Introduction
Trepanning of the bone is one of the oldest known procedures carried out by man and the use of the modern trephine biopsy has a venerable history. Parapia has published an admirable summary of the history of the topic and this should be consulted for the excellent illustrations of historical instruments [Reference Parapia1]. The history is briefly summarized here [Reference Parapia1]. Trepanning of the skull is the oldest known surgical procedure in humans and evidence of this practice has been found in Europe, North Africa, South America, Asia and New Zealand. In Peru, where the procedure is likely to have been carried out to treat headache, mental illness and to relieve intracranial pressure, sharp knives of obsidian, stone and bronze were used for trephination. Celsus, the Roman physician, described a modiolus – an iron instrument with a serrated cylinder that was rotated over a central pin by means of a strap. The early interventions were therapeutic and the first diagnostic biopsy was undertaken in Pianese in Italy in 1903. In 1922, Morris and Falconer used a drill-like instrument to biopsy the tibia, producing similar specimens to modern biopsies and, in the same year, Seyfarth developed a puncture needle for open biopsy of the sternum, producing smears, touch preparations and blocks for sectioning. The modern era probably began in 1958 when McFarland and Dameshek described a technique for biopsy of the right posterior iliac crest using a Silverman needle, which had been described in 1938. Further improvements followed, with modified instruments described by Jamshidi in 1971 and an electric drill technique by Burkhardt in 1971. Recent developments are described later in the chapter.
In modern practice, bone marrow (BM) examination, including both aspiration and biopsy sampling, can be performed on virtually any patient. However, patients with coagulation deficiencies or profound thrombocytopaenia may experience prolonged bleeding, which cannot be controlled by pressure bandages. In these rare cases, specific treatment (e.g. platelet transfusion) may be indicated. In a study of 19 259 procedures, including 13 147 combined procedures and 6112 aspirates, a total of 16 adverse events were reported, representing 0.08% of all reported procedures. The adverse events were eleven haemorrhages, three very serious, with other complications being two infections, two cases of persistent pain and a serous leak persisting for six days [Reference Bain2].
Indications for performing BM examination are summarized in Table 1.1. It has long been accepted that in the vast majority of cases, both aspiration and a biopsy should be performed and are complementary [Reference Bain3, Reference Bain4]. Bone marrow aspiration provides excellent cytological detail; however, marrow architecture cannot be assessed. Bone marrow core biopsy (BMB) allows for an accurate analysis of architecture but fine cytological details may be lost. Table 1.2 shows the accepted indications for performing a BMB. These include cases with inadequate or failed aspiration, a need for accurate assessment of BM cellularity, architecture, stroma, vascularity and bone structure, and cases in which the presence of focal lesions, such as granulomatous disease or metastatic carcinoma, is suspected. In general, patients with hypocellular marrows or BM fibrosis are likely to need a BMB for adequate assessment. In such patients, an aspirate would probably be inadequate or even impossible. Unexplained pancytopaenia and unexplained leukoerythroblastic blood pictures are further indications for a BMB, because they are likely to indicate the presence of BM metastatic disease or fibrosis. Focal skeletal lesions may also require a BMB when focal manifestation of generalized disease, e.g. plasma cell myeloma or metastatic malignancy, may be found. Targeted BMB may also be used.
Table 1.1 Indications for bone marrow aspiration with or without a trephine biopsy.
| Investigation and/or follow-up of: |
|---|
| Unexplained microcytosis |
| Unexplained macrocytosis |
| Unexplained anaemia |
| Unexplained thrombocytopaenia |
| Pancytopaenia (including suspected aplastic anaemia) |
| Leukoerythroblastic blood smear and suspected bone marrow infiltration |
| Suspected acute leukaemia |
| Suspected myelodysplasia or myelodysplastic/myeloproliferative disorder |
| Suspected myeloproliferative neoplasms |
| Suspected chronic lymphocytic leukaemia, hairy cell leukaemia and other leukaemic lymphoproliferative disorders |
| Suspected non-Hodgkin lymphoma |
| Staging of non-Hodgkin lymphoma |
| Suspected plasma cell myeloma or other plasma cell dyscrasias |
| Suspected storage disease |
| Fever of unknown origin (with microbial culture) |
| Investigation of anaemia in immunodeficiency |
| Confirmation of normal bone marrow if bone marrow is being aspirated for allogeneic transplantation |
| Follow-up and monitoring of therapy |
Table 1.2 Indications for performing a bone marrow biopsy.
