Introduction
Avian ectoparasites encompass an exceptionally diverse array of invertebrate taxa, ranging from leeches (Hirudinea) and ticks (Ixodida) to insects from various orders, including lice (Phthiraptera), true bugs (Hemiptera), fleas (Siphonaptera), and flies (Diptera) (Rothschild and Clay Reference Rothschild and Clay1957; Clayton et al. Reference Clayton, Koop, Harbison, Moyer and Bush2010). Many of these ectoparasites are hematophagous, a feeding mode that has evolved independently multiple times within insects, including in Diptera (Deepak and Shruti Reference Deepak and Shruti2024; Cardoso et al. Reference Cardoso, Cunha, Genevcius, Madeira-Ott, Costa and Rossoni2025), and which occurs in many families of both the Nematocera and Brachycera suborders (e.g., Culicidae, Simuliidae, Ceratopogonidae, Hippoboscidae, and Nycteribiidae). However, significant differences exist among dipteran ectoparasites in terms of host selection and specificity, whether they are obligatory blood feeders or not, and regarding diet differentiation across life stages. For instance, the larvae of Protocalliphora spp. (Diptera: Calliphoridae), also known as bird blowflies, are obligatory blood-feeding parasites of nestlings belonging to a wide range of altricial bird species. Little is known of the flies’ adult diet, but they have been known to visit the flowers of herbaceous plants (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989; Bennett and Whitworth Reference Bennett and Whitworth1991). Although the biology and ecology of hematophagous flies parasitising humans and domestic animals are well known (e.g., Marquardt et al. Reference Marquardt, Demaree and Grieve2004), flies parasitising wild animals have received less attention or have mainly been studied according to the costs they can inflict on their hosts. Bird blowflies exemplify this case, with most previous studies focusing on measuring the impacts their larvae may have on the nestlings and reproductive success of altricial birds (Simon et al. Reference Simon, Thomas, Blondel, Perret and Lambrechts2004; Maziarz et al. Reference Maziarz, Broughton, Chylarecki and Hebda2022). In recent years, however, several studies have addressed the environmental factors that drive the prevalence and intensity of bird nest infestations by Protocalliphora spp. (Mennerat et al. Reference Mennerat, Charmantier, Perret, Hurtrez-Boussès and Lambrechts2021; González-Bernardo et al. Reference Gonzalez-Bernardo, Moreno-Rueda, Camacho, Martinez-Padilla, Potti and Canal2025; Merino et al. Reference Merino, García-Del Río, Castaño-Vázquez and Merino2025). However, only a few studies to date have formally investigated the determinants of bird nest infestations by these parasites across multiple years and sites (Mennerat et al. Reference Mennerat, Charmantier, Perret, Hurtrez-Boussès and Lambrechts2021; Maziarz et al. Reference Maziarz, Broughton, Chylarecki and Hebda2022; González-Bernardo et al. Reference Gonzalez-Bernardo, Moreno-Rueda, Camacho, Martinez-Padilla, Potti and Canal2025; Merino et al. Reference Merino, García-Del Río, Castaño-Vázquez and Merino2025). Moreover, these studies, which mainly focused on the effects of weather or climatic factors, were conducted in a small number of forested sites and, therefore, on a limited range of habitat heterogeneity. Because of this, we have limited information on the spatial and temporal variability of infestations and therefore on the influence of abiotic and biotic factors on the host–parasite dynamics involving Protocalliphora species. The lack of long-term monitoring data on bird nest infestations by these parasites also concerns the species of blowflies involved in these interactions. At least 40 species of Protocalliphora have been described in the Holarctic region (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989), many of which have overlapping distribution ranges, resulting in occasional mixed infestations within a bird nest (Daoust et al. Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a). Moreover, the morphological identification of species is challenging in this group, at least for nonspecialists, and has been considered to be reliable only in adults (Heeb et al. Reference Heeb, Kölliker and Richner2000). Furthermore, locating and sampling adult bird blowflies in the field is not easy (Stiner Reference Stiner1969; Bennett and Whitworth Reference Bennett and Whitworth1991), and larvae and puparia often remain the primary evidence of bird blowfly parasitism and parasitic load (Jánošková et al. Reference Jánošková, Országh, Jamriška and Kopáni2010). Despite significant progress regarding the taxonomy, phylogeny (Kutty et al. Reference Kutty, Meusemann, Bayless, Marinho, Pont and Zhou2019; Yan et al. Reference Yan, Pape, Meusemann, Kutty, Meier, Bayless and Zhang2021), and identification of Protocalliphora species, notably through the work of Sabrosky et al. (Reference Sabrosky, Bennett and Whitworth1989) and Whitworth (Reference Whitworth2002, Reference Whitworth2003, Reference Whitworth, Byrd and Tomberlin2019), identification based on puparia morphology is complicated by the fact that key criteria, such as distance between specific spines, prothoracic fringe diameter, and distance between posterior spiracles, can vary widely within and overlap between species (Jánošková et al. Reference Jánošková, Országh, Jamriška and Kopáni2010; Jamriška and Modrý Reference Jamriška and Modrý2013). Furthermore, identification based on DNA barcoding using the Folmer region of the common CO1 gene fails to discriminate between most described Protocalliphora species (Whitworth et al. Reference Whitworth, Dawson, Magalon and Baudry2007). These identification challenges, especially when considering the high parasite load found in some nests, may have led some ecologists to neglect bird blowfly species composition and typically lump all individuals under “Protocalliphora spp.” (e.g., Heeb et al. Reference Heeb, Kölliker and Richner2000; Bańbura et al. Reference Bańbura, Perret, Blondel, Thomas, Cartan-Son and Lambrechts2004; Hannam Reference Hannam2006; Musgrave et al. Reference Musgrave, Bartlow and Fair2019). Lumping all species together can, however, bias species abundance estimates and hinder the study of host–parasite dynamics by neglecting the potential competition that may occur among Protocalliphora species, as well as their interactions with parasitoids within host nests.
Other overlooked factors that may influence parasitoid–parasite interactions include inter- and intraspecific variation in body size. Such variation may reflect differences in larval growth conditions, which in turn can affect metamorphosis, emergence success, and susceptibility to host selection by gregarious jewel wasps, Nasonia spp. (Hymenoptera: Pteromalidae) (Werren and Loehlin Reference Werren and Loehlin2009). These generalist wasps, which primarily parasitise the pupal stage of Protocalliphora spp., are also reported to parasitise several other calyptrate Diptera pupae of agricultural relevance (Rueda and Axtell Reference Rueda and Axtell1985), thereby potentially contributing to the top–down regulation of these flies. Most research on Nasonia spp., including the well-known N. vitripennis (Walker) and N. giraulti Darling, which both attack Protocalliphora spp. (Daoust et al. Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a; Garrido-Bautista et al. Reference Garrido-Bautista, Moreno-Rueda, Baz, Canal, Camacho and Cifrián2020, Reference Garrido-Bautista, Norte, Moreno-Rueda and Nadal-Jiménez2024), have largely focused on their genetics and behavioural ecology under laboratory conditions (e.g., Werren and Loehlin Reference Werren and Loehlin2009; Deveux et al. Reference Deveux, Christiaens, Smagghe and Taning2023). Past studies therefore provide limited insights into the ecological interactions of this parasitoid–parasite relationship in natural habitats and into how the parasitoid’s top–down regulation potential against bird blowflies fluctuates under different conditions in either space or time (Garrido-Bautista et al. Reference Garrido-Bautista, Norte, Moreno-Rueda and Nadal-Jiménez2024). These gaps in our understanding of Protocalliphora spp. extend beyond taxonomy, morphometry, and distribution. Long-term studies focusing on the prevalence and abundance of bird blowfly larvae within nests have noted important annual fluctuations after accounting for some environmental conditions (Wesołowski Reference Wesołowski2001; Musgrave et al. Reference Musgrave, Bartlow and Fair2019; Merino et al. Reference Merino, García-Del Río, Castaño-Vázquez and Merino2025); however, only Merino et al. (Reference Merino, García-Del Río, Castaño-Vázquez and Merino2025) reported patterns suggesting cyclical abundance, although they did not formally analyse cycles. To our knowledge, no details on temporal variation in species composition and parasitoidic pressures have been documented. In the present study, we quantify the spatial and temporal variability in the prevalence, intensity, and species composition of tree swallow, Tachycineta bicolor (Vieillot) (Passeriformes: Hirundinidae), nest infestations by bird blowflies over a 16-year period within a 10 200-km² gradient of agricultural intensity in southern Quebec, Canada. We also report species-specific size distributions for the puparia of three species of Protocalliphora and temporal variation in parasitoidism rates by Nasonia spp., as well as the rate of parasitoid dormancy (i.e., winter diapause) within the host’s puparia.
