Chronic pain – pain lasting for 3 months or longer – is common and a leading cause of disability, notably among US service members and veterans (Dieleman et al., Reference Dieleman, Cao, Chapin, Chen, Li, Liu and Scott2020; Nahin, Reference Nahin2017; Reed et al., Reference Reed, Fischer, Williams, Na and Pietrzak2024). Of US service members and veterans with chronic pain, over half report functional impacts – reduced ability to engage in activities of living – due to chronic pain (Barry et al., Reference Barry, Guo, Kerns, Duong and Reid2003; Reed et al., Reference Reed, Fischer, Williams, Na, Engel and Pietrzak2025).
Exposure to combat-related injuries may be one reason why US service members and veterans are at higher risk for chronic pain and pain-related disability than the general population. Central to the biopsychosocial model of chronic pain (Begum et al., Reference Begum, Veeranki, Chike, Tamang, Simard, Chen and Falasinnu2025; Cook, Brawer, & Vowles, Reference Cook, Brawer and Vowles2006; Gatchel et al., Reference Gatchel, Peng, Peters, Fuchs and Turk2007) is a fear–avoidance cycle, where fear of pain maintains chronic pain (G. J. Asmundson, Norton, & Norton, Reference Asmundson, Norton and Norton1999; Cook et al., Reference Cook, Brawer and Vowles2006). Accordingly, combat-related injuries (e.g. gunshot wounds and explosions), which may provoke sustained fear reactions and avoidance, may increase risk for chronic pain. Evidence suggests that combat injury is related to increased odds of chronic pain (Toblin et al., Reference Toblin, Quartana, Riviere, Walper and Hoge2014), higher frequency of moderate-to-severe chronic pain (Vollert et al., Reference Vollert, Kumar, Coady, Cullinan, Dyball, Fear and Schofield2024), and higher prevalence of post-injury pain (Kumar et al., Reference Kumar, Soliman, Gan, Cullinan, Vollert, Rice and Kemp2024) compared to noncombat injury. However, evidence remains limited on differences in intensity of pain from combat injuries compared to other sources of pain and on the psychological and functional impacts of combat injury–related pain. Investigating the link between combat injuries and psychological outcomes could inform both theory on fear as a mechanism of chronic pain development and treatment of combat injury in ongoing global conflicts.
Specific psychological outcomes relevant to those with chronic pain include post-traumatic stress disorder (PTSD) and major depressive disorder (MDD). PTSD and chronic pain are highly comorbid due to shared precipitating events, such as traumatic injury, and a mutual maintenance of symptoms from avoidance of feared stimuli (A. J. N. Asmundson et al., Reference Asmundson, Paluszek, Sommer, Bolton, Sareen, Afifi and Asmundson2025; Liedl et al., Reference Liedl, O’donnell, Creamer, Silove, McFarlane, Knaevelsrud and Bryant2010; Norman, Stein, Dimsdale, & Hoyt, Reference Norman, Stein, Dimsdale and Hoyt2008; Reed et al., Reference Reed, Fischer, Williams, Na and Pietrzak2024; Sharp & Harvey, Reference Sharp and Harvey2001). Combat injury may play an outsized role in the comorbidity between PTSD and chronic pain, given its occurrence within a fear-inducing context (Afari et al., Reference Afari, Harder, Madra, Heppner, Moeller-Bertram, King and Baker2009; Vollert et al., Reference Vollert, Kumar, Coady, Cullinan, Dyball, Fear and Schofield2024). MDD is also closely connected to chronic pain (Aaron et al., Reference Aaron, Ravyts, Carnahan, Bhattiprolu, Harte, McCaulley and Dudeney2025; Dudeney et al., Reference Dudeney, Aaron, Hathway, Bhattiprolu, Bisby, McGill and Dear2024) and frequently comorbid with PTSD (Androulakis et al., Reference Androulakis, Guo, Zhang, Sico, Warren, Giakas and Reyes2021; Boska, Bishop, & Ashrafioun, Reference Boska, Bishop and Ashrafioun2021). Yet it remains unknown whether combat injury is directly associated with an increased risk of these mental health conditions.
In addition to psychological sequelae, chronic pain following traumatic injuries affects psychosocial functioning, such as the ability to work or engage in social activities (Gironda et al., Reference Gironda, Clark, Ruff, Chait, Craine, Walker and Scholten2009). Pain-related interference in psychosocial functioning (i.e. pain interference) is therefore the primary target of pain-management interventions (Krebs et al., Reference Krebs, Lorenz, Bair, Damush, Wu, Sutherland and Kroenke2009; Ord et al., Reference Ord, Lad, Shura, Rowland, Taber and Martindale2021). Furthermore, psychological features of chronic pain are commonly targeted by interventions. Pain catastrophizing – extreme concerns about pain and its consequences – is a target of evidence-based interventions for chronic pain and associated with psychosocial functioning in veterans with pain (Murphy, Cordova, & Dedert, Reference Murphy, Cordova and Dedert2022). Pain interference and pain catastrophizing are also relevant to the fear–avoidance model of chronic pain, whereby catastrophizing about future pain leads to increased fear of activity and subsequent functional interference. It is paramount to better understand how combat injury may relate to these critical characteristics of pain in order to improve care.
