Introduction
Chicken (Gallus gallus domesticus) is one of the most intensively reared domesticated poultry species, and a fundamental and profitable animal production enterprise (Guèye Reference Guèye2003; FAO 2004). This industry significantly contributes to food security, poverty reduction, and ecological utilisation of natural resources, particularly for smallholder farmers in rural and peri-urban areas (Guèye Reference Guèye2003; Mapiye et al. Reference Mapiye, Mwale, Mupangwa, Chimonyo, Foti and Mutenje2008). In 2022, Africa’s total poultry population was estimated at 2,425 million, of which 2,352 million were chickens (FAOSTAT 2024). Among domestic birds kept for meat and egg production, free-ranging village chickens are the predominant species in the rural poultry sector, where they are raised by village communities for subsistence (Malatji et al. Reference Malatji, Tsotetsi, Van Marle-Koster and Muchadeyi2016; Muchadeyi et al. Reference Muchadeyi, Sibanda, Kusina, Kusina and Makuza2004), with a large proportion kept under a traditional free-range production system.
According to Sarba et al. (2019), free-range chickens contribute over 70% of poultry products and 20% of animal protein intake in Africa. These chickens mainly constitute indigenous breeds, primarily reared successfully in resource-limited rural villages of developing countries due to their adaptation to harsh environmental conditions and extensive husbandry practices (Elson et al. Reference Elson, Gleadthorpe, Vale and Mansfield2011). They scavenge during the day whilst occasionally being fed leftover scrap food and/or rationed formulated chicken feed and are confined to shelters at night (Muchadeyi et al. Reference Muchadeyi, Wollny, Eding, Weigend and Simianer2009). This husbandry system predisposes them to various diseases as they feed and attain food from contaminated environments (Mukaratirwa et al. Reference Mukaratirwa, Hove, Esmann, Hoj, Permin and Nansen2001; Permin and Pedersen Reference Permin and Pedersen2002) and their associated intermediate hosts (Phiri et al. Reference Phiri, Phiri, Ziela, Chota, Masuku and Monrad2007). Several studies have shown that free-ranging chickens are often affected by several bacterial, viral, fungal, and parasitic infections, resulting in reduced production, high morbidity, mortality, and subsequent significant economic losses (Irungu et al. Reference Irungu, Kimani and Kisia2004; Kaingu et al. Reference Kaingu, Kibor, Shivairo, Kutima, Okeno, Waihenya and Kahi2010; Ola-Fadunsin et al. Reference Ola-Fadunsin, Uwabujo, Sanda, Ganiyu, Hussain, Rabiu, Elelu and Alayande2019). Parasitic infections rank high among these infections, and gastrointestinal helminths (cestodes, nematodes, and trematodes) are the most prevalent parasites affecting poultry production (Berhe et al. Reference Berhe, Mekibib, Bsrat and Atsbaha2019; Mikail and Adamu Reference Mikail and Adamu2008; Mohammed et al. Reference Mohammed, Sidi, Saleh, Aliyu, Ibrahim and Sabo2021; Muhairwa et al. Reference Muhairwa, Msoffe, Ramadhani, Mollel, Mtambo and Kassuku2007; Mukaratirwa et al. Reference Mukaratirwa, Hove, Esmann, Hoj, Permin and Nansen2001; Mungube et al. Reference Mungube, Bauni, Tenhagen, Wamae, Nzioka, Muhammed and Nginyi2008; Ngongeh et al. Reference Ngongeh, Onyeabor and Erumaka2012).
In Africa, the prevalence of gastrointestinal helminths in free-ranging chickens is influenced by a combination of environmental, management, and host-related factors (Shiferaw et al. Reference Shiferaw, Tamiru, Gizaw, Atalel, Terfa, Dandecha and Mekibib2016). The warm and humid climates prevalent in many regions of the continent provide ideal conditions for the survival and transmission of helminth parasites (Mungube et al. Reference Mungube, Bauni, Tenhagen, Wamae, Nzioka, Muhammed and Nginyi2008). Numerous studies have reported on gastrointestinal helminth parasites, including cestodes and nematodes (Mwale and Masika Reference Mwale and Masika2011); however, nematodes are considered the most significant due to their high species diversity, wide geographical distribution, and the extent of pathological effects they cause in chickens (Shiferaw et al. Reference Shiferaw, Tamiru, Gizaw, Atalel, Terfa, Dandecha and Mekibib2016).
