Introduction
Candidozyma auris (C. auris) has emerged worldwide as a major healthcare-associated pathogen. C. auris causes serious invasive infections, rapidly contaminates the healthcare environment, and commonly demonstrates resistance to antifungal medications.1–Reference Zhu, Liu and Boekhout5 C. auris has been implicated in outbreaks at many types of healthcare facilities, including short-term and long-term acute care hospitals (STACHS and LTACHs, respectively) and lower acuity skilled nursing facilities (SNFs).Reference Saris, Meis and Voss6–Reference Harris, Pineles and Johnson12
Within the United States, healthcare facilities such as acute care hospitals frequently rely on guidance from the U.S. Centers for Disease Control and Prevention (CDC) to inform their approach and prevent spread of healthcare associated pathogens.13 Current published CDC guidance for C. auris requires some interpretation by each facility based on the local C. auris epidemiology. Implementation of this guidance may vary between hospitals, potentially indicating a lack of knowledge or an inability to implement this guidance for financial or structural reasons. These limitations and knowledge gaps may create variability in infection prevention practices and limit containment.
The Society for Healthcare Epidemiology (SHEA) Research Network (SRN) is a group of healthcare facilities that collaborates to perform research on healthcare epidemiology and antimicrobial stewardship within the United States. We surveyed U.S.-based SRN acute care hospitals to better understand their approach to C. auris prevention practices, delineate perceived barriers to control, and to identify opportunities to improve containment.
Methods
Our team developed a survey to assess facility characteristics, prevention practices, and barriers to C. auris prevention among acute care hospitals in the United States that participate in the SRN. This study was reviewed and approved by the Rush Institutional Review Board and by the SRN prior to email distribution using Research Electronic Data Capture (REDCap).Reference Harris, Taylor, Thielke, Payne, Gonzalez and Conde14 The survey was distributed in 2024; result analysis was performed in 2025. To maximize response potential, surveys were sent out on a rolling basis with initial invitations extended, followed by three reminder emails. The survey consisted of 21 questions, including single or multiple-choice, ranked, and free text questions (Supplemental Methods). The