Introduction
Major depressive disorder (MDD), a prevalent mental health problem in the world, was identified as the third cause of disease burden globally, with projections that it will become the leading cause by 2030 (Malhi & Mann, Reference Malhi and Mann2018). In China, the lifetime prevalence of depression in adults is approximately 7%, with most patients report experiencing insomnia symptoms during the course of depression disease. Previous study has proposed a bidirectional relationship between insomnia and depression, specifically that insomnia not only acts as an independent risk for developing depression but also serving as a core symptom or complication of depression (Lustberg & Reynolds, Reference Lustberg and Reynolds2000). In addition, effective treatment of insomnia can greatly improve depression outcome, including symptom severity, recurrence, and prognosis (Gebara et al., Reference Gebara, Siripong, DiNapoli, Maree, Germain, Reynolds and Karp2018).
Cognitive impairment, particularly attention, memory, and executive function, is another common feature observed in MDD (Rock, Roiser, Riedel, & Blackwell, Reference Rock, Roiser, Riedel and Blackwell2014; Wagner et al., Reference Wagner, Müller, Helmreich, Huss and Tadic2015). And insomnia has also been recognized as a risk factor for cognitive decline (Fortier-Brochu, Beaulieu-Bonneau, Ivers, & Morin, Reference Fortier-Brochu, Beaulieu-Bonneau, Ivers and Morin2012). Previous studies have shown that MDD patients with insomnia symptom would exhibit more pronounced cognitive deficits, which are associated with reduced frontal lobe activity (Li et al., Reference Li, Li, Zhaung, Yu, Wei, Li and Zhang2024). Furthermore, alterations in cingulate-postcentral functional connectivity (FC) in youth with depression mediate the relationship between insomnia and cognitive dysfunction (Hu et al., Reference Hu, Li, Ye, Zhang, Liu, Chen and Zhou2023b), suggesting the potential neural basis underlying these impairments.
Over the past decades, numerous studies had investigated the alterations of brain FC associated with MDD and insomnia. A meta-analysis identified static FC changes associated with depression in brain networks including the default mode network (DMN), frontoparietal network (FPN), attention, and sensorimotor network (SMN) (Zhang et al., Reference Zhang, Zhang, Wang, Lei, Jiang, Xiong and Liu2025). While another study has indicated that abnormalities in static FC among the DMN, FPN, and salience network (SN) may represent shared neurobiological foundations of depression and insomnia (Bagherzadeh-Azbari et al., Reference Bagherzadeh-Azbari, Khazaie, Zarei, Spiegelhalder, Walter, Leerssen and Tahmasian2019). Nevertheless, brain network is a dynamic and time-varying system (Hutchison et al., Reference Hutchison, Womelsdorf, Allen, Bandettini, Calhoun, Corbetta and Chang2013). Analysis of dynamic connectivity can capture the time-dependent characteristics of brain networks (Allen et al., Reference Allen, Damaraju, Plis, Erhardt, Eichele and Calhoun2014; Li et al., Reference Li, Li, Cao, Jiang, Chai, Zhou and Huang2025). One review had pointed out that MDD was marked by abnormal dynamic FC within the DMN and in the dynamic state of weak connection among networks (Sun et al., Reference Sun, Yan, Qu, Luo, Liu, Tian and Hu2024). Moreover, one multicenter study with a large sample size had supported that static DMN FC alteration was only significant in recurrent MDD patients, but not in first-episode drug-naïve MDD (Yan et al., Reference Yan, Chen, Li, Castellanos, Bai, Bo and Zang2019).
In our previous study, insomnia-dominated subtype of depression was identified employing a data-driven method based on combination of clinical characteristics and abnormal network connectivity (Wang et al., Reference Wang, Tang, Zhang, Bu, Lu, Li and Huang2021). We also found that the adolescents with MDD exhibited different static and dynamic network FC patterns according to different insomnia severity (Bao et al., Reference Bao, Gao, Feng, Li, Li, Cao and Huang2025). However, most previous studies had recruited patients with medication history and with recurrent episodes. As far as we know, no study had explored the brain static and dynamic FC comprehensively in first-episode, drug-naïve adult patients with MDD and not to mention use insomnia symptoms as a distinct subtype to distinguish the neurobiological heterogeneity under the disorder.
Thus, in the current study, we recruit a relatively large group of first-episode, drug-naive MDD patients, who were stratified according to insomnia severity aim to(1)evaluate the cognitive performance in MDD patients with different insomnia severity; (2) performed a data-driven method, independent component analysis (ICA), combined with a sliding-window approach and k-means clustering analysis to assess static and dynamic functional network connectivity (FNC); and (3) explore the association between altered brain network functions with clinical symptom and cognitive performance.
Materials and methods
Participants
All participants were recruited at the Mental Health Center of West China Hospital, aged 18–65 years. All adult MDD patients were diagnosed by two experienced psychiatrists using the Structured Clinical Interview for DSM-IV Axis I Disorders. The exclusion criteria were as follows: (1) history of taking antipsychotic medication; (2) history of traumatic brain injury or other severe physical diseases, and first-degree relatives with psychiatric disorders; (3) other current or past Axis I disorders or serious neurological diseases; (4) drug and drug abuse or dependence; and (5) MRI scan contraindication.
