Introduction
The general chemical formula of the amphiboles may be written (Hawthorne and Oberti, Reference Hawthorne and Oberti2007a, Reference Hawthorne and Oberti2007b) as
A B2 C5 T8 O22 W2, where
A = □ (vacancy), Na+, K+, Ca2+, Pb2+, Li+;
B = Na+, Ca2+, Mn2+, Fe2+, Mg2+, Li+;
C = Mg2+, Fe2+, Mn2+, Al3+, Fe3+, Mn3+, Cr3+, Ti4+, Li+;
T = Si4+, Al3+, Ti4+, Be2+;
W = (OH), F, Cl, O2–.
Atoms and ions are indicated by Roman symbols (e.g. Si4+, Si), groups of atoms or ions are indicated by upper-case Roman letters (as in the general formula above); crystallographic sites are indicated by italicised symbols, e.g. T(1), M(4); polyhedra are labelled by the central site, e.g. T(1) tetrahedron and M(4) polyhedron. Data are presented here for cummingtonite-P21/m, the structure of which is illustrated in Fig. 1. When discussing the refinement of site occupancies, atoms are written as neutral because neutral scattering factors were used in the crystal-structure refinement.
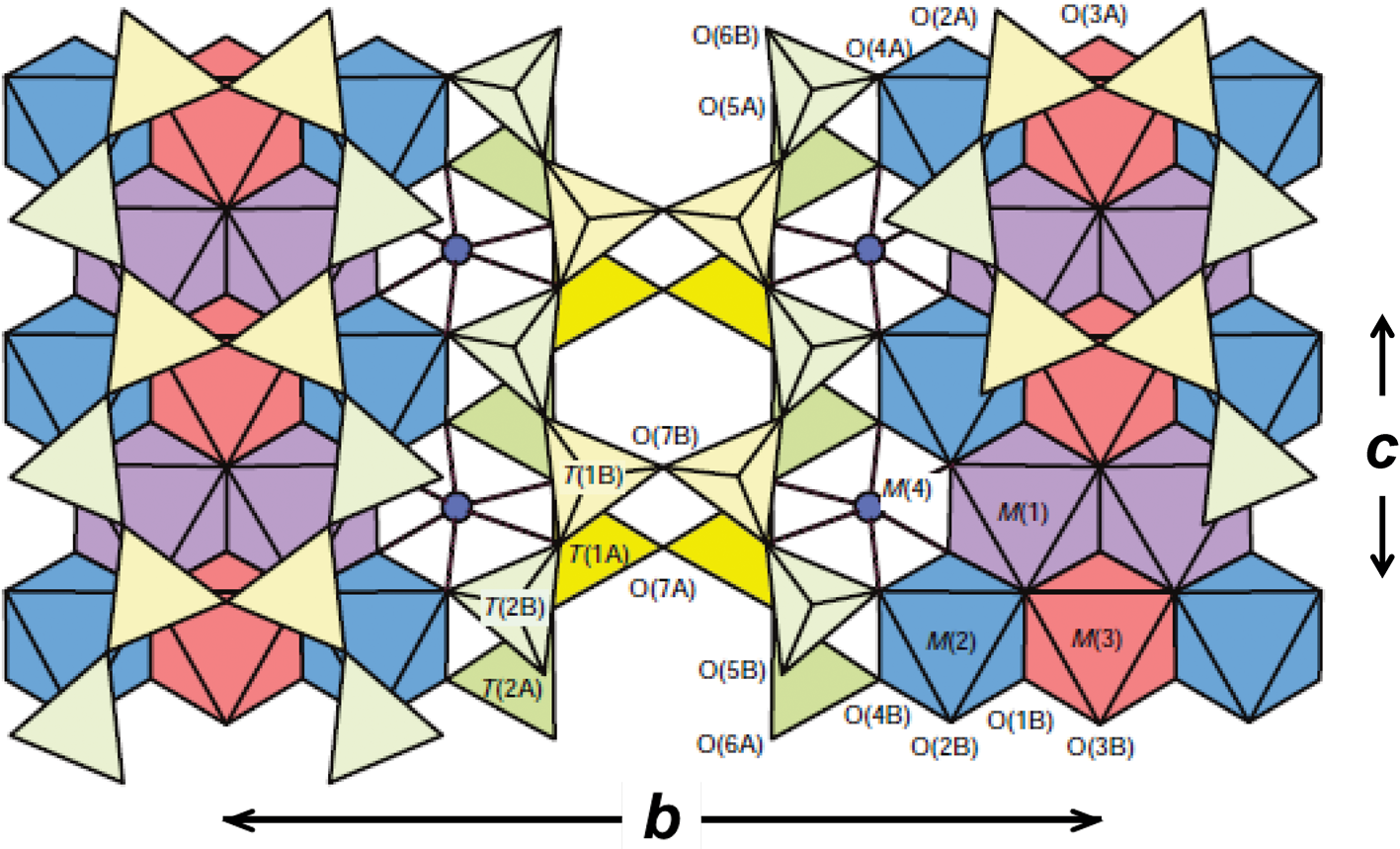
The P21/m amphibole structure projected onto (100); polyhedra: T(1A) = bright yellow, T(2A) = bright green, T(1B) = pale yellow, T(2B) = pale green, M(1) = mauve, M(2) = blue, M(3) = red; sites: M(4) = blue circle. Drawn using ATOMS V6.4.

The Commission on New Minerals, Nomenclature and Classification of the International Mineralogical Association (IMA-CNMNC) rules for naming and classifying amphiboles (Hawthorne et al., Reference Hawthorne, Oberti, Harlow, Maresch, Martin, Schumacher and Welch2012, henceforth referred to as IMA2012) are currently under revision. One of the proposed changes involves the criteria for naming and classifying the magnesium-iron-manganese amphiboles which are currently considered as a single amphibole subgroup. It was well-known at the time that both Mn2+ and Fe2+ are strongly ordered at the M4 and M(4) sites in orthorhombic and monoclinic amphiboles, respectively, but BMg, BFe2+ and BMn2+ were not introduced as classification parameters because of the considerable increase in complexity of the classification and the negative impact that the Amphibole Subcommittee felt that this would have on the petrology community. Any changes to the IMA2012 nomenclature and classification of the magnesium-iron-manganese amphiboles will have several very significant effects and the intent of the current paper is to consider these possible effects prior to any change in nomenclature and classification. The cummingtonite-P21/m described by Kisch (Reference Kisch1969) is ideal for this purpose and a more detailed crystallographic and spectroscopic characterisation is given here.
The P21/m amphibole structure
The P21/m structure is shown in Fig. 1. There are four distinct T-sites, T(1A), T(1B), T(2A) and T(2B), that are tetrahedrally coordinated and link to form two distinct types of double-chain of tetrahedra, the A-chain and the B-chain. Coordination and linkage of these T sites is analogous to that in the C2/m amphibole structure. The A- and B-chains face each other back-to-back across the A cavity, and the B-chain is much more kinked than the A-chain (Fig. 1). There are three distinct M sites that are occupied by the C cations, M(1), M(2) and M(3) with point symmetries 1, 1 and m, respectively. The M(4) site has point symmetry 1 and eight adjacent oxygen atoms. However, the cations occupying this site may not bond to all surrounding oxygen atoms, and the existence of this structure type seems to depend on the bonding requirements of the M(4) cation and the surrounding oxygen atoms.
Some synthetic amphiboles have P21/m symmetry and one synthetic amphibole with ![]() $C\bar 1$ symmetry (Cámara et al., Reference Cámara, Oberti, Della Ventura, Welch and Maresch2004) have been reported. The nature of the P21/m–C2/m transition has been studied in considerable detail: Na(NaMg)Mg5Si8O22(OH)2 (Cámara et al., Reference Cámara, Oberti, Iezzi and Della Ventura2003; Iezzi et al., Reference Iezzi, Della Ventura, Oberti, Cámara and Holtz2004); Na(NaMg)Mg5Si8O22F2 (Cámara et al., Reference Cámara, Oberti and Casati2008); ANaB(LiMg)CMg5Si8O22(OH)2 (Iezzi et al., Reference Iezzi, Tribaudino, Della Ventura, Nestolo and Bellatreccia2005a); Na(NaMg)Mg5Si8O22(OH)2 – Na(LiMg)Mg5Si8O22(OH)2 solid-solutions (Iezzi et al., Reference Iezzi and Tribaudino2006); and Na(NaMg)Mg5Si8O22(OH)2 – Na(NaCa)Mg5Si8O22(OH)2 solid-solutions (Iezzi et al., Reference Iezzi, Della Ventura, Tribaudino, Nemeth, Margiolaki, Cavallo, Gaillard and Behrens2010, Reference Iezzi, Tribaudino, Della Ventura and Margiolaki2011). Welch et al. (Reference Welch, Camara, Della Ventura and Iezzi2007) has reviewed the atomic-scale mechanisms of these transitions in detail.
