Introduction
The incidence of rectal cancer is increasing. Reference Barot, Liljegren, Nordenvall, Blom and Radkiewicz1 Treatment of locally advanced rectal cancer often involves neoadjuvant radiotherapy (RT). In cases of complete clinical response, some patients may not require surgery and can instead be closely monitored in a watch-and-wait program. Reference Rydbeck, Azhar and Blomqvist2
The planning of rectal cancer irradiation involves the manual contouring (MC) of organs at risk (OARs), the gross tumour volume (GTV) and the clinical target volume (CTV). A time-consuming aspect of the CTV delineation is the contouring of the lymphatic nodal levels (LN). Contouring is often performed according to annotation guidelines, such as RTOG Reference Myerson, Garofalo and El Naqa3 or the UK Royal College of Radiologists’ (RCR) guidance. 4 Delineation is a labour-intensive process that requires quality control to prevent excessive incidental irradiation and underdosed volumes. MC is also associated with inter-observer variation, Reference Segedin and Petric5 which is sometimes compensated for by expanding the final treatment volume. Reference van Herk6 To improve the efficiency of this process, the use of image-processing artificial intelligence (AI), auto-segmentation, has become a rapidly emerging tool in RT planning.
Currently, there are numerous available auto-segmentation tools on the market for clinical use, especially for OARs segmentation. Reference Doolan, Charalambous and Roussakis7 Benefits include shorter lead times to treatment, more efficient use of limited professional resources and reduced inter-observer variation. Reference Baroudi, Brock and Cao8 These clinical advantages are all dependent on the quality of the delineations. Reference Doolan, Charalambous and Roussakis7 Poorly auto-segmented structures could require substantial manual correction Reference Doolan, Charalambous and Roussakis7 or may result in the inadvertent acceptance of suboptimal segmentations. Reference Baroudi, Brock and Cao8 As the field of auto-contouring continues to evolve, ongoing in-house quality control and assurance are essential to achieve superior therapeutic outcomes.
The methods used to evaluate the clinical benefit of auto-segmentation differ. Reference Baroudi, Brock and Cao8 While time measurements could be considered representative for clinical benefit, they are themselves time-consuming. Consequently, evaluations are often conducted using various surrogate measures, frequently including quantitative geometric metrics or qualitative assessments of the segmentations. Reference Baroudi, Brock and Cao8 At present, there is no consensus on how these surrogate measures should be evaluated. Reference Baroudi, Brock and Cao8
While the clinical benefits of auto-segmentation of OARs have been studied and reported in earlier publications, the use of auto-segmented CTVs for specific tumours has not been explored to the same extent. A potential advantage of auto-segmenting the CTVs in rectal cancer RT planning has been suggested in only a few published works, Reference Geng, Zhu and Liu9–Reference Wu, Kang and Han14 although these employed varying evaluation methods and yielded different results. In 2024, our department had the opportunity to evaluate two deep learning-based software tools simultaneously in RT planning. The clinical application of these tools in our practice was limited to a few OARs.
In our study, in the setting of RT planning for the treatment of locally advanced rectal cancer, we aimed to investigate the potential use of a constructed CTV structure for rectal tumours by merging some of the auto-generated structures (AI-CTV) and estimate the clinical usefulness of these structures by comparing them to the manually contoured CTVs (MC-CTV) using a combination of quantitative and qualitative methods. We also quantitatively evaluated the auto-segmented OARs in comparison with their manually contoured counterparts.
Methods
Patients: Planning computed tomography images and contour data from ten consecutive rectal cancer patients who underwent preoperative radiotherapy at our department in 2024 were retrospectively collected and anonymised for the performed analyses. The indication for treatment was stage II-III, with no differing treatment protocol between the groups.
CT scanner and software: All patients underwent contrast-enhanced treatment-planning CT using a Siemens Somatom Definition Edge scanner (Siemens Healthineers AG, Forchheim, Germany), following our standard protocol with patient-size-adapted dose parameters and a typical slice thickness of 2 mm.
