Introduction
Maternal mental health is an important determinant of healthy child development (Entringer, Buss, & Wadhwa, Reference Entringer, Buss and Wadhwa2015; Meaney, Reference Meaney2018; O’Donnell, Glover, Barker, & O’Connor, Reference O’Donnell, Glover, Barker and O’Connor2014). Unfortunately, maternal mental health problems during the perinatal period are highly prevalent, affecting roughly one in seven pregnancies (Kendig et al., Reference Kendig, Keats, Hoffman, Kay, Miller, Moore Simas and Lemieux2017). Among different forms of psychopathology, depression is especially frequent and pernicious as it has been associated with a diverse range of adverse offspring neurodevelopmental outcomes, including emotional and behavioral problems and impaired cognitive development (Glover, Reference Glover2014; Urizar & Muñoz, Reference Urizar and Muñoz2021).
These associations between maternal depression during pregnancy and child behavioral outcomes may be mediated by changes in prenatal brain development. Fetal brain development is characterized by rapid growth and a high degree of plasticity and sensitivity to signals from the intrauterine environment, which is thought to promote adaptation to environmental conditions ex utero. Thus, depression-related variation in maternal–placental–fetal stress biology may lead to alterations in fetal brain development, including white matter pathways, which may contribute to increased risk for adverse long-term developmental outcomes. For instance, variation in newborn white matter microstructure has been prospectively associated with language and visuocognitive development, performance IQ, internalizing problems, inattention, and social deficits (Brenner et al., Reference Brenner, Smyser, Lean, Kenley, Smyser, Cyr and Rogers2021; Keunen et al., Reference Keunen, Benders, Leemans, Fieret-Van Stam, Scholtens, Viergever and van den Heuvel2017; Sket et al., Reference Sket, Overfeld, Styner, Gilmore, Entringer, Wadhwa and Buss2019; Stjerna et al., Reference Stjerna, Sairanen, Gröhn, Andersson, Metsäranta, Lano and Vanhatalo2015; Vassar et al., Reference Vassar, Schadl, Cahill-Rowley, Yeom, Stevenson and Rose2020).
Since depressive symptoms often persist during the postnatal period (Dipietro, Costigan, & Sipsma, Reference Dipietro, Costigan and Sipsma2008) and, therefore, continue to influence developmental outcomes, assessing the brain near the time of birth presents a unique opportunity to understand fetal programming effects while limiting the influence of the postnatal environment. Toward this, several studies to date have investigated the association between maternal depression (or depression/anxiety) during pregnancy and newborn offspring white matter microstructure using diffusion tensor imaging (DTI). DTI measures properties of water diffusion in the brain that are themselves reflective of white microstructure (e.g. axon size and density, myelination). Common DTI metrics include fractional anisotropy (FA, the degree of anisotropic diffusion), axial diffusivity (AD, diffusion along the principal eigenvector, typically the white matter bundle), and radial diffusivity (RD, diffusion perpendicular to the principal eigenvector). Variation in these metrics in newborns and infants may be the result of a range of developmental or pathological events. In general, higher FA and lower RD, AD indicate more mature white matter integrity and/or myelination, corresponding to changes in these metrics observed across the first year of life (Rasmussen et al., Reference Rasmussen, Kruggel, Gilmore, Styner, Entringer, Consing and Buss2017; Soares, Marques, Alves, & Sousa, Reference Soares, Marques, Alves and Sousa2013; Stephens et al., Reference Stephens, Langworthy, Short, Girault, Styner and Gilmore2020). Studies of prenatal depression and newborn white matter microstructure have reported heterogeneous findings across a variety of regions, including both lower and higher regional or tract-specific FA in association with maternal depression and anxiety (Demers et al., Reference Demers, Bagonis, Al-Ali, Garcia, Styner, Gilmore and Davis2021; Graham et al., Reference Graham, Jiang, McCorkle, Bellando, Sorensen, Glasier and Ou2020; Lautarescu et al., Reference Lautarescu, Bonthrone, Pietsch, Batalle, Cordero-Grande, Tournier and Counsell2022; Rifkin-Graboi et al., Reference Rifkin-Graboi, Bai, Chen, Hameed, Sim, Tint and Qiu2013, Reference Rifkin-Graboi, Meaney, Chen, Bai, Hameed, Tint and Qiu2015), as well as higher and lower AD and RD (Dean et al., Reference Dean, Planalp, Wooten, Kecskemeti, Adluru, Schmidt and Davidson2018; Lautarescu et al., Reference Lautarescu, Pecheva, Nosarti, Nihouarn, Zhang, Victor and Counsell2020; Rifkin-Graboi et al., Reference Rifkin-Graboi, Bai, Chen, Hameed, Sim, Tint and Qiu2013, Reference Rifkin-Graboi, Meaney, Chen, Bai, Hameed, Tint and Qiu2015). Furthermore, some studies observed no associations between maternal depression and newborn offspring DTI metrics, only under specific conditions – for example, only in treated versus untreated depression or in a sex-specific manner (Hashempour et al., Reference Hashempour, Tuulari, Merisaari, Acosta, Lewis, Pelto and Karlsson2023; Jha et al., Reference Jha, Meltzer-Brody, Steiner, Cornea, Woolson, Ahn and Knickmeyer2016; Lean et al., Reference Lean, Smyser, Brady, Triplett, Kaplan, Kenley and Rogers2022; Roos et al., Reference Roos, Wedderburn, Fouche, Joshi, Narr, Woods and Donald2022). Results have been similarly inconsistent with respect to other diffusion magnetic resonance imaging (MRI) metrics in newborns exposed to maternal depression, such as neurite density and fiber density (Dean et al., Reference Dean, Planalp, Wooten, Kecskemeti, Adluru, Schmidt and Davidson2018; Lautarescu et al., Reference Lautarescu, Bonthrone, Pietsch, Batalle, Cordero-Grande, Tournier and Counsell2022).