| Investigation and/or follow-up of: |
|---|
| Diagnosis and/or staging of suspected Hodgkin lymphoma and non-Hodgkin lymphoma |
| Chronic lymphocytic leukaemia, hairy cell leukaemia and other leukaemic lymphoproliferative disorders |
| Diagnosis of suspected metastatic carcinoma |
| Diagnosis, staging and follow-up of small cell tumours of childhood |
| Myeloproliferative neoplasms, myeloproliferative/myelodysplastic neoplasms, systemic mastocytosis |
| Diagnosis of aplastic anaemia |
| Investigation of an unexplained leukoerythroblastic blood smear |
| Investigation of a fever of unknown origin and/or granulomatous infection |
| Investigation of suspected haemophagocytic syndrome |
| Evaluation of any patient in whom an adequate bone marrow aspirate cannot be obtained |
| Suspected plasma cell myeloma and other plasma cell dyscrasias |
| Suspected acute myeloid leukaemia |
| Suspected myelodysplastic syndrome |
| Investigation of suspected storage disease |
| Suspected primary amyloidosis |
| Investigation of bone diseases |
The BMB specimen differs from biopsy material from most other organs, because a proper interpretation of the BM in haematological disorders requires the incorporation of clinical data, BM aspirate studies, peripheral blood smears and complete blood count data, which should be evaluated in conjunction with flow cytometry and molecular investigations, to form a composite report in order to fulfil WHO disease definitions and comply with current guidelines [Reference Swerdlow, Campo and Harris5–Reference Ramsay, Pomplun and Wilkins8]. Occasionally, BMB imprint smears may be used to assess cytology in cases with inaspirable marrows (dry taps). The benefits of synoptic reporting in a multiparameter diagnosis are well established and check lists and general recommendations have been published in the USA and UK [Reference Sever, Abbott and Baca6, Reference Ramsay, Pomplun and Wilkins8]. The following chapters will emphasize this multifactorial approach to BM evaluation, attempt to highlight the diagnostic questions that require the use of ancillary techniques for accurate diagnosis and provide guidance in their selection.
Obtaining the Bone Marrow Aspirate and Biopsy
The technique used to obtain BMB and aspirate smears has been described in detail in standard publications and the reader is referred to those for more detail [Reference Orazi, Knowles, Foucar and Weis9, Reference Lee, Erber, Porwit, Tomonaga and Peterson10]. Separate guidelines for children have also been published [Reference Abla, Friedman and Doyle11]. If BM aspiration and biopsy are both being performed using the same needle, it is usually preferable to obtain the biopsy first, to avoid distortion of the biopsy specimen by aspiration artefacts. Another approach uses separate needles for each procedure. This requires that the needles be placed in different sites of the bone marrow; good-quality aspirate and biopsy can be obtained in either sequence. When two needles are used, it is often advantageous to aspirate the marrow through a smaller needle specifically designed for that purpose. This minimizes the contamination with peripheral blood, which is often observed when the aspirate is performed through a Jamshidi needle.
In recent years, new and potentially improved BMB devices have been developed, including powered devices. However, a recent study suggested that, despite the ability of powered devices to yield longer BMB lengths, the method is costlier than manual BMB and does not result in a discernible improvement in the quality of BM specimens [Reference Maitia, Short and Verstovsek12]. A recent review of a number of different devices concluded that the newly launched Möller Medical bone marrow biopsy needle (Möller Medical, Fulda, Germany) had definite advantages over the others tested, i.e. the J-needle by Cardinal Health (Cardinal Health, Waukegan, Illinois, USA), the Jamshidi bone marrow biopsy needle with marrow acquisition cradle by CareFusion (CareFusion, Vernon Hills, Illinois, USA) and the OnControl powered drill system by Vidacare (Vidacare, Texas, USA) [Reference Islam13].
The initial aspirate sample should always be used for morphology. Subsequent aspirations may be obtained for flow cytometry, cytogenetics, microbiology culture or molecular studies. Non-anticoagulated specimens should be immediately handed to a technical assistant who will prepare various smears and BM particle crush preparations. The remaining BM aspirate material is allowed to clot and submitted for BM clot sections. Alternatively, aspirate smears can be made in the laboratory after the procedure. To this end, BM aspirate material should be immediately placed into tubes containing ethylenediaminetetraacetic acid (EDTA). This limits the clotting of the aspirate specimen and allows material to be submitted for ancillary studies as well as particle sections. Whether the smears are made at the bedside or from the EDTA tubes, the aspirate should be grossly evaluated for the presence of BM particles. The absence of particles on a smear limits its diagnostic usefulness in many cases. Such smears often show findings consistent with peripheral blood contamination. Many paediatric patients, however, will not demonstrate gross particles in the aspirate material despite numerous BM elements in the smear.