Nematodes affecting free-ranging chickens have been reported in several continents including Asia (Butt et al. Reference Butt, Shaikh, Memon, Narejo and Jalbani2015; Das et al. Reference Das, Laha and Doley2020; Ebrahimi et al. Reference Ebrahimi, Asadpour, Khodaverdi and Borji2014; Nik Nur et al. Reference Nik Nur, Azwan Awang and Shahidur Rahman2015; Salam Reference Salam2015; Vijayalingam et al. Reference Vijayalingam, Rajesh and Latchumikanthan2023; Win et al. Reference Win, Htun, Hmoon, Chel, Thaw, Soe, Oo and Bawm2020; Wuthijaree et al. Reference Wuthijaree, Lambertz, Vearasilp, Anusatsananun and Gauly2019), Oceania (Shifaw et al. Reference Shifaw, Feyera, Sharpe, Elliott, Walkden-Brown and Ruhnke2023), North America (Carrisosa et al. Reference Carrisosa, Jin, McCrea, Macklin, Dormitorio and Hauck2021; Cervantes-Rivera et al. Reference Cervantes-Rivera, Villagómez-Cortés, Arroyo-Lara and Landín-Grandvallet2016; Cornell et al. Reference Cornell, Smith, Crespo, Jones, Crossley, Snyder and Owen2022), South America (da Silva et al. Reference da Silva, Romera, da Silva Conhalato, Soares and Meireles2018; Saraiva et al. Reference Saraiva, Campina, Gonçalves, Melo-Viegas, Santos, Nogueira and Costa2021) and in Africa (Berhe et al. Reference Berhe, Mekibib, Bsrat and Atsbaha2019; Jinga et al. Reference Jinga, Munosiyei, Bobo and Tambura2012; Magwisha et al. Reference Magwisha, Kassuku, Kyvsgaard and Permin2002; Maina et al. Reference Maina, Waruiru, Mbuthia, Munyua, Otieno and Mutune2017; Mukaratirwa and Khumalo Reference Mukaratirwa and Khumalo2010; Mukaratirwa et al. Reference Mukaratirwa, Hove, Esmann, Hoj, Permin and Nansen2001; Mungube et al. Reference Mungube, Bauni, Tenhagen, Wamae, Nzioka, Muhammed and Nginyi2008).
To date, 19 nematode genera have been reported to affect free-range chickens in Africa and these include Ascaridia Dujardin 1845, Heterakis Schrank 1790, Capillaria Zeder 1800, Eucoleus Dujardin 1845, Baruscapillaria Moravec 1982, Aonchotheca López Neyra 1947, Tetrameres Creplin 1846, Strongyloides Grassi 1879, Subulura Molin 1860, Dispharynx Chabaud 1975, Cheilospirura Diesing 1861, Gongylonema Molin 1857, Syngamus Siebold 1836, Cyathostoma Blanchard 1849, Oxyspirura Drasche in Stossich 1898, Trichuris Roederer 1761, Hartertia Seurat 1915, Skrijabinocerca Schikhobalova 1930, and Trichostrongylus Looss 1905 (Magwisha et al. Reference Magwisha, Kassuku, Kyvsgaard and Permin2002; Mukaratirwa and Khumalo Reference Mukaratirwa and Khumalo2010; Mukaratirwa et al. Reference Mukaratirwa, Hove, Esmann, Hoj, Permin and Nansen2001; Nghonjuyi et al. Reference Nghonjuyi, Kimbi and Tiambo2014; Offiong et al. Reference Offiong, Obioku, Umoh, Essien and Idiong2013; Permin et al. Reference Permin, Magwisha, Kassuku, Nansen, Bisgaard, Frandsen and Gibbons1997; Poulsen et al. Reference Poulsen, Permin, Hindsbo, Yelifari, Nansen and Bloch2000; Tay et al. Reference Tay, Emikpe, Ockling, Folitse and Jarikre2017). High prevalence of infections has been reported from the following species: Ascaridia (A.) galli, Heterakis (H.) gallinarum, and Capillaria spp. (Jinga et al. Reference Jinga, Munosiyei, Bobo and Tambura2012; Shiferaw et al. Reference Shiferaw, Tamiru, Gizaw, Atalel, Terfa, Dandecha and Mekibib2016; Wuthijaree et al. Reference Wuthijaree, Lambertz, Vearasilp, Anusatsananun and Gauly2019), where pathology was predominant in juvenile birds with an underdeveloped immune system (Urquhart et al. Reference Urquhart, Duncan, Armour, Dunn and Jennings1996), while heavy infestations in adults allowed them to serve as reservoirs of infection (Permin et al. Reference Permin, Magwisha, Kassuku, Nansen, Bisgaard, Frandsen and Gibbons1997).
Despite the widespread occurrence of gastrointestinal nematodes in free-ranging chickens across Africa, there is limited comprehensive data on their overall prevalence and distribution. Existing studies often focus on specific regions or countries, making it difficult to draw a unified picture of the impact of gastrointestinal nematodes affecting free-range chicken production across the continent (Phiri et al. Reference Phiri, Phiri, Ziela, Chota, Masuku and Monrad2007). Therefore, understanding the epidemiology of gastrointestinal nematodes is crucial for the development of sustainable and cost-effective control strategies tailored to the unique conditions of African poultry farming systems (Dube et al. Reference Dube, Zindi, Mbanga and Dube2010). This review, therefore, aims to provide a comprehensive overview of the prevalence of gastrointestinal nematodes in free-ranging chickens across Africa from 1993 to 2024 through a systematic review and meta-analysis.
