Introduction
Broccoli is a highly nutritious vegetable rich in vitamins, minerals, dietary fiber and sulforaphane (Huang et al. Reference Huang, Wang and Belwal2021). Although global broccoli production is substantial, approximately only 15% of the biomass is utilized during harvest. The remaining stems and leaves are typically discarded and left to decompose, resulting in environmental challenges such as water contamination and foul odors (Yan et al. Reference Yan, Zhou and Shahzad2023). Repurposing broccoli stems and leaves as animal feed offers a dual solution: alleviating the shortage of protein feed resources while mitigating environmental pollution and resource wastage. While previous studies have explored the use of dried broccoli stem and leaf powder in livestock and poultry production (Hu et al. Reference Hu, Zuo and Wang2011, Reference Hu, Wang and Pan2012), the prohibitive costs of drying have severely limited its practical application. Recently, fermented feed from broccoli stems and leaves (FBSL) has garnered increasing global research attention due to its rich nutritional profile, cost-effectiveness and ease of application.
The Jinhua pig, a renowned Chinese indigenous breed, is celebrated for its superior meat quality, characterized by tenderness, high intramuscular fat content and succulence. However, the breed faces limitations, including a slow growth rate, low lean meat percentage and excessive backfat thickness (Ren et al. Reference Ren, Yang and Xiang2020). Consequently, improving growth performance and carcass quality remains a critical priority for the Jinhua pig industry. Currently, data regarding the effects of FBSL on the growth performance, gut microbiota and carcass quality of Jinhua pigs remain limited. Therefore, this study investigated the impact of FBSL supplementation on these parameters, aiming to elucidate the underlying mechanisms involving gut microbiota and lipid metabolism, thereby providing a scientific basis for the application of FBSL in Jinhua pig production.
Materials and methods
The experiment was approved by the Zhejiang University Animal Care and Use Committee (No. ZJU20251202).
Experimental materials and design
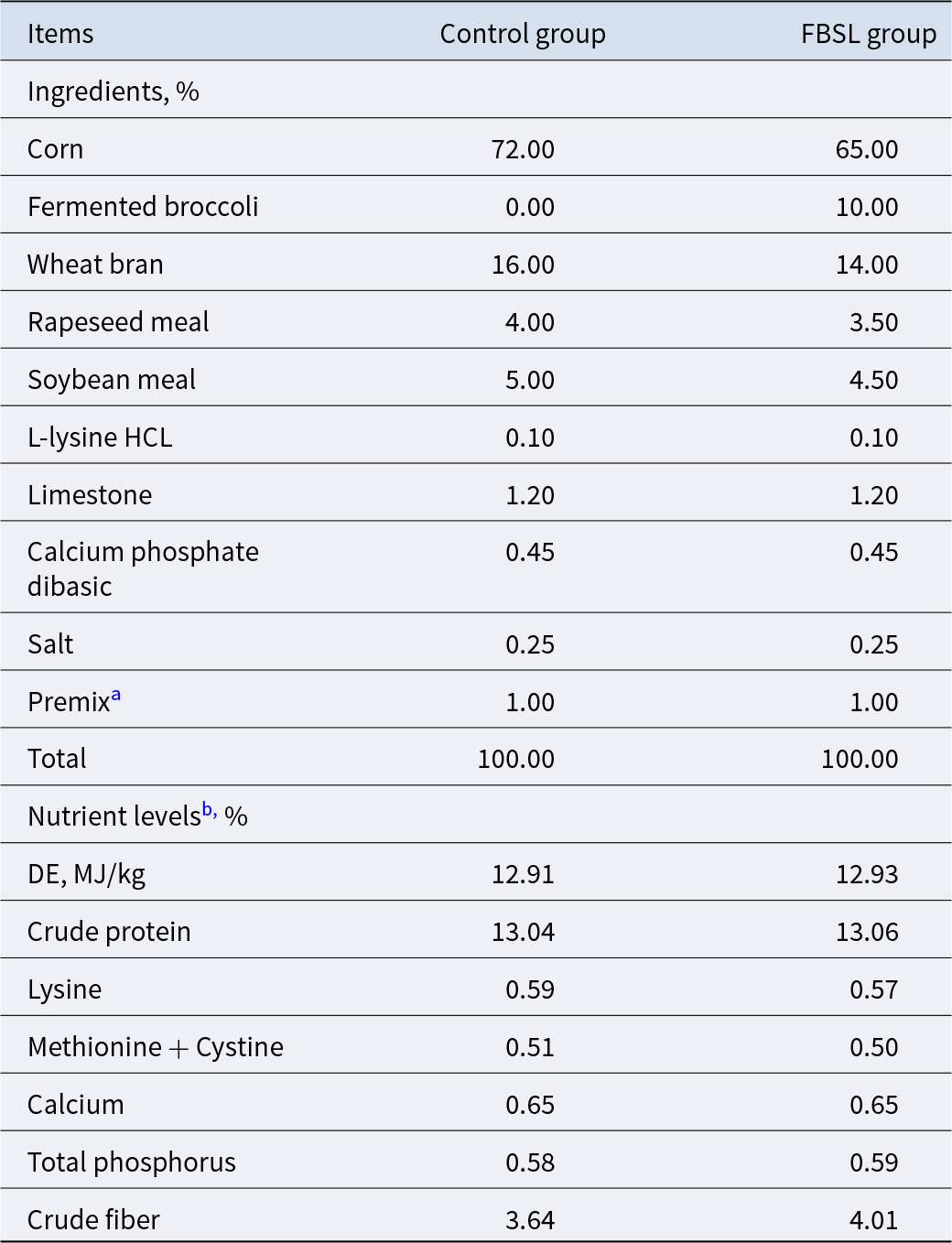
FBSL was produced by fermenting a mixture of broccoli, corn husks, rice bran, wheat bran, Yeast, Bacillus and Lactobacillus. The nutritional composition of the FBSL was as follows: moisture 50.13%, crude protein 12.25%, crude fat 1.21%, crude fiber 8.03%, crude ash 4.75%, crude polysaccharides 5.56%, polyphenols 0.41%, total acids 46.74 mg/g, lactic acid 25.01 mg/g, acid-soluble protein 87.84 mg/g, γ-aminobutyric acid (GABA) 6.07 g/kg, viable Bacillus count 3.58 × 10⁶ CFU/g and viable Lactobacillus count 6.97 × 10⁶ CFU/g.