total number of questions answered by each facility varied, with branching logic utilized to further clarify responses. Completion of all survey questions was not required for form submission, and facilities had the option to leave questions blank. Percentages were calculated against the denominator of the total pool of survey participants (n = 51) unless otherwise noted. This was done as total responses to questions varied and not all participants had the opportunity to view every question due to the nature of the branching logic employed. For ranked-choice questions, responses were analyzed as non-weighted counts. Responses were analyzed using descriptive statistics by SAS version 9.4 (Statistical Analysis System).15 When appropriate, qualitative responses were analyzed using an iterative process. Study team members (HL, SS) reviewed each free text response and qualitatively coded it by consensus.Reference Pope and Mays16
Results
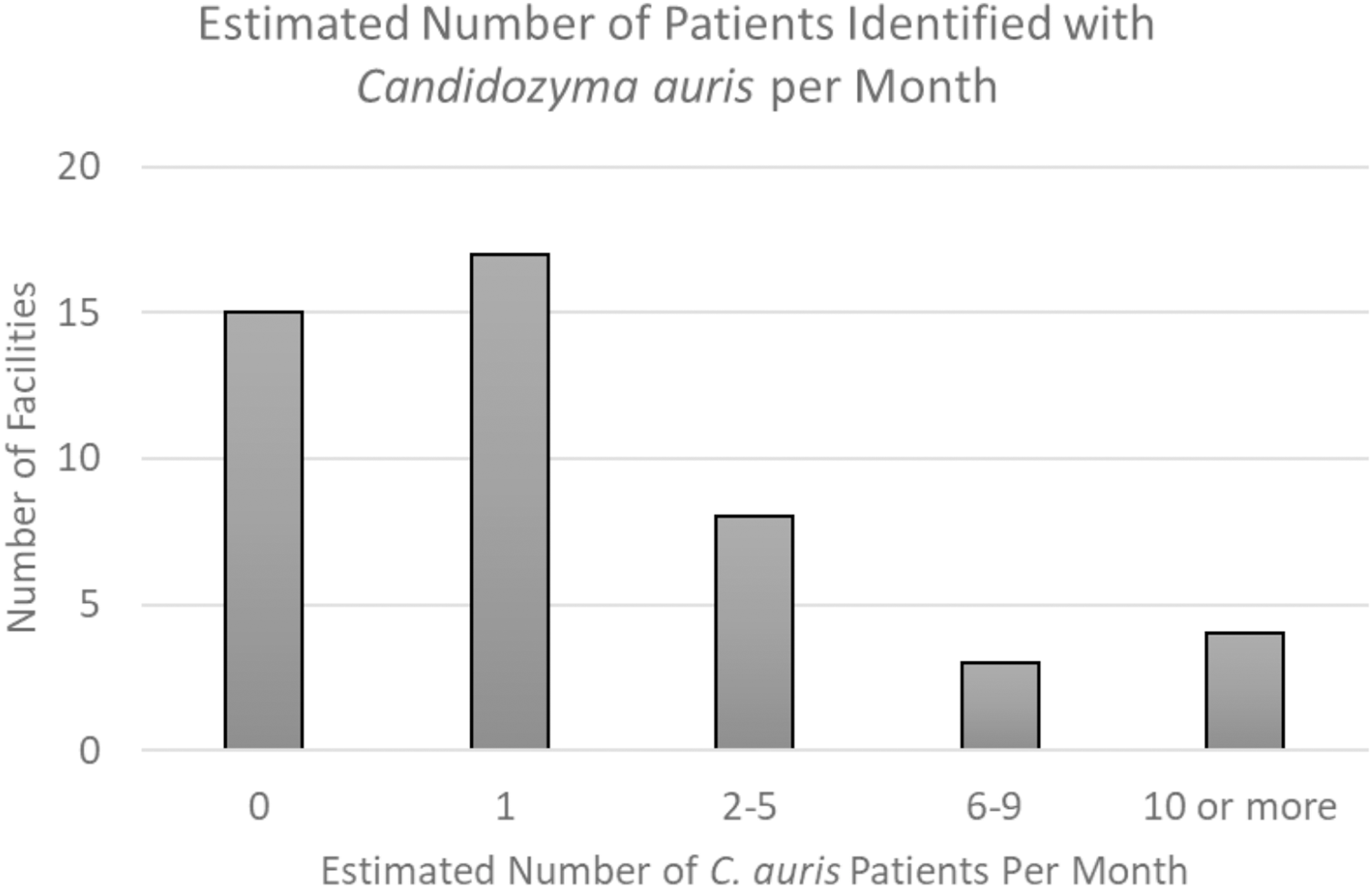
Among 96 acute care U.S.-based hospitals in the SRN, 51 responded (53% response rate) (Table 1). Thirty-four (67%) reported first-hand experience with C. auris, defined as having identified a patient infected or colonized with C. auris within their facility; half of these thirty-four facilities averaged at least one such patient per month (17/34, 50%) (Figure 1). Fifteen hospitals also reported experience with at least one C. auris outbreak, defined as suspected transmission of C. auris within their facility. Ten percent of hospitals (5/51) reported identification of a C. auris isolate with resistance to ≥3 antifungal medication classes (Figure 2).
Estimated number of patients identified with Candidozyma auris per month, by facility.

Locations of participating SHEA Research Network facilities and reported C. auris resistance. The number of participating acute care hospitals within each geographic region are shown in blue circles. Geographic regions include northeast, southeast, midwest, northwest, and southwestern United States. Pink diamonds indicate regions with multidrug-resistant C. auris (resistance ≥3 antifungal classes) reported by survey participants.

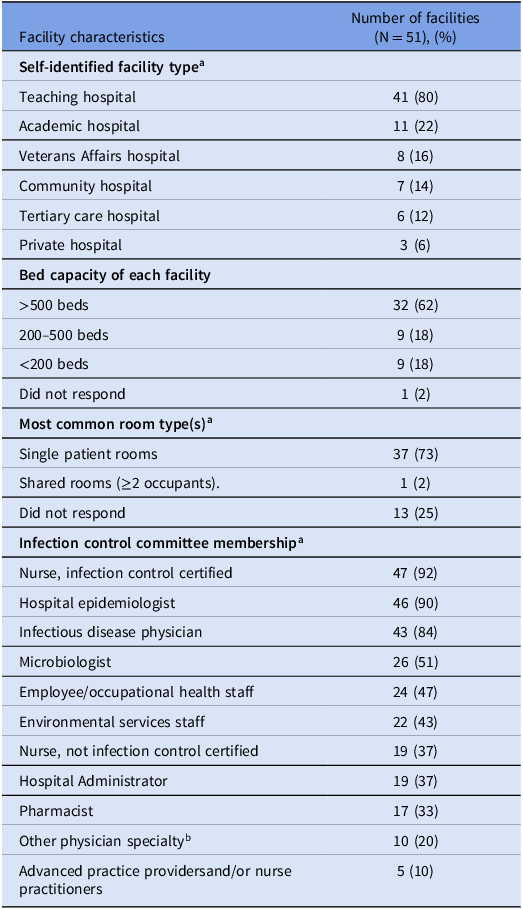
Characteristics of participating SRN facilities

Note. aCategories not mutually exclusive.
b Other physician specialties included by free text: critical care (5), general surgery or surgical subspecialties (4), hospitalist (3), pediatrics (2), anesthesiology (2), cardiology (1), oncology (1).
