Introduction
Restrictive cardiomyopathy (RCM) is an uncommon myocardial disease characterised by impaired ventricular filling with preserved systolic function and normal myocardial thickness. Reference Nihoyannopoulos and Dawson1 Its hallmark is myocardial stiffness causing elevated diastolic pressure. Reference Nihoyannopoulos and Dawson1,Reference Ditaranto, Caponetti and Ferrara2 Paediatric restrictive cardiomyopathy, the rarest cardiomyopathy, usually presents between ages 6 and 10 years and affects both sexes equally. Reference Zampieri, Di Filippo and Zocchi3 Aetiologies include idiopathic, infiltrative, fibrotic, oncological, metabolic, and iatrogenic causes. Reference Ditaranto, Caponetti and Ferrara2,Reference Bogle, Colan and Miyamoto4 Clinical findings range from asymptomatic to severe heart failure with pulmonary hypertension. Reference Ditaranto, Caponetti and Ferrara2 Progressive atrial enlargement leads to regurgitation, arrhythmias, and thromboembolism due to stasis. Reference Ditaranto, Caponetti and Ferrara2,Reference Chen, Williams, Chan and Mondal5 About 15% develop arrhythmias, worsening prognosis. Reference Chen, Williams, Chan and Mondal5 Over half die or need transplantation within 2 years; transplantation remains the definitive therapy. Reference Ditaranto, Caponetti and Ferrara2,Reference Bogle, Colan and Miyamoto4,Reference Mori, Kogaki and Ishida6 This case adds to the limited paediatric literature by illustrating recurrent multiorgan thromboembolism in restrictive cardiomyopathy despite maintained sinus rhythm. The presence of heterozygous prothrombotic genetic variants may represent an additional risk-modifying factor and highlights the complexity of thromboembolic risk stratification in this population.
Case report
The patient is a 14-year-old girl, the fifth child of a non-consanguineous family, born at term with normal development and no relevant family history. At the age of 5 years, she was diagnosed with restrictive cardiomyopathy after evaluation for chest pain.
At the age of 11 years, she presented with left hemiparesis and facial palsy. CT angiography showed thrombotic occlusion of the right middle cerebral artery (Figure 1(a), (b)), and MRI confirmed acute infarction in the right middle cerebral artery territory with additional ischaemic areas (Figure 1(c), (d )). She underwent endovascular thrombectomy and started low-molecular-weight heparin.
Coronal CT angiography demonstrating right Middle Cerebral Artery (MCA) M1 segment occlusion (July 2021) ( a ). Axial CT angiography demonstrating right MCA M1 segment occlusion (July 2021) ( b ). Axial diffusion-weighted MRI demonstrating restricted diffusion in the right MCA territory, consistent with acute ischemic infarction (July 2021) ( c ). Axial Apparent Diffusion Coefficient (ADC) map showing signal reduction in the right MCA territory, consistent with acute infarction (July 2021) ( d ). Axial diffusion-weighted MRI showing restricted diffusion in the left MCA territory, consistent with acute ischemic infarction (November 2023) ( e ). Axial ADC map showing signal reduction in the left MCA territory, confirming acute ischemic infarction (November 2023) ( f ). Coronal CT angiography demonstrating occlusion of the left common and external iliac arteries (November 2023) ( g ). Axial contrast-enhanced CT image demonstrating a filling defect within the right atrium, consistent with right atrial thrombus (November 2023) ( h ).

At the age of 13 years, she was hospitalised for cardiac decompensation and experienced a focal seizure. MRI revealed new ischaemic lesions in the left middle cerebral artery territory (Figure 1( e ), ( f )). After stabilisation, she developed pulseless ventricular tachycardia and received an Implantable Cardioverter Defibrillator (ICD). Subsequently, left lower limb ischaemia occurred due to iliac and popliteal occlusion (Figure 1( g )); thoracic CT showed a right atrial thrombus (Figure 1( h )). Embolectomy was performed.
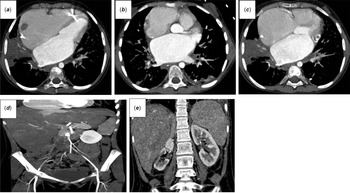
At the age of 14 years, she presented with bilateral leg pain and absent pulses. CT angiography showed thrombi in both atrial appendages (Figure 2( a – c )) and a large thrombus extending from the aortic bifurcation into the iliac arteries (Figure 2( d )), with renal infarctions (Figure 2( e )). Echocardiography revealed biatrial dilatation, reduced ejection fraction, valvular regurgitation, and intracardiac thrombi. Tissue plasminogen activator infusion was given, followed by aspirin and low-molecular-weight heparin.