A total of 36 Jinhua pigs with an average BW of 54.50 ± 1.76 kg (mean ± SD) were randomly allocated into two treatments, resulting in six pigs per pen and three pens per treatment. The control group (CON): basal diet; FBSL group: basal diet containing 10% FBSL. The diet formula met the nutritional recommendations for growing pigs of the NRC(2012), and the nutritional components were shown in Table 1. All pigs were weighed on day 1 (initial) and day 48 (final) of the experiment to calculate average daily gain (ADG). Feed consumption and residues were weighed and recorded on a pen basis to calculate the values of average daily feed intake (ADFI), and feed to gain ratio (F: G).
Composition and nutrient levels of the basal diet

a The premix provided the following per kilogram of the diet: vitamin A, 1350 IU; vitamin D3, 160 IU; vitamin E, 15 IU; vitaminK3, 0.5 mg; vitaminB1, 1.5 mg; vitaminB2, 2.5 mg; vitaminB12, 0.02 mg; folic acid, 0.3 mg; nicotinic acid, 9 mg; D-pantothenic acid, 8 mg; Mn, 3 mg; Zn, 55 mg; Fe, 50 mg; Cu, 3.5 mg; I, 0.15 mg; Se, 0.25 mg.
b Digestive energy (DE) is a calculated value; other nutrient levels are the measured values.
Samples collection and treatments
From each treatment, six pigs (two per pen) per group (with equal gender distribution) were randomly selected for slaughter. The carcasses were longitudinally split along the dorsal midline, with the left half utilized for assessment of carcass traits and meat quality. Following slaughter, 0.5 g of dorsal subcutaneous fat was clipped, and 2 g of colonic digesta were collected and placed into 5 mL Eppendorf tubes. Samples were flash-frozen in liquid nitrogen and stored at −80°C for subsequent analysis of colonic microbiota composition and short-chain fatty acids (SCFAs) content.
Carcass traits and meat quality measurements
Carcass weight was determined by weighing the eviscerated carcass (head, hooves, tail and offal removed, with leaf fat and kidneys retained) after exsanguination and dehairing of the swine. The ratio of post-slaughter carcass weight to pre-slaughter live weight was defined as the dressing percentage. Then, measured the average backfat thickness at three defined points and the loin eye area (Xu et al. Reference Xu, Chen and Huang2022). With reference to Ma et al. (Reference Ma, Liu and Yi2025), the 24 h pH value and 48 h drip loss of the longissimus dorsi muscle were determined. Meat color, including lightness (L*), redness (a*) and yellowness (b*), and the intramuscular fat content were measured according to previous study (Wang et al. Reference Wang, Zhang and Huang2022b, Reference Wang, Zhu and Li2024). The inosine monophosphate (IMP) content in meat samples was determined as described by Hui et al. (Reference Hui, Li and Fang2025).
