Introduction
With approximately 1350 species, the Xyleborini (Coleoptera: Scolytinae) are highly specialised and ecologically successful scolytine ambrosia beetles that bore into the sapwood of dead and dying trees and cultivate fungi (Osborn et al. Reference Osborn, Castro, Duong, Hulcr, Li, Martínez and Cognato2023). Most species carry symbiotic fungi in specialised pockets of the exoskeleton (mycangia). The fungi are evacuated from mycangia and grow in the tunnels constructed by the beetle and serve as food for the adult beetles and developing brood. In general, the beetles are not host specific, and choice of tree most likely depends on degree of decay and moisture content (Smith and Hulcr Reference Smith, Hulcr, Vega and Hofstetter2015). Upon commencement of the fungal garden, the pioneering female lays one (or a few) haploid egg that develops into a male and a majority of diploid eggs that develop into females (Kirkendall Reference Kirkendall, Wrensch and Ebbert1993). The dwarfed and flightless adult male mates with his sisters (Jordal et al. Reference Jordal, Beaver and Kirkendall2001). The haplo–diploid mating system renders reproduction to almost clonal, with occasional mating between unrelated lineages (Kirkendall et al. Reference Kirkendall, Biedermann, Jordal, Vega and Hofstetter2015). Females leave the natal tunnels mated and packed with food, making them well-adapted to colonising new host material in either native or exotic environments. Indeed, there is evidence for prehistoric dispersal of these beetles throughout the world (Cognato et al. Reference Cognato, Hulcr, Dole and Jordal2011, Reference Cognato, Jordal and Rubinoff2018; Gohli et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016), and contemporary human-aided dispersal of xyleborines has resulted in the introduction and establishment of 35 exotic species to the continental United States of America (e.g., Haack and Rabaglia Reference Haack, Rabaglia and Peña2013; Rabaglia et al. Reference Rabaglia, Cognato, Hoebeke, Johnson, Labonte, Carter and Vlach2019; Johnson and Smith Reference Johnson and Smith2023; Osborn et al. Reference Osborn, Castro, Duong, Hulcr, Li, Martínez and Cognato2023; Johnson et al. Reference Johnson, Atkinson, Schnepp, Smith and Hulcr2025). These introductions include species that have impacted southeastern United States of America forest ecology and have threatened California’s and Florida’s avocado industries (Fraedrich et al. Reference Fraedrich, Harrington, Rabaglia, Ulyshen, Mayfield and Hanula2008; Eskalen et al. Reference Eskalen, Gonzalez, Wang, Twizeyimana, Mayorquin and Lynch2012). The exotic species in North America have prompted much research on their ecology, biology, symbiotic fungi, phylogenetics, and population genetics (e.g., Cognato et al. Reference Cognato, Hulcr, Dole and Jordal2011, Reference Cognato, Hoebeke, Kajimura and Smith2015, Reference Cognato, Smith, You, Pham and Hulcr2019; Cooperband et al. Reference Cooperband, Cossé, Jones, Carrillo, Cleary, Canlas and Stouthamer2017; Stouthamer et al. Reference Stouthamer, Rugman-Jones, Thu, Eskalen, Thibault and Hulcr2017; Smith and Cognato Reference Smith and Cognato2022), but little is known of the 18 presumed native species. Of these, seven are adventive or at the northern limits of wider southern ranges, and seven Xyleborus Eichhoff, two Ambrosiodmus Hopkins, and two Anisandrus Ferrari species reside exclusively in Canada and the United States of America.
