Introduction
Maternal undernutrition during pregnancy and lactation adversely affect the long-term health of the child, inducing obesity, type 2 diabetes, and liver disease.Reference Lumey and Stein1,Reference Connor, Kibschull and Matysiak-Zablocki2 [Lumey, 1997, Offspring birth weights after maternal intrauterine undernutrition: a comparison within sibships; Connor, 2020, Maternal malnutrition impacts placental morphology and transporter expression: an origin for poor offspring growth; Mukai, 2014, Maternal fructose intake during pregnancy modulates hepatic and hypothalamic AMP-activated protein kinase (AMPK) signaling in a sex-specific manner in offspring]. The fetal and postnatal nutritional supply may cause systemic inflammation, which may lead to functional and morphological changes in various tissues.Reference Fragoso, Carvalho Jurema Santos and da Silva3–Reference Mohammed, Qadri, Molangiri, Basak and Rajkumar5 Additionally, low protein diets during fetal-neonatal nutrition impaired pancreatic beta-cell function and reduced plasma insulin levels, which may lead to impaired glucose tolerance and insulin resistance in the liver and skeletal muscles of adult life.Reference Wilson and Hughes6–Reference Heywood, Mian, Milla and Lindley8
Nutritional imbalance during gestation and lactation can induce the inflammation. The increased inflammatory markers including interleukin (IL)-6 and tumor necrosis factor (TNF)-α in small-for-gestational-age (SGA) infants were reported in the cord blood when compared with appropriate-for-gestational-age (AGA) controls.Reference Amarilyo, Oren and Mimouni9 In weaning rat offspring, maternal undernutrition is associated with impairment of the hypothalamus.Reference Plagemann, Harder and Rake10 Thus, these previous studies prompt the present study to investigate the protective strategies against the hepatic and hypothalamic inflammation in offspring programmed by maternal undernutrition.
The hypothalamus regulates energy balance and food intake via neurons that respond to hormonal and nutritional variation. In the hypothalamic inflammation, TNF-α signaling impaired insulin secretion and signal transduction in the liver and skeletal muscle.Reference Arruda, Milanski and Coope11 On the other hand, the inhibition of obesity-related hypothalamic inflammation has improved insulin resistance in the liver and reduced hepatic glucose production.Reference Milanski, Arruda and Coope12
AMP-activated protein kinase (AMPK) is known as key regulator of cellular energy homeostasis, glucose uptake, and fatty acid oxidation in various tissues including the liver and skeletal muscles.Reference Herzig and Shaw13 AMPK in the hypothalamus is involved in feeding behavior and energy expenditure such as brown adipose tissue thermogenesis.Reference López14 Therefore, changes in hypothalamic AMPK activation may contribute to the development of metabolic syndrome programmed by maternal undernutrition in adulthood. The mechanistic target of rapamycin (mTOR) is interlinked with AMPK pathway, and the both are involved in energy variation and sensing nutrients.Reference Yi, Yu and Lu15 mTOR, specifically mTOR complex 1 (mTORC1), in the hypothalamus contributes to the regulation of energy expenditure and body weight.Reference Hu, Xu and Liu16 Maternal low protein diets during pregnancy and lactation decreased mTOR phosphorylation in the hypothalamus of rat offspring.Reference Guzmán-Quevedo, Da Silva Aragão and Pérez García17
DNA methyltransferases (DNMT1, DNMT3A, and DNMT3B) catalyze DNA cytosine methylation and contribute to epigenetic control of gene expression involved in fat storage and energy metabolism. For example, in the liver of humans and rodent models, altered DNA methylation patterns have been associated with dysregulation of metabolic and lipid-handling genes and with fatty liver disease progression.Reference Ahrens, Ammerpohl and von Schönfels18,Reference Sookoian, Gianotti, Burgueño and Pirola19 In the hypothalamus of mice, high-fat diet exposure has been shown to modify DNA methylation of genes involved in feeding behavior and energy regulation, indicating epigenetic sensitivity of central metabolic control pathways.Reference Vucetic, Kimmel, Totoki, Hollenbeck and Reyes20 Also DNMT1-dependent methylation has also been shown to regulate inflammatory gene programs, linking DNMT activity with metabolic inflammation in immune and metabolic tissues.Reference Wang, Cao, Yu, Shi, Xue and Shi21
Glucoraphanin (Gr), a glucosinolate enriched in the broccoli, is converted by myrosinase into its active isothiocyanate sulforaphane (SFN) which has beneficial effects on various inflammatory diseases.Reference Yagishita, Fahey, Dinkova-Kostova and Kensler22 The broccoli sprouts consumption decreased the levels of IL-6 and C-reactive protein in overweight adult subjects.Reference López-Chillón, Carazo-Díaz and Prieto-Merino23 In addition, Gr activates the AMPK pathway and suppresses the production of the inflammatory cytokines; IL-1β and TNF-α, in an acute colitis mouse model.Reference Tian, Xu and Sun24 However, little is known about the effect of maternal broccoli intake on inflammation and AMPK activation in offspring programmed by maternal protein restriction. Hence, we aimed to investigate whether maternal broccoli powder (BP) intake during lactation ameliorates inflammation and affects the AMPK signaling in the liver and hypothalamus of female weaning rat offspring exposed to maternal low protein diet.
