Management Implications
Aquatic weed infestations are increasingly problematic in lentic and lotic systems. With relatively few management options, new techniques are needed to effectively and efficiently control the spread of these noxious weeds. Submersed aquatic herbicide applications rely on concentration exposure time for efficacy, commonly performed in a single herbicide application or sequential applications to maintain a single concentration exposure time. This study documented that separating single or sequential applications of herbicides into three applications with a nontreatment interval of no herbicide between each exposure period does not negatively affect efficacy of florpyrauxifen-benzyl or endothall on monoecious Hydrilla verticillata (hydrilla), although nontreatment intervals of 12 d did reduce fluridone efficacy. No difference in efficacy was observed between 10 and 15 µg L−1 of fluridone, indicating lower concentrations may be used when required to meet water use restrictions or desired selectivity levels. These results also indicate that exposure times may be separated or interrupted without negative consequences to efficacy with these herbicides, which may be particularly of relevance to high water flow systems where exposure time is limited. Further investigation is needed to identify optimal nontreatment intervals, herbicide concentrations, and field verification.
Introduction
Hydrilla [Hydrilla verticillata (L.F.) Royle] is an aquatic invasive species that rapidly spreads through fragmentation and persists via axillary and subterranean turions (Netherland Reference Netherland1997). Three biotypes of H. verticillata are present in the United States: monoecious H. verticillata, predominately located from Georgia north along the Atlantic Coast and in the Great Lakes region, as well as Washington and California; dioecious H. verticillata, predominately located in the southeastern United States from Texas to Florida; and clade C H. verticillata in the Connecticut River and the surrounding water bodies (Foley et al. Reference Foley, Stebbins, Doherty, Tippery and Bugbee2024; Steward et al. Reference Steward, Van, Carter and Pieterse1984). Monoecious H. verticillata is characterized by senescence in fall and high subterranean turion production, dioecious H. verticillata tends to have more uniform biomass with subdued senescence in fall compared with monoecious H. verticillata, and clade C hydrilla has prolific axillary turion production with senescence observed in field conditions (Beets et al. Reference Beets, Foley, Sperry, Howell and Richardson2025; True-Meadows et al. Reference True-Meadows, Haug and Richardson2016).
Research and management of H. verticillata has primarily focused on invasions in lentic systems such as lakes and reservoirs; however, H. verticillata invasions in lotic (flowing) systems pose risks to water conveyance, flood control, water quality, and native species distributions (Haller and Sutton Reference Haller and Sutton1975; Langeland Reference Langeland1996). Furthermore, the introduction of H. verticillata to lotic systems that are connected to reservoirs also increases the likelihood that H. verticillata will establish in previously uninvaded systems. Herbicides are a commonly used tool for H. verticillata control. Achieving consistent, effective control of monoecious H. verticillata in lotic systems is challenging due to high flow and water exchange rates, which limit herbicide concentrations and exposure times (Getsinger and Netherland Reference Getsinger and Netherland2018). Additionally, effectiveness of herbicide treatments for long-term H. verticillata control is maximized when herbicides are applied following turion sprouting, but before biomass has peaked and new turions are formed (Nawrocki et al. Reference Nawrocki, Richardson and Hoyle2016; True-Meadows et al. Reference True-Meadows, Haug and Richardson2016). However, optimal application timing for H. verticillata in lotic systems can coincide with high or inconsistent flow, water diversions, dam releases, tidal fluctuations, or major weather events such as hurricanes, which are not conducive for maintaining aquatic herbicide exposure time (Fox et al. Reference Fox, Haller and Shilling1994; Howell et al. Reference Howell, Sperry, Beets, Durham, Glueckert and Riner2026; Sartain et al. Reference Sartain, Haug, Getsinger, Sperry, Heilman and Greer2023).
Herbicide concentration–exposure time (CET) relationships are pivotal to successful submersed aquatic plant management. Increased herbicide exposure time improves efficacy, but the relationship between concentration and exposure time for effective aquatic weed control is unique to each herbicide, for example, endothall, fluridone, and florpyrauxifen-benzyl (Beets et al. Reference Beets, Heilman and Netherland2019; Glomski and Netherland Reference Glomski and Netherland2010; Mudge et al. Reference Mudge, Getsinger and Gray2015; Netherland Reference Netherland2015; Netherland et al. Reference Netherland, Green and Getsinger1991, Reference Netherland, Getsinger and Turner1993). Standard herbicide CET scenarios, in which herbicides are applied a single time (single applications), may not be as applicable for use in lotic systems due to rapid water exchange greatly reducing exposure time. Increasing application concentration may not be possible due to label restrictions, water use restrictions, desired selectivity, or local or state regulations, and is not guaranteed to reduce the required exposure time for efficacy.
