Introduction
Shared decision-making is a joint process in which a healthcare professional works together with an individual to reach a decision about a treatment (National Institute for Health and Care Excellence – NICE 2021). It is one of the key components of person-centred care and involves a discussion in which the individual shares their preferences, beliefs, and values while the clinician shares the treatment options available including risks and benefits (NICE 2021; Grünwald & Thompson Reference Grünwald and Thompson2021). While shared decision-making is recommended at policy level and desired by individuals choosing antipsychotic medicines, its implementation remains limited in those with psychotic illness (Haugom et al. Reference Haugom, Benth, Stensrud, Ruud, Clausen and Landheim2023; Müller et al. Reference Müller, Schuster, Rodolico, Siafis, Leucht and Hamann2023). Although there is ongoing work on identifying and addressing barriers to the implementation of shared decision-making in this group (Haugom et al. Reference Haugom, Benth, Stensrud, Ruud, Clausen and Landheim2023; Siafis et al. Reference Siafis, Bursch, Müller, Schmid, Schuster, Waibel, Huynh, Matthes, Rodolico, Brieger, Bühner, Heres, Leucht and Hamann2022; Zisman-Ilani et al. Reference Zisman-Ilani, Roth and Mistler2021), it is important not to overlook the patient information resources required to support the process. The most used information resources for antipsychotic medicines are patient information leaflets, which are usually supplied by the manufacturer. However, there are several limitations and potential biases associated with these resources that can impede the shared decision-making process.
It is well established that the manufacturers’ patient information leaflets fail to fulfil the criteria expected for effective communication of the potential benefits and harms of medicines (The Academy of Medical Sciences 2017). They have an emphasis on the risks and side-effects of the medicine with little by way of treatment benefits (Hynes-Ryan et al. Reference Hynes-Ryan, Kelleher and Kathryn2025). Additionally, numerous studies have demonstrated that the reading level of these patient information leaflets often exceeds the reading skills of many adults (Brega et al. Reference Brega, Freedman, LeBlanc, Barnard, Mabachi, Cifuentes, Albright, Weiss, Brach and West2015). This holds true for the patient information leaflets for antipsychotic medicines which are ranked as ‘difficult’ to read according to readability tests (Hynes-Ryan et al. Reference Hynes-Ryan, Kelleher and Kathryn2025). They are also lengthy, generally 3,000–5,000 words and thus taking approximately 17 mins for an adult to read (Hynes-Ryan et al. Reference Hynes-Ryan, Kelleher and Kathryn2025). Individuals engaging effectively in shared decision-making using these resources require a high level of health literacy.
Health literacy is the ability to use general literacy skills (reading, writing, numeracy, listening, and speaking) to obtain, understand, appraise, synthesise, communicate, and apply health-related information (Liu et al. Reference Liu, Wang, Liu, Jiang, Wang, Chen, Ju and Zhang2020). Forty per cent of adults in Ireland have limited health literacy and those with severe mental illness experience lower health literacy than the general population (Murray et al. Reference Murray, Smith, Behan and Goss2024; Clausen et al. Reference Clausen, Watanabe-Galloway, Bill Baerentzen and Britigan2016). This is especially true for those experiencing psychotic illness (Clausen et al. Reference Clausen, Watanabe-Galloway, Bill Baerentzen and Britigan2016; Thomson et al. Reference Thomson, Galletly, Prener, Garverich, Liu and Lincoln2022). As those participating in shared decision-making require accurate, accessible, and usable information about the potential benefits and harms of medicines, existing antipsychotic medicines information resources require improvement (The Academy of Medical Sciences 2017).
We know that those seeking mental health information resources explore different avenues to meet their needs and to facilitate decision-making, and that they tend to prefer the internet for ease of access (Grabb Reference Grabb2023; Bergmo et al. Reference Bergmo, Sandsdalen, Manskow, Småbrekke and Waaseth2023; Pate et al. Reference Pate, Heathcote, Simons, Leake and Moseley2020). We also know that search results on platforms like Google®, YouTube®, and TikTok® can often be misleading, inaccurate or provide information on medicines that are unavailable (Osman et al. Reference Osman, Mohamed, Elhassan and Shoufan2022; Nour et al. Reference Nour, Nour, Tsatalou and Barrera2017). We therefore identified a need for accurate and accessible digital resources on antipsychotic medicines to empower those involved in shared decision-making to make an informed choice. The provision of understandable, clear, and relevant information is fundamental to the shared decision-making process.