Finally, it is important to understand whether the attribution of pain to a combat injury is associated with different pain characteristics than the attribution of pain to another source (e.g. a noncombat training injury, medical condition, etc.). In addition to the unique impacts of fear that a combat injury may cause on pain development, pain attributions – what is thought to be the cause(s) of pain – are also known to differentially predict functioning and treatment (e.g. Ashar et al., Reference Ashar, Lumley, Perlis, Liston, Gunning and Wager2023); however, no work at present has examined how a person’s attribution of their pain to a combat injury might associate with mental health outcomes. This gap could be particularly relevant to service members and veterans, who may be exposed to a number of potential sources of pain.
There are several clinical and theoretical implications from a better understanding of combat injury as a source of pain. Clinically, if combat injury is associated with greater pain catastrophizing and pain interference, triaging of service members who are injured in combat to early intervention that emphasizes coping strategies to reduce pain catastrophizing (e.g. cognitive behavioral therapy for chronic pain) and increased focus on early mobility following injury could improve outcomes. Additionally, because of the role of fear as a perpetuating mechanism in the mutual maintenance of pain and PTSD, early treatment of combat injury-related pain may mitigate the risk of PTSD subsequent to a combat injury by reducing vigilance and trauma-related attributions of painful stimuli as threats. Furthermore, the chronic stress resulting from unresolved pain catastrophizing and pain interference could have negative impacts on mood (e.g. MDD) that could be prevented by early intervention. However, these points are currently speculative due to the lack of existing evidence on the link between combat injury–related pain and mental health outcomes.
Given these gaps in knowledge, we first cross-sectionally examined the difference in odds of pain interference and adverse mental health outcomes associated with combat injury–related pain compared to noncombat injury–related pain in a probability sample of current and former US Army soldiers. Next, we longitudinally evaluated (1) the association of combat injury with subsequent pain interference, pain catastrophizing, and mental health outcomes, and (2) the mediation of the relation between combat injury and mental health outcomes by pain interference, pain catastrophizing, and clinically significant pain intensity using data from three US Army combat brigades surveyed before and at several time points following a combat deployment.
Methods
Participants and procedures
Data were from the Army Study to Assess Risk and Resilience in Servicemembers (Army STARRS) and the STARRS-Longitudinal Study (STARRS-LS). The design and procedures of Army STARRS and STARRS-LS are detailed elsewhere (Kessler et al., Reference Kessler, Colpe, Fullerton, Gebler, Naifeh, Nock and Stein2013a; Ursano et al., Reference Ursano, Colpe, Heeringa, Kessler, Schoenbaum and Stein2014). Briefly, STARRS-LS recruited a probability sample of service members who completed baseline surveys while on active duty as part of the Army STARRS New Soldier Study (NSS), All Army Study, or Pre/Post Deployment Study (PPDS; Kessler et al., Reference Kessler, Colpe, Fullerton, Gebler, Naifeh, Nock and Stein2013a; Ursano et al., Reference Ursano, Colpe, Heeringa, Kessler, Schoenbaum and Stein2014) and agreed to their survey responses being linked to their Army/Department of Defense (DoD) administrative data.
Cross-sectional analyses for the current study used data from wave 1 of STARRS-LS (LS1). A total of 14,508 current and former US Army soldiers participated in LS1 between September 2016 and April 2018. The LS1 analysis sample was restricted to participants whose baseline data were collected in the NSS (n = 6333), representing a cohort who enlisted between April 2011 and November 2012 and whose LS1 data were collected ~6 years post-enlistment. The LS1 sample was further constrained to those who reported pain (n = 5003), to enable comparisons of combat injury–related pain to pain from other sources.
Longitudinal analyses used data from the PPDS (January 2012–April 2014), which involved a baseline assessment before deployment to Afghanistan in 2012(T0), a brief assessment within ~1 month of returning(T1), and subsequent follow-ups 3 months(T2) and 9 months(T3) post-deployment. PPDS methods are detailed elsewhere (Campbell-Sills et al., Reference Campbell-Sills, Kautz, Choi, Naifeh, Aliaga, Jain and Ursano2023; Kessler et al., Reference Kessler, Colpe, Fullerton, Gebler, Naifeh, Nock and Stein2013a, Reference Kessler, Heeringa, Colpe, Fullerton, Gebler, Hwang and Schoenbaum2013b; Ursano et al., Reference Ursano, Colpe, Heeringa, Kessler, Schoenbaum and Stein2014). Briefly, 9949 service members were on duty in the 3 brigade combat teams (BCTs) eligible for recruitment, and 9488 (95.3%) consented to participate. Of those who consented, 8558 (86.0%) provided complete data and permitted Army/DoD record linkage. Because the analysis used data from all waves, the sample was restricted to the 60.0% of deployed service members with data at all time points (n = 4645). All participants provided informed consent, and study procedures were approved by the Institutional Review Boards of the collaborating institutions.
