Impact statement
Drylands cover over 40% of Earth’s land surface, supporting billions of people who depend on these fragile ecosystems for their livelihoods. As climate change intensifies and human activities expand, dryland degradation from human-accelerated wildfire cycles and disturbances threatens soil stability and ecosystem health in these areas. Biological soil crusts – thin, cohesive soil surface layers built by and infused with cryptogams – are critical for preventing erosion and maintaining dryland fertility, yet are easily destroyed and require several years to decades to recover naturally. This research addresses a major bottleneck in restoring degraded drylands: cultivating weed-free biocrust products for use in restoration. While scientists have developed innovative methods to grow and transplant these crusts as “sods” (similar to grass sods for lawns), invasive weeds that sprout during the farming process threaten to derail this promising restoration approach. If invasive plants hitchhike on biocrust sods to restoration sites, they could cause more harm than good. Our study demonstrates, for the first time, that specific herbicides can control invasive plants during biocrust farming without inhibiting biocrust development – and in some cases, even enhancing their growth. This breakthrough makes biocrust farming more practical and cost-effective by reducing labor-intensive hand-weeding, potentially enabling restoration at scales that were previously impossible. Additionally, our findings provide crucial guidance for combating stinknet, a rapidly spreading invasive plant that is emerging as a new threat to drylands across the southwestern United States. By making biocrust restoration more economically viable, this research can accelerate efforts to stabilize soils, reduce dust storms, sequester carbon and restore ecosystem function across degraded drylands worldwide, benefiting both natural ecosystems and societies that depend on them.
Introduction
Drylands are vulnerable to ecosystem state changes that leave these ecosystems in degraded conditions, often triggered by land use or anthropogenic disturbance regimes (Bestelmeyer et al., Reference Bestelmeyer, Okin, Duniway, Archer, Sayre, Williamson and Herrick2015). Degradation of drylands is a major problem for human society because they sustain billions of people and collectively comprise nearly half of the land surface and house much of the world’s agriculture and pastoralism (Safriel and Zafar, Reference Safriel, Zafar, El-Kassas and Ezcurra2005; Maestre et al., Reference Maestre, Le Bagousse-Pinguet, Delgado-Baquerizo, Eldridge, Saiz, Berdugo, Gozalo, Ochoa, Guirado, García-Gómez, Valencia, Gaitán, Asensio, Mendoza, Plaza, Díaz-Martínez, Rey, Hu, He, Wang, Lehmann, Rillig, Cesarz, Eisenhauer, Martínez-Valderrama, Moreno-Jiménez, Sala, Abedi, Ahmadian, Alados, Aramayo, Amghar, Arredondo, Ahumada, Bahalkeh, Ben Salem, Blaum, Boldgiv, Bowker, Bran, Bu, Canessa, Castillo-Monroy, Castro, Castro, Castro-Quezada, Chibani, Conceição, Currier, Darrouzet-Nardi, Deák, Donoso, Dougill, Durán, Erdenetsetseg, Espinosa, Fajardo, Farzam, Ferrante, Frank, Fraser, Gherardi, Greenville, Guerra, Gusmán-Montalvan, Hernández-Hernández, Hölzel, Huber-Sannwald, Hughes, Jadán-Maza, Jeltsch, Jentsch, Kaseke, Köbel, Koopman, Leder, Linstädter, le Roux, Li, Liancourt, Liu, Louw, Maggs-Kölling, Makhalanyane, Issa, Manzaneda, Marais, Mora, Moreno, Munson, Nunes, Oliva, Oñatibia, Peter, Pivari, Pueyo, Quiroga, Rahmanian, Reed, Rey, Richard, Rodríguez, Rolo, Rubalcaba, Ruppert, Salah, Schuchardt, Spann, Stavi, Stephens, Swemmer, Teixido, Thomas, Throop, Tielbörger, Travers, Val, Valkó, van den Brink, Ayuso, Velbert, Wamiti, Wang, Wang, Wardle, Yahdjian, Zaady, Zhang, Zhou, Singh and Gross2022; Elouafi, Reference Elouafi2025). Various threats contribute to the dryland degradation challenge, including overgrazing, exotic invasive species, land conversions (to agriculture, energy developments or urban areas), altered disturbance cycles (e.g., fire) and resource extraction. Ecological restoration is one way to combat, decelerate or reverse dryland degradation, but can be challenging because drylands are, by definition, water-limited (Shackelford et al., Reference Shackelford, Paterno, Winkler, Erickson, Leger, Svejcar, Breed, Faist, Harrison, Curran, Guo, Kirmer, Law, Mganga, Munson, Porensky, Quiroga, Török, Wainwright, Abdullahi, Bahm, Ballenger, Barger, Baughman, Becker, Lucas-Borja, Boyd, Burton, Burton, Calleja, Carrick, Caruana, Clements, Davies, Deák, Drake, Dullau, Eldridge, Espeland, Farrell, Fick, Garbowski, de la Riva, Golos, Grey, Heydenrych, Holmes, James, Jonas-Bratten, Kiss, Kramer, Larson, Lorite, Mayence, Merino-Martín, Miglécz, Milton, Monaco, Montalvo, Navarro-Cano, Paschke, Peri, Pokorny, Rinella, Saayman, Schantz, Parkhurst, Seabloom, Stuble, Uselman, Valkó, Veblen, Wilson, Wong, Xu and Suding2021).