Aquatic herbicide applications are commonly performed via boat or airboat using weighted trailing hoses along a parallel track in the littoral zone (Haller et al. Reference Haller, Gettys and Glenn2011; Sperry et al. Reference Sperry, Leary, Jones and Ferrell2021). This targeted delivery method allows the herbicide to access the weed bed and the water column thoroughly, depending on plant density, target species, and the herbicide being applied. Pump systems ensure consistent injection into the water column. In many flowing canals, as well as some reservoirs and rivers, drip or metered applications are used to deliver pulses of herbicide that travel downstream (Dugdale et al. Reference Dugdale, Islam, Hunt, Liu, Bulter, Clements and Netherland2019; Regan Reference Regan2017; Sammons et al. Reference Sammons, Maecina and Partridge2003). For these pulses the exposure time is defined as the period taken from the head to the tail of the plume to pass a specific geographic point.
Traditional sequential applications often use applications spaced months apart (Madsen et al. Reference Madsen, Sartain, Turnage and Marko2013; Turnage et al. Reference Turnage, Byrd, Wersal and Madsen2019) that consider each treatment a separate CET scenario or “bump” treatments to extend a single CET scenario (Getsinger et al. Reference Getsinger, Madsen, Koschnick and Netherland2002). One alternative to single applications or traditional sequential applications to minimize the effects of flow and water exchange on herbicide efficacy is intermittent applications wherein a nontreatment interval of no herbicide exposure is introduced for a specified time period between discrete herbicide applications. Previous mesocosm experiments have indicated that intermittent exposures do not result in decreased efficacy on H. verticillata or Vallisneria taxa (Beets et al. Reference Beets, Haug, Sperry, Thum and Richardson2024; Darnell Reference Darnell2022; Netherland Reference Netherland2015). The functionality of intermittent exposure is not well understood, and there is a lack of information on the influence of nontreatment interval duration. This application method of breaking up exposure times within a single treatment may prove beneficial in both lentic and lotic environments where longer exposure periods are not feasible due to water movement.
Florpyrauxifen-benzyl is the newest registered aquatic herbicide (Anonymous 2018), and publications delineating CET requirements for monoecious H. verticillata are extremely limited. Although it is only registered for use in slow-moving or quiescent waters, intermittent exposures may be beneficial in both systems. Florpyrauxifen-benzyl is a pro-herbicide; pro-herbicides convert to their acid form via hydrolysis, which is bioactive and may be linked to increased efficacy (Miller and Norsworthy Reference Miller and Norsworthy2018; Netherland and Richardson Reference Netherland and Richardson2016; Ortiz et al. Reference Ortiz, Nissen and Dayan2022). Previous mesocosm research has indicated dioecious H. verticillata control at concentrations of 24 µg L−1 for 24 h (Beets and Netherland Reference Beets and Netherland2018). Richardson et al. (Reference Richardson, Haug and Netherland2016) reported a lowest observable effect concentration for monoecious H. verticillata of 3 µg L−1 with a static exposure in a laboratory setting. Few ecosystem-level experiments have been conducted with florpyrauxifen-benzyl due to its relatively recent registration.
In contrast to florpyrauxifen-benzyl, fluridone, a herbicide inhibiting phytoene desaturase, has been widely used for control of monoecious and dioecious H. verticillata. The presence of fluridone-resistant dioecious H. verticillata in Florida has necessitated new use patterns and alternative herbicides (Michel et al. Reference Michel, Scheffler, Arias, Duke, Netherland and Dayan2004; Nawrocki et al. Reference Nawrocki, Richardson and Hoyle2016; Netherland and Jones Reference Netherland and Jones2015; True-Meadows et al. Reference True-Meadows, Haug and Richardson2016). Some formulations are registered for use in flowing systems, and some states have supplemental labels allowing formulations to be used in flowing systems for critical management needs. CET and application timing are limiting factors in fluridone efficacy, especially in hydrodynamic systems and water bodies with water use restrictions (Rasmussen et al. Reference Rasmussen, Conrad, Green, Khanna, Wright, Hoffmann, Caudill and Gilbert2022).
Due to widespread fluridone-resistant biotypes of dioecious H. verticillata in Florida, endothall is heavily relied upon for management (Albrecht et al. Reference Albrecht, Arias, Scheffler, Duke, Netherland and Dayan2004; Sperry et al. Reference Sperry, Leary, Jones and Ferrell2021). Endothall, a contact herbicide inhibiting serine/threonine phosphatase is registered for use in flowing and quiescent ecosystems and can be applied via boat or shoreline injections (Ortiz et al. Reference Ortiz, Nissen and Dayan2022). Endothall CET requirements have been thoroughly investigated, with effective control of dioecious H. verticillata at high concentrations (4 to 5 mg L−1) and 12- to 18-h exposure times or long exposure times (48 h or more) and low concentrations (2 to 3 mg L−1) (Poovey and Getsinger Reference Poovey and Getsinger2010; Skogerboe and Getsinger Reference Skogerboe and Getsinger2010). Although monoecious H. verticillata has not been as thoroughly researched compared with dioecious H. verticillata, endothall efficacy and translocation is reduced for the monoecious biotype (Ortiz et al. Reference Ortiz, Nissen and Dayan2022; Poovey and Getsinger Reference Poovey and Getsinger2010).