Healthy controls (HCs) from comparable sociodemographic backgrounds were recruited via poster advertisements. HCs were excluded if they had any psychiatric disorders or family psychiatric history among first-degree relatives. Individuals with MRI contraindications, such as severe claustrophobia or metal implants, were also excluded.
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2013. All procedures involving human participants/patients were approved by the Ethics Committee of the West China Hospital, Sichuan University (study ethical approval number: 2018(610)), and written informed consent was obtained from all participants before participation.
Clinical assessment
We used the 17-item Hamilton Depression Scale (HAMD-17) and the 14-item Hamilton Anxiety Scale (HAMA-14) to evaluate the severity of depression and anxiety symptoms, respectively, in the participants. The sum of HAMD-17 items 4, 5, and 6 scores, collectively defined as the HAMD-insomnia subscale, was used to assess the severity of insomnia symptoms in MDD patients. Using a cutoff value of 3 on this subscale (Park et al., Reference Park, Kim, Jun, Lee, Kim, Jeong and Park2013), MDD patients with scores ranging from 1 to 3 were defined as low insomnia MDD (LI-MDD), and those with scores >3 were defined as high insomnia MDD (HI-MDD). We also calculated the HAMD-adjusted scores by removing the insomnia subscale to minimize the effect of sleep disturbance on depressive symptom scores.
Neuropsychological test
The neuropsychological test was used to assess three domains of cognitive function relevant to MDD, including episodic memory, attention and processing speed, and executive function (Hu et al., Reference Hu, Li, Zhao, Dong, Qiu, Yang and Gong2023a). Specifically, the Logical Memory I and II, Visual Reproduction I and II for evaluating episodic memory. The Digit Span Forward, Digit Symbol Substitution Test, the Trail Making Test A (TMT_A), and the Trail Making Test B (TMT_B) for assessing attention and processing speed. The Stroop Color and Word Test (SCWT), Digit Span Backward for evaluating Executive function.
MRI data acquisition
All participants were scanned using a 3.0 T SIMENS MR scanner with an eight-channel phased-array head coil. Participants were instructed to rest with their eyes closed, remain awake, and refrain from engaging in structured thinking.
We acquired three-dimensional (3D) T1-weighted images using a spoiled gradient-recall sequence with the following parameters: repetition time = 1900 ms, echo time = 2.26 ms, flip angle = 9°, number of slices = 176, slice thickness = 1 mm, field of view = 256 × 256 mm2, and matrix = 256 × 256.
Functional MRI images were acquired using a gradient-echo echo-planar imaging sequence with the following parameters: repetition time = 2000 ms, echo time = 30 ms, flip angle = 90°, number of slices = 30 slices, slice thickness = 5 mm with no gap, field of view = 240 × 240 mm2, matrix = 64 × 64, and voxel size = 3.75 × 3.75 × 5 mm3. Each functional scanning session lasted 5.8 min, yielding 175 time points per subject.
Imaging preprocessing
We preprocessed the functional MRI data with the Data Processing & Analysis for Brain Imaging toolkit (http://www.restfmri.net; Yan, Wang, Zuo, & Zang, Reference Yan, Wang, Zuo and Zang2016). The main preprocessing steps including first 10 volumes removed, slice timing, head motion correction, normalize, and smoothing and details can be found in the Supplementary Materials.
Network identification
We used group-level ICA (GICA) to identify independent brain networks using group ICA fMRI toolbox (GIFT v4.0). The optimal component number (40) was estimated by minimum description length criterion (Hui et al., Reference Hui, Li, Wen, Yao and Long2011). After reducing the dimension of fMRI data using principal component analysis, we performed ICA with ICASSO (20 runs) to extract 40 components (Himberg, Hyvärinen, & Esposito, Reference Himberg, Hyvärinen and Esposito2004). The detailed steps of ICA are available in Supplementary Material.
Next, we selected 24 meaningful independent components (ICs) by evaluating the spatial correlation between each IC and the standard Yeo brain functional network template (Buckner et al., Reference Buckner, Krienen, Castellanos, Diaz and Yeo2011; Choi, Yeo, & Buckner, Reference Choi, Yeo and Buckner2012; Yeo et al., Reference Yeo, Krienen, Sepulcre, Sabuncu, Lashkari, Hollinshead and Buckner2011). These 24 ICs were sorted into eight networks based on Yeo’s seven-network template, the subcortical template, and the cerebellum template. As there was no IC sorted into limbic network, we finally identified eight networks including the DMN, FPN, SMN, visual network (VIS), dorsal attention network (DAN), ventral attention network (VAN), subcortical network (SC), and cerebellum network (CeN). For the VAN, we defined the component located in insula as lateral VAN and located in dACC as medial VAN (Urchs et al., Reference Urchs, Tam, Orban, Moreau, Benhajali, Nguyen and Bellec2022). Postprocessing steps, including detrending, 3D despiking, and filtering, were implemented to avoid potential effects from noise. Spatial maps of all selected ICs can be seen in Supplementary Figure 1. And the MNI coordinates and network division of 24 ICs are available in Supplementary Table 1.
Static FNC analysis
To evaluate the FC between the identified networks, we extracted the time series of each IC and calculated Pearson correlations among ICs time series from eight networks to obtain static FNC strengths. The coefficients were transformed to Fisher z values to improve the normality of the FNC strengths. Finally, a 24 × 24 static FNC matrix was acquired for each subject for statistical analysis.