$C\bar 1$ symmetry (Cámara et al., Reference Cámara, Oberti, Della Ventura, Welch and Maresch2004) have been reported. The nature of the P21/m–C2/m transition has been studied in considerable detail: Na(NaMg)Mg5Si8O22(OH)2 (Cámara et al., Reference Cámara, Oberti, Iezzi and Della Ventura2003; Iezzi et al., Reference Iezzi, Della Ventura, Oberti, Cámara and Holtz2004); Na(NaMg)Mg5Si8O22F2 (Cámara et al., Reference Cámara, Oberti and Casati2008); ANaB(LiMg)CMg5Si8O22(OH)2 (Iezzi et al., Reference Iezzi, Tribaudino, Della Ventura, Nestolo and Bellatreccia2005a); Na(NaMg)Mg5Si8O22(OH)2 – Na(LiMg)Mg5Si8O22(OH)2 solid-solutions (Iezzi et al., Reference Iezzi and Tribaudino2006); and Na(NaMg)Mg5Si8O22(OH)2 – Na(NaCa)Mg5Si8O22(OH)2 solid-solutions (Iezzi et al., Reference Iezzi, Della Ventura, Tribaudino, Nemeth, Margiolaki, Cavallo, Gaillard and Behrens2010, Reference Iezzi, Tribaudino, Della Ventura and Margiolaki2011). Welch et al. (Reference Welch, Camara, Della Ventura and Iezzi2007) has reviewed the atomic-scale mechanisms of these transitions in detail.
Previous work: general
The magnesium-iron-manganese amphiboles are an important subgroup within the hierarchical classification of the amphiboles (Hawthorne, Reference Hawthorne1983; Hawthorne et al., Reference Hawthorne, Oberti, Harlow, Maresch, Martin, Schumacher and Welch2012) and have the general formula A(□,Na) B(Mg, Fe2+,Mn2+) C(Mg,Fe2+,Mn2+,Al,Fe3+) T(Si,Al)8 O22 W(OH)2. They may be orthorhombic and monoclinic with space groups Pnma, Pnmn, C2/m and P21/m (Hawthorne and Oberti, Reference Hawthorne and Oberti2007a) and the more common compositions range from anthophyllite to gedrite (Rabbitt, Reference Rabbitt1948; Robinson and Jaffe, Reference Robinson and Jaffe1969; Robinson et al., Reference Robinson, Ross and Jaffe1971; Hawthorne et al., Reference Hawthorne, Schindler, Abdu, Sokolova, Evans and Ishida2008; Schindler et al., Reference Schindler, Sokolova, Abdu, Hawthorne, Evans and Ishida2008) and from cummingtonite to grunerite (Winchell, Reference Winchell1938; Mueller, Reference Mueller1960; Klein, Reference Klein1964). Extensive crystallographic (Papike and Ross, Reference Papike and Ross1970; Hawthorne et al., Reference Hawthorne, Schindler, Abdu, Sokolova, Evans and Ishida2008; Schindler et al., Reference Schindler, Sokolova, Abdu, Hawthorne, Evans and Ishida2008; Viswanathan and Ghose, Reference Viswanathan and Ghose1965; Klein and Waldbaum, Reference Klein and Waldbaum1967; Hafner and Ghose, Reference Hafner and Ghose1971), optical (Evans and Medenbach, Reference Evans and Medenbach1997) and thermodynamic (Evans and Ghiorso, Reference Evans and Ghiorso1995; Ghiorso et al., Reference Ghiorso, Evans, Hirschmann and Yang1995) work has been done on both orthorhombic and monoclinic structures.
Previous work: P21/m magnesium-iron-manganese amphiboles
Bown (Reference Bown1966) first identified cummingtonite with P21/m symmetry occurring as fine exsolution lamellae in tremolite from the Wight Talc Mine, Adirondacks, New York State, USA. Papike et al. (Reference Papike, Ross and Clark1969) refined the structure of a cummingtonite with P21/m symmetry and stated that “The M(1), M(2) and M(3) sites are virtually filled by Mg in the primitive structure, according to our refinement”, in agreement with an unpublished infrared spectrum, although they give no detailed results of a site-occupancy refinement. Prewitt et al. (Reference Prewitt, Papike and Ross1970) report the structure of the same sample with all Mn2+ and Fe2+ assigned to the M(4) site: M(1,2,3) = Mg5.00, M(4) = Mn2+0.98Ca0.30Fe0.02 Mg0.56Na0.06 apfu (atoms per formula unit). Ghose and Yang (Reference Ghose and Yang1989) refined the structure of a C2/m amphibole with the formula (Ca0.24Mn2.41Mg4.20Fe0.15)Si8O22(OH)2 from Nsuta, Ghana; the M(4) site is dominated by Mn (although the small amount of Fe was not considered in the assignment of site populations) and small amounts of Mn occur at the M(1,2,3) sites as forced by the chemical formula. Thus at some composition intermediate between M (4)Mn = 0.89 apfu (Prewitt et al., Reference Prewitt, Papike and Ross1970) and M (4)Mn = 1.67 apfu (Ghose and Yang, Reference Ghose and Yang1989), the structure must change from P21/m to C2/m symmetry at room temperature.
Roy (Reference Roy1981) noted that Mn-rich cummingtonite is a common constituent of Mn ore-deposits with compositions containing less than ∼2.3 Mn2+ apfu. Maresch and Czank (Reference Maresch and Czank1983) synthesised orthorhombic amphiboles of composition Mn2+xMg7–x[Si8O22](OH)2 with 0.2 < Mn2+ < 2.3 apfu and noted that, unlike their natural monoclinic compositional analogues, these synthetic amphiboles contain abundant chain-width and chain-arrangement faults. Conversely, Dasgupta et al. (Reference Dasgupta, Miura and Hariya1985) determined the stability of monoclinic amphibole (space group not determined) of composition Mn2+2.35Mg4.65[Si8O22](OH)2 at ∼100–170°C lower than did Maresch and Czank (Reference Maresch and Czank1983), and this temperature difference may account for the difference in the character of the synthesised amphiboles.
Previous work: The P21/m–C2/m transition in magnesium-iron-manganese amphiboles
Much work has been done on crystallographic aspects of the P21/m–C2/m transition in magnesium-iron-manganese amphiboles. This work focused on the structural changes associated with the transition between the two different structure types as a function of temperature (Prewitt et al., Reference Prewitt, Papike and Ross1970; Sueno et al., Reference Sueno, Papike, Prewitt and Brown1972; Yang and Hirschmann, Reference Yang and Hirschmann1995; Yang and Smyth, Reference Yang and Smyth1996), chemical composition (Reece et al., Reference Reece, Redfern, Welch and Henderson2000, Reference Boffa Ballaran, McCammon and Carpenter2002) and pressure (Yang et al., Reference Yang, Hazen, Prewitt, Finger, Lu and Hemley1998; Boffa Ballaran et al., Reference Boffa Ballaran, Angel and Carpenter2000). Later work has focused on the thermodynamic aspects of the P21/m–C2/m transition (Boffa Ballaran et al., Reference Boffa Ballaran, Angel and Carpenter2000, Reference Boffa Ballaran, Carpenter and Domeneghetti2001, Reference Boffa Ballaran, McCammon and Carpenter2002, Reference Boffa Ballaran, Carpenter and Domeneghetti2004; Reece et al., Reference Reece, Redfern, Welch and Henderson2000, Reference Reece, Redfern, Welch, Henderson and McCammon2002) via the behaviour of the order parameter through the phase transition. Note that the property of P21/m symmetry does not warrant an amphibole being considered as a distinct species; species definitions depend on chemical composition although the suffix ‘-P21/m’ is appended to the name of the amphibole or synthetic amphibole where appropriate. Thus monoclinic magnesium-iron-manganese amphiboles with Mn2+ and Fe2+ dominant at the M(4) site are recognised by IMA2012 as distinct species but have no names as, although they are relatively common in Nature, they have not been formally described, proposed and approved by IMA-CNMNC as new mineral species. Monoclinic magnesium-iron-manganese amphiboles with Mg dominant at M(4) are not recognised as distinct species because no natural (unheated) examples have yet been discovered and laboratory-heated minerals are no longer considered as minerals by IMA-CNMNC (with the exception of heated metamict minerals). All amphibole minerals with P21/m symmetry at room temperature refined thus far have Mn2+ or Fe2+ as the dominant cation at the M(4) site. Some chemical compositions have been reported that purportedly have very low Fe/(Fe + Mg) ratios but closer inspection shows this not to be the case: e.g. (1) the amphibole occurs as lamellae in tremolite too small to analyse by electron microprobe and the Fe/(Fe + Mg) ratio reported is that of tremolite; (2) the amphibole is orthorhombic, not monoclinic.