Real-time quantitative PCR analysis
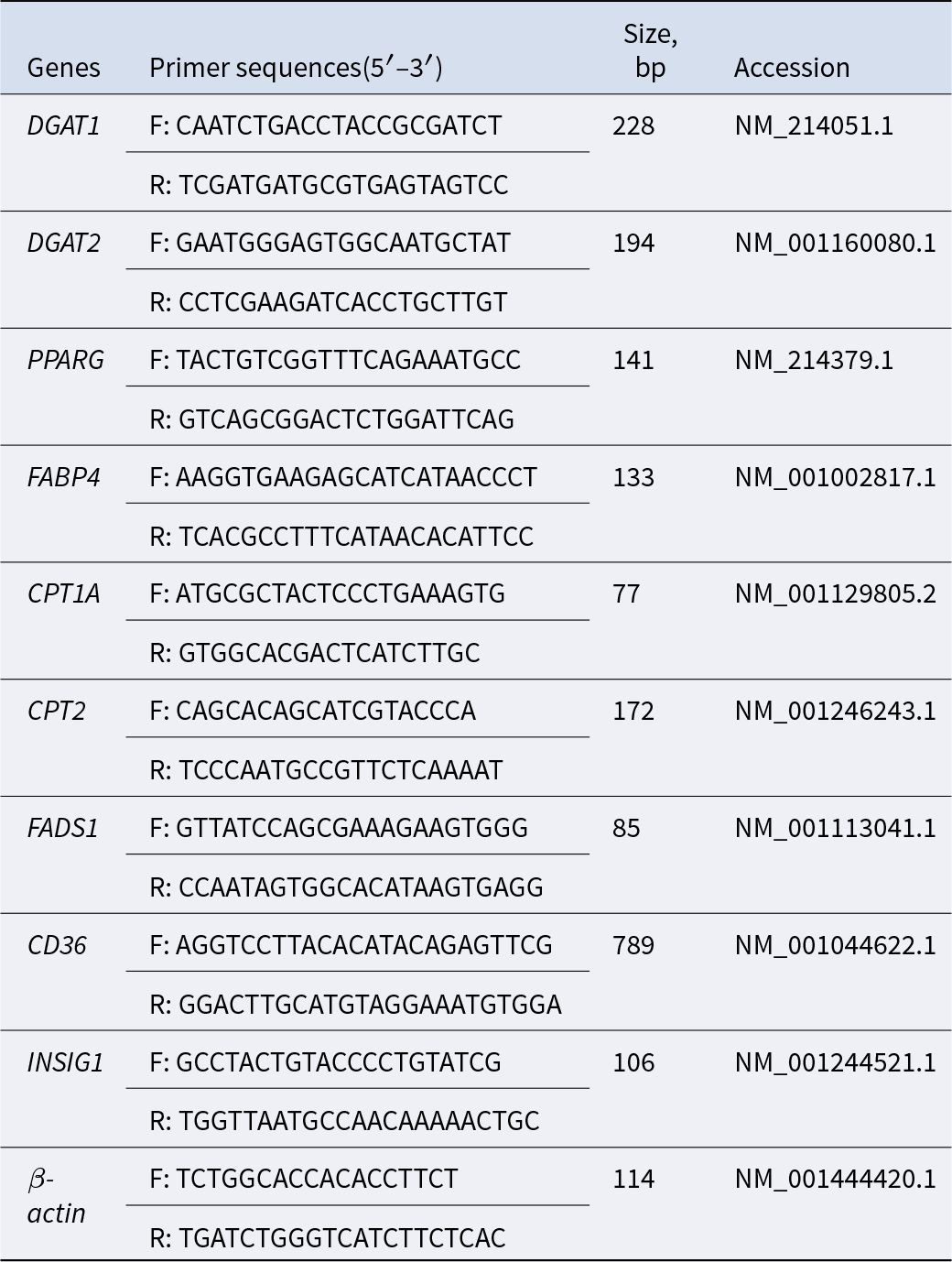
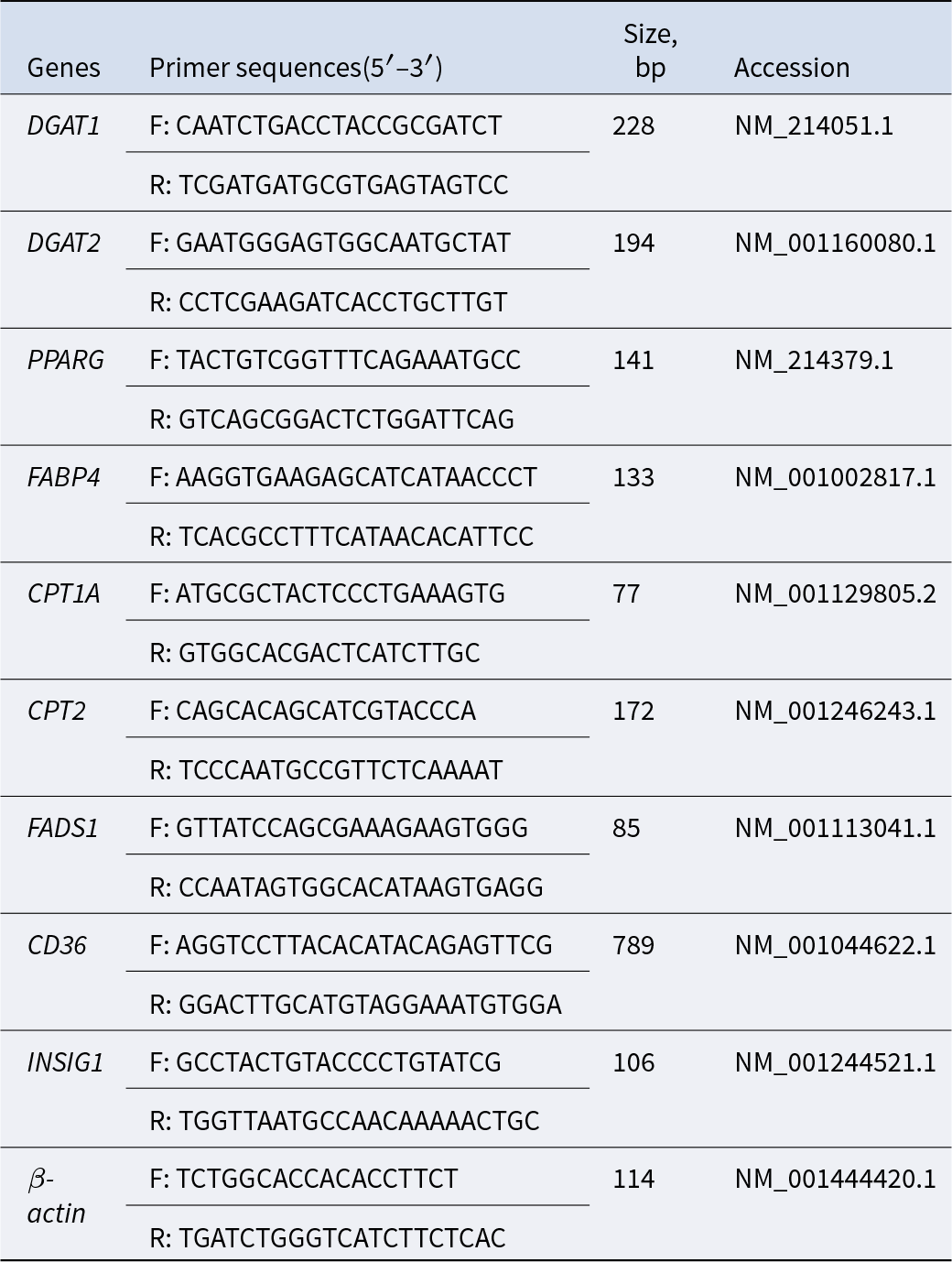
Total RNA was extracted from dorsal subcutaneous fat tissue using the total RNA extraction kit from Invitrogen (USA) in accordance with the manufacturer’s instructions. Complementary DNA (cDNA) was synthesized using the reverse transcription kit from Takara (Japan). Primers were designed based on the conserved regions of porcine genes in GenBank, with an amplification efficiency ranging from 95% to 108%. The data were analyzed following the 2−ΔΔCt method and calculated using β-Actin as the normalization control. The sequences of primers used were presented in Table 2.
Gene primer sequences for RT-qPCR
Short-chain fatty acids in colonic contents
The determination was conducted according to the method described by Zhou et al. (Reference Zhou, Zhang and Zhang2019). Analysis was performed using a 5890-Hewlett Packard gas chromatograph with the internal standard method. Approximately 8 g of colonic content was centrifuged at 4°C and 15,000 g for 15 minutes. One milliliter of supernatant was collected, mixed with 200 μL of a solution containing 25% (w/v) metaphosphoric acid and 2 g/L 2-ethylbutyric acid, incubated on ice for 30 minutes, and centrifuged at 10,000 g for 10 minutes before instrumentalanalysis.
16S rRNA gene sequencing
Genomic DNA was extracted from colonic contents using the CTAB/SDS method, followed by concentration measurement with Nanodrop ND-1000 and purity assessment via 1% agarose gel electrophoresis. The DNA was then diluted to 1 ng/μL with sterile water. The V3–V4 hypervariable region of the bacterial 16S rRNA gene was amplified using barcoded primers (341 F: 5′-CCTAYGGGRBGCASCAG-3′; 806 R: 5′-GGACTACHVGGGTWTCTAAT-3′). An Ion Plus Fragment Library Kit was used to construct the library, and sequencing was performed on the IonS5TMXL platform. Sequencing data underwent quality control with Cutadapt, followed by chimera removal based on the SILVA database via the UCHIME algorithm. Sequences with ≥97% similarity were clustered into operational taxonomic units (OTUs) using Uparse and annotated with the Mothur algorithm. OTU abundances were normalized according to the sample with the fewest sequences, and subsequent α-and β-diversity analyses were performed accordingly.