Dynamic FNC analysis
Sliding window analysis
A sliding window method was applied to obtain the FNC matrix for each time window, with a window length of 30 TRs, which is considered optimal for effectively capturing dynamic fluctuations in FNC (Preti, Bolton, & Van De Ville, Reference Preti, Bolton and Van De Ville2017). A 1 TR step was used, along with a Gaussian filter width of σ = 3 TRs, resulting in 135 windows. Subsequently, the least absolute shrinkage and selection operator technique was utilized to estimate the regularized inverse covariance matrix (Friedman, Hastie, & Tibshirani, Reference Friedman, Hastie and Tibshirani2008), employing the L1-norm with a value of 10 to encourage sparsity. The resulting FNC matrix was then converted to Fisher z scores using the Fisher r-to-z transformation to enhance the normality of the strengths. Finally, 24 × 24 FNC matrices were obtained for each subject across 135 windows for subsequent analysis.
Clustering analysis and temporal properties calculation
To investigate the structure and frequency of the reoccurring FNC configuration over time and across subjects, we applied the K-means algorithm to categorize the dynamic FNC windows into distinct cluster states (Lloyd, Reference Lloyd1982). We repeated the algorithm 100 times with k ranging from 2 to 10 to determine the optimal number of clusters. Based on the elbow criterion (Allen et al., Reference Allen, Damaraju, Plis, Erhardt, Eichele and Calhoun2014), the optimal number of clusters was found to be k = 4 in the current study. Finally, the 135 FNC matrix of each subject was assigned to four different states. Three temporal properties, including fractional state time (the percentage of time spent in the four states), mean dwell time (the time spent in each state), and number of transitions (how many times one state switch to another) were calculated for every subject.
Validation analysis
In addition, we conducted validation analyses to evaluate the reliability of our findings under different window length or step size by using 22 TRs as the window length with 1 TR as step, as well as using 30 TRs as length with 3 TRs as the step. We also validated the findings of static FNC by using Yeo’s 17-network atlas, including 17 cortical networks, one cerebellar network, and one SC.
Statistical analysis
Group differences
We conducted a one-way analysis of variance (ANOVA) to assess differences in age across the three groups, followed by post hoc tests with Bonferroni correction to detect between-group differences. The chi-square test was applied to evaluate differences in gender distribution among the three groups. Two-sample t tests were conducted to detect group difference in clinical symptoms between HI-MDD and LIMDD with age and gender as covariates, and also to detect group difference in neuropsychological tests between HI-MDD and LIMDD after controlling age, gender, and HAMD-adjusted score.
Two-sample t test was conducted to identify differences in static and dynamic FNC strengths as well as temporal properties between whole MDD and HC with age, gender, and mean FD as covariates. A one-way ANOVA model was employed to assess differences in static and dynamic FNC strengths as well as temporal properties across three groups, while adjusting for age, gender, mean FD, and HAMD-adjusted scores. The statistical significance threshold was established at p < 0.05 following false discovery rate (FDR) correction for multiple comparisons. Subsequent to the primary analysis, post hoc pairwise comparisons were conducted with statistical significance defined by Bonferroni-corrected thresholds maintaining p < 0.05/3.
Correlation analysis with clinical features and moderation analysis
Partial correlation analysis was conducted to explore the relationship between insomnia severity and cognitive function (with and without controlling for HAMD-adjusted score), as well as between depression severity and cognitive function (with and without controlling for insomnia score). We also explore the association between altered FNC/temporal properties and clinical symptoms (i.e. depression, anxiety, and insomnia), and cognitive performance with age, gender, and mean FD as covariates, in both the entire MDD group and its subgroups. The 5000 permutation tests were conducted to validate the significance of partial correlations. We set the statistical threshold at p < 0.05, corrected for FDR across all connections. In addition, we also explored the moderation effect of insomnia on relationship between alter FNC and cognitive performance with age, gender, and mean FD as covariates. For the exploratory purpose, we set the statistical threshold at p < 0.05 without multiple comparisons correction for moderation analysis.
To explore potential complex, multivariate relationships between altered FNC and cognitive domains, we performed canonical correlation analysis (CCA) in HI-MDD and LI-MDD groups separately. We set the statistical threshold at p < 0.05 with 5000 permutation tests.
Results
Demographic and clinical characteristics
A total of 203 adult MDD (133 HI-MDD, 70 LI-MDD) and 122 HCs were recruited in our study. There were no significant differences across the LI-MDD, HI-MDD, and HC groups in age and gender. HI-MDD group showed significantly higher HAMD-17 total score, HAMD-insomnia score, HAMD-adjusted score, and the HAMA score compared with LI-MDD group. The HI-MDD group showed significant lower score in the Logical Memory I and II, Visual Reproduction II, Digit Span Backward, and Digit Span forward than the LI-MDD group. The demographic and clinical characteristics can be seen in Table 1 and Supplementary Figure 2.
The demographic and clinical characteristics of HI-MDD, LI-MDD, and HC groups

Note: MDD, major depressive disorder; LI-MDD, Low insomnia MDD; HI-MDD, high insomnia MDD; HC, health control; HAMD, Hamilton Depression Rating Scale; HAMA, Hamilton Anxiety Rating Scale; n, number; n.s., no significant differences; NA, not available.
a ANOVA.
b Chi-square tests.
c Two-sample t test.