Kisch (Reference Kisch1969) reported cummingtonite of composition Na0.09(Ca0.19Mg5.45Fe2+1.23Mn2+0.04Ti4+0.01Al0.07)(Si7.83Al0.17)O22(OH)2 with P21/m symmetry at room temperature. As this is the room-temperature cummingtonite with the most Mg at M(4) (see compilation of analyses in Table 1 of Welch et al., Reference Welch, Camara, Della Ventura and Iezzi2007), we deemed it worthy of further examination.
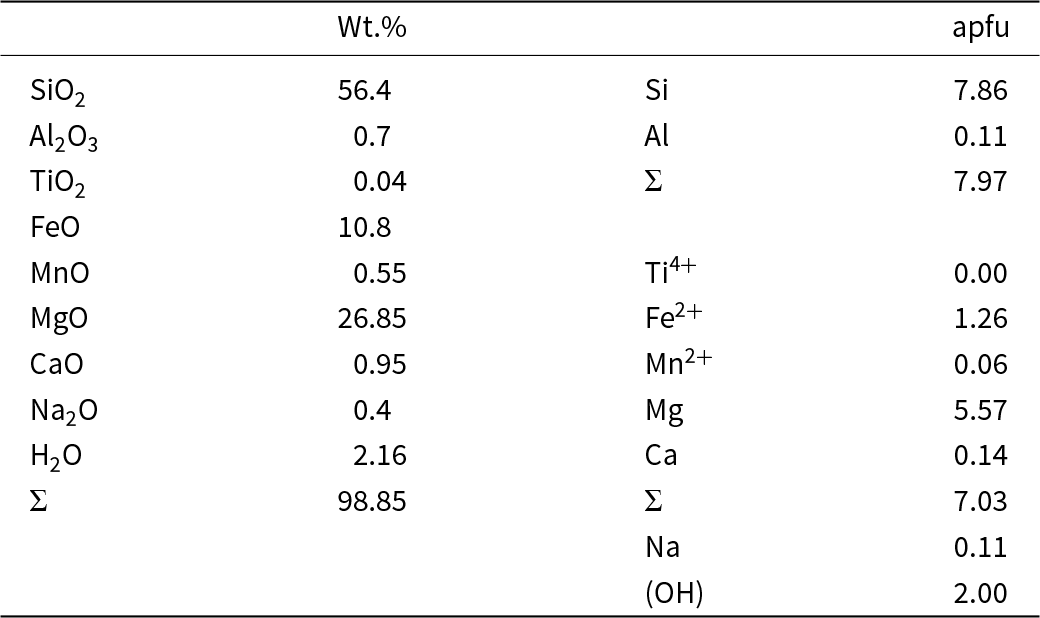
Chemical composition (wt.%)* and unit formula (apfu) for cummingtonite-(P21/m).

* Chemical composition from Kisch (Reference Kisch1969).
Sample provenance
A colourless magnesium-iron-manganese amphibole is associated with a pale bluish-green calcium amphibole and chlorite in a small irregular mass of metamorphosed ultramafic rock within the ‘granitized zones’ west of the Cooma granite-gneiss, Cooma, New South Wales (Joplin, Reference Joplin1942). Kisch (Reference Kisch1969) identified the colourless amphibole as cummingtonite-(P21/m).
Chemical composition
We have used the chemical analysis of cummingtonite-(P21/m) (Table 1) determined by electron-microprobe analysis by Kisch (Reference Kisch1969). The empirical chemical formula (Table 1) was calculated on the basis of 24 (O + OH + F) with OH + F = 2 apfu and Fe3+ = 0.10 apfu as indicated by Mössbauer spectroscopy.
Infrared spectroscopy
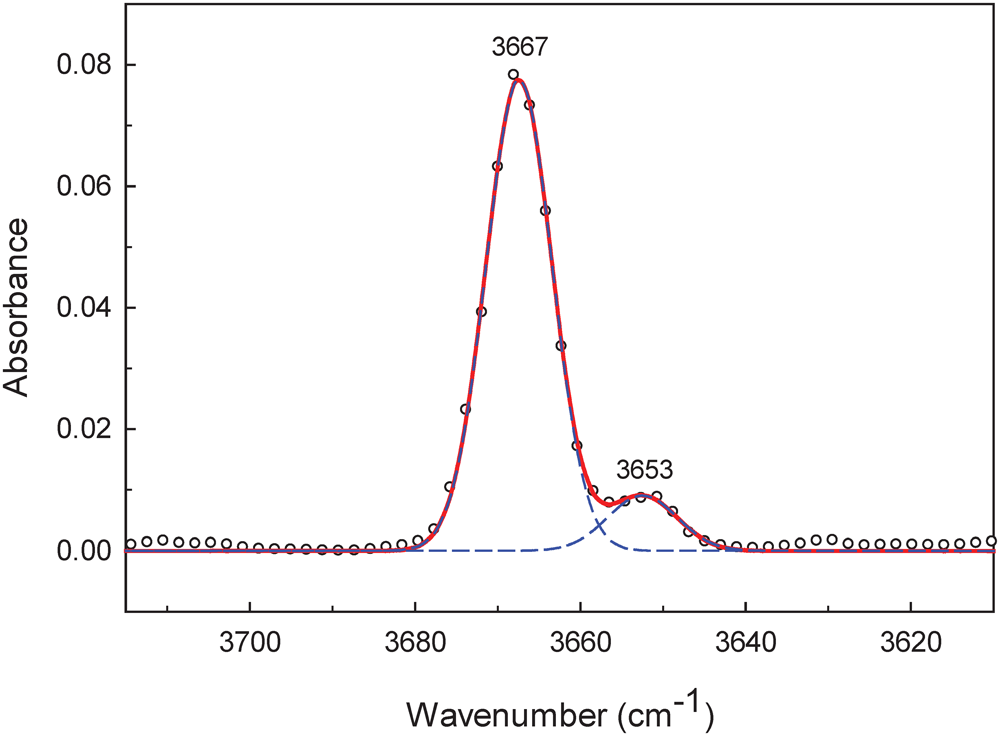
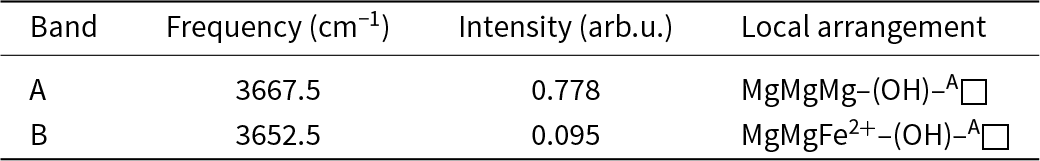
For single-crystal FTIR spectroscopy, a transmission spectrum was collected using a Bruker Hyperion 2000 IR microscope equipped with a liquid-nitrogen-cooled MCT detector. The sample was prepared as a thin film using a diamond micro-compression cell. Data over the range (4000–650 cm–1) were obtained by averaging 100 scans with a resolution of 4 cm–1. Base-line correction and spectral fitting (with a Gaussian function) were done using the program Fityk (Wojdyr, Reference Wojdyr2010). Figure 2 shows the infrared spectrum of cummingtonite-(P21/m) in the principal OH-stretching region fit to two bands with half-widths of 10 cm–1 and fitting parameters as listed in Table 2.