New techniques, herbicides, and use patterns should be evaluated considering that endothall-resistant populations of dioecious H. verticillata have been identified in Florida (Giannotti et al. Reference Giannotti, Egan, Netherland, Williams and Knecht2014). Previous research has identified the potential for metered applications of endothall in rapid-flow lotic systems on sago pondweed [Stuckenia pectinata (L.) Böerner], but this use pattern has not been widely tested on monoecious H. verticillata (Netherland et al. Reference Netherland, Sisneros, Fox and Haller1998; Sisneros et al. Reference Sisneros, Lichtwardt and Greene1998). The aim of this study was to determine the efficacy of intermittent applications of operational concentrations of florpyrauxifen-benzyl, endothall, and fluridone on monoecious H. verticillata biomass. We hypothesized that the nontreatment interval would not negatively impact herbicide efficacy.
Materials and Methods
Three separate experiments were performed on monoecious H. verticillata in greenhouses at North Carolina State University (NCSU), Raleigh, NC. Monoecious H. verticillata tubers were collected from culture tanks at NCSU, originally sourced from an impoundment in Granville County, NC (36.14°N, 78.79°W), and sprouted in dechlorinated tap water (API Tap Water Conditioner®, Mars Fishcare North America, Chalfont, PA, USA). Intermittent exposures were achieved by separating the commonly used exposure periods with different nontreatment intervals between herbicide reapplications. Concentration exposure requirements differ between florpyrauxifen-benzyl, endothall, and fluridone, so reapplication intervals, concentrations, and exposure times were tailored to match each herbicide based on label requirements and historical data (Beets et al. Reference Beets, Heilman and Netherland2019; Netherland Reference Netherland2015; Poovey and Getsinger Reference Poovey and Getsinger2010).
Florpyrauxifen-Benzyl
Once monoecious H. verticillata tubers had sprouted and begun to branch, they were placed into 0.09-L plastic cups (Jolly Chef Cups, Jolly Chef, Yuhang, China) with topsoil (Timberline Soil Top Soil, Oldcastle® Lawn & Garden, Atlanta, GA, USA) amended with slow-release fertilizer (Osmocote Smart Release 15-9-12®, Scotts, Marysville, OH, USA) at 3 g L−1 soil and covered with a sand cap (Quikrete® Play Sand, The Quikrete® Companies, Atlanta, GA, USA) to reduce nutrient leaching into the water column. After an establishment period of 3 wk, a single monoecious H. verticillata plant was placed in a 1-L glass mesocosm with 900 ml of water. Four representative plants were harvested as a pretreatment reference. Each mesocosm was considered an experimental unit. Plants were treated with 10, 30, or 50 µg L−1 of florpyrauxifen-benzyl (ProcellaCOR SCTM, SePRO, Carmel, IN, USA) for three 24-h exposure periods. At the end of the 24-h exposure period, plants were removed from the treated mesocosm and placed in a new a mesocosm with nontreated water. Plants were given nontreatment intervals of 0, 1, 3, or 6 d between each 24-h exposure period for a total exposure time of 72 h. Each application concentration and nontreatment interval combination had four replicates for a total of 48 experimental units. Herbicide was applied via direct injection to the water column using a serial dilution of florpyrauxifen-benzyl for each 24-h exposure. Water temperatures averaged 22.1 C and pH 7.9 at time of treatment, which were the ambient temperature and pH from the municipal water source. A nontreated control was included as a reference in each experimental run, with mesocosms arranged in a completely randomized design.
Mesocosms were placed under 50% shade cloth in a greenhouse to reduce algal proliferation and to standardize light and temperature during the experiment. Observations of plant appearance and visually estimated percent control were taken weekly and compared with nontreated control (0% = no control; 100% = no plant biomass remaining). Destructive biomass harvest occurred 6 wk after study initiation. Aboveground biomass was clipped at the sediment–water interface, washed, and dried in a forced-air dryer for 72 h at 65 C. The experiment was repeated three times (three trials) with Trial 1 initiated on April 8, 2021, Trial 2 on April 19, 2021, and Trial 3 on April 7, 2023. One replication for each treatment from the first trial was excluded due to poor plant health before treatment.