We used the treatment planning software Monaco 6.1.2.0 (Elekta, Stockholm, Sweden) for the MC; Hero 2024.2.0 (Hero Imaging, Umeå, Sweden) for quantitative data acquirement and SPSS v 29.0.2.0 (IBM, Armonk, USA) for statistical analysis. The auto-contouring software tools used in the study were AI-Rad Companion Organs RT VA50 (Siemens Healthineers AG, Forchheim, Germany) and Contour+ v1.2.7 (MVision AI Oy, Helsinki, Finland), henceforth referred to as AI-Rad and Contour+, respectively. Both these AI tools are trained on RTOG-annotated datasets for their rectal cancer structures. During the preparation of the article, we used an AI proofreading tool from Wordwice AI (Wordwice, Des Moines, USA) for language improvement. After using this tool, we reviewed and edited the content as needed.
Ethical approval was granted for the conducted retrospective analyses by the Swedish Ethical Review Authority (Etikprövningsmyndigheten), diary number 2025-04703-01.
Statistical analysis: Due to the low sample size, with presumed non-distribution, we used non-parametric tests, the Wilcoxon signed-rank test, for the difference between quantitative results of the different AI tools as well as the difference in qualitative scores between AI and MC. We used Kendall’s rank correlation coefficient for correlation comparisons.
CTV segmentation: The manual contours, used in the clinical setting, served as ground truth in the comparisons. These contours were delineated and peer reviewed according to local protocol, which followed the RCR guidance, 4 in which the CTV is defined as a combination of relevant nodal volumes, such as the anterior obturator nodes and the clinical target volume of the tumour (CTVT), in turn, defined as the GTV structure with added margins of 2 cm cranially and caudally and 1.5 cm in all other directions.
Comparable AI-CTV and AI-GTV segments were not directly provided by the auto-segmentation tools. Therefore, a CTV was constructed using Contour+ structures in a stepwise manner (Figure 1): the structures labelled ‘rectum’ and ‘recto-sigmoideum’ were fused into a single structure, and slices cranial and caudal to the tumour borders were removed, creating the AI-GTV (Figure 1a–b). This AI-GTV was then expanded by adding margins (2 cm cranially and caudally and 1.5 cm in other directions) and fused with an auto-segmented rectal nodal volume (Figure 1c–d), resulting in the construction of the AI-CTV structure (Figure 1e). This volume could then be compared with the ‘MC-CTV’ (Figure 1f).

Figure 1. Images from a single case illustrating the steps involved in constructing the AI-CTV. (a) AI-rectum + AI-recto-sigmoideum (blue) & MC-GTV (red). (b) MC-GTV (red) & AI-GTV (green). (c) AI-GTV (dark green) & margin (light green). (d) AI-GTV + margin (green) & AI-Ln-Rectal (orange). (e) AI-CTV (yellow). (f) MC-CTV (pink). Abbreviations: AI, artificial intelligence; MC, manual contours; GTV, gross tumour volume; Ln, lymph nodal volume; CTV, clinical target volume.
The intersection between our constructed AI-GTV and MC-CTVT was controlled to exclude any target misses (Figure 2a).

Figure 2. Sagittal view of the intersection between auto-segmentation-based structures and manual contoured target structures. (a) Red colour: MC-CTVT. Green Colour: Constructed AI GTV (Contour+ based). (b) Red colour: MC-CTV. Green Colour: Auto-segmented left internal iliac nodal volume (AI-Rad). Abbreviations: MC, manual contours; CTVT, clinical target volume of tumour; GTV, gross tumour volume; CTV, clinical target volume (including nodal fields); AI, artificial intelligence.
For AI-Rad, a similar approach for constructing an AI-CTV was not possible with the segments provided. However, the intersection between the MC-CTV and the internal iliac nodes suggested by AI-Rad was checked to measure the coverage of these lymph volumes (Figure 2b) and to explore a potential benefit of basing the contouring on this structure.