Materials and methods
Experimental design, animal and experimental diet
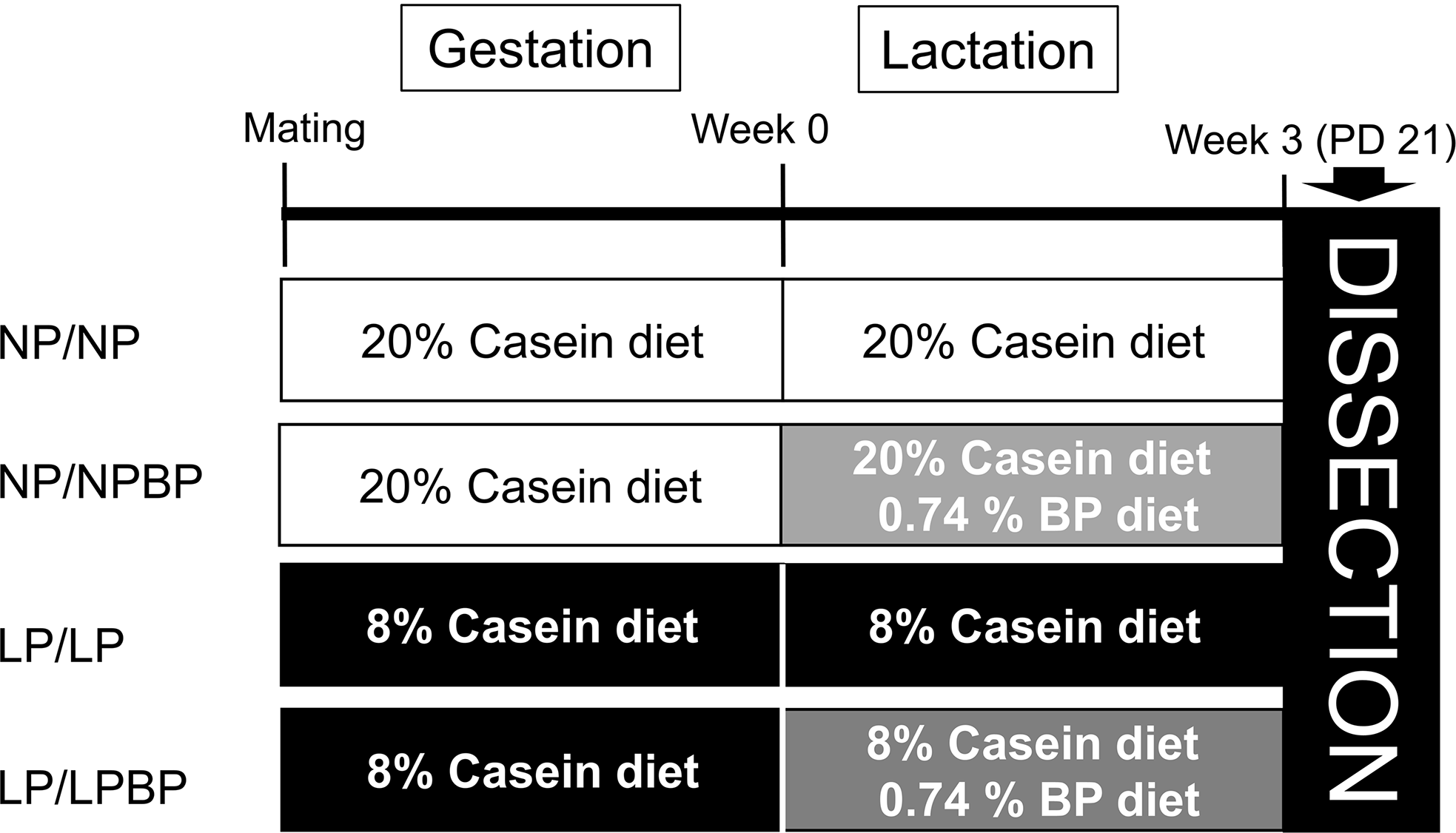
Twelve-week-old male and female Wistar rats obtained from CLEA Japan, Inc. (Tokyo, Japan). They were maintained at ≈23°C with 50%–60% humidity under a 12:12 h light/dark cycle, with free access to a standard diet (CE-2, CLEA Japan, Inc.) and distilled water. At 13–14 weeks, a vaginal impedance reader was used to determine whether the female rats were in the stage of the estrus cycle for mating. One suitable female was mated with one male overnight, and the presence of a vaginal plug the next morning marked gestational day 0. Pregnant rats were randomly divided to two groups and fed diets with 20% casein (normal protein [NP], n = 9) or 8% (low-protein [LP], n = 9) ad libitum during gestation. Post-delivery, dams received diets as follows; normal-protein diet during gestation and lactation (NP/NP, n = 5), normal-protein diet during gestation and 0.74% BP-containing control diet during lactation (NP/NPBP, n = 4), low-protein diet during gestation and lactation (LP/LP, n = 5), and low-protein diet during gestation and 0.74% BP-containing low-protein diet during lactation (LP/LPBP, n = 4) (Fig. 1). The diets were isocaloric (Supplementary Table S1). The BP-containing 135 mg (approximately 0.31 mmol of Gr per gm BP) was supplied by KAGOME Co., Ltd. (Nagoya, Japan). We administered a diet containing 0.74% BP, which was equivalent to a diet containing 0.1% Gr (approximately 2.3 mmol Gr per 1 kg diet), based on a previous study.Reference Fujita, Fujita and Ishima25
Experimental design. NP/NP, 20% casein (normal protein [NP]) diet during gestation and lactation; NP/NPBP, normal-protein diet during gestation, 0.74% broccoli powder (BP)-containing normal-protein diet during lactation; LP/LP, 8% casein (low protein [LP]) diet during gestation and lactation; LP/LPBP, low-protein diet during gestation, 0.74% BP-containing low protein diet during lactation.