Axial contrast-enhanced CT images demonstrating biatrial dilatation with thrombi in the right atrium and left atrial appendage (December 2024) ( a – c ). Contrast-enhanced CT showing an occlusive thrombus extending from the infrarenal abdominal aorta to the aortic bifurcation and right common iliac artery (December 2024) ( d ). CT image showing right renal atrophy and parenchymal loss consistent with sequelae of prior ischemic insult (December 2024) ( e ).

Genetic testing revealed heterozygous β-fibrinogen 455 G/A and MTHFR A1298C variants. Evaluation for vasculitis was negative. Warfarin therapy led to resolution of intracardiac thrombi and improved perfusion. She remains on the heart transplant waiting list with residual left-sided weakness and dystonia.
Discussion
Restrictive cardiomyopathy in the paediatric population is characterised not only by progressive diastolic dysfunction but also by a high burden of complications, notably thromboembolic events. The overall prognosis remains poor, with a 5-year cumulative incidence of heart transplantation reaching 58% in those with a pure restrictive phenotype, reflecting the aggressive natural course of the disease. Reference Zampieri, Di Filippo and Zocchi3 Thromboembolism, in particular, significantly contributes to morbidity and mortality and may be the initial or sole presenting feature in some cases. Reference Al-Shammari, Muslim and Almuslim7
Multiple reports underscore the elevated thromboembolic risk in children, irrespective of underlying rhythm abnormalities. A wide range of thrombus incidence has been reported, from 0% to 42%, with embolism rates between 12% and 33%, highlighting the need for early and routine surveillance strategies. Reference Ditaranto, Caponetti and Ferrara2 The mechanisms predisposing to thrombus formation are multifactorial, including marked biatrial enlargement, low intracavitary flow velocities, and structural distortion of atrial anatomy. While atrial fibrillation has long been recognised as a prothrombotic substrate, paediatric cases demonstrate that even in sinus rhythm, significant thrombotic complications may arise. Reference Al-Shammari, Muslim and Almuslim7–Reference Yamamoto, Itamoto and Hayashi9
Case reports further illuminate the severity of this phenomenon. Turhan et al. described a child who developed a free-floating left atrial ball thrombus during sinus rhythm, leading to the recognition that atrial contractile dysfunction alone may suffice to generate life-threatening emboli. Reference Turhan, Ocal, Erbay, Yasar, Cicekcioglu and Yetkin8 Similarly, Yamamoto et al. reported recurrent pulmonary thromboembolism originating from the right atrial appendage in a patient without atrial fibrillation, underscoring that even in the absence of conventional arrhythmic risk factors, restrictive physiology itself predisposes to thrombus formation. Reference Yamamoto, Itamoto and Hayashi9 Al-Shammari et al. presented a dramatic case of acute bilateral lower limb ischaemia due to complete aortic and iliac artery occlusion by emboli from atrial thrombi in a child with idiopathic RCM, further illustrating the catastrophic potential of this complication. Reference Al-Shammari, Muslim and Almuslim7
Although most literature on atrial thrombosis in RCM stems from adult cohorts, emerging paediatric data support the proactive use of antithrombotic therapy. Given the absence of reliable predictors for thromboembolic events and the unpredictability of their timing, current expert opinion favours the early initiation of anticoagulation upon diagnosis, even in asymptomatic children or those without arrhythmia. Reference Ditaranto, Caponetti and Ferrara2,Reference Chen, Williams, Chan and Mondal5 This approach is bolstered by genetic studies reporting fatal thrombotic events in family members carrying pathogenic sarcomeric gene mutations, suggesting that certain genetic backgrounds may confer an intrinsic thrombotic predisposition. Reference Denfield and Webber10