OAR segmentation: The bladder, bowel bag and femoral heads were manually contoured in the clinical setting and were available as ground truth in comparisons with auto-segmented structures. For the auto-segmented bowel bag, some revisions were made to enable a proper comparison with the MC bowel bag. AI-Rad provided the structure labelled ‘Abdomino-peritoneal space’, which included urogenital organs, which were cropped. For both AI software, all slices cranial and caudal to the MC-Bowel bag, as well as the CTV, were cropped. These revised structures were deemed acceptable for comparison with the MC-Bowel bag.
Metrics used to evaluate auto-segmentation: The quantitative metrics used in this study were the volumetric/surface Dice similarity coefficient (vDSC/sDSC), as well as the maximum/average Hausdorff distance (HD/aHD). The image structures were converted to binary masks prior to the calculation of these metrics. The tolerance value for the sDSC was, for all evaluated structures, set to 2 mm, a value considered acceptable for pelvic organs. Reference Rhee, Akinfenwa and Rigaud15
Qualitative evaluation: Contour quality was reviewed by two experienced oncologists (co-authors A.S. and C.L). We employed two different methods to assess the cases:
Grading test: The structures MC-CTV and AI-CTV were first blinded and randomised, then reviewed separately within the treatment planning module, with one week’s interval between the reviews of structures from the same case. A five-point Likert scale (Table 1) was developed and adapted based on similar scales used by Yu et al. Reference Yu, Anakwenze and Zhao16 and Almberg et al. Reference Almberg, Lervag and Frengen17 In the evaluations, the MC-GTV was highlighted to indicate the tumour’s location. No other information was given during the review. In cases graded 3 or lower, reviewers were also asked to specify the anatomical area requiring improvement and the nature of the needed adjustment, viz., expansion or reduction of contours. To facilitate the correlation analysis, the results from the five-point scale were converted symmetrically into a three-point scale, where scores of 1–2 corresponded to 1 (unacceptable), 3 to 2 (intermediate) and 4–5 to 3 (acceptable).
Table 1. Grading test: questions assessing clinical benefit of CTV structures

CTV, clinical target volume.
Preference test: As the grading test provided information about the entire segmentation, the reviewer could infer which structure had been delineated by AI, potentially introducing bias. Therefore, a second test was devised based on the ‘Turing test’ also designed by Wu et al. Reference Wu, Kang and Han14 In each case, 10 slices of the CTV were randomly selected, with contours of MC-CTV and AI-CTV randomly assigned the colours red or green in each individual slice (Figure 3). Each slice was then scored for preference of either colour using a bipolar 5-point Likert scale: ‘strongly for red/green’, ‘for red/green’ or ‘neither for nor against red/green’. With ten cases, 100 images were thus reviewed.

Figure 3. Examples of slices reviewed in the preference test. The slices were arranged from caudal to cranial direction. The colour given to structures was individually randomised in each slice. (a) AI: red, MC: green. (b) AI: green, MC: red. (c) AI: green, MC: red. (d) AI: red, MC: green. Abbreviations: AI, constructed clinical target volume (CTV); MC, manually contoured CTV.