The infrared spectrum of cummingtonite-(P21/m) in the principal (OH)-stretching region.

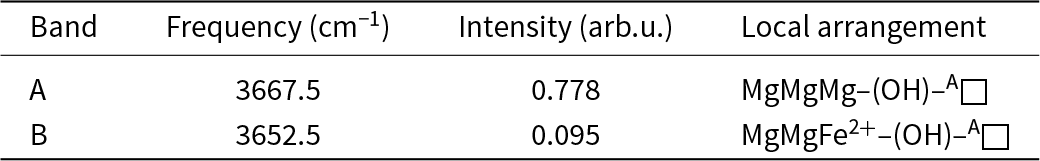
Infrared band positions, observed intensities and associated local arrangements for cummingtonite-(P21/m).
Mössbauer spectroscopy
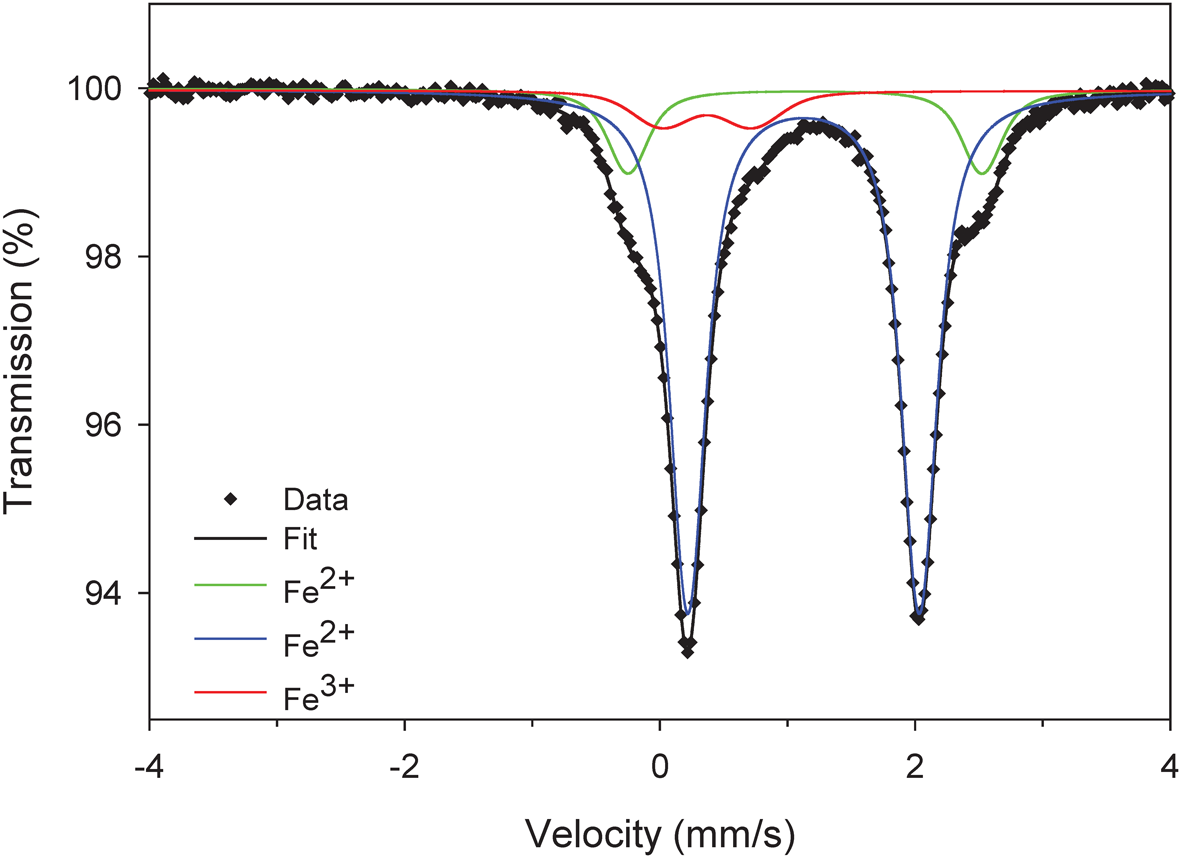
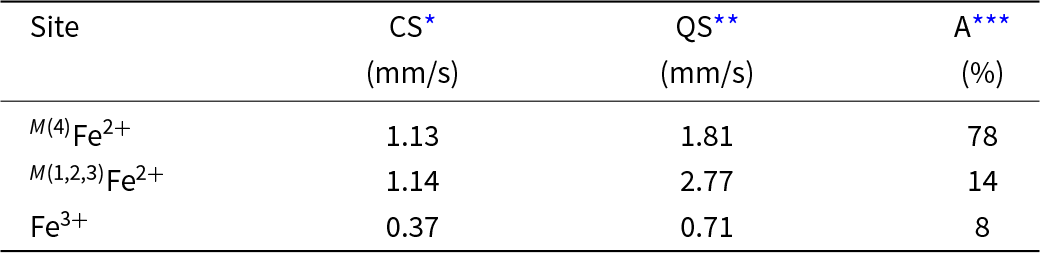
The Mössbauer spectrum was acquired in transmission geometry at room temperature using a 57Co(Rh) point source, and the spectrometer was calibrated with the room-temperature spectrum of α–Fe. The spectrum was analysed in terms of a Voigt-function-based quadrupole-splitting distribution (Rancourt and Ping, Reference Rancourt and Ping1991) using the RECOIL® software package. Figure 3 shows the Mössbauer spectrum of cummingtonite-(P21/m) fit to three bands and details of the fitted spectrum are given in Table 3.
The 57Fe Mössbauer spectrum of cummingtonite-(P21/m). Blue doublet: Fe2+ at M(4); green doublet: Fe2+ at M(1,2,3); red doublet: Fe3+ at M(2) in exsolution lamellae.

Mössbauer parameters for cummingtonite-(P21/m).
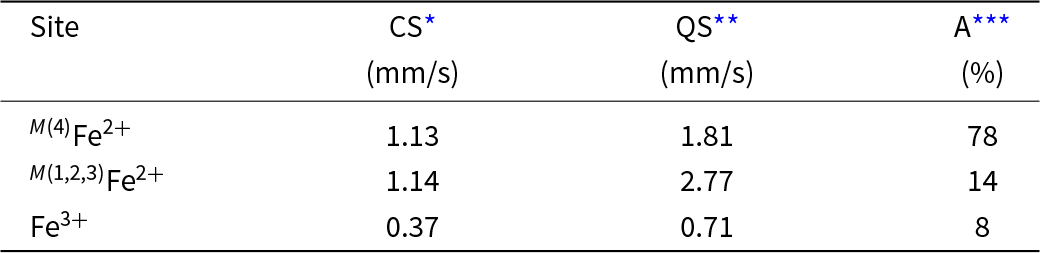
* CS = Centre Shift relative to α–Fe at RT; standard deviation = 0.02 mm/s;