Xyleborini originated 23–25 million years ago, likely in tropical Asia or Oceania (Jordal and Cognato Reference Jordal and Cognato2012). Dispersal throughout the pantropics began approximately 15 million years ago, with the beetles colonising and radiating throughout South America, Africa, and remote islands, including the Hawaiian archipelago (Gohli et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016; Cognato et al. Reference Cognato, Jordal and Rubinoff2018). Given the diversity of unrelated genera in these areas, multiple lineages likely colonised the continents at different times, whereas only one dispersal event has been documented for Hawaii (Golhi et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016; Cognato et al. Reference Cognato, Jordal and Rubinoff2018). In the Nearctic, at least three dispersal events of Xyleborini took place, based on the occurrence of native species of three genera, Ambrosiodmus, Anisandrus, and Xyleborus. Most interesting are the seven Xyleborus species native to the Nearctic, whose general morphological similarity to each other suggests a single origin and subsequent diversification (Fig. 1). However, considerable morphological difference occurs in the elytral declivity and pronotal shape to suggest that the native species may not share a common ancestor (Fig. 1). In the present study, we reconstructed a DNA-based phylogeny of the native Nearctic Xyleborus species and of a sample of worldwide Xyleborini to test the monophyly of the native Nearctic species.
Xyleborus species native to the Nearctic: A, X. intrusus, B, X. pubescens, C, X. planicollis, D, X. viduus, E, X. impressus, F, X. xylographus, and G, X. celsus; Xyleborus species with other distributions: H, X. affinis, I, X. perforans, J, X. volvulus, K, X. ferrugineus, and L , X. monographus.

Materials and methods
Taxa and DNA sequencing
Previously collected adult specimens were stored in 100% ethanol below –20 °C before molecular analysis (Table 1). Because it was not possible to exhaustively sample the 270 Xyleborus species, we sampled species representative of major geographic areas and species with worldwide distributions from a selection of ethanol-preserved specimens in the A.J. Cook Arthropod Research Collection, Michigan State University (East Lansing, Michigan, United States of America). One arguably Nearctic species, X. horridus Eichhoff, was not available for DNA sequencing. Specimens were identified using published keys (Wood Reference Wood1982, Reference Wood2007; Gomez et al. Reference Gomez, Rabaglia, Fairbanks and Hulcr2018; Rabaglia et al. Reference Rabaglia, Smith, Rugman-Jones, Digirolomo, Ewing and Eskalen2020). Samples of DNA were extracted from 41 specimens representing 26 Xyleborus species using a Qiagen DNEasy Blood and Tissue Kit (Qiagen, Hilden, Germany) following the manufacturer’s protocols (Table 1). Several species were represented by two or more individuals due to the potential of pseudocryptic species known to exist in Xyleborini (e.g., Smith and Cognato Reference Smith and Cognato2022). The remaining body parts were pinned and vouchered at the A.J. Cook Arthropod Research Collection. Using the purified DNA, partial gene regions of mitochondrial cytochrome c oxidase 1 (CO1), nuclear ribosomal 28S (D2 and D3 regions), and nuclear protein–coding carbamoylphosphate synthetase 2–aspartate transcarbamylase–dihydroorotase (CAD) were amplified and prepared for sequencing using published primers and protocols (Cognato Reference Cognato2013). Polymerase chain reaction amplicons were sequenced with the BigDye Terminator, version 1.1, cycle-sequencing kit (Applied Biosystems, Foster City, California, United States of America) and analysed on an ABI 3730 or 3700 platform (Applied Biosystems, Thermo Fisher Scientific, Carlsbad, California) at the Research Technology Support Facility, Michigan State University. The resulting DNA sequences were assembled with Sequencher (GeneCodes, Ann Arbor, Michigan) and submitted to GenBank (Table 1). No length differences were detected in the protein-coding regions; however, length variation was present in the 28S rDNA sequences, which were aligned by using the MUSCLE using default settings (Edgar Reference Edgar2004). An additional 123 sequences for Xyleborus and related taxa were retrieved from GenBank (Table 1; Cognato et al. Reference Cognato, Hulcr, Dole and Jordal2011; LeMay et al. Reference LeMay, Johnson and Hulcr2024; Cognato and Smith Reference Cognato and Smith2025).