Increasingly, dryland restoration ecologists are investigating the potential of biological soil crusts (biocrusts) to help reverse dryland degradation. Biocrusts are thin, cohesive and photosynthetic soil surface layers built and inhabited by biota highly adapted to aridity and drought, commonly including cyanobacteria, lichens and mosses (Weber et al., Reference Weber, Belnap, Büdel, Antoninka, Barger, Chaudhary, Darrouzet-Nardi, Eldridge, Faist, Ferrenberg, Havrilla, Huber-Sannwald, Malam Issa, Maestre, Reed, Rodriguez-Caballero, Tucker, Young, Zhang, Zhao, Zhou and Bowker2022). Biocrusts protect dryland soil from erosion (Rodriguez-Caballero et al., Reference Rodriguez-Caballero, Stanelle, Egerer, Cheng, Su, Canton, Belnap, Andreae, Tegen, Reick, Pöschl and Weber2022), build soil fertility (Elbert et al., Reference Elbert, Weber, Burrows, Steinkamp, Büdel, Andreae and Pöschl2012), strongly influence local hydrology and water balance (Li et al., Reference Li, Bowker and Xiao2022) and shape other biotic communities (Bacovcin et al., Reference Bacovcin, McIntyre and Havrilla2025). Biocrust biota are being grown as bio-inoculants for use in degraded drylands, but the modes of production and field deployment are still under active investigation (Antoninka et al., Reference Antoninka, Faist, Rodriguez-Caballero, Young, Chaudhary, Condon and Pyke2020; Roncero-Ramos et al., Reference Roncero-Ramos, Román, Acién and Cantón2022; Heredia-Velásquez et al., Reference Heredia-Velásquez, Giraldo-Silva, Nelson, Bethany, Kut, González-de-Salceda and Garcia-Pichel2023; Bailey et al., Reference Bailey, Lauria, Gugel, Mayorga, Anenberg, Griffen, Condon, Doherty, Tucker, Ramsey, Reed, Antoninka, Durham, Cooke, Gibson, Olney, Charley, Covington, Nelson, Gonzalez, Pablo, Bia, Sloan, Kebede, Fule, Grover, Dohrenwend and Bowker2026).
One successful bio-inoculant approach has been to grow biocrusts with irrigation to form biocrust “sods” (Antoninka et al., Reference Antoninka, Rowe, Weser, Langenfeld, Brady, Sprague and Day2024; Olney, Reference Olney2025; Bailey et al., Reference Bailey, Lauria, Gugel, Mayorga, Anenberg, Griffen, Condon, Doherty, Tucker, Ramsey, Reed, Antoninka, Durham, Cooke, Gibson, Olney, Charley, Covington, Nelson, Gonzalez, Pablo, Bia, Sloan, Kebede, Fule, Grover, Dohrenwend and Bowker2026) and transfer them to the field (Rowe et al., Reference Rowe, Antoninka, Langenfeld, Brady, Sprague, Fastiggi, Kollath, Ramsey and Barker2026). Biocrust sods are patches of biocrust grown in a thin layer over fabrics ex situ (e.g., in farms) that can be rolled, transported and unfurled at sites undergoing ecological restoration. However, one limitation to this practice is that in the process of farming sods with irrigation, invasive non-native plants from the seedbank of the biocrust salvage site or dispersed by wind onto the growing sods may also thrive. Thus, weed control is an essential quality control step in biocrust sod farming in some sites.
In recent biocrust “sod” production, the most prevalent invasive, non-native plant on the sods has been Oncosiphon pilulifer (stinknet), a winter annual forb. Stinknet has been present in the southwestern United States since the 1980s in California and the 1990s in Arizona, but has become a widespread invasive more recently, becoming listed as an Arizona Noxious Weed in 2020 (Hedrick and McDonald, Reference Hedrick and McDonald2020). It is likely to spread across more of the southwest United States, including the Mojave Desert (Hedrick and McDonald, Reference Hedrick and McDonald2020). Dense stands formed by stinknet reduce native plant diversity, threatening critical habitat for sensitive species, and are unpalatable for cattle and likely wildlife (Rutherford and Powrie, Reference Rutherford and Powrie2010; McDonald et al., Reference McDonald, Larios and Rodriguez2023). In previous sod farming efforts, we clipped plants by hand to prevent them from seeding because pulling out roots could have damaged sods. This practice was both overly labor-intensive and did not guarantee a weed-free product since the invasive annuals can regrow from roots, especially in the case of a late spring precipitation event. Because we transfer sods to natural areas, it is of particular urgency that the sods do not become a seed source for invasive species in restoration sites.