Results
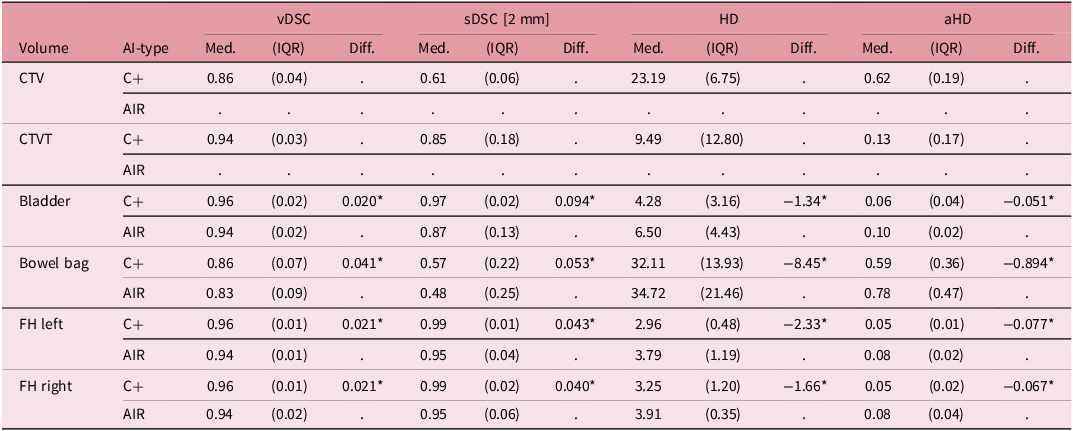
Quantitative metrics: In Table 2, summary statistics of the quantitative metrics of the constructed CTVs and CTVTs (Contour+-based) as well as for OARs from both evaluated AI tools are presented. For the constructed CTVs, the median values of vDSC, sDSC, HD and aHD were 0.86, 0.61, 23.2 mm and 0.62 mm, respectively. For the OARs, the differences between Contour+ and AI-Rad contours were tested for zero median, with p-values listed in the footnotes. There was a significant difference between the AI tools analysed for most OARs and metrics, except for the bowel bag, where the Contour+ OARs had a greater similarity to their MC counterpart, with higher vDSC and sDSC values and lower HD and aHD values compared to the AI-Rad OARs.
Table 2. Summary of statistics for quantitative metrics

*p < 0.01.
vDSC, volumetric Dice similarity coefficient; sDSC, surface Dice similarity coefficient; HD, Hausdorff distance; aHD, average Hausdorff distance; Med, median; IQR, inter-quartile range; CTV, clinical target volume; CTVT, clinical target volume of the tumour; FH, femoral head.
Qualitative tests: Table 3 presents both qualitative and quantitative metrics of our constructed AI-CTV, based on Contour+ auto-segmentations. Median grading test scores are displayed on a case-by-case basis. The median score on the five-point grading scale was 2.0 (range 1–3) for the constructed AI-CTV, indicating overall poor quality and a need for ‘major corrections with little to no time saved’. All constructed AI-CTVs were deemed to require revision by both reviewers. The median score for the MC-CTV was 4.3 (range 3–5), indicating overall acceptance of the MC.
Table 3. Case-by-case display of qualitative metrics and qualitative test scores for the constructed AI and MC CTV, including nodal fields

a Scored according to the question sheet in Table 1.
b A score of 1 indicates a strong preference for MC over AI contours, a score of 5 indicates the opposite, and a score of 3 indicates an equal preference.
*p < 0.01.
MC, manual contour; AI, constructed clinical target volume; vDSC, volumetric Dice similarity coefficient; sDSC, surface Dice similarity coefficient; HD, Hausdorff Distance; aHD, average Hausdorff distance.
For the preference test, the median of the reviewers’ mean scores for each case’s ten reviewed slices is presented. A score of 1 indicates a strong preference for MC, 3 indicates neutrality (neither for nor against MC or AI), and 5 indicates a strong preference for AI. A total median score of 2.0 represents an average, but not strong, preference for MC over AI.
There was a consistent difference between AI and MC segmentations, favouring MC in both the grading and preference tests, p-value <0.01. Some differences were observed between the two reviewers, although the variation was mostly limited to single steps on the scale.
In the poorly graded CTV structures, a frequently noted omission was the cranial-anterior nodal levels, that is, the nodes around the superior rectal artery and the anterior internal iliac artery, either due to the entire intended level being excluded or an insufficient margin around these vessels.
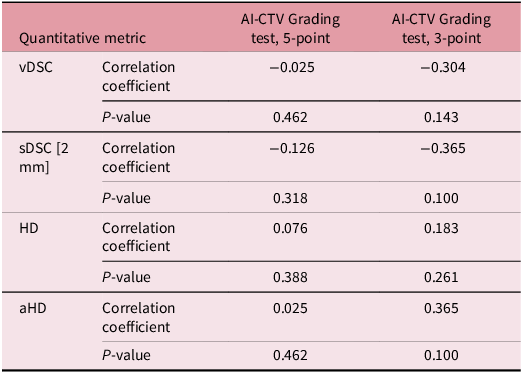
The correlation values between the metrics and the grading test score for the constructed AI-CTV are presented in Table 4. We found no statistical significance in this analysis. The correlation coefficients trended as negative for vDSC/sDSC and positive for HD/aHD, which is contrary to the expected relationship between a good metric score and a high grading score. This pattern was more pronounced when using the three-point scale compared to the five-point scale, although it was still not statistically significant.
