Introduction
The Developmental Origins of Health and Disease (DOHaD) hypothesis postulates that environmental exposures, particularly during critical periods of fetal and early postnatal development, have profound and long-lasting consequences for health and disease risk across the life course. Reference Barker and Osmond1–Reference Gluckman, Hanson and Buklijas4 In Japan, the prevalence of low birth weight (LBW; <2500 g) has remained around 10% since the 1980s. Reference Itoh, Aoyama, Kohmura-Kobayashi and Nemoto5 LBW is associated with an increased risk of metabolic and cardiovascular diseases Reference Itoh, Aoyama, Kohmura-Kobayashi and Nemoto5,Reference Nemoto and Sagawa6 ; one proposed mechanism is a mismatch between a thrifty, in utero-programmed phenotype and nutrient-rich postnatal environments. Reference Gluckman, Hanson, Buklijas, Low and Beedle7,Reference Gluckman, Hanson, Cooper and Thornburg8
Our previous study using a rat model of fetal undernutrition demonstrated that LBW offspring exhibited impaired postnatal growth and reduced hepatic insulin-like growth factor 1 (IGF-1) expression, despite unaltered plasma growth hormone (GH) levels. Reference Nemoto and Kakinuma9 Additionally, LBW rats show exaggerated corticosterone responses to stress, indicating impaired negative feedback regulation of the hypothalamic–pituitary–adrenal (HPA) axis, particularly at the pituitary level. Reference Nemoto, Kakinuma and Shibasaki10 Mechanistically, increased expression of growth arrest-specific 5 (Gas5) long non-coding RNA (lncRNA), a competitive inhibitor of glucocorticoid receptor (GR) function, and decreased expression of miR-23b, which interacts with Gas5, prevent the suppression of corticotropin-releasing factor receptor expression in the pituitary gland, thereby attenuating glucocorticoid negative feedback. Reference Nemoto, Kakinuma and Shibasaki10–Reference Nemoto, Morita and Kakinuma12
We have previously demonstrated that dietary supplementation with methyl-donor nutrients (e.g., folate and vitamin B12) and related one-carbon cofactors (e.g., betaine/choline) during late pregnancy or early lactation normalizes stress-induced corticosterone elevation in LBW offspring. Reference Nemoto and Kakinuma13 In this context, the present study was designed to determine whether nutritional modulation during the lactational period can attenuate the adverse developmental programming induced by fetal undernutrition. Specifically, we sought to investigate whether dietary supplementation with soy protein isolate (SPI) during lactation can reprogram growth and stress axis regulation in offspring born with LBW, ultimately reducing disease risk in later life. SPI, a high-quality protein source with unique amino acid composition contains bioactive components such as isoflavones and methyl donors, has been shown to modulate endocrine and metabolic regulation. Reference Humayun, Elango, Moehn, Ball and Pencharz14–Reference Torre-Villalvazo, Gonzalez, Aguilar-Salinas, Tovar and Torres19 We hypothesized that SPI given to lactating dams would (i) improve offspring growth and circulating IGF-1, (ii) restore pituitary GR-mediated feedback (Gas5/miR-23b/Pomc axis), and (iii) attenuate stress-induced corticosterone hypersecretion. This study examined whether SPI supplementation during lactation ameliorates growth impairment, improves IGF-1 signaling, and normalizes stress-induced corticosterone hypersecretion in LBW offspring. These findings support the DOHaD theory, which posits that early-life nutritional exposures elicit long-term effects by reprogramming gene expression and neuroendocrine pathways through epigenetic mechanisms. As sex steroids modulate hypothalamic corticotropin-releasing factor (CRF) circuitry, Reference Kuriyama and Shibasaki20,Reference Sekino, Ohata, Mano-Otagiri, Arai and Shibasaki21 we assessed stress responsiveness in males to minimize hypothalamic estrogen-receptor confounding; female growth outcomes were analyzed separately. We further investigated whether the impaired glucocorticoid feedback was pituitary-specific and evaluated the peripheral metabolic consequences of prolonged glucocorticoid exposure.
Materials and methods
Animal model and dietary intervention
Wistar rats were housed at 23 ± 2°C under a 12-h light/dark cycle (lights on at 08:00 h, off at 2000 h) with ad libitum access to food and sterile water. All procedures were approved by the Laboratory Animals Ethics Review Committee of Nippon Medical School (approval nos. 27-067 and 2020-003) and conducted in accordance with institutional and national guidelines.