An alternative strategy for controlling stinknet and other invasive plants without disturbing the soil is herbicides, but the effects of herbicides on biocrusts have not been fully documented. Published studies on the subject found positive (Youtie et al., Reference Youtie, Ponzetti, Salzer and Freudenberger1999), minimal (Bailey et al., Reference Bailey, Antoninka, Kobelt, Poff and Bowker2025), negative (Zaady et al., Reference Zaady, Levacov and Shachak2004) or neutral (Condon and Gray, Reference Condon and Gray2020; Slate et al., Reference Slate, Durham, Casper, Mummey, Ramsey and Pearson2023) effects of herbicides on biocrusts. Bailey et al. (Reference Bailey, Antoninka, Kobelt, Poff and Bowker2025) found that mosses were reduced by glyphosate, a post-emergent herbicide, but not aminopyralid, which can be applied as a pre- or post-emergent herbicide. Lichens were not affected by any of the herbicides tested (Bailey et al., Reference Bailey, Antoninka, Kobelt, Poff and Bowker2025). Similarly, glyphosate had no negative effects on bryophyte cover, but may have indirectly improved growth by reducing competition with plants for space and light compared with the controls (Youtie et al., Reference Youtie, Ponzetti, Salzer and Freudenberger1999; Zaady et al., Reference Zaady, Eldridge and Bowker2016). However, the application of the preemergent herbicide, simazine, decreased chlorophyll levels and moss density and had residual effects 16 years after treatment (Zaady et al., Reference Zaady, Levacov and Shachak2004, Reference Zaady, Arbel, Barkai and Sarig2013, Reference Zaady, Eldridge and Bowker2016). Finally, grass-specific imazapic and shrub-specific tebuthiuron, applied as preemergent herbicides, did not significantly change lichen or moss cover across study sites (Condon and Gray, Reference Condon and Gray2020). The evaluation of herbicide effects on late successional biocrust taxa is important because of their distinct functional roles. Early colonizers often include lightly pigmented bundle-forming filamentous cyanobacteria that knit together the soil surface (Belnap et al., Reference Belnap, Phillips, Witwicki and Miller2008b). Once these taxa have proliferated, another suite of darkly pigmented cyanobacterial taxa colonize the upper surface of the biocrust, giving it a gray to black appearance (Weber et al., Reference Weber, Bowker, Zhang, Belnap, Weber, Budel and Belnap2016). These taxa shield the entire community from ultraviolet radiation with their sunscreen pigments (Belnap et al., Reference Belnap, Phillips, Flint, Money and Caldwell2008a). Also, many of these dark-pigmented taxa are nitrogen fixers (Belnap, Reference Belnap2002), and their dominance coincides with altered hydrological function (Belnap et al., Reference Belnap, Wilcox, Van Scoyoc and Phillips2013). If the environment permits, other groups of biota, such as lichens or mosses, may colonize and become major community members late in the successional process (Dojani et al., Reference Dojani, Büdel, Deutschewitz and Weber2011). Until now, the effects of herbicides on Sonoran Desert biocrusts, typically dominated by cyanobacteria and lichens, have not been studied. Furthermore, herbicides have not been tested on any biocrust sod farm in which biocrusts are actively growing.
Perhaps owing to its recent arrival in the southwest, few herbicide trials on stinknet have been conducted. However, one extension study reported that clopyralid, aminopyralid, glyphosate and aminopyralid + triclopyr provide >90% control of stinknet in California, reporting 1-year control with clopyralid and aminopyralid (McDonald, Reference McDonald2019). Indaziflam has also been suggested as an effective preemergent herbicide (McDonald et al., Reference McDonald, Larios and Rodriguez2023). Of these herbicides, the effects of aminopyralid and glyphosate on biocrusts have been tested (Bailey et al., Reference Bailey, Antoninka, Kobelt, Poff and Bowker2025). Aminopyralid is a plant growth regulator and belongs to the Group 4 herbicides (synthetic auxins); when absorbed, it deregulates metabolic pathways, causing abnormal plant growth through uneven cell division and growth (Beyond Pesticides, Reference Pesticides2011). Aminopyralid selectively targets broadleaf forbs through uptake by plant foliage and translocation through the plant and can act as a preemergent herbicide as seedlings emerge from the soil (Ratnayake et al., Reference Ratnayake, Prieto and Smearman2013). Glyphosate is a widely available, nonselective, broad-spectrum Group 9 herbicide that works by inhibiting the synthesis of amino acids and is rapidly inactivated and immobilized by adsorption to soil (Beyond Pesticides, Reference Pesticides2011; Control Solutions Inc., 2012). Although Bailey et al. (Reference Bailey, Antoninka, Kobelt, Poff and Bowker2025) found adverse effects on moss by glyphosate, we included it because of its availability and widespread use. Finally, Indaziflam is a group 29 preemergent herbicide that inhibits cellulose biosynthesis (Neal, Reference Neal2023). When applied to the soils, it creates a barrier that prevents germination of grasses, annual sedges and broadleaf herbs by preventing cell wall formation, which is crucial for plant growth (Bayer CropScience, 2020; Chemical Warehouse, 2025). It provides extended residual control and has a low use rate, which makes it economical (Bayer CropScience, 2020).
