Introduction
A growing body of research is dedicated to understanding the experiences of individuals participating in health research, including their perspectives regarding the consent process [Reference O. Sullivan, Feeney, Crowley, Sukumar, McAuliffe and Doran1], concerns around data sharing and privacy [Reference Hendricks-Sturrup and Lu2], the importance of supportive logistics [Reference Cartmell, Bonilha, Simpson, Ford, Bryant and Alberg3], and the foundational role of trust [Reference Kraft, Cho and Gillespie4]. Literature consistently demonstrates that research participation is influenced by a complex interplay of personal, social, and structural factors. For example, Kost et al. conducted focus groups with research participants and found that motivations for participation often centered around altruism and the personal relevance of the study, with financial incentives serving as a less important motivator [Reference Kost, Lee, Yessis, Coller and Henderson5]. However, many participants expressed dissatisfaction over not receiving individual or aggregate study results and a growing gap in research translation, a frustration also substantiated in our recent work [6,Reference Poger, Abrams Schrodel, McCormick, Bordner and Kraschnewski7]. While altruism and trust enhanced research engagement, barriers such as distrust, logistical challenges, and a lack of clear communication remain significant [Reference Kost, Lee, Yessis, Coller and Henderson5,Reference Mouslim, Johnson and Dean8,Reference Kehoe, Locke, McClellan, Landray, Hernandez and Okun9].
Additionally, rural perspectives on research are likely shaped by health and healthcare experiences differing from their urban counterparts. For example, rural counties in PA have fewer primary care physicians (PCP), medical specialists, and allied health professionals, including five counties without maternal care [10]. Unfortunately, this geographic isolation leads to numerous health disparities, including higher infant mortality rates, as well as higher rates of chronic conditions such as cancer and obesity [11]. Such barriers are further exacerbated in rural communities with limited access to healthcare and research infrastructure due to hospital closures and the absence of other community safety nets [Reference Mudaranthakam, Gajewski and Krebill12,Reference Morgan, Fahs and Klesh13]. National data show disparities in research engagement between rural and urban populations [Reference Graves, Beese, Abshire and Bennett14], underscoring the importance of understanding the unique motivators for, and barriers to, research participation experienced by rural residents. Existing studies largely rely on qualitative data, focus on specific diseases, or reflect investigator perspectives rather than participant viewpoints [Reference Tanner, Kim, Friedman, Foster and Bergeron15,Reference Chichua, Mazzoni, Marzorati and Pravettoni16].
We developed and administered a human-centered, evidence-informed survey to assess motivators and barriers to research participation among adults in Pennsylvania. The survey was specifically designed to capture differences between individuals with and without prior research participation and between those living in rural versus urban areas. We hypothesized that individuals with prior research experience would report different motivators and barriers than those who have never participated in research. We also anticipated that rural and urban respondents would differ in their reported motivators and barriers to participation. A key goal of this study is to inform the development of targeted strategies to improve research engagement, particularly among rural populations.
Methods
Recruitment and outreach
Participants were recruited by email invitations, website postings, flyers, and word-of-mouth to complete a one-time deidentified survey capturing their perceptions of research and basic demographic characteristics. Names and contact information were collected separately in a post-survey form to facilitate compensation. The most effective method of recruitment for this study was leveraging the Volunteer Repository, a platform to quickly and easily identify studies across Penn State with searchable filters. Built onto the Penn State StudyFinder infrastructure, the Volunteer Repository gives interested volunteers the opportunity to self-select research topics of interest, enhancing their awareness of research opportunities and connecting them with relevant studies through targeted communication. The email invitation described the purpose of the survey in lay terms, provided eligibility criteria (18 years of age+), and estimated participant commitment (approximately 15 minutes). Almost half of the study sample (N = 122) was obtained from email invitations to the Volunteer Repository participants. Additionally, a study flyer was created and disseminated via the Penn State University Park and College of Medicine Clinical Research Centers, Penn State Health clinics, and the LION Mobile Clinic, which reaches rural central Pennsylvania communities [Reference Brant, Segel and McShane17,18].