** QS = Quadrupole Splitting; standard deviation = 0.02 mm/s;
*** A = relative area; standard deviation = 1%.
Crystal-structure refinement
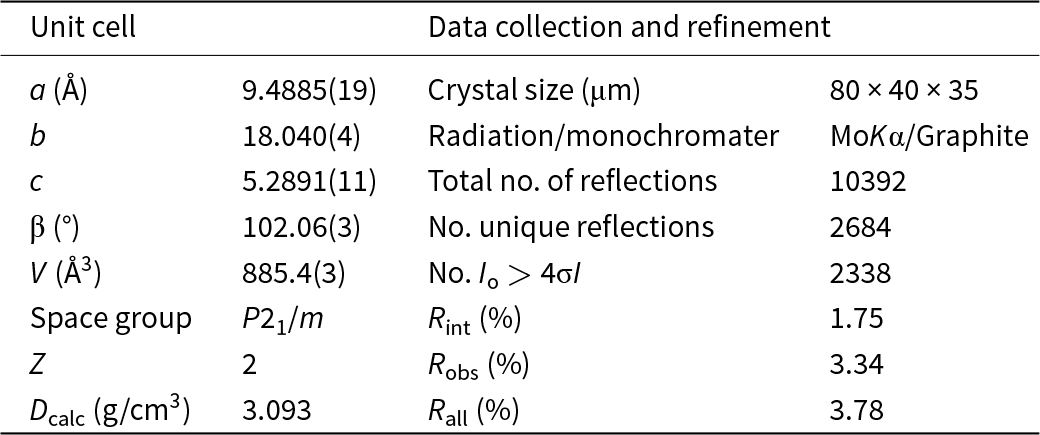
A crystal was attached to a tapered glass fibre and mounted on a Bruker D8 three-circle diffractometer equipped with a rotating-anode generator (MoKα radiation), multilayer optics and an APEX-II detector. A total of 31960 intensities was collected to 65°2θ using 6 s per 0.3° frame, with a crystal-to-detector distance of 5 cm. Empirical absorption corrections (SADABS; Sheldrick, Reference Sheldrick2008) were applied and reflections were corrected for Lorentz, polarisation and background effects, averaged and reduced to structure factors. The unit-cell dimensions were refined by least-squares from the positions of 4061 reflections with I > 10σI and are given in Table 4, together with other information on data collection and structure refinement. All calculations were done with the SHELXTL PC (Plus) system of programs; R indices are expressed as percentages. The structure was refined to convergence by full-matrix least-squares methods with anisotropic-displacement parameters for all atoms except the H atoms. At the later stages of refinement, difference-Fourier maps showed weak density maxima ∼1 Å from the O(3A) and O(3B) anions. These maxima were entered into the refinement as H atoms and their positional parameters were refined with the restraint that the O(3A)–HA and O(3B)–HB distances be ∼0.96 Å. In crystal structures refined by X-ray diffraction, the Odonor–H distances obtained are too short as a significant fraction of the electron density of H is delocalised into the Odonor–H bond. This has the effect of giving poor H···Oacceptor hydrogen-bond distances, often making it difficult to interpret the role of hydrogen bonds in a structure. This may be avoided by restraining the Odonor–H distances during refinement to their expected values based on neutron diffraction. In the present case, the Odonor···H distances were restrained to 0.96 Å, close to the Odonor–H distances found by neutron diffraction for tremolite (Hawthorne and Grundy, Reference Hawthorne and Grundy1976) and kaersutite (Gatta et al., Reference Gatta, McIntyre, Oberti and Hawthorne2017). The structure converged to a final R obs index of 3.34%. Refined atom coordinates and anisotropic-displacement parameters are listed in Table 5, and selected interatomic distances are given in Table 6. The crystal structure of cummingtonite-(P21/m) is illustrated in Fig. 1 and will be helpful in discussions involving bond lengths and cation order. A table of structure factors and a Crystallographic Information File (CIF) for cummingtonite-(P21/m) have been deposited with the Principal Editor of Mineralogical Magazine and are available as Supplementary Material (see below).
Miscellaneous information for cummingtonite-(P21/m).

Site coordinates and anisotropic-displacement parameters for cummingtonite-(P21/m).

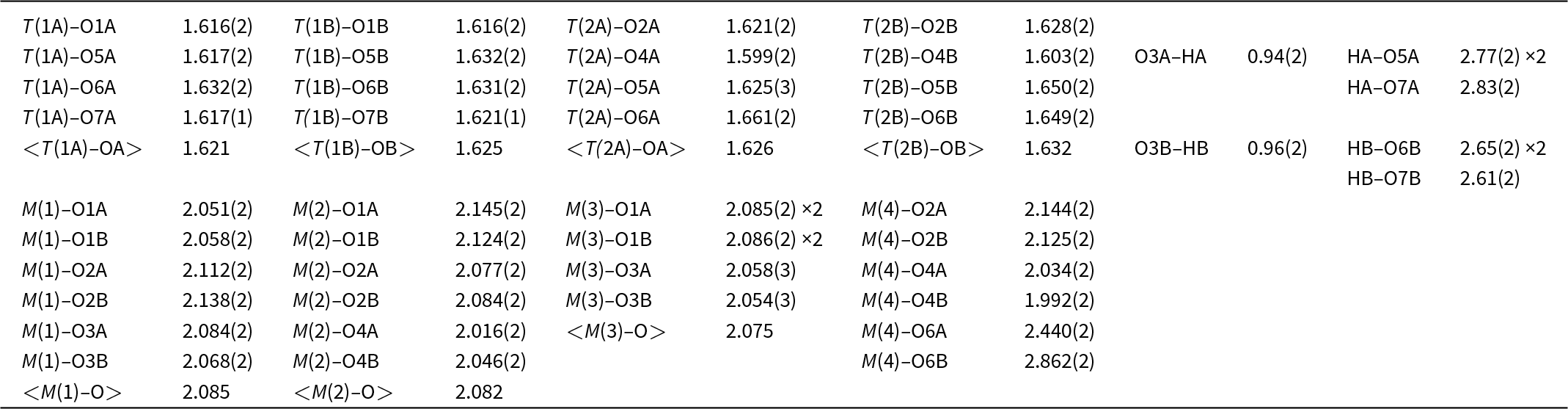
Selected interatomic distances (Å) in cummingtonite-(P21/m).

Derivation of site populations
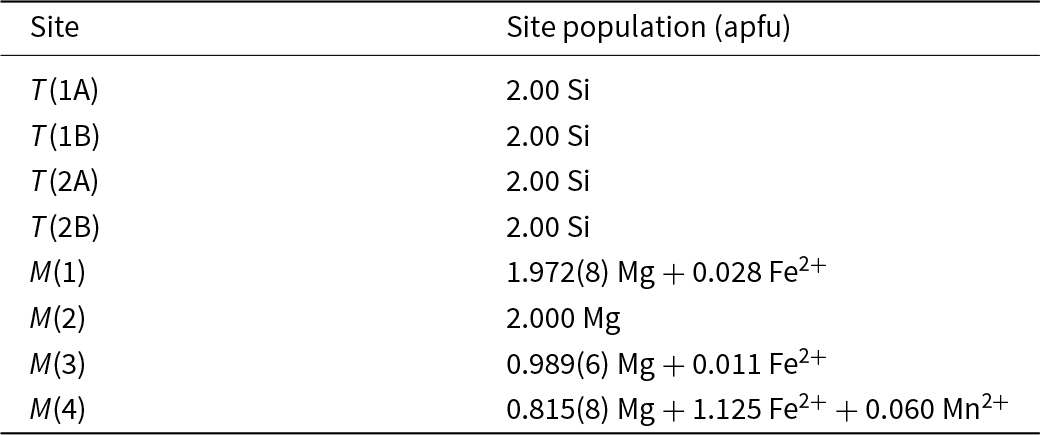
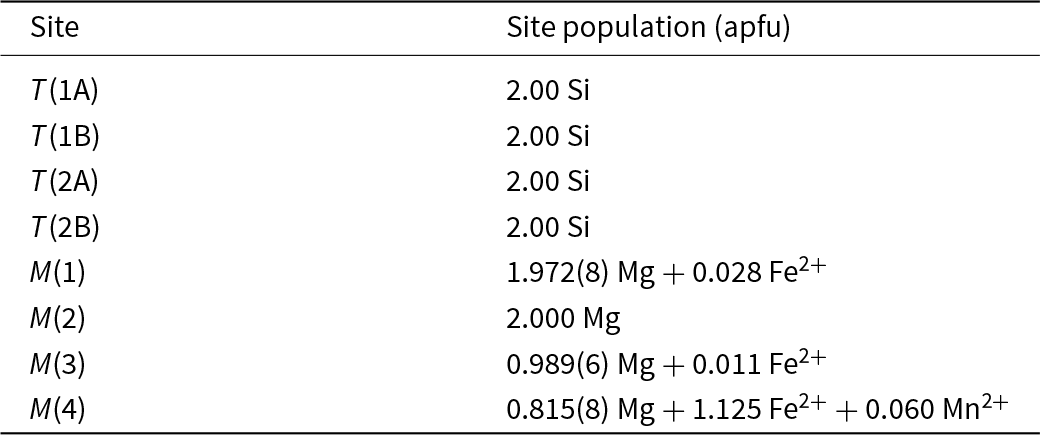
The chemical analysis of Table 1 lists significant Na as an A-group cation. However, there was no significant density in the A-cavity at the final stages of refinement and attempts to insert ANa and refine its occupancy led to near-zero occupancy and nonsensical atom coordinates. As stated in the section on sample provenance, cummingtonite-(P21/m) is associated with a pale bluish-green calcium amphibole. Coexisting magnesium-iron-manganese and calcium amphiboles generally show mutual exsolution (e.g. Asklund et al., Reference Asklund, Brown and Smith1962; Jaffe et al., Reference H-W, Robinson and Klein1968; Ross et al., Reference Ross, Papike and Shaw1969; Klein et al., Reference Klein, Schumacher and Czank1996). Let us examine the formula from Table 1 (modified for the amount of Fe3+ determined here by Mössbauer spectroscopy, Table 3, Fig. 3): Na0.11(Ca0.14Mg5.57Fe2+1.16Mn0.06Fe3+0.10)7.03(Si7.86Al0.11)7.97O22(OH)2. The constituents of this formula that do not fit the form (Mg,Fe2+,Mn)7Si8O22(OH)2 are ANa0.11BCa0.14CFe3+0.10TAl0.11. These constituents we may reasonably assign to calcium amphibole exsolved from cummingtonite-(P21/m), particularly as no density was observed at the A-site and attempts to refine Na at the A-site were unsuccessful. If we remove these extra constituents from the formula and renormalise, we get the following formula for cummingtonite-(P21/m): (Mg5.66Fe2+1.28Mn0.06)Σ7.00Si8.00.
