Highlights
What is already known?
-
• ITC, including population-adjusted indirect comparison (PAIC) and network meta-analysis, are widely used to estimate the comparative effectiveness of interventions when direct head-to-head trials are unavailable.
-
• HR are commonly used as effect measures in ITCs involving time-to-event outcomes.
What is new?
-
• We introduce the concept of statistical transitivity as a key criterion for evaluating effect measures in ITC and highlight its importance in interpreting comparative effectiveness.
-
• Through theoretical proofs and practical examples, we demonstrate the risks of using HRs in ITC, especially when the proportional hazard assumption does not hold across all treatment arms.
Potential impact for RSM readers
-
• We caution against the indiscriminate use of HRs in ITC for time-to-event outcomes and recommend alternative measures with better transitivity properties, such as RMST, landmark survival probability, or the AH-SW differences or ratios.
-
• We present a decision-making flowchart for survival outcome ITCs and emphasize explicit evaluation of both clinical and statistical transitivity to assess validity.
1 Introduction
In medical product development, there are often multiple approved treatments for the same disease condition. Comparing their relative effectiveness within specific populations is crucial, particularly for securing reimbursement from public or private sectors, where a drug must be evaluated against the standard of care (SoC) through a health technology assessment (HTA).Reference Phillippo, Ades, Dias, Palmer, Abrams and Welton1 While randomized controlled trials (RCTs) are the gold standard for treatment comparisons, it is often impractical to conduct head-to-head trials for all available drugs. As an alternative, indirect treatment comparison (ITC) methods leverage existing trial data to compare treatments.
In a typical anchored indirect comparison of drug A versus drug B, a common comparator C such as a placebo, is present in both trials (referred to as the AC and BC trials). Given the potential imbalance of effect modifiers, ITC requires balancing the covariate distributions across the two trial populations. However, due to data availability constraints, individual participant data (IPD) are often available for only one trial, while the other provides only aggregate-level data (AgD). To address this issue, researchers have developed the population-adjusted indirect comparison (PAIC) methods, which account for covariate distribution differences between the AC and BC trials. Among various PAIC approaches,Reference Caro and Ishak2, Reference Signorovitch, Wu and Yu3 matching-adjusted indirect comparison (MAIC) has gained popularity in the HTA submissions.Reference Phillippo, Dias, Elsada, Ades and Welton4, Reference Jiang, Liu and Alemayehu5 In an anchored indirect comparison of drug A versus drug B, MAIC estimates a set of balancing weights for the AC trial (where IPD is available) to ensure the weighted population matches the BC trial population based on summary statistics from AgD. Comparative effectiveness is then calculated by subtracting or dividing the weighted relative effect of drug A versus drug C by the relative effect of drug B versus drug C.
An essential requirement for the validity of indirect comparison between drug A and B through a common comparator C is the transitivity of relative effects. This assumes that the true relative effect of drug A versus drug B equals the difference (or ratio) between the true relative effects of drug A versus drug C and drug B versus drug C. This assumption (sometimes described as exchangeability, consistency, or similarity across studies) has been well known in other ITC scenarios such as network meta-analysis.Reference Song6– Reference Jansen and Naci11 SalantiReference Salanti10 highlights two key requirements for the validity of transitivity in ITC. First, the drug C must be comparable in both the AC trial and BC trials. For instance, if drug C represents the current SoC at the time of the trials, differences in SoC due to advancements over time could violate transitivity if the trials are not conducted simultaneously. Second, all effect modifiers must be balanced between the AC trial and BC trial populations. Salanti et al.Reference Salanti, Marinho and Higgins9 provide a cautionary example showing that violations of transitivity, particularly differences in study populations, can fundamentally compromise network meta-analysis (NMA) results. Song et al.Reference Song6 systematically compared results from direct head-to-head meta-analyses with those from adjusted indirect comparisons and underscored that the validity of such comparisons relies on the similarity of the trial populations and interventions. Ades et al.Reference Ades, Welton, Dias, Phillippo and Caldwell8 discussed the subtle differences between the network meta-analysis and pairwise meta-analysis and argued that their validity depends on the exchangeability (encompassing concepts of homogeneity, similarity, and consistency) of included trials. Cipriani et al.Reference Cipriani, Higgins, Geddes and Salanti12 underscores that the reliability of network meta-analysis depends critically on the assumption that studies are comparable in all important respects and provides guidance for evaluating, documenting, and, where necessary, adjusting for differences to support valid indirect comparisons. Donegan et al.Reference Donegan, Williamson, Gamble and Tudur-Smith13 emphasize that systematically evaluating and transparently reporting population similarity are essential for ensuring the reliability of indirect comparisons in evidence synthesis.
Existing literature discussed the transitivity assumption primarily from a scientific or clinical perspective, particularly focused on the similarity and consistency of the interventions, study populations, or effect measures. In this article, we further examine the transitivity assumption from a statistical perspective. Specifically, for a relative effect to be transitive, the effect measure itself must be a statistically transitive measurement. In other words, the validity of the ITC depends not only on the specific scientific background, but also on the selection of statistical measurement. In Section 2, we give a formal definition of statistical transitivity in the context of ITC and offer insights into its implications.
This article focuses on anchored ITC for time-to-event outcomes. The hazard ratio (HR)Reference Cox14 is one of the most popular measures for comparing the efficacy of two drugs in survival analysis. In the comparison of drug A versus drug C, the Cox model treats the baseline hazard (i.e., the hazard for drug C) as a nuisance parameter and only estimates the HR between the two drugs. Our literature review indicates that most existing studies use the HR as the metric for ITC when dealing with time-to-event outcomes. For example, Aouni et al.Reference Aouni, Gaudel-Dedieu and Sebastien15 compared different matching strategies and penalty factors for MAIC, using the ratio of HR as the indirect comparison measure. Remiro-Azocar et al.Reference Remiro-Azócar, Heath and Baio16 and Weber et al.Reference Weber, Jensen and Kieser17 conducted comprehensive simulation studies to compare various indirect comparison methods, while both of which used the log HR as the effect measure. Leahy and WalshReference Leahy and Walsh18 proposed using MAIC in Bayesian network meta-analysis for covariate adjustment with an HR model. Park et al.Reference Park, Campbell and Towle19 introduced a doubly robust approach for indirect comparison with time-to-event outcomes, also using HR as the comparison metric. Beyond MAIC, HR has also been widely adopted as the primary comparison metric in network meta-analysis.Reference Nazarzadeh, Bidel and Canoy20– Reference Zhao, He and Wang24
While we acknowledge and appreciate these contributions to ITC literature, this cautionary note highlights that the HR is not a transitive measure and should be used with caution for ITC. Alternatively, we propose using the restricted mean survival time (RMST) difference, the landmark survival probability difference (or ratio) or the average hazard with survival weights (AH-SW) difference as more appropriate measures.
2 The statistical transitivity of an effect measurement
In an anchored ITC, transitivity refers to the property that the comparative effectiveness of drugs A versus B can be inferred through a common comparator C, that is,
![]() ${\mu}_{AB}={\mu}_{AC}-{\mu}_{BC}$
(or
${\mu}_{AB}={\mu}_{AC}-{\mu}_{BC}$
(or
![]() ${\mu}_{AB}={\mu}_{AC}/{\mu}_{BC}$
for a ratio effect measure) where
${\mu}_{AB}={\mu}_{AC}/{\mu}_{BC}$
for a ratio effect measure) where
![]() ${\mu}_{AB}$
represents the comparative effectiveness measurement (e.g., mean difference, risk difference, or risk ratio) between drug A and drug B.Reference Salanti10 However, the validity of transitivity in an anchored indirect comparison relies on several factors: 1) the common comparator drug C must be consistent (i.e., similar) in both the AC and BC trials, 2) all effect modifiers must be balanced (after adjustment) in the populations of the AC and BC trials, and 3) the effect measurement itself must be statistically transitive (the focus of this article).
${\mu}_{AB}$
represents the comparative effectiveness measurement (e.g., mean difference, risk difference, or risk ratio) between drug A and drug B.Reference Salanti10 However, the validity of transitivity in an anchored indirect comparison relies on several factors: 1) the common comparator drug C must be consistent (i.e., similar) in both the AC and BC trials, 2) all effect modifiers must be balanced (after adjustment) in the populations of the AC and BC trials, and 3) the effect measurement itself must be statistically transitive (the focus of this article).
The first requirement can be addressed by carefully examining the design and timing of both trials. For a detailed discussion, readers are referred to Salanti.Reference Salanti10 Various ITC approachesReference Signorovitch, Wu and Yu3, Reference Phillippo, Dias and Ades25– Reference Jackson, Rhodes and Ouwens28 have been proposed to address the second requirement, typically assuming that all relevant effect modifiers are included in the adjustment. For a comprehensive review of MAIC approaches, readers can refer to Jiang et al.Reference Jiang, Cappelleri, Gamalo, Chen, Thomas and Chu29
This article primarily focuses on the third requirement: the statistical transitivity of an effect measurement. To illustrate this concept, consider a randomized three-arm trial that includes drugs A, B, and C. First, we define two key concepts in this context: the direct comparison and the indirect comparison. The direct comparison of drug A versus drug B, denoted as
![]() ${\mu}_{AB}^D$
, is calculated directly from the outcome data of drugs A and B using metrics such as risk difference, HR, or other measures. In contrast, the indirect comparison, denoted as
${\mu}_{AB}^D$
, is calculated directly from the outcome data of drugs A and B using metrics such as risk difference, HR, or other measures. In contrast, the indirect comparison, denoted as
![]() ${\mu}_{AB}^I$
, is derived by contrasting the effects of drug A versus drug C and drug B versus drug C (i.e.,
${\mu}_{AB}^I$
, is derived by contrasting the effects of drug A versus drug C and drug B versus drug C (i.e.,
![]() ${\mu}_{AB}^I={\mu}_{AC}^D-{\mu}_{BC}^D$
or
${\mu}_{AB}^I={\mu}_{AC}^D-{\mu}_{BC}^D$
or
![]() ${\mu}_{AB}^I={\mu}_{AC}^D/{\mu}_{BC}^D$
). Notably, a three-arm trial allows for both direct and indirect comparisons of drug A versus drug B. The indirect comparison in a three-arm trial mirrors the ITC by hypothetically creating the AC and BC trials, ensuring that drug C is identical in both trials.