Survey methodology
A 33-item questionnaire was developed through previously cited themes around barriers/motivators to research participation [Reference Kost, Lee, Yessis, Coller and Henderson5,Reference Boyd, Sternke, Tite and Morgan19,Reference Allison, Patel and Kaur20]. Five motivation themes emerged and were adapted during survey design: altruism or contribution [Reference Kost, Lee, Yessis, Coller and Henderson5,Reference Boyd, Sternke, Tite and Morgan19,Reference Bergmann, Matzneller and Weber21,Reference Burt, Dhillon and Sharma22], incentive [Reference Bergmann, Matzneller and Weber21], personal relevance [Reference Boyd, Sternke, Tite and Morgan19,Reference Shaver, Nikita and Banks23], relationships, and trust [Reference Bergmann, Matzneller and Weber21,Reference Burt, Dhillon and Sharma22]. Similarly, six barrier themes were selected or adapted to better understand the needs and perceptions of Pennsylvanians: access [Reference Boyd, Sternke, Tite and Morgan19,Reference McKinney, Bell and Samborski24], incentive, ethics [Reference Allison, Patel and Kaur20], health concerns [Reference Boyd, Sternke, Tite and Morgan19,Reference Allison, Patel and Kaur20,Reference McKinney, Bell and Samborski24,Reference Shaver, Cao and Noyes25], management [Reference Boyd, Sternke, Tite and Morgan19,Reference McKinney, Bell and Samborski24], withholding of study outcomes [Reference Kost, Lee, Yessis, Coller and Henderson5,Reference Boyd, Sternke, Tite and Morgan19], and distrust of site or team [Reference Allison, Patel and Kaur20,Reference Burt, Dhillon and Sharma22].
Questions (Table 1) regarding potential barriers and motivations for participating in research were structured as multiple-choice questions where participants could select all barriers/motivators that applied to them. Each barrier/motivator was then coded as “Yes” or “No” for analysis based on whether the participant selected it or not. The survey was conducted using REDCap, a secure, HIPAA-compliant web application for survey data collection [Reference Harris, Taylor, Thielke, Payne, Gonzalez and Conde26,Reference Harris, Taylor and Minor27]. After finishing the survey, participants were asked to complete a separate secure form via REDCap to provide contact information to enter a drawing for one of ten $100 gift cards. All identifying information was collected solely for compensation purposes, and their main survey responses remained deidentified.
Motivators and barriers to research participation

Classification of rurality
Rurality was determined by the state-based definition set forth by the Center for Rural Pennsylvania [28], which is based on population density. Using the Center for Rural Pennsylvania’s definitions, there are 48 rural counties (26% of the state’s population) and 19 urban counties in PA. We intentionally used Pennsylvania’s statutory rurality definition because the study is situated within a state-specific policy and healthcare delivery context. Unlike federal classifications (e.g., USDA), which define rurality as “non-urban,” Pennsylvania’s definition operationalizes rurality directly and is the basis for state health, workforce, and funding decisions. Using this framework therefore aligns the analysis with how rural populations are served in practice. According to the 2020 Census, the population density of Pennsylvania is 291 people per square mile. The Center for Rural Pennsylvania defines a county as rural if the population density of that county is fewer than 291 people per square mile. Participants were asked to provide their zip code in the demographic part of the survey. The zip codes were then mapped onto their corresponding counties. A separate rural vs. urban variable was then created based on county.
Human participant protections
This survey study received an IRB exempt determination. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. A summary explanation of research was provided to participants at the beginning of the survey. Survey completion was taken as implied consent to participate.