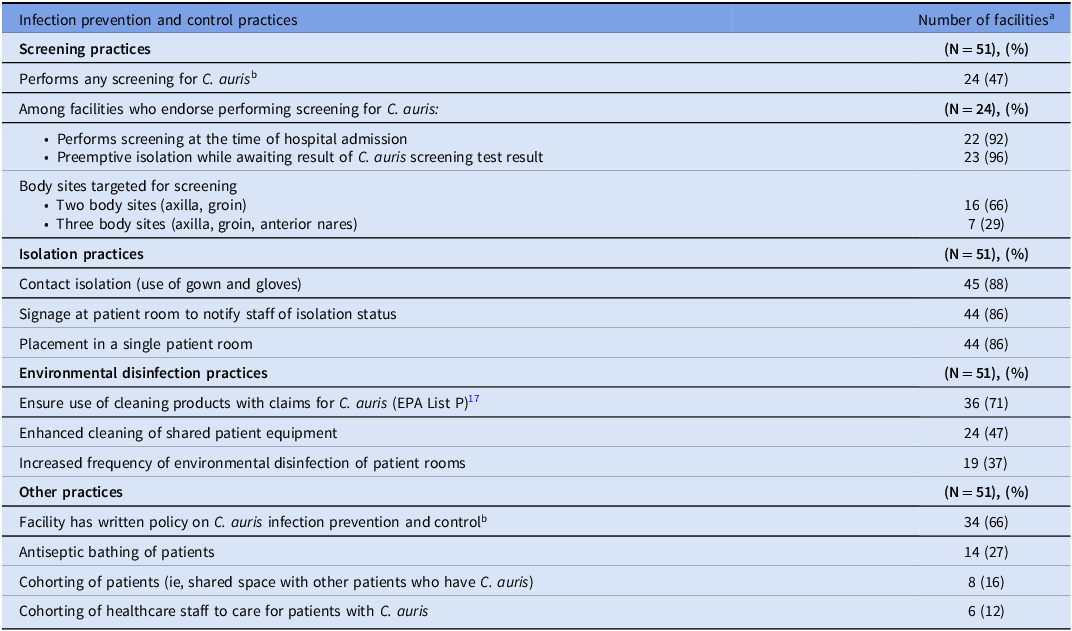
C. auris screening was performed in 24 (47%) acute care hospitals, typically at the time of admission (22/24, 92%). Facilities were asked to describe only their own screening practices, excluding ongoing public health screening. Sixteen facilities screened by two body sites (axillae and groin), while seven screened by three body sites (axillae, groin, and anterior nares). Polymerase chain reaction was the most used testing method (17/24, 71%). Acute care hospitals using targeted C. auris screening strategies (14/24, 58%) were asked to share screening criteria in a free text response; approaches included screening patients who were exposed to other healthcare facilities (eg, SNF, LTACH) (n = 12), history of a multidrug-resistant organism (n = 7), healthcare exposure outside of the U.S. (n = 6), presence of a tracheostomy or chronic ventilation (n = 5), admission from a facility with a known C. auris outbreak (n = 5), or presence of a chronic wound (n = 1).
Thirty-four acute care hospitals (67%) reported having a written policy regarding C. auris prevention; four respondents declined to answer this question. Isolation was commonly reported as a prevention practice for patients with C. auris, including contact isolation (ie, use of gloves and gowns) (88%), placement into a single patient room (86%), use of signage to notify staff of isolation status (86%), and preemptive isolation while awaiting screening results (45%). Most hospitals reported the use of cleaning products on the Environmental Protection Agency (EPA) list P for C. auris disinfection (71%).17 Targeted prevention practices related to environmental cleaning were reported in less than half of participants (Table 2).
Infection prevention and control practices for Candidozyma auris

Note. aRespondents were not required to complete every question, and percentages are calculated against total pool of respondents (N = 51) unless otherwise specified.
b Four respondents declined to answer this question.