We experimentally grew biocrust sods to test the efficacy and cost of two preemergent and two post-emergent herbicides, hand clipping and no treatment (control) for controlling stinknet while monitoring the effects on the growing biocrusts. Therefore, this study represents a novel study of both an herbicide trial for stinknet and a test of the toxicity of those herbicides to biocrust. We addressed the questions: Which combinations of herbicide and application method (pre- or post-emergent) effectively control stinknet and are most compatible with biocrust sod production? Are herbicides a more economical method for weed control in sod farms than manual control? Do herbicide-treated biocrust sods retain toxicity to plants 2–4 months after application? Answering these questions could lead to significant upgrades in efficiency in biocrust sod production, enabling wider use and adoption of this promising technique.
Methods
Study site
We performed the sod cultivation and herbicide trial on the grounds of Scottsdale Community College (33.513788° N, 111.879778° W; Arizona, USA). The average precipitation for this Sonoran Desert site is 220.15 mm. The temperature averages 22.9°C with highs reaching over 40°C in the summer (1991–2020 Normals, PRISM data downloaded 8-11-2025). The high temperatures ranged from 12.2°C to 36.7°C during the study period (PRISM data downloaded 8-11-2025). The site was cleared of debris and leveled for a previous experiment (Antoninka et al., Reference Antoninka, Rowe, Weser, Langenfeld, Brady, Sprague and Day2024). The site was dominated by the previous year’s stinknet (O. pilulifer) standing crop. In late November (23), 2024, we set up 60 miniature (0.25 m2) biocrust sods using previously proven methods (Antoninka et al., Reference Antoninka, Rowe, Weser, Langenfeld, Brady, Sprague and Day2024; Rowe et al., Reference Rowe, Antoninka, Langenfeld, Brady, Sprague, Fastiggi, Kollath, Ramsey and Barker2026) with a 50 cm buffer between plots. Briefly, we set up plots with the following elements on top of a heavy plastic 20-year weed-blocker: (1) 50 × 50 cm biodegradable weed cloth (sheets of maize-derived poly-lactid acid, Vigoro), (2) 1.25 L of substrate (Quikrete graded and washed coarse sand) spread out thinly for each 0.25 m2 plot, (3) 50 × 50 cm fine woven jute burlap fabric, (4) biocrust inoculum sieved through 0.4 cm mesh and applied evenly over the burlap at a calculated rate of 40% cover of the surface area to 0.5 cm depth (100 mL per unit by volume), (5) 7.4 mL stinknet seed collected from the plants on site. Biocrust inoculum was salvaged before the construction of the Pima-Dynamite Trailhead of the nearby McDowell Sonoran Preserve (33.7464, −111.8883) before trailhead construction in November 2022 and stored in the dark at ambient temperatures (mean annual temp 24.1°C). Layers were held in place with four landscape staples in the corners of each plot. To sustain nearly continuous hydration to maintain active biocrusts during daylight hours, we created a 50 cm length loop of ¼ inch drip tape (emitters every 6 inches) placed over each sod. This supplied complete irrigation coverage across the sods. We irrigated 7× per day (every 2 hours) until April 3, 2025, for 3-minute intervals during daylight hours from 6 am to 6 pm, which resulted in near-saturation. Some water was lost between pulses to evaporation, but not enough to dessicate the biota. Finally, we laid 62% tan shade cloth flat over the length of the plots and attached it to the ground with ground staples outside of the sods.
Experimental design and treatment applications
We delineated 10 linear blocks with six plots each to control for any unevenness in irrigation or position, then randomly assigned one of the six treatments to each plot within each block in a full factorial, random block design. The treatments included four herbicide applications using rates recommended on the label (5 mL/10 L aminopyralid [preemergent]; 5 mL/10 L aminopyralid [post-emergent]; 1.225 ml/3 L Indaziflam [preemergent, Esplanade]; 3% Glyphosate [post-emergent]), hand cutting, or no removal (control). To avoid overspray, a cardboard cutout was placed around the plot, then we sprayed herbicide evenly across the plot for 10 seconds at a spray rate that dispensed a total of 150 mL per plot. Preemergent herbicide was sprayed within 48 hours after initial plot setup. Post-emergent herbicide was sprayed early in the season when stinknet covered about 50% of the plots on January 17, 2025 (Figure 1B,C), because post-emergent herbicides are considered most effective when applied to young, actively growing stinknet plants (Chamberland, Reference Chamberland2020). We had expected to conduct a second spray, but there was too little additional growth (<10% cover on post-emergent herbicide plots). We used 5 cm-long scissors in the “hand-cut” plots to trim all plants to 2 cm in early February and early April, when stinknet in plots reached a height of at least 15 cm or began to flower.
Stinknet cover means by stinknet removal treatment estimated almost two months after seeding (A) and photos taken on January 16, 2025, prior to application of post-emergent herbicides (B-D). Filled bars indicate the mean and error bars indicate one standard error of the mean. Photos show one row of biocrust sods (B), stinknet growth on the untreated sods prior to the application of post-emergent herbicides (C), and sod with a pre-emergent indaziflam treatment (D). Photo credit: A. Wiggins.