To address this problem, we adopted Design Thinking, a human-centred approach to innovation. (Brown Reference Brown2008). Design Thinking prioritises engaging with service users to better understand their needs and has been increasingly studied in health services research (Flood et al. Reference Flood, Ennis, Ludlow, Sweeney, Holton, Morgan, Clarke, Carroll, Mellon, Boland, Mohamed, De Brún, Hanratty and Moriarty2021; Kim et al. Reference Kim, Myers and Allen2017). It is an iterative, non-linear process that considers both technology and available resources to ensure solutions generated to address problems are both desirable and feasible (Flood et al. Reference Flood, Ennis, Ludlow, Sweeney, Holton, Morgan, Clarke, Carroll, Mellon, Boland, Mohamed, De Brún, Hanratty and Moriarty2021). In this study, we aimed to develop accessible and reliable antipsychotic medicines information resources to support the share decision-making process using a Design Thinking approach.
Methods
We adopted the Double Diamond Design Thinking Model to co-design a suite of antipsychotic medicines information videos. The Double Diamond Model is a design process framework that highlights the importance of translating a need into a statement of challenge before developing solutions (Jun et al. Reference Jun, Hignett and Clarkson2024). The model identifies four core activities – Discover, Define, Develop, Deliver – that involve divergent and convergent thinking to explore the full range of stakeholder needs for improvement before refining them into a clear problem statement and providing the best solution (Jun et al. Reference Jun, Hignett and Clarkson2024). Co-design methodologies have been increasingly incorporated into mainstream research frameworks as principles of inclusion, empowerment, and impact and have been promoted by research and funding institutions (Jun et al. Reference Jun, Hignett and Clarkson2024). The epistemology is constructivist, with knowledge and understanding built in a pragmatic, participatory way valuing the tacit knowledge of the service user as the expert (Flood et al. Reference Flood, Gleeson, Flynn, Ennis, Ludlow, Sweeney, Holton, Morgan, Clarke, Carroll, Mellon, Boland, Mohamed, De Brún, Hanratty, Kelly, Moriarty, Desselle, García–Cárdenas, Anderson, Aslani, Chen and Chen2022). Our study operationalised the Double Diamond framework as a mixed-methods, participatory design process (Flood et al. Reference Flood, Gleeson, Flynn, Ennis, Ludlow, Sweeney, Holton, Morgan, Clarke, Carroll, Mellon, Boland, Mohamed, De Brún, Hanratty, Kelly, Moriarty, Desselle, García–Cárdenas, Anderson, Aslani, Chen and Chen2022). Quantitative data were generated in the Discover phase through structured assessments of existing written and digital medicines information resources. Qualitative data were collected through content review of existing resources and open-ended feedback from a multidisciplinary expert panel and experts-by-experience during iterative prototyping. Quantitative scores were summarised descriptively to identify gaps in accessibility and usability, while qualitative feedback was collated, reviewed by the research team, and incorporated through consensus into revisions of scripts and videos until final sign-off was achieved.
Expert panel of key stakeholders and reflexivity
This study was conducted within a constructivist, participatory framework. The first author – an advanced specialist mental health pharmacist – has a clinical, academic, and health literacy background which shaped the initial identification of the problem. The study focussed on accuracy, safety, and accessibility of antipsychotic medicines information and the information needs of service users. The study was conceptualised with three specialist mental health pharmacists, one consultant psychiatrist and two adult experts-by-experience. These participants found that the medicines information resources that support shared decision-making were insufficient. The research team also included a second consultant psychiatrist and two mental health nurses with experience in supporting those taking antipsychotic medicines and research. We acknowledge that the perspectives of the clinical stakeholders carried implicit assumptions about information needs and priorities, reflexivity was embedded through the co-design approach. Cycles of iterative prototyping and review enabled participants to reshape and adjust the emerging output. This ensured that lived experience was treated as an equal form of expertise.
Discover – explore current needs and solutions, and gather insights into the challenge
The Discover phase of the Double Diamond model is the first divergent thinking phase. It involves exploring current needs and solutions to better understand the problem. Our first step was to review existing antipsychotic medicines information resources and rate their readability, understandability, and actionability. Readability refers to how easy a text is to read based on factors like sentence length, word difficulty, and structure. Understandability goes beyond just reading words as it measures how well the reader can grasp the meaning of the text. Actionability is a measure of what the reader can do based on the information presented. During the Discover phase, we also looked at health literacy guidelines and toolkits to identify standards to which patient information resources should adhere. Quantitative and qualitative data were collected in this phase to identify gaps in the accessibility, understandability, and actionability of existing antipsychotic medicines information resources. Quantitative data comprised PEMAT and readability scores, while qualitative data consisted of structured observations on content omissions, language, tone, and usability across written and video resources.