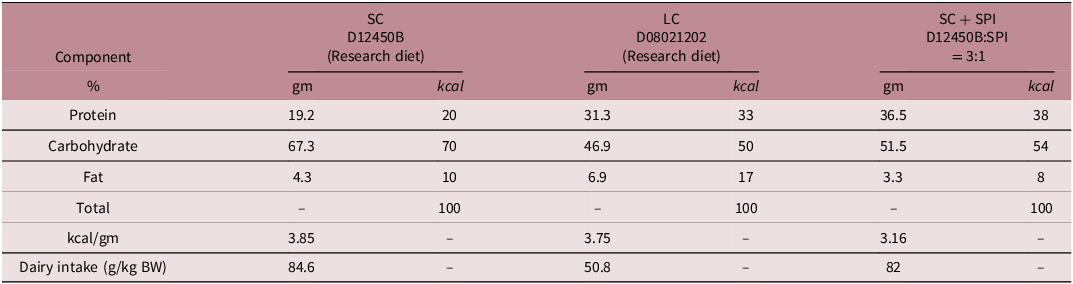
Female Wistar rats (12–15 weeks of age) were mated with untreated 15-week-old control males and randomly allocated to one of three dietary groups: (1) standard chow (SC; D12450B, Research Diets), (2) low-carbohydrate, calorie-restricted diet (LC; D08021202, Research Diets), Reference Nemoto and Kakinuma9 or (3) LC during gestation followed by SPI-supplemented diet during lactation (3 parts D12450B + 1 part SPI, w/w; Table 1 and Figure S1). Estrous cycle stage was determined using the vaginal impedance method; females identified as being in proestrus were co-housed with males overnight. The presence of a vaginal plug the following morning was designated as gestational day (GD) 0, at which point dietary restriction was initiated in the LBW groups.
Composition of the diets consumed by the rats

Throughout gestation, maternal food intake and body weight were recorded daily. Parturition occurred on GD 22. At birth, litter size and pup body weight were recorded. Litter size ranged from 11 to 15 pups, with no significant differences among groups, and sex ratio did not differ between groups. All pups remained with their biological dams (no cross-fostering). Litters were standardized to 10–12 pups within 24 h to minimize variation in postnatal nutrition.
For the SPI intervention, dams in the LBW + SPI group received the SPI-containing diet ad libitum from the day of parturition (postnatal day, PND 0) through PND 7. From PND 8 onward, all dams were returned to standard chow. Offspring were maintained with their biological dams throughout lactation.
For glucocorticoid challenge experiments, additional male pups from the same litters were allocated to hydrocortisone, dexamethasone, or vehicle treatment groups. Dexamethasone-treated groups consisted of 7 pups (from 3 litters) per experimental group. Details of sample sizes (number of litters, n) for each experiment are provided in the figure legends.
Sample collection and experimental procedures
At 3 weeks, male offspring (n = 6 per group) were euthanized via decapitation, and trunk blood was collected for hormone assays. For glucocorticoid challenge experiments, additional pups received intraperitoneal injections of hydrocortisone (100 µg/kg BW; #H0396, Sigma-Aldrich, St. Louis, MO, USA) or dexamethasone (200 µg/kg BW; ZENOAQ, Fukushima, Japan) dissolved in sterile saline. Reference Nemoto, Mano and Shibasaki22 Post-injection sampling was performed at 4 hours (hydrocortisone) or 24 hours (dexamethasone) to assess glucocorticoid receptor (GR)–responsive gene expression and systemic effects.
Experimental unit, randomization, and blinding
The litter was treated as the experimental unit for all pre-weaning endpoints. For pre-weaning outcomes (e.g., birth and weaning weights, circulating hormone levels), four pups per litter were randomly selected for analysis to avoid pseudoreplication. For post-weaning analyses, up to four males and four females per litter were randomly selected, unless otherwise specified. Sample sizes were determined a priori (α = 0.05, power = 0.8) based on previous variance estimates of IGF-1, targeting a 20% detectable difference. Exact n values (litters) are reported in the Methods and figure legends. Investigators responsible for body-weight measurements, hormone assays, and gene-expression analyses were blinded to group allocation until completion of data collection and analysis.