In mid-March (March 14), we broadcast a native seed mixture (Table 1) on each plot as a bioassay of remaining toxicity from herbicide applications. We selected seeded plant species based on seed availability and the establishment success of the species in previous experiments (Rowe et al., Reference Rowe, Sprague, Ball, Langenfeld and Rivera2022; Lyu et al., Reference Lyu, Rowe, Broatch, Brady, Fastiggi, Fitts, Langenfeld and Munson202 Reference Bailey, Lauria, Gugel, Mayorga, Anenberg, Griffen, Condon, Doherty, Tucker, Ramsey, Reed, Antoninka, Durham, Cooke, Gibson, Olney, Charley, Covington, Nelson, Gonzalez, Pablo, Bia, Sloan, Kebede, Fule, Grover, Dohrenwend and Bowker6). We also grew some of each species in the greenhouse to facilitate plant ID in the field.
Amounts of native seed added to each plot

Note: Seeds were purchased from Borderlands Nursery and Seed (Patagonia, AZ) in March 2025.
Plant and biocrust sampling
On January 16 (before applying post emergent herbicide), we visually estimated stinknet cover using 10 cover class estimates (Peet et al., Reference Peet, Wentworth and White1998). At the end of the experiment (April 3), we visually estimated total plant cover and stinknet cover using the same cover classes and counted the number of individuals of native seeded species. After plant sampling, we cut the plants in the plots to the ground to enable biocrust sampling.
To sample biocrust, we centered a 30 × 30 cm quadrat frame in each plot and used a gridded point intercept quadrat sampling method (25 intersections per quadrat). The categories used in these procedures were: bare soil, cyanobacteria (light and dark), lichen, moss, jute, litter and gravel. If there was biocrust at the point of the pin, even if under litter or jute, it was recorded. Biocrust taxa in the plot were recorded as trace (2% cover, the mid-point between 0 and the 4% resolution of our method) if visibly present but not captured as a point interception.
Statistical analysis and cost accounting
To evaluate the effect of the treatments on the response variables biocrust cover, stinknet cover, total native seedling establishment (counts) and bare ground cover, we used a linear mixed model with treatments (six levels) and block as the random effect. Biocrust cover was analyzed in three ways: as total cover of all categories of biocrust, light cyanobacteria and as the sum of dark-pigmented cyanobacteria, mosses, or lichens (typical late colonizers and indicators of a high degree of development; hereafter “late successional”). Although total biocrust is not independent of the other variables, we included it as a summary variable across the biocrust constituents. Although we estimated total plant cover and stinknet cover separately, the difference between the two estimates was negligible (<3% total), so we report only stinknet cover. We checked model assumptions with residuals and used a rank transformation for stinknet cover and total seedling count, and a square root transformation for non-biocrust cover and bare ground to normalize the data. We used Tukey’s test for analyzing post hoc differences among treatment groups. We used JMP Pro 18 (JMP Statistical Discovery LLC, Cary, NC, USA) for all statistical analysis and graphs.
To compare costs across treatments, we added together all labor and supply costs based on the per-plot scale and then scaled up to a larger management scale of 0.2 ha (~1 acre). We estimated the labor time of applying treatments by taking the mean time of treatment application in the 25 m2 plot and scaling that to 0.2 ha (multiplier = 10,000). To the timed labor, we added fixed labor time associated with herbicide preparation and application. We estimated mixing the herbicide at the beginning of the day and cleaning the equipment at the end of the day as 1 hour, and added 10 minutes for every refill required for a 4-gallon herbicide backpack sprayer for a 0.2 ha area. We multiplied the labor time by $19.16/hour based on the average pesticide applicator wage (https://www.ziprecruiter.com/Salaries/Pesticide-Applicator-Salary--in-Arizona, accessed 11-18-2025) to derive labor costs. We calculated supply costs for 0.2 ha based on actual supplies (herbicides, scissors) used in the 25 m2 plots and applied the scaling multiplier. Supply costs and labor costs were summed for a total cost of each treatment at 0.2 ha scale.
Results
After two months, stinknet had grown to a mean cover of 49.5 ± 5.5% in untreated plots and 0.35 ± 0.13% in the preemergent herbicide treatments (Figure 1A). At the end of the experiment in early April, all herbicides controlled stinknet more than the control treatments (Figure 2). Indaziflam controlled stinknet more than the other herbicides, except glyphosate (Figure 2A). Native seedlings from the seed mix established best with the two herbicides applied as post-emergent compared to the other treatments, except the preemergent application of aminopyralid (Figure 2A).
Total biocrust, light cyanobacteria, late successional biocrust (dark cyanobacteria, lichen, and moss), stinknet, and bare ground cover and total native seedlings means by stinknet removal treatment estimated at the end of the experiment, April 2025 (A). Photos of hand-cut treatment plot (B) and sod row (treatments from forefront: post-emergent aminopyralid, pre-emergent aminopyralid, and hand cut) (C) at the end of the experiment, prior to the final hand cut treatment, April 1, 2025. Different letters within the y axis response variables indicate differences among means based on Tukey’s post hoc test. Filled bars indicate the mean and error bars indicate one standard error of the mean. Photo credit: A. Wiggins.