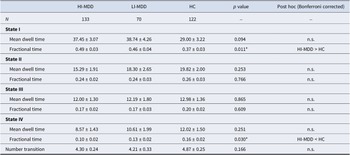
The results of dynamic properties among HI-MDD, LI-MDD, and HC groups

Note: MDD, major depressive disorder; LI-MDD, Low insomnia MDD; HI-MDD, high insomnia MDD; HC, health control; n, number; n.s., no significant differences.
Static FNC
There was no significant difference in static FNC (connectivity averaged over the entire session) between the entire aMDD group and HCs, and also no group difference among three groups after multiple comparison corrections (FDR corrected p > 0.05). This situation changed distinctly once we considered dynamic measures of connectivity.
Dynamic FNC
Dynamic states identified by clustering analysis
The FNC matrix of 135 windows with eight networks for each subject could be clustered into four configuration states. In all participants, State I, which occurred most frequently (19,312 times, 44%), was characterized by widely sparse and weak positive connections among the eight networks. State II, the second most frequent state (10,964 times, 25%), was characterized by strong connection within both DMN and VIS, as well as more dense positive connections among eight networks than state I. State III (7979 times, 18%) was characterized by strong connection within the SMN and VIS, also between SMN and VIS. State IV (5620 times, 13%) was characterized by strong positive and negative connections among eight networks. The strong positive connections primarily involved the SMN and VIS intranetwork and internetwork connections, while the strong negative connections mainly involved the DMN, SMN, SC, and VIS internetwork connections.
Temporal properties of dynamic states
For temporal properties of dynamic FNC states, we discovered that whole MDD group showed increased the percentage of time spent in state I and significantly decreased the percentage of time spent in state IV than HC. We found significant differences in the fractional state time of states I and IV across the three groups (p < 0.05, FDR corrected). Post hoc tests revealed that HI-MDD performed significantly increased the percentage of time spent in state I and significantly decreased the percentage of time spent in state IV than HC (Bonferroni corrected). No significant differences were found between HI-MDD and LI-MDD, or between LI-MDD and HC. There were no significant group differences for dwell time and transition number. And the dynamic state properties can be seen in Figure 1 and Table 2.
Temporal properties of dynamic functional connectivity states. The four dynamic functional connectivity states of all participants (MDD and HC) identified by clustering analysis (A). Group comparisons of temporal properties (fractional time, mean dwell time and transition number) of each state between whole MDD and HC groups (B), and between HI-MDD, LI-MDD and HC groups (C). * represents p < 0.05; HI-MDD, high insomnia major depressive disorder; LI-MDD, low insomnia major depressive disorder; HC, health control.

Group differences in functional network connectivity strength of dynamic state I and state II among HI-MDD, LI-MDD and HC groups (A). Three columns on the left show the ANOVA and post-hoc results of FNC comparison. The fourth column shows common FNC alterations found in both HI-MDD and LI-MDD groups. The right column shows distinct FNC alterations between HI-MDD and LI-MDD groups. Summary of distinct and common FNC alterations in HI-MDD and LI-MDD groups (B). Partial correlations between altered FNC and clinical and cognitive measures in HI-MDD and LI-MDD groups (C). * represents p < 0.05. Moderation effect of insomnia in relationship between DMN-VAN FC and logical memory in whole MDD group (D). FNC, functional netweork connectivity; DMN, default mode network; FPN, frontoparietal network; SMN, sensorimotor network; VIS, visual network; DAN, dorsal attention network; VAN, ventral attention network; SC, subcortical network; CeN, cerebellum network; dACC, dorsal anterior cingulate cortex.

FC strength in dynamic states
On analyzing the FNC strength for each dynamic state, we observed increased DMN-CeN connectivity and decreased DMN-DAN connectivity in state I in entire MDD group relative to HC. ANOVA results showed significant differences in the following FNC pairs (p < 0.05, FDR corrected) in state I across the three groups: (1) DMN-DAN/SMN/FPN/VIS/VAN/CeN, (2) DAN-SMN/FPN/VIS/CeN, and (3) SMN-VIS. Post hoc tests showed that HI-MDD exhibited significantly increased dynamic FNC in the DMN-lateral VAN (insula), DMN-VIS, DMN-CeN, and DAN-VIS network pairs, as well as decreased dynamic FNC in the DAN-FPN, DAN-CeN, DAN-DMN, DAN-VIS, and SMN-VIS network pairs compared to HC (Bonferroni corrected). LI-MDD showed significantly increased dynamic FNC in the DMN-FPN, DMN-lateral VAN (insula), DAN-SMN, and DAN-VIS network pairs, as well as decreased dynamic FNC in the DMN-SMN, DMN-DAN, and DAN-CeN network pairs compared to HC (Bonferroni corrected). HI-MDD exhibited significantly increased dynamic FNC in the DMN-VIS, DMN-SMN network pairs, and significantly decreased dynamic FNC in the SMN-DAN network pairs compared to LI-MDD (Bonferroni corrected).