Representative aspirate smears and imprints are stained with a Romanovsky type of stain. The actual stain type varies among laboratories and includes Giemsa, Wright–Giemsa and May–Grünwald–Giemsa stains. Rapid review of these smears helps in determining the need for ancillary studies, such as cytochemistry, immunophenotyping, cytogenetic analysis and molecular genetic study.
Clot biopsy sections are often made from coagulated aspirate material. This material contains predominantly blood as well as small BM particles that can be embedded in paraffin, sectioned and stained with haematoxylin and eosin (H&E) or other stains. Alternatively, when EDTA-anticoagulated aspirate specimens are used, the BM particles that are left after smears are prepared can be filtered and embedded for histologic evaluation [Reference Arber, Johnson and Rainer14]. This method provides a more concentrated collection of BM particles but may not yield any material, particularly in paediatric patients [Reference Brunning, Bloomfield, McKenna and Peterson15].
Trephine core biopsy is not performed for all patients, but these specimens are essential in the evaluation of patients suspected of having disease that may only involve the BM focally, such as malignant lymphoma or metastatic carcinoma, and are preferred in all patients. Core biopsies allow for architectural assessment of the BM and offer a number of other benefits. The incidences of BM involvement by various types of malignancies are proportional to the amount of BM evaluated. The minimum adequate length is in the range 15 to 20 mm [Reference Ramsay, Pomplun and Wilkins8]. To increase the yield, bilateral BMBs have been recommended for patients undergoing BM staging by several authors [Reference Brunning, Bloomfield, McKenna and Peterson15, Reference Wang, Weiss and Chang16] but this is no longer regularly performed [Reference Ramsay, Pomplun and Wilkins8].
Imprint slides from the biopsy specimens may yield positive same-day results in some of the aspirate-negative or dry-tap cases [Reference James, Stass and Schumacher17]. The imprints can be made either at the bedside or in the laboratory. To make them in the laboratory, the BM core is submitted fresh, on saline-dampened gauze, with the imprints made immediately to allow adequate fixation of the biopsy specimen. Otherwise, the imprints are made at the bedside and the biopsy specimen is submitted in fixative.
Fixation, Decalcification and Processing of the Biopsy
Bone marrow biopsies differ from most others in that decalcification is mandatory and excellent morphology is obligatory. Furthermore, molecular diagnostic techniques, covered in detail in Chapter 20, can be applied to BM biopsies and preservation of DNA and RNA is thus required to maximize the diagnostic yield. Archival BM biopsies provide an opportunity to examine previous biopsies for cytogenetic abnormalities that may shed light on progression of the disease and to use new diagnostic techniques unavailable at the time of the original diagnosis. However, methods for the preparation, processing and reporting of BM aspirates and BMB specimens can vary considerably and these differences may thus result in inconsistencies in disease diagnosis or classification that may affect treatment and clinical outcomes [Reference Lee, Erber, Porwit, Tomonaga and Peterson10]. An international Working Party for the Standardization of Bone Marrow Specimens and Reports was formed by the International Council for Standardization in Haematology (ICSH) to prepare a set of guidelines based on preferred best practices, which are summarized here, and the reader is referred to this comprehensive overview for further detail [Reference Lee, Erber, Porwit, Tomonaga and Peterson10].
Decalcification using methods including EDTA, formic acid, acetic acid, nitric acid and various commercial agents varies in time required for complete decalcification and in the preservation of cytomorphology, tissue iron and the ability to perform reliable, quantifiable immunohistochemistry and molecular diagnostic analysis [Reference Lee, Erber, Porwit, Tomonaga and Peterson10]. A recent study of the methods used by 28 different laboratories revealed that eight different fixatives and nine different decalcification methods were used [Reference Torlakovic, Naresh and Kremer18]. While 93% of participants believed that they produced excellent results in BMB immunohistochemistry (IHC), only 21% of laboratories did not have any poor results. This study led to a call for a reduction in the number of processing methods and calibration of results in external quality assessment (EQA) programmes [Reference Torlakovic, Naresh and Kremer18]. Elliot et al. have also addressed the variability of IHC staining in general and stressed the need for IHC laboratories to bring the same level of robust validation seen in the molecular pathology laboratories and called for the principles to be applied to all routine IHC tests participation, assessed in appropriate EQA schemes [Reference Elliott, McQuaid, Salto-Tellez and Maxwell19]. ICSH guidelines for the standardization of BM IHC have now been published based on currently available published evidence and modern understanding of quality assurance principles as applied to IHC in general [Reference Torlakovic, Brynes and Hyjek20]. The reader is referred to the article for detailed guidance but, in summary, the following core principles are defined.