Endothall
Monoecious H. verticillata plants were established and handled as in florpyrauxifen-benzyl protocol, except that they were placed into a 0.57-L pot (Square Pot HC Companies 4” Kord Lite Green, Hummert™ International, Earth City, MO, USA) following branching. To account for the larger experimental unit, the establishment period was increased to 5 wk to attain sufficient biomass. After the establishment period, a single monoecious H. verticillata plant was placed in a plastic 15-L mesocosm with 12 L of water treated with water conditioner (API Tap Water Conditioner®, Mars Fishcare North America). Plants were treated with 2, 3, or 4 mg L−1 of endothall (Aquathol® K, United Phosphorus, Inc. [now UPL Aquatics], King of Prussia, PA, USA) for three 8-h exposure periods or a single 24-h exposure period. Due to logistical constraints, herbicide was not reapplied every 8 h when there was not a nontreatment interval. Plants were given nontreatment intervals of 0, 16, 40, or 64 h between each 8-h exposure period for a total exposure time of 24 h. Each concentration and nontreatment interval combination had four replicates. Water temperatures averaged 22.4 C and pH 7.38 at the time of treatment. This experiment was repeated twice (two trials) with Trial 1 initiated July 9, 2021, and Trial 2 initiated April 18, 2023.
Fluridone
Monoecious H. verticillata plants were established as in the endothall protocol, except that they were placed in a 1.8-L pot (Dillen 6.0 Azalea Thinwall Terracotta, Griffin Greenhouse Supplies, Tewksbury, MA, USA) following branching. After the establishment period, a single monoecious H. verticillata plant was placed in a plastic 18-L mesocosm with 16 L of water. Plants were treated with 5, 10, or 15 µg L−1 of fluridone (Sonar GenesisTM, SePRO) for three 15-d exposure periods. At the end of the 15-d exposure period, plants were placed in a mesocosm with nontreated water. Plants were given nontreatment intervals of 0, 3, 6, or 12 d between each 15-d exposure period for a total exposure time of 45 d, with a new serial dilution for each reapplication. Each concentration and nontreatment interval combination had four replicates. Water temperature was 25.3 C and pH 7.6 at time of treatment. A nontreated control was included as reference for each harvest period. Final harvest occurred 6 wk after the final treatment to account for the slow plant response to fluridone (Arnold Reference Arnold1979; Netherland et al. Reference Netherland, Getsinger and Turner1993). This experiment was repeated twice (two trials) Trial 1 was initiated on July 18, 2023, and Trial 2 on August 1, 2023.
Statistical Analysis
Aboveground biomass reduction was calculated by the following equation:
Values that resulted in negative reduction (sample was larger than mean of the control) were corrected to zero. Biomass reduction and visual control data were subjected to a mixed-model two-way ANOVA and a Tukey’s honest significant difference (HSD) test for mean separations between significant factors in JMP Pro (JMP Pro v. 17, SAS Institute, Cary, NC, USA). Herbicide concentration and nontreatment interval were considered fixed effects for a total of two factors and trial was considered a random effect. Biomass data were also subjected to a separate mixed-model two-way ANOVA with a Dunnett’s test to evaluate significant differences from the control. Results from the trials were pooled for each herbicide, based on lack of significance when included in a mixed-model ANOVA (P > 0.05).
Results and Discussion
Florpyrauxifen-Benzyl
Florpyrauxifen-benzyl applications resulted in synthetic auxin symptoms. Epinasty was the most common symptom at all concentrations, with fragmentation and decreased pigmentation occurring at 30 and 50 µg L−1. There was a significant interaction between herbicide concentration and nontreatment interval for visually estimated control and biomass reduction (P < 0.01). Minimal differences were observed among treatments at 2 and 4 wk after initial treatment (WAIT) (Table 1). At 6 WAIT, plants treated with 30 µg L−1 with a 6-d nontreatment interval exhibited the highest control (91%), significantly more than observed in plants treated with 10 µg L−1 florpyrauxifen-benzyl, 30 µg L−1 with a 3-d nontreatment interval or 50 µg L−1 without a nontreatment interval (Supplementary Material). Plants degraded during the experimental period; however, some minor recovery (color change from bleached to green and straightening of stems) was observed 6 WAIT, resulting in discrepancies between visually estimated control and measured biomass reduction. Heavily injured plants compressed and fragmented to a high degree, despite biomass remaining in the mesocosm, and may have confounded observations resulting in higher control estimation than actual biomass reduction. Viable biomass was not differentiated from nonviable, which highlights the need for further investigation into regrowth potential following treatments from florpyrauxifen-benzyl, especially in plant apical fragments that may serve as a dispersal method for H. verticillata (Heidbüchel and Hussner Reference Heidbüchel and Hussner2019).