Measures
Combat injury–related pain and combat injury
Combat injury–related pain was defined in the LS1 survey by an item assessing the primary source of pain, for which ‘A combat injury’ was an option. Comparisons in LS1 were made between those who endorsed combat injury as their primary source of pain (1) and those with pain who did not endorse a combat injury as the primary source of that pain (0). In PPDS, combat injury was defined as responding ‘1 time’ or greater to the T1 survey item that queried being wounded during the index deployment. Comparisons were made between those who endorsed being wounded (1) and those who did not endorse being wounded (0).
Pain intensity, pain interference, and pain catastrophizing
Pain intensity was assessed in both LS1 and PPDS with the numeric rating scale (NRS; average pain from [0] No pain to [10] Pain as bad as could be). The NRS is reliable for the assessment of pain intensity (Ferreira-Valente, Pais-Ribeiro, & Jensen, Reference Ferreira-Valente, Pais-Ribeiro and Jensen2011; Jensen, Chen, & Brugger, Reference Jensen, Chen and Brugger2003). Clinically significant pain intensity (≥4 on the NRS) was defined per established guidelines (Jones, Vojir, Hutt, & Fink, Reference Jones, Vojir, Hutt and Fink2007).
Pain interference was assessed in the LS1 survey by the item, ‘How much did pain interfere with your normal work (including work outside the home and housework) in the past 30 days?’ with a five-point response scale ranging from (0) Not at all to (4) Extremely. Due to right-skewed distributions, responses were dichotomized, with pain interference defined as (2) Moderately or greater. At PPDS T0 and T2, pain interference was assessed with two items from the Graded Chronic Pain Scale (Von Korff, Ormel, Keefe, & Dworkin, Reference Von Korff, Ormel, Keefe and Dworkin1992) assessing interference with work and social activities on a scale from (0) None of the time to (4) All or almost all the time. Respondents were coded as having pain interference if they endorsed (2) Some of the time or greater on either item. Internal consistency of the dichotomized items was high (α = 0.86). At T3, the pain interference assessment was restricted to interference with work, with pain interference coded as present if the item was rated (2) Some of the time or greater.
Pain catastrophizing was also assessed in PPDS, through three items from the Pain Catastrophizing Scale (Sullivan, Bishop, & Pivik, Reference Sullivan, Bishop and Pivik1995) rated from (0) None of the time to (4) All or almost all the time. These same items were assessed at T0 and T2. Pain catastrophizing was coded as present if any of these three items were rated (2) Some of the time or greater, with the overall score demonstrating acceptable internal consistency (α = 0.78).
Mental health
PTSD was assessed via adapted versions of the PTSD Checklist-Civilian Version (NSS and PPDS T0 surveys) and PTSD Checklist for DSM-5 (PCL-5; LS1 and PPDS T2 and T3 surveys; Blevins et al., Reference Blevins, Weathers, Davis, Witte and Domino2015; Weathers et al., Reference Weathers, Litz, Herman, Huska and Keane1993). Major depressive episodes (MDEs) were evaluated in all surveys using items adapted from the Composite International Diagnostic Interview Screening Scales (CIDI-SC; Kessler & Üstün, Reference Kessler and Üstün2004). Questions were asked about the experience of mental health symptoms over the respondent’s lifetime in the NSS and PPDS T0 surveys and over the past 30 days in the PPDS T2 and T3 and LS1 surveys. The outcomes used for this study were binary variables of (1) meets probable diagnosis or (0) does not meet probable diagnosis. Army STARRS survey-based diagnoses demonstrated good agreement with diagnoses based on clinical interviews in a previous study (Kessler et al., Reference Kessler, Colpe, Fullerton, Gebler, Naifeh, Nock and Stein2013a, Reference Kessler, Heeringa, Colpe, Fullerton, Gebler, Hwang and Schoenbaum2013b).
Covariates
Covariates in each model included baseline sociodemographic features (age, sex, race, and ethnicity). Raw race and ethnicity responses were categorized as Black, Hispanic, Other, and Non-Hispanic White. Baseline lifetime diagnosis of PTSD and MDE were also included in all models to account for pre-enlistment (the NSS baseline for the LS1 sample) or pre-deployment (PPDS T0) status on the outcome of interest. In the LS1 analysis, the models also adjusted for current military status (active duty, activated Guard/Reserve, deactivated Guard/Reserve, separated, or retired) and history of combat deployment (yes or no). In PPDS, the models also adjusted for BCT, number of previous combat deployments (0, 1, or > 1), and number of deployment stressors (minus the focal exposure of being wounded; Stein et al., Reference Stein, Kessler, Heeringa, Jain, Campbell-Sills, Colpe and Schoenbaum2015).
Data analysis
Weights-adjusted values were used for sample description and statistical models. For the LS1 sample, weights included nonresponse and poststratification adjustments for Army STARRS baseline and STARRS-LS follow-up survey data, oversampling of baseline respondents with mental health concerns, and underrepresentation of difficult-to-recruit participants in STARRS-LS. The weighting procedures for LS1 have previously been described (Naifeh et al., Reference Naifeh, Capaldi, Chu, King, Koh, Marx and Stanley2023).