Comparing to HC, the whole MDD group exhibited increase DMN-lateral VAN, SMN-SC, and within-CeN connectivity, but decrease DMN-SMN connectivity in state II. We also discovered significant differences in the following FNC pairs (p < 0.05, FDR corrected) in state II across the three groups: (1) DMN-SMN/FPN/lateral VAN, (2) SMN-SC, and within the CeN. And post hoc tests indicated that HI-MDD performed significantly increased dynamic FNC in the DMN-lateral VAN (insula), SMN-SC network pairs, and within CeN, as well as significantly decreased FNC in the DMN-SMN network pair compared to HC (Bonferroni corrected). LI-MDD exhibited significantly increased dynamic FNC in the DMN-lateral VAN (insula), SMN-SC, SMN-DAN network pairs, and within CeN, along with significantly decreased dynamic FNC in the DMN-SMN network pair compared to HC (Bonferroni corrected). In addition, HI-MDD exhibited significantly increased dynamic FNC in the DMN-medial VAN (dACC) network pair compared to LI-MDD (Bonferroni corrected). All dynamic FNC results can be seen in Table 3 and Figure 2a,b. Given the significant differences in depressive and anxiety symptoms severity between the HI-MDD and LI-MDD groups, we included depressive and anxiety symptom severity as covariates when examining the differences in dynamic FNC between these two groups. The between-group differences remained significant after this adjustment.
The results of dynamic FNC among HI-MDD, LI-MDD, and HC groups

Note: MDD, major depressive disorder; LI-MDD, Low insomnia MDD; HI-MDD, high insomnia MDD; HC, health control; FNC, functional network connectivity; IC, independent component, DMN, default mode network; FPN, frontoparietal network; SMN, sensorimotor network; VIS, visual network; DAN, dorsal attention network; VAN, ventral attention network; SC, subcortical network; CeN, cerebellum network; n, number.
Correlation analysis with clinical features and moderation analysis
Correlations between clinical symptom and cognitive functions
In the entire MDD group, controlling age and gender, insomnia severity was negatively correlated with Digit Span Forward (attention and processing speed), Digit Span Backward (executive function), and logical memory I and II (episodic memory), but positively correlated with SCWT (executive function) and TMT_B (processing speed). When controlling age, gender, and HAMD-adjusted score, insomnia was negatively correlated with logical memory I and II, visual reproduction I and II, Digit Span Forward, and Digit Span Backward in whole MDD group. Controlling age and gender, the HAMD-adjusted score showed positive correlation with TMT_A and TMT_B (processing speed), and SCWT (executive function). After controlling age, gender, and insomnia score, HAMD-adjusted score also showed a positive correlation with TMT_A and TMT_B. These relationships did not exist in either of the MDD subgroup. Based on the outlier removal criterion (mean ± 3 SD), we removed the outliers of the variates and reanalyzed the correlations, and it did not influence most of the findings. The correlation results can be acquired in Supplementary Figures 3–6.
Correlations between dynamic FNC and clinical symptom
There was no significant correlation between dynamic FNC and clinical symptom in the whole MDD and HI-MDD group. In LI-MDD, DMN-lateral VAN (insula) (r = 0.299, p = 0.029, pFDR = 0.131) and DMN-FPN connectivity in state I (r = 0.375, p = 0.006, pFDR = 0.054) were positively correlated with the HAMA score, as well as DAN-SMN connectivity in state I was positively correlated with the HAMD-adjusted score (r = 0.317, p = 0.021, pFDR = 0.189).
Correlations between dynamic FNC and cognitive functions
The DMN-CeN dynamic FNC in state I was negatively correlated with Logical Memory I (episodic memory, r = −0.192, p = 0.042, pFDR = 0.140) in the HI-MDD group. In the HI-MDD group, DMN-lateral VAN (insula) connectivity in state I was negatively correlated with Logical Memory I (r = −0.231, p = 0.014, pFDR = 0.110; r = −0.216, p = 0.022, pFDR = 0.110) and II (r = −0.217, p = 0.021, pFDR = 0.120; r = −0.213, p = 0.024, pFDR = 0.120), Visual reproduction II (r = −0.216, p = 0.022, pFDR = 0.110), Digit Span Forward (r = −0.235, p = 0.013, pFDR = 0.130) and Digit Span Backward (r = −0.236, p = 0.012, pFDR = 0.120), as well as positively correlated with TMT_A (r = 0.236, p = 0.012, pFDR = 0.120) and TMT_B (r = 0.207, p = 0.029, pFDR = 0.190; r = 0.197, p = 0.038, pFDR = 0.190), mainly involving episodic memory, processing speed, and executive function. And in the LI-MDD group, we discovered DMN-lateral VAN (insula) connectivity in state I was negatively correlated with Visual reproduction I (r = −0.300, p = 0.029, pFDR = 0.189) and II (r = −0.297, p = 0.031, pFDR = 0.279) (episodic memory), but positively correlated with TMT_B (r = 0.340, p = 0.013, pFDR = 0.117) (processing speed). And DMN-DAN connectivity was negatively correlated with TMT_B (r = −0.271, p = 0.049, pFDR = 0.221) (processing speed), as well as DMN-FPN connectivity was negatively correlated with Visual reproduction I (r = −0.281, p = 0.042, pFDR = 0.189) (episodic memory). The correlation results can be seen in Figure 2c and Supplementary Tables 2 and 3. In addition, the fractional time of state IV was negatively correlation with TMT_A score (processing speed) in the HI-MDD group, and results available in Supplementary Figure 7. In addition, the CCA did not yield significant canonical modes between FNC and cognitive domains in two subgroups (p > 0.05). The results of CCA between FNC and cognitive functions in HI-MDD and LI-MDD can be seen in Supplementary Figure 8.