${\mu}_{AB}^I={\mu}_{AC}^D/{\mu}_{BC}^D$
). Notably, a three-arm trial allows for both direct and indirect comparisons of drug A versus drug B. The indirect comparison in a three-arm trial mirrors the ITC by hypothetically creating the AC and BC trials, ensuring that drug C is identical in both trials.
The primary rationale for ITC is to estimate the comparative effectiveness of two treatments when direct comparison results are unavailable. Therefore, we ideally expect the direct comparison of drug A versus drug B to match the indirect comparison of drug A versus drug B with a common comparator drug C. This leads to the following definition of the statistical transitivity of a measurement.
Definition: (Statistical transitivity of an effect measurement).
A comparative effect measurement, denoted as
![]() $\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^B\right\}}_{i=1}^{n_B}\right)$
, where
$\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^B\right\}}_{i=1}^{n_B}\right)$
, where
![]() ${\left\{{Y}_i^A\right\}}_{i=1}^{n_A}$
represents the data from drug A and
${\left\{{Y}_i^A\right\}}_{i=1}^{n_A}$
represents the data from drug A and
![]() ${\left\{{Y}_i^B\right\}}_{i=1}^{n_B}$
represents the data from drug B, is considered transitive in an ITC if, in the three-arm trial scenario, the direct comparison of drug A versus drug B is identical to the indirect comparison of drug A versus drug B. For a ratio effect measure, this means that
${\left\{{Y}_i^B\right\}}_{i=1}^{n_B}$
represents the data from drug B, is considered transitive in an ITC if, in the three-arm trial scenario, the direct comparison of drug A versus drug B is identical to the indirect comparison of drug A versus drug B. For a ratio effect measure, this means that
![]() $\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^B\right\}}_{i=1}^{n_B}\right)=\frac{\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)}{\mu \left({\left\{{Y}_i^B\right\}}_{i=1}^{n_B},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)}$
for arbitrary data of drugs, A, B, and C.
$\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^B\right\}}_{i=1}^{n_B}\right)=\frac{\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)}{\mu \left({\left\{{Y}_i^B\right\}}_{i=1}^{n_B},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)}$
for arbitrary data of drugs, A, B, and C.
Note that the indirect comparison of drug A versus drug B can also be expressed as the difference (instead of the ratio) of two treatment effects, in which case,
![]() $\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^B\right\}}_{i=1}^{n_B}\right)=\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)-\mu \left({\left\{{Y}_i^B\right\}}_{i=1}^{n_B},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)$
, depending on the chosen effect measure. It is evident that many effect measurements used in ITC are transitive under this definition. For example, the risk/mean difference
$\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^B\right\}}_{i=1}^{n_B}\right)=\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)-\mu \left({\left\{{Y}_i^B\right\}}_{i=1}^{n_B},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)$
, depending on the chosen effect measure. It is evident that many effect measurements used in ITC are transitive under this definition. For example, the risk/mean difference
![]() $\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)=\frac{1}{n_A}\sum \limits_{i=1}^{n_A}{Y}_i^A-\frac{1}{n_B}\sum \limits_{i=1}^{n_B}{Y}_i^B$
is a transitive measurement. In Section 3, we will show that the HR is not a transitive measurement for time-to-event outcomes and thus should be used with caution in ITC.
$\mu \left({\left\{{Y}_i^A\right\}}_{i=1}^{n_A},{\left\{{Y}_i^C\right\}}_{i=1}^{n_C}\right)=\frac{1}{n_A}\sum \limits_{i=1}^{n_A}{Y}_i^A-\frac{1}{n_B}\sum \limits_{i=1}^{n_B}{Y}_i^B$
is a transitive measurement. In Section 3, we will show that the HR is not a transitive measurement for time-to-event outcomes and thus should be used with caution in ITC.
We emphasize that the transitivity of measurements is crucial in ITCs. Transitivity refers to the property that allows us to integrate over the common comparator C when comparing drugs A and B indirectly, provided all other assumptions are met. If a measurement is transitive, then the information from comparator C can be completely accounted for, assuming that 1) drug C is similar in both the AC and BC trials, and 2) there is no difference in the distribution of effect modifiers in the populations of the AC and BC trials.
However, if a measurement is not statistically transitive, then the choice of comparator C can affect the result of indirect comparison of A versus B, even in the ideal scenario of a randomized three-arm trial. Therefore, we recommend that indirect comparisons be performed only using statistically transitive measurements whenever possible.
3 HR should be used with caution in indirect treatment comparisons
3.1 HR derived from the Cox Proportional Hazard (PH) model is not a transitive measurement: an illustrative example
In this section, we focus on the transitivity of the HR within the context of ITC. Consider a three-arm trial with arms A, B, and C. Denote the hazard functions for drugs A, B, and C as
![]() ${h}_A(t)$
,
${h}_A(t)$
,
![]() ${h}_B(t)$
, and
${h}_B(t)$
, and
![]() ${h}_C(t),$
respectively. The “model free” HR
${h}_C(t),$
respectively. The “model free” HR
![]() ${\lambda}_{AB}(t)$
at each specific time point
${\lambda}_{AB}(t)$
at each specific time point
![]() $t$
can be defined as
$t$
can be defined as
![]() ${\lambda}_{AB}(t)=\frac{h_A(t)}{h_B(t)}$
. It should be noted that the HR function,
${\lambda}_{AB}(t)=\frac{h_A(t)}{h_B(t)}$
. It should be noted that the HR function,
![]() $\lambda (t)$
, is a transitive measurement if each hazard function is estimated independently (e.g., using the Kaplan–Meier estimator):
$\lambda (t)$
, is a transitive measurement if each hazard function is estimated independently (e.g., using the Kaplan–Meier estimator):
 $$\begin{align*}{\widehat{\lambda}}_{AB}(t)=\frac{{\widehat{h}}_A(t)}{{\widehat{h}}_B(t)}=\frac{{\widehat{h}}_A(t)}{{\widehat{h}}_C(t)}/\frac{{\widehat{h}}_B(t)}{{\widehat{h}}_C(t)}=\frac{{\widehat{\lambda}}_{AC}(t)}{{\widehat{\lambda}}_{BC}(t)}\end{align*}$$
$$\begin{align*}{\widehat{\lambda}}_{AB}(t)=\frac{{\widehat{h}}_A(t)}{{\widehat{h}}_B(t)}=\frac{{\widehat{h}}_A(t)}{{\widehat{h}}_C(t)}/\frac{{\widehat{h}}_B(t)}{{\widehat{h}}_C(t)}=\frac{{\widehat{\lambda}}_{AC}(t)}{{\widehat{\lambda}}_{BC}(t)}\end{align*}$$
for any
![]() $t$
. However, the HR function
$t$
. However, the HR function
![]() ${\lambda}_{AB}(t)$
is hard to interpret and thus rarely reported in real clinical trials. Instead, it is common to assume
${\lambda}_{AB}(t)$
is hard to interpret and thus rarely reported in real clinical trials. Instead, it is common to assume
![]() $\lambda (t)$
does not vary with time
$\lambda (t)$
does not vary with time
![]() $t$
(i.e., the Cox PH assumption) and defines this common ratio as HR
$t$
(i.e., the Cox PH assumption) and defines this common ratio as HR
![]() $\lambda$
. In the remainder of this article, “hazard ratio” refers to this constant
$\lambda$
. In the remainder of this article, “hazard ratio” refers to this constant
![]() $\lambda$
rather than the time-varying
$\lambda$
rather than the time-varying
![]() $\lambda (t)$
.
$\lambda (t)$
.