Besides methodological differences between previously published studies and small sample sizes that may have led to inconclusive results, one potential reason for these contrary findings may be that depression is a heterogeneous disease in terms of symptom patterns, course trajectories, comorbidities, but also in underlying biology (Buch & Liston, Reference Buch and Liston2021; Milaneschi, Lamers, Berk, & Penninx, Reference Milaneschi, Lamers, Berk and Penninx2020). Exposure to childhood maltreatment (CM) is an important risk factor for the development of depression across the lifespan and has been shown to moderate many aspects of the condition that exhibit heterogeneity, such as age at onset and treatment response (Nelson, Klumparendt, Doebler, & Ehring, Reference Nelson, Klumparendt, Doebler and Ehring2017; Teicher & Samson, Reference Teicher and Samson2013). There is also ample evidence for differential biological dysregulation in relation to depression, depending on the presence or absence of maltreatment, which may also extend into pregnancy, having the potential to ultimately guide fetal development (Danese et al., Reference Danese, Moffitt, Pariante, Ambler, Poulton and Caspi2008; de Punder et al., Reference de Punder, Entringer, Heim, Deuter, Otte, Wingenfeld and Kuehl2018; Epstein et al., Reference Epstein, Houfek, Rice, Weiss, French, Kupzyk and Pullen2020; Heim et al., Reference Heim, Mletzko, Purselle, Musselman and Nemeroff2008; Walsh et al., Reference Walsh, Basu, Werner, Lee, Feng, Osborne and Monk2016). Furthermore, CM has repeatedly been shown to have widespread and long-lasting endocrine, immune, and epigenetic consequences. These changes may be carried forward into a later pregnancy (Blackmore et al., Reference Blackmore, Moynihan, Rubinow, Pressman, Gilchrist and O’Connor2011; Moog et al., Reference Moog, Heim, Entringer, Simhan, Wadhwa and Buss2022) and may interact with additional prenatal exposures, including depression, potentially influencing fetal brain development. As such, maternal depression during pregnancy may be associated with different gestational biological profiles depending on a history of maltreatment, which, in turn, may result in differential effects on fetal white matter development.
The objective of the present study is to test the corollary and novel hypothesis that the association of maternal depressive symptomatology with offspring white matter microstructure is moderated by maternal CM history. Specifically, we aim to test whether the relationship between prenatal depressive symptoms and newborn white matter microstructure in the group of mother–child dyads with maternal history of CM differs from the relationship in the group without maternal CM history. To limit confounding by postnatal influences, we used DTI data from infants acquired close to birth, and because this type of data is still scarce, the sample was combined across two cohorts with an identical imaging setup to increase power for statistical inference. We focused on average global metrics of white matter microstructure due to the heterogeneous findings in previous studies regarding regions and tracts involved and limited power to perform analyses of a variety of individual tracts.
Methods
Study sample and design
The research question was investigated in a subsample of two longitudinal prospective cohort studies in the United States that are part of the NIH Environmental Influences on Child Health Outcomes program (Knapp et al., Reference Knapp, Kress, Parker, Page, McArthur and Gachigi2023). One study was conducted at the University of Pittsburgh Medical Center (subsequently referred to as “Cohort 1”). The study enrolled pregnant women attending antenatal care at clinics affiliated with the UPMC Magee-Women’s Hospital in the Pittsburgh metropolitan area. The second study was conducted at the University of Rochester Medical Center (subsequently referred to as “Cohort 2”). The study enrolled pregnant women attending antenatal care at clinics affiliated with the Medical Center in Rochester, New York (O’Connor et al., Reference O’Connor, Best, Brunner, Ciesla, Cunning and Kapula2021). For inclusion and exclusion criteria of both cohorts, please refer to Supplementary Materials. The present analysis included n = 55 mother–child dyads from Cohort 1 enrolled between November 2019 and May 2021, and n = 35 mother–child dyads from Cohort 2 enrolled between December 2015 and December 2020, whose children underwent MRI of the brain shortly after birth. Both cohorts performed serial assessments of psychological, biological, and biosocial measures across pregnancy. In Cohort 1, two study visits were conducted during pregnancy (T1: <27 weeks’ gestation and T2: 30–35 weeks’ gestation), and in Cohort 2, study visits were conducted three times across pregnancy (T1: 12.34 ± 1.5 weeks’ gestation, T2: 20.69 ± 1.7 weeks’ gestation, and T3: 31.48 ± 1.4 weeks’ gestation). After birth, an MRI scan of the offspring’s brain was performed within 90 days of birth (mean: 36.24 ± 16.2 days). All participants provided written informed consent (parents on behalf of their children), and all assessments were approved by the respective institutional review boards.