The refined <T–O> distances (Table 6) are in accord with the lack of Al at the T sites. As expected, Fe2+ is strongly ordered at the M(4) site relative to the M(1,2,3) sites (Table 7). The Fe2+ content at M(2) refined to very close to zero and was set at this value in the final stages of refinement. Small positive occupancies for Fe2+ at M(1) and M(3) were obtained with a preference for M(1).
Assigned site populations (apfu) for cummingtonite-(P21/m).
Cation order from infrared and Mössbauer spectroscopies
The fitted infrared spectrum in the principal OH-stretching region is shown in Fig. 2 and details regarding the fitted spectrum are given in Table 2. The spectrum has a strong symmetrical peak at 3667 cm–1 and a much weaker peak at 3653 cm–1. These peaks are assigned to configuration symbol M (1)M2+ M (1)M2+ M (3)M2+–OH–A□–TSiTSi (Hawthorne and Della Ventura, Reference Hawthorne and Della Ventura2007) which simplifies to M (1)M2+ M (1)M2+ M (3)M2+–OH as the other sites are completely ordered, and the resulting local arrangements are M (1)MgM (1)MgM (3)Mg–OH and M (1)MgM (1)Fe2+M(3)Mg–OH (≈ M (1)MgM (1)MgM (3)Fe2+–OH) in accord with Burns and Strens (Reference Burns and Strens1966) and Strens (Reference Strens and Farmer1974). In the P21/m-amphibole structure, there are two distinct (OH) groups (Table 5), O(3A) associated with HA and O(3B) associated with HB, and each local arrangement around each distinct (OH) group will give rise to two peaks of (approximately) the same intensity. In some other P21/m amphibole structures, this is the case: in both synthetic ANaB(LiMg)CMg5Si8O22(OH)2 and synthetic Na(NaMg)Mg5Si8O22(OH)2 (Iezzi et al., Reference Iezzi, Tribaudino, Della Ventura, Nestolo and Bellatreccia2005a, Reference Iezzi, Gatta, Kockelmann, Della Ventura, Rinaldi, Schäfer, Piccinini and Gaillard2005b), there are two well-resolved peaks assigned to the local arrangement M (1)MgM (1)MgM (3)Mg–OH (Table 8). However, in cummingtonite-(P21/m) (this work), there is a single peak at 3667 cm–1 with a half-width of 10 cm–1, resembling the spectrum of C2/m cummingtonite (Reece et al., Reference Reece, Redfern, Welch and Henderson2000; Boffa Ballaran et al., Reference Boffa Ballaran, Carpenter and Domeneghetti2004) (Table 8). We may get a measure of the strengths of the hydrogen bonds in amphiboles with M(1,2,3) ≈ 5 apfu by examining the M(1,2,3)–OH distances: the shorter the bonds from CMg to the (OH) group, the stronger the Odonor–H bonds, the higher the principal-stretching frequency, and the weaker the H···Oacceptor hydrogen bond. In synthetic Na(NaMg)Mg5Si8O22(OH)2 (Table 8), the <O3A–Mg3> and <O3B–Mg3> distances are very different: 1.996 and 2.030 Å, respectively, indicating that the corresponding peaks in the infrared should be well-resolved, as is the case (Table 8). In cummingtonite-(P21/m) and cummingtonite (Table 8), the corresponding <O3A–Mg3>, <O3B–Mg3> and <O3–Mg3> distances are very similar: 2.075, 2.063 and 2.061 cm–1 with very close half-widths: 10 and 8 cm–1, respectively. The similarity of these values and their considerable difference from those of Na(NaMg)Mg5Si8O22(OH)2 suggest that there is a small difference in the frequencies of the peaks corresponding to the O3A–HA and O3B–HB arrangements. This peak separation is small compared to the half-widths of the peaks, and results in overlap of the two peaks in the spectrum of cummingtonite-(P21/m) with just a slight broadening of the resultant composite peak.
Mean OH–M distances (Å) and peaks (cm–1) in the principal OH-stretching region of the infrared for selected amphiboles.

The Mössbauer spectrum (Fig. 3) shows three quadrupole-split doublets. The dominant doublet and the intermediate-intensity doublet may be assigned to octahedrally coordinated Fe2+ at M(4) and M(1,2,3), respectively, in accord with Bancroft et al. (Reference Bancroft, Burns and Maddock1967), Hafner and Ghose (Reference Hafner and Ghose1971), Ghose and Weidner (Reference Ghose and Weidner1972) and Barabanov and Tomilov (Reference Barabanov and Tomilov1973). The weakest doublet has a much lower centre shift (0.37 mm/s, Table 3), indicative of octahedrally coordinated Fe3+ (Hawthorne, Reference Hawthorne1988) that occurs in exsolved calcium amphibole. Both the infrared spectrum and the Mössbauer spectrum are in accord with the strong order of Fe2+ at M(4) but also a small amount of Fe2+ at M(1,2,3) as indicated by the structure refinement.
The argument for a revised classification for the magnesium-iron-manganese amphiboles
Some years ago, there was external pressure on IMA-CNMNC to include BMg, BFe2+ and BMn2+ as classification criteria, and the chair of IMA-CNMNC at the time directed the Amphibole Subcommittee to revise IMA2012 in this respect. This revision has raised some tricky issues concerning the classification of amphiboles (and minerals in general) and the senior (oldest) author of both this paper and IMA2012 thought it appropriate to air these issues in the scientific literature prior to possible implementation.
The current classification of the magnesium-iron-manganese amphiboles
Table 9 shows the current IMA2012 classification of the magnesium-iron-manganese amphiboles. When most amphiboles are analysed, their site occupancies are not determined and cations are assigned to the A-, B-, C- and T-groups in the following order: (1) T: Si, Al, Ti4+; (2) C: Ti4+, Al, Fe3+, Mn3+, V3+, Cr3+, Mg, Fe2+, Mn2+, Li; (3) B: Mn2+, (Fe2+, Mg), Li, Ca, Na; and (4) A: Ca, Na, K. From the work cited above, it was recognised over 50 years ago that Mn2+ shows a strong preference for the M(4) site in monoclinic magnesium-iron-manganese amphiboles. The relative amounts of Mg and Fe2+ in the B- and C-cation groups of the magnesium-iron-manganese amphiboles cannot be determined without crystal-structure refinement or Mössbauer or infrared spectroscopy. Hence for this subgroup, IMA2012 treated the divisions between Mg–Fe2+ homovalent analogues in terms of the sum of the B and C cations. However, it was recognised that Mn2+ has a significant preference for the M(4) site, and hence distinct species were recognised in IMA2012 with Mn2+ assigned as the dominant B-cation with the rider that “where direct experimental data are available, they take precedence over such an assignment”.