For the PPDS sample, weights included (1) a propensity-based adjustment for baseline attrition; (2) post-stratification based on key demographic and Army service characteristics to generalize to soldiers who deployed to Afghanistan after T0; and (3) a propensity-based attrition adjustment to account for incomplete data in one or more of the three follow-up waves. More information about the weighting of Army STARRS data is available elsewhere (Kessler et al., Reference Kessler, Heeringa, Colpe, Fullerton, Gebler, Hwang and Schoenbaum2013b).
For the PPDS sample, we assessed the association of combat injury at T1 with the new onset of pain interference and catastrophizing at T2 and T3 using weights-adjusted logistic regression models.
For both LS1 and PPDS samples, Spearman correlations were used for bivariable associations, and weights-adjusted multivariable logistic regression was conducted to assess the independent associations of combat injury–related pain (in LS1), combat injury (in PPDS), clinically significant pain intensity, pain interference, and pain catastrophizing (only in PPDS) with mental health outcomes adjusted for relevant covariates. In the LS1 sample, the interaction of combat injury-related pain and pain interference was included to explore the potential increased mental health impact of functional interference due to combat injury compared to other pain sources. Adjusted odds ratios (ORs) with 95% confidence intervals were reported. Variance inflation factors ranged between 1.10 and 2.31 across all models and terms, indicating that multicollinearity was unlikely among the variables included.
For the PPDS sample, we also examined the extent to which associations between combat injury and mental health outcomes were explained by pain variables (clinically significant pain intensity, pain interference, and pain catastrophizing) using the ‘mma’ package in R to run multiple mediation analysis (Yu & Li, Reference Yu and Li2017). The models also adjusted for the relevant covariates. The estimated total, direct, and indirect (mediation) effects on the log-odds of the outcome were reported with confidence intervals estimated using bootstrapping. All analyses were performed between November 2024 and July 2025 in R version 4.4.1 (R Core Team, 2018).
Results
Sample characteristics
The descriptive statistics, including raw frequencies and weights-adjusted percentages, for both samples are in Table 1. Of the 6333 NSS respondents who completed surveys at LS1, 5003 (80%) endorsed past-30-day pain ‘a little of the time’ or more. Of these respondents, 2453 (50.5%) endorsed clinically significant pain intensity, 1442(29.7%) endorsed pain interference, 1101 (22.5%) endorsed MDE, and 1180 (22.9%) endorsed PTSD. In the LS1 sample with pain, clinically significant pain intensity and pain interference were significantly, positively correlated (ρ = .54).
Baseline descriptive statistics for the study samples

Note: Table contains raw frequencies and weights-adjusted percentages/means based on the sampling approaches taken to each study. LS1 survey data were used in the cross-sectional analysis. PPDS was a longitudinal survey comprised of a baseline (T0) assessment, and 1 (T1), 3 (T2), and 9 months (T3) following return from deployment. LS1, STARRS Longitudinal Survey-Wave 1; PPDS, pre–post-deployment survey; PTSD, post-traumatic stress disorder; MDE, major depressive episode.
In the longitudinal sample (PPDS), of the 4645 soldiers who completed all waves, 1741 (36.7%) endorsed clinically significant pain intensity at T2, 1136 (23.6%) endorsed pain interference at T2 and 1106 (18.2%) at T3, 861 (17.9%) endorsed pain catastrophizing at T2 and 599 (13.8%) at T3, 357 (7.7%) reported PTSD and 330 (6.7%) endorsed MDE at T2, and 547 (9.6%) endorsed PTSD and 314 (5.7%) endorsed MDE at T3. In this sample, clinically significant pain intensity was significantly correlated with pain interference (T2: ρ = .60; T3: ρ = .59 and pain catastrophizing (T2: ρ = .53; T3: ρ = .45) at T2 and T3.
Combat injury–related pain in LS1
Among those endorsing pain at LS1, combat injury–related pain relative to pain from other injuries or conditions was associated with a 2.17 (95% CI: 1.44–3.27) times increase in odds of past-12-month MDE after controlling for covariates. When pain intensity and interference were added to the model, combat injury–related pain was no longer significantly associated with MDE (Table 2). Instead, pain interference (OR = 3.04, 95% CI: 2.47–3.74) and clinically significant pain intensity (OR = 2.18, 95% CI: 1.64–2.91) were associated with increased odds of MDE. There was, however, a significant interaction between combat injury–related pain and pain interference, such that those respondents with combat injury–related pain and pain interference had magnified odds of MDE compared to those with pain interference alone (OR = 5.46, 95% CI: 1.71–17.44).
Weights-adjusted multivariable logistic regression of past-12-month MDE and PTSD in LS1

Note: Total N = 4987. OR = odds ratio; CI, confidence interval; LL, lower limit; UL, upper limit; MDE, major depressive episode; PTSD, post-traumatic stress disorder. Models with main effects only were retained when interaction terms were nonsignificant.