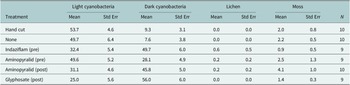
Total biocrust was higher in all herbicide treatments compared to no treatment (control) and was not different from the hand cut treatment (Figure 2A). Light cyanobacteria were lower in plots with glyphosate herbicide compared with no herbicide treatment plots, but no differences were detected among the preemergent herbicide treatments and the no herbicide treatment plots (Figure 2A). Late successional biocrust grew more in plots with herbicide compared with the no herbicide treatment plots (hand cut or control; Figure 2A). Lichens were not found on control treatment plots and rarely on herbicide-treated plots (Table 2). All herbicide treatments had higher bare ground than the no herbicide treatments (Figure 2A).
Means and standard errors for biocrust cover of the taxonomic groups by treatment

Note: Pre and post refer to whether the herbicide was applied as a pre- or post-emergent herbicide. N is the number of plots included in each treatment.
The hand-cut treatment took 0.04 ± 0.00 decimal hours (mean ± se) for the first treatment (February 10) and 0.70 ± 28.39 decimal hours for the second treatment (April 2), for a total of 0.11 ± 0.02 decimal hours per plot or 1.07 decimal hours across the 10 hand-cut plots. By contrast, the herbicide treatment took 0.003 decimal hours (10 seconds) in each plot or 0.03 decimal hours across the 10 replicate plots for each herbicide treatment. Thus, hand-cutting is a factor of 38.6 times more time-consuming at the plot scale. When we scaled up these costs to the application of 0.2 ha of sods and added supply costs and fixed labor costs (time for mixing chemicals and cleaning the sprayer tanks afterwards), the hand-cut treatment was still the most expensive treatment (Table 3). Glyphosate was the most expensive herbicide application and the only herbicide to have a significantly inhibitory effect on light cyanobacteria compared with both controls (Table 3 and Figure 2). Overall, all herbicide treatments were better than the hand-cut treatment in terms of cost, seedling germination, and total and late successional biocrust (Table 3 and Figure 2). Only the post-emergent herbicides aminopyralid and glyphosate resulted in less-light cyanobacteria than the hand-cut treatment (Figure 2).
Effort and supply costs for each treatment at the management scale of 0.2 ha, based on the per-plot time and supply costs
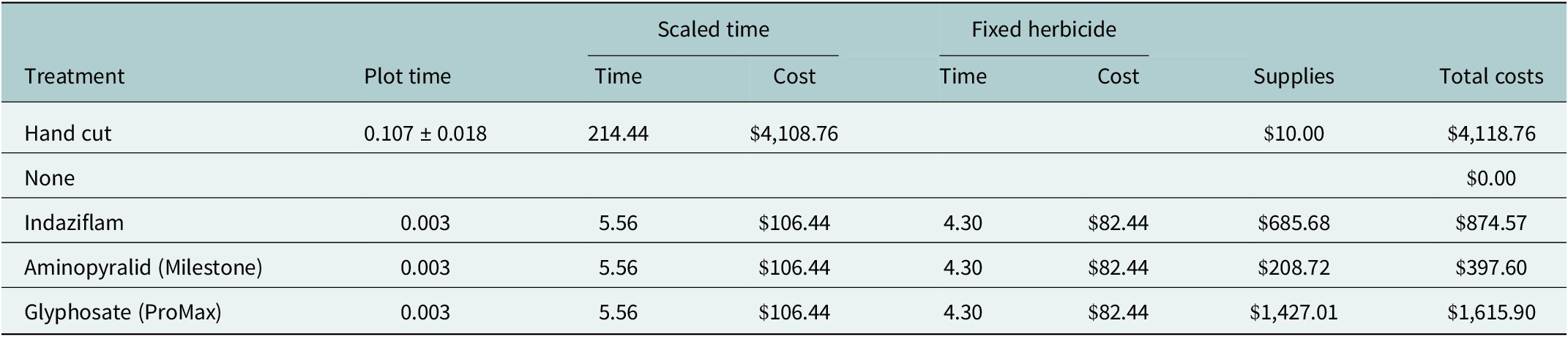
Note: Time and costs are reported in decimal hours and US dollars, respectively. Column headings are abbreviated as follows: “Plot time” indicates the application time of each treatment per 25 m2 plot and is reported as mean ± se. “Scaled time” includes the labor costs (time and cost) of the treatment application based on the plot time scaled up to 0.2 ha; “Fixed herbicide” includes preparation costs (time and cost) of 1 hour for mixing the herbicide at the beginning of the day and cleaning the equipment at the end of the day, plus 10 minutes for every refill required for a 4-gallon herbicide backpack sprayer, “Supplies” includes the scaled up costs of the supplies for each treatment (materials for hand-cut treatments are scissors, price shown is for a two-pack). “Total costs” is the sum of the scaled time cost, fixed herbicide cost and supply cost for 0.2 ha.
Discussion
In this study, we demonstrated that multiple herbicides outperform hand cutting as a tactic for invasive plant control during biocrust sod production. Further, hand-cutting methods were 3–12 times more costly than herbicide applications for controlling invasive plants. None of the herbicides affected total or late successional biocrusts negatively, relative to hand cutting or no control of stinknet, but some herbicides allowed greater biocrust growth than others. Finally, the various herbicides differed in their residual toxicity for plant germination of the native seed mix at the end of the biocrust sod farming period. Overall, accounting for cost, control of invasives, effects on biocrusts and residual toxicity, Indaziflam, applied as a preemergent, and aminopyralid, applied as a post-emergent, emerged as the best options to keep biocrust sods free of stinknet in our study system.