Methods
Search strategy
A systematic search of the literature was conducted on Google Scholar, PubMed, and Web of Science databases to retrieve studies reporting on gastrointestinal nematodes of free-ranging chickens from 1993 to 2024. The search combined the following predetermined search terms and Boolean operators (AND, OR): gastrointestinal (GIT) nematode parasites OR GIT worms OR GIT nematode OR GIT parasitic infections AND prevalence AND free-ranging chickens in African countries. Further relevant studies were identified by cross-referencing or screening through bibliographies of relevant articles. Full-text articles were retrieved and managed on EndNote reference manager version X8 (Clarivate Analytics, Philadelphia, PA, USA).
Selection criteria
Articles were included if they met the following predetermined criteria during appraisal for meta-analysis: (i) peer-reviewed studies conducted in Africa and published between 1993 and 2024, (ii) studies conducted specifically on free-ranging chickens, (iii) the number of free-ranging chickens surveyed and infected by GIT nematodes were clearly stated for prevalence calculation, (iv) GIT nematodes were identified up to species level, (v) the diagnostic/detection method(s) used were clearly stated. Studies were excluded if they reported on GIT nematodes of other poultry species or of intensively reared chickens, were conducted outside Africa, were not conducted within the time frame, were published in languages other than English, and lacked sufficient information or relevant outcomes.
Data extraction
Literature search and selection were conducted by I.W.; then M.M.P. and I.N. independently checked selected articles and confirmed the extracted data. Thereafter, duplicates and articles that did not meet the inclusion criteria were excluded from analysis. Relevant information, such as the author’s names, year of publication, country of study, number of free-range chickens screened, number of positive cases and prevalence, nematode species detected, and the diagnostic method used, was extracted from text, tables, and figures and computed on MS Excel for meta-analysis.
Quality assessment of the articles
The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach was used to assess the overall quality of each article for meta-analysis (Guyatt et al. Reference Guyatt, Oxman, Vist, Kunz, Falck-Ytter, Alonso-Coello and Schünemann2008). Studies were allocated a one-point score for each inclusion criterion met; therefore, each study received a score ranging from 0 to 5 points. Publications were deemed of good quality if they received a total score of 5 points, moderate quality a score of 4, and low quality a score of ≤3, and all articles with a low quality score were excluded from the study (Doi and Thalib, Reference Doi and Thalib2008). The standardized quality index score (between 0 and 1) was then computed by calculating scores using five domains: reporting clarity, locality, diagnostic validation, sample size, and sampling strategy. The total score was calculated by adding the points from each domain, with higher scores indicating a lower risk of bias.
Meta-analysis and meta-regression
To avoid overestimating the weight of individual studies, the prevalence data were transformed using the double arcsine approach (Barendregt et al. Reference Barendregt, Doi, Lee, Norman and Vos2013). This approach uses the arcsine transformation twice to measure the prevalence to account for any heterogeneity which may be caused by extremely large or small sample sizes. The MetaXL add-in for Microsoft Excel was used to compute a quality effects model to account for any heterogeneity. Heterogeneity between estimates was evaluated using the inverse variance statistic (I2 index), and its significance was assessed using Cochrane’s Q test. The I2 score of 25%, 50%, or 75% was interpreted as low, moderate, or high heterogeneity following Higgins et al. (Reference Higgins, Thompson, Deeks and Altman2003) protocol, respectively. The estimated prevalence and the 95% confidence interval (CI) of the nematode species among the free-range chickens were graphically demonstrated on forest plots. Sources of variability in prevalence estimates were examined using a multivariate random-effects meta-regression in R Studio with the metafor package. Multivariate meta-regression was employed to assess the collective and individual effects of all predefined study-level characteristics simultaneously, while accounting for potential confounding among moderators. The meta-regression was treated as linear model on the logit-transformed prevalence data. The linear regression analysis was conducted to evaluate publication bias, and the funnel plots were used to visualize the publication bias.
Results
Eligibility of search results
A literature search yielded 1701 records, and 1613 articles were excluded due to duplications or deemed ineligible based on their title and abstract (Fig. 1). Full-texts of 88 articles were retrieved and assessed for eligibility based on the predetermined inclusion criteria, and 12 studies were ineligible. Thus, the remaining 76 articles met the inclusion criteria for review and meta-analysis. These were distributed across 17 African countries from five regions, of which West Africa (Ghana and Nigeria) contributed more studies (n = 28), followed by East Africa (Eritrea, Ethiopia, Kenya, and Tanzania) (n = 27), Southern Africa (Botswana, Lesotho, South Africa, Zambia, and Zimbabwe) and North Africa (Algeria, Egypt, Libya, Morocco, and Tunisia) both with 10 studies each, and the least studies were from Central Africa (Cameroon) (n = 1) (Supplementary Table S1).

Figure 1. PRISMA flow diagram showing the search and selection process.