We begin by demonstrating that the HR
![]() $\lambda$
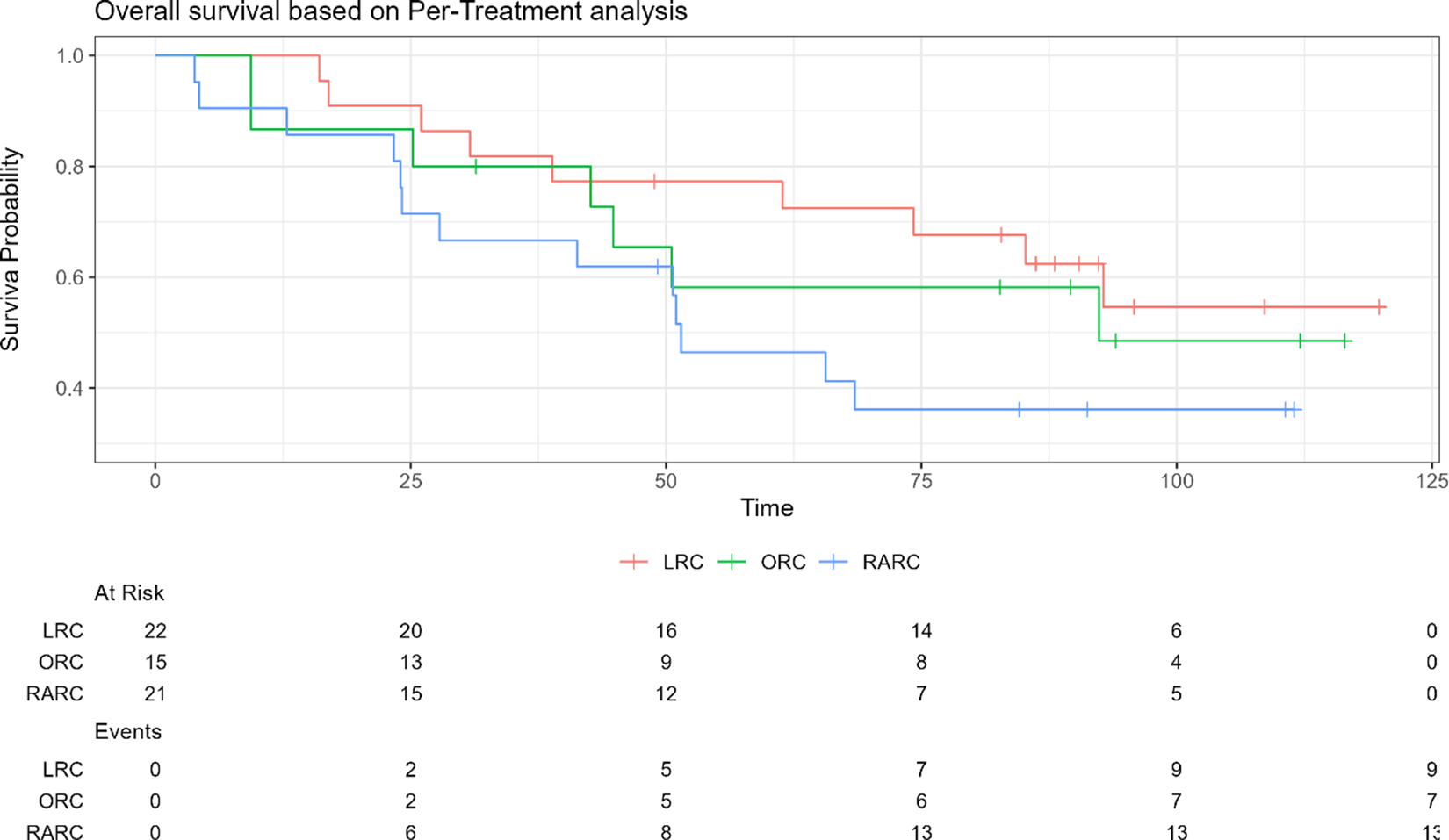
is not a transitive measurement through an illustrative example involving bladder cancer. Open radical cystectomy (ORC) is widely regarded as the gold standard treatment for patients with muscle-invasive bladder cancer. Recent advancements in medical technology have introduced minimally invasive techniques, such as laparoscopic radical cystectomy (LRC) and robotic-assisted radical cystectomy (RARC).Reference Bochner, Dalbagni and Sjoberg30 In this example, we use data from a randomized three-arm trial comparing ORC, LRC, and RARC in terms of the overall survival outcomes.Reference Khan, Omar and Ahmed31 The IPD for the time-to-event outcome is reconstructed from the published Kaplan–Meier survival curves using the R package IPDfromKM.Reference Liu, Zhou and Lee32 Figure 1 displays the reconstructed Kaplan–Meier survival curves. All the analyses are performed using R version 4.3.2.33
$\lambda$
is not a transitive measurement through an illustrative example involving bladder cancer. Open radical cystectomy (ORC) is widely regarded as the gold standard treatment for patients with muscle-invasive bladder cancer. Recent advancements in medical technology have introduced minimally invasive techniques, such as laparoscopic radical cystectomy (LRC) and robotic-assisted radical cystectomy (RARC).Reference Bochner, Dalbagni and Sjoberg30 In this example, we use data from a randomized three-arm trial comparing ORC, LRC, and RARC in terms of the overall survival outcomes.Reference Khan, Omar and Ahmed31 The IPD for the time-to-event outcome is reconstructed from the published Kaplan–Meier survival curves using the R package IPDfromKM.Reference Liu, Zhou and Lee32 Figure 1 displays the reconstructed Kaplan–Meier survival curves. All the analyses are performed using R version 4.3.2.33
Kaplan–Meier survival curves for the illustrative example of radical cystectomy. ORC, LRC, and RARC refer to the open radical cystectomy, laparoscopic radical cystectomy, and robotic-assisted radical cystectomy.

Denote RARC as drug A, ORC as drug B, and LRC as drug C. We calculate the pairwise HR for A versus B, A versus C, and B versus C using the coxph function in the R package survival.
Reference Therneau34 The estimated pairwise log HRs are
![]() ${\widehat{\mu}}_{AC}^D=0.701$
,
${\widehat{\mu}}_{AC}^D=0.701$
,
![]() ${\widehat{\mu}}_{BC}^D=0.247$
, and
${\widehat{\mu}}_{BC}^D=0.247$
, and
![]() ${\widehat{\mu}}_{AB}^D=0.394$
, which are consistent with the published results. It is evident that, in this three-arm trial, the direct comparison (log HR) of drug A versus B,
${\widehat{\mu}}_{AB}^D=0.394$
, which are consistent with the published results. It is evident that, in this three-arm trial, the direct comparison (log HR) of drug A versus B,
![]() ${\widehat{\mu}}_{AB}^D=0.394$
, does not equal the indirect comparison of A versus B through drug C, because
${\widehat{\mu}}_{AB}^D=0.394$
, does not equal the indirect comparison of A versus B through drug C, because
![]() ${\widehat{\mu}}_{AB}^I={\widehat{\mu}}_{AC}^D-{\widehat{\mu}}_{BC}^D=0.701-0.247=0.454$
. This discrepancy indicates that neither the log of the HR nor the HR itself is a transitive measurement for ITC. Based on this illustrative example, we have the following observations:
${\widehat{\mu}}_{AB}^I={\widehat{\mu}}_{AC}^D-{\widehat{\mu}}_{BC}^D=0.701-0.247=0.454$
. This discrepancy indicates that neither the log of the HR nor the HR itself is a transitive measurement for ITC. Based on this illustrative example, we have the following observations:
Property 1 HR from Cox PH model is not a statistically transitive measurement
HR does not satisfy the definition of a transitive measurement for ITC as outlined in Section 2 and, therefore, is not a transitive measurement.
Property 1 shows that in a randomized three-arm trial, the indirect HR does not always match the direct HR, indicating that the HR
![]() $\lambda$
under the Cox proportional hazard model is not inherently transitive.
$\lambda$
under the Cox proportional hazard model is not inherently transitive.
3.2 The expectation of a HR does not maintain transitivity
One might suspect that this discrepancy is simply due to sampling variability, implying that the two HRs could be identical with an infinitely large sample size. However, as we will show in the next theorem, even the expected values of the HRs are non-transitive if the proportional hazards assumption does not hold among drugs A, B, and C. Therefore, the indirect comparison of HR is not an unbiased estimator if the proportional hazards assumption is violated, which also depends on the common comparator C.
Theorem 2 Transitivity of HR under expectation
Let
![]() ${h}_A(t),{h}_B(t),$
and
${h}_A(t),{h}_B(t),$
and
![]() ${h}_C(t)$
be the hazard function for treatments A, B, and C, respectively, where we assume that they have the same follow-up time
${h}_C(t)$
be the hazard function for treatments A, B, and C, respectively, where we assume that they have the same follow-up time
![]() $t\in \left[0,T\right]$
. Denote the sample size for each treatment group as
$t\in \left[0,T\right]$
. Denote the sample size for each treatment group as
![]() ${n}_A,{n}_B,$
and
${n}_A,{n}_B,$
and
![]() ${n}_C$
. Additionally, denote the expectation of the estimated HR of treatment A versus treatment B under the Cox proportional hazards model as
${n}_C$
. Additionally, denote the expectation of the estimated HR of treatment A versus treatment B under the Cox proportional hazards model as
![]() ${\mathrm{HR}}_{AB}$
(with
${\mathrm{HR}}_{AB}$
(with
![]() ${\mathrm{HR}}_{AC}\;\mathrm{and}\;{\mathrm{HR}}_{BC}$
defined similarly). Then, the expectation of the HR is transitive in the following manner:
${\mathrm{HR}}_{AC}\;\mathrm{and}\;{\mathrm{HR}}_{BC}$
defined similarly). Then, the expectation of the HR is transitive in the following manner:
for any
![]() ${n}_A,{n}_B,$
and
${n}_A,{n}_B,$
and
![]() ${n}_C$
, if and only if the proportional hazard assumption is satisfied among the three arms, that is,
${n}_C$
, if and only if the proportional hazard assumption is satisfied among the three arms, that is,
for some scalars
![]() $\alpha, \beta >0$
.
$\alpha, \beta >0$
.
Proof: See the Appendix for the proof.
In other words, the expected value of HR is transitive only when the proportional hazards assumption holds for all the three included groups. The proof of Theorem 2 further clarifies why the direct and indirect HRs may differ. Because the HR represents an average of the pointwise HRs over time, the influence of drug C is not fully neutralized in the indirect comparison. Consequently, the direct and indirect comparisons yield different values. Essentially, the detailed information embedded in the baseline hazard functions of the A-C and B-C comparisons cannot be cancelled out, rendering the HR a non-transitive measurement in ITC.
3.3 A simulated paradoxical example for using the HR in ITC
In this subsection, we present a simulated data example to illustrate how a paradox can arise between the indirect and direct HRs when the proportional hazards assumption is violated. Specifically, in a three-arm trial, the direct HR comparing drug A with drug B favors drug B, while the indirect HR, derived through drug C as a common comparator, suggests an advantage for drug A.
To demonstrate this, consider three drugs A, B, and C with corresponding hazard functions
![]() ${h}_1(t),{h}_2(t),$
and
${h}_1(t),{h}_2(t),$
and
![]() ${h}_3(t)$
where
${h}_3(t)$
where
![]() $t\in \left[0,12\right]$
. For drug A and drug C, we assume proportional hazards with
$t\in \left[0,12\right]$
. For drug A and drug C, we assume proportional hazards with
![]() ${h}_1(t)=0.2$
and
${h}_1(t)=0.2$
and
![]() ${h}_3(t)=0.8$
for all
${h}_3(t)=0.8$
for all
![]() $t\in \left[0,12\right]$
. For drug B, we assume the hazard function changes with
$t\in \left[0,12\right]$
. For drug B, we assume the hazard function changes with
![]() ${h}_2(t)=0.1$
for
${h}_2(t)=0.1$
for
![]() $t\in \left[0,3\right]$
and
$t\in \left[0,3\right]$
and
![]() ${h}_2(t)=0.35$
for
${h}_2(t)=0.35$
for
![]() $t\in \left(3,12\right]$
. Using the hazard functions, we simulate
$t\in \left(3,12\right]$
. Using the hazard functions, we simulate
![]() $n=\mathrm{2,000}$
subjects for each group (we select a large sample size to ensure that all the estimated relative effects are not impacted by random variability) and estimate the HRs
$n=\mathrm{2,000}$
subjects for each group (we select a large sample size to ensure that all the estimated relative effects are not impacted by random variability) and estimate the HRs
![]() ${\widehat{\mathrm{HR}}}_{AB}$
,
${\widehat{\mathrm{HR}}}_{AB}$
,
![]() ${\widehat{\mathrm{HR}}}_{AC}$
, and
${\widehat{\mathrm{HR}}}_{AC}$
, and
![]() ${\widehat{\mathrm{HR}}}_{BC}$
. The estimated direct HRs are
${\widehat{\mathrm{HR}}}_{BC}$
. The estimated direct HRs are
![]() ${\mu}_{AB}^D={\widehat{\mathrm{HR}}}_{AB}=0.896$
with the corresponding 95% confidence interval
${\mu}_{AB}^D={\widehat{\mathrm{HR}}}_{AB}=0.896$
with the corresponding 95% confidence interval
![]() $\left(0.840,0.956\right),$
suggesting that drug A is statistically significant worse than drug B. However, for the indirect HR, we have
$\left(0.840,0.956\right),$
suggesting that drug A is statistically significant worse than drug B. However, for the indirect HR, we have
![]() ${\mu}_{AB}^I=\frac{{\widehat{\mathrm{HR}}}_{AC}}{{\widehat{\mathrm{HR}}}_{BC}}=0.238/0.184=1.296$
with a 95% confidence interval of
${\mu}_{AB}^I=\frac{{\widehat{\mathrm{HR}}}_{AC}}{{\widehat{\mathrm{HR}}}_{BC}}=0.238/0.184=1.296$
with a 95% confidence interval of
![]() $\left(1.117,1.440\right)$
, suggesting that drug A is significantly better than drug B. The corresponding R code for simulating the data is included in the Supplementary Material.