Methods
Study system and field sampling
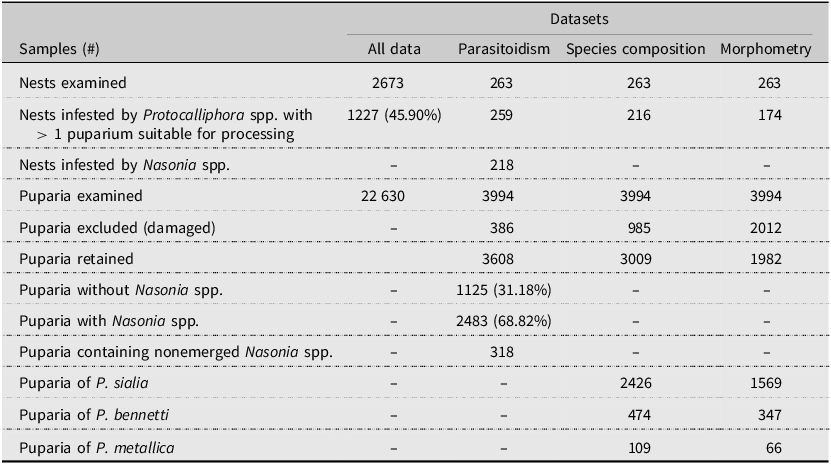
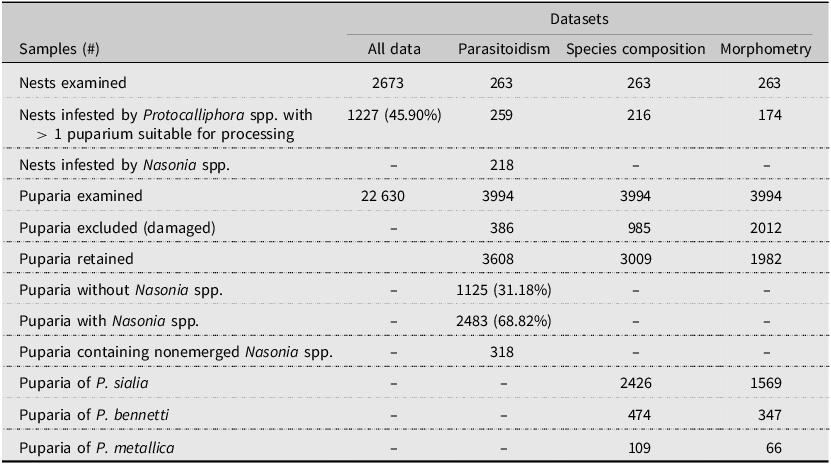
We collected Protocalliphora spp. puparia between 2004 and 2019 within a network of 400 nest boxes distributed equally among 40 farms located along a 10 200-km² gradient of agricultural intensity in southern Quebec, Canada (Ghilain and Bélisle Reference Ghilain and Bélisle2008; Supplementary material, File S1). Nest boxes were placed about 1.5 m above ground and spaced by about 50 m apart along linear transects that followed field edges. Although the primary objective of the above network initiated in 2004 was to quantify the impact of land use on tree swallow prey availability (Powell et al. Reference Powell, Garrett, Roy, Oliver, Larrivée and Bélisle2024) and breeding ecology (Garrett et al. Reference Garrett, Pelletier, Garant and Bélisle2022a), it evolved into a comprehensive platform for diverse investigations, including the effects of bird blowfly parasitic load on the swallows’ breeding success (Daoust et al. Reference Daoust, Bélisle, Savage, Robillard, Baeta and Brodeur2012b; Pigeon et al. Reference Pigeon, Baeta, Bélisle, Garant and Pelletier2013; Sigouin et al. Reference Sigouin, Bélisle, Garant and Pelletier2021). At the end of each breeding season, we collected and bagged the entire contents of nest boxes exclusively occupied by tree swallows, a single-brooded species. Collected nests were stored at 4 °C prior to processing, which included two days at –80 °C to kill any remaining living organisms, before sorting under a ventilated hood. Protocalliphora spp. puparia found within nest material were counted and stored in vials filled with 75% ethanol and referenced according to farm, nest box ID, and year of sampling. Note that puparia from 2008 and 2009 were collected under two protocols by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a) to rear some material in the lab and to collect emerging flies or Nasonia spp. wasps. As a result, some specimens for these two years were no longer available for further examination in the present study (i.e., for parasitoidism detection or for bird blowfly species composition analyses and morphometry). Nonetheless, our dataset allowed us to investigate nest infestation and parasitic load using nests from all nest boxes occupied by tree swallows with a single clutch event that resulted in at least one nestling on a given year between 2004 and 2019 (including 2008 and 2009), for a total of 2673 nests (Table 1).
Number of tree swallow nests and Protocalliphora spp. puparia collected and analysed between 2004 and 2019 in southern Quebec, Canada. No puparia from 2008 to 2009 could be included in our parasitoidism, species composition, and morphometry estimates because they were used in a previous study by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a) and were no longer suitable for further processing. Instead, proportions for 2008 and 2009 were directly derived from Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a)
Specimen examination
Due to the considerable number of Protocalliphora spp. puparia and the significant time required for individual specimen examination, a subsample of 263 nests infested by Protocalliphora spp. was selected for parasitoidism assessment, species identification, and morphometric measurements of puparia found therein (Table 1). Infested nests were subsampled as follows: 10 farms were selected by stratified random sampling to represent the gradient of agricultural intensity of the study area across years. (For details, see Supplementary material, File S2.) For each year (excluding 2008 and 2009), two infested nests (when available) were randomly sampled on each of the above 10 farms. All puparia found within these nests were retained. If fewer than 100 puparia could be obtained for a given year, we randomly selected an additional farm from which we randomly selected up to two nests to extract all puparia therein. This process was then repeated until at least 100 puparia could be processed for a given year. The number of additional farms thereby sampled averaged 1.2 per year, a number unlikely to alter the general patterns provided by the core group of 10 farms.
All Protocalliphora spp. specimens from the resulting subsample of 263 nests (n = 3994 puparia) were examined, and only those with adequate structural integrity for the determination or measurement of a given attribute were considered in data analyses regarding parasitoidism, species composition, and morphometry (Table 1).
Parasitoidism of Protocalliphora spp. by Nasonia spp.
Species-level identification of Nasonia spp. wasps was not conducted in the present work due to fall sample collections, when most parasitoids had already emerged or when those that had not were killed before completing their development. However, early subsampling and rearing of Protocalliphora spp. pupae by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a) within the same study system allowed the post-emergence identification of two Nasonia species – N. vitripennis and N. giraulti. Nasonia vitripennis accounted for more than 90% of parasitoid events in Protocalliphora spp.
The occurrence of Nasonia spp. parasitoidism on a host puparium is typically diagnosed by the presence of one or more exit holes of 0.5–1 mm in diameter. Although other parasitoids can create similar emergence holes, N. vitripennis and N. giraulti were the only two parasitoid species to emerge from 956 pupae sampled by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a) across the same study system in 2008–2009. We therefore presume that most, if not all, emergence holes noted in sampled Protocalliphora spp. resulted from individuals of this genus. Because Nasonia spp. can die or overwinter as pupae inside their host instead of emerging at the end of the active season (Floate and Skovgård Reference Floate and Skovgård2004; Garrido-Bautista et al. Reference Garrido-Bautista, Moreno-Rueda, Baz, Canal, Camacho and Cifrián2020), puparia containing a dead Protocalliphora spp. pupa and those containing unemerged Nasonia spp. look identical. For this reason, we systematically dissected Protocalliphora spp. puparia showing no sign of either fly or wasp emergence to verify the occurrence of Nasonia spp. parasitoids to discriminate pupation failures linked to parasitoidism from those resulting from other causes. All 3608 Protocalliphora spp. puparia assessed for Nasonia spp. infestation were therefore classified into one of the following categories: no evidence of Nasonia spp. parasitoidism; presence of Nasonia spp. emergence holes; and presence of unemerged Nasonia spp. pupae without exit holes.