Herbicides provide a higher-quality and less expensive biocrust sod product
Herbicide treatments appear to be a promising way to scale up weed-free biocrust sod production and even improve late successional biocrust development compared with hand cutting and no treatment. High stinknet cover in both the hand-cut and control treatments corresponded with lower late successional biocrust cover, likely by outcompeting biocrust for light and space and creating a disturbed environment. While researchers have actively studied the effects of biocrust on plants (Godínez-Alvarez et al., Reference Godínez-Alvarez, Morín and Rivera-Aguilar2012; Havrilla et al., Reference Havrilla, Chaudhary, Ferrenberg, Antoninka, Belnap, Bowker, Eldridge, Faist, Huber-Sannwald, Leslie, Rodriguez-Caballero, Zhang and Barger2019), the reversal has not received much attention. Biocrusts are primarily found in less productive ecosystems (e.g., drylands) in which vascular plants cannot attain complete canopy coverage (Weber et al., Reference Weber, Belnap, Büdel, Antoninka, Barger, Chaudhary, Darrouzet-Nardi, Eldridge, Faist, Ferrenberg, Havrilla, Huber-Sannwald, Malam Issa, Maestre, Reed, Rodriguez-Caballero, Tucker, Young, Zhang, Zhao, Zhou and Bowker2022). Therefore, it stands to reason that biocrusts were reduced by the dense and near-complete stinket cover in the control plots. Similarly, we found a relationship in which at certain plant and biocrust cover values, there was a positive growth relationship, but higher levels resulted in a negative growth relationship (Rowe et al., Reference Rowe, Antoninka, Langenfeld, Brady, Sprague, Fastiggi, Kollath, Ramsey and Barker2026). The dense stinknet displaced the area that otherwise could have been occupied by biocrusts. It is also possible that the ground disturbance caused by stinknet emergence and growth slowed biocrust development. It is important to note that we intentionally seeded stinknet into our experiment, creating dense mats of stinknet in untreated controls and disrupting the biocrust surface, which possibly exaggerated the differences between herbicide treatments and the hand-cut alternatives that might have been seen had we not seeded. This worst-case scenario simulates a farm site with extreme invasive plant pressure.
As observed in some other recent studies (Condon and Gray, Reference Condon and Gray2020), herbicides did not thwart biocrust development on the sods. Rather, biocrusts were able to progress beyond dominance by early colonizers. Continuous hydration farming combined with the weed control provided by all herbicide treatments allowed the development of a high coverage of dark cyanobacterial biocrusts in a few months; in nature, this transition would require years to decades.
Cyanobacteria and lichens tend to dominate Sonoran Desert biocrusts more than mosses (Rosentreter and Belnap, Reference Rosentreter, Belnap, Belnap and Lange2001), in contrast to cooler semi-arid regions, possibly because mosses prefer longer hydration periods relative to the other groups (Antoninka et al., Reference Antoninka, Bowker, Reed and Doherty2016; Bowker and Antoninka, Reference Bowker and Antoninka2016; Ayuso et al., Reference Ayuso, Giraldo-Silva, Barger and Garcia-Pichel2020). Thus, a biocrust sod dominated by a mixture of cyanobacteria and lichens would be an appropriate biocrust restoration product for many Sonoran Desert ecosystems. Earlier sod-farming efforts at the same farm using the same initial inoculum source achieved 1–3% moss cover and 1–2% lichen cover in the comparable jute and sand quesadilla treatment (Antoninka et al., Reference Antoninka, Rowe, Weser, Langenfeld, Brady, Sprague and Day2024). However, in this study, although moss levels were similar, lichen abundance was very low. We cannot determine whether the difference is due to the use of herbicides, climate differences between the two growing years, the age of the inoculum, or another factor. Based on an ongoing long-term biocrust storage experiment we are conducting, lichen growth began to decline after 3 years of storage (unpublished data). Bailey et al. (Reference Bailey, Antoninka, Kobelt, Poff and Bowker2025) tested biocrust response to herbicides and found that mosses were most sensitive to some herbicides (of those herbicides studied here, glyphosate reduced moss growth, but not aminopyralid). Lichens were not affected by any of the herbicides (Bailey et al., Reference Bailey, Antoninka, Kobelt, Poff and Bowker2025). However, other studies in areas where moss biocrusts are prevalent have not found negative effects on biocrusts (Condon and Gray, Reference Condon and Gray2020; Slate et al., Reference Slate, Durham, Casper, Mummey, Ramsey and Pearson2023). As herbicide use in biocrust farming operations becomes more common, it will be important to definitively demonstrate that the herbicides do not slow or prevent moss or lichen establishment, especially in areas where mosses are a more important component of the biocrust community.