$\left(1.117,1.440\right)$
, suggesting that drug A is significantly better than drug B. The corresponding R code for simulating the data is included in the Supplementary Material.
This example highlights the issue of non-transitivity when using HRs in ITC. When direct and indirect comparisons can yield conflicting conclusions, it raises concerns about the reliability of ITC results. In this case, while the hazards for drugs A and C remain constant and follow a proportional pattern, the hazard for drug B changes over time. As demonstrated in Section 3.2, a key driver of this paradoxical phenomenon is the violation of the proportional hazards assumption. However, in real-world applications of ITC, individual patient data (IPD) for the BC trial are often unavailable, making it challenging to assess the validity of this assumption. As illustrated in this example, even if the proportional hazards assumption holds in the AC trial, it does not necessarily prevent the occurrence of such paradoxical results. This highlights the need for caution when interpreting ITC findings.
3.4 Different follow-up times in the ITC
In the previous subsections, we focused on the transitivity issue under the assumption of a common follow-up time for all three drugs in a hypothetical three-arm trial. We demonstrated through Property 1 and Theorem 2 that the HR is not a transitive measure and should be used with caution in ITC. However, there is another important issue related to the HRs that can affect the validity of ITC, which arises from differences in follow-up time between trials.
The HR depends on follow-up time: As demonstrated by previous studies (e.g., HernanReference Hernán35), the HR is highly sensitive to the duration of follow-up. The estimated HR is essentially a weighted average of the time-dependent HR over the entire follow-up period. This means that the value of HR can change depending on the length of the follow-up time considered. This matters for ITC because the AC and BC trials often have different follow-up periods. Consequently, the C arm in each trial is observed over different time windows, producing HR estimates that are not directly comparable, even if drug C is identical across trials and populations are well balanced.
The discrepancy in follow-up times further disrupts the transitivity of the HR in ITC. If follow-up time is different in the AC trial compared to the BC trial, then the HR derived from the AC trial (comparing drug A with drug C) and the HR from the BC trial (comparing drug B with drug C) may not be directly comparable. Even if drug C is the same in both trials, the HR estimates in both trials will be influenced by the respective follow-up times. Thus, if HRs are used in ITC (despite the concerns in previous sections), they should be estimated over a common follow-up period across the AC and BC trials. This can be done by digitizing the KM survival curve in the BC trial and re-estimating the HR during a comparable follow-up time period.
4 Alternative measurements for ITC with time-to-event outcomes
As demonstrated in Section 3, the HR is not a statistically transitive measurement for ITC. This limitation highlights the need for alternative approaches to compare treatments in a time-to-event context. In this section, we recommend three alternative measurements, RMST and landmark survival probability difference (or ratio), and the average hazard with survival weights difference (or ratio) which are fully transitive and easy to compute. We illustrate how these measurements can be used in the scenario of matching-adjusted treatment comparison (MAIC), though they are generally applicable for other ITC approaches.
4.1 Notation and setup for anchored MAIC
Let us consider an anchored MAIC, focusing on the comparison between drugs A and B with a common comparator drug C. We assume that researchers only have the individual-level data for AC trial:
![]() ${\left\{{\boldsymbol{X}}_i^t,{T}_i^t,{\delta}_i^t\right\}}_{i=1}^{n_t}$
, where
${\left\{{\boldsymbol{X}}_i^t,{T}_i^t,{\delta}_i^t\right\}}_{i=1}^{n_t}$
, where
![]() $t=A,C$
indicate the treatment (control) allocation; ,
$t=A,C$
indicate the treatment (control) allocation; ,
![]() $i=1,\dots, {n}_t$
the index of trial participants;
$i=1,\dots, {n}_t$
the index of trial participants;
![]() ${\boldsymbol{X}}_i^t$
is the vector of all effect modifiers that need to be adjusted;
${\boldsymbol{X}}_i^t$
is the vector of all effect modifiers that need to be adjusted;
![]() ${T}_i^t$
is the right-censored event time;
${T}_i^t$
is the right-censored event time;
![]() ${\delta}_i^t$
is the censoring indicator with
${\delta}_i^t$
is the censoring indicator with
![]() ${\delta}_i^t=0$
if
${\delta}_i^t=0$
if
![]() ${T}_i^t$
is censored and
${T}_i^t$
is censored and
![]() ${\delta}_i^t=1$
if
${\delta}_i^t=1$
if
![]() ${T}_i^t$
corresponds to an event. For the BC trial, researchers only have the published summary-level data
${T}_i^t$
corresponds to an event. For the BC trial, researchers only have the published summary-level data
![]() $\left\{{\widehat{\boldsymbol{X}}}^t,{\widehat{\mu}}^{BC(BC)}\right\}$
where
$\left\{{\widehat{\boldsymbol{X}}}^t,{\widehat{\mu}}^{BC(BC)}\right\}$
where
![]() ${\widehat{\boldsymbol{X}}}^t,t=B,C$
denote the sample mean of the covariates in group
${\widehat{\boldsymbol{X}}}^t,t=B,C$
denote the sample mean of the covariates in group
![]() $t=B,C$
and
$t=B,C$
and
![]() ${\widehat{\mu}}^{BC(BC)}$
be the estimated treatment effect of drug B versus drug C in the population of BC trial. For time-to-event outcomes, the summary-level data usually includes published Kaplan–Meier survival curves with sample size information (number of individuals at risk). If the BC trial does not report the RMST (or landmark survival probability) difference, but does report Kaplan–Meier survival curves, one can easily digitalize the KM curves and calculate the corresponding measurement based on the reconstructed IPD. For further details on how to digitize a Kaplan–Meier curve and calculate the corresponding measurements, one can refer to Liu et al.,Reference Liu, Zhou and Lee32 which provides a comprehensive guide, along with an R package for digitizing KM curves.
${\widehat{\mu}}^{BC(BC)}$
be the estimated treatment effect of drug B versus drug C in the population of BC trial. For time-to-event outcomes, the summary-level data usually includes published Kaplan–Meier survival curves with sample size information (number of individuals at risk). If the BC trial does not report the RMST (or landmark survival probability) difference, but does report Kaplan–Meier survival curves, one can easily digitalize the KM curves and calculate the corresponding measurement based on the reconstructed IPD. For further details on how to digitize a Kaplan–Meier curve and calculate the corresponding measurements, one can refer to Liu et al.,Reference Liu, Zhou and Lee32 which provides a comprehensive guide, along with an R package for digitizing KM curves.
MAIC aims to estimate a set of balancing weights
![]() $\boldsymbol{w}=\left\{{w}_1^A,\dots, {w}_{n_A}^A,{w}_1^C,\dots, {w}_{n_C}^C\right\}$
, with
$\boldsymbol{w}=\left\{{w}_1^A,\dots, {w}_{n_A}^A,{w}_1^C,\dots, {w}_{n_C}^C\right\}$
, with
![]() ${{\sum}_{\boldsymbol{i}}{w}_i^A=1}$
, and
${{\sum}_{\boldsymbol{i}}{w}_i^A=1}$
, and
![]() ${\sum}_{\boldsymbol{i}}{w}_i^C=1$
, for each participant in the AC trial such that the weighted population of treatment and control groups in the AC trial aligns with that in the BC trial in terms of the reported sample mean
${\sum}_{\boldsymbol{i}}{w}_i^C=1$
, for each participant in the AC trial such that the weighted population of treatment and control groups in the AC trial aligns with that in the BC trial in terms of the reported sample mean
![]() ${\widehat{\boldsymbol{X}}}^B$
and
${\widehat{\boldsymbol{X}}}^B$
and
![]() ${\widehat{\boldsymbol{X}}}^C$
, that is,
${\widehat{\boldsymbol{X}}}^C$
, that is,
![]() ${\sum}_{i=1}^{n_A}{w}_i^A{\boldsymbol{X}}_i^A={\widehat{\boldsymbol{X}}}^B$
and
${\sum}_{i=1}^{n_A}{w}_i^A{\boldsymbol{X}}_i^A={\widehat{\boldsymbol{X}}}^B$
and
![]() ${\sum}_{i=1}^{n_C}{w}_i^C{\boldsymbol{X}}_i^C={\widehat{\boldsymbol{X}}}^C$
. There is also an alternative matching strategy which, instead of matching the treated groups and control group separately, matches the entire AC trial population with the entire BC trial population. The weights can be estimated through various approaches, including the original MAIC with the method of moments, the MAIC with the largest effective sample size,Reference Jackson, Rhodes and Ouwens28 and two-stage MAIC method,Reference Remiro-Azócar27 see Jiang et al.Reference Jiang, Cappelleri, Gamalo, Chen, Thomas and Chu29 for a comprehensive review of the approaches. Under the assumption that all effect modifiers are included and that the correlations between covariates are similar between the AC trial and BC trial, the weighted IPD can be used to generate the comparative effectiveness of drug A versus drug C in the population of the BC trial.