qPCR-based microbiota analysis
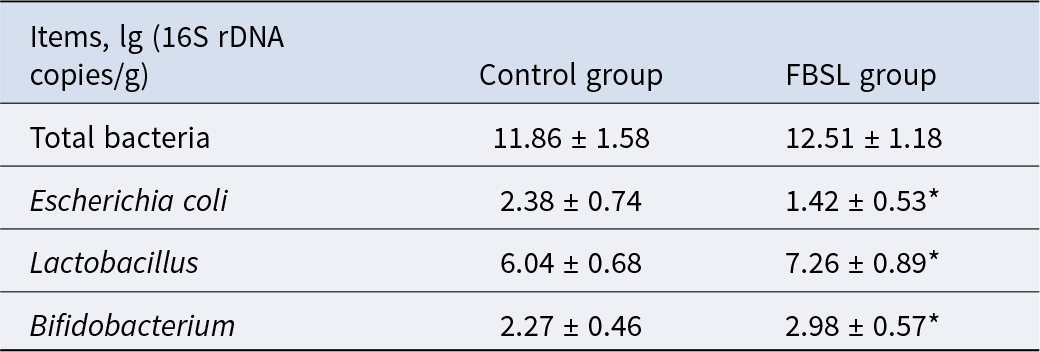
The P-MA qPCR Viable Bacteria Detection Kit was used to detect total bacteria, Escherichia coli, Lactobacillus, Bacillus and Bifidobacterium in colonic contents, with the primer sequences listed in Table 3. The qPCR reaction program was set as follows: pre-denaturation at 98°C for 1 minute, followed by 40 cycles of denaturation at 95°C for 15 seconds, annealing at 50°C for 45 seconds and extension at 72°C for 30 seconds, with a final extension at 72°C for 5 minutes. The copy numbers of target bacterial DNA were calculated based on the measured cycle threshold values (Ct value).
Bacterial primer sequences for qPCR

Statistical analysis
Data were analyzed using an independent samples t-test by SPSS 27.0 (SPSS Inc. Chicago, IL, USA) for Windows. All data were presented as means ± SD (standard deviation). These values were considered significant when P < 0.05. Figures were plotted by GraphPad Prism 10.3.1 software.
Results
Growth performance
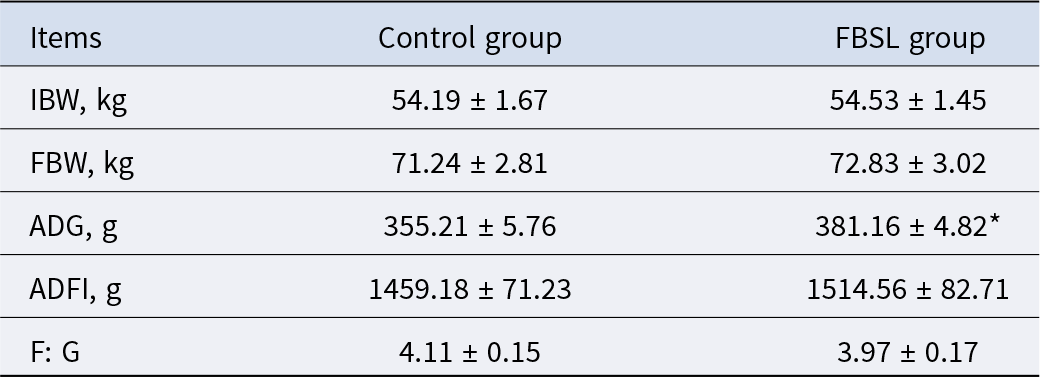
The effect of FBSL on growth performance of Jinhua pigs is shown in Table 4. Compared with the CON group, the average daily gain (ADG) of FBSL-fed Jinhua pigs increased by 7.31% (P < 0.05); however, there were no significant differences in average daily feed intake (ADFI) and feed to gain ratio (F: G) between the two groups (P > 0.05).
Effect of FBSL on growth performance of Jinhua pigs

IBW, initial body weight; FBW, final body weight; ADG, average daily gain; ADFI, average daily feed intake; F: G, the ratio of feed intake to weight gain. The data were expressed as mean ± SD (three replicates per group of six pigs per replicate). In the same line, values with superscript * mean significant difference (P < 0.05).
Carcass quality
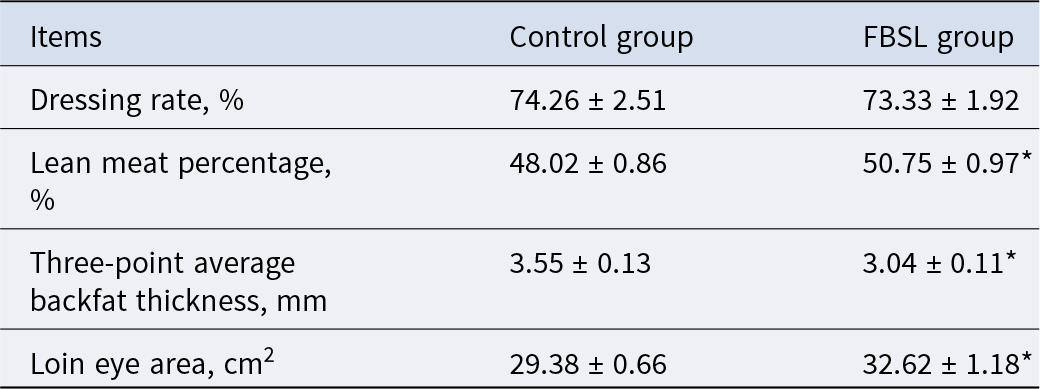
The effect of FBSL on carcass quality of Jinhua pigs is shown in Table 5. Compared with the CON group, the lean meat percentage and loin eye area of pigs in the FBSL group increased by 5.69% (P < 0.05) and 11.03% (P < 0.05), respectively; the average backfat thickness at three points decreased by 14.37% (P < 0.05), while there was no significant difference in dressing percentage between the two groups (P > 0.05).
Effect of FBSL on carcass quality of Jinhua pigs

The data were expressed as mean ± SD (n = 6). In the same line, values with superscript * mean significant difference (P < 0.05).