Statistical analyses
Descriptive statistics were calculated for demographic variables like age, race, and biological sex of the participants. The primary outcome was a binary (Yes/No) variable that indicated a participant’s past participation in research while the primary independent variables were binary (Yes/No) responses related to the different motivators and barriers to research participation. Chi-squared tests of independence were used to explore the relationship between the primary outcome variable and each of the primary independent variables. The results were only considered reliable when the chi-squared test assumptions were fulfilled (i.e., expected cell size > 5). Statistical significance was evaluated at α = 0.05.
Results
Descriptives of sample
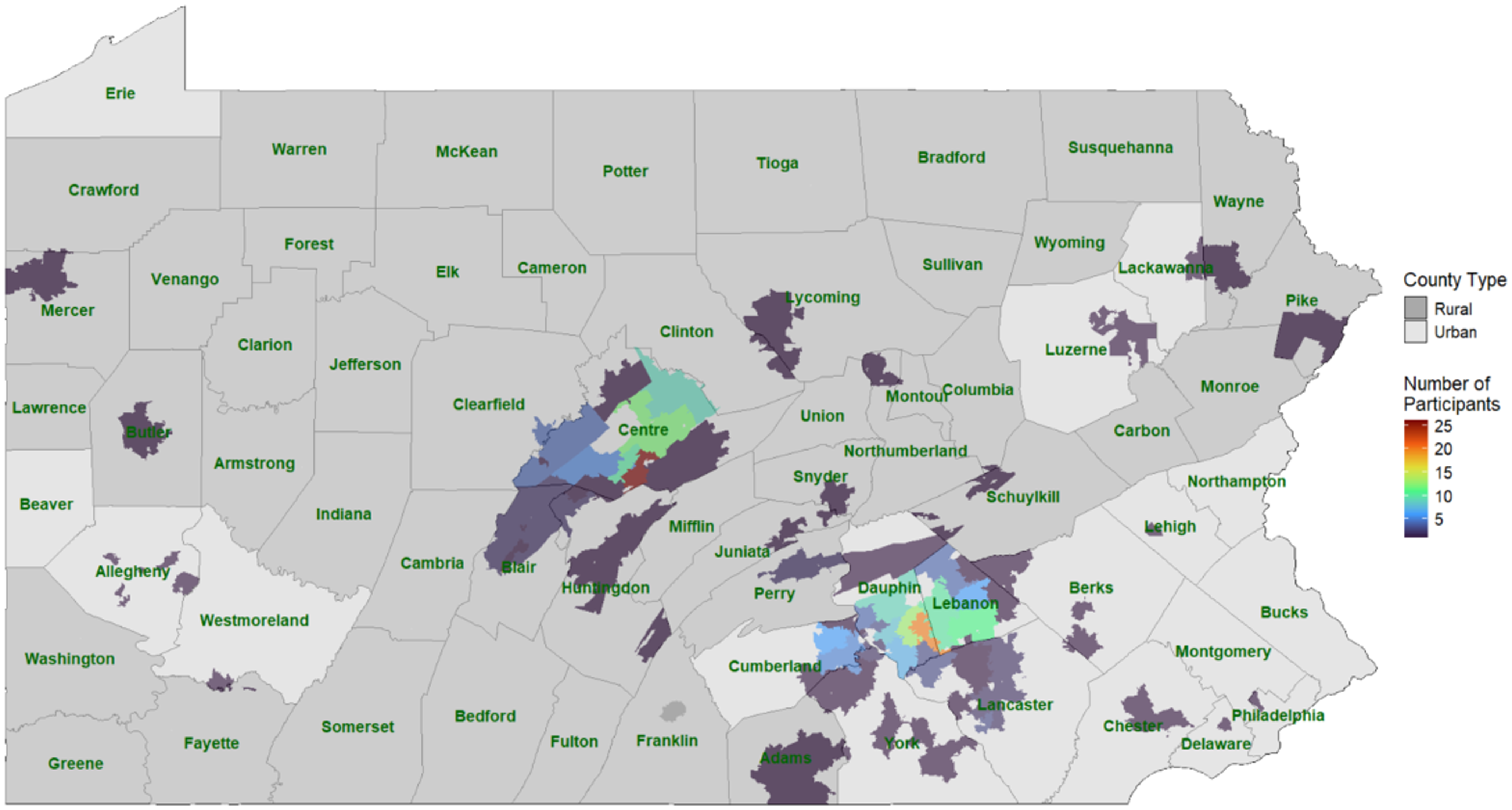
Out of 345 completed surveys, 284 reported residing in Pennsylvania and answered the question about prior participation in research. All analyses were performed using data from these 284 participants. Most of the participants were White (81.3%), and 74.6% of participants reported their legal sex at birth to be female (See Table 2). Most (63%) of participants lived in urban areas in Pennsylvania (see Figure 1, Pennsylvania map with locations of study participants).