Patient information leaflet assessment
We examined data on the most commonly prescribed and dispensed antipsychotic medicines in Ireland over the last five years and compiled our own list of six medicines. This list was then reviewed and approved by our expert panel. We collectively assessed the understandability and actionability of the existing information resources for these medicines (the manufacturers’ patient information leaflets) using the PEMAT (Shoemaker et al. Reference Shoemaker, Wolf and Brach2014). The PEMAT is a systematic method for health providers/librarians to evaluate and compare the understandability and actionability of patient education materials. It can assess both printable (PEMAT-P) and audiovisual (PEMAT-A/V) materials. For materials to be understandable and actionable, thus supporting shared decision-making, individuals with diverse backgrounds and varying levels of health literacy should be able to process the content, explain key messages and identify what they can do based on the information presented (Shoemaker et al. Reference Shoemaker, Wolf and Brach2014). The higher the score, the more understandable and/or actionable the material. PEMAT-P item results were added and converted into percentage scores for understandability and actionability for each leaflet. These quantitative results were tabulated and compared across medicines to identify weaknesses in existing resources that limit the shared decision-making process. According to the authors of the PEMAT, it does not assess accuracy or comprehensiveness or perform readability tests. As a result, additional assessments are recommended to ensure the content is clinically correct, comprehensive, and readable. Manufacturers’ patient information leaflets are regulatory documents and therefore have a high degree of inherent accuracy. The readability of the manufacturer’s patient information leaflets for antipsychotic medicines has been assessed and classified as ‘difficult’ to read (Hynes-Ryan et al. Reference Hynes-Ryan, Kelleher and Kathryn2025). Thus, we did not repeat this assessment. We did, however, review the comprehensiveness of each leaflet in line with the questions in the Satisfaction with Information About Medicines Scale (SIMS) (Horne et al. Reference Horne, Hankins and Jenkins2001).
Internet and digital resource search
As those seeking information on antipsychotic medicines may also be exploring online resources given their ease of access, our next step was to review antipsychotic medicines information available online to determine the readability (using the Flesch–Kincaid Reading Ease Score) (Flesch Reference Flesch1948), understandability, and actionability, as well as the accuracy of content. This review examined both written and video information resources from the most used search engines and video platforms. We searched each engine/platform using the generic medicine name before carrying out our review and assessment on the most popular results. To ensure the search results were not biased towards the search history of the lead author, these medicine key word searches were carried out by three independent non-healthcare professionals. For each written and video-based resource identified, quantitative data were assessed using the Flesch–Kincaid Reading Ease Score and the PEMAT-P or PEMAT-A/V, as appropriate. These scores were tabulated and presented comparatively in to illustrate differences in readability, understandability, and actionability across platforms. Qualitative observations regarding content gaps, visual support, and action-oriented guidance were recorded to inform subsequent script development.