Table 4. Correlation coefficient values of quantitative metrics versus grading test results of the constructed AI-CTV, 5-point and 3-point scales

AI-CTV, constructed clinical target volume; vDSC, volumetric Dice similarity coefficient; sDSC, surface Dice similarity coefficient; HD, Hausdorff distance; aHD, average Hausdorff distance.
Intersection analysis: The volumes of the constructed AI-GTV were covered by the structure MC-GTV+margin to an average extent of 99.6% (Figure 2a). The AI-Rad-segmented external iliac nodal level was, on average, 97.2% (dx) and 96.2% (sin) covered by the MC-CTV (Figure 2b).
Discussion
Interpretation of CTV results: The median vDSC of 0.86 for our constructed AI-CTV was comparable to mean values reported in other rectal cancer studies, e.g., Geng: 0.859, Wu: 0.914, Men: 0.8712 and Song: 0.8711. In contrast, the sDSC result, median 0.61, was lower than that reported by Song: 0.7911; however, they used a higher tolerance value of 4 mm. Our result for the HD, with a median value of 23.19 mm, was considerably higher than the mean values in the aforementioned studies, viz., between 7 and 9 mm. The maximum HD is sensitive to outlier values, Reference Taha and Hanbury18 and the shape of the CTV and the process of its construction may have resulted in more outliers, suggesting that maximum HD might not be a suitable metric for this structure. We have not found comparable results of aHD. Based on data from other organ structures, our result, a median of 0.62, was interpreted as intermediate (0.4–0.6). Reference Van Dijk, Van Den Bosch and Aljabar19
While our vDSC results for the constructed AI-CTV were acceptable, the corresponding qualitative grading score was low. In contrast, Geng et al. report considerably higher grading scores for their AI-CTV. This discrepancy between the qualitative results and vDSC values provides further evidence that vDSC results should be interpreted with caution.
The correlation analysis did not align as expected, indicating a relationship between bad metric scores and higher qualitative grades. However, since none of the analysed AI-CTVs were given a good grade, the variation reflects only different degrees of bad gradings. Moreover, this result was not statistically significant. Therefore, no conclusion can be drawn regarding the correlation between quantitative metrics and qualitative assessments.
Interpretation of OARs results: Contour+ performed slightly better than AI-Rad across all OARs. However, both tools produced acceptable results, and our findings were comparable to those of other studies assessing OARs in the pelvic region. Reference Song, Hu and Wu11,Reference Men, Dai and Li12 For the bowel bag structures, there was a greater degree of variation. The constructed bowel bag structures were not qualitatively reviewed in this study. However, similar results, with less degree of agreement between the auto-segmented bowel bag structures and their MC counterparts compared to other pelvic OARs, have been reported in previous reports as well, Reference Song, Hu and Wu11,Reference Men, Dai and Li12 suggesting that this structure is more difficult to auto-segment.
Evaluation of AI-CTV construction: Although our constructed AI structures were graded poorly, there was almost complete coverage of the AI-GTV by the MC-GTV with margins (Figure 2a), a test designed to safeguard against an overall miss of the target by the structure constructed by the auto-segmented rectum structure (Figure 1f).
