter wellbeing in high-latitude communities is partly determined by cultural framing of darkness as meaningful rather than purely adverse (Johnsen et al., Reference Johnsen, Wynn, Allebrandt and Bratlid2013; Leibowitz & Vittersø, Reference Leibowitz and Vittersø2020). It also supports a biopsychosocial account of adaptation: blackout blinds and light boxes as biological levers, fixed mealtimes and family schedules as social levers and seasonal meaning-making as a cognitive-affective lever (Antonovsky, Reference Antonovsky1996; Engel, Reference Engel1977).
For policy, framing these behaviours as infrastructure rather than lifestyle preference matters. Public-health messaging in northern regions often centres on vitamin D and generic advice to “get outside at midday.” Our observations point to a broader, more explicit legitimisation of temporal and circadian self-management: blackout in summer as protective rather than antisocial, morning bright-light exposure in winter as prevention rather than “overreaction” and pre-season planning for mood/routine as anticipatory care rather than catastrophising (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Dawson & McCulloch, Reference Dawson and McCulloch2005; Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Golden et al., Reference Golden, Gaynes, Ekstrom, Hamer, Jacobsen, Suppes, Wisner and Nemeroff2005; Terman et al., Reference Terman, Terman, Quitkin, McGrath, Stewart and Rafferty2005). In this sense, circadian and temporal hygiene may be better treated as routine high-latitude self-management (and in some contexts, fatigue-risk management) than as clinical aftercare following breakdown (Chang et al., Reference Chang, Aeschbach, Duffy and Czeisler2015; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022).
Implications
For Arctic and sub-Arctic public health
These data suggest that sleep and mood in northern populations should not be treated as static traits but as seasonally strained systems that many people actively manage. Primary-care encounters could include seasonally framed questions (e.g. winter hypersomnia-like fatigue and summer early waking) and, where relevant, brief seasonality and chronotype screens (Adan & Almirall, Reference Adan and Almirall1991; Kasper et al., Reference Kasper, Wehr, Bartko, Gaist and Rosenthal1989; Roenneberg et al., Reference Roenneberg, Wirz-Justice and Merrow2003). Public messaging can also move beyond generic “get daylight” advice towards targeted routines: morning bright-light exposure in winter to support phase advance and sleep stability (Golden et al., Reference Golden, Gaynes, Ekstrom, Hamer, Jacobsen, Suppes, Wisner and Nemeroff2005; Khalsa et al., Reference Khalsa, Jewett, Cajochen and Czeisler2003; Terman et al., Reference Terman, Terman, Quitkin, McGrath, Stewart and Rafferty2005), strict blackout and evening light minimisation in summer to protect melatonin and sleep continuity (Chang et al., Reference Chang, Aeschbach, Duffy and Czeisler2015; Duffy & Czeisler, Reference Duffy and Czeisler2009; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011) and maintenance of social structure year-round to buffer mood via social zeitgebers (Ehlers et al., Reference Ehlers, Frank and Kupfer1988; Frank et al., Reference Frank, Kupfer, Thase, Mallinger, Swartz, Fagiolini, Grochocinski, Houck, Scott, Thompson and Monk2005; Monk et al., Reference Monk, Flaherty, Frank, Hoskinson and Kupfer1990). These recommendations are consistent with evidence linking irregular light exposure and circadian misalignment to psychiatric risk (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023).
For extreme and isolated operations (polar stations, offshore platforms and analogue space habitats)
These environments already recognise fatigue risk, but the present findings reinforce that circadian misalignment and temporal disorientation are not cosmetic morale issues; they are core psychological and operational risks. Winter-over programmes increasingly combine scheduled lighting, protected downtime and group routines to stabilise sleep and mood (Palinkas & Suedfeld, Reference Palinkas and Suedfeld2008; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022). Similar patterns are reported in other ICE contexts, including circadian-aligned watchkeeping and engineered schedules in maritime/submarine settings and sleep management needs in spaceflight and analogue missions (Barger et al., Reference Barger, Flynn-Evans, Kubey, Walsh, Ronda, Wang, Wright, Czeisler and Klerman2014; Basner et al., Reference Basner, Dinges, Mollicone, Ecker, Jones, Hyder, Kan, Morukov and Sutton2014; Erez et al., Reference Erez, Lieberman, Rafael, Ketko and Moran2025; Shattuck et al., Reference Shattuck, Lawrence-Sidebottom, Matsangas, Arendt and Belenky2024). Civilian accounts here show the same principles operating in ordinary homes. This supports the argument for circadian-aware rostering and dynamic light management in any long-duration ICE environment in which external zeitgebers are weak or absent (Chang et al., Reference Chang, Aeschbach, Duffy and Czeisler2015; Dawson & McCulloch, Reference Dawson and McCulloch2005; Gooley et al., Reference Gooley, Chamberlain, Smith, Khalsa, Rajaratnam, Van Reen, Zeitzer, Czeisler and Lockley2011; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022).