Overall prevalence of gastrointestinal nematode species of free-range chickens
The frequency of nematode species affecting free-range chickens is presented in Table 1. Out of a total of 74 789 free-ranging chickens screened, 13 625 were infected with one or more GIT nematode species, and the overall pooled prevalence was 15% (95% CI: 13–18%) (Supplementary Table S1). A total of 27 nematode species were documented, and A. galli and H. gallinarum were the most recorded nematode species, with the former reported in all 76 studies and the latter reported in 68 studies. The highest prevalence of 100% was reported with Baruscapillaria (B.) obsignata in Zimbabwe, and the lowest prevalence of 0.2% was reported with B. obsignata in Lesotho and Syngamus (S.) trachea in Nigeria and Kenya, with 0.2% and 0.3%, respectively. The quality effects model revealed a significantly high degree of heterogeneity between studies (Q = 19 756.59, P <0.01), with an I 2 index of 98%.
Table 1. Frequency of nematode species affecting free-ranging chickens from 1993 to 2024

Prevalence of nematode infections in free-ranging chickens by region
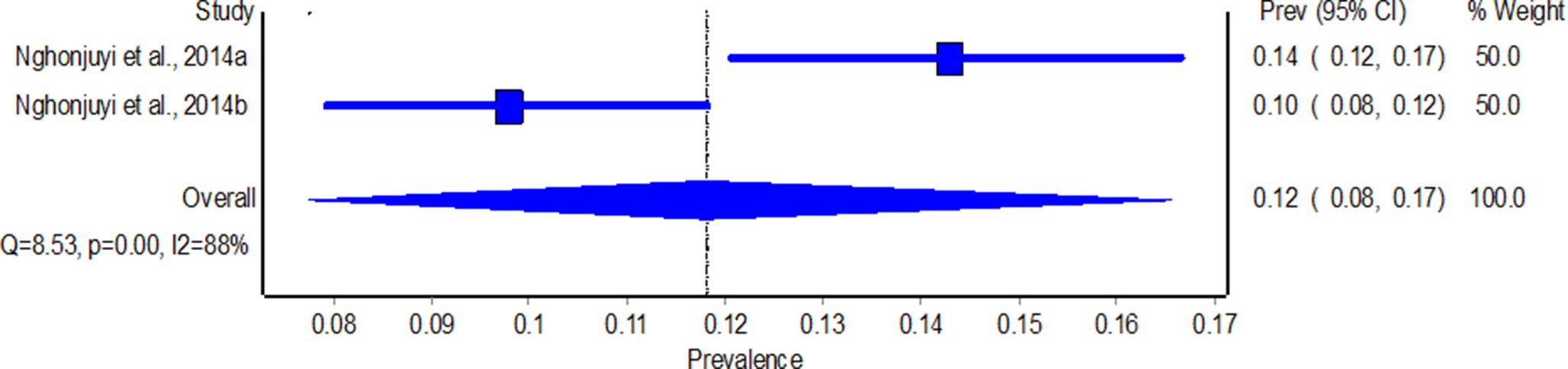
Southern African region recorded the highest pooled prevalence of GIT nematode infections in free-ranging chickens 22% (95% CI: 13–33%) (Fig. 2), followed by eastern Africa with 16% (95% CI: 13–20%) (Fig. 3), northern African region with 14% (95% CI: 9–21%) (Fig. 4), central Africa with 12% (95% CI: 8–17%) (Fig. 5), and western Africa recorded the least pooled prevalence of 5% (95% CI: 0–2) (Fig. 6). Western Africa had the highest species diversity (n = 23) and central Africa had the least (n = 2). The quality effects model revealed a significantly high degree of heterogeneity between regions, with eastern and southern Africa recording the highest heterogeneity (I 2 = 99%, P <0.01) (Fig. 2 and 3), followed by western Africa (I 2 = 97%, P <0.01) (Fig. 6), northern Africa (I 2 =96%, P <0.01) (Fig. 4), and central Africa (I 2 = 88%, P <0.01) (Fig. 5).

Figure 2. Forest plot for the prevalence of gastrointestinal nematode species in free-range chickens from Southern Africa from 1993 to 2024.

Figure 3. Forest plot for the prevalence of gastrointestinal nematode species in free-range chickens from Eastern Africa from 1993 to 2024.

Figure 4. Forest plot for the prevalence of gastrointestinal nematode species in free-range chickens from Northern Africa from 1993 to 2024.

Figure 5. Forest plot for the prevalence of gastrointestinal nematode species in free-range chickens from Central Africa from 1993 to 2024.

Figure 6. Forest plot for the prevalence of gastrointestinal nematode species in free-range chickens from Western Africa from 1993 to 2024.
Prevalence of gastrointestinal nematodes at family level
The prevalence of nematodes affecting free-range chickens was computed according to their families (Table 2), and the Tetrameridae family recorded the highest overall pooled prevalence of 46% (95% CI: 28–64%), while Syngamidae and Spiruridae families recorded the lowest pooled prevalence of 2% (95% CI: 1–4%) and 1% (95% CI: 0–3%), respectively. However, these families had low species diversity, with the Tetrameridae family consisting of two species, Tetrameres (T.) americana and T. fissispina, and the former species recorded the highest prevalence of 51.1% (Table 1). Syngamidae and Spiruridae families had one representative species each, S. trachea and Hartertia (Ha.) gallinarum, respectively. Results from the quality effects model across all nematode families revealed a high heterogeneity of I 2 > 80% (P <0.01), except for Spiruridae family, which recorded I 2 = 0% (P >0.01) (Table 2). It was not possible to conduct a meta-analysis for the family Thelaziidae due to the limited number of studies.