Health literacy guideline review
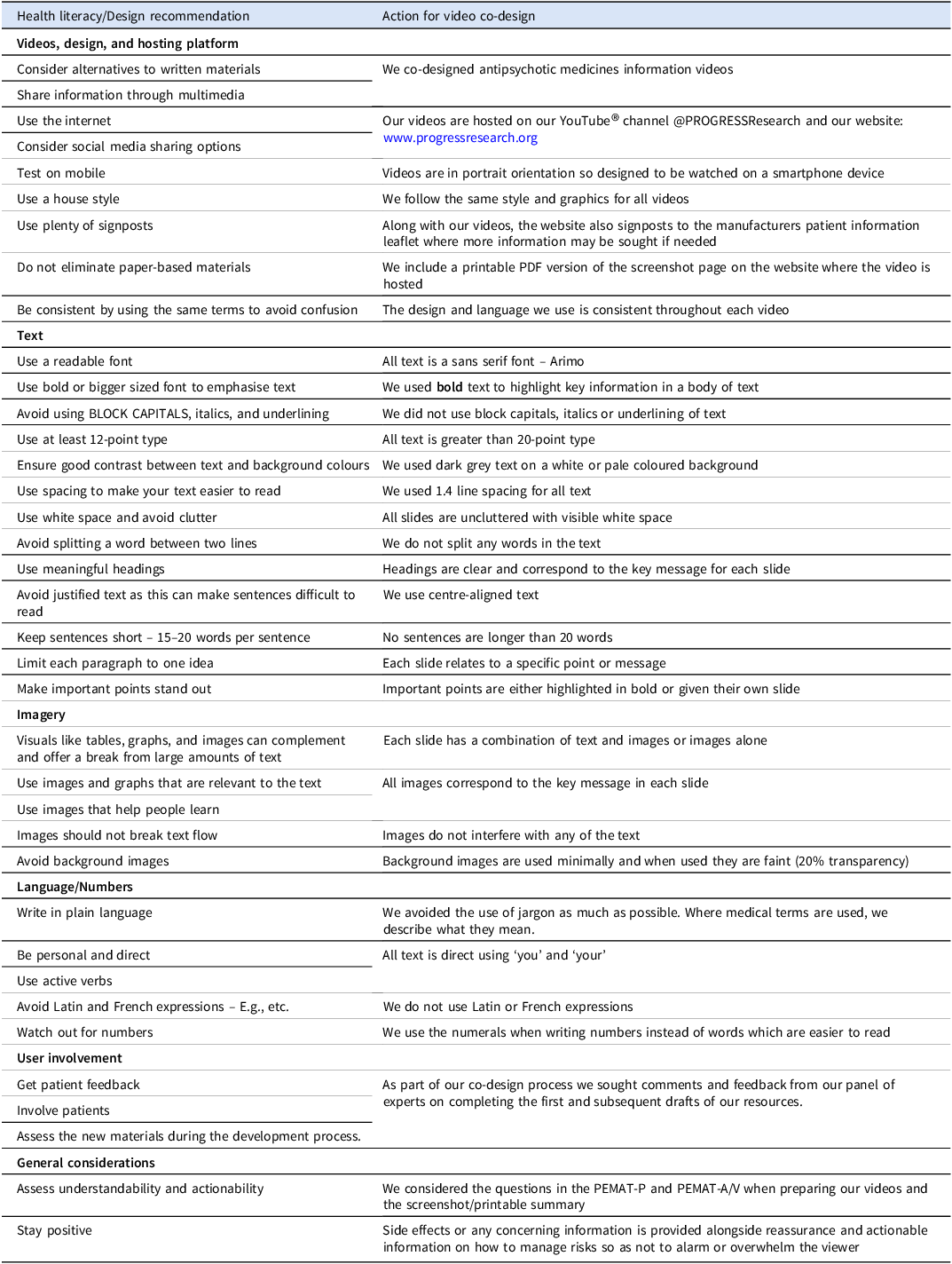
The final step of our Discover phase involved exploring the health literacy standards to which patient information materials should align. We reviewed national and international toolkits and guidelines on producing written and digital materials that are accessible and understandable which were recommended by a group of Health Literacy experts. Using these toolkits/guidelines we compiled our own list of standards to which we would align our co-designed antipsychotic medicines information resources.
Define – balance the range of needs and articulate a clear statement of the challenge to be resolved
In the Define phase of the Double Diamond Design Thinking model, the insights gathered in the Discover phase are reviewed and refined, and a clear statement of the problem to be addressed is outlined. Quantitative assessment results and qualitative observations from the Discover phase were collated and synthesised to define the challenge at hand, which then guided content scope and design priorities for the co-designed videos.
Develop – generate and evaluate solutions to the challenge
In the Develop phase, we diverged again to create a solution to our problem by integrating qualitative stakeholder feedback into successive prototypes through structured iteration cycles, with all feedback documented, reviewed, and incorporated through consensus.
First draft of antipsychotic information resources
Using clinically accurate information from manufacturers’ patient information leaflets and other digital resources, along with the clinical expertise of our expert panel, we drafted a script for each of the six antipsychotic medicines. The content and structure of the scripts were informed by the qualitative observations from our review of existing written and video-based medicines information resources, including identified omissions (e.g. lack of symptom descriptions, time to effect, and actionable guidance), language complexity, limited visual support, and poor actionability. These qualitative findings were used to prioritise key messages, plain language, and the inclusion of clear, user-centred information to support shared decision-making. The content was further guided by a previously co-designed medicines information resource for young people (www.youthmed.info) (Hynes-Ryan et al. Reference Hynes-Ryan, Kelleher and Kathryn2025). We then produced prototype antipsychotic medicines information videos using the online design platform www.canva.com and recorded the voiceover for each video on a smartphone device. Each key message from the scripts corresponded to a slide in the videos with associated graphics. We selected pictogram images with a consistent style and colour throughout.