Maternal depressive symptoms
In both cohorts, maternal depressive symptoms during pregnancy were assessed with the Edinburgh Postnatal Depression Scale (EPDS) (Cox, Holden, & Sagovsky, Reference Cox, Holden and Sagovsky1987), which has been developed as a screening tool for postnatal depression and has also been validated for use during pregnancy (Cox, Holden, & Henshaw, Reference Cox, Holden and Henshaw2014). The EPDS is a self-report questionnaire comprising 10 items that are aggregated into a sum score with a possible range of 0–30. The EPDS was completed by the pregnant women at each pregnancy visit. In Cohort 1, n = 4 participants had missing data at the first time point, and n = 9 had missing data at the second time point. In Cohort 2, n = 2 subjects had missing data at the first time point. EPDS scores correlated strongly between the time points (r: 0.57–0.79). A mean depression score was computed across all available time points and used in analyses.
Maternal CM
Maternal exposure to CM was assessed during pregnancy with the Childhood Trauma Questionnaire (CTQ) (Bernstein & Fink, Reference Bernstein and Fink1998) in Cohort 1 and with the Adverse Childhood Experiences (ACE) scale (Felitti et al., Reference Felitti, Anda, Nordenberg, Williamson, Spitz, Edwards and Marks1998) in Cohort 2. Both measures assess exposure to five forms of CM before the age of 18 years: emotional, physical, and sexual abuse, as well as emotional and physical neglect. The ACE scale also assesses five additional exposures termed household challenges, which were not considered for the purpose of the present study. The CTQ measures each form of CM with five items on a 5-point Likert scale, indicating frequency of exposure. Cut scores exist for each subscale to categorize exposures based on severity. The ACE measures each form of CM via one item with a binary response option (yes/no). CTQ responses for each subscale were categorized using the moderate to severe cut score for the respective subscale. CM exposures measured with the CTQ and with the ACE were then harmonized into one binary score, indicating exposure to at least one form of CM (CM+) versus no exposure to all forms of CM (CM-). See Supplementary Materials for details on the harmonizing procedure.
Magnetic resonance imaging
At both sites, MRI scans were acquired during natural sleep using a 64-channel head coil at 3T field strength on a Siemens PRISMA system in unsedated neonates. After feeding and soothing to the point of sleep, infants were swaddled and placed in a vacuum pillow. Trained study personnel observed infants throughout the duration of scans, and a pulse-oximeter was attached to the infant’s foot, monitoring heart rate and oxygen saturation. In Cohort 1, an MRI scan was attempted in N = 130 newborns. (Partial) MRI data were acquired in n = 101 newborns, and DTI data were available for n = 62, of which n = 7 had to be excluded due to quality issues, resulting in a sample size of n = 55. In Cohort 2, an MRI scan was attempted in N = 105 newborns. (Partial) MRI data were acquired in n = 98 newborns, and DTI data were collected in n = 76. Of these, n = 41 were excluded during quality control, resulting in a sample size of n = 35 (see below for more information regarding quality control). The high proportion of excluded subjects in this cohort was likely due to increased motion and perception of environmental changes related to older infant age. The same scanning protocol was used at both sites. High-resolution anatomical scans, including T1-weighted (Magnetization-Prepared Rapid Gradient-Echo, repetion time (TR)/echo time (TE)/inversion time (TI) = 2,400/2.22/1,000 ms, Flip Angle = 8 degrees, Matrix = 208 × 300 × 320, Resolution = 0.8 × 0.8 × 0.8 mm, 6m38s) and T2-weighted (Turbo Spin Echo, TR/TE = 3,200/563 ms, Matrix = 208 × 300 × 320, Resolution = 0.8 × 0.8 × 0.8 mm, 5m57s) images were acquired. The 80-direction diffusion weighted protocol (Echo Planar Imaging, TR/TE = 3,222/89.2 ms, Matrix = 140 × 140 × 92 mm, Resolution = 1.5 × 1.5 × 1.5 mm, Partial Fourier = 6/8, GRAPPA Phase Encoding Acceleration Factor = 4, 37 unique directions at b = 1,500s/mm2, 37 unique directions at b = 3,000 s/mm2, 6 at b = 0 s/mm2) was collected in a forward/reverse phase-encode manner.
Image processing of diffusion-weighted data
A rigorous diffusion MRI quality control was applied to all diffusion-weighted imaging (DWI) data using a combination of DTIPrep (www.nitrc.org/projects/dtiprep), which performs slice-wise and gradient-wise artifact detection and removal, as well as FSL (v6.0.3), jointly correcting for eddy current, motion, susceptibility, and related artifacts. As an additional quality control step, interactive tractography was performed in Slicer (http://www.slicer.org) and visually assessed for artifacts undetectable by voxel-wise inspection, such as any consistently observed directional biases. Skull and non-brain tissue were masked using FSL’s Brain Extraction Tool on the geometric mean of the DWI image, followed by a manual correction.
Using the UNC − Utah National Alliance for Medical Image Computing DTI framework (Verde et al., Reference Verde, Budin, Berger, Gupta, Farzinfar, Kaiser and Styner2014), a study-specific DTI atlas was created from all high-quality subject DTI data (Cohort 1 [n = 55] and Cohort 2 [n = 35]). The framework represents a coherent, end-to-end toolset for atlas fiber tract-based DTI analysis encompassing data conversion, quality control, atlas building, fiber tractography, and fiber parameterization (see Supplementary Information for details). Diffusion tensor metrics were extracted at evenly spaced points (arc lengths) along major fiber tracts. Fiber profiles of FA diffusivity values were computed and averaged across each fiber tract. Fiber profiles of AD and RD were also produced. Fiber profiles were cropped to mask out low signal-to-noise areas at both ends.