Inclusion and exclusion criteria
Inclusion criteria were live-born pups from term pregnancies. Exclusion criteria were stillbirths, litters with fewer than 10 pups, or technical assay failures (eg, RNA extraction or qPCR).
Restraint stress protocol
At 6 weeks of age, the rats were subjected to restraint stress by enclosure in a flexible wire mesh (12 mm × 12 mm) for 120 min between 09:00 and 12:00 h in an isolated room. Reference Nemoto, Kakinuma and Shibasaki10 Immediately after restraint, the animals were sacrificed in an adjacent room, and trunk blood was collected. Non-stressed control rats were housed in separate rooms and handled identically without restraint exposure.
RNA isolation and real-time RT-PCR
Total RNA was extracted from the pituitary gland, liver, soleus muscle, and perirenal white adipose tissue using RNAiso Plus (Takara, Shiga, Japan). RNA purity was confirmed by spectrophotometric analysis, with all samples showing an A260/A280 ratio > 1.7. For miRNA expression analysis (miR-23b), 500 ng of total RNA was reverse-transcribed using the Mir-X® miRNA First-Strand Synthesis and SYBR® qRT-PCR Kit (Clontech Laboratories, Mountain View, CA). For mRNA analysis, 250 ng of RNA was reverse-transcribed using the PrimeScript® RT Reagent Kit with gDNA Eraser (Takara). PCR amplification was performed using SYBR Premix Ex Taq (Takara) with gene-specific primers for Crhr1, Pomc, Fkbp5 (FK506-binding protein 5), Gpt1 (Glutamic Pyruvic transaminase), Pck1 (phosphoenolpyruvate carboxykinase), Igf1, Trim63 (tripartite motif-containing 63), Gapdh (internal control), and Gas5 lncRNA. Primer sets were obtained from Takara (RA025834, RA013901, RA049269, RA091999, RA065599, RA028844, RA073736, and RA015380). Quantification was performed using the second-derivative Ct method. Reference Nemoto, Mano and Shibasaki22
Hormone assays
Serum concentrations of corticosterone, GH, and IGF-1 were measured using the following ELISA kits: rat corticosterone ELISA (#501320, Cayman Chemical, Ann Arbor, MI, USA), rat GH ELISA (#AKRGH-010; FUJIFILM Wako, Osaka, Japan), and mouse/rat IGF-1 ELISA (#MG100; R&D Systems, Minneapolis, MN, USA). All terminal blood collections were performed between 09:00 and 12:00 h to minimize circadian variability. Because GH secretion is pulsatile, single terminal measurements were used only as a surrogate index of somatotropic activity and interpreted with caution.
Plasma corticosterone concentrations were measured with an assay range of 8.2–5,000 pg/mL and a sensitivity of 30 pg/mL (defined at 80% B/B0). Plasma IGF-1 concentrations were measured with a range of 31.2–2,000 pg/mL and a sensitivity of 8.4 pg/mL. The calibration range of the GH assay was 31.3–2,000 pg/mL; assay sensitivity was not reported by the manufacturer.
Statistical analysis
Data are presented as means ± standard error of the mean (SEM). Statistical significance was assessed using one-way ANOVA followed by Tukey’s post-hoc test, two-way ANOVA, or Pearson’s correlation, as appropriate. Assumptions of ANOVA (normality and homoscedasticity) were verified before testing. Real-time RT-PCR results were normalized to the control set at 100%. A p-value of <0.05 was considered statistically significant. Analyses were conducted using the Prism 9 software (GraphPad Software, La Jolla, CA, USA).
Results
Soy protein intervention improves growth and IGF-1 levels in LBW offspring
Offspring born to dams fed a low-carbohydrate, calorie-restricted diet (LC group) exhibited significantly lower body weights at birth (Fig. 1a) and weaning (Fig. 1b) than the control offspring. Plasma IGF-1 concentrations were markedly reduced in LC offspring. However, postnatal dietary intervention with SPI during lactation normalized the body weight of female offspring at the time of weaning and restored IGF-1 levels to control values (Fig. 1c, d). In males, body weight and circulating IGF-1 levels at weaning tended to improve with SPI supplementation (p = 0.35 for body weight and p = 0.13 for IGF, vs LC, respectively); however, these changes were not statistically significant.