Moderation analysis
Moderation analysis showed that, in state I, insomnia severity significantly moderated the association between DMN-lateral VAN (insula) connectivity and logical memory I/II scores in the MDD group (p = 0.0218 and p = 0.0485, uncorrected). The moderation result is shown in Figure 2d.
Validation analysis
Our validation analyses demonstrated that the between-group differences in dynamic properties remained robust when different window lengths or step sizes were applied. Moreover, the negative findings in static FNC persisted when the Yeo-17 network parcellation was used. Detailed results of the validation analyses are provided in Supplementary Tables 4 and 5 and Supplementary Figures 9–11.
Discussion
Our study investigated both the static and dynamic FNC characteristic alterations of adult MDD with different insomnia severity. While there is no significant group difference in static FNC, dynamic FNC analysis indicated insomnia-related specific cortical functional network alteration patterns. In dynamic analysis, HI-MDD patients spent more proportion of time in states representing overall weak network integration which was not observed in LI-MDD patients. Referring to network connectivity strength, both HI-MDD and LI-MDD shared common increased DMN-lateral VAN (insula) connectivity in states I and II, accompanied by decreased DAN-related connectivity with CeN and DMN in state I comparing to HCs. Moreover, HI-MDD exhibited more alterations as increased DMN-medial VAN (dACC) connectivity in state II, along with generally increased DMN-related connectivity with VIS and SMN in state I relative to LI-MDD. Our findings support that dynamic analysis may be more sensitive in capturing the brain FNC alterations relative to static method in drug naïve patients with MDD. In addition, we identified insomnia-related memory deficits and depression-related processing speed impairment in MDD. Furthermore, insomnia severity significantly moderated the association between DMN-lateral VAN (insula) and logical memory function in the MDD group.
Our findings support that dynamic analysis may be more sensitive in capturing the brain FNC alterations relative to static method in drug naïve patients with MDD. The insensitive of static connectivity may be interpretated as brain large-scale function still retain a certain degree of stability, whereas more pronounced change occur in recurrent patients with prolonged disease duration (Kendler, Thornton, & Gardner, Reference Kendler, Thornton and Gardner2001; Moylan, Maes, Wray, & Berk, Reference Moylan, Maes, Wray and Berk2013; Yan et al., Reference Yan, Chen, Li, Castellanos, Bai, Bo and Zang2019). In addition, previous studies have revealed that 8 weeks of antidepressant treatment can significantly reduce FNC among large-scale networks, when no significant FNC differences were detected between MDD patients and HC at baseline (Li et al., Reference Li, Su, Wu, Castellanos, Li, Li and Yan2021). Finally, from the perspective of analysis method, static FNC is derived by averaging functional coupling across the entire scanning period, under the assumption that brain connectivity remains temporally stable during data acquisition (Allen et al., Reference Allen, Damaraju, Plis, Erhardt, Eichele and Calhoun2014). As such, static connectivity reflects the overall, time-invariant organization of large-scale brain networks, which cannot capture transient and subtle abnormalities.
Common dynamic alterations in HI-MDD and LI-MDD
Dynamic FNC analysis revealed that both HI-MDD and LI-MDD showed increased DMN and lateral VAN (insula) relative to HC. And this DMN-lateral VAN FNC alteration appeared both in states I and II. VAN plays a crucial role in detecting and integrating emotional information (Bagherzadeh-Azbari et al., Reference Bagherzadeh-Azbari, Khazaie, Zarei, Spiegelhalder, Walter, Leerssen and Tahmasian2019; Bao et al., Reference Bao, Gao, Cao, Li, Liu, Liang and Huang2021), the DMN-lateral VAN (insula) connection is vital for emotion regulation. And previous study has reported the relationship between sleep and emotion regulation (Palmer & Alfano, Reference Palmer and Alfano2017). Our study finds stable DMN-lateral VAN (insula) FNC alteration may account for the disruption of sleep and emotion regulation process, which caused insomnia and impaired emotion regulation. Furthermore, the correlation between DMN-lateral VAN FNC and cognitive performance also indicated potential target region of DMN, lateral VAN for cognitive impairment in MDD with insomnia symptom. Interestingly, we also observed more cognitive functional items correlated with DMN-lateral VAN FNC in HI-MDD, which may suggest DMN and lateral VAN may serve as a key neural substrate linking insomnia severity to cognitive dysfunction in adult MDD.