Survey respondent spatial distribution in commonwealth of Pennsylvania. Heat map density by number of participants in zipcode. Rural urban counties are depicted as darker gray, urban counties lighter grey.

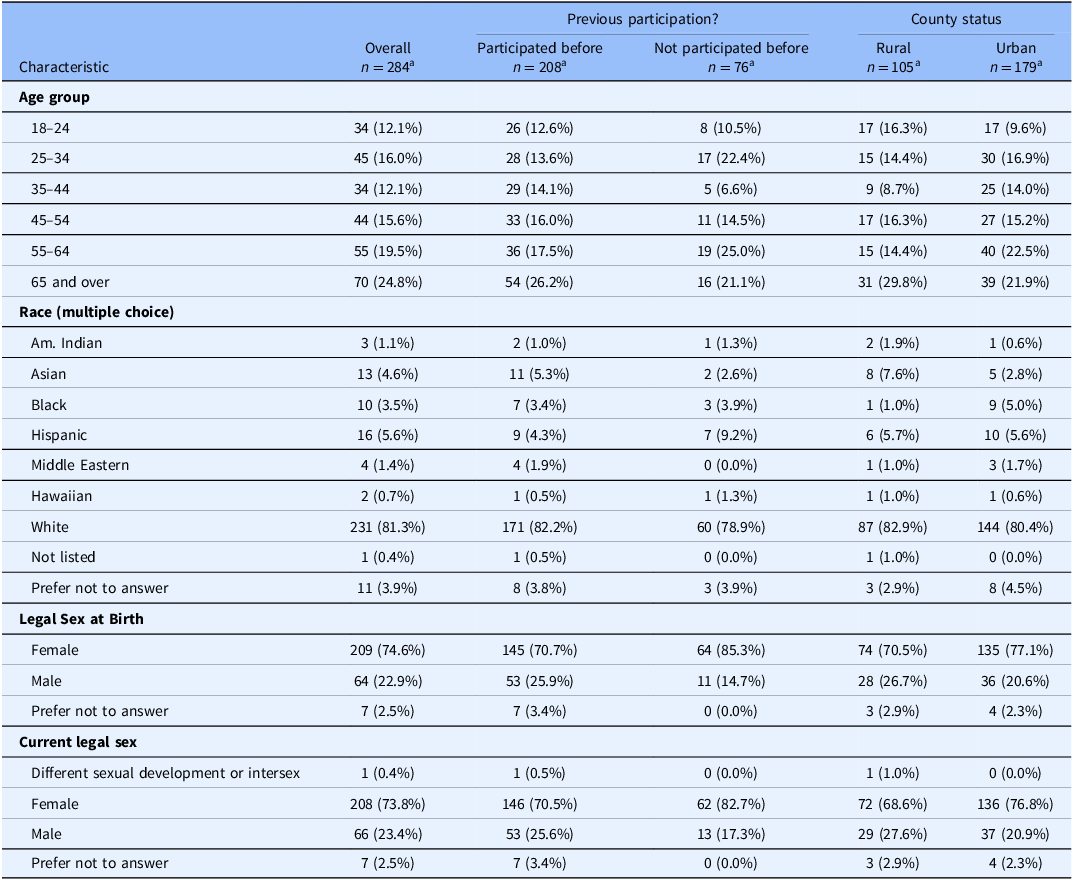
Demographics of the survey participants from Pennsylvania

Note: Out of 286 Pennsylvania participants, 2 did not answer the “Participate” question. Therefore, the following tables only includes 284 participants.
a n (%).