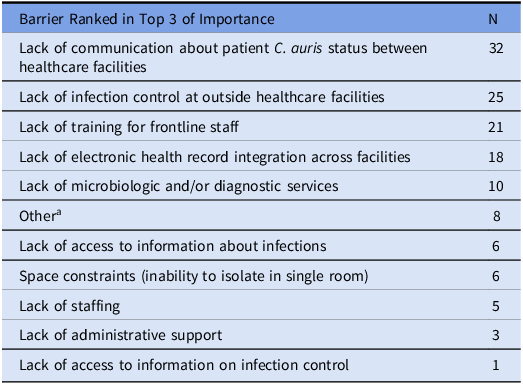
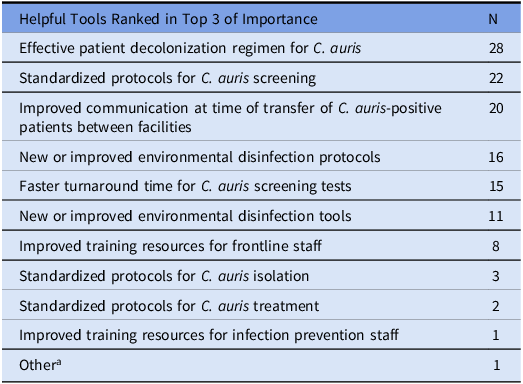
Barriers to control of C. auris were queried as a ranked choice question; the three most commonly selected barriers included lack of communication of C. auris colonization or infection status at the time of patient transfer between healthcare facilities (n = 32), lack of infection control at outside healthcare facilities (n = 25), and lack of training for frontline staff (n = 21) (Table 3). Participants were also asked to rank which resources or tools would best support C. auris prevention in their hospital; the three most commonly selected tools were C. auris decolonization regimens (n = 28), standardized protocols for C. auris screening (n = 22), and improved communication between healthcare facilities (n = 20) (Table 4).
In your opinion, what are the most important barriers that should be addressed to prevent the spread of C. auris in your facility?

Note. aFree text responses included public health guidance or risk stratification tools to select patients for screening (n = 4); communication with public health departments (e, lack of data sharing, delay in obtaining genomic data) (n = 3); non-adherence with infection control measures (n = 2); lack of availability of microbiologic resources to support screening (n = 1).
In your opinion, development of which of the following tools would be most helpful to support your ability to respond to C. auris in your facility?

Note. aFree text response: coordinated regional surveillance program.
Discussion
Candidozyma auris continues to spread across U.S. healthcare facilities despite containment efforts.Reference Lyman, Forsberg and Sexton7 We surveyed C. auris prevention practices in U.S.-based acute care SRN hospitals to better understand barriers to control and identify opportunities to improve prevention strategies. Two-thirds of participating hospitals reported having provided care to a patient with C. auris colonization or infection, with half of those also experiencing at least one C. auris outbreak. Prevention strategies commonly included contact isolation, with approximately half of hospitals surveyed also performing C. auris screening. Barriers to effective C. auris prevention focused on challenges when accepting patients from other healthcare facilities, including poor communication regarding C. auris status and perceived suboptimal infection control practices at the originating healthcare facility. The most helpful tools to improve C. auris containment included the development of effective decolonization regimens and standardized screening strategies.
Reported C. auris prevention practices varied among participating facilities, and there was no single element that was universally applied across SRN acute care hospitals. There was strong, but not universal, alignment across facilities regarding use of contact isolation, placement into single occupant rooms, and use of signage to notify staff of isolation status. Future prevention efforts should focus on aligning these critical prevention practices, as C. auris is known to rapidly contaminate the healthcare environment and may serve as an important intermediary of transmission.Reference Sansom, Gussin and Schoeny18 In contrast to other prevention practices, the use of targeted environmental disinfection was less consistent, although over 70% of participants did report ensuring the use of EPA list P products for environmental disinfection.17 Survey questions were not tailored to explore the reasoning behind facilities’ use of disinfectants outside of EPA list P products, and this represents an area for future research. Our current findings suggest that additional education and training in environmental disinfection is an area of opportunity to prevent C. auris spread, potentially through multimodal training approaches (ie, in-person teaching by experts as well as self-guided modules).Reference Sansom, King and Lucky19
Our study adds to the growing body of literature regarding screening practices for C. auris. In a survey of 253 Emerging Infection Network facilities, 36% reported screening patients for C. auris.Reference Hennessee, Forsberg and Beekmann20 Another survey of French mycologists from facilities in France (n = 36) found that 31% were aware of C. auris screening being conducted in their facility.Reference Guitard, Bellanger and Dorin21 In the current survey, nearly 50% of participants reported screening for C. auris, which may reflect growing awareness of C. auris or the greater resources available to teaching hospitals,Reference Valencia, Arora, Ranji, Meza and Moriates22 who comprised most survey participants. Barriers to screening for C. auris in this study aligned with prior work,Reference Hennessee, Forsberg and Beekmann20,Reference Guitard, Bellanger and Dorin21 including limited microbiologic laboratory capabilities. Participants noted the development of standardized screening protocols for C. auris as the second most commonly reported tool to help improve C. auris containment. Expanded engagement of local public health departments to provide guidance and microbiologic support for screening may help contain C. auris spread. Additionally, development of strong regional collaborations between healthcare facilities to contain C. auris may be needed, which would facilitate alignment of screening protocols and communication of screening results.