Body weight and growth-related endocrine parameters in low birth weight (LBW) offspring. (a) Birth and weaning body weights of male and female offspring from dams fed a control, low-carbohydrate (LC), or soy protein isolated-supplemented diet (3 parts D12450B + 1 part SPI, w/w) during lactation (SPI). Body weights were measured at birth and weaning; no intermediate time-point data were collected. Thus, only these two developmental milestones are presented. (b) Plasma insulin-like growth factor-1 (IGF-1) concentrations in offspring at weaning. Statistical differences among groups were analyzed by one-way ANOVA followed by Tukey’s post-hoc test. Bars represent means ± SEM (n = litters). n = 20 for panels (a) and (b); n = 8 for panels (c) and (d). *P < 0.05, ***P < 0.0005, and ****P < 0.0001.

SPI Normalizes Pituitary Gene Expression and Basal Corticosterone Regulation
In the pituitary glands of LBW offspring, the expression of the glucocorticoid receptor (GR) decoy Gas5 lncRNA was elevated (Fig. 2a), whereas expression miR-23b and proopiomelanocortin (Pomc) mRNA was suppressed (Fig. 2b, c). Although basal serum corticosterone concentrations did not differ significantly between normal birth weight (NBW) and LBW rats, SPI intervention reduced Gas5 lncRNA levels and significantly increased miR-23b and Pomc mRNA expression (Fig. 2).
Pituitary expression of growth arrest–specific 5 (Gas5) long non-coding RNA (lncRNA) (a), miR-23b (b), and proopiomelanocortin (Pomc) mRNA (c), and serum corticosterone concentrations (d) in normal birth weight (NBW), birth weight (LBW), and SPI-treated LBW offspring. Values are presented as mean ± SEM. n = 5. *P < 0.05, and ****P < 0.0001.

SPI attenuates stress-induced corticosterone hypersecretion in LBW rats
When subjected to 120 min of restraint stress, LBW rats exhibited a markedly exaggerated increase in serum corticosterone levels relative to NBW controls, suggesting impaired negative feedback regulation of the HPA axis. Stress-induced corticosterone hypersecretion was significantly reduced in LBW rats that received SPI during lactation (Fig. 3).
Serum corticosterone concentrations following 120 min of restraint stress in normal birth weight (NBW), low birth weight (LBW), and SPI-treated LBW offspring. Values are presented as mean ± SEM. n = 5. *P < 0.05 vs. non-stressed, **P < 0.001 vs. non-stressed, ***P < 0.0005 vs. non-stressed, ****P < 0.0001 vs. non-stressed, and †† P < 0.05 vs. NBW.

Tissue-specific glucocorticoid sensitivity and metabolic dysregulation in LBW offspring
Gas5 lncRNA expression was selectively upregulated in the pituitary glands of LBW offspring, with no significant changes detected in the liver, skeletal muscle, or white adipose tissue. Following hydrocortisone administration, the pituitary expression of Fkbp5 mRNA, a GR target gene, was significantly reduced in LBW rats, indicating impaired receptor responsiveness. In contrast, dexamethasone treatment increased hepatic mRNA expression of Gpt1 and Pck1 and elevated blood glucose levels, reflecting enhanced hepatic glucocorticoid sensitivity. Additionally, hepatic Igf1 expression was decreased, and skeletal muscle Trim63 mRNA expression was upregulated in both NBW and LBW offspring, consistent with impaired growth signaling and increased proteolysis. These changes were attenuated by SPI treatment (Fig. 4).
(a) Tissue-specific expression of Gas5 lncRNA in the pituitary gland, liver, skeletal muscle, and white adipose tissue. (b) Pituitary Fkbp5 mRNA expression 4 h after intraperitoneal administration of hydrocortisone administration (100 µg/kg body weight). (c) Hepatic Gpt1 and Pck1 mRNA expression and blood glucose concentrations 24 h after dexamethasone administration (200 µg/kg body weight). (d) Hepatic Igf1 and skeletal muscle Trim63 mRNA expression 4 h after hydrocortisone administration (100 µg/kg body weight). NBW indicates normal birth weight; LBW, low birth weight. Values are presented as mean ± SEM. n = 7. *P < 0.05, **P < 0.001, ***P < 0.0005, and ****P < 0.0001.