Ischaemic Time
Depending on the availability of on-site technical support, preparation of BM biopsy imprints and aspirate smears at the bedside, to minimise cold ischaemic time and hence preserve protein epitopes relevant to IHC and RNA and DNA integrity, constitutes optimal practice. Transportation of fresh samples to the laboratory is suboptimal and increases epitope degradation and the risk of false negative results [Reference Torlakovic, Brynes and Hyjek20]. The BM biopsy should be rapidly placed into fixative for onward transmission to the laboratory after any additional techniques such as trephine biopsy roll preparations have been made.
Fixation
To avoid zoning, with areas of absent or suboptimal staining, fixation must be even and complete. 10% buffered formalin remains the common standard fixative but acetic acid–zinc–formalin (AZF) has been proposed as an alternative that improves laboratory throughput due to decreased decalcification time, while preserving IHC and molecular testing [Reference Naresh, Lampert and Hasserjian21, Reference Bonds, Barnes, Foucar and Sever22]. Local experience has not supported AZF as a method due to poor molecular results and careful evaluation is recommended before adoption [unpublished data]. Bouin’s solution has fallen out of favour due to laboratory safety issues and poor IHC results, and Zenker’s solution and B5 are now unavailable in many countries due to their mercuric chloride content [Reference Torlakovic, Brynes and Hyjek20].
Decalcification
Current optimal practice requires that recommendations for the IHC processing methods also take into account DNA and RNA integrity, and this applies to decalcification as well as fixation, and there may be a conflict between the ability to perform both IHC and molecular tests and turnaround time (TAT). Decalcification is best achieved by 14% EDTA, which sequesters metallic ions, including calcium, in aqueous solutions as a chelating factor for 16–24 hours [Reference Torlakovic, Brynes and Hyjek20]. Strong mineral acids such as 10% hydrogen chloride (HCl) or weak organic acids, such as 5–10% formic acid (HCOOH) form soluble calcium salts in an ion exchange that moves calcium into the decalcification solution and are used for decalcification, particularly when rapid TAT is required. However, questions remain about suitability for IHC and molecular tests, and acid-based decalcification is not recommended unless steps are taken to ensure that processing is rigorously monitored and material is preserved for IHC and molecular tests, such as the aspirate or peripheral blood [Reference Torlakovic, Brynes and Hyjek20]. Improved TAT is achieved by shortening EDTA decalcification using a magnetic stirrer in a microwave or hot plate at 37–40°C [Reference Torlakovic, Brynes and Hyjek20]. Decalcification can be accelerated by using ultrasound energization at lower temperatures (e.g. 18°C), which reduces the decalcification time to approximately 6–12 hours and even as short as 2 hours [Reference Torlakovic, Brynes and Hyjek20, Reference Reineke, Jenni and Abdou23]. Novel decalcification solutions used at 37°C that resulted in superior nucleic acid preservation have been described and deserve further evaluation [Reference Anagnostopoulos, Lenze, Hummel, Dietel and Joehrens24].
Whatever method is chosen, IHC results must be validated for both descriptive and quantitative tests according to international guidelines [Reference Torlakovic, Brynes and Hyjek20]. Methodology should take into account that decalcified BM biopsies may require a modified protocol in comparison with non-decalcified tissues. Bone marrow IHC should be validated by an experienced haematopathologist who is aware of the full range of IHC staining in haematological tissues, e.g. the difference in intensity of staining in CD117 staining for proerythroblasts versus mast cells, adequate staining of both being required for validation (Figure 1.1) [Reference Torlakovic, Brynes and Hyjek20]. External quality assurance is necessary for validation but many general schemes do not include decalcified BM biopsies and this is an issue that requires further action [Reference Torlakovic, Brynes and Hyjek20].
Similar considerations apply to the use of BM biopsies for fluorescence in situ hybridization (FISH) and polymerase chain reaction (PCR)-based molecular techniques for examination of DNA and RNA, and the technical aspects have been comprehensively reviewed [Reference Fend, Tzankov and Bink25, Reference Neat, Moonim, Dunn, Geoghegan and Foot26].