For cognitive models of time in extreme environments
Finally, and central to a temporal-cognition agenda, these accounts show that high-latitude life does not just involve seasonal fatigue and restlessness but also temporal displacement. Participants described manufacturing day and night, anticipating winter months in advance and experiencing weeks that “blur.” Under unstable photoperiods, temporal experience, how time is segmented, anticipated, endured and made meaningful, appears actively constructed rather than simply read from a clock (Flaherty, Reference Flaherty2003; Wearden, Reference Wearden2016; Zerubavel, Reference Zerubavel1981). Antarctic and other ICE narratives report similar patterns of temporal disorientation and deliberate temporal scaffolding (Palinkas, Reference Palinkas1992; Stuster, Reference Stuster2010; Suedfeld & Weiss, Reference Suedfeld and Weiss2000; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). Studying how people build and maintain temporal order should therefore be a priority for models of human functioning in environments with weak external time cues, including polar overwintering, prolonged submersion and spaceflight (Palinkas, Reference Palinkas1992; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022).
Limitations
This study is qualitative and based on an opportunity sample of 28 adults; it is not intended to estimate prevalence (e.g. “X% of high-latitude residents experience SAD-like symptoms”). The sample is not demographically balanced across age, gender, occupation or socioeconomic position, and clinical diagnoses were not systematically recorded. Data were collected asynchronously in writing, which likely enhanced candour and reflection but did not permit follow-up probing. Participants also varied in latitude: some reported near-total winter darkness or midnight sun, whereas others lived in highly seasonal but not fully polar conditions. We therefore interpret these accounts as experiences of strong seasonal light variation, not exclusively of true polar night.
An additional limitation concerns the framing of some survey prompts. Although the study was designed to elicit lived experiences of seasonal light, sleep, mood and coping, several prompts explicitly referenced difficulties (e.g. SAD symptoms and insomnia/hypersomnia), which may have increased the salience of problem-focused or dysfunction-framed responses. This does not invalidate the accounts reported here, but it may have influenced which aspects of experience participants foregrounded and may have reduced opportunities for neutral or positive experiences to be elaborated unless offered spontaneously. Subsequent research should use more balanced prompt sets that explicitly invite accounts of strain, adaptation, benefit, identity and seasonal meaning-making and should compare whether different elicitation framings yield different thematic emphases.
It is also important to recognise that this sample largely reflects Western/Northern European and Canadian settler perspectives. It does not attempt to represent Indigenous or land-based temporalities in the Arctic, where seasonal light, activity and cultural practice are embedded in particular historical, subsistence and colonial contexts. Future work should treat those contexts on their own terms, rather than assuming that “winter coping” generalises, and should be developed in ways that respect Indigenous knowledge systems and community priorities.
In our data, participants occasionally used diagnostic terms (“seasonal depression,” “anxiety,” “insomnia” and “SAD”) informally. We report these self-descriptions without treating them as verified clinical diagnoses. All coding and interpretation were conducted manually using a reflexive thematic analysis approach; no automated text analysis was used (Braun & Clarke, Reference Braun and Clarke2006, Reference Braun and Clarke2019, Reference Braun and Clarke2021). Raw participant text remains confidential and was not shared outside the research team; the analysis presented here is human, reflexive and accountable.
Despite these constraints, the accounts provide an ecological description of how circadian stress, mood and temporal experience are lived and managed in everyday high-latitude life, and they triangulate well with physiological, clinical and operational evidence from polar and other extreme environments (Burns et al., Reference Burns, Windred, Rutter, Olivier, Vetter, Saxena, Lane, Phillips and Cain2023; Palinkas, Reference Palinkas1992; Pattyn et al., Reference Pattyn, Van Puyvelde, Fernández-Téllez, Roelands and Mairesse2018; Rosenthal et al., Reference Rosenthal, Sack, Gillin, Lewy, Goodwin, Davenport, Mueller, Newsome and Wehr1984; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022).
Conclusion
Living through prolonged darkness and prolonged daylight is not simply “a bit tiring” but a whole-body negotiation with light. Participants described winter as a period of inertia, heavy sleep and lowered affect and summer as a period of curtailed or fragmented sleep, overstimulation and irritability. They also described temporal disorientation and the active rebuilding of day and night through routines, family schedules, rituals, light boxes and blackout practices. Seasonal light at high latitudes should therefore be understood as an environmental health pressure rather than just a cultural curiosity.
What is often described as “coping” is, in practice, a form of temporal and circadian engineering: residents actively construct biological day and night, emotional steadiness, and temporal order. This resembles the formal self-regulatory strategies used in polar and other ICE environments, where lighting schedules, protected downtime and shared routines are used to support functioning (Palinkas, Reference Palinkas1992; Sletten et al., Reference Sletten, Sullivan, Arendt, Palinkas, Barger, Fletcher, Arnold, Wallace, Strauss, Baker, Kloza, Kennaway, Rajaratnam, Ayton and Lockley2022; Van Puyvelde et al., Reference Van Puyvelde, Gijbels, Van Caelenberg, Smith, Bessone, Buckle-Charlesworth and Pattyn2022). The translational implication is clear: circadian-aware scheduling, morning-light protocols and social-rhythm supports should be normalised within northern public health, not reserved for specialist extreme operations (Ehlers et al., Reference Ehlers, Frank and Kupfer1988).
Data availability statement
To support research transparency and reproducibility, study materials and de-identified data associated with this manuscript are openly available on the Open Science Framework (OSF) at osf.io/3j7kp. The repository includes the survey materials (including the Qualtrics file), a de-identified dataset and related study documentation. All shared data were anonymised prior to upload.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
The authors declare none.
Ethical standards
The authors declare that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. Ethical approval was granted by the School of Psychology Student Ethics Committee, Keele University. All participants provided informed consent and were able to withdraw within three weeks of participation.