Combat injury–related pain relative to pain from other injuries or conditions was also associated with a 3.96 (95% CI: 2.71–5.79) times increase in odds of past-12-month PTSD. When pain intensity and interference were added to the model, combat injury–related pain remained significantly associated with PTSD (Table 2; OR = 2.89, 95% CI: 1.85–4.51). Additionally, pain interference (OR = 2.68, 95% CI: 2.07–3.46) and clinically significant pain intensity (OR = 1.93, 95% CI: 1.51–2.46) were associated with increased odds of PTSD. The interaction term was nonsignificant in this analysis, so the main-effects model was accepted as the final model.
Combat injury in PPDS
New-onset pain interference and catastrophizing
Combat injury was associated with increased odds of new-onset pain interference and catastrophizing at both T2 and T3. See Supplementary Material for details.
PTSD
Combat injury was associated with increased odds of past-30-day PTSD at T2 (OR = 4.06, 95% CI: 2.68–6.16) compared to no combat injury. When T1 deployment stress and T2 pain variables were added to the model, combat injury remained associated with increased odds of PTSD (Table 3; OR = 1.94, 95% CI: 1.19–3.14), as were pain interference (OR = 2.54, 95% CI: 1.65–3.92) and pain catastrophizing (OR = 2.10, 95% CI: 1.43–3.07). Combat injury was again associated with past-30-day PTSD at T3 (OR = 2.86, 95% CI: 1.99–4.09). When pain variables were added to the model, the association was attenuated but still significant, with pain variables also exhibiting significant associations with past-30-day PTSD (Table 3).
Weights-adjusted multivariable logistic regression of past-30-day PTSD and MDE in PPDS T2 and T3

Note: Total N = 4456. OR, odds ratio; CI, confidence interval; LL, lower limit; UL, upper limit; MDE, major depressive episode. Reference categories: Female: Male; Race: non-Hispanic White; Deployments: Not previously deployed. Models also adjusted for Brigade Combat Team. Significant associations are italicized.
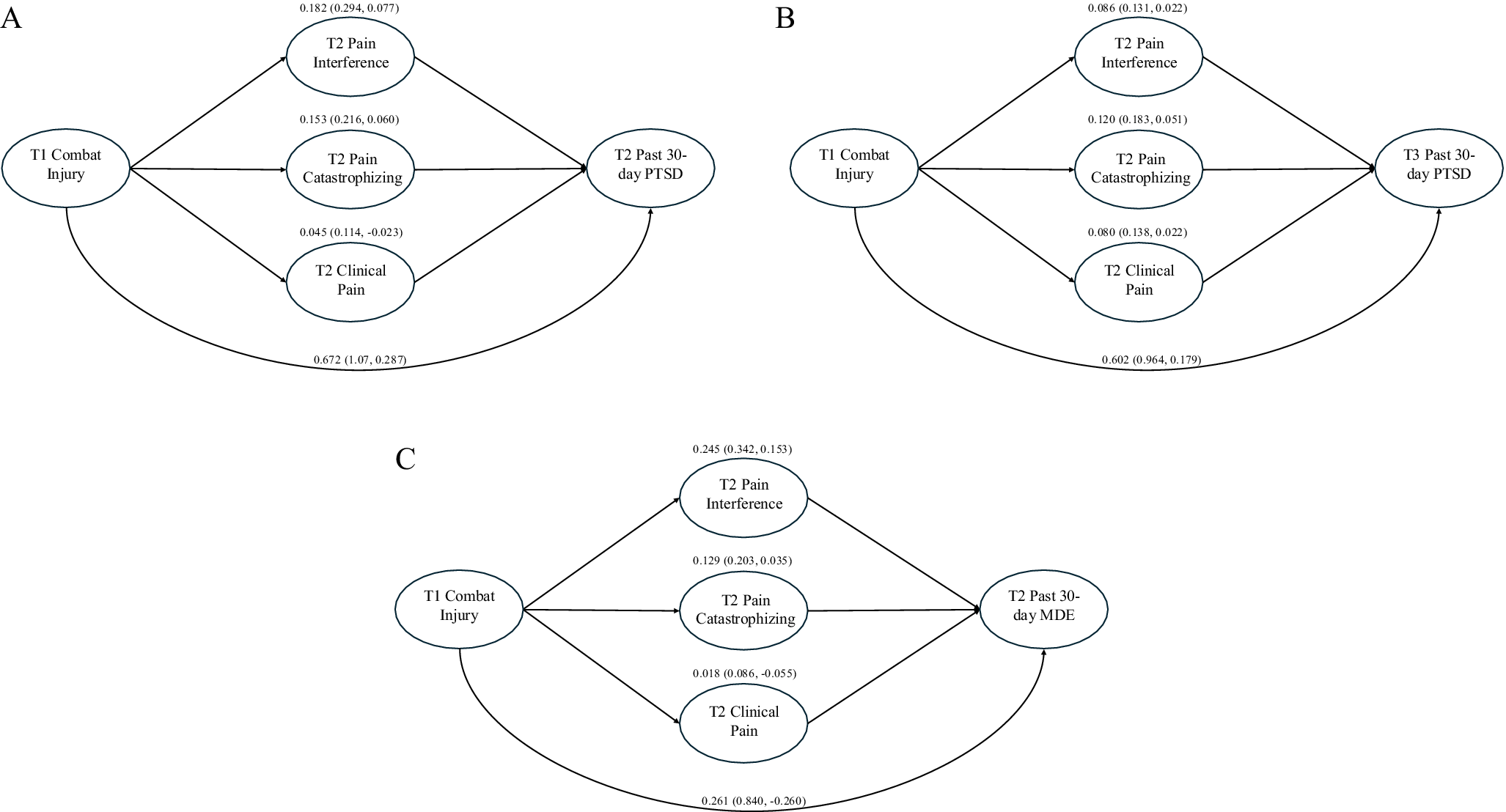
Joint mediation of the relation between combat injury and PTSD by pain variables
In the multiple mediation analysis (Figure 1a), the total effect of combat injury on past-30-day PTSD at T2 indicated that those with a combat injury had 2.89 times greater odds of PTSD than those without a combat injury (log odds = 1.06, 95% CI: 0.61–1.50). The joint mediation effect (i.e. total indirect effect) indicated 34% of the increase in odds of PTSD associated with combat injury was explained by the pain variables (log odds = 0.36, 95% CI: 0.22–0.52). Most of the mediation effect was attributable to pain interference (17.2%) and pain catastrophizing (14.4%). For T3 PTSD (Figure 1b), the odds of past-30-day PTSD were 2.43 times greater for those with a combat injury than those without (log odds = 0.89, 95% CI: 0.45–1.25). The joint mediation effect indicated 33% of the increase in odds of T3 PTSD due to combat injury was explained by the pain variables (log odds = 0.29, 95% CI: 0.18–0.37). The mediation effect in the model of T3 PTSD was more equally distributed among the three pain variables than in the model of T2 PTSD (Figure 1b).