Evaluation of the constructed CTV: In our trial, the MC-CTV was found to be significantly superior to our constructed AI-CTV in both quantitative methods and previously described quantitative metrics, strongly suggesting a low possibility for clinical use. This outcome may be attributed to the generally lower quality of the constructed AI structure compared to the MC; however, other factors could also have influenced the results:
-
• Contouring guidelines: One possible factor that could explain differences observed is the use of different contouring guidelines. While our results did not favour our constructed AI-CTV, similar studies, e.g., Wu et al. Reference Wu, Kang and Han14 and Geng et al., Reference Geng, Zhu and Liu9 reported no significant differences between their MC and AI-CTV structures. A key distinction is that the model used by Geng was based on a dataset from the study site itself, whereas the Contour+ AI employed in our study was trained on datasets using contouring guidelines different from those adopted by our department. The poor performance of the AI-CTV around the superior rectal artery exemplifies this, as this structure is not fully included in the RTOG guidelines, Reference Myerson, Garofalo and El Naqa3 whereas a greater portion is included in the RCR guidance. 4 Although Contour+ provided an additional structure covering the anterior nodal volume around the internal iliac artery, which could encompass this area, this structure instead encompassed a wider region anteriorly; therefore, it was not incorporated into our constructed AI-CTV.
-
• The error resulting from the use of an AI tool based on contouring guidelines that differ from those of the local department is of importance. In another study, Geng et al. Reference Geng, Sui and Du20 present a method of adapting commercial software to the clinic’s own cases and contouring conventions, offering an alternative approach to reducing these discrepancies rather than rejecting an otherwise useful AI tool.
-
• Intra-observer variation: Although the MC-CTVs were peer-reviewed and approved for treatment in our clinical setting, a few cases still required minor correction before they were deemed perfect by the two reviewers for use in RT. This further highlights the existing interobserver variation in both manual contouring and target review. While this variation may have influenced the results, the AI-CTVs in these cases still performed worse than their MC-CTV counterparts.
-
• Choice of qualitative methodology: There is considerable variation in the designs of grading tests used in similar studies, so direct comparisons of results between different studies should be approached with caution. While Baroudi et al. have proposed the use of their five-point scale in future research, Reference Baroudi, Brock and Cao8 we found, during the design and testing of their scale, a lack of nuance between certain grade levels, i.e., levels 1 and 2, rendered similar conclusions. A five-point scale was employed for the review owing to its lower risk of bias compared to scales with fewer points. Reference Baroudi, Brock and Cao8 The results were condensed to a three-point scale primarily for reducing parameters for the association analysis. These three categories correspond to clinically relevant distinctions. Since the reduction was symmetrical, the risk of bias was considered low.
-
• Bias: The grading test employed is considered more clinically relevant than the preference test. Additionally, reviewing both AI-CTV and MC-CTV in a blinded manner allowed for the performance of statistical tests of independence, despite the low number of cases. However, the preference test is less susceptible to bias, as it is easier to blind and results in a greater total number of evaluated images. Given the consistent preference for the MC across both tests, we interpret that the influence of bias on our results was minor.
Limitations: Our study is limited by the small sample size, partly due to the time-restricted trial licence of Contour+. Furthermore, only two reviewers were involved. Likert scales are inherently subjective, disproportionate and susceptible to bias. To partially mitigate these limitations, two distinct qualitative methods were employed. Given the significant differences observed between our constructed AI-CTV and MC-CTV structures across both methods, some conclusions can still be drawn.
Conclusion: In conclusion, our study did not find clinical utility of our constructed AI-CTV structures, in contrast to the few previously published data on auto-segmentation of rectal cancer target volumes. However, we observed that the contouring errors in these structures were limited in magnitude and may partly reflect differences in guidelines used to set up the AI tools employed and our local protocol. We hope that the findings of our study will provide novel and valuable guidance for the further developments and applications of auto-segmentation software, as well as for study design and metric interpretation. The use of AI tools still holds considerable promise for reducing the time spent contouring OARs and CTVs in RT planning following future improvements, and we are currently pursuing this objective through further research and development.
Acknowledgements
We thank Mikael Andersson Franko, biostatistician at the Department of Clinical Science and Education, Södersjukhuset, Karolinska Institutet, for helpful input on our statistical analysis and representatives of MVision AI Oy (Helsinki, Finland) for the use of their software in this research report.
Financial support
This work was supported by grants from the Swedish Cancer Society (Cancerfonden), 22 2052 S, and Stockholm Gotland Regional Cancer Centre, project number 2025/44.
Competing interests
The authors declare none.