Embedding and Sectioning
Historically, BM biopsies have been embedded in either paraffin or in plastic. Both methods have certain advantages and disadvantages and are suitable for IHC and molecular tests [Reference Torlakovic, Brynes and Hyjek20, Reference Krenacs, Bagdi, Stelkovics, Bereczki and Krenacs27]. However, excellent morphology may be obtained from carefully processed and sectioned paraffin-embedded sections and most laboratories no longer possess the expertise for plastic processing and sectioning (Figures 1.2–1.4). Modern fully automated systems that perform microwave-enhanced fixation, decalcification and paraffin impregnation in a closed system (LOGOS J, Milestone, Bergamo, Italy) are available and enable overnight protocols, improving TAT to sign-out within two days [Reference De Laak–de Vries, Siebers and Burgers28].
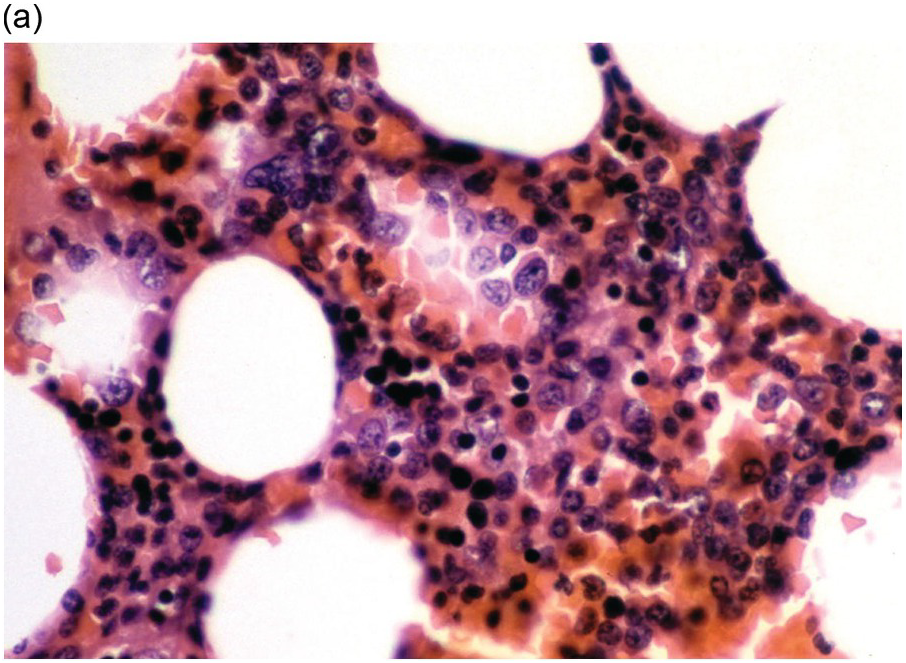
(a) An example of a poorly prepared bone marrow core biopsy. The specimen is too thick and not well stained, preventing adequate identification of different cell types and their stages of maturation.
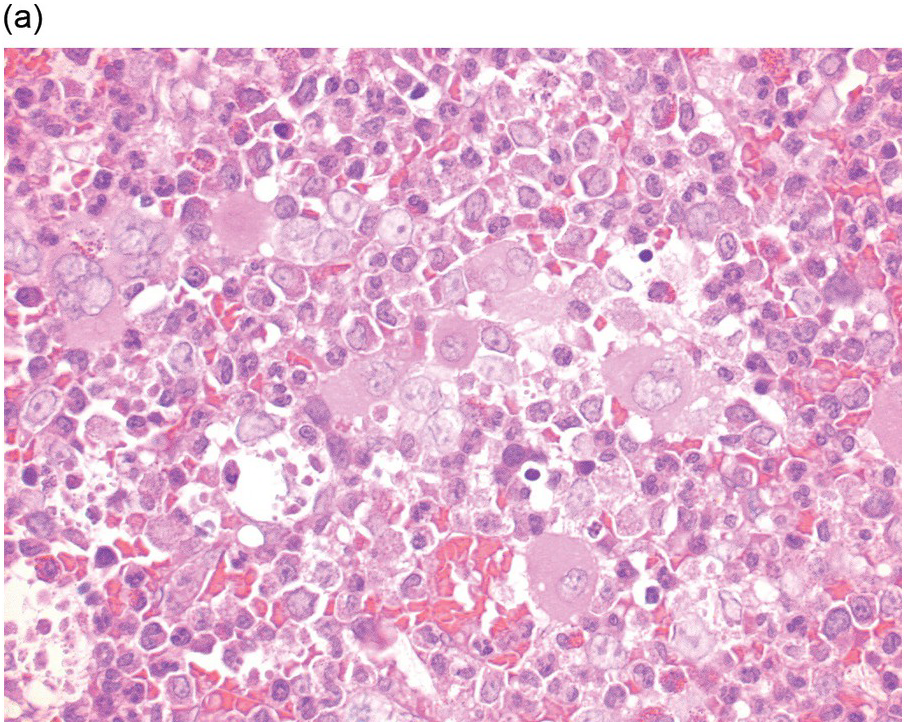

(b) CD61 highlights the dwarf megakaryocytes seen in (a). Good-quality sections are needed for interpretation of IHC.