Meat quality
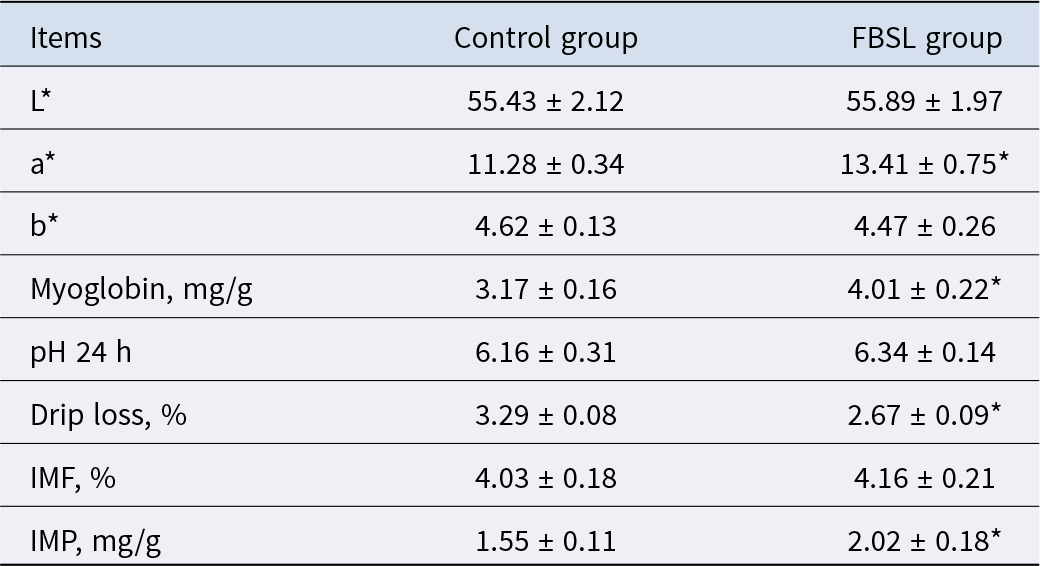
As shown in Table 6, compared with the CON group, the pork redness (a* value), myoglobin content, and inosine monophosphate content of pigs in the FBSL group increased by 18.88% (P < 0.05), 26.50% (P < 0.05) and 30.32% (P < 0.05), respectively; while the drip loss decreased by 18.84% (P < 0.05).
Effect of FBSL on meat quality of Jinhua pigs

L*, Lightness; a*, Redness; b*, Yellowness; IMF, intramuscular fat; IMP, inosine monophosphate. The data were expressed as mean ± SD (n = 6). In the same line, values with superscript * mean significant difference (P < 0.05).
Short-chain fatty acids in colonic contents
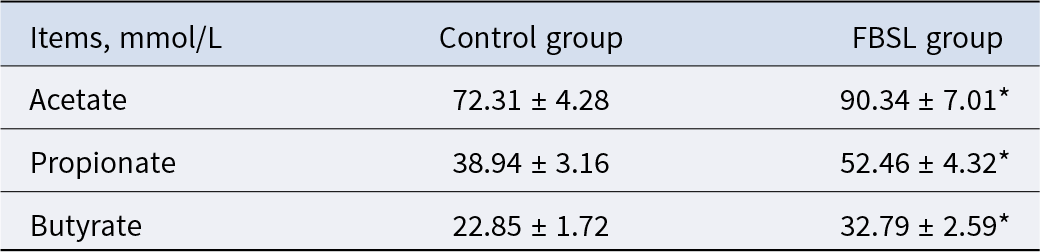
As shown in Table 7, compared with the control group, the contents of acetic acid, propionic acid and butyric acid in the colonic contents of pigs in the FBSL group increased by 24.93% (P < 0.05), 34.72% (P < 0.05) and 43.50% (P < 0.05), respectively.
Effect of FBSL on SCFAs in the colon of Jinhua pigs

The data were expressed as mean ± SD (n = 6). In the same line, values with superscript * mean significant difference (P < 0.05).
Lipid metabolism-related genes expression
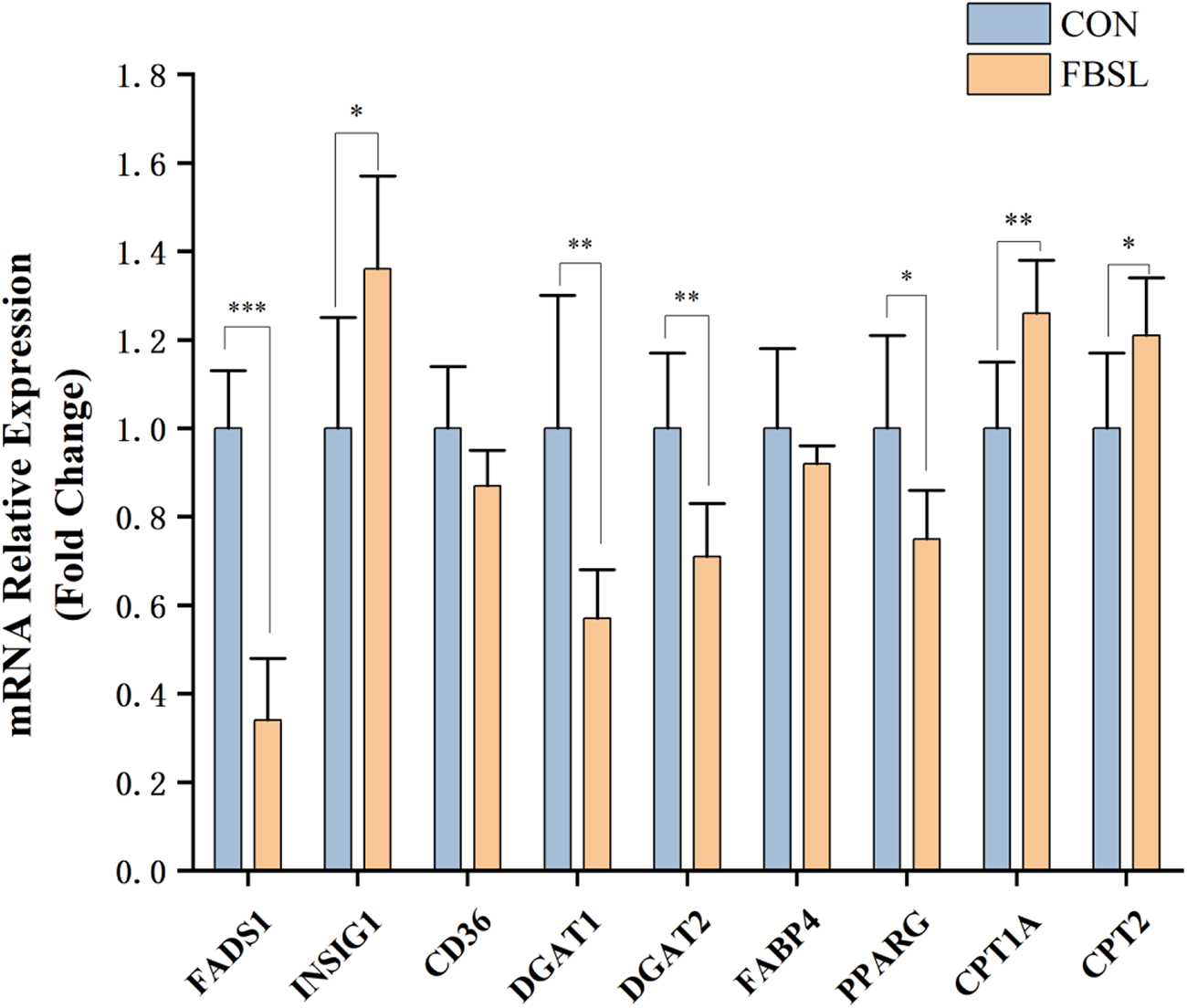
Compared with the control group, the relative mRNA expression levels of DGAT1, DGAT2, FADS1 and PPARG in the dorsal subcutaneous fat tissue of pigs in the FBSL group decreased by 43.31% (P < 0.05), 28.75% (P < 0.05), 66.32% (P < 0.05) and 24.67% (P < 0.05); while the relative mRNA expression levels of INSIG1, CPT1A and CPT2 increased by 35.83% (P < 0.05), 26.28% (P < 0.05) and 21.12% (P < 0.05) (Fig. 1).