Xyleborini species included in this study and associated DNA vouchers, localities, and GenBank numbers. N/A, not available; CO1, cytochrome c oxidase 1; CAD, carbamoylphosphate synthetase 2–aspartate transcarbamylase–dihydroorotase; USA, United States of America; AZ, Arizona; FL, Florida; GA, Georgia; KS, Kansas; MD, Maryland; MI, Michigan; MO, Missouri; NY, New York; OH, Ohio; PA, Pennsylvania; SC, South Carolina; TX, Texas

Phylogenetic analysis and ancestral state reconstruction
Phylogenetic relationships were inferred using combined datasets with Bayesian and parsimony analyses. Bayesian inference was performed with MrBayes, version 3.2.2 (Ronquist et al. Reference Ronquist, Teslenko, van der Mark, Ayres, Darling and Höhna2012), employing two independent runs of four Metropolis-coupled Markov chain–Monte Carlo analyses (one cold and three heated chains). Data were partitioned by CO1 and CAD codon positions, as well as by 28S sequences, with each partition modelled under a general time-reversible substitution scheme incorporating invariant sites and gamma-distributed rate variation (GTR + I + Γ). These model parameters were chosen based on previous examination of model fit for Xyleborini (Cognato et al. Reference Cognato, Hulcr, Dole and Jordal2011). Each analysis was executed for 10 million generations, sampling every 100th generation. Convergence was achieved within this timeframe, with runs showing minimal divergence (average standard deviation of split frequencies = 0.0045). Posterior probabilities were derived from a majority-rule consensus of post–burn–in trees, totalling 150 002 trees. The estimated sample sizes of parameters were larger than 500. Parsimony optimality consisted of a heuristic search, with 500 stepwise random addition replicates holding only 500 suboptimal trees per replicate and the default settings (PAUP* 4.0 b10 PPC; Swofford Reference Swofford2002). Bootstrap values were calculated with 500 pseudoreplicates. Intraspecific and interspecific pairwise uncorrected “p” distances were calculated in PAUP*.
Based on Wood and Bright (Reference Wood and Bright1992), biogeographic regions, as defined by Darlington (Reference Darlington1957), were scored for the terminal taxa in Mesquite, version 3.81 (Maddison and Maddison Reference Maddison and Maddison2023). For species with worldwide distributions (specifically, X. affinis, X. bispinatus, X. ferrugineus, X. perforans, and X. volvulus), we scored their terminals with hypothesised ancestral distributions to reflect possible regions of origin (Gohli et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016). The geographic region character states included 0 – Eastern Palearctic and Oriental, 1 – Neotropical, 2 – Nearctic, 3 – Afrotropic, and 4 – Western Palearctic, and were traced using the likelihood criterion onto the phylogeny (Figs. 2 and 3). Ancestral geographic states were estimated using the Markov k-1 (mk-1) model, which allows for equal rates of change between character states, and marginal probability reconstruction was estimated with rate of 0.89381612 and –logL: 70.20962133 (Opt.: width 0.0), using the default Mesquite settings. Likelihoods were reported as proportional likelihoods.
Phylogeny of Xyleborus and other xyleborine species based on Bayesian analysis of DNA data. Solid black circles represent posterior probabilities greater than 90%. Pie diagrams represent proportional likelihood of ancestral geographic ranges. Numbers at nodes are discussed in the text and Table 2. The numbered clades may not indicate the same clades in Figure 3. Asterisks indicate Xyleborus species with worldwide distributions.

The strict consensus of the 36 most parsimonious trees of Xyleborus and other xyleborine species based on a parsimony analysis of DNA data. Solid black circles represent bootstrap values greater than 90%. Pie diagrams represent proportional likelihood of ancestral geographic ranges. Numbers at nodes are discussed in the text. The numbered clades may not indicate the same clades in Figure 2. Asterisks indicate Xyleborus species with worldwide distributions.