Expert panel of key stakeholder review and iteration
Following completion of the initial drafts of the information videos for all six antipsychotic medicines, we sought feedback and comments from our group of key stakeholders. We sent the videos to each member of the expert panel along with a list of survey-style question which were open-ended and panel members were invited to include any other comments or feedback in their replies (Table 1). As this was an iterative review process with seven clinicians and two experts-by-experience, all feedback and comments were considered collectively to ensure equal weight was attributed to all input.
Survey questions for expert panel

Data analysis
All qualitative feedback from the expert panel was collated into a single table and reviewed independently by the specialist mental health pharmacists. Through team discussion and consensus, feedback was categorised into actionable revisions or deferred content for future videos. Quantitative assessment findings from the Discover phase were used to guide design standards, content, and formatting decisions. Subsequent drafts of the videos were sent to all members of the expert panel for a final sign-off. Non-adopted recommendations were discussed with the relevant expert panel member and alternative actions were agreed.
Deliver – launch the new organisation, product, or service
The Deliver phase of the Double Diamond Design Thinking Model involves the provision of the solution. It is where the results of the initial three phases come together, and the new resource can be launched. For this final phase, we determined the most appropriate hosting platform for our resources and planned how we could best ensure broad availability to support the shared decision-making process.
Results
Discover
Patient information leaflet assessment – PEMAT
Aripiprazole, clozapine, olanzapine, paliperidone, quetiapine, and risperidone were the six antipsychotic medicines we chose based on prescribing and dispensing data and expert panel review (Vaughan et al. Reference Vaughan, Lucey and Sahm2024; Hynes et al. Reference Hynes, McWilliams, Clarke, Fitzgerald, Feeney, Taylor, Boland and Keating2020; Keating et al. Reference Keating, McWilliams, Boland, Doyle, Behan, Strawbridge and Clarke2021). The manufacturers’ patient information leaflets for each of these six medicines are in a standardised format as detailed in Supplement 1. While the content of these leaflets is clinically accurate, they lack some important information relevant to shared decision-making. For example, they do not contain any descriptions of the symptoms that medicines target beyond their licensed indications (schizophrenia, bipolar disorder, mania, etc.). Moreover, they do not describe how the medicines work, when to take them, time to effect, duration of treatment, physical health monitoring requirements, reassurance around the time-limited nature of side-effects and support available for persistent/distressing side-effects or any summary of all this information. The readability of these leaflets is categorised as ‘difficult’ (Hynes-Ryan et al. Reference Hynes-Ryan, Kelleher and Kathryn2025), and the results of the PEMAT-P indicate the understandability and actionability of the manufacturer’s patient information leaflets is low (31 and 40%, respectively) (Fig. 1).
Readability, understandability, and actionability of medicines information resources from manufacturer, Google®, ChatGPT® and YouTube®.

Internet and digital resource search and assessment
The five most used search engines and video platforms as of January 2025 are Google®, YouTube®, Facebook®, Instagram® and ChatGPT® (Similar Web 2025). The average click-through-rate (a metric of how often people click on a link after seeing it) for the first position of results following a search in Google® is up to 40%, with the second- and third-ranking results getting up to 20 and 10%, respectively (SISTRIX Content Team 2021; Dean Reference Dean2019). We therefore only reviewed the content on the first page of results for each Google® search performed for each antipsychotic medicine and reviewed the top five videos returned following our YouTube® search. Results of our Google® search review are available in Supplement 1. While the resources following the medicine key word search were mostly reliable (government bodies, academic/medical centres), the content was similar to that in the manufacturers’ patient information leaflets with the exception of the NHS.UK medicines information which included details of how the medicines work, symptoms they help with and time to effect. However, the NHS.UK resource did not have a page for paliperidone, clozapine or many other commonly prescribed second-generation antipsychotic medicines. All resources identified through our Google® search were categorised as ‘difficult’ or ‘very difficult’ to read and all but one (NHS.UK) did not score well on the PEMAT-P (understandability ranged from 14–71% and actionability ranged from 0–60%).
On searching ChatGPT® for information on each of our antipsychotic medicines, we noted the results scored moderately for understandability (50%) but very low for actionability (0%) and had a readability score of ‘very difficult’ (see Fig. 1).
YouTube® has the capacity to highlight those results that are ‘From Health Sources’, and these are displayed in a separate tab. We identified five YouTube® profiles that created video content on antipsychotic medicines (Supplement 1), two of whom were nurses, one was a pharmacist and two were psychiatrists. Videos were 3–15 minutes long and, while the information discussed by each clinician was clinically accurate, according to the PEMAT-A/V, the videos had similar understandability and actionability scores to the written information we reviewed on Google® and ChatGPT® (understandability ranged from 21 to 53% and actionability was 0%). In these videos, clinicians spoke directly to the camera and used little or no imagery to support their verbal communication.