Overall, n = 7 datasets were excluded from further analysis from Cohort 1 and n = 41 datasets from Cohort 2. The global mean over all tracts of each DTI measurement was computed for the association analyses.
Covariates
Race and ethnicity are considered social constructs of racial identity that mirror differences in social experiences, such as inequities in health outcomes and access to health care. To account for these unmeasured inequities, the analyses incorporated maternal race and ethnicity as a proxy covariate. Maternal race and ethnicity, annual household income, and use of anxiety/depression medication during pregnancy were assessed by maternal self-report. Use of anxiety/depression medication was assessed at the second time point (30–35 weeks’ gestation) in Cohort 1 and at each study visit in Cohort 2. A dichotomous variable was created, indicating the use of anxiety/depression medication at any time during pregnancy versus no use of anxiety/depression medication. See Supplementary Materials for details on the harmonization procedure regarding household income. Gestational age at birth and sex assigned at birth were taken from medical records. To account for systematic differences in MRI acquisition, as well as unmeasured and smaller, nonsignificant differences in covariates between the cohorts, the site of data collection was included as a covariate in all analyses.
Statistical analyses
Since the DTI metrics are strongly associated with infant age, measures were residualized for gestational age at birth and postnatal age at scan, and residualized measures were used in the analyses. While no extreme outliers were present in the data, for each of the residualized DTI metrics, some values fell 1.5 × inter-quartile range (IQR) below quartile 1 and/or 1.5 × IQR above quartile 3 of the distribution (FA: n = 2, RD: n = 5, AD: n = 1). Sensitivity analyses were run for all models excluding these cases.
We used a three-step model-building approach. For each residualized global white matter DTI metric (FA, RD, and AD), three linear regression models were fit. Model 1 included the predictors of interest, maternal depressive symptoms, and CM, as well as site, maternal race and ethnicity, and infant sex as covariates to estimate independent effects of CM and depressive symptoms. Model 2 additionally included the interaction term between maternal depressive symptoms and CM. Model 3 additionally adjusted for household income and use of anxiety/depression medication during pregnancy. Model 3 was run separately to avoid overfitting and because both additional covariates have been identified in previous