Possible end-member schemes for magnesium-iron-manganese amphiboles.

An alternative classification
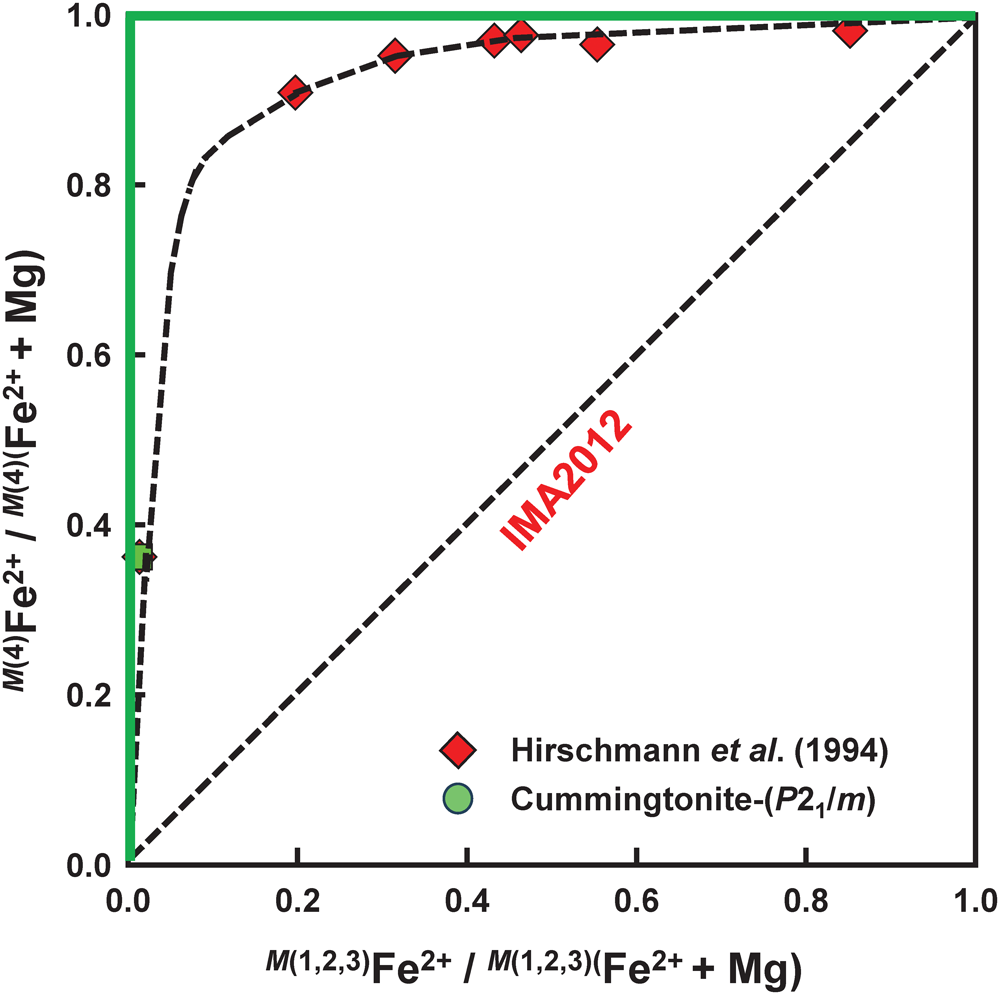
For amphiboles of the cummingtonite–grunerite series, Hirschmann et al. (Reference Hirschmann, Evans and Yang1994) and Evans et al. (Reference Evans, Ghiorso, Yang and Medenbach2001) showed that Fe2+ is very strongly ordered at the M(4) site (Fig. 4), relative to the M(1,2,3) sites. Orthorhombic magnesium-iron amphiboles (anthophyllite–‘ferro-anthophyllite’) show analogous behaviour (Seifert, Reference Seifert1978). In Fig. 4, the red diamonds show compositions of unheated amphiboles of the cummingtonite–grunerite series of Hirschmann et al. (Reference Hirschmann, Evans and Yang1994). IMA2012 treats these compositions as having complete disorder of Mg and Fe2+ between the B- and C-groups of cations, i.e. the M(4) and M(1,2,3) sites; according to this model, the data should follow the straight broken black line in Fig. 4. There is no correlation between the behaviour of the data (red diamonds) and the model used by IMA2012; the latter is obviously not adequate. An alternative model to that used by IMA2012 is as follows: C-group cations are assigned as indicated above, and if the total amount exceeds 5 apfu, the cations are assigned to the B-group in the following order: Mn2+, Fe2+, Mg2+. When this is done, the site-occupancy data will follow the solid green lines in Fig. 4 which are in much closer accord with the experimental data than the assignment used in IMA2012.
Order of Fe2+ and Mg between the M(4) and M(1,2,3) sites in monoclinic low-Mn magnesium-iron-manganese amphiboles. The red diamonds are the unheated data of Hirschmann et al. (Reference Hirschmann, Evans and Yang1994) and the green circle is the data of cummingtonite-(P21/m) from this work. IMA2012 treats these compositions as having no order of Mg and Fe2+ between the B- and C-groups of cations [i.e. the M(4) and M(1,2,3) sites], and according to this model, the data should follow the straight dashed black line labelled IMA2012. An alternative model is to assume complete order of Fe2+ and Mg between the B- and C-groups of cations [i.e. the M(4) and M(1,2,3) sites], as indicated by the solid green lines that more closely approximate the experimental data.