${\sum}_{i=1}^{n_C}{w}_i^C{\boldsymbol{X}}_i^C={\widehat{\boldsymbol{X}}}^C$
. There is also an alternative matching strategy which, instead of matching the treated groups and control group separately, matches the entire AC trial population with the entire BC trial population. The weights can be estimated through various approaches, including the original MAIC with the method of moments, the MAIC with the largest effective sample size,Reference Jackson, Rhodes and Ouwens28 and two-stage MAIC method,Reference Remiro-Azócar27 see Jiang et al.Reference Jiang, Cappelleri, Gamalo, Chen, Thomas and Chu29 for a comprehensive review of the approaches. Under the assumption that all effect modifiers are included and that the correlations between covariates are similar between the AC trial and BC trial, the weighted IPD can be used to generate the comparative effectiveness of drug A versus drug C in the population of the BC trial.
4.2 Matching-adjusted indirect comparison with restricted mean survival time difference
The RMSTReference Royston and Parmar36 is a model-free measurement that reflects the average survival time up to a prespecified fixed follow-up time. Compared to the HR, RMST is more flexible as it does not require the proportional hazards assumption. RMST is also easier to interpret clinically, since it directly relates to the average survival time within a fixed time interval.Reference Royston and Parmar36–
Reference Jiang, Lu and Liu38 As RMST represents the area under the survival curve up to a specified time, the RMST difference of drug B versus drug C (and its corresponding standard error) can be easily calculated using the reconstructed IPD from the reported Kaplan–Meier survival curve. For estimating the RMST difference of drug A versus drug C using the weighted IPD with weights
![]() $\boldsymbol{w}=\left\{{w}_1^A,\dots, {w}_{n_A}^A,{w}_1^C,\dots, {w}_{n_C}^C\right\}$
estimated from one of the MAIC methods, the RMST can be calculated as the area under the estimated weighted Kaplan–Meier survival curve.Reference Winnett and Sasieni39–
Reference Conner, Sullivan, Benjamin, LaValley, Galea and Trinquart41
$\boldsymbol{w}=\left\{{w}_1^A,\dots, {w}_{n_A}^A,{w}_1^C,\dots, {w}_{n_C}^C\right\}$
estimated from one of the MAIC methods, the RMST can be calculated as the area under the estimated weighted Kaplan–Meier survival curve.Reference Winnett and Sasieni39–
Reference Conner, Sullivan, Benjamin, LaValley, Galea and Trinquart41
Suppose the event in the drug A’s arm occurs at
![]() $D$
distinct times
$D$
distinct times
![]() ${t}_1^A<{t}_2^A<\dots <{t}_D^A$
, then the weighted Kaplan–Meier estimator of the survival function can be expressed as
${t}_1^A<{t}_2^A<\dots <{t}_D^A$
, then the weighted Kaplan–Meier estimator of the survival function can be expressed as
 $$\begin{align*}{\widehat{S}}^A(t)=\left\{\begin{array}{ll}1,& \mathrm{if}\;t<{t}_1^A,\\ {}{\prod}_{t_j^A\le t}\left(1-\frac{{\overset{\sim }{\theta}}_j^A}{\theta_j^A}\right),& \mathrm{otherwise},\end{array}\right.\end{align*}$$
$$\begin{align*}{\widehat{S}}^A(t)=\left\{\begin{array}{ll}1,& \mathrm{if}\;t<{t}_1^A,\\ {}{\prod}_{t_j^A\le t}\left(1-\frac{{\overset{\sim }{\theta}}_j^A}{\theta_j^A}\right),& \mathrm{otherwise},\end{array}\right.\end{align*}$$
where
![]() ${\overset{\sim }{\theta}}_j^A=\sum \nolimits_{i:{T}_i^A={t}_j^A}{w}_i^A{\delta}_i^{AC}$
and
${\overset{\sim }{\theta}}_j^A=\sum \nolimits_{i:{T}_i^A={t}_j^A}{w}_i^A{\delta}_i^{AC}$
and
![]() ${\theta}_j^A=\sum \nolimits_{i:{T}_i^A>{t}_j^A}{w}_i^A$
be the weighted number of events and the weighted number of individuals at risk for drug A at time
${\theta}_j^A=\sum \nolimits_{i:{T}_i^A>{t}_j^A}{w}_i^A$
be the weighted number of events and the weighted number of individuals at risk for drug A at time
![]() ${t}_j^A$
. Then, the weighted RMST of drug A with threshold time
${t}_j^A$
. Then, the weighted RMST of drug A with threshold time
![]() $\tau$
can be calculated as the area under
$\tau$
can be calculated as the area under
![]() ${\widehat{S}}^A(t)$
with
${\widehat{S}}^A(t)$
with
![]() $t\le \tau$
, that is,
$t\le \tau$
, that is,
![]() ${\widehat{\mu}}^A\left(\tau \right)={\int}_{t=0}^{\tau }{\widehat{S}}^A(t) dt$
. The corresponding variance can be estimated either through nonparametric bootstrap method or using the formula provided in Conner et al.,Reference Conner, Sullivan, Benjamin, LaValley, Galea and Trinquart41 namely,
${\widehat{\mu}}^A\left(\tau \right)={\int}_{t=0}^{\tau }{\widehat{S}}^A(t) dt$
. The corresponding variance can be estimated either through nonparametric bootstrap method or using the formula provided in Conner et al.,Reference Conner, Sullivan, Benjamin, LaValley, Galea and Trinquart41 namely,
 $$\begin{align*}\widehat{V}\left(\widehat{\mu}\right)=\sum \limits_{j:{t}_j^A\le \tau }{\left[\sum \limits_{i=j}^{\tau }{\widehat{S}}^A\left({t}_i\right)\left({t}_{i+1}-{t}_i\right)\right]}^2\frac{{\overset{\sim }{\theta}}_j^A}{M_j^A\left({\theta}_j^A-{\overset{\sim }{\theta}}_j^A\right)},\end{align*}$$
$$\begin{align*}\widehat{V}\left(\widehat{\mu}\right)=\sum \limits_{j:{t}_j^A\le \tau }{\left[\sum \limits_{i=j}^{\tau }{\widehat{S}}^A\left({t}_i\right)\left({t}_{i+1}-{t}_i\right)\right]}^2\frac{{\overset{\sim }{\theta}}_j^A}{M_j^A\left({\theta}_j^A-{\overset{\sim }{\theta}}_j^A\right)},\end{align*}$$
where
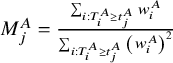 ${M}_j^A=\frac{\sum_{i:{T}_i^A\ge {t}_j^A}{w}_i^A}{\sum_{i:{T}_i^A\ge {t}_j^A}{\left({w}_i^A\right)}^2}$
. Similarly, the weighted RMST of drug C
${M}_j^A=\frac{\sum_{i:{T}_i^A\ge {t}_j^A}{w}_i^A}{\sum_{i:{T}_i^A\ge {t}_j^A}{\left({w}_i^A\right)}^2}$
. Similarly, the weighted RMST of drug C
![]() ${\widehat{\mu}}^C\left(\tau \right)$
can be estimated using
${\widehat{\mu}}^C\left(\tau \right)$
can be estimated using
![]() ${\widehat{S}}^C(t)$
and the RMST difference of drug A versus drug C in the population of the BC trial is defined as
${\widehat{S}}^C(t)$
and the RMST difference of drug A versus drug C in the population of the BC trial is defined as
and the indirect comparison of the RMST of drug A versus drug B in the population of the BC trial can be derived as the difference between
![]() ${\widehat{\mu}}^{AC(BC)}\left(\tau \right)$
and
${\widehat{\mu}}^{AC(BC)}\left(\tau \right)$
and
![]() ${\widehat{\mu}}^{BC(BC)}\left(\tau \right)$
.
${\widehat{\mu}}^{BC(BC)}\left(\tau \right)$
.
4.3 MAIC with landmark survival probability difference
Landmark survival probability refers to the survival probabilities
![]() $S(t)$
for groups of patients at specific time points, or landmarks
$S(t)$
for groups of patients at specific time points, or landmarks
![]() $t$
, during a follow-up period. For each prespecified time point, the landmark survival probability serves as a transitive measurement of the survival outcome. Based on the estimated survival function
$t$
, during a follow-up period. For each prespecified time point, the landmark survival probability serves as a transitive measurement of the survival outcome. Based on the estimated survival function
![]() $\widehat{S}(t)$
introduced in the previous section, the landmark survival probability can be calculated for any treatment arm with any time point
$\widehat{S}(t)$
introduced in the previous section, the landmark survival probability can be calculated for any treatment arm with any time point
![]() $t<{t}_{\mathrm{max}}$
, where
$t<{t}_{\mathrm{max}}$
, where
![]() ${t}_{\mathrm{max}}$
be the maximum time point with the observed event. The corresponding variance of
${t}_{\mathrm{max}}$
be the maximum time point with the observed event. The corresponding variance of
![]() $\widehat{S}(t)$
can be estimated using the variance formula in Xie and Liu.Reference Xie and Liu40
$\widehat{S}(t)$
can be estimated using the variance formula in Xie and Liu.Reference Xie and Liu40
4.4 Matching-adjusted indirect comparison with the average hazard with survival weights difference (or ratio)
Uno and HoriguchiReference Uno and Horiguchi42 recently proposed an alternative measurement that summarizes the group-specific hazard information, which they termed as the AH-SW. The AH-SW ratio for a given threshold time
![]() $\tau$
is defined as
$\tau$
is defined as
where
![]() $k\in \left\{A,B,C\right\}$
,
$k\in \left\{A,B,C\right\}$
,
![]() ${F}_k\left(\tau \right)$
is the cumulative density function at time
${F}_k\left(\tau \right)$
is the cumulative density function at time
![]() $\tau$
, and
$\tau$
, and
![]() ${R}_k\left(\tau \right)$
is the RMST with threshold time
${R}_k\left(\tau \right)$
is the RMST with threshold time
![]() $\tau$
. AH-SW represents a person-time incidence rate that is independent of random censoring. Uno and HoriguchiReference Uno and Horiguchi42 proposed to estimate
$\tau$
. AH-SW represents a person-time incidence rate that is independent of random censoring. Uno and HoriguchiReference Uno and Horiguchi42 proposed to estimate
![]() ${\eta}_k\left(\tau \right)$
by
${\eta}_k\left(\tau \right)$
by
![]() ${\widehat{\eta}}_k\left(\tau \right)=\frac{{\widehat{F}}_k\left(\tau \right)}{{\widehat{R}}_k\left(\tau \right)}$
where
${\widehat{\eta}}_k\left(\tau \right)=\frac{{\widehat{F}}_k\left(\tau \right)}{{\widehat{R}}_k\left(\tau \right)}$
where
![]() ${\widehat{F}}_k\left(\tau \right)$
and
${\widehat{F}}_k\left(\tau \right)$
and
![]() ${\widehat{R}}_k\left(\tau \right)$
are estimated nonparametrically through the Kaplan–Meier estimator. By estimating
${\widehat{R}}_k\left(\tau \right)$
are estimated nonparametrically through the Kaplan–Meier estimator. By estimating
![]() ${\eta}_k\left(\tau \right)$
through the nonparametric Kaplan–Meier estimator,
${\eta}_k\left(\tau \right)$
through the nonparametric Kaplan–Meier estimator,
![]() ${\widehat{\eta}}_k\left(\tau \right)$
is clearly a transitive measurement. Since we introduced the variance estimation for
${\widehat{\eta}}_k\left(\tau \right)$
is clearly a transitive measurement. Since we introduced the variance estimation for
![]() ${\widehat{F}}_k\left(\tau \right)$
and
${\widehat{F}}_k\left(\tau \right)$
and
![]() ${\widehat{R}}_k\left(\tau \right)$
under MAIC weights, the variance of
${\widehat{R}}_k\left(\tau \right)$
under MAIC weights, the variance of
![]() ${\widehat{\eta}}_k\left(\tau \right)$
can either be calculated analytically or through bootstrap.