Pups were weighed on postnatal day 4 and housed. In this study, we used female pups, because we reported that the increased hypothalamic AMPK phosphorylation levels and the decreased hepatic AMPK phosphorylation levels in 3-week-old female offspring from fructose-fed rat dams.Reference Mukai, Ozaki, Serita and Sato26
At 21 days of age, after fasting for 15–16 h, the female pups were randomly selected from each of the four groups and were weighed. Thereafter, blood samples were collected from the abdominal aorta under anesthesia (mixed anesthetic agents: medetomidine, midazolam, and butorphanol), as previously described.Reference Kawai, Takagi, Kaneko and Kurosawa27 The hypothalamus and liver were quickly removed, and liver was weighed. Portions of the liver were placed in formaldehyde for immunohistochemical analysis. The regions including arcuate nucleus and ventromedial hypothalamus and the left lateral lobe of liver were immediately frozen in liquid nitrogen and stored at –80°C for other analyses later.
Measurements of plasma biochemical parameters
Plasma samples were separated by centrifugation and tested for glucose (Glc) and triglyceride (TG) levels using a commercially available kit (FUJIFILM Wako Pure Chemical Co. Japan). Plasma insulin levels were measured using an LBIS rat insulin ELISA kit (AKRIN-010T; FUJIFILM Wako Shibayagi Co., Gunma, Japan).
Immunohistochemistry
Paraformaldehyde-fixed liver tissues were dehydrated through a series of graded ethanol, cleared in xylene, and embedded in paraffin. Paraffin-embedded liver tissues were sectioned, and sections were subsequently deparaffinized and rehydrated. Sections were pretreated with 0.1% pepsin solution and 3% hydrogen peroxide (H2O2) followed by blocking with 1.5% skimmed milk in PBS. The sections were incubated with an anti-CD68 antibody (1.0 mg/ml; 1:60; MCA341R, mouse monoclonal antibody, clone; ED1, Bio-Rad, Hercules, CA, USA), and subsequently with Histofine Simple Stain Rat MAX PO (Nichirei Biosciences Inc., Tokyo, Japan). Positive reactions were visualized using 3,3′-diaminobenzidine tetrahydrochloride in 50 mM Tris-HCl buffer (pH 7.4) containing 3% (v/v) H2O2. The sections were lightly counterstained with hematoxylin. To count the cells showing CD68 immunopositivity in the liver, 10 randomly selected fields were examined. Quantification was performed using an Olympus BX43 microscope with a digital camera and the CellSens Dimension software (Olympus Co., Tokyo, Japan).
Western blotting
Hypothalamic and liver tissues were homogenized in a homogenizing buffer (Radioimmunoprecipitation Assay buffer; RIPA buffer) containing 1 mM phenylmethylsulfonyl fluoride and a protease inhibitor cocktail (Roche Applied Science, Indianapolis IN, USA), respectively. The resulting homogenates were centrifuged, and the supernatants were collected for analysis of protein concentration. These proteins were electrophoresed on 10%–15% SDS-polyacrylamide gels and then transferred to PVDF membranes. A single membrane was cut into specific molecular weight regions, and each membrane segment was probed with one primary antibody. No membrane stripping or reprobing was performed. The membrane segments were blocked with non-fat dry milk and incubated overnight with antibodies targeting specific proteins such as rabbit antibodies against AMPK (#2532), phospho-AMPK-Thr172 (#2531), mTOR (#2972), phospho-mTOR-Ser2448 (#2971), protein kinase B (Akt #9272), phospho-Akt-Ser473 (#9271), and nuclear factor kappa B (NFκB) p65 (#8242; Cell Signaling Technology, Danvers, MA, USA). Protein bands were visualized using enhanced chemiluminescence (ECL) western blot detection reagents (Global Life Science Solution, Buckinghamshire, UK) on Hyperfilm (Cytiva, Sweden), and the density of specific bands was quantified (ATTO Corp., Tokyo, Japan). Protein levels were normalized to β-actin levels within the same sample and mentioned as fold change with reference to control group.
RT-PCR
Total RNA was extracted from the hypothalamus and liver using the Direct-zol RNA MicroPrep Kit (Zymo Research, Irvin, CA, USA) according to the manufacturer’s instructions. A High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific, Waltham, MA, USA) was used to synthesize cDNA. TNF-α, DNA methyltransferase (Dnmt) 1, Dnmt 3a, ionized calcium binding adaptor molecule 1 (Iba1), glial fibrillary acidic protein (GFAP), and β-actin mRNA levels were analyzed using the primers (Supplementary Table S2). Real-time polymerase chain reaction was performed using StepOnePlus™ (Applied Biosystems, Foster city, CA, USA) with the PowerUp™ SYBR™ Green Master Mix (Thermo Fisher Scientific) according to the manufacturer’s instructions. Gene expression levels are expressed relative to those of β-actin levels. Fold differences were calculated using the delta–delta CT method using the StepOne™ (Applied Biosystems) software.
Statistical analysis
All data were analyzed using IBM SPSS Version 29.0. Data are expressed as the mean ± standard error of the mean (SEM). Statistical analyses were performed using one-way analysis of variance followed by post hoc Tukey’s test. Statistically significance was considered at p < 0.05.