The insula, as core hub of the VAN or the SN (Kucyi, Hodaie, & Davis, Reference Kucyi, Hodaie and Davis2012), plays a pivotal role in cognitive process (Menon & Uddin, Reference Menon and Uddin2010) by switch the function of DMN and the FPN. Specifically, during high-level cognitive demands, the insula generates control signals to suppress the activation of DMN while facilitating the activation of task-positive networks (like FPN), thereby supporting complex cognitive processing (Goulden et al., Reference Goulden, Khusnulina, Davis, Bracewell, Bokde, McNulty and Mullins2014; Sridharan, Levitin, & Menon, Reference Sridharan, Levitin and Menon2008). Furthermore, insomnia is closely associated with aberrant activity and FC in insula, accompanied by dysregulated salience processing (Chen, Chang, Glover, & Gotlib, Reference Chen, Chang, Glover and Gotlib2014). Therefore, our observation of hyperconnectivity between the insula and the DMN in high-insomnia MDD may reflect a diminished inhibitory control of the insula over the DMN which represent a disrupted switching process leading to the impairment of higher-order cognitive functions. It also explains why insomnia severity significantly moderates the association between DMN–insula connectivity and memory deficits in MDD in the current study. In summarize, insomnia may aggravate depression-related cognitive impairment by attenuating the insula’s capacity for network interaction as a gatekeeper.
We also observed both HI-MDD and LI-MDD showed brain network FNC alteration patterns, primarily characterized by decreased DMN/CeN-DAN FNC, as well as increased FNC within the CeN and DMN-SC in state I, and decreased DMN-SMN FNC in state II. The connectivity in DMN and DAN is responsible for maintaining effective cognitive processes (Yang et al., Reference Yang, Tian, He, Qiu, Feng, Chen and Lei2021), cerebellum is associated with the regulation of sleep–wake cycle and rhythm and also involved in emotional regulation process (Canto et al., Reference Canto, Onuki, Bruinsma, van der Werf and De Zeeuw2017; Xu et al., Reference Xu, Wang, Li, Qian, Wang, Wang and Niu2023). Our result revealed decreased dynamic FNC in DMN/CeN-DAN may lead to attention orientation abnormal, the development of insomnia and emotional regulation dysfunction (Yang et al., Reference Yang, Tian, He, Qiu, Feng, Chen and Lei2021), which manifested pay more attention to negative emotion, insomnia symptom in MDD patients with insomnia symptoms. In our study, the SC primarily includes the striatum, whose function is closely associated with the reward circuit (Wang, Wang, Liu, & Wang, Reference Wang, Wang, Liu and Wang2018). The DMN-SC dynamic FNC alteration in our study may be associated with the reward process disrupts MDD with insomnia symptoms. And DMN-SMN FNC alteration may contribute to sustained sensory processing of environmental stimuli, which may lead to insomnia and hyperarousal. Taken together, our study identified the stable DMN-lateral VAN connection abnormal in MDD patient, which supported the critical role of DMN and lateral VAN in neural mechanism of adult MDD with insomnia symptom.
Distinct dynamic alterations in HI-MDD and LI-MDD
Referring to dynamic properties, we found significant increased fractional time in weak-connection state, as well as decreased fractional time in strong-connection state, in the HI-MDD group compared to HC. But no significant difference was found in the LI-MDD group compared to HC. According to these findings, we speculated that MDD patients with high insomnia spent more time in a weak network connection state, which indicates insufficient functional interaction between brain network regions and may lead to impaired maintenance of brain function. Furthermore, serious insomnia symptom seems to prolong the time spent in this state in MDD. In other words, serious insomnia symptom in adult MDD may inhibit the normal functional interaction among brain network. This may also support the tight relationship between insomnia and depressive.
The observed temporal shift toward weak-connection state and away from strong-connection state in HI-MDD patients suggests a disruption in global network efficiency. This tendency entrapment in weak-connection state can be interpreted within the framework of the economy of brain network organization (Bullmore & Sporns, Reference Bullmore and Sporns2012). High-efficiency functional integration, as represented by strong-connection state requires substantial neural resources to maintain (Bullmore & Sporns, Reference Bullmore and Sporns2012). Chronic insomnia and sleep deprivation have been reported to deplete cognitive reserves and impair the brain’s capacity to sustain high-demand FC (Killgore, Reference Killgore2010; Zhou et al., Reference Zhou, Yu, Liu, Wang, Bao, Li and Zhou2025). Consequently, the increased fractional time in weak-connection state may reflect the impairment of neural flexibility, where the brain prefers to a low-energy, segregated configuration due to an inability to mobilize the resources required for the strong-connection state (Javaheripour et al., Reference Javaheripour, Colic, Opel, Li, Maleki Balajoo, Chand and Walter2023).