${\widehat{\eta}}_k\left(\tau \right)$
can either be calculated analytically or through bootstrap.
4.5 Revisit the ORC example
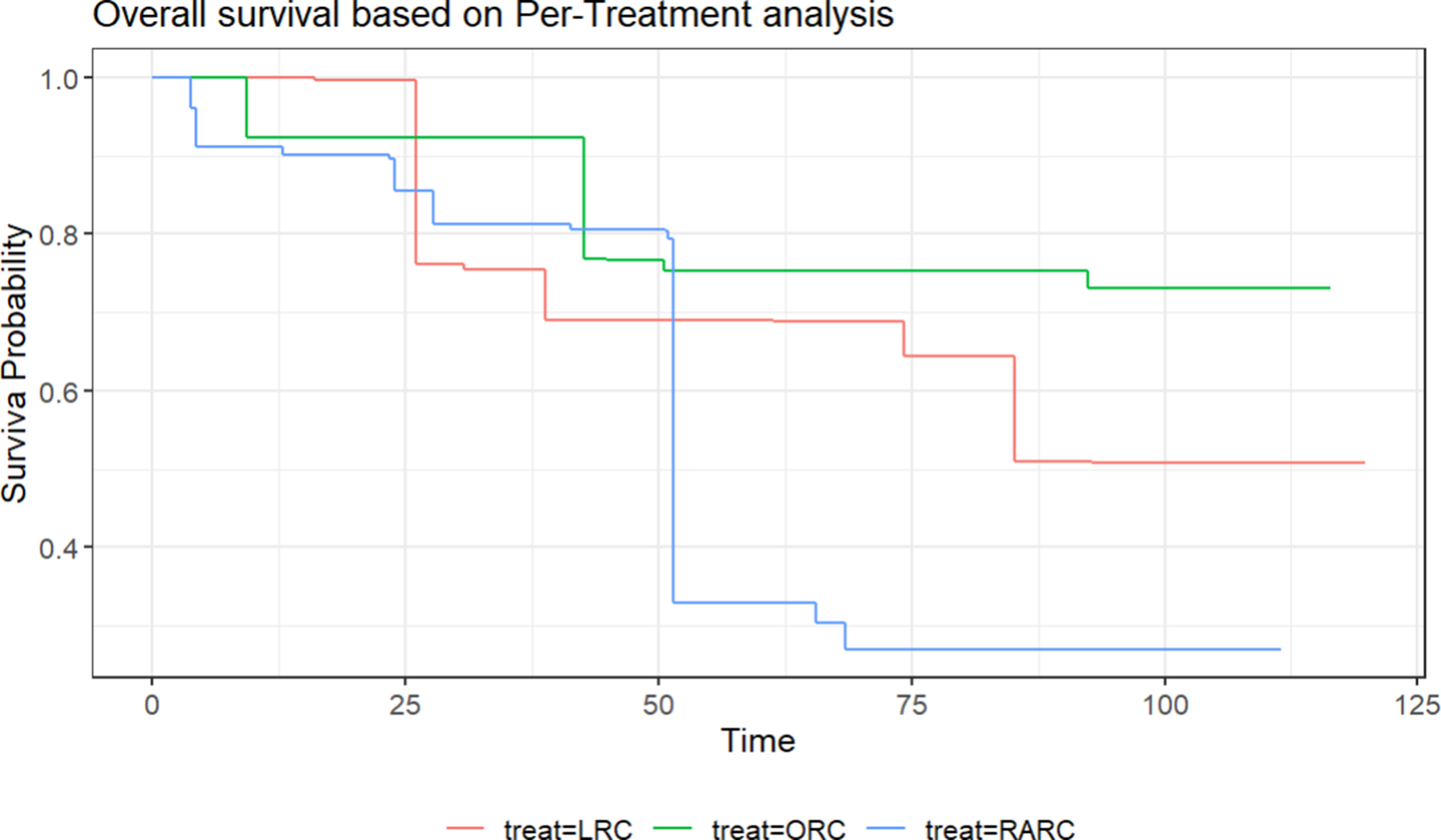
Here, we revisit the illustrative ORC example and illustrate how to compute the weighted RMST/landmark survival probability and the corresponding variance. As the data are from a three-arm randomized trial, we randomly simulate the weights for each subject following a log-normal distribution. Then, we estimate the weighted Kaplan–Meier survival curve using the simulated weights. Figure 2 displays the estimated KM curve. The weighted RMST and its standard error are estimated using the “akm.rmst” function provided by Conner et al.Reference Conner, Sullivan, Benjamin, LaValley, Galea and Trinquart41 The estimated RMST after adjustment with
![]() $\tau =70$
are 57.17 for LRC group (with standard error 8.50), 60.75 for the ORC group (with standard error 8.47), and 50.44 for the RARC group (with standard error 9.99). The landmark survival probability at
$\tau =70$
are 57.17 for LRC group (with standard error 8.50), 60.75 for the ORC group (with standard error 8.47), and 50.44 for the RARC group (with standard error 9.99). The landmark survival probability at
![]() $t=70$
is 0.689 for LRC (with standard error 0.203), 0.752 for ORC (with standard error 0.169), and 0.269 for the RARC group (with standard error 0.166). It is clear to see that both RMST and landmark survival probability are transitive, since
$t=70$
is 0.689 for LRC (with standard error 0.203), 0.752 for ORC (with standard error 0.169), and 0.269 for the RARC group (with standard error 0.166). It is clear to see that both RMST and landmark survival probability are transitive, since
where
![]() $\mu$
is either the RMST or the landmark survival probability.
$\mu$
is either the RMST or the landmark survival probability.
Weighted Kaplan–Meier survival curve with randomly generated weights.

It should be noted that RMST and landmark survival probability are transitive only if they are estimated nonparametrically (i.e., without using the Cox PH model) which is the common approach for the estimation. In the Supplementary Code (see Supplementary Material), we illustrate that the RMST can be non-transitive if it is estimated by the Cox proportional hazard model.
5 Discussion
In this article, we highlight a critical issue with the use of HR in ITCs: the HR is not a statistically transitive measurement. This discrepancy primarily arises due to the differences in how the baseline hazard functions are estimated in the direct and indirect comparisons. Specifically, the Cox proportional hazards model, which is commonly used to estimate HRs, treats the baseline hazard function as a nuisance parameter. Because of this, the baseline hazard functions are implicitly different in each pairwise comparison (A vs. B, A vs. C, and B vs. C). As a result, the indirect comparison of drugs A and B can be affected by the choice of the common comparator C. Theorem 2 demonstrated that if the proportional hazards assumption holds for all three drugs (A, B, and C) in the three-arm trial, the expected value of the estimated HR would be transitive. This means that the HRs calculated through direct and indirect comparisons would align. However, in practice, ensuring that the proportional hazards assumption holds across both the AC and BC trials is difficult. This assumption cannot be assessed using aggregate data alone, especially when individual patient data (IPD) is not available for the BC trial.
Even if both the AC and BC trials have tested the assumption (i.e., failed to reject the PH assumption), the HR should still be used with caution because the power of the PH test is likely very low.Reference Austin43 Indeed, Stensrud and HernánReference Stensrud and Hernán44 argued that the proportional hazard assumption seldom holds in medical studies. In addition to its non-transitivity, the HR also suffers from non-collapsibility; its value can change when conditioning on additional covariates, even in the absence of confounding,Reference Daniel, Zhang and Farewell45 and lack causal interpretability, as it represents the average of instantaneous relative risk among individuals who remain at risk, a quantity influenced by the evolving composition of the risk set over time. Given these limitations, we recommend that the HR be used with caution in ITC.
While RMST and landmark survival probability difference (or ratio) offer alternatives to the HR, they are not without limitations. As discussed in the literature, RMST can only summarize survival information up to a specific time point, typically the smallest follow-up time across the groups. This limitation means that when comparing drug A and drug B using RMST in a scenario where the AC trial follows up for 4 years and the BC trial follows up for 3 years, only data for the first 3 years can be included in the comparison. This results in the exclusion of potentially valuable information from the AC trial, especially if the treatment effects extend beyond the shorter follow-up period.