Multiple mediation models of (a) the relation between T1 combat injury and T2 past-30-day PTSD, (b) the relation between T1 combat injury and T3 past-30-day PTSD, and (c) the relation between T1 combat injury and T2 past-30-day MDE. The coefficients for the full effect of combat injury on PTSD/MDE and the mediation effect of each mediator are shown on the log odds scale.

MDE
Combat injury was associated with increased odds of past-30-day MDE at T2 (OR = 2.56, 95% CI: 1.74–3.76) compared to no combat injury. When T1 deployment stress and T2 pain variables were added to the model, pain interference (OR = 3.87, 95% CI: 2.58–5.81) and pain catastrophizing (OR = 1.74, 95% CI: 1.16–2.60) were associated with increased odds of MDE, but combat injury was no longer associated (Table 3; OR = 1.32, 95% CI: 0.81–2.15).
Combat injury also exhibited a significant association with past-30-day MDE at T3 (OR = 1.74, 95% CI: 1.07–2.85) that was eliminated when T2 pain interference (OR = 1.84, 95% CI: 1.35–2.52) and clinically significant pain intensity (OR = 1.80, 95% CI: 1.30–2.49) were included in the model (Table 3).
Joint mediation of the relation between combat injury and MDE by pain variables
Multiple mediation analysis (Figure 1c) revealed that service members with a combat injury had 1.91 times greater odds of past-30-day MDE at T2 than those without a combat injury (log odds = 0.65, 95% CI: 0.11–1.24). The joint mediation effect indicated 60% of the increase in odds of MDE due to combat injury was explained by the pain variables (log odds = 0.39, 95% CI: 0.26–0.52). Most of the mediation effect was attributable to pain interference (37.9%) and pain catastrophizing (19.9%). For T3 MDE, the total effect was not significant (log odds = 0.37, 95% CI: −0.19–0.79).
Discussion
Results suggest that combat injury is associated with increased odds of new-onset pain interference, new-onset pain catastrophizing, PTSD, and MDE compared to no combat injury (irrespective of the presence of another painful condition) among combat-deployed US service members. In order to disambiguate the impact of combat injury broadly and pain specifically related to that injury, we assessed the cross-sectional relation of pain primarily attributed to a combat injury (i.e. combat injury–related pain) with pain experiences and mental health outcomes. We found that, compared to pain from other injuries and conditions, combat injury–related pain was associated with increased odds of PTSD and MDE, with the relation between combat injury–related pain and MDE potentiated by pain interference. This significant interaction of pain interference and combat injury–related pain on odds of MDE suggests that the functional impact of combat injury–related pain in particular may increase the odds of developing MDE. This finding is consistent with prior work demonstrating that the interference of pain with activities drives the association between pain and depression (Von Korff & Simon, Reference Von Korff and Simon1996). Taken together with the elevated odds of PTSD among those with combat injury–related pain, this is evidence of a disproportionate mental health impact of pain from combat injuries versus other attributed causes, and suggests that the combination of chronic pain from a combat injury and pain-related functional impairment is associated with a particularly elevated risk of mood difficulties.