Results
Body weights during lactation
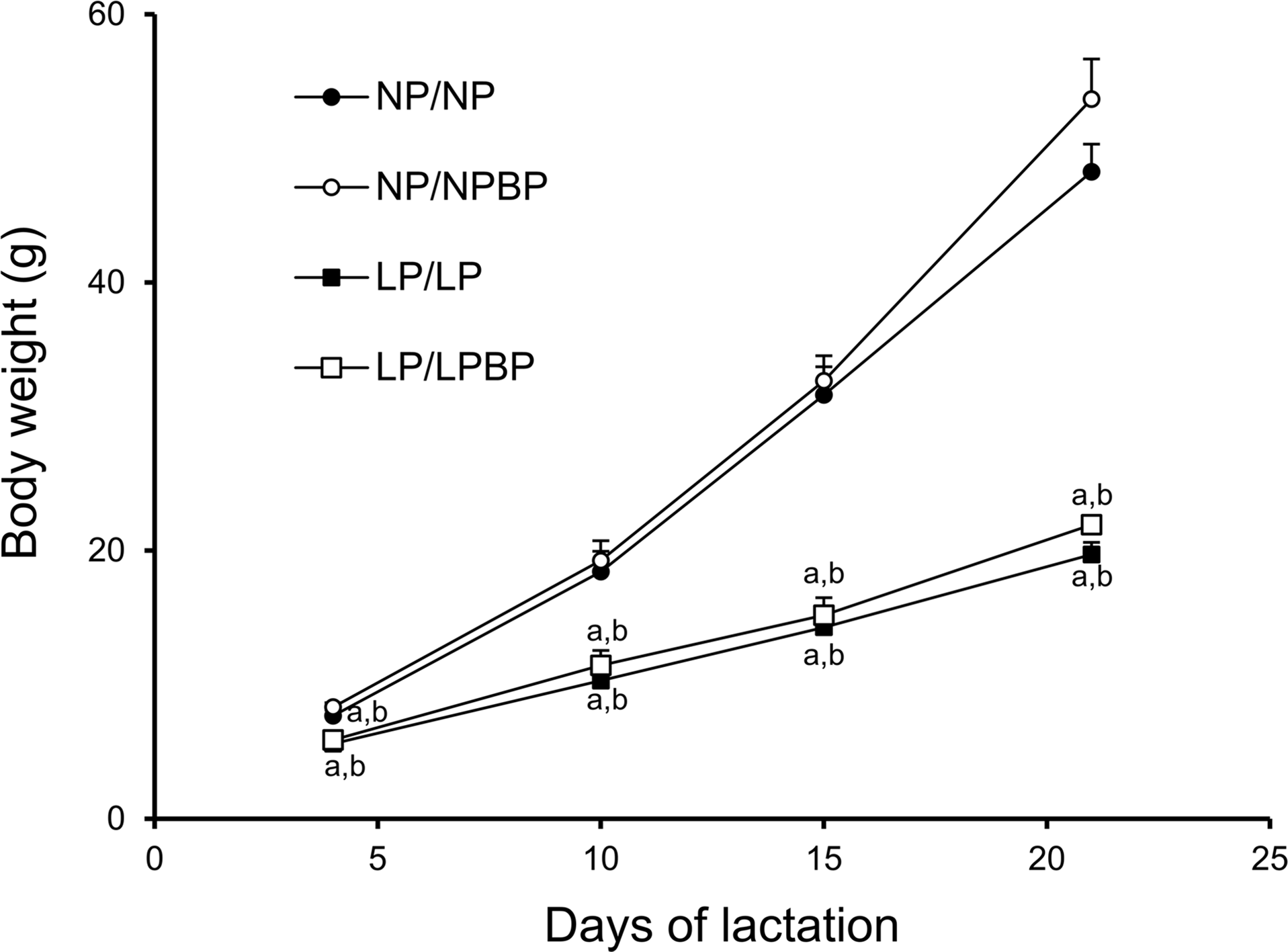
The body weights of female pups during lactation were significantly lower in the LP/LP and LP/LPBP group compared to both NP/NP and NP/NPBP groups (Fig. 2). No significant difference was found between LP/LP and LP/LPBP groups and NP/NP and NP/NPBP groups, respectively, though the body weights of NP/NPBP tended to be higher (p = 0.1).
Effect of broccoli powder (BP) intake during lactation on body weights of female offspring during the lactation period. Values are presented as mean ± SEM (n = 3–9 pups/litter). ap < 0.05 compared with NP/NP, bp < 0.05 compared with NP/NPBP.

Liver weights, plasma glucose, triglyceride, and insulin levels
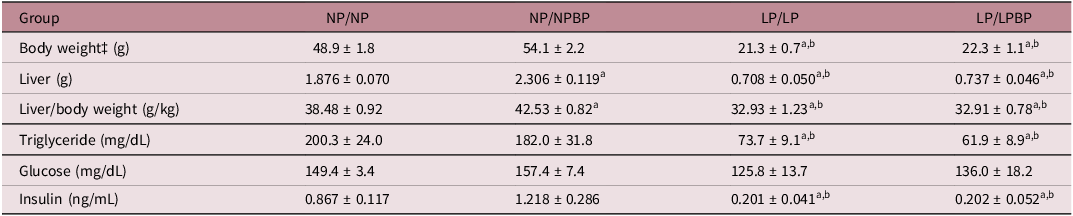
The relative liver weights were significantly lower in the LP/LP and LP/LPBP groups compared to the NP/NP and NP/NPBP groups, respectively, no significant changes were found between the LP/LP and LP/LPBP groups (Table 1). The relative liver weights were significantly heavier in the NP/NPBP compared to the NP/NP group. The plasma Glc levels were not significantly changed between the different groups. The plasma insulin levels were significantly lower in the LP/LP and LP/LPBP groups than in the NP/NP and NP/NPBP groups. There was no significant difference in the insulin levels between LP/LP and LP/LPBP groups and NP/NP and NP/NPBP groups, respectively. The plasma TG levels were significantly lower in the LP/LP and LP/LPBP compared to both the NP/NP and NP/NPBP, though no significant difference was observed between the LP/LP and LP/LPBP groups (Table 1).
Morphological characteristics and plasma parameters of female offspring

‡ At sacrifice. Values are presented as mean ± SEM. (n = 9–10).
a p < 0.05 compared with NP/NP.
b p < 0.05 compared with NP/NPBP.