Another limitation of RMST is that it is a nonparametric measure, and thus, it cannot inform about survival beyond the observed study period. This makes RMST unsuitable for long-term analyses, especially when policymakers or healthcare require evidence for extended timeframes. For this reason, we suggest that when performing ITC, multiple measures—RMST, landmark survival probability, and HR—should be considered together. These measures are relatively easy to compute when individual patient data can be reconstructed from Kaplan–Meier curves.
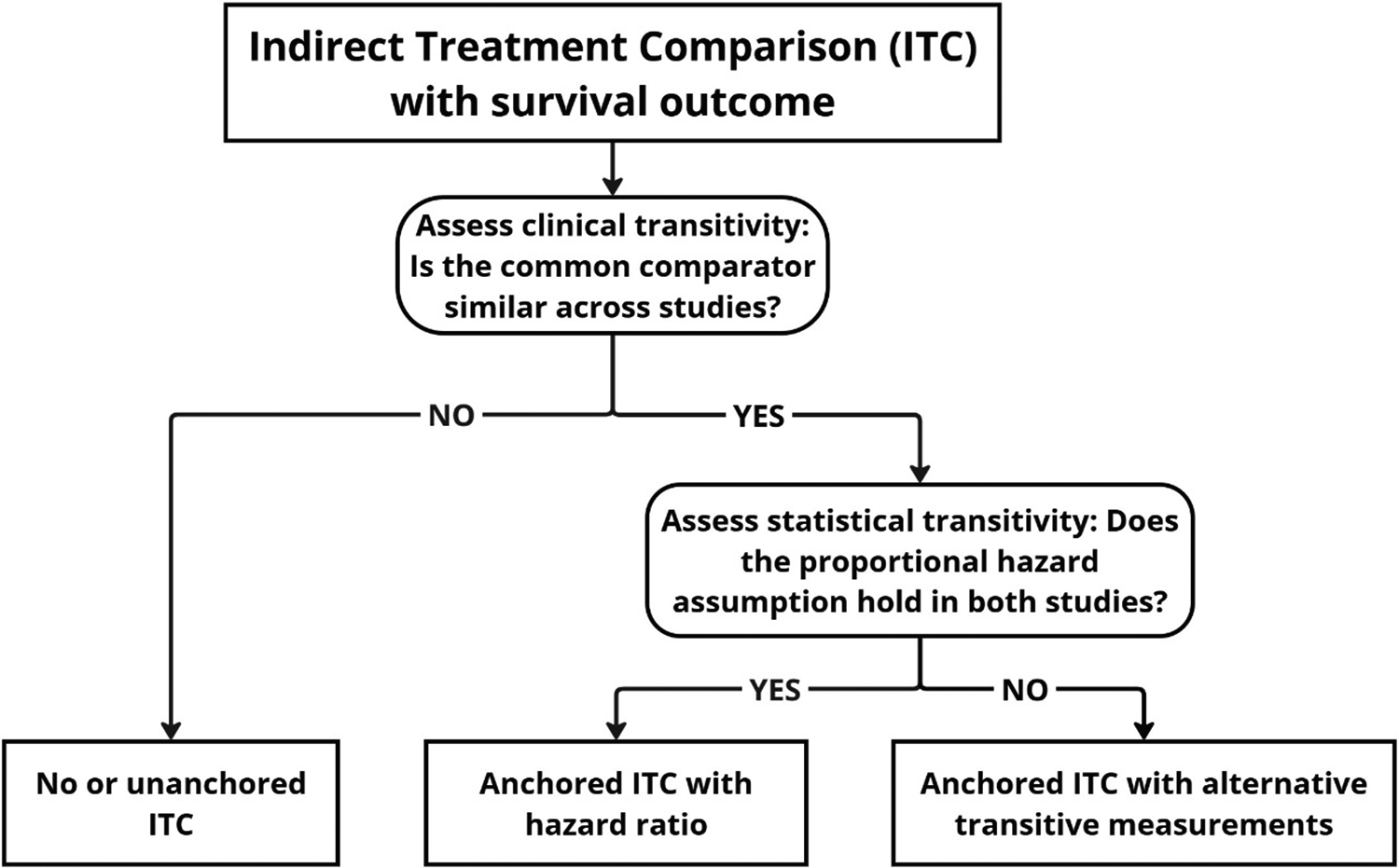
Beyond this manuscript’s focus on statistical transitivity, clinical transitivity is also frequently overlooked in ITC practice. Truong et al.Reference Truong, Tran, Le, Pham and Vo46 found that only 4.9% of population-adjusted ITCs explicitly evaluated whether the common comparator was consistent across trials. Accordingly, we propose a decision-making flowchart (Figure 3) for ITC with survival outcomes: First, assess clinical transitivity by verifying the comparability of the common comparator across studies; if satisfied, assess statistical transitivity before using the HR. When the proportional hazards assumption fails in either trial, alternative metrics with better transitivity properties—such as RMST differences (or ratios), landmark survival probability differences (or ratios), or other measures (e.g., AH-SW difference or ratio) — should be considered. Given the limitations of any single metric, we recommend sensitivity analyses using alternative measures regardless of the primary choice, because they can provide complementary insights.
Decision-making flowchart for performing indirect treatment comparison with survival outcomes.

This article primarily focuses on anchored indirect comparison where there exists a common comparator group in two separate trials. For unanchored indirect comparisons, survival outcomes can be directly compared between drugs A and B after adjusting for covariates. In this case, HRs can be calculated by weighting the IPD from trial A and the reconstructed IPD from trial B. However, as pointed out by Phillippo et al.,Reference Phillippo, Ades, Dias, Palmer, Abrams and Welton1 conducting an unanchored indirect comparison requires including all prognostic variables and effect modifiers that influence the outcome, which can be very challenging in practice. Therefore, we also strongly recommend performing an anchored indirect comparison whenever possible, as it offers a more robust and reliable approach.
Author contributions
Conceptualization: Z.J., H.C.; Investigation: Z.J., J.L., W.H., J.C., S.R., Y.C., H.C.; Methodology: Z.J., J.L., H.C.; Resources: W.H., J.C., S.R., Y.C., H.C.; Supervision: W.H., J.C., S.R., Y.C., H.C.; Validation: J.L., W.H., J.C., S.R., Y.C., H.C.; Visualization: Z.J., J.L.; Writing—original draft: Z.J., J.L.; Writing—review and editing: Z.J., J.L., W.H., J.C., S.R., Y.C., H.C.
Competing interest statement
W.H. is employed by Abbvie; J.C., S.R., and H.C. are employed by Pfizer. They may own stocks of their company. However, all of the contents in this manuscript are strictly educational, instructive, and methodological, not involving any real medicinal intervention. The remaining authors declare no competing interests.
Data availability statement
The data used in the illustrative example were digitalized from the cited paper using the R IPDfromKMReference Liu, Zhou and Lee32 package.
Funding statement
The authors declare that no specific funding has been received for this article.
Supplementary material
To view supplementary material for this article, please visit http://doi.org/10.1017/rsm.2025.10059.
Appendix
Theorem 2 Transitivity of hazard ratio under expectation
Let
![]() ${h}_A(t),{h}_B(t),$
and
${h}_A(t),{h}_B(t),$
and
![]() ${h}_C(t)$
be the hazard function for drugs A, B, and C, respectively, where
${h}_C(t)$
be the hazard function for drugs A, B, and C, respectively, where
![]() $t\in \left[0,T\right]$
denotes time. Let the sample size for treatment groups be denoted as
$t\in \left[0,T\right]$
denotes time. Let the sample size for treatment groups be denoted as
![]() ${n}_A,{n}_B,$
and
${n}_A,{n}_B,$
and
![]() ${n}_C$
. The expectation of the estimated hazard ratio (HR) of drug A versus drug B under the Cox model is denoted as
${n}_C$
. The expectation of the estimated hazard ratio (HR) of drug A versus drug B under the Cox model is denoted as
![]() ${\mathrm{HR}}_{AB}$
, with
${\mathrm{HR}}_{AB}$
, with
![]() ${\mathrm{HR}}_{AC}\;\mathrm{and}\;{\mathrm{HR}}_{BC}$
defined similarly. Then, the expectation of the HR follows a transitive property such that
${\mathrm{HR}}_{AC}\;\mathrm{and}\;{\mathrm{HR}}_{BC}$
defined similarly. Then, the expectation of the HR follows a transitive property such that
for any values of
![]() ${n}_A,{n}_B,$
and
${n}_A,{n}_B,$
and
![]() ${n}_C$
, if and only if the hazard functions for three groups are proportional. Specifically, this means that
${n}_C$
, if and only if the hazard functions for three groups are proportional. Specifically, this means that
![]() ${h}_A(t)=\alpha {h}_B(t)=\beta {h}_C(t)$
where
${h}_A(t)=\alpha {h}_B(t)=\beta {h}_C(t)$
where
![]() $\alpha$
and
$\alpha$
and
![]() $\beta >0$
are constants.
$\beta >0$
are constants.
Proof: According to Schemper et al.,Reference Schemper, Wakounig and Heinze47 under the nonproportional hazards, the Cox model estimates the average HR across all event times, with an expectation given by
 $$\begin{align*}{\mathrm{HR}}_{AB}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt},\end{align*}$$
$$\begin{align*}{\mathrm{HR}}_{AB}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt},\end{align*}$$
where
![]() ${h}_A(t)$
and
${h}_A(t)$
and
![]() ${h}_B(t)$
are the hazard functions for drugs A and B, and
${h}_B(t)$
are the hazard functions for drugs A and B, and
![]() ${f}_{AB}(t)$
is the event density for the combined population of drugs A and B. Similarly, the corresponding relationships for other HRs can be expressed in a similar form.