studies as modifiers of the depression association (Jha et al., Reference Jha, Meltzer-Brody, Steiner, Cornea, Woolson, Ahn and Knickmeyer2016; Lean et al., Reference Lean, Smyser, Brady, Triplett, Kaplan, Kenley and Rogers2022) and may also be part of the mechanistic pathway in the association between maternal depression and/or CM and offspring brain outcomes. Corrections for multiple testing across DTI metrics were performed by controlling the false discovery rate using the p.adjust function from the ‘stats’ package. All analyses were conducted in R (R Core Team, Reference Team2022).
Results
Descriptives and preliminary analyses
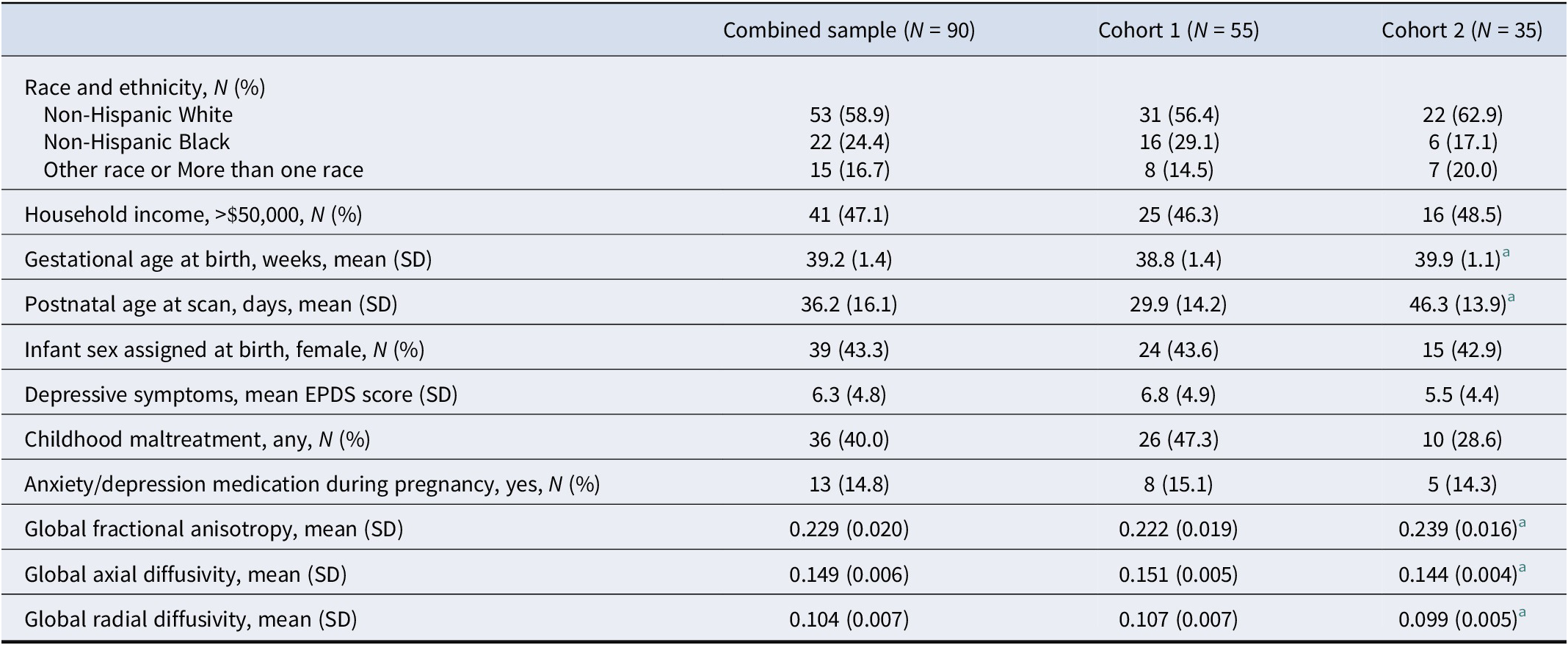
Table 1 presents an overview of descriptive information regarding both cohorts. Race and ethnicity, and household income of the study sample were comparable between the two cohorts. Approximately 59% of participants identified as Non-Hispanic White (56% in Cohort 1 and 63% in Cohort 2); 24% identified as Non-Hispanic Black (29% in Cohort 1 and 17% in Cohort 2); another race or more than one race was reported by 14.5% and 20% of the sample, respectively. Forty-seven percent reported an annual household income >$50,000 (46% in Cohort 1 and 48.5% in Cohort 2). Exposure to any type of CM was reported by 47% of the sample in Cohort 1 and 29% in Cohort 2. This difference was statistically not significant (χ 2 = 2.39, p = .122). Approximately 43% of newborns were assigned female sex at birth in both cohorts. The two cohorts differed significantly with respect to gestational age at birth and postnatal age at scan. Newborns in Cohort 2 were significantly older at birth and at the time of the MRI scan. Since the DTI metrics are strongly associated with age, there was a significant difference between the cohorts in these measures (see Table 1); however, after residualizing the DTI metrics for gestational age at birth and postnatal age at scan, no statistical differences remained (see Supplementary Material for details).
Cohort characteristics

Note: Means and SDs for diffusivity measures are multiplied by a factor of 100 to improve readability. EPDS, Edinburgh Postnatal Depression Scale.
a Significantly different (p < .05) from Cohort 1 in independent sample t-tests or chi-square tests.
Bivariate correlations between the main predictors and covariates can be found in Supplementary Table S1 available online.
Association between maternal depression, maternal CM, and newborn global white matter microstructure
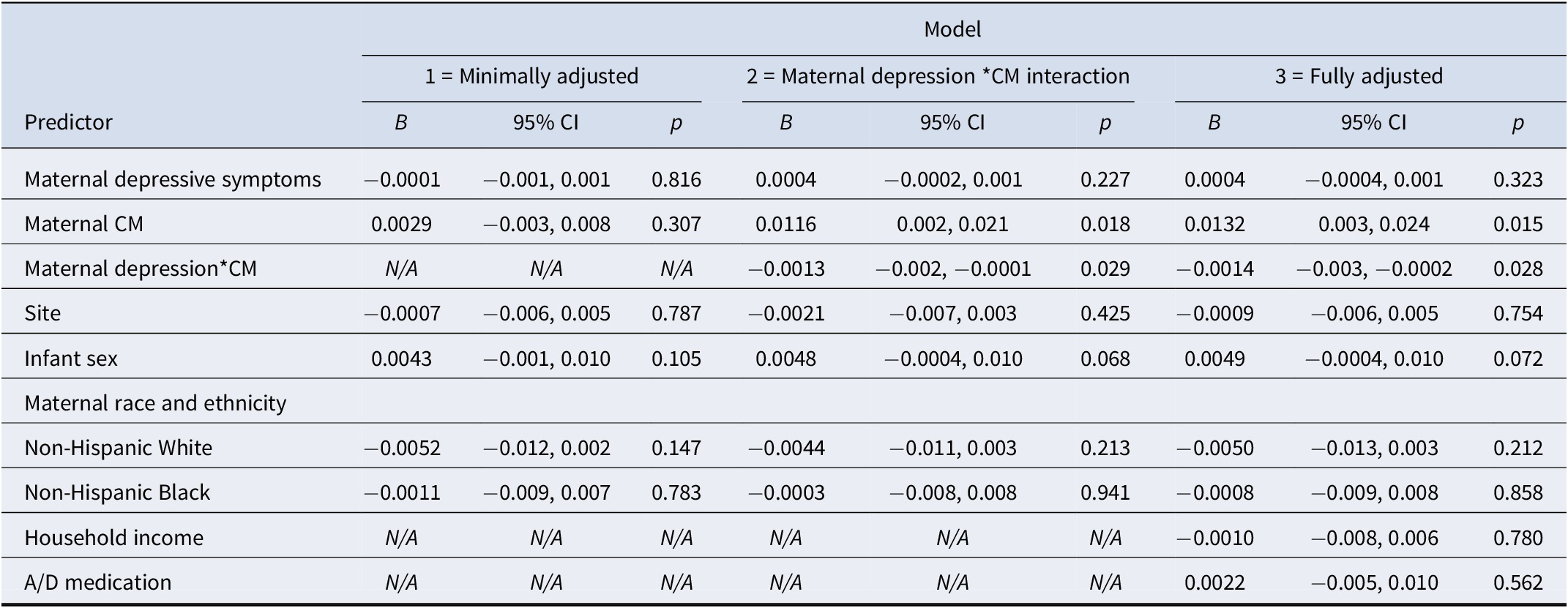
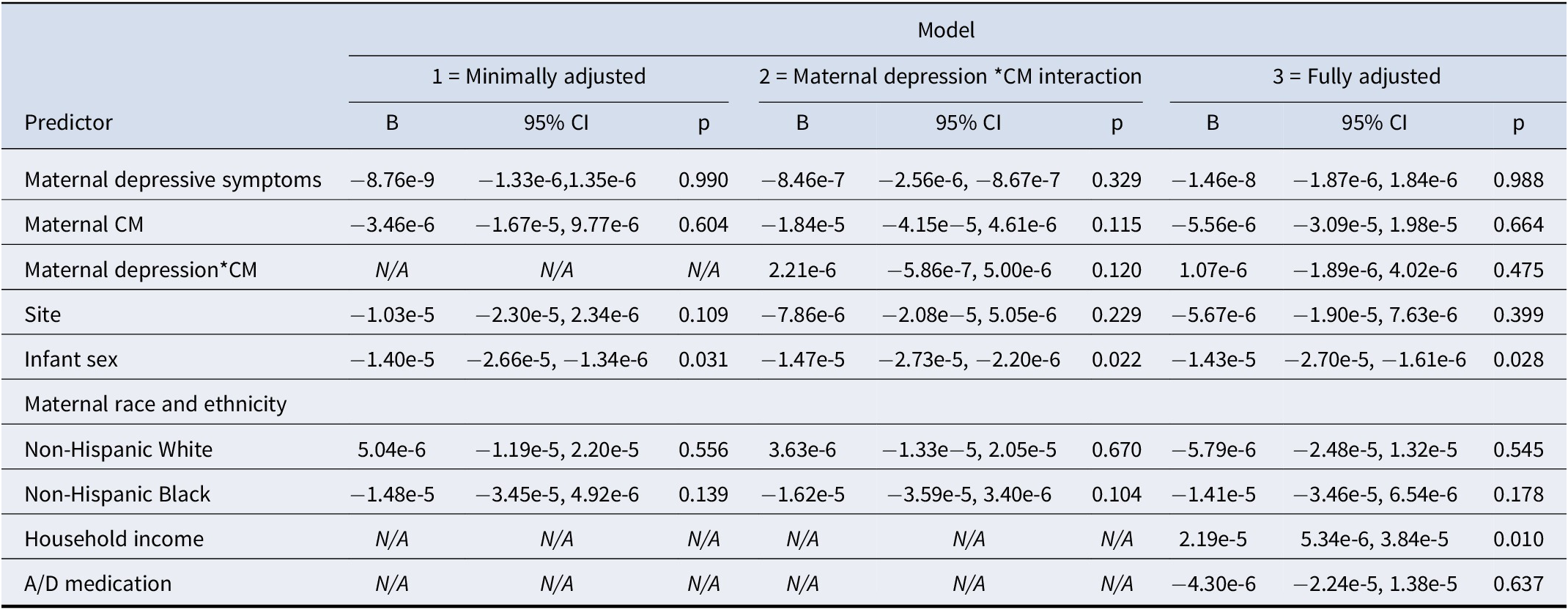