Effect of BP intake on the inflammatory status in the liver
The CD68-positive macrophages number was significantly higher in the LP/LP group compared to the NP/NP group (Fig. 3a and 3b). Conversely, the number in the LP/LPBP group was significantly lower than the LP/LP group. The levels of NFκB p65 protein expression was significantly lower in the LP/LPBP group compared to the LP/LP group though no significant difference was observed between the LP/LP and NP/NP groups (Fig. 3c). The levels of TNF-α mRNA expression were significantly higher in the LP/LP group than in the NP/NP group. In contrast, the TNF-α mRNA levels in the LP/LPBP group were significantly lower compared to the LP/LP group (Fig. 3d).
Effect of broccoli powder (BP) intake during lactation on the inflammatory status in the liver of offspring. (a and b) CD68-positive macrophage infiltration and counts, (c) nuclear factor-kappa B (NFκB) p65 protein expression levels, and (d) tumor necrosis factor (TNF)-α mRNA expression levels. The arrows indicate CD68-positive macrophages (a). Counterstaining was performed with hematoxylin (scale bars: 50 μm in the upper images and 20 μm in the lower images). Values are presented as the mean ± SEM (n = 9–10). ap < 0.05 compared with NP/NP, bp < 0.05 compared with NP/NPBP, cp < 0.05 compared with LP/LP. scale bar: 50 μm for upper images, 20 μm for bottom images.

Effect of BP intake on the inflammatory status in the hypothalamus
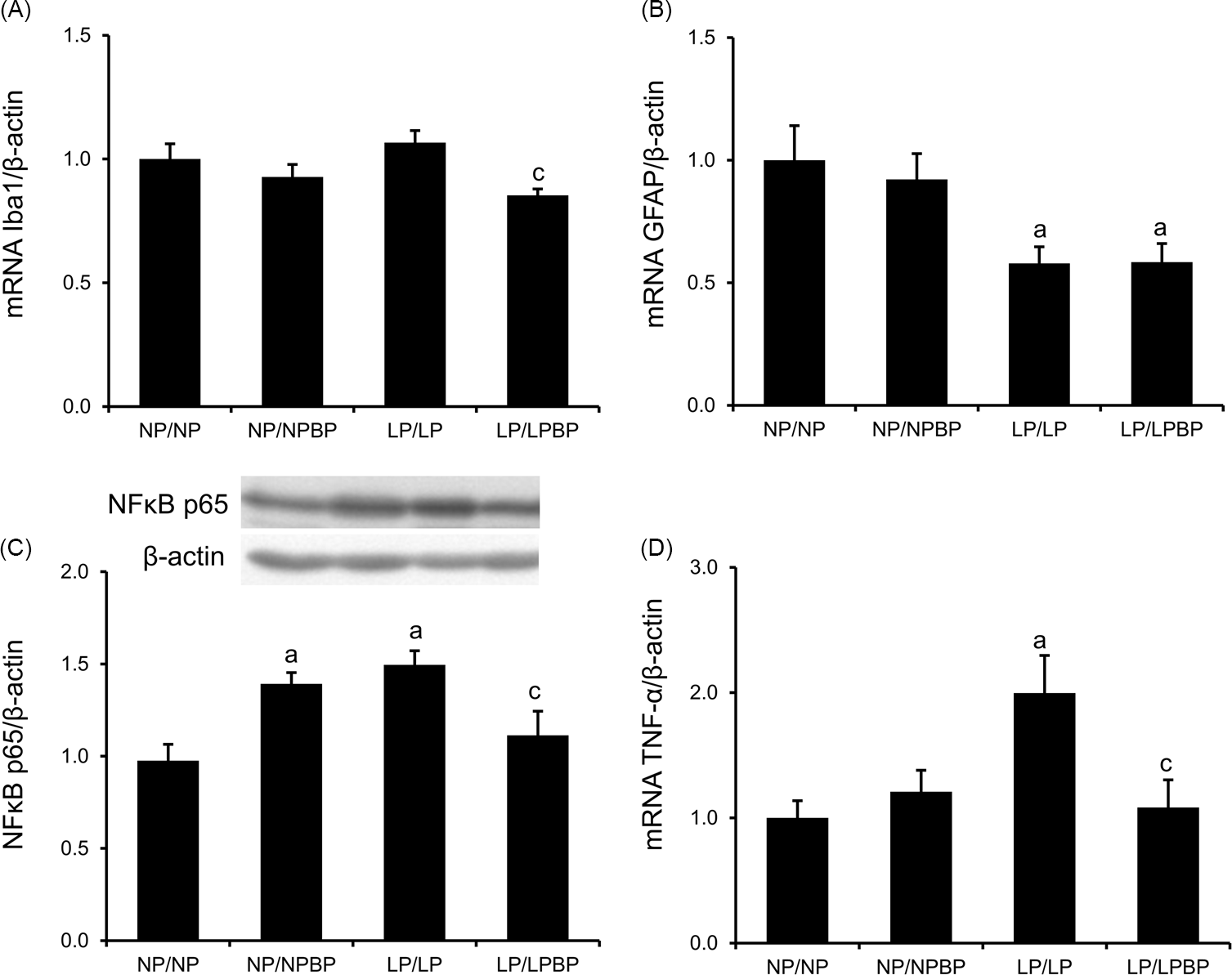
The mRNA expression levels of Iba1, a microglia marker, in the LP/LPBP group were significantly lower than in the LP/LP group, though no significant difference was found between the LP/LP and NP/NP groups (Fig. 4a). The mRNA expression levels of GFAP, an astrocyte marker, in the LP/LP and LP/LPBP groups were significantly lower compared to the NP/NP and NP/NPBP, respectively (Fig. 4b). No significant differences were shown between the LP/LP and LP/LPBP groups. The expression levels of NFκB p65 protein as well as the TNF-α mRNA were significantly higher in the LP/LP group compared to the NP/NP group (Fig. 4c and 4d). Conversely, these levels of in the NFκB p65 protein and TNF-α mRNA in the LP/LPBP group were significantly lower than in the LP/LP group, respectively. In addition, the levels of NFκB p65 in the NP/NPBP group was significantly higher than the NP/NP group.