Protocalliphora species composition
We identified 3009 well-preserved Protocalliphora spp. puparia based on morphology, following Sabrosky et al. (Reference Sabrosky, Bennett and Whitworth1989) and Whitworth (Reference Whitworth2002, Reference Whitworth2003, Reference Whitworth, Byrd and Tomberlin2019). Consistent with Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a), who conducted research within the same study system, only three species were identified: Protocalliphora sialia Shannon and Dobroscky, Protocalliphora bennetti (Whitworth), and Protocalliphora metallica (Townsend). Ten puparia (five with and five without Nasonia exit holes) from each of the three Protocalliphora species were kept as voucher specimens and deposited in the Bishop’s University Insect Collection (BUIC; Sherbrooke, Quebec, Canada), along with the remains of five dissected specimens containing unmerged parasitoid wasps. All material is filed in the voucher collection under “Coroller-Chouraki Protocalliphora spp. 2025.”
Protocalliphora spp. morphometry
To assess inter- and intraspecific variation in the maximum length and width of Protocalliphora spp. puparia (in millimetres), each puparium was photographed before species identification using a Leica M165FC stereomicroscope equipped with a 0.63× PlanApo objective, a Leica MC170HD colour camera, and Leica LAS× image-capture software, version 3.0.3 (Leica Camera AG, Wetzlar, Germany). The microscope being already factory-encoded, all acquired images were automatically calibrated in terms of pixel size (µm/pixel). All image-based measurements were subsequently performed using Fiji image analysis software (Schindelin et al. Reference Schindelin, Arganda-Carreras, Frise, Kaynig, Longair and Pietzsch2012). Image analysis was automated using a homemade Fiji script developed using the built-in Fiji/ImageJ API. For each image, a threshold-based image segmentation was performed to isolate individual puparia, and a Fit Ellipse function was applied to each object. This allowed us to model any photographed puparium as an ellipsoid-shaped object and to calculate its theoretical maximum length and width. This approach minimised measurement biases that might result from puparia from which an adult fly had emerged otherwise appearing shorter due to the missing emergence cap.
Data analysis
Interannual variability in tree swallow nest infestation by Protocalliphora spp. between 2004 and 2019 was evaluated by calculating, for each sampling year, the proportion of infested nests and the average number of Protocalliphora spp. puparia per infested nest (± standard deviation). We then assessed how the annual mean nest infestation rate across farms varied in time between 2004 and 2019 and to what level each farm followed that global trend, if any, using a generalised linear additive mixed model fitted with the mgcv package, version 1.9-1 (Wood Reference Wood2023) in R, version 4.3.1 (R Core Team 2023). More specifically, we modelled the proportion of infested nests over time based on two components: (1) a global smooth term for year to capture the overall temporal trend, and (2) farm-specific smooth terms to account for temporal patterns unique to each farm. The farm-specific (local) trends could differ in shape and complexity from the global one (see Pedersen et al. Reference Pedersen, Miller, Simpson and Ross2019, model GI). The generalised linear additive mixed model was fitted using a binomial distribution on farm–year counts of infested nests (with the farm–year nest counts as weight argument) and logit-link function. We performed the same exploratory analyses for the average number of Protocalliphora spp. puparia per infested nest with a generalised linear additive mixed model that used a tweedie distribution and a log-link function due to substantial overdispersion with respect to a (truncated) Poisson distribution. Temporal autocorrelation in the residuals was estimated to be weak (r k < 0.15) at all k (≤ 10) time lags using the acf function in R (Simpson Reference Simpson2018), suggesting that the fitted generalised linear additive mixed models adequately captured the temporal structure within the response variables. Detailed codes, model diagnostics, and results for these analyses are provided in Supplementary material, File S3.
Interannual variability in parasitoid–parasite dynamics was evaluated by contrasting the yearly proportion of noninfested to infested puparia and the proportion of infested puparia from which wasps had emerged against those still containing Nasonia spp. pupae or adults. Interannual variability in Protocalliphora species composition was assessed by calculating the yearly relative proportion of each species for all specimens that could be unambiguously identified. Parasitoidism rates and species composition for 2008–2009 were obtained from Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a).
Intra- and interspecies variability in the maximum length and width of puparia of P. sialia, P. bennetti, and P. metallica were assessed first by using density plots and boxplots based on the raw data. We then assessed interspecific differences in mean puparium maximum length and width using generalised linear mixed models fitted with the glmmTMB R package (Brooks et al. Reference Brooks, Bolker, Kristensen, Maechler, Magnusson, McGillycuddy and van Benthem2023). In this analysis, P. bennetti, the intermediate-size species (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989; Whitworth Reference Whitworth2002), was designated as the reference category. Farm ID and year were included as random effects to account for environmental variation and to control for potential confounding effects. Nest box ID was included as a random effect, but this led to convergence issues due to the low number of repeated measures within each nest: for this reason, this variable was omitted from the analysis. The proportion of variance explained by Farm ID and year random effects was estimated following Nakagawa and Schielzeth (Reference Nakagawa and Schielzeth2013). Because species-specific maximum length and width were approximately normally distributed, generalised linear mixed models were fitted using a Gaussian distribution and identity link.
Results
Spatiotemporal variation in Protocalliphora spp. prevalence and load
Nest infestation by bird blowflies occurred widely throughout the study area. All 40 monitored farms harboured at least one infested tree swallow nest at some point between 2004 and 2019 and between 67.65% and 92.31% of farms had at least one infested nest, depending on the year. Overall, 45.90% (1227) of the 2673 tree swallow nests with a single clutch event leading to at least one nestling were infested by at least one Protocalliphora spp. larva, based on counts of puparia. The proportion of infested nests varied substantially both among years (range: 28.76% in 2012 to 69.33% in 2008; Fig. 1A) and among farms (range: 13.37–79.49%). Although most farms showed yearly fluctuations in nest infestation rate that followed the global, nonlinear trend suggestive of a recurrent pattern with an approximately 3-year period at the scale of the study area, about 30% of farms showed mild to strong departures from that global trend (n = 587 farm–years; GAMM R 2 = 48.8%; Supplementary material, File S3).