Estimated visual control and biomass reduction of Hydrilla verticillata following three 24-h exposures of florpyrauxifen-benzyl.a

a Means with same letter within each column are not statistically different from each other according to Tukey’s honest significant difference (HSD) (α = 0.05; n = 11)
b Control estimates based on visual estimation (0% = no injury; 100% = complete control)
c Mean nontreated biomass = 0.18 g; asterisks indicate treatments are significantly different from nontreated control according to Dunnett’s test (α = 0.05)
Nontreated control biomass increased 4.9 times over the study period compared with nontreated plants harvested on the day of the initial treatment (preharvest). Aboveground biomass reduction was greatest (89%) following exposure to 30 µg L−1 with a 6-d nontreatment interval (Table 1). Dunnett’s test confirmed biomass of all treatments was significantly different from that of the nontreated control (P < 0.001). High efficacy of florpyrauxifen-benzyl has been observed in dioecious H. verticillata at similar concentrations with 72 h of exposure (Beets and Netherland Reference Beets and Netherland2018; Mudge et al. Reference Mudge, Sartain, Getsinger and Netherland2021). Previous mesocosm data indicate absorption is lower and EC50 values are higher for florpyrauxifen-benzyl in monoecious H. verticillata compared with its dioecious counterpart (Haug et al. Reference Haug, Ahmed, Gannon and Richardson2021; Richardson et al. Reference Richardson, Haug and Netherland2016). Beets et al. (Reference Beets, Haug, Sperry, Thum and Richardson2024) observed 96% to 100% control of monoecious H. verticillata with florpyrauxifen-benzyl at 30 µg L−1 in single application and intermittent exposures.
Furthermore, Sperry et al. (Reference Sperry, Leary, Jones and Ferrell2021) reported large biovolume reductions of dioecious H. verticillata in a small Florida lake with an application of florpyrauxifen-benzyl at 48 µg L−1 when approximately 20% of the lake was treated, further corroborating high efficacy in dioecious H. verticillata treatments. Selectivity for native desirable species, including Vallisneria sp. and spatterdock [Nuphar lutea (L.) Sm. ssp. advena (Aiton) Kartesz & Gandhi], despite the rapidly declining concentrations of florpyrauxifen-benzyl in treated areas was also observed (Sperry et al. Reference Sperry, Leary, Jones and Ferrell2021). However, small-plot treatments of florpyrauxifen-benzyl on monoecious H. verticillata in a high-flow reservoir resulted in no reduction in H. verticillata abundance despite the presence of injury symptoms at 72 h after treatment (HAT) (Sartain et al. Reference Sartain, Haug, Getsinger, Sperry, Heilman and Greer2023). Residue analysis by Sartain et al. (Reference Sartain, Haug, Getsinger, Sperry, Heilman and Greer2023) revealed florpyrauxifen-benzyl concentration had dissipated within 5 HAT to 4.2 µg L−1 from the initial target concentration of 48 µg L−1, which was likely tied to dam operation, as well as plant absorption and herbicide degradation. The dichotomy between Sperry et al. (Reference Sperry, Leary, Jones and Ferrell2021) and Sartain et al. (Reference Sartain, Haug, Getsinger, Sperry, Heilman and Greer2023) may be further explained by the plot size relative to the size of the lake and the impact this water exchange potential (relative size of treated and nontreated area) has on herbicide exposure time.
In CET mesocosm studies, intermittent exposure scenarios, and systems where sudden water exchange may occur, such as in Sartain et al. (Reference Sartain, Haug, Getsinger, Sperry, Heilman and Greer2023), uptake of the acid form may be decreased, thereby reducing efficacy of florpyrauxifen-benzyl treatments. Netherland and Richardson (Reference Netherland and Richardson2016) observed differential efficacy of the ester (florpyrauxifen-benzyl) and acid forms on some species, indicating that the acid product likely has a role in efficacy with florpyrauxifen-benzyl and factors into CET requirements.
Endothall
There was no significant interaction between herbicide concentration and nontreatment interval for visually estimated control or biomass reduction (P > 0.05). Additionally, nontreatment interval did not have a significant effect on biomass reduction or estimated control (P > 0.05), but herbicide concentration did have a significant effect (P < 0.001). Monoecious H. verticillata visually estimated control was significantly different from the nontreated control in plants exposed to 3 mg L−1 endothall (25%) and 4 mg L−1 endothall (41%) at 2 WAIT (Supplementary Material; Table 2). Monoecious H. verticillata control progressed by 4 WAIT, with estimated control increasing with herbicide concentration, and this trend continued at 6 WAIT.
Estimated visual control and biomass reduction of Hydrilla verticillata following a single 24-h application or three 8-h applications of endothall.a

a Means with same letter within each column are not statistically different from each other according to Tukey’s honest significant difference (HSD) (α = 0.05; n = 8).
b Control estimates based on visual estimation (0% = no injury; 100% = complete control).