Full model results are displayed in Tables 2–4. We observed no independent associations between maternal depressive symptoms or maternal CM and newborn white matter microstructure (model 1). However, model 2 indicated that maternal history of CM moderated the association between maternal depressive symptoms and newborn global mean FA (B = -0.001, standard error [SE] = 0.0006, β = −0.523, p = .029, p adj = 0.044, f 2 = 0.06) and global mean RD (B = 5.94e-6, SE = 2.36e-6, β = 0.590, p = .014, p adj = 0.042, f 2 = 0.08). Specifically, the association between maternal depressive symptoms and newborn FA was positive in the CM- group and negative in the CM+ group (see Figure 1a). The direction of this interaction was reversed for newborn RD (see Figure 1b), which is consistent with the relationship of these metrics with each other. The interaction between maternal depressive symptoms and CM on global mean AD followed the same pattern as the interaction effect on RD, but was not significant at p < .05.
Regression models testing the association between maternal depressive symptoms, maternal childhood maltreatment, and offspring global fractional anisotropy

Regression models testing the association between maternal depressive symptoms, maternal childhood maltreatment, and offspring global radial diffusivity

Regression models testing the association between maternal depressive symptoms, maternal childhood maltreatment, and offspring global axial diffusivity

Note: Complete model results, including multiple regression coefficients, 95% confidence intervals, and p-values for all predictors and covariates. CM, childhood maltreatment; A/D medication, anxiety/depression medication.
Interaction between maternal depressive symptoms during pregnancy and maternal history of childhood maltreatment on newborn global white matter microstructure.
Note: DTI metrics are residualized for gestational age at birth and postnatal age at scan. CM+ represents the group with maternal childhood maltreatment history (N = 36), CM- represents the group without maltreatment history (N = 54). CM significantly moderated the association between maternal depressive symptoms during pregnancy and newborn global FA (panel a) and RD (panel b) but not AD (panel c). CM, childhood maltreatment; EPDS, Edinburgh Postnatal Depression Scale.

In the fully adjusted model, which additionally controlled for household income and anxiety/depression medication (model 3 in Tables 2–4), the results remained consistent, although the strength of the interaction of maternal depressive symptoms and maternal CM on global RD was slightly attenuated (B = 5.46e-6, SE = 2.52e-6, β = 0.526, p = .033, p adj = 0.049).