Effect of FBSL on expression of genes related to lipid metabolism in Jinhua pigs.

α and β Diversity of colonic contents microbiota analysis
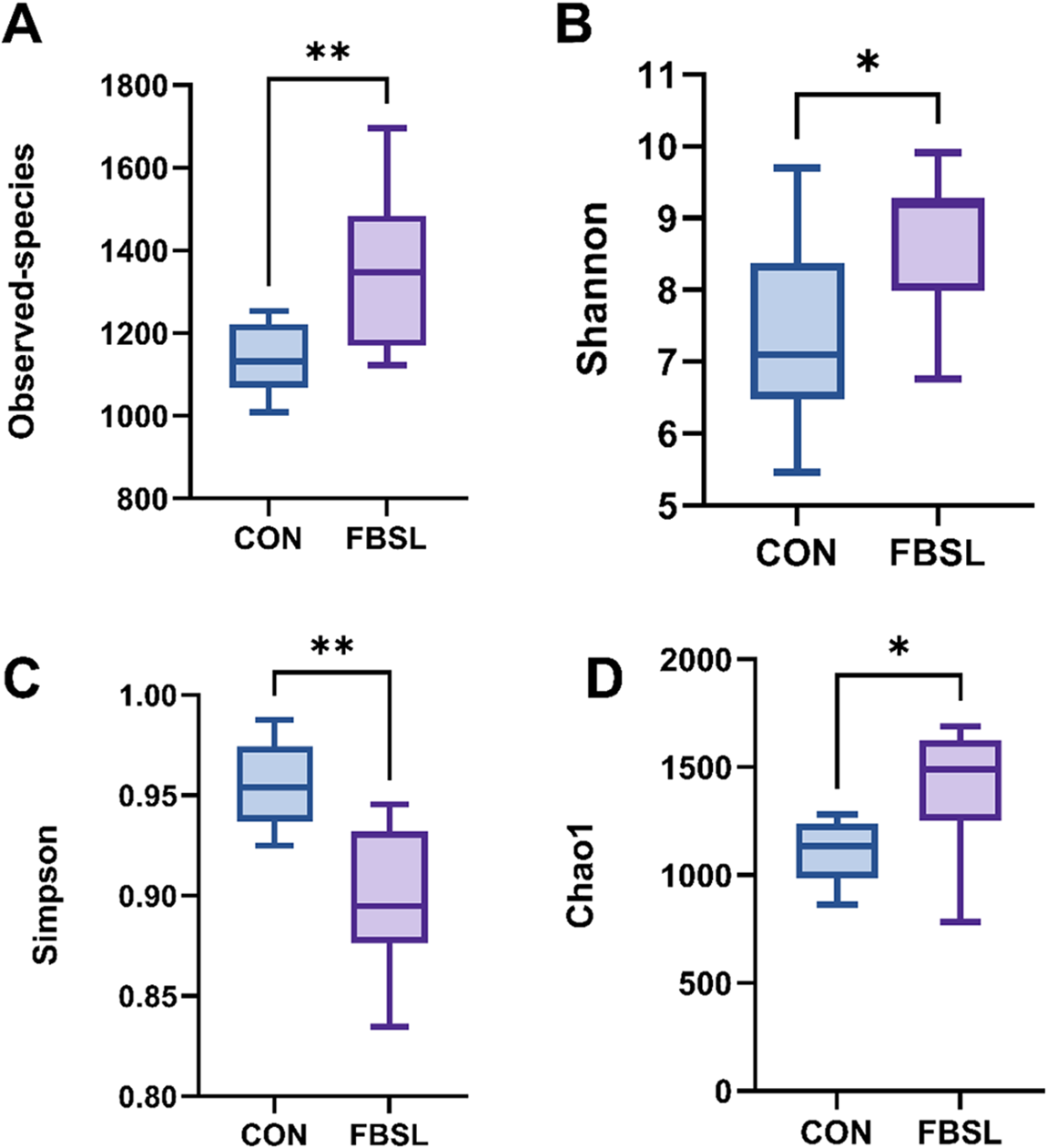
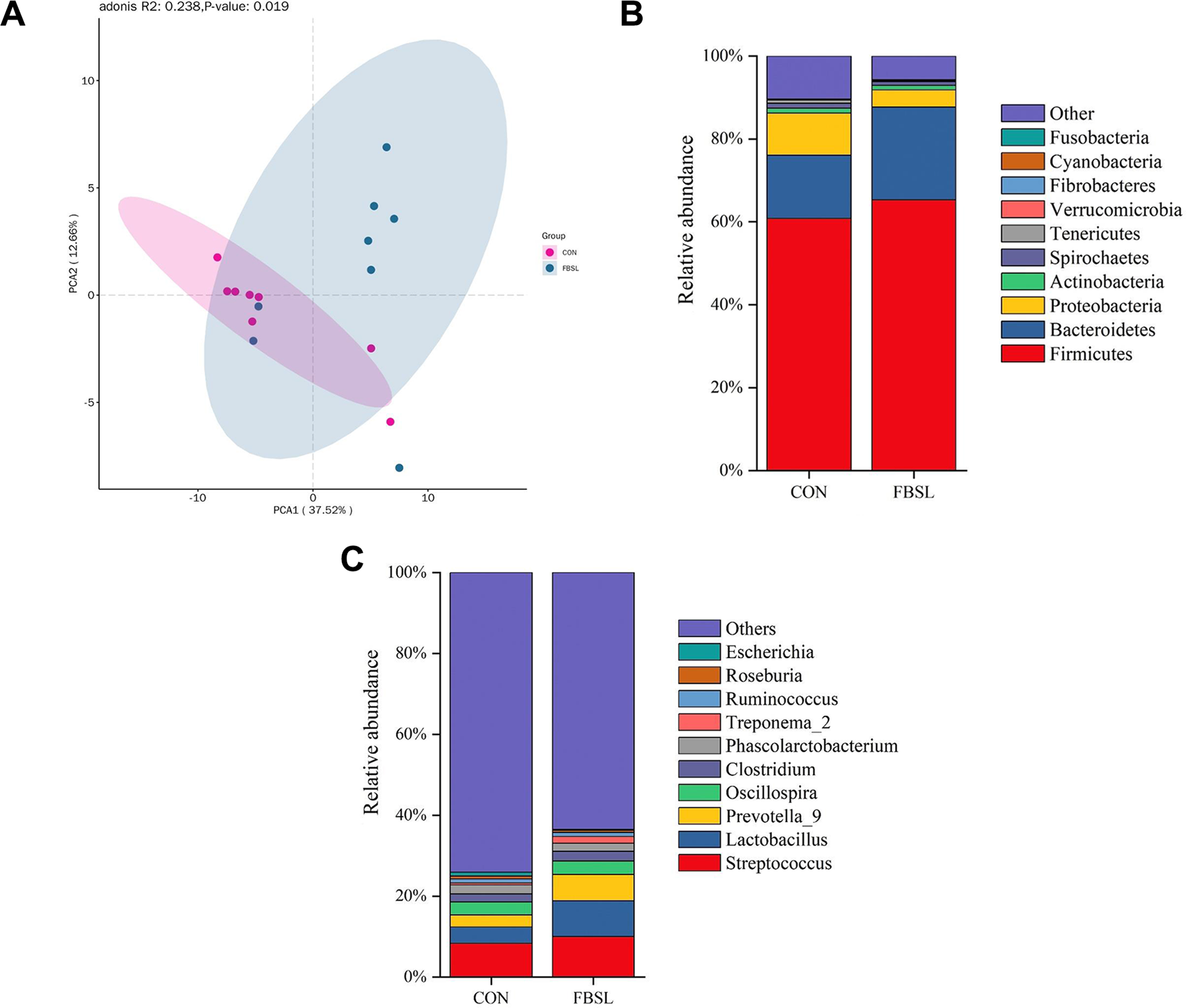
The results of α-diversity analysis indicated that compared with the CON group, the Observed species index, Shannon index and Chao1 index of the FBSL group were significantly increased (P < 0.05), while the Simpson index was significantly decreased (P < 0.05) (Fig. 2). These results suggested that FBSL could effectively enhance the richness of the colonic microbial community in Jinhua pigs. In the β-diversity analysis, the results of principal coordinate analysis based on the linear microbial community structure (Fig. 3A) showed that there was a significant difference in the colonic microbiota structure between the control group and the FBSL group (P < 0.05).
Effect of FBSL on α diversity of colonic contents in Jinhua pigs.

Effects of FBSL on β diversity and dominant flora of colonic contents in Jinhua pigs.

Abundance of colonic contents microbiota
FBSL could alter the community structure composition of the colonic contents’ microbiota in Jinhua pigs. At the phylum level, the colonic microbiota composition of finishing pigs in the two groups was consistent (Fig. 3B), with the main dominant phyla being Firmicutes, Bacteroidetes and Proteobacteria, which is consistent with the known characteristics of the core gut microbiota composition in pigs. Compared with the CON group, the relative abundance of Bacteroidetes in the FBSL group increased by 46.61%, while the relative abundance of Proteobacteria decreased by 59.41% (P < 0.05).
At the genus level, the main dominant genera included Streptococcus, Lactobacillus, Prevotella-9, etc. Among them, compared with the CON group, the relative abundances of Lactobacillus, Prevotella-9 and Treponema in the FBSL group were significantly increased (P < 0.05), while the relative abundance of Escherichia was significantly decreased (P < 0.05) (Fig. 3C). As shown in Table 8, the results of qPCR analysis of specific microbiota indicated that compared with the CON group, the relative abundance of Escherichia coli in the FBSL group decreased by 40.34% (P < 0.05), and the relative abundances of Lactobacillus and Bifidobacterium increased by 20.20% and 31.28%, respectively (P < 0.05).
Effects of FBSL on flora of colonic contents in Jinhua pigs

The data were expressed as mean ± SD (n = 6). In the same line, values with superscript *mean significant difference (P < 0.05).
Discussion