Table 2. Prevalence of nematode families affecting free-ranging chickens from 1993 to 2024

Prevalence of gastrointestinal nematode species in free-range chickens by time in years
Variation in the prevalence of gastrointestinal nematode species in free-range chickens was evaluated according to eras, and articles were divided into three groups: 1993–2002, 2003–2013, and 2014–2024. Results showed that most studies were conducted between 2014 and 2024, and the least between 1993 and 2002. Species diversity corresponded with the frequency of studies available, with the highest diversity noted during the period of 2014–2024, followed by 2003–2013, and the least during 1993–2002. However, the pooled prevalence of gastrointestinal nematode species in free-range chickens was highest during the era 1993–2002 (17%; 95% CI: 11–24%), followed by 2003–2013 and 2014–2024 (with 14%; 95% CI: 10–19% and 14%; 95% CI: 11–17%, respectively). Results from the quality effects model revealed a high heterogeneity in reports of GIT nematodes in free-range chickens over time, with all eras recording I 2 > 95% (P <0.01) (Table 3).
Table 3. Pooled prevalence of gastrointestinal nematode infection in free-ranging chickens in Africa based on different risk factors

Prevalence of gastrointestinal nematodes in the free-range chickens by diagnostic/detection methods
To detect the infections of the gastrointestinal nematodes in the free-range chickens, three diagnostic techniques were used individually or in combination, viz necropsy, coproscopy, and molecular (PCR). Results showed that most studies used necropsy (n = 48) to detect infections, while 27 studies used coproscopy, 4 studies used a combination of necropsy and molecular (Table 3), and only one study used combined coproscopy and molecular methods and was not included in the meta-analysis. The pooled prevalence of gastrointestinal nematodes in free-range chickens based on techniques was higher with necropsy, with 17% (95% CI: 14–20%), followed by necropsy and molecular, with 15% (95% CI: 3–33%), and lastly coproscopy with 10% (95% CI: 7–14%) (Table 3). Results revealed a high heterogeneity between necropsy and coproscopy, both with I 2 = 98% (P <0.01), and necropsy and molecular I 2 = 92% (P <0.01) (Table 3).
Prevalence of gastrointestinal nematode species in free-range chickens by sex, age, and mixed infections
The prevalence of GIT nematode species in free-range chickens was evaluated according to sex, age, and mixed infections. Seven studies reported on the infection of the nematode species with regard to sex of the chickens, three on age, and six on mixed infections (Table 3; Fig. 7). The pooled prevalence of GIT nematodes in free-range chickens based on sex was higher in females (46%; 95% CI: 27–66%) compared to males (45%; 95% CI: 30–60%). Heterogeneity was high for both sexes (I 2 >90%, P <0.01) (Table 3) and young (≤18 weeks) free-range chickens recorded a higher pooled prevalence of GIT nematodes (45%; 95% CI: 21–71%) compared to adults (>18 weeks) (42%; 95% CI: 14–72%). However, a high heterogeneity was recorded in adult free-ranging chickens (I 2 = 98%, P <0.01) compared to the one recorded for the young ones (I 2 = 96%, P <0.01) (Table 3). Pooled prevalence for mixed infections of GIT nematode species in free-range chickens recorded 11% (95% CI: 6–18%), and a relatively average heterogeneity of I 2 = 72% (P <0.01) (Fig. 7).

Figure 7. Forest plot for the prevalence of gastrointestinal nematode species in free-range chickens according to mixed infections.
Publication bias and multiple linear regression
Asymmetric funnel plots were observed, indicating the presence of publication bias (Supplementary Figure S1), which may be due to a small-sample-size bias or publication bias in the articles. The multivariate meta-regression model, incorporating region, parasite family, study period, and diagnostic method, provided a statistically significant fit to the data (QM = 194.57, df = 4, P <0.0001). This confirms that the selected moderators collectively have a significant effect on the observed prevalence estimates. The meta-regression analysis revealed that several nematode families were significantly associated with higher effect sizes, including Ascaridiidae (estimate = 2.08, P <0.001), Heterakidae (estimate = 1.56, P <0.001), and Tetrameridae (estimate = 2.14, P <0.001), while Gongylonematidae and Subuluridae showed weaker but significant positive associations. Among diagnostic methods, necropsy alone yielded a significant positive effect (estimate = 0.57, p = 0.004), whereas coproscopy-based methods did not significantly influence the estimates. No significant differences were observed across study periods or African regions (Supplementary Table S2).
Discussion
The overall prevalence of the nematode species infection in free-ranging chickens in Africa was 15%. The prevalence could have been influenced by several factors, such as distribution and availability of intermediate hosts and/or environmental contamination with infective parasite eggs or larvae (Opara et al., Reference Opara, Osowa and Maxwell2014). Additionally, the GIT nematode prevalence might have been attributed to the differences in the feeding patterns of the birds, the incidence of the infective stages, and the intermediate hosts of the parasites picked up by the birds or found in places where the chickens free ranged.