Sensitivity analyses
Excluding outliers (defined using the IQR method) from the models generated only minor changes in results. Most importantly, the interaction effect between maternal depression and maternal CM on global FA and global RD remained statistically significant in both the minimally adjusted model 2 (FA: B = -0.001, SE = 0.0005, p = .015; RD: B = 4.49e-6, SE = 1.83e-6, p = .016) and the fully adjusted model 3 (FA: B = -0.001, SE = 0.0005, p = .015; RD: 4.00e-6, SE = 1.96e-6, p = .045). The interaction effect on global AD remained nonsignificant.
Discussion
This study investigated the moderating role of maternal CM experience on the association between depressive symptoms during pregnancy and white matter integrity in offspring shortly after birth. We observed no independent associations between maternal depressive symptoms during pregnancy and infant global white matter microstructure. Similarly, infants’ white matter microstructural properties were unrelated to whether their mother had been exposed to CM. However, in infants born to women with a history of CM, higher depressive symptoms were associated with lower FA and higher RD, a pattern that was reversed in infants of women without CM. Results were consistent even after adjusting for household income and use of anxiety/depression medication, two variables that may be part of the mechanistic pathway underlying this association.
DTI metrics are indirect measures of microstructure and cannot be linked directly to specific neurobiological alterations; however, AD has been related to axonal organization and fragmentation, whereas RD has been related to myelination and axonal density (Harsan et al., Reference Harsan, Poulet, Guignard, Steibel, Parizel, de Sousa and Ghandour2006; Song et al., Reference Song, Sun, Ramsbottom, Chang, Russell and Cross2002). From a developmental perspective, all measures of diffusivity decrease strongly across early life (particularly the first year of life) while FA shows a dramatic increase (Stephens et al., Reference Stephens, Langworthy, Short, Girault, Styner and Gilmore2020). Thus, among infants of women with CM exposure, higher depressive symptomatology during pregnancy was associated with a pattern indicating lower white matter microstructural maturity; in contrast, in infants of women without CM, prenatal depressive symptoms were related to a pattern indicative of greater white matter maturity. While the direction of the association was the same for both diffusivity measures (i.e. RD and AD), only the association between maternal depressive symptoms and maternal CM with RD was statistically significant, which may suggest a certain specificity in the nature of the microstructural variation.
Although the current study does not permit conclusions about the functional or clinical significance of the observed differences, it is important to note that both slower and accelerated development may be advantageous or disadvantageous depending on the developmental context. If the brain develops more quickly, windows of heightened plasticity may close earlier, reducing the brain’s sensitivity to environmental exposures, which may lead to less optimal adaptation due to decreased opportunity to sample the environment but may also limit the influence of harmful exposures (Tooley, Bassett, & Mackey, Reference Tooley, Bassett and Mackey2021). The ‘stress acceleration hypothesis’ posits that accelerated development may reflect an adaptive strategy for harsh and low nurturing developmental contexts (Callaghan & Tottenham, Reference Callaghan and Tottenham2016), whereas a safer or more positive environment but also a higher variability in environmental conditions (independent of the valence of these experiences) has been suggested to result in a protracted period of brain development (Tooley et al., Reference Tooley, Bassett and Mackey2021). Empirically, the direction of the observed changes in neurodevelopmental pacing does not always correspond to these theoretical models and likely depends on a range of additional factors, including the specific type of adversity, the specific brain outcome that is measured, and the age or developmental stage of the child. In the context of the present study, the pattern of higher maturity associated with more depressive symptomatology among the group of infants born to women without CM exposure may indeed be explained in terms of a developmental acceleration in response to signals reflecting a harsh or stressful environment. However, this is not the case for the pattern observed among CM-exposed women. Whether the lower white matter microstructural maturity in this group is the result of processes that cannot be easily explained by the currently available models or whether it reflects a developmental insult rather than an adaptive process warrants further investigation with longitudinal follow-up.