Given the dearth of fossil records for xyleborines (e.g., Osborn et al. Reference Osborn, Smith and Cognato2022), we did not calculate a time-calibrated phylogeny. We discuss the age of clades based on the time-calibrated phylogeny of Xyleborus species with worldwide distributions (Gohli et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016).
Results
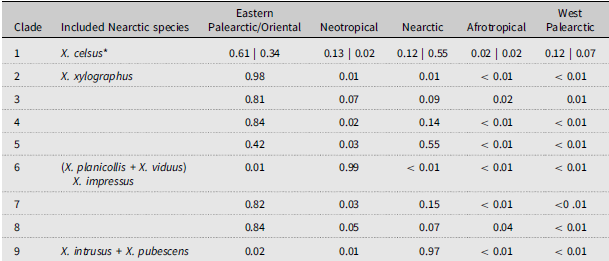
The Bayesian analysis resulted in a resolved phylogeny except for the species relationships in clade 2 (Fig. 2). Many clades had greater than 0.9 posterior probabilities. In general, the species relationships were similar to those of the more extensive phylogeny of Xyleborini reported by Cognato et al. (Reference Cognato, Hulcr, Dole and Jordal2011). Notably, Xyleborus was paraphyletic in relation to the endemic Neotropical genera, Coptoborus Hopkins and Sampsonius Eggers. The Nearctic Xyleborus species were not collectively monophyletic, and they occurred in four separate clades: 1 – X. celsus Eichhoff, 2 – X. xylographus (Say), 6 – X. planicollis Zimmermann + X. viduus Eichhoff + X. impressus Eichhoff, and 9 – X. intrusus Blandford + X. pubescens Zimmermann. Clades 2 and 5 include currently worldwide-distributed species, and clade 9 was found to be sister to clade 4, which includes species from most geographic regions (Fig. 2). Xyleborus celsus was found to be sister to Eastern Palearctic–Oriental and Western Palearctic species (Fig. 2). The proportional likelihood of the clades’ ancestral geographic ranges suggests that most Nearctic clades derived from Eastern Palearctic–Oriental or Nearctic ancestors, although Neotropical, Nearctic, or Western Palearctic ancestral ranges for X. celsus are possible (Table 2). Independent diversification of endemic species occurred twice in the Nearctic with X. planicollis + X. viduus + X. impressus and with X. intrusus + X. pubescens.
Proportional likelihood of ancestral geographic ranges of Nearctic Xyleborus species. Clade numbers refer to the Bayesian phylogeny, Figure 2.

* Left number refers to the sister relationship of X. celsus and X. monographus + X. festivus; right number refers to the sister relationship of X. celsus and X. festivus observed in the parsimony phylogeny, clade 12, Figure 3.
Parsimony analysis resulted in 36 most parsimonious trees, which were mostly resolved in the strict consensus of those trees (Fig. 3). Only the relationships of X. intrusus individuals and some of the X. pubescens individuals were unresolved. This phylogeny differs from that of the Bayesian tree in the sister relationship between X. celsus and X. festivus Eichhoff and the sister relationship between clade 11 (X. celsus + X. festivus + X. monographus Eichhoff) and clade 9 (X. intrusus + X. pubescens; Fig. 3). Bootstrap values were less than 90% for most internal nodes (Fig. 3). The Nearctic Xyleborus species were not collectively monophyletic, and they occurred in four separate clades, as they did in the Bayesian analysis. However, reconstruction of the ancestral ranges suggested an Eastern Palearctic–Oriental origin for X. xylographus, an Eastern Palearctic–Oriental or Nearctic origin for X. celsus, and Nearctic origins for X. planicollis + X. viduus + X. impressus and for X. intrusus + X. pubescens (Fig. 3). Furthermore, intraspecific CO1 DNA differences less than 10% and monophyly of species represented by more than one individual did not suggest the possibility of cryptic species (see Cognato et al. Reference Cognato, Sari, Smith, Beaver, Li and Hulcr2020 for discussion concerning Xyleborine species concept).