Tetrameridae was found to be a common nematode family parasitising free-range chickens, particularly species from the genus Tetrameres. Agüero et al. (Reference Agüero, Gilardoni, Cremonte and Diaz2016) reported the Tetrameres spp. to have worldwide distribution, and one of its representative species, T. americana, has been reported as a common parasite of free-range chickens in tropical and subtropical areas with high prevalence (Fink et al. Reference Fink, Permin, Magwisha and Jensen2005). In tropical Africa, Tetrameres genus usually results in high prevalence infections in free-ranging chickens (Wamboi et al. Reference Wamboi, Waruiru, Mbuthia, Nguhiu and Bebora2020), and the present study recorded a pooled prevalence of 54% for the Tetrameridae, which included species such as T. americana and T. fissispina. According to Magwisha et al. (Reference Magwisha, Kassuku, Kyvsgaard and Permin2002), the observed high prevalence from this family may be attributed to the parasite’s transmission dynamics, which involves a variety of arthropods that may be season-dependent, aiding in its proliferation. Tetrameres americana was documented in 12 countries from eastern Africa, western Africa, and southern Africa (Chilinda et al. Reference Chilinda, Lungu, Phiri, Chibinga and Simbaya2020; Fink et al. Reference Fink, Permin, Magwisha and Jensen2005; Jinga et al. Reference Jinga, Munosiyei, Bobo and Tambura2012; Magwisha et al. Reference Magwisha, Kassuku, Kyvsgaard and Permin2002; Permin et al. Reference Permin, Magwisha, Kassuku, Nansen, Bisgaard, Frandsen and Gibbons1997; Permin et al. Reference Permin, Esmann, Hoj, Hove and Mukaratirwa2002), whereas T. fissispina was documented in three countries from eastern Africa and western Africa (Magwisha et al. Reference Magwisha, Kassuku, Kyvsgaard and Permin2002; Olwande et al. Reference Olwande, Okuthe, Ogara and Bebora2016; Poulsen et al. Reference Poulsen, Permin, Hindsbo, Yelifari, Nansen and Bloch2000). These two species primarily infect the proventriculus and oesophagus of chickens (Mungube et al. Reference Mungube, Bauni, Tenhagen, Wamae, Nzioka, Muhammed and Nginyi2008; Permin et al. Reference Permin, Magwisha, Kassuku, Nansen, Bisgaard, Frandsen and Gibbons1997), which may potentially cause severe pathological lesions within these organs.
In contrast, Syngamidae and Spiruridae had the least recorded prevalence, with most reported studies for Syngamidae recording S. trachea as the main species affecting free-range chickens, and all three studies for Spiruridae reported Ha. gallinarum. Despite the low prevalence, S. trachea showed a wider distribution, recorded in 19 countries across the eastern, western, and southern regions, whereas Ha. gallinarum was recorded in three countries from the western region of Africa (Tay et al. Reference Tay, Emikpe, Ockling, Folitse and Jarikre2017). Syngamus trachea infects birds through multiple transmission mechanisms, such as directly ingesting third-stage larvae (L3) from embryonated eggs or free L3 in contaminated environments (soil, feed, water) or indirectly through ingestion of paratenic hosts such as earthworms and other invertebrates, including snails, slugs, beetles, and flies (Gethings et al. Reference Gethings, Sage and Leather2016). According to Ngongeh et al. (Reference Ngongeh, Onyeabor and Erumaka2012), the high pre-parasitic stage of the parasite can be maintained within the environment due to bionomic factors such as high temperatures and high humidity. However, the low infection percentage revealed seems to be due to less favourable conditions associated with free-ranging poultry management systems, which is a possibility, according to Thapa et al. (Reference Thapa, Hinrichsen, Brenninkmeyer, Gunnarsson, Heerkens, Verwer, Niebuhr, Willett, Grilli and Thamsborg2015), who further highlighted the complexities of on-farm transmission dynamics in relation to helminth infections.
The most reported nematode species were A. galli and H. gallinarum, which occurred in all reported African regions. These species have been reported globally with a high prevalence (García Cuadrado et al. Reference García Cuadrado, Martínez-Moreno, Zafra Leva and Acosta García2021; Kaufmann et al. Reference Kaufmann, Das, Sohnrey and Gauly2011; Thapa et al. Reference Thapa, Hinrichsen, Brenninkmeyer, Gunnarsson, Heerkens, Verwer, Niebuhr, Willett, Grilli and Thamsborg2015; Wuthijaree et al. Reference Wuthijaree, Lambertz, Vearasilp, Anusatsananun and Gauly2019). Similarity in rate of infection could be accounted for by their co-occurrence and co-infection as both parasites have similar life cycles and have the same environmental conditions requirements for transmission (Das et al. Reference Das, Hennies, Sohnrey, Rahimian, Wongrak, Stehr and Gauly2017). Ascaridia galli and H. gallinarum are among poultry’s most common and important helminth infections, with the former having the capability of causing diarrhoea, poor growth, egg drop in laying chickens, and in severe infections, blood loss due to intestinal mucosa damage, secondary bacterial infections, and intestinal obstruction in cases of heavy infections (Ashenafi and Eshetu Reference Ashenafi and Eshetu2004). Comparatively, H. gallinarum is non-pathogenic; however, it plays a crucial role as a potential vector of Histomonas meleagridis, a highly pathogenic etiologic agent of ‘blackhead disease’ (histomoniasis) in many avian species, including chickens (Pinckney et al. Reference Pinckney, Coomansingh, Bhaiyat and Sharma2008). Ashenafi and Eshetu (Reference Ashenafi and Eshetu2004) noted a colossal infection by H. gallinarum in free-range backyard chickens which led to severe debility, morbidity, and mortality in extreme cases.