c Mean nontreated biomass = 1.68 g; asterisks indicate treatments are significantly different from nontreated control according to Dunnett’s test (α = 0.05)
Mean biomass of the nontreated control increased seven times over the study period compared with preharvest biomass. Dunnett’s test confirmed biomass of all treatments was significantly different from that of the nontreated control (P < 0.001). Monoecious H. verticillata treated with 2 mg L−1 resulted in the lowest observed efficacy (66%; Table 2). Endothall applications with 3 mg L−1 resulted in 81% reduction in biomass, while monoecious H. verticillata exposed to 4 mg L−1 endothall had 93% biomass reduction. Previous research with endothall has found 2.0 mg L−1 for 48 h or 3 to 5 mg L−1 for 24 h provides effective control of dioecious H. verticillata (Netherland et al. Reference Netherland, Green and Getsinger1991). Ortiz et al. (Reference Ortiz, Nissen and Gray2019) observed accumulation of endothall in monoecious H. verticillata greater than treatment solution concentrations and evidence of translocation of endothall to root tissue. Previous studies have also indicated absorption of endothall into H. verticillata is low when exposed for less than 2 to 4 d (Haller and Sutton Reference Haller and Sutton1973; Ortiz et al. Reference Ortiz, Nissen and Dayan2022; Van and Conant Reference Van and Conant1988). However, Poovey and Getsinger (Reference Poovey and Getsinger2010) observed reductions in monoecious and dioecious H. verticillata biomass at 1 ppm with at least 96 h of exposure. Overall, previous research indicates endothall benefits from increased exposure times, and the results of this study documented that intermittent applications of endothall may provide an option if continuous exposure is not possible. Intermittent applications provided equivalent control when a single 24-h exposure is split into three intermittent 8-h applications, regardless of nontreatment interval.
Fluridone
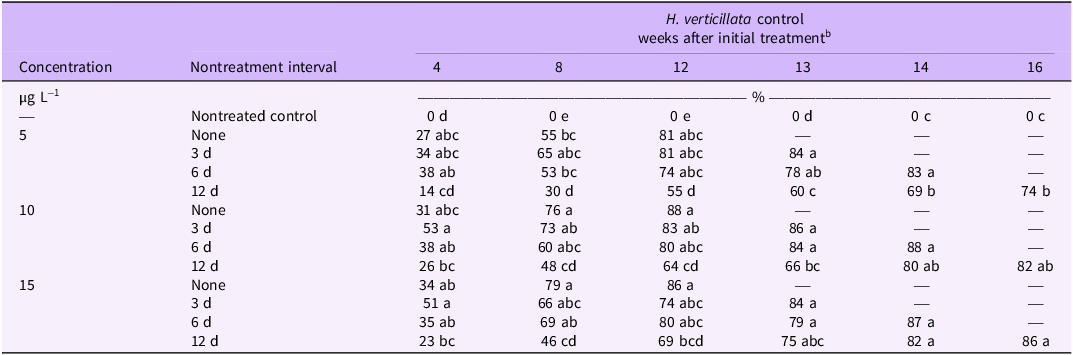
There was a significant interaction between fluridone herbicide concentration and nontreatment interval on visually estimated control. All treatments except 5 µg L−1 with a 12-d nontreatment interval resulted in 23% to 53% visually estimated control at 4 WAIT (Table 3). Previous research has also observed plant responses at this time point (Macdonald et al. Reference Macdonald, Shilling, Doong and Haller1993; Netherland Reference Netherland2015). Differences between treatments remained consistent as the study progressed, at 8 and 12 WAIT, significant differences were observed between zero nontreatment interval and 12-d nontreatment intervals (P < 0.05).
Estimated visual control of Hydrilla verticillata following three 15-d applications of fluridone.a

a Means with same letter within each column are not statistically different from each other according to Tukey’s honest significant difference (HSD) (α = 0.05; n = 8).
b Control estimates based on visual estimation (0% = no injury; 100% = complete control). Single dashes indicate final biomass harvest had occurred.