A, Patterns of tree swallow nest infestation by Protocalliphora spp. observed between 2004 and 2019 in southern Quebec, Canada. Left y-axis and corresponding point ranges (red dots ± error bars) show the yearly average (± standard deviation; SD) number of Protocalliphora spp. puparia per infested nest. Right y-axis and corresponding stacked bar graph show the yearly number of infested nests (grey) over the yearly total number of examined nests (i.e., nests that harboured a single clutch that experienced at least one hatching event; white). B, Histogram showing the frequency distribution of the number of Protocalliphora spp. puparia found within infested nests (n = 1227)
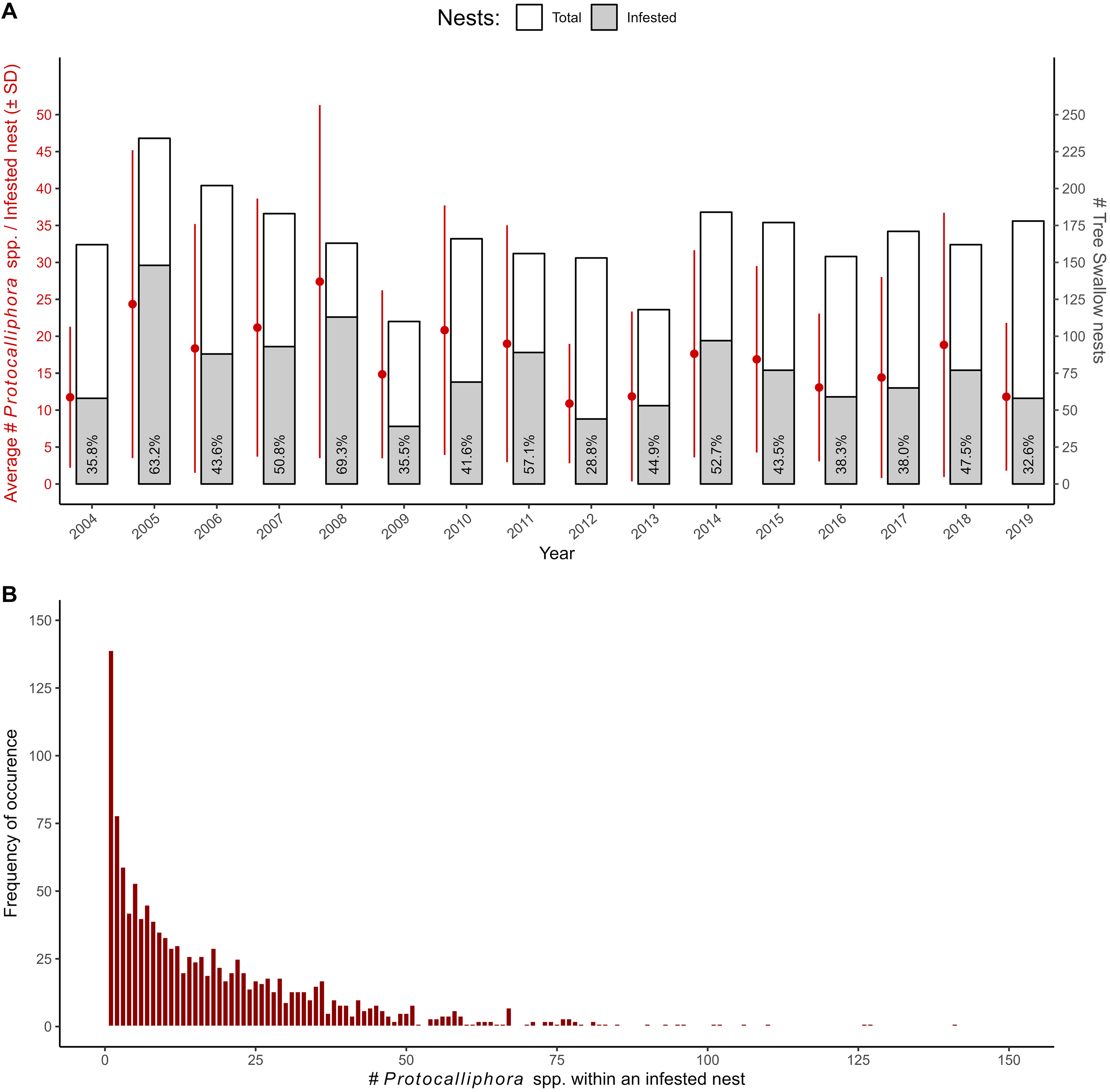
A total of 22 630 puparia were counted among infested nests, resulting in an overall estimated average load (± standard deviation) of 18.44 ± 19.16 Protocalliphora spp. per infested nest. The average number of Protocalliphora spp. per infested nest exhibited variability both within and across years, fluctuating between 10.89 ± 8.08 (2012) and 27.40 ± 23.90 (2008), with a yearly coefficient of variation ranging between 74.21 and 97.11% (Fig. 1A). The overall frequency distribution of the number of Protocalliphora spp. per infested nest was, however, highly skewed to the right with a median of 12 puparia (range: 1–141 puparia; Fig. 1B). Interestingly, most farms showed yearly fluctuations in average Protocalliphora spp. loads per infested nest that followed a global, nonlinear trend, the peaks and troughs of which coincided with those observed for the proportion of nests infested by Protocalliphora spp. (n = 1227 infested nests; GAMM R 2 = 12.2%; Supplementary material, File S3). However, the farms that deviated from the global load trend (i.e., 22.5% of farms) were not the same as those for which the proportion of infested nests and the generalised linear additive mixed model residuals showed no temporal autocorrelation pattern.
Parasitoidism of Protocalliphora spp. by Nasonia spp.
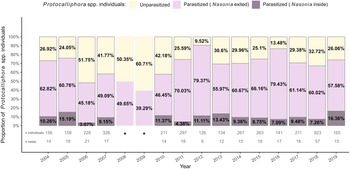
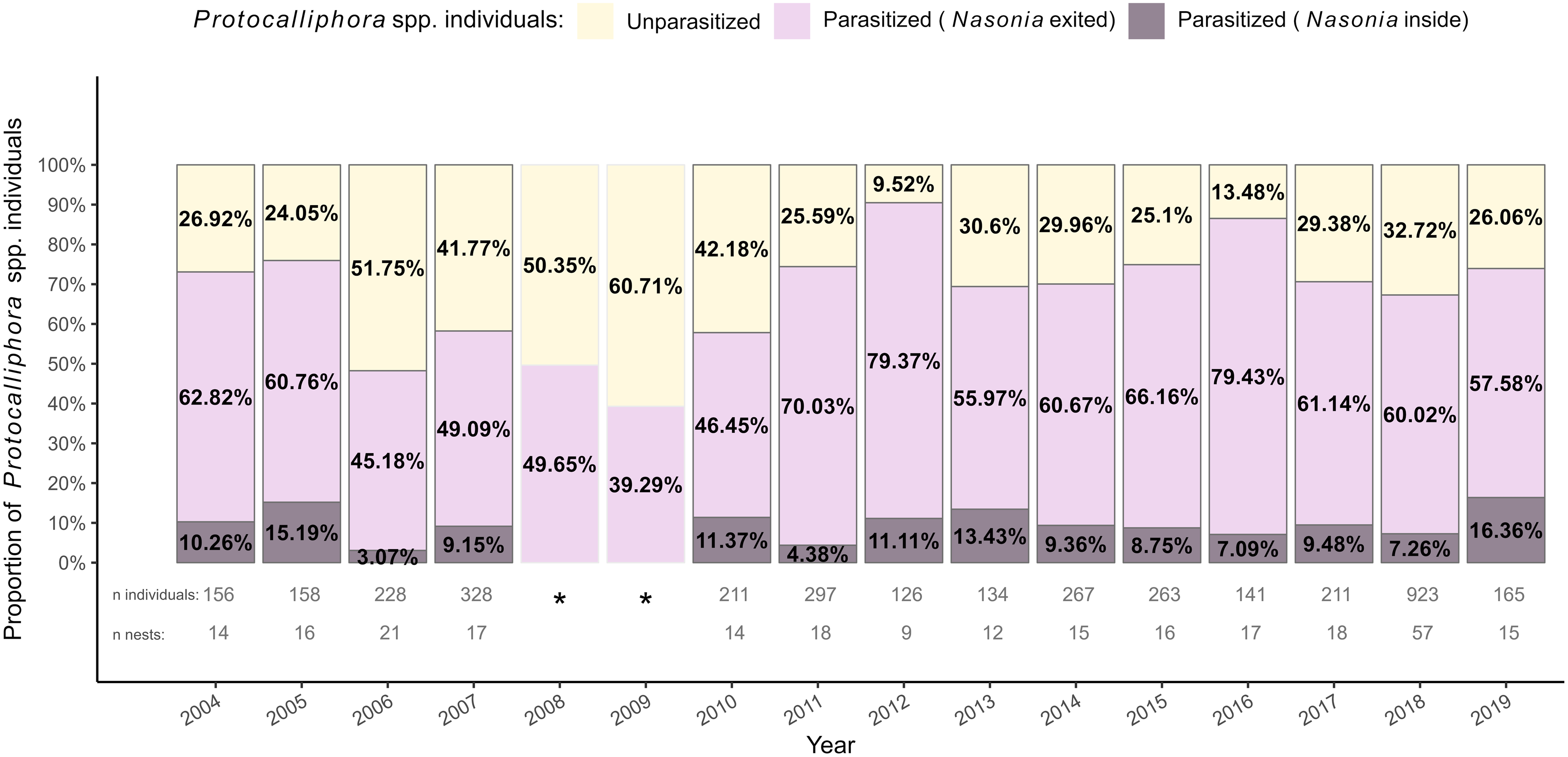
Nasonia spp. likely occurred throughout the study area because they were detected at least once in 30 of the 31 farms from which Protocalliphora spp. puparia were subsampled to estimate parasitoidism rate. Overall, Nasonia spp. parasitised at least one puparium in 85.08% of nests in which Protocalliphora spp. were found and 68.82% of the 3608 subsampled puparia that could be examined (Table 1). Importantly, 12.81% of parasitised Protocalliphora spp. puparia contained nonemerged Nasonia spp. individuals. These nonemerged individuals occurred nearly exclusively at the pupal stage, suggesting that they either had failed to produce adults or had entered diapause for the coming winter. Parasitoidism by Nasonia spp. remained high across years, ranging from 48.25% (2006) to 90.48% (2012) of Protocalliphora spp. puparia (Fig. 2). Note that Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a) estimated parasitoidism rates in 2008 and 2009 based on a mix of reared Protocalliphora spp. pupae and on the presence of exit holes made by Nasonia spp. to exit puparia. They reported among the lowest parasitoidism rates of the study period (i.e., 50.35% and 39.29%, respectively). The proportions of infested Protocalliphora spp. puparia from which Nasonia spp. did not emerge before winter varied substantially, ranging from 3.07% in 2006 to 16.36% in 2019 (Fig. 2).