The video content we reviewed on Facebook® and Instagram® was quite variable. Some were clinician-led videos where psychiatrists, pharmacists or nurses discussed the medicines and outlined licensed indications and side-effects. Others were made either by those taking the medicines themselves or caring for someone taking the medicine and gave a subjective account (positive or negative) of the experience. While these videos provide a useful insight into the experience of taking antipsychotic medicines, they lacked some of the clinical details around how the medicines work, the symptoms they treat, the time to effect, and so forth. As the quality and duration of both types of video on Facebook® and Instagram® were so variable, it was not possible to assess their understandability or actionability using the PEMAT-A/V.
Health literacy guideline review
We were unable to identify any guidelines available that were specific to the co-design of accessible video materials. As a result, we used existing national and international literacy toolkits and guidelines on producing written and digital materials (HSE 2025; NALA 2024; ODPHP 2025; Brach Reference Brach2024; Centre for Excellence in Universal Design 2017). Table 2 describes these recommendations and how we adopted them in the co-design of our video resources.
Health literacy toolkit and guideline recommendations and actions

Define
Our Discover phase gave us a clear picture of the challenge those participating in shared decision-making are facing when it comes to accessible and reliable antipsychotic medicines information resources. This allowed us to define our problem – how might we collaboratively and cost-effectively develop accurate, understandable, and actionable antipsychotic medicines information resources to support the shared decision-making process.
Develop
The outcomes of our Discover and Define phases presented us with a clear objective on what we were required to do to overcome the challenge at hand, namely to co-design a suite of antipsychotic medicines information videos.
First draft of antipsychotic information resources
While the information in the manufacturers’ patient information leaflets, on Google®, ChatGPT®, and YouTube® is mostly accurate, it was clear that the content is difficult to read and understand, and the actionability could be improved. We identified key pieces of information from these resources to guide the content of our video scripts (Example in Supplement 2). As those engaging in shared decision-making involving antipsychotic medicines may not have a formal diagnosis, may be prescribed antipsychotic medicines outside the licensed indication, may lack insight into their condition or may feel stigmatised and marginalised by a mental health diagnosis (Timimi Reference Timimi2014), we avoided as much as possible using diagnoses and instead described the symptoms to which people can relate. Our first draft videos include a description of symptoms, details of how each medicine works, when to take it, the time to effect, how long it may be needed for, physical health monitoring requirements, the most common side-effects, warning signs, and what to do if they are observed, stopping the medicine, where more information may be sought, and a summary page that can be captured with a screenshot and saved on the smart device.
Expert panel feedback, comments, and recommendations
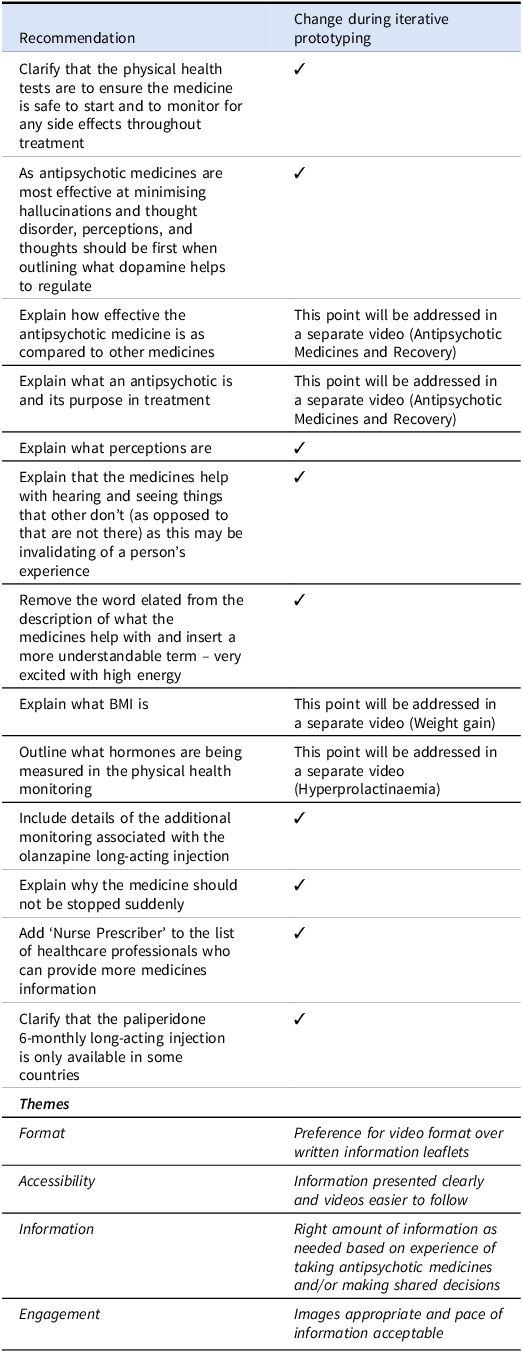
As this was an iterative co-design process, we considered all feedback and comments collectively to ensure equal weight was attributed to all input. The recommendations are outlined in Table 3 along with the changes made through iterative prototyping. The reasons that some recommendations were not accepted is because they are due to be covered in more detail in future videos.