It is essential to formulate a protocol for the sections to cut that avoids multiple recuts and loss of tissue from the block. The first batch of sections should therefore include the shallow levels and sufficient haematoxylin and eosin and/or Giemsa-stained sections, routine tinctorial stains such as reticulin and Perls’ stains, a Ziehl–Neelsen stain in all immunodeficiency cases, a Congo red in plasma cell dyscrasias and other cause of amyloidosis, and other stains for organisms when required (see Chapter 6), plus enough unstained sections (USS) for the expected IHC panel [Reference Ramsay, Pomplun and Wilkins8]. Preselection of tinctorial stains and USS can have important advantages in TAT and can be protocol driven following consensus between haematopathology team members. The use of IHC panels for defined conditions, selected according to the proposed diagnosis, is well suited to automated IHC staining and can improve consistency within teams and compliance with guidelines, as well as improving TAT.
An Approach to Bone Marrow Evaluation of Neoplastic and Proliferative Processes
It is often easiest to evaluate a BM specimen by comparing it to what would be expected in the normal BM [Reference Brown and Gatter29, Reference Bain, Clark and Wilkins30]. The initial evaluation on low magnification includes the assessment of sample adequacy and BM cellularity. The latter is usually based on the biopsy. Estimates of cellularity on aspirate material have been described [Reference Fong, Okafor, Schmitz, Thomas and Westerman31] but may be unreliable in variably cellular BMs [Reference Gruppo, Lampkin and Granger32]. The normal cellularity varies with age and evaluation of cellularity must always be made in the context of the patient’s age. The BM is approximately 100% cellular during the first three months of life; 80% cellular in children up to age 10 years; it then slowly declines in cellularity until age 30 years, when it remains about 50% cellular. The usually accepted range of cellularity in normal adults is 40–70% [Reference Bain, Clark and Wilkins30, Reference Hartsock, Smith and Petty33–Reference Friebert, Shepardson, Shurin, Rosenthal and Rosenthal35]. The BM cellularity declines again in elderly patients to about 30% at 70 years. Because of the variation in cellularity by age, the report should clearly indicate whether the stated cellularity in a given specimen is normocellular, hypocellular or hypercellular and to what degree (Figure 1.5).
Subcortical BM is normally hypocellular and the first three subcortical trabecular spaces are usually ignored in the cellularity estimate. Superficial core biopsies may contain only these subcortical areas and such biopsy specimens should be considered inadequate for purposes of cellularity evaluation, as well as for the assessment of focal BM infiltrates (Figure 1.6). Avulsion of the BM during biopsy, and technical artefacts such as tearing of the BM during section cutting, may lead to false diagnoses of hypocellularity. Likewise, crush artefact may give a false impression of hypercellularity (Chapter 3).