Stacked bar graph representing the interannual rates of Protocalliphora spp. puparia parasitoidism by Nasonia spp. within tree swallow nests collected in southern Quebec, Canada, between 2004 and 2019. Protocalliphora spp. puparia were classified as unparasitised when they showed no signs of parasitoidism (yellow) and as parasitised if Nasonia spp. exit holes were observed on the puparia (pink) or if Nasonia spp. first instars were found when Nasonia spp. exited Protocalliphora spp. (purple). *Proportions for 2008 and 2009 were calculated from statistics reported by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a, tables 3 and 4). Parasitoidism was determined by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a) using a mix of reared Protocalliphora spp. pupae and based on the presence of holes made by Nasonia spp. to exit puparia
Protocalliphora species composition
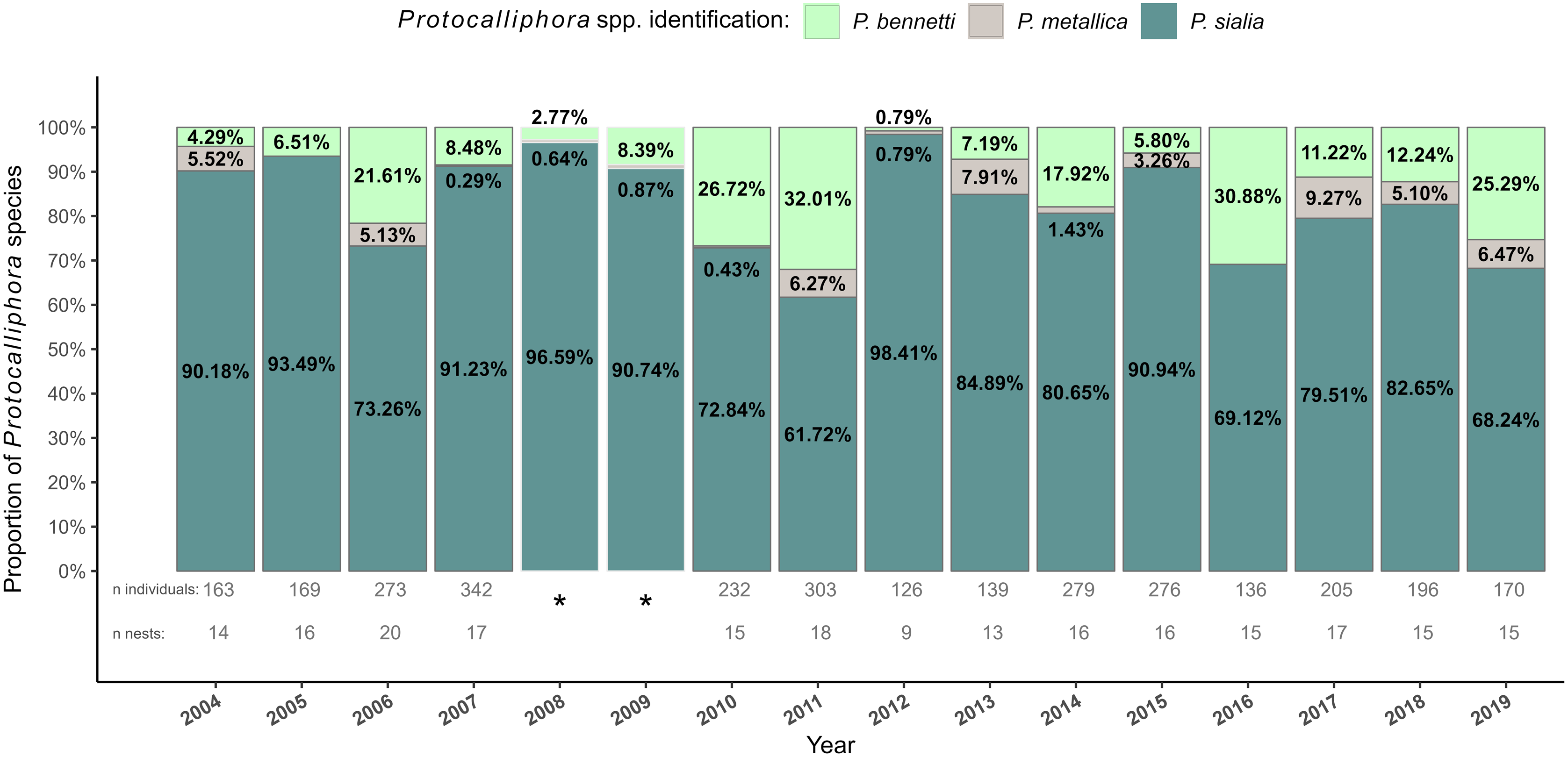
Of the 3009 Protocalliphora spp. puparia suitable for species identification, 82.62% belonged to P. sialia, 15.75% belonged to P. bennetti, and 3.62% belonged to P. metallica (Table 1). Protocalliphora sialia was consistently the most abundant species in each year, ranging from 61.72% in 2011 to 98.41% the following year (Fig. 3). In contrast, P. bennetti exhibited more pronounced fluctuations in relative abundance, ranging from quasi-absence (0.79%) in 2012 to 32.01% in 2011. Protocalliphora metallica, the least abundant species in our study system, also showed important variation in its relative abundance: it was either completely absent from our subsamples in 2005 and 2016 or as or more abundant than P. bennetti was in 2004, 2012, and 2017, but it never exceeded 9.27% (2017) of Protocalliphora puparia in any given year (Fig. 3). Data extrapolated from Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a) for 2008 and 2009 also showed a strong dominance of P. sialia, with proportions of 96.59% and 90.74%, respectively. Overall, all three species were found over most of the study area (Supplementary material, File S4).