Recommendations and emergent themes arising during iterative prototyping

Final draft sign-off
All members of the expert panel provided a final sign-off on the videos before they were made available for upload to our video sharing platform. An additional six videos including five antipsychotic medicines (brexpiprazole, cariprazine, lurasidone, asenapine, and ziprasidone) and another medicine used to prevent antipsychotic-induced weight gain (metformin) were subsequently developed. These additional videos underwent the same rigorous development and evaluation process as the initial six.
Deliver
To close out our design thinking process, we brought together the results of our discover, define, and develop phases to deliver our solution – six antipsychotic medicines information videos on aripiprazole, clozapine, olanzapine, paliperidone, quetiapine, and risperidone. We uploaded the final videos to our new YouTube® channel @PROGRESSResearch and embedded these videos on our website: www.progressresearch.org. The videos can be accessed freely here alongside a printable summary leaflet and clickable link(s) to the manufacturer’s patient information leaflet(s). This is to ensure those using the videos to support shared decision-making have access to more reliable information as needed.
Discussion
Summary
Although shared decision-making involving antipsychotic medicines is complex, there are clear opportunities to make it more achievable, particularly through the provision of accurate and accessible medicines information. Existing resources including the manufacturer’s patient information leaflets and widely used digital platforms fall short when it comes to readability, understandability, actionability, and sometimes reliability limiting their usefulness in communicating risks and benefits. Addressing this gap required an interdisciplinary, collaborative Design Thinking approach (Flood et al. Reference Flood, Ennis, Ludlow, Sweeney, Holton, Morgan, Clarke, Carroll, Mellon, Boland, Mohamed, De Brún, Hanratty and Moriarty2021). By adopting the Double Diamond Model, we reviewed existing resources to identify unmet needs, defined the central challenge of supporting shared decision-making, and developed new information videos. These were evaluated by an expert panel and subsequently launched via our YouTube® channel (@PROGRESSResearch) and website (www.progressresearch.org). As health system challenges grow in complexity, interdisciplinary, and human-centred design approaches are increasingly important to ensure solutions remain patient-centred, particularly in psychosis research given the severity and burden of psychotic disorders. Our co-design methodology prioritised the perspectives of experts-by-experience and clinicians, placing users at the centre of the research process.
Clinical implications
Research indicates that learning from video can enhance retention and comprehension compared with reading text alone (Abdalla et al. Reference Abdalla, Azzani, Rajendren, Hong, Balachandran, Hassan, Wei, Yahaya, En, Ajaykumar, Roger and Haja2021; Nassar et al. Reference Nassar, Aboalshamat, Alsanei, Alghamdi, Fudah and Alhilou2024). As a result, those who watch our antipsychotic medicines information videos instead of reading the information in leaflets or online will be better equipped to engage in meaningful discussions with their clinicians when it comes to treatment options. As well as supporting the individual, our resources will also provide information and reassurance to family or caregivers and other mental healthcare professionals who are less familiar with the risks and benefits of antipsychotic medicines.
Future directions
The use of digital resources to access health information is increasing. However, there is currently no oversight regarding the quality of video resources making misinformation difficult to identify. Our co-design project is the first step towards using digital technology to support shared decision-making in an accurate and accessible way. We plan on adding more content including videos on antipsychotic side effects (weight gain, dyslipidaemia, and hyperprolactinaemia). We are also working on shorter videos to answer frequently asked questions including one on ‘Antipsychotic medicines and recovery’. Future work will focus on evaluating the impact of our resources on the experience of those engaging in shared decision-making and carrying out a stakeholder mapping exercise to engage strategically with professional bodies, service providers and advocacy groups who will endorse our videos as trustworthy and informative. We also plan on utilising these videos in a shared decision-making tool for people experiencing psychotic illness which will draw together all the information an individual and their caregivers will need to make an informed choice.