Discussion
This study demonstrated that offspring born to dams fed a low-carbohydrate, calorie-restricted diet during gestation exhibited impaired postnatal growth, reduced IGF-1 signaling, and dysregulated HPA-axis function, features consistent with a stress-sensitive, thrifty phenotype. Lactational supplementation with SPI during the early postnatal period partially or fully normalized these abnormalities, indicating that early-life nutritional modulation can counteract adverse developmental effects established in utero.
Postnatal growth and the somatotropic axis
LBW offspring displayed reduced body size at birth and weaning, with lower circulating IGF-1 levels despite unaltered plasma GH levels. This dissociation indicates suppression of the somatotropic axis, consistent with the thrifty phenotype model in which energy is preferentially allocated to immediate survival at the expense of growth. SPI supplementation increased circulating IGF-1 levels and improved body weight at weaning, effects that were most pronounced in females. Amino acids abundant in SPI, such as arginine and glutamine, are known GH secretagogues, and their presence may have contributed to the observed increase in IGF-1 through nutrient signaling pathways. Reference Collier, Casey and Kanaley23–Reference Reid, Dai and Locasale25 These findings align with evidence that early postnatal nutritional interventions can recalibrate growth trajectories disrupted by prenatal undernutrition. Reference Balbus, Barouki and Birnbaum26
The sex-specific growth response observed here parallels previous reports that early-life nutritional interventions can produce differential outcomes depending on the hormonal milieu and early-life environment. Reference Lee, Thompson and Vanden Heuvel27–Reference Tarry-Adkins and Ozanne29 Although males exhibited only modest improvements in growth, the biological basis of these sex differences warrants further study, including the potential influence of estrogen receptor-mediated modulation of GH and IGF-1 signaling.
Notably, circulating GH concentrations were not significantly altered by SPI intervention (Fig. 1), suggesting that the observed restoration of IGF-1 levels and postnatal growth cannot be attributed solely to enhanced GH secretion. Instead, these findings raise the possibility that SPI acts downstream of GH, potentially through hepatic IGF-1 expression and/or sensitivity to nutritional cues. Hepatic IGF-1 expression and secretion are highly responsive to amino acid availability and overall protein nutrition, independent of changes in circulating GH, and the improved IGF-1 profile in SPI-exposed offspring likely reflects such direct nutritional regulation at the hepatic level. At the same time, SPI was provided only during the first postnatal week, yet its effects on growth persisted beyond the intervention period, suggesting that early-life amino acid-rich nutrition may also influence longer term regulatory set points of the somatotropic axis.
It is important to distinguish between the direct nutritional effects of amino acids and developmental programming. Although certain amino acids in SPI might act as acute secretagogues for growth-related hormones, the fact that the ameliorative effects on the GH-IGF-1 axis were sustained even after the cessation of SPI supplementation supports the occurrence of nutritional programming. This suggests that early postnatal SPI exposure during a critical window may reset the developmental trajectory of LBW offspring, potentially through lasting functional changes in the pituitary-hepatic axis. Clarifying the relative contributions of acute nutrient signaling versus more durable programming mechanisms will require future studies incorporating direct epigenetic analyses.
HPA axis dysregulation and SPI-mediated restoration
At the molecular level, LBW offspring exhibited increased expression of Gas5 and decreased levels of miR-23b in the pituitary. Gas5 acts as a decoy GR response element, inhibiting GR-mediated transcription, Reference Kino, Hurt, Ichijo, Nader and Chrousos30 and its upregulation likely contributes to impaired glucocorticoid negative feedback. Reduced Pomc mRNA, a precursor of ACTH, further supports blunted pituitary GR responsiveness. SPI supplementation reversed these molecular changes by downregulating Gas5, upregulating miR-23b, and restoring Pomc expression and basal corticosterone concentrations, indicating improved pituitary sensitivity to glucocorticoid feedback. These alterations in pituitary Gas5 lncRNA and miR-23b expression suggest that early-life diet can modulate regulators of GR sensitivity and thereby influence programming of the stress axis. Given that miR-23b has been identified as a post-transcriptional regulator of glucocorticoid receptor signaling, Reference Shen, Liu and Fu31 the restoration of miR-23b levels by SPI supplementation may contribute to the normalization of HPA axis feedback sensitivity. Furthermore, considering the established interaction between Gas5 lncRNA and glucocorticoid receptors, these findings suggest that SPI recalibrates a coordinated non-coding RNA network to mitigate early-life programming of stress responsiveness. In the present study, the restoration of miR-23b expression levels in the SPI-supplemented group suggests that early nutritional intervention can stabilize post-transcriptional regulatory mechanisms that are otherwise disrupted by fetal undernutrition.