Stacked bar graph representing the interannual Protocalliphora species composition within tree swallow nests collected in southern Quebec, Canada, between 2004 and 2019. Three species were identified (P. bennetti, light green; P. metallica, grey; P. sialia, dark green). *Proportions for 2008 and 2009 were derived from Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a, table 1)
Protocalliphora spp. morphometry
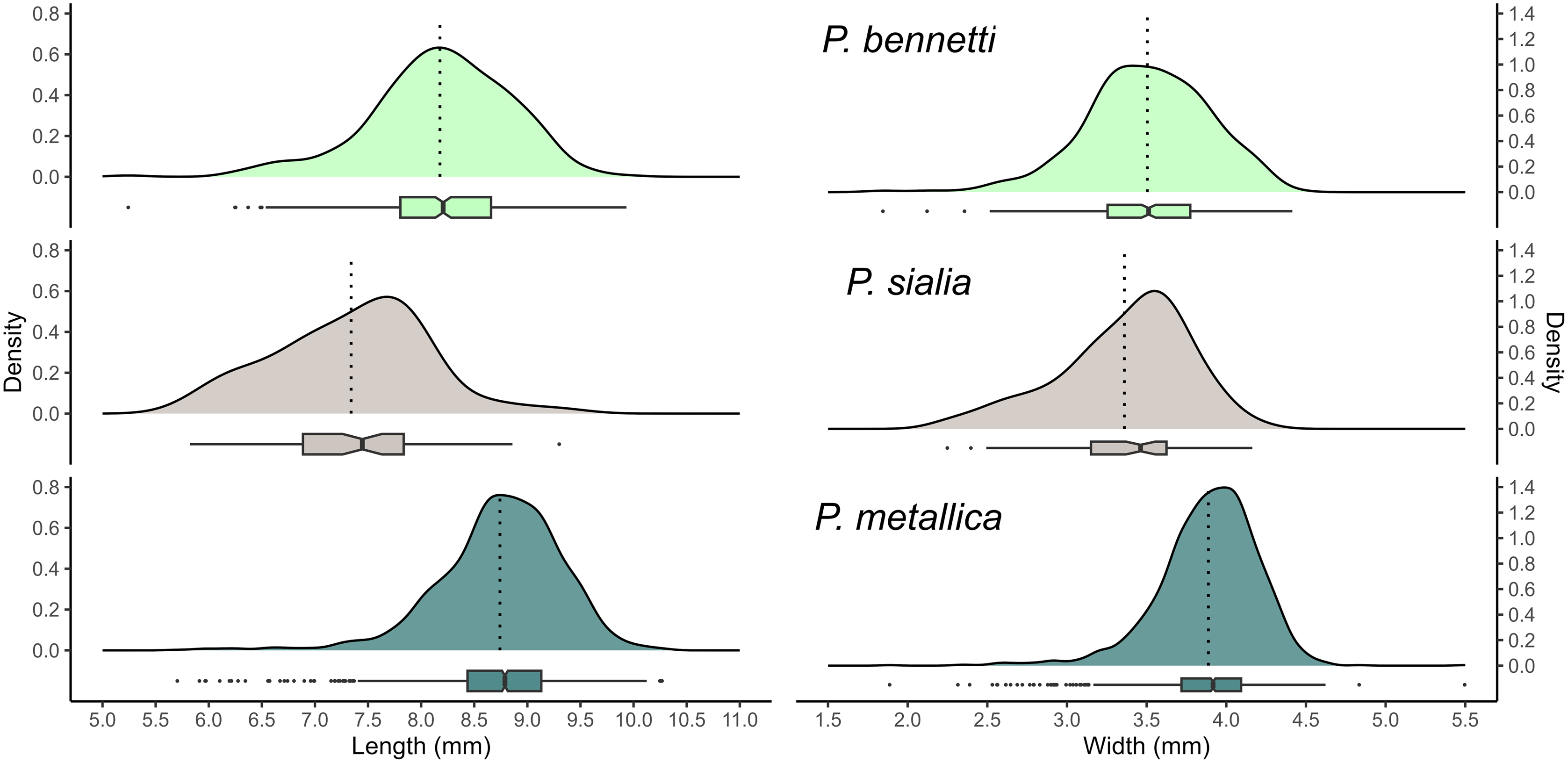
The maximum puparium lengths and widths of the three Protocalliphora species were obtained from a subsample of 1982 individuals (Table 1). The distribution of maximum lengths and widths of the three species was unimodal and relatively symmetrical, although slightly skewed to the left in P. metallica (Fig. 4). Despite substantial intraspecific variation leading to distributional overlap, the generalised linear mixed models showed the species differed in their average morphometry. Protocalliphora bennetti exhibited a mean puparium length (± standard deviation) of 8.18 ± 0.67 mm (range: 5.24–9.93 mm), whereas P. metallica showed a significantly shorter mean length (7.34 ± 0.71 mm; range: 5.82–9.29 mm; P < 0.001) and P. sialia showed a longer one (8.74 ± 0.58 mm; range: 5.70–10.27 mm; P < 0.001). Regarding width, P. metallica had a narrower puparium (3.36 ± 0.41 mm, range: 2.25–4.16 mm; P < 0.001) and P. sialia a wider puparium (3.89 ± 0.32 mm, range: 1.89–5.50 mm; P < 0.001) than P. bennetti did (3.50 ± 0.39 mm, range: 1.84–4.41 mm). The proportion of variance in length and width explained by both the random (i.e., Farm ID and year) and fixed (i.e., species) effects of the linear mixed models was 36.45% and 30.57% for the maximum puparium length and width, respectively. Farm ID and year alone accounted for 51.58% and 44.16% of these proportions, respectively.
Density distribution representing the morphometric measurements (length and width in millimetres) of puparia for Proocalliphora bennetti (light green, n = 347), P. metallica (grey, n = 66), and P. sialia (dark green, n = 1,569) collected in southern Quebec, Canada, between 2004 and 2019 (excluding 2008–2009). Mean (dotted line) and quartiles (via boxplot) are shown
Discussion
Our study presents a rare time series on the prevalence of bird nest infestation by bird blowflies and their abundance within infested nests. We found that infestation rates and loads of Protocalliphora spp. within tree swallow nests were high over a 16-year period (2004–2019), yet highly variable in both space and time along a 10 200-km2 gradient of agricultural intensity in southern Quebec, Canada. Moreover, we observed that these fluctuations potentially followed a recurrent pattern that was shared by approximately 75% of the sampled locations (i.e., farms), suggesting that both large-scale and local conditions or processes may be at play. Our study also shows that, although bird blowfly pupae were subjected to parasitoidic pressures by Nasonia spp. wasps that can vary as much as double among years, these were high enough to kill more than half of the Protocalliphora spp. pupae produced in most years. Such an impact suggests that Protocalliphora spp. and Nasonia spp. may be part of host–parasitoid dynamics that are responsible for the oscillations in bird blowfly infestation rates and loads that we report. Consistent with the previous two-year investigation in our study system by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a, Reference Daoust, Bélisle, Savage, Robillard, Baeta and Brodeur2012b), we found both mono- and multispecific infestations in nests, involving three species of Protocalliphora spp., with P. sialia dominating (> 80% of puparia). However, we found higher proportions of P. bennetti (∼2×) and P. metallica (∼4×) than reported by Daoust et al. (Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a), emphasising the value of multiyear studies in revealing the complete range of species composition variability in these flies. Lastly, we report highly variable puparium length and width, with overlapping frequency distributions, among Protocalliphora species. This finding suggests that these morphometric traits are not reliable as discrete diagnostic characters and should not to be used in isolation for identifying Protocalliphora species (Whitworth Reference Whitworth2002). It also raises important questions about the ecological factors driving this variability and the potential consequences of size variation for emergence success and host selection by parasitoids.