Evidence also indicates that pain-related experiences mediate the relation between combat injury and subsequent mental health difficulties. For example, pain catastrophizing and interference (but not intensity) explained significant proportions of the effect of combat injury on risk of concurrent PTSD and MDE (i.e. assessed at the same time point as the pain variables). This may reflect bidirectional effects of pain-related factors and mental health symptoms, whereby mental health symptoms magnify the psychological and functional burdens of pain and vice versa. Furthermore, all three pain-related factors mediated the relations between combat injury and PTSD, evaluated 6 months after the pain assessment. This suggests that the intensity of pain assessed several months after injury – along with its psychological and functional impacts – impacts the risk of chronic PTSD among those who sustain combat injury.
Findings are consistent with the theory that threat-based cues are relevant to both the initiation of chronic pain (i.e. threat of reinjury/increased pain) and the mutual-maintenance model of pain and mental health comorbidities (e.g. threat of reexperiencing trauma in PTSD; McAndrew et al., Reference McAndrew, Lu, Phillips, Maestro and Quigley2019; Sharp & Harvey, Reference Sharp and Harvey2001). The significant mediation of the prospective relation between combat injury and PTSD by psychological factors of pain highlights the potentially mechanistic role of fears about pain in the relation between initial fearful experiences (i.e. combat injury) and long-term risk of PTSD. The unique contextual factors of military service may be particularly relevant to the role of threat-based cues and pain in the relation between injury and PTSD among service members and veterans. For example, training to remain vigilant could lead to a chronic sense of threat that could potentiate pain experiences and maintain PTSD. Furthermore, several psychological (e.g. suppression of emotions) and behavioral (e.g. lack of pacing of physical activity, pushing physical limits) predisposing factors for chronic pain are situationally adaptive in military culture. Taken together, findings support the supposition that aspects of military service could predispose chronic pain that could be precipitated by combat injury and perpetuated by psychological reactions to pain (e.g. pain catastrophizing). The study findings also broadly align with the chronic pain cycle, in which pain catastrophizing leads to less functional activity (e.g. work, social activities), which negatively impacts mood, social support, and physical fitness, which in turn leads to increased pain intensity and catastrophizing.
This study has limitations. We did not have a measure of injury severity or the type of injury experienced (e.g. penetrating, blast, etc.). While pain-related variables were collected more than 3 months following combat injury in both samples, indicating that any persisting pain interference or catastrophizing from the injury was consistent with a chronic pain process (i.e. pain lasting more than 3 months), future work including explicit measures of injury severity (e.g. the Injury Severity Score; Baker, ’Neill, Haddon, & Long, Reference Baker, ’Neill, Haddon and Long1974) will better disambiguate the impact of injury severity versus pain sources and experiences. Furthermore, measures of combat injury and mental health outcomes were based on the service member’s report and not derived from a medical record or confirmed by a provider. Additionally, pain interference and catastrophizing were derived from a brief set of items. While these items are derived from validated measures and the internal consistencies of the items are satisfactory, further work examining the pain experiences of service members with more detailed assessments is warranted. Another limitation is the dichotomization of pain interference, pain catastrophizing, PTSD, and MDE, which artificially reduces these complex constructs into binary experiences. To mitigate this concern, the probable diagnoses of PTSD and MDE used in this study have been validated previously (e.g. Kessler et al., Reference Kessler, Colpe, Fullerton, Gebler, Naifeh, Nock and Stein2013a, Reference Kessler, Heeringa, Colpe, Fullerton, Gebler, Hwang and Schoenbaum2013b). While they do not necessarily reflect the severity of symptoms, these carry the benefit of clinical relevance. Our results offer important insight into the impact of the presence of pain interference and pain catastrophizing on likely diagnosis of PTSD and MDE; however, future work would benefit from examining these constructs as dimensional variables to understand the impact of the severity of interference and catastrophizing on the severity of PTSD and MDE symptom burden. In particular, examination of the ways in which combat injury and pain factors affect each symptom cluster in PTSD (reexperiencing, avoidance, negative alterations of cognition and mood, and changes in arousal and reactivity) could lead to greater understanding of how combat injury and chronic pain mechanisms may drive comorbidity of chronic pain and PTSD.
Several treatment implications arise from these findings. First, results suggest that effective, early treatment of pain in service members may have benefits for both overall functioning and prevention of negative mental health outcomes. The early and comprehensive treatment of acute pain could prevent the transition to chronic pain through reduction in nociceptive distress, which can, in turn, reduce the burden of allodynia and hyperalgesia (i.e. increased pain sensitivity) following injury. Psychoeducation regarding risk factors and treatment for chronic pain delivered proactively to service members and reinforced after injury could also be beneficial. Second, traumatic sources of pain (e.g. combat injury) may respond to psychosocial pain treatments, particularly treatments that specifically target the mutual maintenance of PTSD and pain through fear processes (e.g. Emotion Awareness and Expression Therapy [EAET] for Chronic Pain; Yarns et al., Reference Yarns, Jackson, Alas, Melrose, Lumley and Sultzer2024). Relatedly, the finding that attribution of pain to a combat injury is associated with pain-related functioning and mental health outcomes compared to other pain attributions suggests that treatments, such as EAET, that focus on reattributing pain from physical sources to mind/brain sources could also be effective (Ashar et al., Reference Ashar, Lumley, Perlis, Liston, Gunning and Wager2023). Finally, the impact of pain interference and pain catastrophizing proximal to the injury on the development of later mental health conditions indicates that psychosocial treatments that target interference and catastrophizing (e.g. cognitive behavioral therapy for chronic pain) in the immediate aftermath of a combat injury may have broad-reaching benefits (Murphy et al., Reference Murphy, Cordova and Dedert2022).