Effect of broccoli powder (BP) intake during lactation on the inflammatory status in the hypothalamus of offspring. (a) Iba1 mRNA expression levels, (b) GFAP mRNA expression levels, (c) nuclear factor-kappa B (NFκB) p65 protein expression levels, and (d) tumor necrosis factor (TNF)-α mRNA expression levels. Values are presented as the mean ± SEM (n = 8–10). ap < 0.05 compared with NP/NP, bp < 0.05 compared with NP/NPBP, cp < 0.05 compared with LP/LP.

Effect of BP intake on phosphorylation and protein expression of AMPK, mTOR, and Akt in the liver and hypothalamus
In the liver, the phosphorylated AMPK levels were significantly higher in the LP/LP group compared to the NP/NP group (Fig. 5a). In the LP/LPBP group, the phosphorylated AMPK levels were significantly higher than in the LP/LP group. The phosphorylated levels of mTOR as well as Akt in the LP/LPBP group were significantly lower than in the LP/LP group (Fig. 5b and 5c). No significant changes were found in the levels of total AMPK, mTOR, and Akt protein across different groups.
Effect of broccoli powder (BP) intake during lactation on phosphorylation and protein expression of AMP-activated protein kinase (AMPK), mechanistic target of rapamycin (mTOR), and Akt in the liver of offspring. (a, b, and c) levels of expression and phosphorylation of AMPK, mTOR, and Akt, respectively. Values are presented as the mean ± SEM (n = 9–10). ap < 0.05 compared with NP/NP, bp < 0.05 compared with NP/NPBP, cp < 0.05 compared with LP/LP.

In the hypothalamus, the phosphorylated AMPK levels were significantly higher in the NP/NPBP and the LP/LPBP groups than in the NP/NP and LP/LP groups, respectively (Fig. 6a). No significant changes were found in the phosphorylated and total mTOR and Akt protein levels in the four groups, respectively (Fig. 6b and 6c).
Effect of broccoli powder (BP) intake during lactation on phosphorylation and protein expression of AMP-activated protein kinase (AMPK), mechanistic target of rapamycin (mTOR), and Akt in the hypothalamus of offspring. (a, b, and c) levels of expression and phosphorylation of AMPK, mTOR, and Akt, respectively. Values are presented as the mean ± SEM (n = 8–10. ap < 0.05 compared with NP/NP, bp < 0.05 compared with NP/NPBP, cp < 0.05 compared with LP/LP.

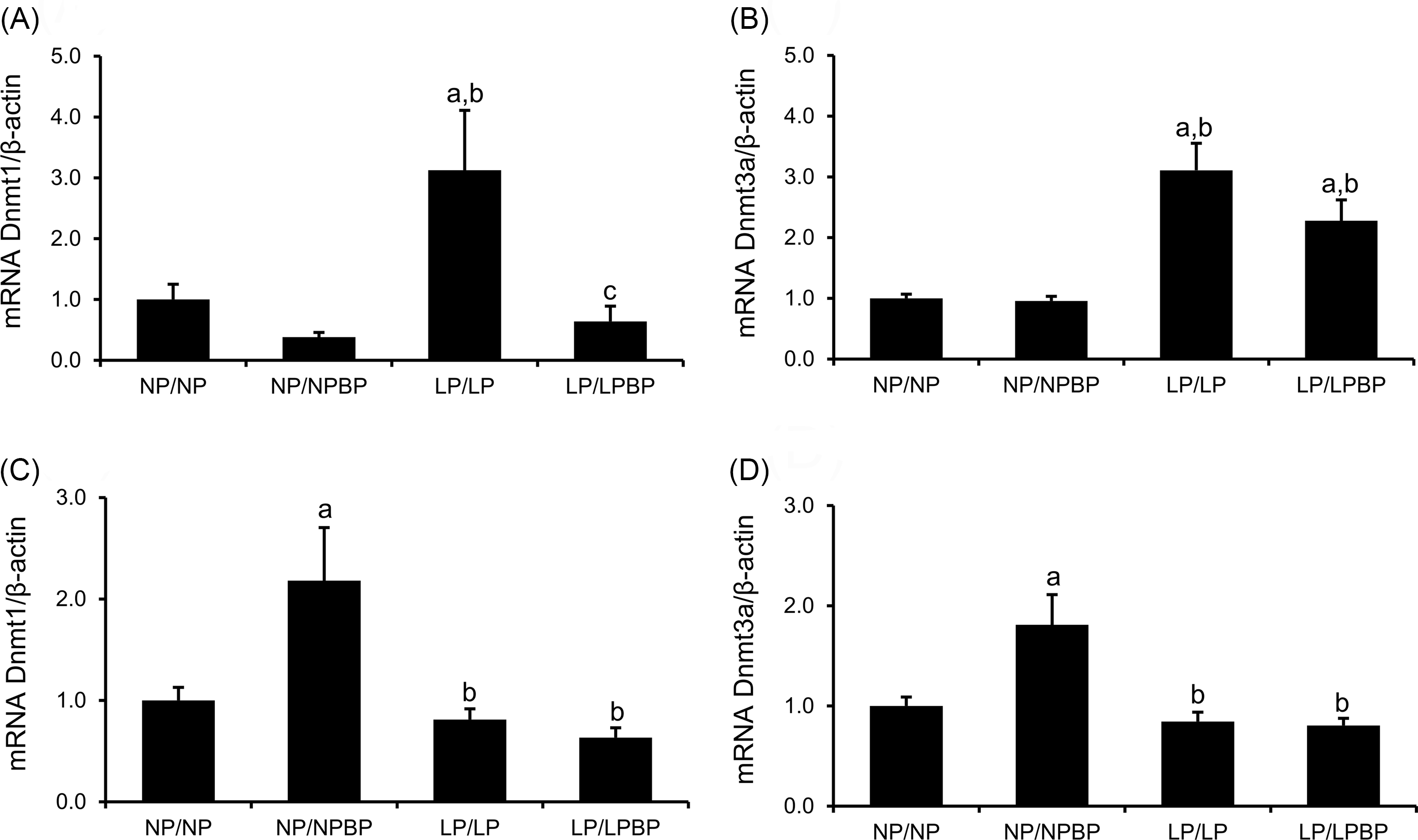
Effect of BP intake on mRNA expression of Dnmt1 and Dnmt3a in the liver and hypothalamus
In the liver, the mRNA expression levels of Dnmt1 and Dnmt3a in the LP/LP group were significantly higher than in the NP/NP group (Fig. 7a and 7b). Conversely, the Dnmt1 levels in the LP/LPBP was significantly decreased compared to the LP/LP group, however, no significant differences were found in the level of Dnmt3a.