Current research regarding the effects of FBSL on porcine growth performance remains inconclusive. Zhao et al. (Reference Zhao, Sun and Yao2025) reported that supplementation with 5% FBSL had no significant effect on the growth performance of Duroc × Landrace × Yorkshire (DLY) finishing pigs. Conversely, Sun et al. (Reference Sun, Qian and Wu2023) observed that 5% FBSL supplementation significantly increased the ADFI of DLY growing pigs. In the present study, supplementation with 10% FBSL significantly enhanced the ADG of Jinhua pigs. Discrepancies in feeding efficacy may be attributed to factors such as fermentation quality, pig breed, age and inclusion levels. The FBSL used in this study was characterized by high viable counts of Bacillus and Lactobacillus, as well as an abundance of bioactive components, including crude polysaccharides, polyphenols, lactic acid, acid-soluble protein and GABA, which likely contributed to the observed improvements in growth performance.
Pork is the most consumed meat globally, and its quality is intrinsically linked to human health (Wang et al. Reference Wang, Zhao and Liu2023). As a premium indigenous breed, Jinhua pigs are noted for their fine muscle texture and tolerance to coarse feed; however, they are also associated with thick backfat, low lean meat percentage and slow growth. In this experiment, FBSL significantly reduced average backfat thickness at three measurement points and markedly increased the lean meat percentage. To elucidate the mechanism by which FBSL regulates lipid metabolism, we analyzed the mRNA expression of genes involved in lipid synthesis, lipolysis and transport in dorsal subcutaneous adipose tissue. DGAT1 and DGAT2 are pivotal acyltransferases in the triglyceride synthesis pathway; their downregulation directly suppresses triglyceride biosynthesis and reduces lipid precursor accumulation (Ji et al. Reference Ji, Chen and Zhang2022). FADS1, a fatty acid desaturase, is essential for polyunsaturated fatty acid biosynthesis, and its downregulation limits substrate availability for lipogenesis (Ghooray et al. Reference Ghooray, Xu and Shi2024). Furthermore, PPARG is a key transcriptional regulator of adipocyte differentiation, and its suppression inhibits adipogenesis and lipid deposition (Lei et al. Reference Lei, Li and Li2024). Our findings demonstrate that FBSL significantly downregulates the mRNA expression of DGAT1, DGAT2, FADS1 and PPARG in dorsal subcutaneous adipose tissue. Additionally, the upregulation of INSIG1 indirectly inhibits the transcriptional activation of adipogenesis-related genes, thereby reducing lipid synthesis capacity. Concurrently, increased expression of CPT1A and CPT2 – key enzymes in mitochondrial fatty acid β-oxidation – promotes fatty acid transport into the mitochondrial matrix for degradation (Han et al. Reference Han, Wei and Zhang2019). Collectively, these results indicate that FBSL mitigates fat accumulation in Jinhua pigs by suppressing lipid synthesis and enhancing lipolysis, leading to reduced backfat thickness and improved lean meat percentage.