Comparisons by prior participation
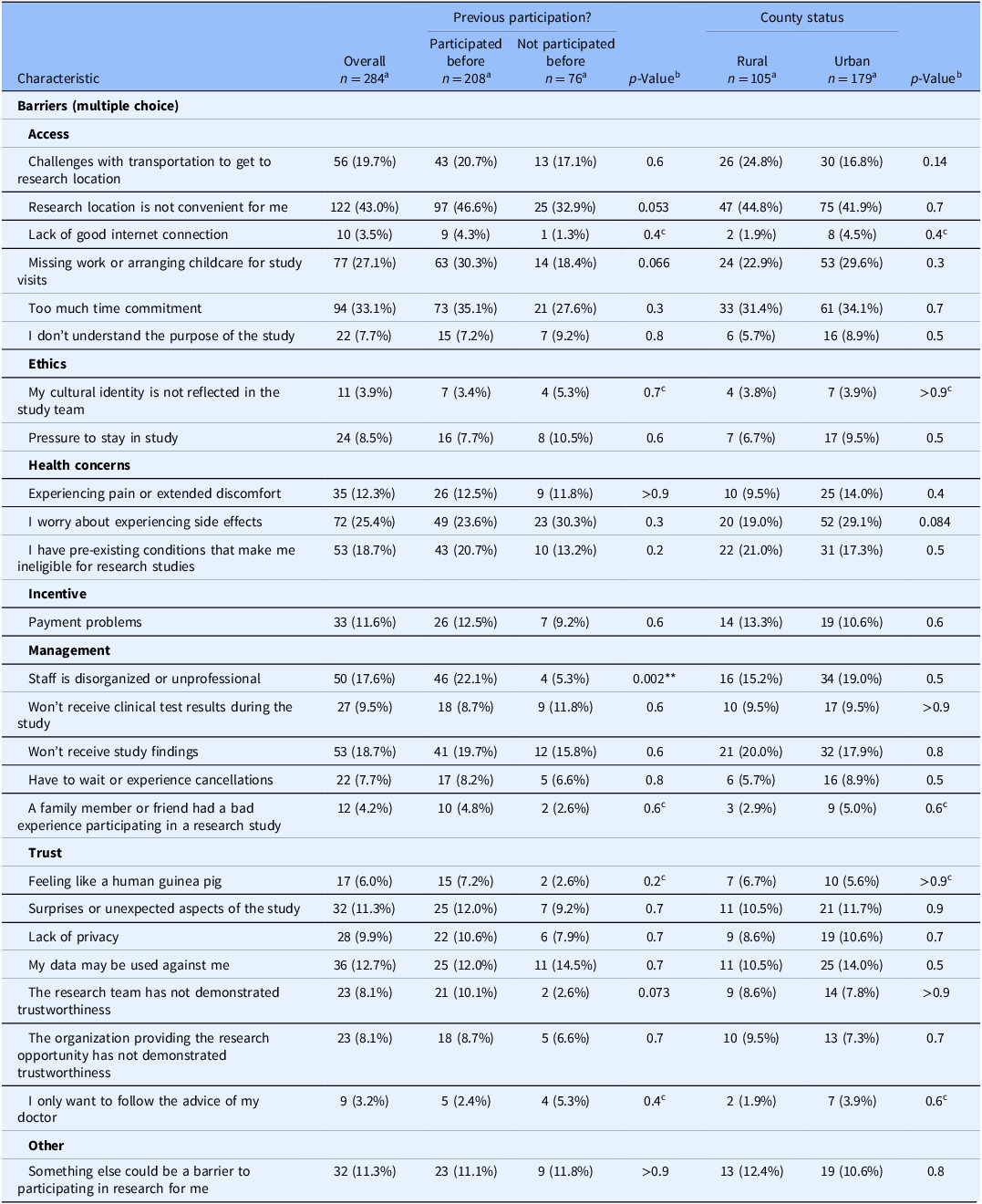
Overall, 208 participants reported prior participation in research while 76 reported having never participated before. Group comparisons of motivators are shown in Table 3. Overall, participants with prior research participation experience were more likely to report a commitment to volunteering as a motivator. Participants without prior participation were more likely to endorse “free healthcare,” “study topic is relevant to me or my loved ones,” and “getting to know the research team better” as motivators. “It’s the only option left to help my condition” was also more likely to be endorsed by participants without any prior research participation, but the data did not meet the requirements for a reliable analysis, so we cannot be confident in this result. The comparisons of reported barriers are shown in Table 4. Overall, participants with prior research participation were more likely to report “staff is disorganized or unprofessional” as a barrier. There were no significant differences between the two groups for any other barrier to research participation. However, there were trends suggesting that participants with prior participation might be more likely to consider “inconvenient research location,” “missing work,” and “research team’s trustworthiness” as barriers. See Figure 2, left panels.
Motivators for participation in research

a n (%).
b Pearson’s Chi-squared test.
c Chi-squared approximation may be incorrect because of low expected cell size.
p < 0.05; **p < 0.01; ***p < 0.001.
Barriers to participation in research

a n (%).
b Pearson’s Chi-squared test.
c Chi-squared approximation may be incorrect because of low expected cell size.
p < 0.05; **p < 0.01; ***p < 0.001.