Nevertheless, dynamic FNC analysis indicated that HI-MDD and LI-MDD exhibit different brain network FNC alteration patterns. HI-MDD showed more extensive network connectivity alterations than LI-MDD, especially in state I. Specifically, the HI-MDD performed increased DMN-VIS/SMN FNC and decreased DAN-SMN FNC in state I, as well as increased DMN-medial VAN (dACC) FNC in state II compared to LI-MDD. DMN involved in regulating consciousness, self-referential activity, emotional processing (Li et al., Reference Li, Chen, Guan, Wang, Qian, Zhao and Li2018), VIS is essential for visual perception of visual stimuli (Zhu et al., Reference Zhu, Chen, Zheng, Zhao, Xia, Tang and Wang2025), and SMN involves in motor and sensory processing (Bao et al., Reference Bao, Gao, Cao, Li, Liu, Liang and Huang2021; Santarnecchi et al., Reference Santarnecchi, Del Bianco, Sicilia, Momi, Di Lorenzo, Ferrone and Rossi2018). The integration of the VIS and SMN is crucial for appropriate sensory response to visual stimuli. Abnormalities in the DMN-VIS/SMN interaction may be linked to overactivity in visual regions, leading to prolonged sensory processing of visual stimuli, which could disrupt sleep maintenance in MDD. The dorsal anterior cingulate cortex, a node of VAN, is associated with cognitive processing and emotional expression (Bao et al., Reference Bao, Gao, Cao, Li, Liu, Liang and Huang2021; Bush et al., Reference Bush, Vogt, Holmes, Dale, Greve, Jenike and Rosen2002). Abnormal DMN-dACC connection may contribute to the impairment of emotional expression in patients. Furthermore, previous study has reported that altered dACC activity contributes to heightened negative emotion in MDD with insomnia compared to those without insomnia (Dai et al., Reference Dai, Liu, Hu, Ren, Jin, Xu and Cao2024). This also supported that DMN-dACC connectivity alteration may underlie more severe insomnia symptoms and greater cognition impairment in HI-MDD. DAN mainly involved in attention orientation (Lei, Wang, Yuan, & Mantini, Reference Lei, Wang, Yuan and Mantini2014). Dysfunction in the DAN-SMN in HI-MDD may be associated with heightened attention to negative stimuli, making patients more sensitive to such stimuli, which could ultimately exacerbate insomnia symptoms in individuals with MDD. In general, we found that HI-MDD and LI-MDD exhibit distinct patterns of brain network connectivity, primarily involving the DMN, medial VAN (dACC), VIS, and SMN networks, which may serve as key nodes underlying differences in the severity of insomnia in MDD.
Relationship between cognition, insomnia, and FNC in adult depression
HI-MDD group demonstrated more severe cognitive dysfunction including episodic memory impairment, attention and executive dysfunction compared to LI-MDD, reflecting as lower score in the Logical Memory I/II and Visual Reproduction II, and Digit Span Backward and Digit Span forward comparing to the LI-MDD group. Consistent with a previous system review (Pearson et al., Reference Pearson, Uglik-Marucha, Miskowiak, Cairney, Rosenzweig, Young and Stokes2023), our findings also confirmed that more severe insomnia in MDD is associated with more pronounced cognitive impairment. Moreover, when controlling for depression or insomnia severity, we identified symptom specific cognitive impairment in MDD. Namely, episodic memory impairment was consistently associated with insomnia severity, while processing speed (TMT A/B) were linking to depressive severity. This aligned with prior evidence that sleep promotes memory consolidation (Cellini, Reference Cellini2017), whereas depressive symptoms contribute to processing speed (Kriesche et al., Reference Kriesche, Woll, Tschentscher, Engel and Karch2023).
More importantly, we further discovered that insomnia moderate the association between DMN-lateral VAN (insula) connectivity and logical memory. This means that DMN-lateral VAN connectivity associated with memory impairment mainly in the HI-MDD group with only mild or absent relationship in LI-MDD patients. These results suggested that insomnia amplifying network’s cognitive impact in MDD the treatment of insomnia may be an effective way to preserve cognitive function in MDD patients.
Limitation
There are also several limitations in present study. First, recruiting first-episode, medication-naïve MDD, our study minimized potential confounding influences of medication and episode frequency. However, this may limit the applicability of our results to individuals with recurrent depressive episodes or those undergoing therapy. Second, although our findings underscored the neural network patterns of MDD insomnia-related subtype, future research should adopt a more comprehensive assessment of sleep, including factors such as daytime fatigue, to better clarify the neural basis of sleep disturbances in MDD. Third, correlation results cannot survive the FDR correction and should be regarded as exploratory findings and interpreted with caution. The moderate effect sizes (r ≈ 0.3) of most results suggest potentially clinical meaningful associations that warrant further investigations in larger cohorts.
Conclusion
This study focused on alterations of dynamic FNC associated with different insomnia severity in MDD. In our study, static FNC analysis found no significant group difference revealed that dynamic can better capture the FNC alteration in MDD related to insomnia severity. Our main findings suggested that insomnia-related FNC alterations in MDD mainly involved in abnormal DMN-medial VAN (dACC) connection and DAN-related connectivity with CeN/DMN, whereas depression-related FNC alteration mainly involved in altered DMN-lateral VAN (insula) and DMN-related connectivity with CeN/DMN. We also identified insomnia-related episodic memory impairment and depression-related processing speed decline in MDD with insomnia. Furthermore, the moderation of insomnia on relationship between FNC alteration and cognitive impairment emphasized the necessity of treating insomnia to preserve cognitive function in MDD patients.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0033291726103900.
Data availability statement
Any additional information required to reanalyze the data reported in this article is available from the lead contact upon request.
Acknowledgments
This study is supported by grants from the National Natural Science Foundation of China (82372080) and Natural Science and technology Project of Tibet Autonomous Region (XZ202401ZR0081).
Author contribution
X.H. conceptualized and designed the study. W.B. drafted the initial manuscript and carried out the initial analyses. Y.G. and X.H. critically reviewed and revised the manuscript. Q.G. designed the data collection instruments. H.L., Z.Z., Y.W. and X.H. collected and preprocessed data.
Funding statement
This study is supported by grants from the National Natural Science Foundation of China (82372080) and Natural Science and technology Project of Tibet Autonomous Region (XZ202401ZR0081).
Competing interests
The authors have nothing to disclose.