Meat color, drip loss and flavor compounds are critical indicators of pork quality (Liu et al. Reference Liu, Long and Zhang2021). Pork redness (a* value), primarily determined by myoglobin concentration, directly influences consumer sensory acceptance. Drip loss indicates the water-holding capacity of muscle tissue and is a key parameter for assessing tenderness. Additionally, IMP is recognized as a pivotal compound responsible for the characteristic umami flavor of pork (Zhang et al. Reference Zhang, Du and Shen2025). Our study found that dietary supplementation with FBSL significantly improved meat quality attributes in Jinhua pigs, a* value and myoglobin content were markedly increased, drip loss was significantly reduced, and IMP content was substantially elevated. Previous studies suggest that Lactobacillus fermentation broth can increase muscle IMP content in finishing pigs (Lin et al. Reference Lin, Zhou and Lu2025), while polyphenols can inhibit myofibrillar protein oxidation, preserving muscle structure and myoglobin stability (Zhu et al. Reference Zhu, Han and Bu2022). Our results suggest that the synergistic action of various bioactive compounds in FBSL significantly enhances the pork quality of Jinhua pigs.
The balance of the digestive tract microbiota is crucial for nutrient digestion, absorption and the maintenance of gut microenvironmental stability. Zhao et al. (Reference Zhao, Sun and Yao2025) reported that FBSL supplementation improved the cecal microbial structure in DLY finishing pigs and reduced the abundance of harmful bacteria. Higher gut microbiota diversity is generally associated with greater community stability and enhanced gut health (Upadhaya and Kim Reference Upadhaya and Kim2022). In this study, FBSL significantly increased the observed species, Shannon and Chao1 indices of the colonic microbiota, while reducing the Simpson index, indicating a notable enhancement in microbial richness and diversity. Furthermore, β-diversity analysis revealed distinct structural differences in the microbiota between the control and FBSL groups. The high abundance of Bacillus and Lactobacillus in the FBSL likely drove these regulatory effects. High-throughput sequencing revealed that at the phylum level, the FBSL group exhibited a significantly higher relative abundance of Bacteroidetes and a lower relative abundance of Proteobacteria compared to controls. Bacteroidetes facilitate the degradation of complex carbohydrates via polysaccharide-degrading enzymes, thereby enhancing nutrient utilization (Wang et al. Reference Wang, Hu and Zijlstra2019). Conversely, the Proteobacteria phylum contains many opportunistic pathogens (e.g. Escherichia coli), and its reduction may lower the risk of gut inflammation (Carvalho et al. Reference Carvalho, Koren and Goodrich2012). At the genus level, FBSL significantly increased the relative abundances of Lactobacillus and Prevotella-9 while decreasing Escherichia. As a core beneficial genus, Lactobacillus lowers gut pH through lactic acid production, thereby inhibiting pathogen proliferation (Jiang et al. Reference Jiang, Cheng and Liu2020), while Prevotella-9 contributes to fiber degradation and short-chain fatty acid (SCFA) synthesis (Bai et al. Reference Bai, Zhou and Li2021). Their synergistic interaction reinforces gut microecological balance. Meanwhile, the marked reduction in Escherichia contributes to improved swine gut health (Zhao et al. Reference Zhao, Sun and Yao2025).
Quantitative PCR analysis confirmed that FBSL supplementation significantly increased the abundance of Lactobacillus and Bifidobacterium, while reducing E. coli populations in the colon. Lactobacillus enhances gut metabolic activity and homeostasis (Wang et al. Reference Wang, Zhu and Chen2021), while Bifidobacterium modulates glucose and lipid metabolism and alleviates colitis (Wang et al. Reference Wang, Huang and Tan2022a). The reduction of E. coli, a common opportunistic pathogen, may mitigate the risk of gut infections (Li et al. Reference Li, Zhao and Guo2024). These findings suggest that FBSL inhibits pathogenic growth while promoting beneficial microbial proliferation. Furthermore, SCFAs exert anti-inflammatory effects and promote gut health (Van Bockstal et al. Reference Van Bockstal, Prims and Van Cruchten2024). In this experiment, FBSL supplementation significantly elevated colonic concentrations of acetic acid, propionic acid and butyric acid. The crude fiber in FBSL serves as a substrate for microbial fermentation, driving SCFA production, which plays a critical role in modulating colonic physiology and reshaping the gut environment (Cong et al. Reference Cong, Zhou and Zhang2022).
Conclusions
In conclusion, our research has shown that supplementation of 10% FBSL significantly increases the ADG and lean meat percentage of Jinhua pigs, reduces backfat thickness, and improves pork a* value, myoglobin content and IMP content. Moreover, FBSL enhances the growth performance and carcass quality of Jinhua pigs by optimizing the colonic microbiota structure, increasing the gut SCFAs content and regulating the expression of lipid metabolism-related genes.
Acknowledgements
This research was supported by Zhejiang Provincial San Nong Jiu Fang Project (2025SNJF005) and Zhejiang Provincial Agricultural Industry Technology Team Project.
Author contributions
L.L.L.: Conceptualization, Investigation, Methodology, Formal analysis, Writing. J.Q.H., Y.J.L., Y.D.W., J.F.: Investigation, Formal analysis, Writing – review & editing. C.R.Z., C.H.H.: Conceptualization, Investigation, Formal analysis, Writing – review & editing, Supervision, Funding acquisition. All authors read and approved the final manuscript. Cenrong Zhang can also be contacted for correspondence, email: zhangcenrong@163.com
Conflict(s) of interests
The authors declare no conflict of interest.