Although sustained effects following early-life nutritional intervention are often discussed in the context of epigenetic programming, our data do not demonstrate direct epigenetic modifications at the Gas5 or Pomc loci. Targeted analysis of DNA methylation across Gas5 coding and promoter-associated regions revealed no significant differences between groups, suggesting that altered Gas5 expression is not mediated by local CpG methylation at these sites (Fig. S2). Importantly, Pomc mRNA expression remained inducible following stress exposure, Reference Nemoto, Kakinuma and Shibasaki10 indicating preserved transcriptional competence of the Pomc gene and supporting the interpretation that elevated Pomc expression in LBW offspring reflects impaired GR-mediated negative feedback rather than intrinsic Pomc dysregulation. Thus, while we cannot exclude epigenetic mechanisms acting at distal regulatory regions or through chromatin-level modifications, our findings more directly support a model in which early lactational nutrition alters endocrine set points and hormone sensitivity.
Central glucocorticoid resistance and peripheral hypersensitivity
Tissue-specific analyses revealed that Gas5 upregulation was restricted to the pituitary gland, with no changes in peripheral organs, suggesting a central origin of glucocorticoid resistance. Consistent with this, pituitary expression of Fkbp5, a marker of GR activation, Reference Davies, Ning and Sanchez32,Reference Kageyama, Iwasaki, Watanuki, Niioka and Daimon33 was blunted after hydrocortisone injection. In contrast, peripheral glucocorticoid sensitivity appeared to be exaggerated; Gpt1 and Pck1 were overexpressed, and dexamethasone treatment induced hyperglycemia. Additionally, suppression of hepatic Igf1 and upregulation of muscle Trim63, a ubiquitin ligase associated with muscle atrophy, indicated reduced anabolic signaling and enhanced catabolism in peripheral tissues. This combination of central glucocorticoid resistance and peripheral hypersensitivity is a well-recognized consequence of adverse intrauterine environments and has been linked to increased disease susceptibility later in life. Reference Poore, Boullin and Cleal34–Reference Martin, Wang and Lye36
SPI supplementation mitigated these maladaptive peripheral responses, likely through normalization of circulating corticosterone levels and improved central regulation of the HPA axis. The glucocorticoid challenge experiments further clarify the level at which GR signaling is altered. Hydrocortisone, which is metabolized and acts through endogenous corticosterone production, elicited an exaggerated corticosterone response in LBW males, indicating impaired negative feedback within the central HPA axis. In contrast, dexamethasone, a synthetic glucocorticoid that potently suppresses ACTH via pituitary and hypothalamic GR, produced only partial suppression of corticosterone in LBW rats, again consistent with reduced central GR sensitivity. SPI supplementation normalized the corticosterone response to both hydrocortisone and dexamethasone, supporting the interpretation that lactational SPI improves GR-mediated feedback rather than altering Pomc transcriptional competence per se.
Implications for DOHaD and early life intervention
Our findings fit squarely within the DOHaD paradigm, which emphasizes that environmental exposures during critical developmental windows can have lasting consequences for health and disease risk. The combination of impaired somatotropic signaling, central glucocorticoid resistance, and peripheral hypersensitivity observed in LBW offspring provides a mechanistic basis for their increased vulnerability to metabolic and neuroendocrine disorders. Targeting the lactational period – a critical postnatal window – through SPI supplementation partially reversed these abnormalities, indicating that safe, food-based interventions may enhance endocrine and metabolic resilience in at risk offspring.