Our case highlights the coexistence of significant thromboembolic complications and malignant arrhythmias in paediatric restrictive cardiomyopathy. The patient developed recurrent, multisystem embolic events, including ischaemic stroke, bilateral lower limb ischaemia, renal infarctions, despite being in sinus rhythm for most of the clinical course. Serial imaging revealed intracardiac thrombi, emphasising the contribution of atrial dilation and impaired flow dynamics to thrombus formation in the absence of atrial fibrillation. Also genetic testing identified heterozygous variants in β-fibrinogen 455 G/A and MTHFR A1298C, suggesting a prothrombotic predisposition. The β-fibrinogen 455 G/A polymorphism has been shown to be associated with left atrial thrombus formation and severe spontaneous echo contrast in patients with atrial fibrillation, suggesting a role as a genetic risk modifier for thromboembolism. Reference Bozdemir, Kirimli and Akdeniz11 In addition, meta-analytic data indicate that the β-fibrinogen 455 G/A polymorphism is associated with an increased risk of ischaemic stroke, supporting its contribution to thromboembolic susceptibility in genetically predisposed individuals. Reference Luo, Li and Jiang12 Parallel to these findings, studies in non-valvular atrial fibrillation-related cardioembolic stroke cohorts have demonstrated that MTHFR polymorphisms, especially the A1298C variant, are associated with greater stroke severity and recurrence. Reference Chita, Tudor and Christodorescu13 Moreover, MTHFR variants, including A1298C, have been linked to an increased risk of venous thromboembolism independent of plasma homocysteine levels, pointing toward a genotype-driven thrombotic risk rather than a purely metabolic mechanism. Reference Liu, Silva, Malone and Seetharaman14 In our patient, the coexistence of MTHFR A1298C and β-fibrinogen 455 G/A variants may have acted as synergistic genetic risk modifiers, contributing to the pronounced thromboembolic phenotype observed despite the absence of atrial arrhythmias.
Arrhythmias, although not always the primary trigger, remain closely linked to thromboembolism. Approximately 15% of paediatric patients develop clinically significant rhythm disturbances, Reference Denfield and Webber10 while some studies report arrhythmia rates as high as 60%, including atrial fibrillation, Atrioventricular blocks, supraventricular tachycardias, and ventricular tachyarrhythmias. Reference Wittekind, Ryan and Gao15 Continuous rhythm monitoring is thus essential for risk stratification and management. Although Wittekind et al. found no direct association between arrhythmias and outcomes, likely due to preemptive ICD or pacemaker implantation, rhythm disturbances may exacerbate intracardiac turbulence and increase the risk of thrombus embolisation. Reference Wittekind, Ryan and Gao15 Moreover, Walsh et al. emphasised the association between conduction abnormalities and sudden cardiac events, reinforcing the need for prophylactic pacing in selected cases.Reference Walsh, Grenier, Jefferies, Towbin, Lorts and Czosek 16
In our patient, pulseless ventricular tachycardia occurred and required ICD placement, a life-threatening arrhythmic complication that has also been described in previous paediatric cases. This observation highlights the malignant arrhythmic potential of restrictive cardiomyopathy in children and supports the need for early rhythm surveillance and timely consideration of device therapy, even in the absence of preceding atrial arrhythmias.
Our case differs from previously reported paediatric restrictive cardiomyopathy cases in several important aspects. Unlike many reports in which thromboembolic events were isolated, preceded by atrial arrhythmias, or limited to a single vascular territory, our patient experienced recurrent, multisystemic thromboembolism involving the cerebral, peripheral, renal, and intracardiac circulations despite sustained sinus rhythm for most of the disease course. Furthermore, the thromboembolic burden progressed over time and persisted despite initial anticoagulation, ultimately coinciding with malignant ventricular arrhythmia requiring ICD implantation. In addition to advanced atrial remodelling and restrictive physiology, the identification of concomitant heterozygous prothrombotic genetic variants (β-fibrinogen 455 G/A and MTHFR A1298C) distinguishes this case and suggests a cumulative genetic contribution to thromboembolic susceptibility.
Conclusion
In conclusion, thromboembolism is a central and potentially fatal complication of paediatric restrictive cardiomyopathy. Its occurrence is not limited to those with arrhythmias and may manifest early in the disease course. Given the unpredictability and high clinical burden of these events, early initiation of anticoagulation, close rhythm surveillance, and timely consideration of heart transplantation remain cornerstones of management. Future research should aim to elucidate predictive markers and tailor prevention strategies to individual patient profiles.
Financial support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Competing interests
No potential conflicts of interest relevant to this article were reported.
Consent statement
Written informed consent was obtained from the patient’s legal guardians for the publication of this case report and any accompanying images.