The mechanisms underlying these differential associations between maternal depression during pregnancy and offspring white matter development in mothers with and without CM are not well understood, but variation in maternal–placental–fetal stress biology is a likely candidate. Biological correlates of depression, such as the activity of the endocrine stress system and the immune system, may differ during pregnancy depending on CM status (Epstein et al., Reference Epstein, Houfek, Rice, Weiss, French, Kupzyk and Pullen2020; Walsh et al., Reference Walsh, Basu, Werner, Lee, Feng, Osborne and Monk2016). One potential pathway involves inflammatory mediators, including the cytokine interleukin (IL)-6 and tumor necrosis factor-alpha. In pregnant individuals with abuse history or higher maltreatment severity, depressive symptoms have been shown to be positively associated with these inflammation biomarkers across pregnancy but depressive symptoms were not associated with inflammation when maltreatment severity was low (Kleih et al., Reference Kleih, Entringer, Scholaske, Kathmann, DePunder, Heim and Buss2022; Walsh et al., Reference Walsh, Basu, Werner, Lee, Feng, Osborne and Monk2016). Importantly, these inflammatory mediators may directly or indirectly influence the developing fetal brain. For instance, maternal prenatal IL-6 concentrations have been associated with lower FA in the uncinate fasciculus of their newborn children (Rasmussen et al., Reference Rasmussen, Graham, Entringer, Gilmore, Styner, Fair and Buss2019). Studies in mice suggest that the effects of maternal prenatal immune activation on white matter microstructure may be mediated by long-term decreased expression of myelin basic protein and other genes involved in myelination (Makinodan et al., Reference Makinodan, Tatsumi, Manabe, Yamauchi, Makinodan, Matsuyoshi and Wanaka2008; Richetto et al., Reference Richetto, Chesters, Cattaneo, Labouesse, Gutierrez, Wood and Riva2017) or by decreasing oligodendrogenesis (Velloso et al., Reference Velloso, Wadhwa, Kumari, Carcea, Gunal and Levison2022) or both. In the absence of elevated inflammatory markers (as observed in pregnant women without exposure to CM), other biological factors may be involved in mediating the suggested effect of maternal depressive symptoms on offspring white matter development, with contrasting consequences for developmental pacing. For instance, glucocorticoids are known to accelerate maturation at the expense of proliferation in many organs, including the brain (Eachus & Ryu, Reference Eachus and Ryu2024), and in a mouse model, early life stress, which was accompanied by elevated glucocorticoid concentrations, resulted in an earlier increase in myelin basic protein in the hippocampus (Bath, Manzano-Nieves, & Goodwill, Reference Bath, Manzano-Nieves and Goodwill2016). The mechanistic pathway may also involve epigenetic modifications. Prenatal maternal depression has been associated with infant DNA methylation changes (albeit not consistently, Drzymalla et al., Reference Drzymalla, Crider, Wang, Marta, Khoury and Rasooly2023) and DNA methylation in cord blood has been related to the microstructure in several brain regions previously associated with maternal depression/anxiety (Dean et al., Reference Dean, Madrid, Planalp, Moody, Papale, Knobel and Kling2021).
A limitation of the current study is the relatively modest sample size, which limited the ability to test the contribution of specific white matter tracts in addition to the investigation of global white matter microstructure. Furthermore, the sample size may have impacted the robustness of our findings, and the complexity of the models increases the risk of overfitting. Consequently, the observations need to be replicated in independent as well as larger, more well-powered study populations (e.g. within the HEALthy Brain and Child Development study or the ENIGMA ORIGINs consortium; Alex et al., Reference Alex, Buss, Davis, Campos, Donald, Fair and Knickmeyer2023; Volkow et al., Reference Volkow, Gordon, Bianchi, Chiang, Clayton, Klein and Weiss2024) to determine whether these associations are driven by specific regions or whether they truly represent a global effect. At the same time, it is possible that our observed associations may not be specific to depression but rather reflect the effects of poor mental health or stress more broadly. In our nonclinical sample, we were not able to disentangle potential differential associations of specific disorders. Furthermore, longitudinal assessments of white matter microstructure and development are paramount to determine whether the observed patterns are stable, adaptive versus maladaptive, and underlie later-emerging behavioral health outcomes. Another limitation is the combination of the study sample from study populations at two distinct sites. While combining samples across cohorts may increase the ecological validity of our findings, the two cohorts differed in several important aspects, including the measure of CM and the age range of the neonates that underwent the MRI scan. Although we used the DTI metrics residualized for gestational age at birth and postnatal age at scan in all analyses, we cannot rule out that aspects of the pre- and postnatal environment may have affected neonatal white matter microstructure more strongly with more time spent in utero or ex utero. Lastly, we have tested only a limited number of covariates on the mechanistic pathway that have been implicated in previous studies (Jha et al., Reference Jha, Meltzer-Brody, Steiner, Cornea, Woolson, Ahn and Knickmeyer2016; Lean et al., Reference Lean, Smyser, Brady, Triplett, Kaplan, Kenley and Rogers2022); however, the influence of other, untested, prenatal exposures should be investigated in future studies, including substance use, nutrition, and social support.
The results of this study underscore the interplay between maternal depressive symptoms during pregnancy, CM exposure, and their combined impact on offspring brain development. They further suggest differential mechanisms underlying the association between maternal depressive symptoms during pregnancy and offspring neurodevelopment depending on maternal history of CM exposure. Assuming a singular biological risk profile in the context of maternal depression may therefore lead to flawed conclusions regarding the seemingly inconsistent findings in the previous literature. Our findings also have substantial implications for public health initiatives. The identification of specific risk factors, particularly CM, provides a basis for the design and implementation of more precise and effective interventions aimed at mitigating the potential adverse effects on offspring neurodevelopment. Furthermore, these findings highlight the importance of monitoring the psychosocial well-being of pregnant women. Routine screening for maternal depressive symptoms, coupled with an assessment of a woman’s history of CM, into prenatal care protocols may enable early identification and targeted support for at-risk individuals, thereby contributing to improved maternal and child outcomes.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0033291726103468.
Acknowledgments
This work was financially supported by the National Institutes of Health grants UG3/UH3OD023349 (PI: TGO’C), R01MH105538 (PI: PDW, CB, DAF), P50HD103573 (PI: MS), 4R00HD100593–03 (PI: JMR), R01HD083369 (PI: ESB), P30ES005022 (PI: ESB), and U01DA055363 (PI: AMG); The Wynne Center for Family Research (PI: TGO’C); and the University of Rochester CTSA award number UL1 TR002001 from the National Center for Advancing Translational Sciences of the National Institutes of Health and the German Research Foundation (DFG) award number 441735381 (PI: NKM). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or other funding bodies.
Competing interests
The authors declare none.
Ethical standard
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.