Taken together, the results of this study suggest that both combat injury and the attribution of pain to a combat injury are closely, positively related to mechanisms of chronic pain – pain catastrophizing and interference – and may warrant enhanced screening and pain management interventions at the time of injury to mitigate the development of pain interference and pain catastrophizing, and subsequent chronic pain and mental health concerns. These findings are particularly relevant in the context of ongoing global conflicts, increasing the exposure of both civilians and military personnel to combat-related injury.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0033291726103584.
Acknowledgments
The Army STARRS Team consists of Co-Principal Investigators: Robert J. Ursano, MD (Uniformed Services University) and Murray B. Stein, MD, MPH (University of California San Diego and VA San Diego Healthcare System); Site Principal Investigators: James Wagner, PhD (University of Michigan) and Ronald C. Kessler, PhD (Harvard Medical School); Army scientific consultant/liaison: Kenneth Cox, MD, MPH ([Office of the Assistant Secretary of the Army (Manpower and Reserve Affairs]); Other team members: Pablo A. Aliaga, MA (Uniformed Services University); David M. Benedek, MD (Uniformed Services University); Laura Campbell-Sills, PhD (University of California San Diego); Catherine L. Dempsey, PhD (Uniformed Services University); Carol S. Fullerton, PhD (Uniformed Services University); Nancy Gebler, MA (University of Michigan); Meredith House, BA (University of Michigan); Paul E. Hurwitz, MPH (Uniformed Services University); Sonia Jain, PhD (University of California San Diego); Tzu-Cheg Kao, PhD (Uniformed Services University); Chris J. Kennedy, PhD (Massachusetts General Hospital); Lisa Lewandowski-Romps, PhD (University of Michigan); Alex Luedtke, PhD (University of Washington and Fred Hutchinson Cancer Research Center); Holly Heberman Mash, PhD (Uniformed Services University); James A. Naifeh, PhD (Uniformed Services University); Matthew K. Nock, PhD (Harvard University); Nancy A. Sampson, BA (Harvard Medical School); Rachel Shor (Uniformed Services University); and Alan M. Zaslavsky, PhD (Harvard Medical School).
Funding statement
The Army Study to Assess Risk and Resilience in Servicemembers (STARRS) was supported by the Department of the Army and funded under cooperative agreement number U01MH087981 (2009–2015) with the National Institute of Mental Health (NIMH). STARRS-Longitudinal Survey was then sponsored and funded by the Department of Defense (USUHS grant number HU0001-15-2-0004). MGW is also supported by the United States Department of Veterans Affairs (VA) VISN 17 Center of Excellence for Research on Returning War Veterans, the Central Texas Veterans Affairs Healthcare System, and the VA Suicide Prevention Actively Managed Portfolio – project SPR-001-25F.
Competing interests
In the past 3 years, Dr. Kessler was a consultant for Cambridge Health Alliance, Canandaigua VA Medical Center, Child Mind Institute, Holmusk, Massachusetts General Hospital, Partners Healthcare, Inc., RallyPoint Networks, Inc., Sage Therapeutics, and the University of North Carolina. He has stock options in Cerebral Inc., Mirah, PYM (Prepare Your Mind), Roga Sciences, and Verisense Health. In the past 3 years, Dr. Stein has received consulting income from atai Life Sciences, BigHealth, Biogen, Bionomics, Boehringer Ingelheim, Delix Therapeutics, EmpowerPharm, Engrail Therapeutics, Janssen, Jazz Pharmaceuticals, Karuna Therapeutics, Lundbeck, Lykos Therapeutics, NeuroTrauma Sciences, Otsuka US, PureTech Health, Roche/Genentech, Sage Therapeutics, Seaport Therapeutics, and Transcend Therapeutics. Dr. Stein has stock options in Oxeia Biopharmaceuticals and EpiVario. He has been paid for his editorial work on Depression and Anxiety (Editor-in-Chief), Biological Psychiatry (Deputy Editor), and UpToDate (Co-Editor-in-Chief for Psychiatry). He has also received research support from the National Institutes of Health, the Department of Veterans Affairs, and the Department of Defense. He is on the scientific advisory board of the Brain and Behavior Research Foundation and the Anxiety and Depression Association of America. Dr. Benedek has received royalties from UpToDate. The other authors have no known conflicts of interest to declare. Since the authors are employees of the U.S. Government and contributed to the manuscript as part of their official duties, the work is not subject to US copyright.
Ethical standard
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Disclaimer
The views expressed in this manuscript are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs, the Department of Defense, the Department of the Army, the National Institute of Mental Health, or the United States Government.