Discussion
The present study is the first to place the Nearctic Xyleborus in phylogenetic context within a selection of Xyleborus and other Xyleborini species from worldwide localities. We demonstrate that the seven Nearctic species are not collectively monophyletic and that they have four independent evolutionary origins. Based on the Bayesian analysis, three of these clades most likely derived from species with Eastern Palearctic–Oriental or Nearctic distributions, whereas the origin of X. celsus is uncertain and suggests a dispersal event from the Palearctic, in general, to the Nearctic (0.72 proportional likelihood; Table 2; Fig. 2). Alternatively, the parsimony analysis suggests a possible Nearctic origin for the X. monographus, X. celsus + X. festivus clade (0.5 proportional likelihood; Fig. 3; clade 11). This is the first suggestion of dispersal between these biogeographical regions for xyleborines, which is an uncommon direction of dispersal among scolytines, given the prevalence of Palearctic-to-Nearctic dispersal (e.g., Godefroid et al. Reference Godefroid, Meseguer, Saune, Genson, Streito and Rossi2019; Cognato et al. Reference Cognato, Smith and Jordal2021). We prefer the Palearctic-to-Nearctic dispersal hypothesis for X. celsus, given the higher-than-0.9 posterior probabilities for most clades in the Bayesian tree (Fig. 2) as compared to the poor bootstrap values of the parsimony tree for internal clades (10–12) that connect X. celsus and X. intrusus + X. pubescens (Fig. 3).
The Nearctic species were phylogenetically placed with morphologically similar species based on the shape of the elytral declivity and the size and placement of declivital granules (Gomez et al. Reference Gomez, Rabaglia, Fairbanks and Hulcr2018). Xyleborus celsus, X. festivus, and X. monographus (clade 1; Fig. 2) have granules on interstriae 1 and 3 (none on interstriae 2) on the declivital face (Fig. 1). Their sister relationship to a specific Xyleborus species is ambiguous, given their phylogenetic placement varied between the trees (Figs. 2 and 3). Xyleborus impressus, X. planicollis, and X. viduus (clade 6; Figs. 2 and 3) lack a granule at the bases of interstriae 1 (Fig. 1). This clade and its sister relationship to X. ferrugineus (clade 5; Figs. 2 and 3) were previously unknown, and, except for X. planicollis, these species share a larger granule on interstriae 3 (Fig. 1). Xyleborus xylographus, X. intrusus, and X. pubescens have steep declivital slopes with rounded lateral margins, but their affinity to non-Nearctic endemic species remains ambiguous (Figs. 1, 2 and 3).
Given the xyleborine traits of extreme inbreeding and fungus farming, Jordal and Cognato (Reference Jordal and Cognato2012), Gohli et al. (Reference Gohli, Selvarajah, Kirkendall and Jordal2016), and Cognato et al. (Reference Cognato, Jordal and Rubinoff2018) have hypothesised that prehistorical dispersal (as opposed to vicariance) and colonisation of the world may explain much of xyleborine diversity. This diversification began approximately 20 million years ago, in a time of global warming, through long-distance dispersal, and it continued into the present, although recent colonisation events have been human-aided (Jordal and Cognato Reference Jordal and Cognato2012; Gohli et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016; Rabaglia et al. Reference Rabaglia, Cognato, Hoebeke, Johnson, Labonte, Carter and Vlach2019). The Nearctic endemic species potentially arrived in the New World within the past 10 million years, given that is the estimated beginning of the dispersal of globally distributed species that share ancestry with most Nearctic endemics (Fig. 2; X. xylographus, clades 6 and 9; Gohli et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016). The colonisation events of about 10 million years ago on the Hawaiian Islands and in Madagascar resulted in a radiation of 21 and 32 xyleborine species, respectively (Cognato et al. Reference Cognato, Jordal and Rubinoff2018; Eliassen and Jordal Reference Eliasson and Jordal2021). Dispersal between South America and Africa also contributed to the tribe’s diversification and resulted in hundreds of species, some of which occurred within 2 million years (Osborn et al. Reference Osborn, Smith and Cognato2022). These observations suggest that dispersal of xyleborines can result in species radiations within a few million years.