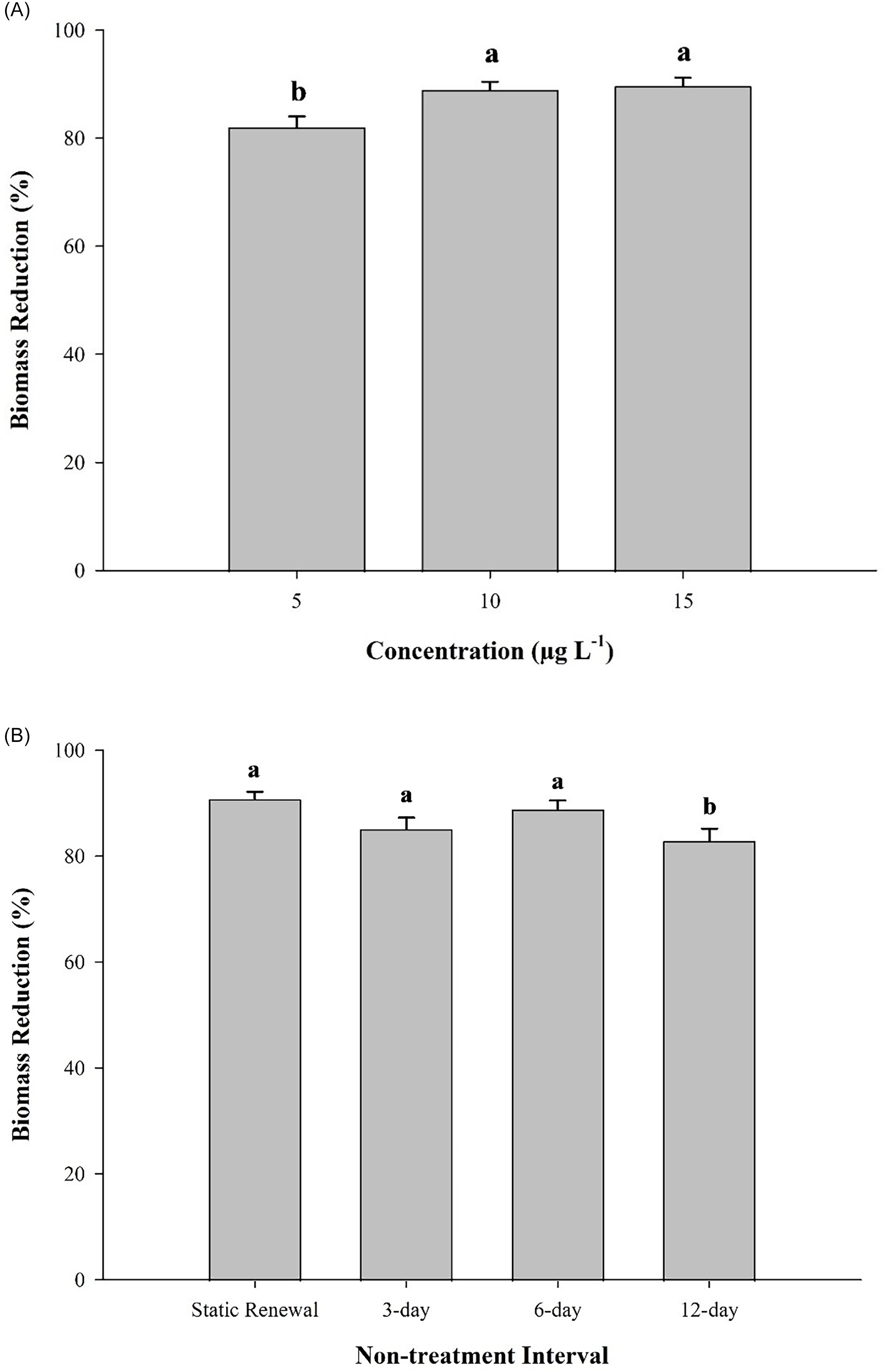
There was no interaction between herbicide concentration and nontreatment interval on biomass reduction (P = 0.50); however, main effects of concentration and nontreatment interval did have a significant influence on biomass reduction (P < 0.001). Fluridone treatments at 5 µg L−1 resulted in 82% biomass reduction, whereas treatments at 10 and 15 µg L−1 resulted in 89% biomass reduction (Figure 1A). Biomass reduction did not differ between zero nontreatment interval, 3-d nontreatment interval, and 6-d nontreatment interval (85% to 91%) but was slightly lower in fluridone applications with a 12-d nontreatment interval (83%; Figure 1B). These findings corroborate previous research that intermittent applications of fluridone with 3-d and 6-d nontreatment intervals do not decrease efficacy (Netherland Reference Netherland2015). The slight decrease in efficacy observed from the 12-d nontreatment interval suggests that plants are able to recover between exposures.
Average (±SE) biomass reduction of Hydrilla verticillata following three 15-d applications of fluridone by concentration (A) and nontreatment interval (B). Bars that share a letter are not significantly different at α = 0.05 based on a Tukey’s honest significant difference (HSD) post hoc test. Mean nontreated biomass was 3.8, 3.3, 3.7, and 3.7 g for 0, 3-d, 6-d, and 12-d nontreatment interval controls, respectively, and Dunnett’s test confirmed biomass of all treatments was significantly different from that of the nontreated control (P < 0.001).

Previous research has shown that 12, 24, and 48 µg L−1 of fluridone provide effective control of dioecious H. verticillata in growth chambers with 60 to 90 d of exposure, although regrowth was observed with a 30-d recovery period in untreated water (Netherland et al. Reference Netherland, Getsinger and Turner1993). This may allow for treatment prescriptions with lower target concentrations, which would reduce herbicide costs and can improve selectivity. However, the identification of fluridone-resistant dioecious H. verticillata supports that caution should be taken when utilizing low doses of fluridone for long-term H. verticillata management and that herbicide rotations and combinations are an important aspect of herbicide stewardship and resistance management (Michel et al. Reference Michel, Scheffler, Arias, Duke, Netherland and Dayan2004; Puri et al. Reference Puri, Haller and Netherland2009).