Effect of broccoli powder (BP) intake during lactation on mRNA expression of DNA methyltransferase (Dnmt) 1 and Dnmt3a in the liver and the hypothalamus of offspring. (a and b) levels mRNA expression of Dnmt1 and Dnmt3a in the liver. (c and d) levels mRNA expression of Dnmt1 and Dnmt3a in the hypothalamus. Values are presented as the mean ± SEM (a and b; n = 9–10, c and d; n = 8–10). ap < 0.05 compared with NP/NP, bp < 0.05 compared with NP/NPBP, cp < 0.05 compared with LP/LP.

In the hypothalamus, there was no significant difference in the Dnmt1 and Dnmt3a levels between the LP/LP and NP/NP groups, respectively (Fig. 7c and 7d). The mRNA expression levels of Dnmt1 and Dnmt3a were significantly increased in the NP/NPBP group compared to the NP/NP groups. No significant differences in the levels of Dnmt1 and Dnmt3a were observed between the LP/LP and LP/LPBP groups.
Discussion
Maternal malnutrition or protein restriction during pregnancy is involved in the long-term metabolic consequences such as obesity, type 2 diabetes, and liver disease. Additionally, nutritional imbalance during gestation and lactation can cause inflammation. The action of Gr is involved in obesity-related inflammation, insulin resistance, and energy homeostasis.Reference Xu, Nagata and Ota28 However, there is limited information about the effect of maternal BP intake during lactation on inflammation and AMPK phosphorylation in the liver and hypothalamus in weaning offspring programmed by maternal protein restriction.
Our key highlights of the present study are as follows: in female rat offspring programmed by maternal protein restriction, when BP was given during lactation, (a) macrophage counts, the expression levels of NFκB p65 protein and TNF-α mRNA decreased in the liver, (b) in the hypothalamus, the expression levels of NFκB p65 protein, Iba1 and TNF-α mRNA decreased, (c) AMPK phosphorylation upregulated in the liver though the mTOR and Akt phosphorylation downregulated, (d) in the hypothalamus, AMPK phosphorylation upregulated though no changes on the mTOR and Akt phosphorylation, (e) the expression levels of Dnmt1 mRNA decreased in the liver but not in the hypothalamus.
We confirmed that maternal low protein diet during pregnancy and lactation reduced body and liver weights in weaning offspring, even when BP diet was given during lactation. Several studies showed maternal undernutrition can lead to the intrauterine growth restriction and resulting in the low birth weight of offspring in human and animal studies.Reference Lumey and Stein1,Reference Connor, Kibschull and Matysiak-Zablocki2 In addition, we showed that the plasma insulin levels in the LP diet groups decreased significantly compared to the NP diet groups. A maternal low protein diet has reported to impair insulin secretion in offspring.Reference Leandro, Fidalgo and Bento-Santos29,Reference Alejandro, Jo and Akhaphong30 Therefore, BP intake during lactation may not affect plasma insulin concentration in the weaning offspring whose insulin secretion was already impaired by maternal protein restriction. In the NP/NPBP group, the increased liver weights and a tendency of increased plasma insulin levels (p = 0.4) were observed. SFN was reported to enhance insulin-like growth factor binding protein-3 (IGFBP-3) expression.Reference Dang, Huang and Chen31 IGFBP-3, which binds to insulin-like growth factor (IGF), which is intimately associated with the organism’s growth and development processes.Reference Baxter32 Thus, in the NP/NPBP group, BP intake during lactation might affect the increase of the liver weights via increased IGFBP-3 expression, though further studies are required to validate these effects of BP during lactation on the liver.
In this study, we demonstrated that BP intake during lactation decreased the NFκB p65 protein and TNF-α mRNA expression levels in the liver as well as hypothalamus of weaning offspring from LP-fed mothers. Gr attenuated activated macrophage accumulation and inflammatory mediators; TNF-α and chemokine (C–C motif) ligand 2 (Ccl2) mRNA in the liver of high-fat diet-fed mice.Reference Nagata, Xu and Kohno33 In addition, SFN was reported to decrease the mRNA levels of proinflammatory mediators; IL-1β and IL-6 in the liver and brain of LPS-challenged mice.Reference Townsend and Johnson34 The consumption of broccoli sprouts showed the decreased IL-6 levels and C-reactive protein in obese subjects.Reference López-Chillón, Carazo-Díaz and Prieto-Merino23 The inhibition of TNF-α reduced hypothalamic inflammation and improved insulin signal transduction in the liver.Reference Milanski, Arruda and Coope12 Taking together, we hypothesized that maternal BP intake during lactation may prevent inflammation in the liver as well as hypothalamus of the weaning offspring triggered by maternal low protein diet.