Motivators and Barriers to research participation. (A) Motivators and (B) Barriers to research participation, comparison by research participation history. (C) Motivators and (D) Barriers to research participation, comparison by rurality.

Comparisons by rurality
Group comparisons of motivators and barriers by rurality are shown in Tables 3 and 4, respectively. Rural participants were more likely to endorse “It’s the only option left to help my condition” as a motivator. Aside from this, there were no significant differences between the proportion of rural and urban participants who endorsed any of the other motivators or barriers. However, there were trends suggesting that urban participants might be more likely to endorse “worry about experiencing side effects” as a barrier. See Figure 2, right panels.
Discussion
The unique constellation of challenges, motivators, and barriers faced by rural participants necessitates that their lived realities be made central in any study design involving these communities. We conducted a quantitative survey of motivators and barriers, extracting key findings from prior research participant studies, and adapted to a rural context. Our goal was to surface distinct perspectives from both prior research participants and non-research participants, particularly in rural communities often underrepresented in research engagement. This 33-item REDCap survey included five motivation themes: contribution, incentive, personal relevance, relationships, and trust; and six barrier themes: access, incentive, ethics, health concerns, management, and distrust of site or team. Despite the anticipated skew toward urban participants (63%), the study secured substantial rural representation (37%). Adults who were older than fifty-five (44.0%), white (81.3%), and who identified as female (74.6%) comprised most of the sample. Of interest, women were even more heavily represented in the research participant group (70.7%).