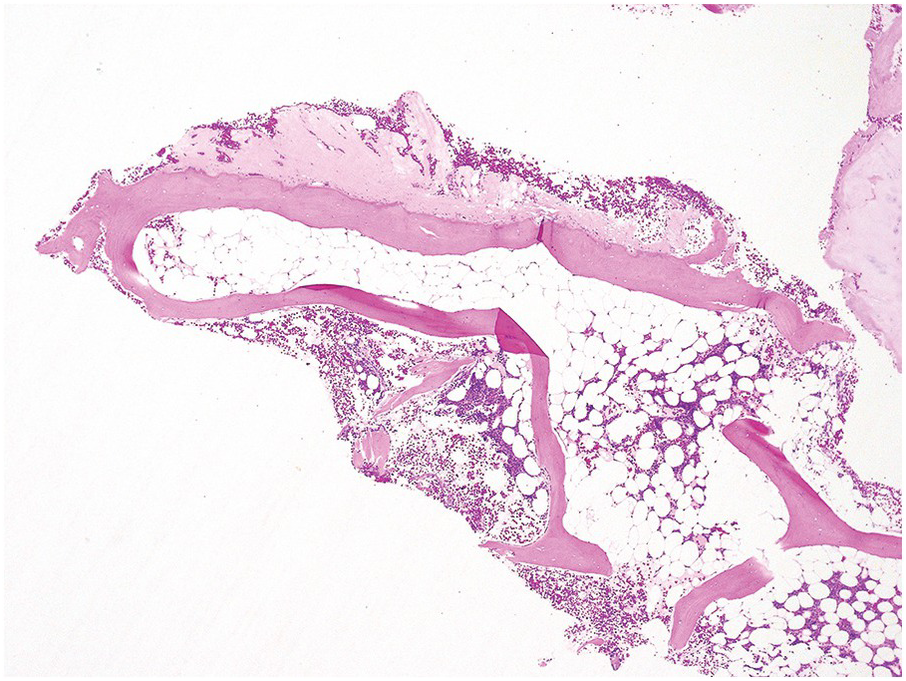
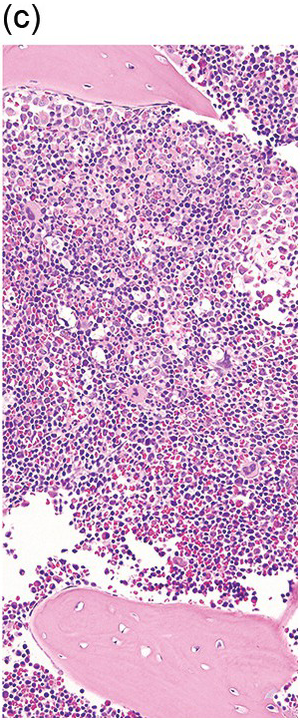
Figure 1.6 An example of a core biopsy taken in a subcortical location. Note the periosteum (upper portion of the photograph), suggesting that this is the outer cortical layer of the bone. Beneath this outer cortex of bone there is an area of hypocellular bone marrow, which includes the first three subcortical trabecular spaces. Deeper in the specimen (lower right corner), the cellularity is considered to be more representative.
After the BM cellularity has been evaluated the cellular elements must be considered (Chapter 2). The three main BM cell lineages, erythroid, granulocytic (myeloid) and megakaryocytic, should be evaluated first and quantified in relative and absolute terms. The various stages of cell maturation are best evaluated from the aspirate smear material but the distribution pattern of different cell lineages is best evaluated on the clot or core biopsy specimen [Reference Bain, Clark and Wilkins30]. Lymphocytes normally represent 10 to 15% of cells on aspirate smears. Lymphoid precursor cells or haematogones are present at all ages but decrease with increasing age [Reference Sevilla, Colovai, Emmons, Bhagat and Alobeid36, Reference Chantepie, Cornet, Salaün and Reman37]. An increased number of haematogones may be seen in autoimmune disorders (idiopathic thrombocytopaenic purpura and autoimmune or other blood cytopaenias), congenital BM disorders, AIDS, following CMV infection and in neoplastic diseases, such as lymphomas and in BM regenerative states following chemotherapy or BM transplantation [Reference Chantepie, Cornet, Salaün and Reman37]. Haematogones may be recognized in the BM biopsy by TdT and CD10 staining but these markers are only present in a subset of haematogones. Chantepie et al. have comprehensively reviewed the topic and haematogone recognition by flow cytometry [Reference Chantepie, Cornet, Salaün and Reman37]. The differential diagnosis is covered in the relevant chapters.
Mature lymphocytes may be normally increased in the elderly. Assessment of the lymphoid population should include IHC where relevant, particularly when the diagnosis of lymphoid neoplasia is in consideration (Figure 1.7) Lymphoid aggregates are common in biopsy material of elderly patients and are non-paratrabecular in location (Figure 1.8). The aggregates are more commonly predominantly composed of T-lymphocytes and germinal centres are rarely present. Cells that are present at a lower frequency in the BM include monocytes, macrophages, plasma cells, mast cells, eosinophils, basophils and osteoblasts. These cells normally represent less than 5% of BM cells on smears and the diagnosis of reactive and neoplastic disorders is discussed in the relevant chapters. They can be identified by IHC (Figure 1.9).