Strengths and limitations
Our videos were not designed by a professional media company, however our experts-by-experience reported that they found the graphics and voiceover to be acceptable and relatable. We reviewed and assessed only the resources on the first page of our Google® search for readability, understandability, actionability, and reliability, and we acknowledge that there may be other resources that are more acceptable on subsequent pages. However, to our knowledge, ours are the only antipsychotic medicines information videos co-designed with key stakeholders in the shared decision-making process including pharmacists, psychiatrists, nurses, and experts-by-experience.
Our video resources require a level of digital literacy to access. For those experiencing active psychosis, this may present a challenge. This represents another limitation of their role in facilitating shared decision-making about antipsychotic medicines. However, as the resources are freely available, we would expect that access may be supported by clinicians and family members/carers to ensure those without digital literacy skills can use them appropriately.
Conclusions
Our antipsychotic medicine information videos mark a significant step towards fostering shared decision-making for those who are underserved by current resources. By embracing a human-centred and innovative Design Thinking approach, we ensured that the voices of key stakeholders – psychiatrists, pharmacists, nurses, and, most importantly, experts-by-experience – were at the heart of every phase. These resources may empower individuals on their mental health recovery journey, providing them with clear, accurate, and accessible information to make informed choices about antipsychotic medicines.
Supplement 1
Format and Content of Medicines Information Resources from Manufacturer, Google®, ChatGPT®, and YouTube®

Supplement 2
Aripiprazole Script
This is an information video about aripiprazole, also as known as Abilify.
Aripiprazole is an antipsychotic medicine that lowers the activity of dopamine in your brain.
Dopamine is a natural chemical that carries messages between your brain cells.
It helps you to regulate your perceptions (how you see, hear, or become aware of something through your senses), your thoughts, behaviour, and emotions.
Aripiprazole helps with feeling agitated and very excited with high energy, strongly believing things you usually don’t, hearing or seeing things that others don’t and having intrusive and distressing thoughts.
When you start aripiprazole, your dose may be increased so that you get the best out of the medicine. It can take 1–2 weeks for your symptoms to get better and 4–6 weeks for the full effects. During this time, you may notice some side effects.
Side effects are more common when you start aripiprazole, and many go away as you keep taking the medicine. Some side effects may not go away but if you are worried about them or if you feel that the medicine isn’t helping, your doctor can make changes to find the right fit for you.
The most common side effects of aripiprazole include feeling restless, shaky hands, trouble sleeping, headache, constipation, stomach ache, or blurred vision. Drowsiness is not a common side effect but can happen to some people and may affect your ability to drive.
There are some physical health tests you will need to have before you start aripiprazole and during your treatment. These tests will make sure it’s safe to start the medicine and check for side effects so that any unwanted changes are found and managed early. They include a heart check, weight and body mass index and blood tests to check how your body handles sugar and cholesterol.
There are some other things you should know about aripiprazole.
Don’t stop taking aripiprazole suddenly because your symptoms may come back. Your doctor can advise you on the best way to stop the medicine.
You can take aripiprazole as a tablet every day or an injection once a month or every two months.
It’s a good idea for you to take aripiprazole tablets at the same time each day – usually once a day in the morning.
For you to have the best chance of staying well, you should keep taking aripiprazole for 1–3 years or longer depending on the reason why you’re taking it.
If you notice that you become much more impulsive and start spending more money, eating more or gambling, you should speak to your doctor straight away.
For more information on aripiprazole, you can speak to your doctor, nurse prescriber or pharmacist, or read the patient information leaflet.
If you want to save the important information from this video, take a screenshot now!
This video was co-designed by the Progress Research Group.
Acknowledgements
The authors gratefully acknowledge the contributions and support of our anonymous expert-by-experience, as well as Laura Sahm and Michelle O’Driscoll of University College Cork for their Health Literacy expertise, Ciaran Carolan, and the nursing staff from Cluain Mhuire Community Mental Health Services for their in-use feedback and comments. We would like to acknowledge the support of the HSE Spark Innovation Programme.
Financial support statement
This work was supported by the Health Research Board (APRO-2023-005).
Competing interests
No competing interests.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committee on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. The authors assert that the local ethics committee has determined that ethical approval for publication of this co-design project was not required by their local Ethics Committee.