${f}_{AB}(t)$
is the event density for the combined population of drugs A and B. Similarly, the corresponding relationships for other HRs can be expressed in a similar form.
 $$\begin{align*}{\mathrm{HR}}_{AC}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_C(t)}{f}_{AC}(t) dt}{\int_0^T\frac{h_C(t)}{h_A(t)+{h}_C(t)}{f}_{AC}(t) dt},\end{align*}$$
$$\begin{align*}{\mathrm{HR}}_{AC}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_C(t)}{f}_{AC}(t) dt}{\int_0^T\frac{h_C(t)}{h_A(t)+{h}_C(t)}{f}_{AC}(t) dt},\end{align*}$$
and
 $$\begin{align*}{\mathrm{HR}}_{BC}=\frac{\int_0^T\frac{h_B(t)}{h_B(t)+{h}_C(t)}{f}_{BC}(t) dt}{\int_0^T\frac{h_C(t)}{h_B(t)+{h}_C(t)}{f}_{BC}(t) dt}.\end{align*}$$
$$\begin{align*}{\mathrm{HR}}_{BC}=\frac{\int_0^T\frac{h_B(t)}{h_B(t)+{h}_C(t)}{f}_{BC}(t) dt}{\int_0^T\frac{h_C(t)}{h_B(t)+{h}_C(t)}{f}_{BC}(t) dt}.\end{align*}$$
It is important to note that the event density for the combined population of drugs A and B,
![]() ${f}_{AB}(t)$
, depends on the relative sample sizes of treatment groups A and B. If
${f}_{AB}(t)$
, depends on the relative sample sizes of treatment groups A and B. If
![]() ${n}_A$
is changed while
${n}_A$
is changed while
![]() ${n}_B$
remains fixed, the density
${n}_B$
remains fixed, the density
![]() ${f}_{AB}(t)$
will be different unless the hazard functions for both groups are equal
${f}_{AB}(t)$
will be different unless the hazard functions for both groups are equal
![]() ${h}_A(t)={h}_B(t)$
.
${h}_A(t)={h}_B(t)$
.
Now, assuming the proportional hazard assumption holds for the three treatment groups,
![]() ${h}_A(t)=\alpha {h}_B(t)=\beta {h}_C(t)$
for some constants
${h}_A(t)=\alpha {h}_B(t)=\beta {h}_C(t)$
for some constants
![]() $\alpha, \beta >0$
, we can derive the following:
$\alpha, \beta >0$
, we can derive the following:
 $$\begin{align*}{\mathrm{HR}}_{AB}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}=\frac{\int_0^T\frac{{\alpha h}_B(t)}{{\alpha h}_B(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{{\alpha h}_B(t)+{h}_B(t)}{f}_{AB}(t) dt}=\alpha .\end{align*}$$
$$\begin{align*}{\mathrm{HR}}_{AB}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}=\frac{\int_0^T\frac{{\alpha h}_B(t)}{{\alpha h}_B(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{{\alpha h}_B(t)+{h}_B(t)}{f}_{AB}(t) dt}=\alpha .\end{align*}$$
Similarly,
![]() ${\mathrm{HR}}_{AC}=\beta$
and
${\mathrm{HR}}_{AC}=\beta$
and
![]() ${\mathrm{HR}}_{BC}=\beta /\alpha$
. Thus, we have the relation
${\mathrm{HR}}_{BC}=\beta /\alpha$
. Thus, we have the relation
![]() ${\mathrm{HR}}_{AB}={\mathrm{HR}}_{AC}/{\mathrm{HR}}_{BC}$
, which is independent of the sample sizes
${\mathrm{HR}}_{AB}={\mathrm{HR}}_{AC}/{\mathrm{HR}}_{BC}$
, which is independent of the sample sizes
![]() ${n}_A,{n}_B,$
and
${n}_A,{n}_B,$
and
![]() ${n}_C$
.
${n}_C$
.
To demonstrate the opposite direction of the assertion, assume
![]() ${h}_A(t)$
and
${h}_A(t)$
and
![]() ${h}_B(t)$
are not proportional, meaning that
${h}_B(t)$
are not proportional, meaning that
![]() ${h}_A(t)/{h}_B(t)$
varies with time
${h}_A(t)/{h}_B(t)$
varies with time
![]() $t$
. We will show that the relationship
$t$
. We will show that the relationship
![]() ${\mathrm{HR}}_{AB}={\mathrm{HR}}_{AC}/{\mathrm{HR}}_{BC}$
cannot hold for all values of
${\mathrm{HR}}_{AB}={\mathrm{HR}}_{AC}/{\mathrm{HR}}_{BC}$
cannot hold for all values of
![]() ${n}_A,{n}_B,$
and
${n}_A,{n}_B,$
and
![]() ${n}_C$
.
${n}_C$
.
Assume that
![]() ${\mathrm{HR}}_{AB}={\mathrm{HR}}_{AC}/{\mathrm{HR}}_{BC}$
holds for the sample sizes with the ratio
${\mathrm{HR}}_{AB}={\mathrm{HR}}_{AC}/{\mathrm{HR}}_{BC}$
holds for the sample sizes with the ratio
![]() ${n}_A:{n}_B:{n}_C=a:b:c$
with the corresponding densities
${n}_A:{n}_B:{n}_C=a:b:c$
with the corresponding densities
![]() ${f}_{AB},{f}_{AC},$
and
${f}_{AB},{f}_{AC},$
and
![]() ${f}_{BC}$
. Now, consider the scenario where the sample size ratio is changed to
${f}_{BC}$
. Now, consider the scenario where the sample size ratio is changed to
![]() ${n}_A:{n}_B:{n}_C=a^{\prime }:b:c$
, resulting in new density functions
${n}_A:{n}_B:{n}_C=a^{\prime }:b:c$
, resulting in new density functions
![]() ${f}_{AB}^{\prime}\;\mathrm{and}\;{f}_{AC}^{\prime }$
. Since the
${f}_{AB}^{\prime}\;\mathrm{and}\;{f}_{AC}^{\prime }$
. Since the
![]() ${h}_A(t)$
and
${h}_A(t)$
and
![]() ${h}_B(t)$
are not proportional, the term
${h}_B(t)$
are not proportional, the term
![]() $\frac{h_A(t)}{h_A(t)+{h}_B(t)}$
will change over time
$\frac{h_A(t)}{h_A(t)+{h}_B(t)}$
will change over time
![]() $t$
. Therefore, for some value of
$t$
. Therefore, for some value of
![]() ${n}_a^{\prime }$
, the following relationship will hold:
${n}_a^{\prime }$
, the following relationship will hold:
 $$\begin{align*}{\mathrm{HR}}_{AB}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}\ne \frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}^{\prime }(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}^{\prime }(t) dt}={\mathrm{HR}}_{AB}^{\prime }.\end{align*}$$
$$\begin{align*}{\mathrm{HR}}_{AB}=\frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}(t) dt}\ne \frac{\int_0^T\frac{h_A(t)}{h_A(t)+{h}_B(t)}{f}_{AB}^{\prime }(t) dt}{\int_0^T\frac{h_B(t)}{h_A(t)+{h}_B(t)}{f}_{AB}^{\prime }(t) dt}={\mathrm{HR}}_{AB}^{\prime }.\end{align*}$$
Now consider the new hazard ratio
![]() ${\mathrm{HR}}_{AC}^{\prime }$
with the updated density function
${\mathrm{HR}}_{AC}^{\prime }$
with the updated density function
![]() ${f}_{AC}^{\prime }$
. If
${f}_{AC}^{\prime }$
. If
![]() ${h}_A(t)$
and
${h}_A(t)$
and
![]() ${h}_C(t)$
are proportional,
${h}_C(t)$
are proportional,
![]() ${\mathrm{HR}}_{AC}^{\prime }$
will be the same as
${\mathrm{HR}}_{AC}^{\prime }$
will be the same as
![]() ${\mathrm{HR}}_{AC}$
as it is unaffected by the sample sizes. In this case, we have
${\mathrm{HR}}_{AC}$
as it is unaffected by the sample sizes. In this case, we have
This shows that the HR is not transitive.
If
![]() ${h}_A(t)$
and
${h}_A(t)$
and
![]() ${h}_C(t)$
are not proportional,
${h}_C(t)$
are not proportional,
![]() ${\mathrm{HR}}_{AC}^{\prime }$
can differ from
${\mathrm{HR}}_{AC}^{\prime }$
can differ from
![]() ${\mathrm{HR}}_{AC}$
. For the HR to be transitive, we would need
${\mathrm{HR}}_{AC}$
. For the HR to be transitive, we would need
![]() ${\mathrm{HR}}_{AC}^{\prime }/{\mathrm{HR}}_{BC}={\mathrm{HR}}_{AB}^{\prime }$
. However, this cannot hold for all ratios of
${\mathrm{HR}}_{AC}^{\prime }/{\mathrm{HR}}_{BC}={\mathrm{HR}}_{AB}^{\prime }$
. However, this cannot hold for all ratios of
![]() $a^{\prime}\ne a$
. To understand why, note that the density function
$a^{\prime}\ne a$
. To understand why, note that the density function
![]() ${f}_{AB}(t)$
is the mixed density of the combined populations of treatments A and B. A change in the sample size for treatment A will result in a similar change in both
${f}_{AB}(t)$
is the mixed density of the combined populations of treatments A and B. A change in the sample size for treatment A will result in a similar change in both
![]() ${f}_{AB}(t)$
and
${f}_{AB}(t)$
and
![]() ${f}_{AC}(t)$
. However, since the ratios
${f}_{AC}(t)$
. However, since the ratios
![]() ${h}_A(t)/{h}_B(t)$
and
${h}_A(t)/{h}_B(t)$
and
![]() ${h}_A(t)/{h}_C(t)$
are different over some intervals, the change in sample size
${h}_A(t)/{h}_C(t)$
are different over some intervals, the change in sample size
![]() ${n}_A$
will certainly have different magnitudes of influence on
${n}_A$
will certainly have different magnitudes of influence on
![]() ${\mathrm{HR}}_{AC}^{\prime }$
and
${\mathrm{HR}}_{AC}^{\prime }$
and
![]() ${\mathrm{HR}}_{AB}^{\prime }$
. As a result, the equation
${\mathrm{HR}}_{AB}^{\prime }$
. As a result, the equation
![]() ${\mathrm{HR}}_{AC}^{\prime }/{\mathrm{HR}}_{BC}={\mathrm{HR}}_{AB}^{\prime }$
cannot always hold.
${\mathrm{HR}}_{AC}^{\prime }/{\mathrm{HR}}_{BC}={\mathrm{HR}}_{AB}^{\prime }$
cannot always hold.
Therefore, the only scenario in which the HR is transitive under expectation is when all treatment groups have proportional hazards.