Rather than simply repeating the observed results, these data suggest that early-life soy protein supplementation acts at multiple levels of endocrine regulation: supporting hepatic IGF-1 production, normalizing GR-mediated feedback in the pituitary–hypothalamic axis and reducing peripheral glucocorticoid over-responsiveness. Such multi-level modulation of hormonal pathways provides a plausible mechanistic framework for how early nutrition can shape long-term health trajectories.
Future directions
Long-term studies are needed to determine whether the benefits of SPI persist into adulthood and whether they influence reproductive fitness, lifespan, or transgenerational outcomes. Comprehensive omics analyses, including transcriptomics, proteomics, and epigenomics, can clarify the molecular pathways through which SPI affects the somatotropic and HPA axes. Additionally, extending these findings to other protein sources and mixed diets could help identify whether the effects are specific to soy protein or generalizable to other functional proteins. The present findings underscore the need for life-course approaches to disease prevention, emphasizing nutritional modulation during sensitive developmental windows. While our study focused on a rat model, future research should assess the translatability and durability of these molecular signatures in human populations and across generations. Such knowledge will facilitate the development of effective nutritional interventions for NCD prevention, aligned with public health priorities at both population and policy levels. Translational studies linking early nutrition to adult metabolic health will be essential to advance DOHaD-informed interventions.
Limitations
This study has several limitations. First, growth hormone secretion is pulsatile, and our use of single terminal blood samples did not allow assessment of pulsatility dynamics, although this limitation is common in small-animal studies. We therefore interpreted growth outcomes primarily through circulating IGF-1 concentrations and somatic growth parameters. Second, female offspring were not included in the stress reactivity tests; although our focus was on pituitary-specific glucocorticoid resistance in males, future studies should address sex-specific responses more comprehensively. Third, dietary SPI contained isoflavones and vitamins in addition to amino acids, and it is difficult to fully disentangle the relative contribution of these components to the observed effects. Moreover, we did not analyze milk composition in this study, which is an additional limitation. Maternal diet has been shown to alter rat milk composition, including macronutrients and bioactive components, with consequent effects on offspring growth and metabolism. Reference Del Prado, Delgado and Villalpando37,Reference Hallam, Barile, Meyrand, German and Reimer38 It is therefore plausible that SPI may have exerted part of its effects indirectly through changes in milk composition, which warrants direct investigation in future studies. Finally, while our experimental design minimized pseudoreplication by treating the litter as the unit of analysis, replication in additional cohorts is warranted to confirm generalizability.
Despite these limitations, our revisions – including detailed diet composition tables, explicit randomization and blinding procedures, and power-based sample-size determination – enhance the rigor and reproducibility of the study. These methodological refinements enhance the reproducibility and interpretability of our findings.
Conclusion
Together, the present results highlight that nutritional modulation during early postnatal life – particularly via functional dietary proteins such as SPI – can partially reverse adverse developmental programming induced by fetal undernutrition. By establishing resilient endocrine and metabolic trajectories, such early-life nutritional modulation fits squarely within the DOHaD paradigm, and these interventions may hold promise for reducing disease risk and improving health outcomes across the life course. Our results suggest that SPI does not merely act as a transient nutritional support but functions as a developmental programming agent that resets the endocrine trajectory of LBW offspring.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S2040174426100531.
Data availability statement
Data described in the manuscript, code book, and analytic code are not publicly available at present, but will be provided by the corresponding author upon reasonable request. At present, no data have been deposited in a public repository.
Acknowledgements
SPI was provided by Fuji Oil Co., Ltd We would like to thank Editage (www.editage.jp) for English language editing.
Author contributions
The authors’ responsibilities were as follows: TN designed the research; TN and HY conducted the research; TN and YK analyzed data; TN wrote the manuscript; all authors read and approved the final manuscript.
Funding statement
This work was supported by the Fuji Foundation for Protein Research (TN grant nos.2023-005 and 2024-021).
Competing interests
The authors declare no conflicts of interest.
Ethical standard
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national guides on the care and use of laboratory animals (rat) and has been approved by the institutional committee (approval nos. 27-067 and 2020-003).”
Declaration of Generative AI and AI assisted technologies in the writing process
During the preparation of this manuscript, the authors used ChatGPT-5, developed by OpenAI, to assist with proofreading and to enhance the clarity of text. After using this tool, the authors carefully reviewed and edited the content and take full responsibility for the content of the manuscript.