Regardless of prior research participation or rural setting, the top six motivators to research participation included a willingness to “contribute to knowledge and medicine” (74%), to “help others” (67%), “because the research was a personally important topic” (63%), to “make a difference” (62%), “financial compensation” (60%), and “working with a trustworthy organization” (50%). Participants’ motivations support previous work suggesting that altruism[Reference Kost, Lee, Yessis, Coller and Henderson5,Reference Boyd, Sternke, Tite and Morgan19,Reference Burt, Dhillon and Sharma22,Reference Shaver, Nikita and Banks23] and willingness to contribute to meaningful causes[Reference Kraft, Cho and Gillespie4,Reference Boyd, Sternke, Tite and Morgan19,Reference Bergmann, Matzneller and Weber21] are key facilitators to research participation. Furthermore, personal relevance[Reference Boyd, Sternke, Tite and Morgan19,Reference Shaver, Nikita and Banks23] and a trustworthy organization[Reference Bergmann, Matzneller and Weber21,Reference Shaver, Nikita and Banks23] have both been shown to increase research participation. This is particularly true when studies align with participants’ own experiences or their communities’ needs[Reference Kraft, Cho and Gillespie4,Reference Boyd, Sternke, Tite and Morgan19] and are conducted by researchers and organizations perceived to be credible and trustworthy [Reference Bergmann, Matzneller and Weber21,Reference Shaver, Nikita and Banks23].
Despite the similarities in the top six motivators to participate in research, some differences based on prior research experience emerged. Participants with prior research experience were more likely to endorse volunteerism as a motivator than their non-research-participating counterparts (42% and 26%, respectively). In contrast, participants without prior research experience were more likely to endorse the following four motivators than those with prior research experience: (1) the study topic is “relevant to me and my loved ones” (78% and 57%, respectively); (2) “free healthcare” (30% and 13%); (3) the opportunity to “get to know the research team better” (22% and 10%); and (4) “it’s the only option left to help my condition” (13% and 2%, a small cell size limiting confidence in the chi-square test). However, for the last motivator, the chi-square assumption of expected cell size greater than 5 was not met when comparing participants with and without prior research experience, limiting confidence in that specific result. This assumption was met when comparing rural and urban participants.
Our findings align with prior research identifying common participant burdens as major barriers to participating in research regardless of prior research experience. An inconvenient research site location emerged as one of the most reported barriers. Several studies have identified research site location as a barrier, often reflecting transportation issues such as limited access to reliable transportation [Reference Cartmell, Bonilha, Simpson, Ford, Bryant and Alberg3,Reference Boyd, Sternke, Tite and Morgan19,Reference Tanner, Kim, Friedman, Foster and Bergeron29] and inability to afford travel costs [Reference Cartmell, Bonilha, Simpson, Ford, Bryant and Alberg3]. Time constraints and inflexible scheduling also emerged, supporting earlier studies that identified time commitment [Reference Cartmell, Bonilha, Simpson, Ford, Bryant and Alberg3,Reference Friedman, Foster, Bergeron, Tanner and Kim30,Reference McKinney, Bell and Samborski24], work-related conflicts[Reference Cartmell, Bonilha, Simpson, Ford, Bryant and Alberg3,Reference Byrne, Tannenbaum, Glück, Hurley and Antoni31,Reference McKinney, Bell and Samborski24], and other competing commitments such as a lack of child and elderly care[Reference Byrne, Tannenbaum, Glück, Hurley and Antoni31] as significant barriers to research participation. Further, concern about side effects was an endorsed barrier, which may emphasize participants’ ongoing worries about the perceived risks and safety of participating in research [Reference Cartmell, Bonilha, Simpson, Ford, Bryant and Alberg3,Reference Kost, Lee, Yessis, Coller and Henderson5,Reference Cartmell, Bonilha, Simpson, Ford, Bryant and Alberg3,Reference Byrne, Tannenbaum, Glück, Hurley and Antoni31]. “Disorganized and unprofessional staff” was one of the most frequently reported barriers by prior research participants, amplifying previous findings that associate this factor with negative research experiences [Reference Kost, Lee, Yessis, Coller and Henderson5]. Overall, our findings emphasize barriers that continue to limit research participation across a range of populations and study contexts.