Effective communication of patient C. auris status between healthcare facilities at the time of patient transfer was identified as an important barrier across multiple questions in our survey. Interfacility transfer communication is a major challenge due to gaps, inconsistencies and heterogeneity in practice.Reference King, Gilmore-Bykovskyi, Roiland, Polnaszek, Bowers and Kind23–Reference Ellingson, Noble and Buser25 Despite its importance, communication of C. auris status at the time of interfacility transfer has not been standardized and most jurisdictions in the U.S. do not have such interfacility communication requirements.Reference Ellingson, Noble and Buser25 Among states mandating interfacility communication of multidrug-resistant organism status, compliance is heterogeneous, with <50% using standardized communication processes.Reference Ellingson, Noble and Tran26 Some strategies to overcome this barrier have been developed, such as statewide web-based registries (eg, Illinois XDRO registryReference Trick, Lin and Cheng-Leidig27) for data exchange, but additional work is needed. Standardization and automation of clinical information sharing may improve the quality and safety of patient transitions, while simultaneously preventing C. auris spread at receiving facilities due to rapid initiation of prevention measures.
This study has several limitations. The survey was distributed to U.S.-based acute care hospitals, which may limit generalizability to global health systems. The SRN, as a consortium of hospitals with explicit interest in infection control practices, may not represent all acute care facilities in the U.S. and thus generalizability may be limited. Most participants self-identified as teaching hospitals; this may also limit generalizability because teaching hospitals may have more resources than community hospitals to implement prevention programs.Reference Valencia, Arora, Ranji, Meza and Moriates22 As such, our findings may overestimate the true prevalence of some practices, such as C. auris screening. The survey instructed participating facilities to describe their own screening practices, but not those performed by the public health department. Facilities that were reliant on public health for surveillance would not have reported this practice, and this should be taken into consideration when interpreting results. Inherent limitations of survey-based research should also be considered, such as non-response, recall and social desirability biases.Reference Vetter and Mascha28,Reference Vesely and Klöckner29 The influence of non-response bias is anticipated to be low within this report, as the majority of respondents completed all questions: four declined to answer detailed questions about screening for C. auris, but all respondents completed the sections detailing infection prevention practices. Strengths of this study include alignment of reported data with the previously described epidemiology of C. auris; for example, reports of antifungal-resistant strains are present in previously described geographic regions of the Northeastern U.S.1–Reference Zhu, Liu and Boekhout5 This study reflects the experience of the SRN facilities at the time of survey completion in 2024, and Figure 2 should not be interpreted with the implication that resistant isolates of C. auris are limited to regions marked within the figure.
In this survey of U.S.-based acute care SRN hospitals, we identified multiple barriers and potential solutions to improve C. auris infection prevention and control. Increased education and support for effective environmental disinfection and interfacility communication are potential targets for future interventions. Additionally, development of effective C. auris decolonization regimens and more discrete guidance for surveillance programs should be prioritized.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/ice.2026.10396
Acknowledgements
The authors thank the SHEA Research Network participants and staff, as well as the participants of this study. We are grateful to Maroya Walters, Meghan Lyman, Karen Coffey, and Erica Shenoy for their critical review of the survey questions.
Financial support
This work was supported by the SHEA Research Scholar Award and by the cooperative agreement U54CK000607 from the Centers for Disease Control and Prevention (CDC). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of SHEA or CDC.
Competing interests
All authors report no conflicts of interest relevant to this article.