Next, we demonstrated that BP intake increased the phosphorylated AMPK levels in the liver of the weaning offspring exposed to maternal protein restriction. The increased AMPK activation contributes to the improved insulin resistance, anti-inflammatory effects and antioxidant defense.Reference Ruderman, Carling, Prentki and Cacicedo35 For instance, in high-fat diet fed mice, Gr reduced hepatic steatosis and inflammation via upregulation of AMPK phosphorylation.Reference Promsuwan, Sawamoto and Xu36 Notably, the phosphorylated mTOR and Akt levels in the liver decreased in the LP/LPBP group compared to the LP/LP group. The upregulated mTOR activation in the muscle of obese and high-fat-fed rodents leads to inhibition of insulin signaling, which suppresses glucose uptake by the muscle, contributing to the increased insulin resistance.Reference Laplante and Sabatini37 Impairment of Akt activity is associated with obesity and insulin resistance induced by a high-fat diet.Reference Satoh38 Therefore, BP intake during lactation may contribute to the prevention of insulin resistance in the liver through the upregulated AMPK activation and the downregulated mTOR and Akt activation in offspring programmed by maternal low protein diet.
We showed that in the hypothalamus, the phosphorylated AMPK levels were upregulated in the NP/NPBP and LP/LPBP groups, respectively, though no significant differences in the phosphorylated mTOR and Akt levels. SFN treatment has reported to increase AMPK phosphorylation levels in rat brain after carbon monoxide poisoning.Reference Yue, Zhou and Song39 In the hypothalamus, AMPK modulates whole-body energy homeostasis and body weight by inducing appetite to increase food intake and diminishing adaptive thermogenesis in adipose tissues.Reference Andersson and Filipsson40 Therefore, we assumed that in the hypothalamus, the increased AMPK phosphorylation caused by BP diet during lactation in both the NP and LP groups may lead to higher appetite and lower energy expenditure.
Interestingly, the Dnmt1 mRNA levels increased in the liver of the LP group compared to NP group. Conversely, maternal BP decreased the Dnmt1 levels of weaning offspring born to low-protein diet fed mothers. Dnmt1 levels have been reported to increase in the liver of patients with steatohepatitis.Reference Pirola, Gianotti and Burgueño41 Pro-inflammatory cytokines such as TNF-α and IL-1β in obesity-induced mice increased the Dnmt1 expression and its activity. When the Dnmt1 activity was inhibited, the obesity-induced glucose intolerance and insulin resistance were attenuated.Reference Kim, Park and Pan42 Thus, the hepatic suppression of Dnmt1 levels in the offspring fed BP during lactation may contribute to the prevention of insulin resistance in adulthood.
In the hypothalamus, maternal BP intake increased the mRNA levels of Dnmt1 and Dnmt3a in weaning offspring born to the NP diet fed mothers but not the LP diet. Dnmt1 in the hypothalamus regulates energy homeostasis via controlling food intake and energy expenditure. The deletion of Dnmt1 in the hypothalamus improved insulin tolerance in high-fat diet-fed mice by reducing feed intake and increasing energy expenditure.Reference Bruggeman, Garretson, Wu, Shi and Xue43 Dnmt3a is thought to generate the patterns of DNA methylation in the hypothalamic feeding center during the early postnatal period in rodents.Reference Yoshikawa, Ariyani and Kohno44 Therefore, the increased Dnmt1 and Dnmt3a mRNA in the hypothalamus of the NP/NPBP group may be associated with the modification of feed intake and energy expenditure.
Conclusion
In conclusion, we demonstrated that maternal BP intake during lactation suppressed the expression levels of NFκB p65 protein and TNF-α mRNA in the liver as well as the hypothalamus of offspring programmed by maternal protein restriction. Furthermore, we showed that maternal BP intake upregulated AMPK phosphorylation and downregulated the mTOR and Akt phosphorylation in the liver. In the hypothalamus, maternal BP intake upregulated the AMPK phosphorylation in offspring fed maternal normal- and low- protein diets. Thus, maternal BP diet during lactation may be a useful preventive strategy, at least, for the inhibition of inflammation in the liver as well as hypothalamus in weaning offspring programmed by maternal protein restriction. However, further investigation is needed to elucidate the underlying molecular mechanisms of the increased AMPK activation in the hypothalamus of weaning offspring received BP diet during lactation, and impacts of BP diet during lactation on appetite and energy expenditure in adulthood.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S2040174426100543.
Acknowledgments
The authors thank Keiko Tamakuma for providing technical assistance.
Author contributions
K.A was responsible for conceptualization, data analysis, writing of the original draft. K.S carried out data acquisition and data analysis. M.Y was responsible for provision of resources, data acquisition and review of the original draft. S.S was responsible for provision of resources, writing, review and editing of the original draft, supervision and conceptualization. All authors read and approved the manuscript for publication.
Financial support
This study was supported by JSPS KAKENHI Grant (No.23K10940).
Competing interests
None.
Ethical standard
Our study was approved by the Animal Research Committee of Aomori University of Health and Welfare (permission number: 21008), and all procedures followed Institutional Guidelines for Animal Experimentation. It was carried out in accordance with the spirit of animal welfare in accordance with the “Guidelines for Animal Experiments at Aomori Prefectural University of Health.” The Guidelines for animal experiments are created based on the “Action on the Protection and Management of Animals,” “Basic Guidelines for the Implementation of Animal Experiments at Research Institutions,” and “Standards for Breeding and Storage of Laboratory Animals and Relief of Pain.”