Differences in overall motivator and barrier patterns across municipality types were insignificant, with rural respondents indicating slightly more trust in research teams and site than urban respondents (38% and 34%, respectively). Rural respondents also were more concerned with the trustworthiness of the research organization than their urban counterparts (53.7% and 45.7%). Respondents without prior participation were more likely to endorse motivators to research participation, including “free healthcare,” “study topic is relevant to me or my loved ones,” and “getting to know the research team better.” Previous work has identified positive relationships with research staff and financial compensation as key factors in continued research participation [Reference Kost, Lee, Yessis, Coller and Henderson5]. Respondents without prior participation may view these identified motivators as anticipated benefits based on their own perceptions of research involvement. Respondents with prior research participation were more likely to report “staff is disorganized or unprofessional” as a barrier; other barriers across cohorts were not significantly different. Urban respondents were more sensitive to convenience, time burden, and financial incentives. These barriers contrast with those from a prior qualitative study of 212 rural and urban participants in a southeastern state [Reference Friedman, Foster, Bergeron, Tanner and Kim30], which found both rural and urban groups endorsed fear, side effects, lack of understanding of clinical trials, time commitment, and distrust as barriers. These mixed findings highlight the need for future research to better understand how barriers to research participation intersect with the geographic location and the demographic composition of the sample.
While there were no significant differences based on rurality, there were trends suggesting that urban participants might be more likely to endorse “worry about experiencing side effects” as a barrier. Prior work highlights that individuals from rural populations experience significant barriers to research participation[Reference Bergmann, Matzneller and Weber21,Reference Bergeron, Foster, Friedman, Tanner and Kim32]. Our findings serve as an avenue for future work to inform the development of targeted strategies to improve research engagement, particularly among those who have not participated in research in rural populations.
Limitations
This study has several limitations. The cross-sectional design and snowball recruitment methods limit the representativeness of the sample and causal inferences that can be drawn. Future studies of rural Pennsylvanians could employ systematic sampling methods that cover more or randomly selected regions/counties to allow for weighted estimations of motivators and barriers to research. It would have been interesting to look at the comparisons between motivators/barriers within subgroups of participants split by rural/urban and previous participation/no prior participation in research. However, the small sample size of rural participants without prior research participation limited our ability to make reliable comparisons within these subgroups. Although informed by qualitative studies, quantitative surveys such as ours are limited in improving understanding on how to address identified research barriers, warranting additional qualitative studies. Future studies could include ratings of the strength of agreement with the motivators/barriers in order to identify the most impactful direction for interventions.
Conclusion
Enhancing the research experience for rural communities – particularly through strategies that reflect their needs and concerns – is essential to improving participation in research. A positive research experience can also have an outsized impact on participant engagement in future studies.
Author contributions
Jennifer B. McCormick: Conceptualization, Methodology, Writing-review & editing; Jennifer Mary Poger: Conceptualization, Project administration, Writing-original draft, Writing-review & editing; Sandesh Bhandari: Formal analysis, Visualization, Writing-original draft, Writing-review & editing; Courtney Whetzel: Supervision, Writing-review & editing; Angel Collie: Visualization, Writing-original draft, Writing-review & editing; Alyson G. Eggleston: Conceptualization, Writing-original draft, Writing-review & editing; Jennifer L. Kraschnewski: Conceptualization, Funding acquisition, Project administration, Supervision, Writing-review & editing; Orfeu M. Buxton: Supervision, Writing-original draft, Writing-review & editing.
Funding statement
The project described was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through grant UL1-TR002014. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Competing interests
None. Outside of the current work, OMB discloses that he received subcontract grants to Penn State from Mobile Sleep Technologies (NIH/NIA SBIR R43-AG056250, R44-AG056250); received honoraria/travel support for lectures/mentoring from Tufts School of Dental Medicine, University of Utah, University of Arizona, Spencer Study Club, and University of Miami; consulting fees from Georgia State University, National Institute on Aging, and Harvard Chan School of Public Health; and receives an honorarium for his role as the Editor in Chief of Sleep Health sleephealthjournal.org.
Use of AI
No generative AI or other AI methodologies were used in this manuscript.