Spatiotemporal variation in Protocalliphora spp. prevalence and load
The prevalence and intensity of bird nest infestations by Protocalliphora spp. have generally been found to vary widely in both space and time. For example, Bennett and Whitworth’s (Reference Bennett and Whitworth1992) comprehensive examination across 73 bird species and 4668 sampled nests revealed infestation rates of 52% within the Rocky Mountain region (Utah, United States of America) versus 24% in the Great Lakes region (Ontario, Canada). The mechanisms underlying such variation likely involve multiple dependent and interactive factors, and few studies have attempted to quantify the importance and spatiotemporal scales at which they operate. Although large-scale geographic variation may result from differences in climate, landscape habitat composition, bird host community (species composition and abundance), and parasitoid pressure (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989; Bennett and Whitworth Reference Bennett and Whitworth1992; Daoust et al. Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a, Reference Daoust, Bélisle, Savage, Robillard, Baeta and Brodeur2012b; Garrido-Bautista et al. Reference Garrido-Bautista, Moreno-Rueda, Baz, Canal, Camacho and Cifrián2020), local variation may originate in factors as specific as bird host brood size, bird host hatching date (Williams Reference Williams2017; Wolfe-Merritt et al. Reference Wolfe-Merritt, Hartman, Barragan, Hellman, Pigott, Rodríguez-Ferraro and Tori2022), and nest type and material affecting the nest microclimate (Dawson et al. Reference Dawson, O’Brien and Mlynowski2011; Griebel et al. Reference Griebel, Dawson and Clark2020). With respect to tree swallows breeding within nest boxes, previous research reported Protocalliphora spp. infestation rates ranging from 89% to 100% in most study sites across Canada (Rendell and Verbeek Reference Rendell and Verbeek1996; Dawson et al. Reference Dawson, Hillen and Whitworth2005; Gentes et al. Reference Gentes, Whitworth, Waldner, Fenton and Smits2007; Griebel et al. Reference Griebel, Dawson and Clark2020). Those reported infestation rates are generally much higher than those we found either across years or across farms within our study area (overall rate = 45.90%). In contrast to other study sites, many of which were located near wetlands, ours were located on farms distributed along a gradient of agricultural intensity with little, if any, wetland cover (Garrett et al. Reference Garrett, Pelletier, Garant and Bélisle2022a). Because of this, we hypothesise that the much lower infestation rates we observed are linked to intensive agricultural practices that may impact Protocalliphora spp. via multiple pathways, including directly through wetland drainage or pesticide use or indirectly by reducing the density of floral resources and bird hosts (e.g., Robert et al. Reference Robert, Hachey, Lepage and Couturier2019; Wagner et al. Reference Wagner, Grames, Forister, Berenbaum and Stopak2021; Powell et al. Reference Powell, Garrett, Roy, Oliver, Larrivée and Bélisle2024). Evidence for such negative impacts has also been reported in our study area by Daoust et al. (Reference Daoust, Bélisle, Savage, Robillard, Baeta and Brodeur2012b), who found lower loads of Protocalliphora spp. in tree swallow nests located within more agro-intensively worked landscapes during a two-year period (2008–2009).
Annual infestation rates of tree swallow nests by Protocalliphora spp. varied between 29% and 63% across the 16 years of our study. This level of interannual variation, which was observed across the entire study area (i.e., 10 200 km2), was comparable to what we observed among nest boxes from a single farm (i.e., < 1 km2; Supplementary material, File S5). Wesołowski (Reference Wesołowski2001) also reported highly variable annual nest infestation rates by Protocalliphora spp. in marsh tits, Parus palustris (Linnaeus) (Passeriformes: Paridae), that ranged from 27% to 88% in four 0.5-km2 plots separated by 1–2 km during an eight-year period in eastern Poland. Similarly, Musgrave et al. (Reference Musgrave, Bartlow and Fair2019) found annual infestation rates between 30% and 91% in western bluebird, Sialia mexicana Swainson (Passeriformes: Turdidae), nests monitored over 17 years in an approximately 100-km2 area of northern New Mexico, United States of America. Despite potential confounding effects, the above comparisons suggest that nest infestation rates by Protocalliphora spp. typically vary substantially among years across a range of spatial scales. Given the lack of reported time series of infestation rates and loads in the literature (but see Merino et al. Reference Merino, García-Del Río, Castaño-Vázquez and Merino2025), it remains largely unknown how these variables are structured in time and how such temporal structures vary across spatial scales and ecological contexts, hindering investigation into the ecological mechanisms underlying their origin. Our observation that both yearly fluctuations in bird blowfly nest infestation rates and loads were to some degree recurrent and synchronous among approximately 75% of the farms sampled over 10 200 km2 nonetheless suggests that regional (e.g., climatic), local (e.g., crop rotations), and density-dependent (e.g., parasitoidic pressure by Nasonia spp.) processes may be at play.
The observed loads of Protocalliphora spp. within infested nests exhibited a right-skewed distribution, a phenomenon commonly observed in the field in parasitology (Wilson and Grenfell Reference Wilson and Grenfell1997). Furthermore, a high degree of variability between nests, between sites, and across years has been reported (Griebel and Dawson Reference Griebel and Dawson2019; Remeš and Krist Reference Remeš and Krist2005). Part of the interannual variation is often associated with weather (Merino and Potti Reference Merino and Potti1996; Dawson et al. Reference Dawson, Hillen and Whitworth2005; Castaño-Vazquez et al. Reference Castaño-Vázquez, Schumm, Bentele, Quillfeldt and Merino2021; Mennerat et al. Reference Mennerat, Charmantier, Perret, Hurtrez-Boussès and Lambrechts2021; Merino et al. Reference Merino, García-Del Río, Castaño-Vázquez and Merino2025), and spatial variation is typically linked to microhabitat and nest-specific factors, such as the amount of feather lining or the species and number of nestling hosts (Eeva et al. Reference Eeva, Lehikoinen and Nurmi1994; Tomás et al. Reference Tomás, Merino, Moreno, Morales and Martínez-de La Puente2007; Dawson et al. Reference Dawson, O’Brien and Mlynowski2011; Williams Reference Williams2017; Wolfe-Merritt et al. Reference Wolfe-Merritt, Hartman, Barragan, Hellman, Pigott, Rodríguez-Ferraro and Tori2022). In our study system, part of the between-farm variation likely results from the diversified agricultural contexts in which tree swallows breed. Daoust et al. (Reference Daoust, Bélisle, Savage, Robillard, Baeta and Brodeur2012b) found fewer Protocalliphora spp. in tree swallow nests located in more agro-intensive areas, particularly in maize and soybean monocultures. This pattern may result from several direct and indirect effects associated with intensive agriculture, such as reduced shelter and food resources for adult flies, lower avian host availability at both the landscape and brood scale, and increased toxicological effects from pesticide exposure either directly or through their avian hosts (Daoust et al. Reference Daoust, Bélisle, Savage, Robillard, Baeta and Brodeur2012b). Because Protocalliphora spp. loads are typically inferred from puparia counts after nestlings have left their nests (Jánošková et al. Reference Jánošková, Országh, Jamriška and Kopáni2010), estimates reflect only the fraction of individuals that successfully pupated. In our case, they may underestimate the prevalence and load of Protocalliphora spp. in harsher environments. Given the scarcity of studies that have examined the effects of agricultural intensity on ectoparasites of wild birds, more extensive and long-term research is needed to determine the mechanisms by which agriculture generates spatiotemporal variation and ultimately shapes host–parasite dynamics.
Parasitoidism of Protocalliphora spp. by Nasonia spp.
Interannual variation in the proportions of puparia infested by Nasonia spp. showed no clear patterns while ranging between 48.25% and 90.48% among years, with an overall rate of 68.82%. These proportions are generally higher than those reported by Jamriška and Lučeničová (Reference Jamriška and Lučeničová2019), who observed an average parasitoidism rate of 37.91% over four years in Slovakia, and those reported by Bennett and Whitworth (Reference Bennett and Whitworth1991), who reported an overall rate of about 17% from Utah and Ontario, in North America. In contrast, Grillenberger et al. (Reference Grillenberger, Van De Zande, Bijlsma, Gadau and Beukeboom2009) reported a rate of approximately 44% in New York State, United States of America, and Garrido-Bautista et al. (Reference Garrido-Bautista, Moreno-Rueda, Baz, Canal, Camacho and Cifrián2020) reported rates from 47% to 56% for southern Spain and from 61% to 79% for central Spain, figures much closer to our own. Although more extensive longitudinal and ecological studies such as González-Bernardo et al. (Reference Gonzalez-Bernardo, Moreno-Rueda, Camacho, Martinez-Padilla, Potti and Canal2025) are needed, current findings suggest that geographical and large-scale environmental contexts may significantly influence the likelihood of Nasonia spp. parasitoidism (Garrido-Bautista et al. Reference Garrido-Bautista, Norte, Moreno-Rueda and Nadal-Jiménez2024), a primary determinant of Protocalliphora spp. emergence success. However, it is crucial to recognise that, in addition to the substantial control exerted by Nasonia spp. on bird blowfly communities, uninfested Protocalliphora spp. individuals may also die during pupation due to other ecological factors, such as environmental contamination. The specific impacts of those factors on Protocalliphora spp. emergence success have yet to be thoroughly investigated and quantified.