The relatively rapid dissipation of florpyrauxifen-benzyl in field studies indicates intermittent applications at concentrations at or below the maximum label concentration (50 µg L−1) may provide an alternative method for H. verticillata control. Our experiment documented intermittent applications of florpyrauxifen-benzyl can provide equivalent control to 72 continuous hours of herbicide exposure. We did not, however, investigate a nontreatment interval at the 14-d reapplication interval indicated on the label. Future studies should compare the shorter intervals studied here to the labeled reapplication interval and recognize the interaction between florpyrauxifen-benzyl concentration and nontreatment interval.
Understanding the absorption and physiological dynamics of our findings can also improve understanding of the management options. No relationship was observed between the efficacy of endothall concentration and the nontreatment interval between intermittent applications, indicating endothall may be less reliant on continuous exposure and a key herbicide in lotic or hydrodynamic systems where continuous CET cannot be achieved. Further studies are warranted to investigate an expanded range of nontreatment intervals, as well as the efficacy of these treatments on other aquatic macrophyte species. It should be noted that the use patterns tested in this study would exceed the 5 mg L−1 per 7-d period specified on endothall labels. Future research should seek to compare intermittent applications to those within label compliance, including decreasing concentrations of subsequent intermittent applications following initial application. Because endothall is labeled for use in flowing systems, including rivers and canals, intermittent applications can easily be adapted for use in these settings, building off existing drip and pulse methodologies that have previously been utilized (Dugdale et al. Reference Dugdale, Islam, Hunt, Liu, Bulter, Clements and Netherland2019; Regan Reference Regan2017; Sammons et al. Reference Sammons, Maecina and Partridge2003).
Further research is needed to identify the most optimal concentrations, exposure periods, and nontreatment intervals for fluridone applications, considering the large gap between the 15-d exposures tested in this experiment and 3-d exposures tested previously (Netherland Reference Netherland2015). Biomass reduction of monoecious H. verticillata did not differ between 10 and 15 µg L−1, providing some flexibility in application concentrations when degradation occurs or regulations require reduced use rates. Fluridone concentration can decrease rapidly depending on discharge values (11 to 13 d after treatment) prompting reapplication for efficacious control, especially in lotic systems (Fox et al. Reference Fox, Haller and Shilling1994). Metered drip application of fluridone was effective in suppressing monoecious H. verticillata biomass in the Eno River (NC), and intermittent exposures can be integrated into this treatment method to improve efficacy ([NCDEQ] North Carolina Department of Environmental Quality 2014; Regan Reference Regan2017).
These small-scale mesocosm experiments indicated no loss in efficacy compared with single applications with endothall or sequential applications with florpyrauxifen-benzyl and fluridone, warranting further research. Intermittent applications may provide a particularly useful management technique for lotic systems where long exposures may not be feasible, and an intermittent application approach is more realistic to these field conditions. Additional research in large outdoor mesocosms and field plots would help to identify the most efficacious intermittent applications. Small-mesocosm studies such as these are beneficial for narrowing down efficacious treatments, focusing on replication and cost efficiency, and identifying treatments to be focused on in outdoor mesocosm facilities and field trials (Netherland and Getsinger Reference Netherland and Getsinger2018). However, small-scale mesocosm trials can overestimate efficacy compared with operational settings due to the lack of complex biotic and abiotic factors that can affect herbicide efficacy, including wave action, temperature, pH, and plant size. In addition to scaling up mesocosm size, future research should prioritize other target aquatic weed species. Hydrilla verticillata might provide priority information for management of this noxious weed in the eastern and midwestern United States, but many other aquatic weeds persist in lotic and lentic environments, and their management programs may also benefit from intermittent exposures. In rapidly flowing lotic systems, it is possible that most or all water treated with a herbicide application has left the target site, and reapplication intervals could be shortened in a special needs situation using a 24(c) label. These short nontreatment intervals may also have implications for tidal systems, where treated water may move downstream and backflow. Such special needs labels may also allow use of formulations in lotic systems when they otherwise may be restricted to quiescent systems. New research is needed on the efficacy, non-target, and environmental impacts of intermittent herbicide treatments.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/inp.2026.10040
Acknowledgments
The authors thank Kara Foley, Andrew Howell, Michael Punt, Delaney Davenport, and Logan Wilson for their assistance with plant propagation, treatment, and harvesting. This article was reviewed in accordance with U.S. Army Engineer Research and Development Center policy and approved for publication. Citation of trade names does not constitute endorsement or approval of the use of such commercial products. The content of this work does not necessarily reflect the position or policy of the U.S. government, and no official endorsement should be inferred. United States Department of Agriculture is an equal opportunity employer and provider.
Funding statement
Funding for this research was provided by the U.S. Army Corps of Engineers, Engineer Research and Development Center’s Aquatic Plant Control Research Program, the Aquatic Plant Management Society’s Michael D. Netherland Graduate Student Scholarship, USDA ARS internal funding, and North Carolina State University internal funding.
Competing interests
The authors declare no conflicts of interest.