Southern Africa recorded the highest pooled prevalence of gastrointestinal nematode species of free-ranging chickens, and according to Marumbwa et al. (Reference Marumbwa, Cho and Chirwa2019), the subtropical and temperate zones of this region, along with its moderate to high rainfall and optimal temperature range (20–30°C) create ideal conditions which may accelerate the development and survival of nematode eggs and larvae on soil and vegetation (Wuthijaree et al. Reference Wuthijaree, Lambertz, Vearasilp, Anusatsananun and Gauly2019). Furthermore, Marumbwa et al. (Reference Marumbwa, Cho and Chirwa2019) also highlighted that southern Africa faces climate variability where the El Niño-induced droughts, followed by floods, can stress chickens and weaken their immune systems, leading to increased parasite proliferation with the return of the rains. Despite recording the highest pooled prevalence, southern Africa had a lower GIT nematode species diversity (n = 13) compared to the western Africa region (n = 23). However, individual species prevalence ranges differed distinctly, with southern Africa reaching as high as 100%, while western Africa ranged from 6% to 16%. Several authors noted that these variations in prevalence may be linked to seasonality and region, which also influence the occurrence and intensities of some species (Ashenafi and Eshetu Reference Ashenafi and Eshetu2004; Jinga et al. Reference Jinga, Munosiyei, Bobo and Tambura2012; Magwisha et al. Reference Magwisha, Kassuku, Kyvsgaard and Permin2002). Wuthijaree et al. (Reference Wuthijaree, Lambertz, Vearasilp, Anusatsananun and Gauly2019) also reported that adequate moisture and temperature in the environment are indeed the most determinant factors for the development of the infective stage of the helminth parasite, and that may influence the epidemiology of parasite infection.
Despite most studies and the highest species diversity recorded between 2014 and 2024, the highest pooled prevalence of nematode species in free-range chickens was demonstrated between 1993 and 2002. A lack of proper vector-controlling methods and the inadequate supply of vaccine administration to free-range chicken production could have played a role in attaining high infections during that particular period (Malatji et al. Reference Malatji, Tsotetsi, Van Marle-Koster and Muchadeyi2016). Conversely, frequently communicated information from the campaigns on the sustainable development goals promoted globally, regionally, and nationally could have contributed to the lower prevalence in the recent decade (2014–2024). This increase is driven by the high demand for free-range chickens as reported in many regions in Africa (Malatji et al. Reference Malatji, Tsotetsi, Van Marle-Koster and Muchadeyi2016; Mtileni et al. Reference Mtileni, Muchadeyi, Maiwashe, Phitsane, Halimani and Chimonyo2009; Muchadeyi et al. Reference Muchadeyi, Sibanda, Kusina, Kusina and Makuza2004) and awareness of the economic impact of nematode parasites on the health of free-range chicken production (Berhe et al. Reference Berhe, Mekibib, Bsrat and Atsbaha2019; Mukaratirwa et al. Reference Mukaratirwa, Hove, Esmann, Hoj, Permin and Nansen2001; Mwale and Masika Reference Mwale and Masika2011).
The pooled prevalence of gastrointestinal nematodes in free-range chickens based on sex was slightly higher in females (46%) compared to males (45%). Physiological, morphological, and behavioural differences between females and males could influence the susceptibility to parasite infections (Kaboudi et al. Reference Kaboudi, Rhif, Romdhane, Abdallah and Dhibi2024). Furthermore, the higher infection in females could be attributed to the fact that male chickens under any condition of management tend to court the females by allowing them to feed first before the males, and in so doing, they are more likely to easily acquire infective pathogens than the males (Opara et al. Reference Opara, Osowa and Maxwell2014). In addition, the females are known to be more voracious in their feeding habits, especially during egg production, compared to males, which remain largely selective (Mohammed et al. Reference Mohammed, Ojo, Opara, Jegede and Agbede2019). However, according to Kaboudi et al. (Reference Kaboudi, Rhif, Romdhane, Abdallah and Dhibi2024), the prevalence of helminth infections did not differ between female and male birds, indicating that helminth species have no natural affinity for either sex of host chicken.