Solid solution in magnesium-iron-manganese amphiboles
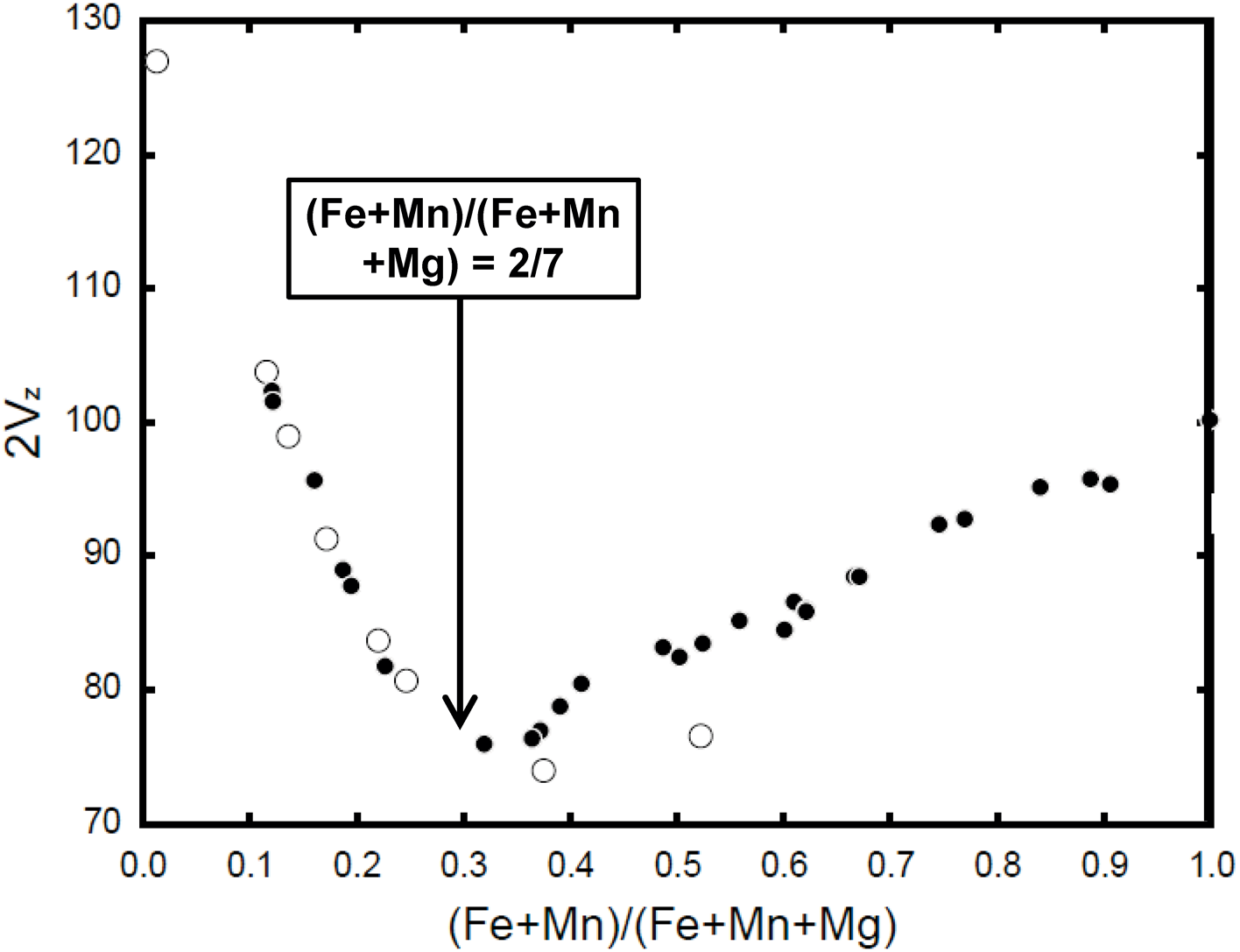
It has been customary in the past to regard the compositional range Mg7Si8O22(OH)2 – Fe2+7Si8O22(OH)2 in monoclinic amphiboles as a binary solid-solution series. However, it is better considered as two separate solid-solution series, BMg2CMg5Si8O22(OH)2 – BFe2+2CMg5Si8O22(OH)2 and BFe2+2CMg5Si8O22(OH)2 – BFe2+2CFe2+5Si8O22(OH)2 as the behaviour of the M(4) and M(1,2,3) sites is quite different in relation to the preferred cations substituting at the different sites. Doing so brings the magnesium-iron-manganese amphiboles into line with related amphibole subgroups involving B-group Ca, Na and Li. Moreover, this view is supported by the optical data of Evans et al. (Reference Evans, Ghiorso, Yang and Medenbach2001) in which there is a radical change in optical properties between the solid solutions BMg2CMg5Si8O22(OH)2 – BFe2+2CMg5Si8O22(OH)2 and BFe2+2CMg5Si8O22(OH)2 – BFe2+2CFe2+5Si8O22(OH)2 (Fig. 5).
Variation of 2V z of anthophyllite-ferro-anthophyllite (hollow circles) and metamorphic cummingtonite–grunerite (solid circles) as a function of (Fe + Mn)/(Fe + Mn + Mg); modified from Evans et al. (Reference Evans, Ghiorso, Yang and Medenbach2001).

Thus there seems to be a good scientific reason to treat Mg, Fe2+ and Mn2+ as distinct B-group constituents with regard to the classification and nomenclature of the amphiboles. However, if we were to treat Mg, Fe2+ and Mn2+ in exactly the same way as we treat other constituents, particularly Ca, we end up with some undesirable features. For example, if we were to adhere to the dominant-constituent rule of IMA (Hatert and Burke, Reference Hatert and Burke2008), the composition □(Ca0.53Mg0.49Fe2+0.49Mn2+0.49)Σ2Mg5Si8O22(OH)2 = □(Ca0.53BΣM2+1.47)Σ2Mg5Si8O22 (OH)2 would be assigned the name tremolite. Such a change is undesirable from many aspects, as it would drastically extend the compositional field of tremolite and contract the collective compositional field of the magnesium-iron-manganese amphiboles. One way around this problem is to retain the criterion used in IMA2012 for this purpose: the boundary between the calcium- and the collective magnesium-, iron- and manganese-amphibole subgroup is assigned as BCa/ΣB ≥ BΣM2+/ΣB, and then the individual magnesium-, iron- and manganese-amphibole subgroups are assigned as BMg/ΣB ≥ BΣFe2+/ΣB, BΣMn2+/ΣB, etc. within the collective subgroup of the magnesium-iron-manganese amphiboles. Note that, unlike IMA2012, this new nomenclature adheres to the definition of ‘ferro-’: CFe2+ > CMg, CMn2+ except where well-established names (i.e. grunerite) are retained.
Nomenclature
So how should the resultant amphibole species be named (Table 9)? At the present time, there is no well-characterised monoclinic amphibole with the dominant end-member composition A□BMg2CMg5TSi8O22(OH)2. If this is assigned as the end-member formula of cummingtonite, there will be no cummingtonite known in the geological record and all the cummingtonites in the literature will be renamed ‘name 1ʼ as in Table 9. Few if any mineralogists and petrologists would consider this a satisfactory procedure. It seems more practical to redefine cummingtonite as having the end-member formula A□BFe2+2CMg5TSi8O22(OH)2: all amphiboles named cummingtonite in the literature will remain cummingtonite and monoclinic amphiboles with the end-member formula A□BMg2CMg5TSi8O22(OH)2 (if found) will be assigned a new name.
Current ambiguity in naming magnesium-iron-manganese amphiboles
IMA2012 contains the following statements: “The new classification presented here is based on the chemical formula of an amphibole measured by electron microprobe or wet-chemical techniques, possibly augmented by additional analytical, structural and spectroscopic data” (our italics). Some degree of ambiguity is introduced by the inclusion of “structural and spectroscopic data”. As noted above, if we assign excess C-group cations to the B-group in the order Mn2+, Fe2+, then Mg, this assumes that there is no Mn2+ or Fe2+ at M(1,2,3). However, we have seen from the structural and spectroscopic results for cummingtonite-(P21/m) that this is not the case: some Fe2+ occurs at the M(1,2,3) sites even though [6]Mg exceeds 5.00 apfu. Moreover, Reece et al. (Reference Reece, Redfern, Welch and Henderson2000) showed that Mn2+ is not completely ordered at the M(4) site in an Fe-free C2/m cummingtonite. Thus the boundaries between the possible end-members listed in Table 9 depend on the experimental method used to determine the assignment of cations in the structure. In terms of the approval of new species, this is not a problem as use of crystal-structure refinement and spectroscopy is almost always required. However, work on the chemical characterisation of amphiboles commonly involves electron microprobe analysis (EMPA) only, and then ambiguity can arise in the naming of amphiboles with these compositions depending on the experimental techniques available to particular scientists. We can live with this ambiguity in the description and approval of new magnesium-iron-manganese amphiboles as this requires crystal-structure refinement and spectroscopy data on site occupancies. However, naming of already approved magnesium-iron-manganese amphiboles, as is the case for much compositional and petrological work on these amphiboles, assigns excess C-group cations to the B-group in the order Mn2+, Fe2+, then Mg, and the amphiboles are named according to the dominance of BMg, BFe2+ or BMn2+. Use of EMPA data only versus the collective use of EMPA data, crystal structure refinement and/or spectroscopic data may result in the assignment of different names for the same material. How to resolve this ambiguity is not clear.
A major issue with naming and classifying amphiboles is striking a balance between ease of use and rigour. Overemphasis on ease of use is accompanied by the risk of misrepresenting their crystal chemistry and compromising their use in Petrology. Overemphasising rigour restricts the effective use of amphiboles to those who have sophisticated instrumentation and expertise in its use. These opposing issues tend to lead to a split in the amphibole community, but this is not necessarily a bad thing. Two camps pulling in opposite directions will hopefully lead to solutions that are semi-acceptable to the general community.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1180/mgm.2025.14
Acknowledgements
We thank Mark Welch and Gianluca Iezzi for their excellent reviews of this paper. FCH was supported by a Tier-I Canada Research Chair in Crystallography and Mineralogy, Research Tools and Equipment, and Discovery grants from the Natural Sciences and Engineering Research Council of Canada, and by Canada Foundation for Innovation Grants for instrumentation.
Competing interests
The authors declare none.