Considerations for choosing the right herbicide
Choosing which herbicide to apply in biocrust-producing farms depends on multiple factors, including maximal effectiveness against target plants, minimal inhibition of biocrust development, costs of the product and labor and desired end use of the sods. The choice may differ by location because these factors might vary. While the other factors are self-explanatory, the desired end use of the sods is a factor because, in some cases, it will be desirable for the sods to be colonized by native plants, and in other scenarios, it may not. Therefore, the residual toxicity of the herbicides after the biocrusts have grown to a high coverage and level of development becomes important.
In some cases, such as erosion control, the goal may be to combine the effect of biocrust sods with plant growth to stabilize soils, for example, after a wildfire or construction. Biocrust sods can be produced to be permeable to vascular plants either by omitting or removing the weed-barrier base (Bailey et al., Reference Bailey, Lauria, Gugel, Mayorga, Anenberg, Griffen, Condon, Doherty, Tucker, Ramsey, Reed, Antoninka, Durham, Cooke, Gibson, Olney, Charley, Covington, Nelson, Gonzalez, Pablo, Bia, Sloan, Kebede, Fule, Grover, Dohrenwend and Bowker2026). Residual toxicity of herbicides used in sod production would be counterproductive. In this case, the best and most cost-effective herbicide treatment of those studied may be aminopyralid (milestone). As a preemergent, aminopyralid was not as effective in controlling stinknet and suppressed late successional biocrust cover more than the other herbicide treatments. But when applied as a post-emergent herbicide, it resulted in the highest late successional and light cyanobacterial biocrust cover, effective stinknet control (but not as effective as indaziflam) and the best rate of native seedling establishment. Alternatively, if the management goal is fire suppression, weed-blocking biocrust sods with weed-barrier bases to inhibit invasive plants that serve as fine fuels and enhance fire spread may be the best strategy. In such cases, residual toxicity of herbicides, such as with indaziflam, would enhance the ability of biocrust sods to inhibit flammable plant biomass (exotic or native). Although more than twice as expensive as aminopyralid, indaziflam was the most effective at preventing stinknet emergence, but also at suppressing native seedlings. Plots with indaziflam were high in late successional biocrust cover and had no detectable differences with other treatments in terms of light cyanobacteria cover.
New weapons to combat new invaders
Stinknet is a relatively new invader in the southwestern United States. Because of the alarming speed of its spread, it has already become one of the most concerning invasives in the Sonoran Desert region of Arizona, and is poised to spread to neighboring ecoregions and new states in the United States and Mexico (Hedrick and McDonald, Reference Hedrick and McDonald2020). Our study is one of the first herbicide trials on stinknet. One unpublished study reported that preemergent indaziflam and aminopyralid were much more effective at controlling stinknet than post-emergent herbicides (Rodriguez and Larios, Reference Rodriguez and Larios2021). Our study supports the high degree of efficacy of preemergent indaziflam, but we did not detect differences between stinknet cover treated with indaziflam compared to post-emergent glyphosate. The difference between the two studies’ findings may be attributed to the application timing. Whereas we applied the post-emergent herbicides early, the previous study sprayed post-emergent herbicides when stinknet was starting to bud, and some of the plant was flowering (Rodriguez and Larios, Reference Rodriguez and Larios2021). Although Glyphosate (Roundup) had similar effects on stinknet as aminopyralid applied as a post-emergent and with indaziflam, and is widely available, its drawback is that it is 4× as expensive as aminopyralid when scaled to 0.2 ha. The previous study and ours supported different findings related to aminopyralid. In the previous study, aminopyralid applied as a pre-emergent was one of the most effective treatments (Rodriguez and Larios, Reference Rodriguez and Larios2021). However, although we applied aminopyralid at the same rate as their study, it was not as effective as indaziflam when applied as a preemergent or a post-emergent. Our study also documents the efficacy of aminopyralid as a post-emergent for control of stinknet, which did substantially reduce stinknet coverage, though it was not one of the best performers. This difference may also be a result of herbicide application timing. Our study provides managers with actionable knowledge of effective herbicide treatments that can be applied to stinknet and will be safe for biocrusts in the field or farm setting. As a result, managers can more effectively integrate biocrust restoration materials into their restoration and weed management plans.
Open peer review
For open peer review materials, please visit http://doi.org/10.1017/dry.2026.10023.
Data availability statement
Data and analysis files will be available on https://github.com/HelenIvy/Biocrust_Stinknet upon publication.
Acknowledgments
The authors are grateful to Scottsdale Community College for providing facilities for the experiment and the McDowell Sonoran Conservancy for providing experimental materials. Darren Olney assisted in the experiment setup. The authors used claude.ai (https://claude.ai/new) to shorten and reformat the abstract to the journal’s specifications; the revision was then edited by the authors.
Author contribution
H.I.R. wrote the first draft, led the data analyses and designed figures. M.B. conceptualized the experiment and contributed to writing. H.I.R. managed the project. J.W. and A.W. applied the treatments. All authors contributed to the field work, revised and approved the final version of the manuscript.
Financial support
This work was funded by the Salt River Project (SRP). Elvy Barton served as our SRP advisor.
Competing interests
The authors declare none.







Comments
No accompanying comment.