Nasonia spp. parasitoid wasps have mostly served as model species in numerous laboratory studies addressing themes in genetics, developmental biology, and behavioural ecology (Werren and Loehlin Reference Werren and Loehlin2009; Niehuis et al. Reference Niehuis, Gibson, Rosenberg, Pannebakker, Koevoets and Judson2010; Nadal-Jimenez et al. Reference Nadal-Jimenez, Frost, Norte, Garrido-Bautista, Wilkes and Connell2023). The ecological factors driving host selection and parasitoidism by Nasonia spp. under natural conditions are thus poorly understood (Peters Reference Peters2010; Peters and Abraham Reference Peters and Abraham2010; Garrido-Bautista et al. Reference Garrido-Bautista, Moreno-Rueda, Baz, Canal, Camacho and Cifrián2020, Reference Garrido-Bautista, Norte, Moreno-Rueda and Nadal-Jiménez2024). This is particularly true given the wide diversity of hosts in which Nasonia spp. can lay eggs and the potential for regional variations due to their widespread distribution (Skovgård and Jespersen Reference Skovgård and Jespersen1999). This leaves the causes of yearly fluctuations in Protocalliphora spp. puparia parasitoidic events and of winter diapause within the host puparia unresolved. Future research on Nasonia spp. ecology would likely benefit from considering factors such as vegetation type and density, because these features may influence host parasitoidism by affecting the availability of food and shelter (Wylie Reference Wylie1958). Avian nest availability associated with these environmental factors could also potentially induce host-dependent effects, contributing to an increased incidence of Nasonia spp. parasitoidism on Protocalliphora spp.
The present study reports the first quantitative exploration of dormancy and mortality of Nasonia spp. within their host in the wild. We noted consistently low yearly proportions of Protocalliphora spp. puparia containing dormant or dead Nasonia spp. (range: 3.07–16.36%), with no discernible temporal trend. Several biotic and abiotic factors, such as weather, luminosity, or host and parasitoid body condition, may affect the production of diapausing Nasonia spp. offspring (Saunders Reference Saunders and Waddington1969; Yoder et al. Reference Yoder, Rivers and Denlinger1994; Bertossa et al. Reference Bertossa, van Dijk, Beersma and Beukeboom2010; Paolucci et al. Reference Paolucci, Van De Zande and Beukeboom2013; Li et al. Reference Li, Zhang, Zhang, Chen and Denlinger2014). Mechanisms linked to phenology could thereby affect the dormancy and survival of Nasonia spp. larvae. For instance, bird blowfly pupation phenology, which in turn determines host availability and development conditions for Nasonia spp., must partly depend on avian host breeding phenology. Moreover, agricultural intensity may affect Nasonia spp. development and emergence success because pesticide use can alter parasitoid wasps’ cognition and fitness (Schöfer et al. Reference Schöfer, Ackermann, Hoheneder, Hofferberth and Ruther2023, Reference Schöfer, Ratschmann and Ruther2024; Sulg et al. Reference Sulg, Kaasik, Kallavus and Veromann2023) and the availability and quality of their hosts (Daoust et al. Reference Daoust, Bélisle, Savage, Robillard, Baeta and Brodeur2012b; Li et al. Reference Li, Zhang, Zhang, Chen and Denlinger2014).
Protocalliphora species composition and morphometry
Bird nestlings from the Nearctic region are hosts to at least 28 Protocalliphora species (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989; updated by Whitworth Reference Whitworth2002). Although some bird blowfly species appear to specialise on specific hosts through behavioural adaptations, some parasitise a wide array of bird species (Bennett and Whitworth Reference Bennett and Whitworth1992), and the occurrence of multiple Protocalliphora species within a single host nest is not rare (Daoust et al. Reference Daoust, Savage, Whitworth, Bélisle and Brodeur2012a). Our longitudinal study, one of the few that describe Protocalliphora species composition, reports that such compositions can vary substantially across years. Indeed, our results show that certain species (in this case, P. metallica) may be practically absent in some years, only to reappear in relatively high numbers the following year. Even the highly prevalent P. sialia fluctuated widely, representing 58.93% of the Protocalliphora specimens in 2011 but being almost the only species found in infested nests the following year (98.32%). The results show that longitudinal studies considering variability in species composition are essential before conclusions about host specialisation to avian parasites and mixed infestations can be drawn.
Although descriptive statistics of some morphometric measurements of Protocalliphora spp. puparia can be found in the literature, few, if any, studies report the actual frequency distributions of these features that may help discriminate among species of this cryptic group (Heeb et al. Reference Heeb, Kölliker and Richner2000; Whitworth Reference Whitworth2002, Reference Whitworth2003; Jamriška and Modrý Reference Jamriška and Modrý2013). The mean, standard deviation, and range estimates of puparium length and width for P. bennetti, P. metallica, and P. sialia that we obtained via photogrammetry align well with published estimates (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989; Whitworth Reference Whitworth2002). However, the fact that density plots highlighted substantial intraspecific variation in both puparium length and width, as well as significant overlap among the three studied species, indicates that these metrics should not be used on their own for Protocalliphora spp. identification, even within a small regional species pool. Nonetheless, the ecological factors contributing to this high intraspecific variation, with certain puparia being more than twice as large and long as others, are unclear. Sexual dimorphism (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989) or factors such as host availability and quality, as well as developmental period and temperature, which are often interconnected, have been hypothesised as causes of size variation in Protocalliphora spp. For instance, underfed larvae may conclude their development as ‘runts’ (Sabrosky et al. Reference Sabrosky, Bennett and Whitworth1989; Bennett and Whitworth Reference Bennett and Whitworth1991). Alternatively, intensive agricultural practices, including pesticide use, have been reported as either direct or indirect drivers of phenotypic variation (Wisniewska and Prokopy Reference Wisniewska and Prokopy1997; Verheyen and Stoks Reference Verheyen and Stoks2019; Gérard et al. Reference Gérard, Baird, Breeze, Dominik and Michez2022; Ponce-Méndez et al. Reference Ponce-Méndez, García-Martínez, Serna-Lagunes, Lasa-Covarrubias, Presa-Parra, Murguía-González and Llarena-Hernández2022) and pupation success (Heneberg et al. Reference Heneberg, Bogusch, Astapenková and Řezáč2020; Mahdjoub et al. Reference Mahdjoub, Blanckenhorn, Lüpold, Roy, Gourgoulianni and Khelifa2020) in nontargeted arthropod species.
All the ecological parameters investigated in the present study showed substantial variation in space and time or among individuals, indicating that future studies addressing the mechanisms driving Protocalliphora spp. host–parasite dynamics adopt a long-term monitoring and multitrophic approach in contrasting environments. Investigating the tri-trophic relationships involving birds, Protocalliphora spp., and Nasonia spp. across diverse agroecosystems presents a promising avenue for such research. Given the complex interplay among environmental conditions and multitrophic interactions, we further suggest that such endeavours be tackled using a causal inference framework to limit biases, especially because each trophic level may respond to different factors at different spatiotemporal scales and potentially nonlinearly or in different directions, which has the potential to amplify, attenuate, mask, and even create effects (Arif and MacNeil Reference Arif and MacNeil2023; Grace Reference Grace2024).
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.4039/tce.2026.10052.
Acknowledgements
The authors are deeply grateful to the many farm owners who have collaborated in our long-term monitoring programme. The authors also sincerely thank T.L. Whitworth for his helpful exchanges and expert guidance in the identification of challenging Protocalliphora individuals. They thank the Université de Sherbrooke for its institutional support and the funding agencies that made this research possible, including the Natural Sciences and Engineering Research Council of Canada (NSERC), the Fonds de recherche du Québec – Nature et technologies (FRQ-NT), and the Canada Research Chairs programme. They warmly acknowledge the contribution of the many students, field assistants, and technicians, and especially of L. Glaude and Y. Sageau, who participated in the collection and sorting of nests over the years. The authors specially thank D. Garneau for his generous assistance with all aspects of imagery. This study was conducted with the approval of the Université de Sherbrooke animal care committee, and the authors gratefully recognise all those whose efforts on the ground and behind the scenes contributed to making the work possible.