However, Xyleborus colonisation of the Nearctic did not result in comparable endemic diversity, as is observed in the previous mentioned scenarios. Only two instances of colonisation led to diversification: clade 9, with two species (X. intrusus + X. pubescens), and clade 6, with three species (X. planicollis + X. viduus + X. impressus; Fig. 2). In the case of X. intrusus + X. pubescens, Pinus host specificity and geographic isolation may have influenced speciation, given the more Central American–western United States of America distribution of X. intrusus, as compared to the eastern United States of America distribution of X. pubescens (Cognato and Smith Reference Cognato and Smith2025). Based on the phylogenetic relationship of Guatemalan, Arizonan, and X. intrusus specimens from the eastern United States of America, X. intrusus likely originated in Central America and dispersed northwards through Mexico and the southwestern United States of America, through the western United States of America, across southern Canada and into the eastern United States of America (Figs. 2 and 3; Wood and Bright Reference Wood and Bright1992; Gomez et al. Reference Gomez, Rabaglia, Fairbanks and Hulcr2018). Factors contributing to speciation for X. planicollis + X. viduus + X. impressus are less certain, given the distributions of species overlap in the eastern United States of America and that these species share many angiosperm hosts.
Details concerning the low diversity of Nearctic Xyleborus species are uncertain. There would have been ample time for natural selection or genetic drift (or both) to initiate speciation, given that Xyleborini speciation is estimated to have occurred within 2–10 million years (Gohli et al. Reference Gohli, Selvarajah, Kirkendall and Jordal2016; Eliasson and Jordal Reference Eliasson and Jordal2021; Osborn et al. Reference Osborn, Smith and Cognato2022). Potentially suboptimal pre-Holocene climates may have impeded Xyleborus diversification in the Nearctic and in the western Palearctic, which harbours only four endemic Xyleborus species (Knížek Reference Knížek, Löbl and Smetana2011). Xyleborines, in general, are more diverse in tropical and subtropical regions (Beaver Reference Beaver1979). The mild or nonexistent winters and humid summers found there promote survival of the beetles’ nutritional fungi and thereby the survival of the beetles (Atkinson et al. Reference Atkinson, Rabaglia and Bright1990). These optimal fungus-growing climates may have been limited during the climatically variable Pliocene and the colder and drier Pleistocene (Webb and Bartlein Reference Webb III and Bartlein1992; Salzmann et al. Reference Salzmann, Haywood, Lunt, Valdes and Hill2008). Only approximately 1.5% of all Xyleborus species are endemic to the Holarctic region (Osborn et al. Reference Osborn, Castro, Duong, Hulcr, Li, Martínez and Cognato2023). Currently, 6–7 endemic Xyleborus species and the highest diversity of xyleborines introduced to the Nearctic occur in the warm humid climate of the southeastern United States of America (Atkinson et al. Reference Atkinson, Rabaglia and Bright1990; Gomez et al. Reference Gomez, Rabaglia, Fairbanks and Hulcr2018; Rabaglia et al. Reference Rabaglia, Cognato, Hoebeke, Johnson, Labonte, Carter and Vlach2019).
Although we demonstrate collective polyphyly for the endemic Nearctic species and resolve relationships among some of these species, a few relationships remain ambiguous. Specifically, the sister-species to X. xylographus, X. intrusus + X. pubescens, and the X. celsus clade are unknown. A more complete sample of Xyleborus species and analysis of additional gene regions would produce a phylogeny with greater resolution and would allow further exploration of the endemic Nearctic species and their relationships within Xyleborus diversity.
Acknowledgements
This study was funded in part by a Cooperative Agreement from United States Department of Agriculture, Forest Service (16-CA-11420004-072) with A.I.C. Suggestions from anonymous reviewers helped to improve the manuscript.