Age is also considered a risk factor associated with the occurrence of GIT helminth infections (Kaboudi et al. Reference Kaboudi, Rhif, Romdhane, Abdallah and Dhibi2024). The pooled prevalence of GIT nematodes was higher in young free-range chickens compared to adults. Younger animals are highly susceptible to helminth infections compared to adults, possibly due to their immature immune system and poor acclimatisation to their immediate environment (Ola-Fadunsin et al. Reference Ola-Fadunsin, Uwabujo, Sanda, Ganiyu, Hussain, Rabiu, Elelu and Alayande2019).
The quality effects model revealed a relatively average heterogeneity with mixed infections, and the pooled prevalence according to mixed infections of gastrointestinal nematodes was ascertained. The endemicity of mixed infections could be related to environmental factors and poor hygienic conditions in the backyard system, which are favourable for developing parasitic infections (Kaboudi et al. Reference Kaboudi, Rhif, Romdhane, Abdallah and Dhibi2024). Gastrointestinal tract helminths are either commonly over-dispersed or aggregated within the host population. Aggregation can be attributed to several factors, including heterogeneity in the host behaviour influencing the uptake of infective stages and spatial heterogeneity in the distribution of infective stages (Hussen et al. Reference Hussen, Chacka, Deneke and Bitew2012). Mixed infections in rural free-ranging chickens have been reported in several studies (Mohammed et al. Reference Mohammed, Ojo, Opara, Jegede and Agbede2019; Ohaeri and Okwum Reference Ohaeri and Okwum2013; Yoriyo et al. Reference Yoriyo, Adang, Fabiyi and Adamu2008). According to Smyth (Reference Smyth1976), the establishment of mixed infection involving one or multiple species might be attributed to the food selected and consumed by the chicken at a particular time, which may be contaminated and ultimately leads to infection.
Substantial heterogeneity was observed between prevalence studies across the continent. The potential source of variation, as indicated by the meta-regression results, was the differences in diagnostic/detection methods employed when carrying out the studies, which showed a significant difference in the pooled prevalence of nematode infections. The highest prevalence was attained through necropsy compared to coproscopy. Our results concur with those attained by Shifaw et al. (Reference Shifaw, Feyera, Walkden-Brown, Sharpe, Elliott and Ruhnke2021), where they found that the widely used diagnostic method was necropsy examination. Necropsy is known for its accuracy and reliability for detection and identification of GIT helminths in chickens (Macklin Reference Macklin2013); however, the method may not be the most economically viable method for routine diagnosis, as it involves sacrificing individual chickens. Varying prevalence rates noted in the cited studies could have been a result of them being conducted in different seasons, use of different ages of the chickens, environmental sources with varying availability of intermediate hosts, and study chickens with different individual host resistance (Sahu et al. Reference Sahu, Anand and Sinha2016).
Strengths and limitations
The current study is the first systematic review to document the pooled prevalence of GIT nematode species in free-ranging chickens from various sub-regions of Africa covering a broad research period. In addition, the strengths of the systematic review and meta-analysis were adherence to the criteria and the principles of the PRISMA guidelines, the provision of estimates based on a quality effects model, and the analysis conducted per subgroup for heterogeneity. Despite using a standardised analysis process, it was difficult to achieve a consistent meta-analysis due to the different attributes that were included for the accountability towards the heterogeneity. Therefore, there was a limitation of publication bias. A subsequent number of studies in this review primarily identified most of the nematode species based on morphological approaches. The inability to identify nematodes to the species level led to the exclusion of several articles in the meta-analysis that could have aided in the estimation of the prevalence of the GIT nematodes. The low number of published articles that were eligible for the review was not evenly distributed across the subregions of Africa. Meta-analysis could not be conducted on some of the nematode families due to the limited number of studies carried out. Thus, in this review, the prevalence data may not fully represent the prevalence of GIT nematode species affecting free-ranging chickens across Africa.
Conclusions
This review provides insights into the occurrence and prevalence of GIT nematode infections in free-ranging chickens across African sub-regions. The study highlighted the lessening of parasitic nematode species burden in free-range chickens; however, there is a higher GIT nematode species diversity and a wider distribution of the most prevalent species across Africa. Such evidence indicates a serious long-term and regional threat to sustainable free-range chicken production due to increased pathogenic potential and subsequent complications in disease control, ultimately resulting in significant reductions in flock productivity. The study also recommended future studies that involve the integration of both morphological and molecular techniques to increase the accuracy of parasite identification to the species level. Furthermore, this will aid in determining species-specific distribution and regional species biodiversity, which will assist in investigating the transmission dynamics of these parasites and the development of sustainable control strategies.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0022149X26101400.
Acknowledgements
The authors would like to acknowledge the University of KwaZulu-Natal librarian and library staff for assisting with full-text reprints of some articles.
Author contribution
I.W. conceptualized the study under the guidance of S.M., D.T., and M.P.M. I.W. developed the concept note, conducted the search, selected studies, and wrote the first draft of the manuscript under M.P.M.’s guidance. S.M and M.M.P independently confirmed the article selected and data extraction. I.N. performed the statistical analysis and prepared figures and tables. All authors contributed to the article, agreed on the final draft, and approved the submitted version.
Financial support
No financial assistance was received for this study.
Competing interests
The authors declare no conflict of interest.