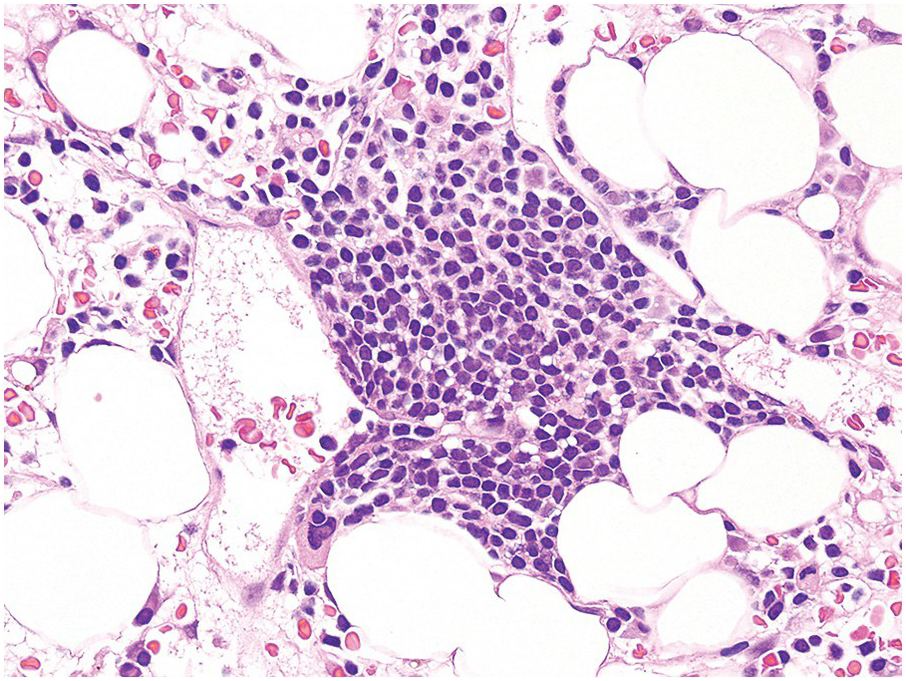
Figure 1.8 A benign lymphoid aggregate within a bone marrow biopsy. This lymphoid aggregate, which is composed predominantly of small lymphocytes, most likely represents a reactive lymphoid follicle. Note that the aggregate is in a perivascular location, which is most often associated with benign lymphoid aggregates.
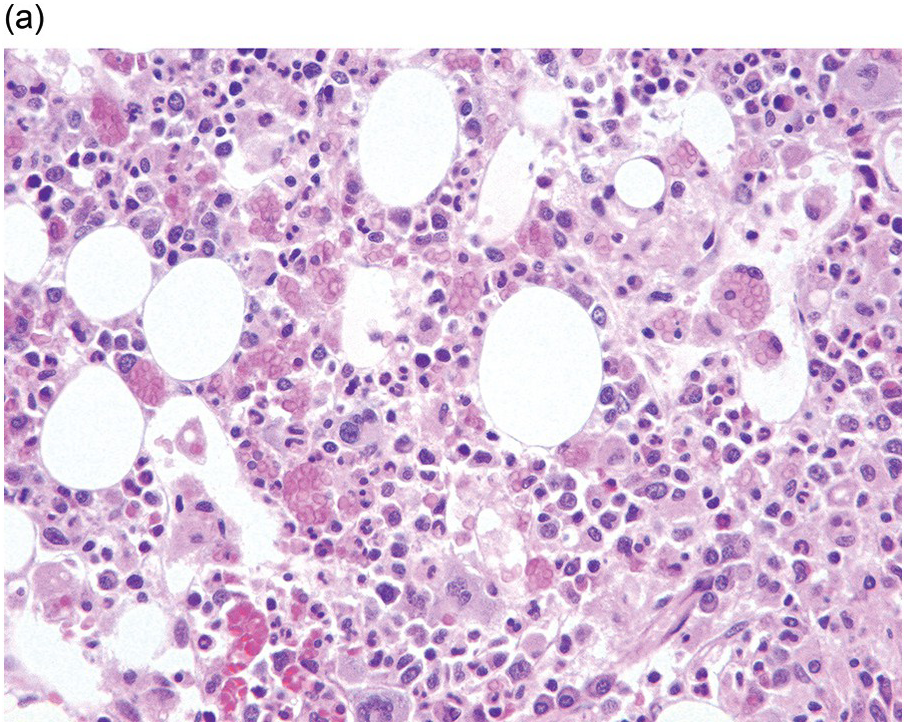
(a) Haemophagocytosis in a case of cutaneous T-cell lymphoma.

(b) Numerous macrophages distended by red cells are shown by CD68 IHC.
The bone trabeculae should also be evaluated for evidence of osteopaenia, osteoblastic proliferations and changes of Paget’s disease [Reference Malcolm38]. Bone changes in renal failure [Reference Carvalho, Alves and Frazão39], which may be seen in plasma cell myeloma and biopsies for the assessment of anaemia in renal failure, should be noted. A detailed discussion of bone pathology is beyond the scope of this book, which is primarily devoted to BM interpretation, but further discussion is found in Chapter 3.
Evaluation of Stainable Iron and Bone Marrow Stroma
In a normal BM aspirate smear stained by Perls’ Prussian blue, iron is predominantly found in histiocytes embedded inside BM particles (Figure 1.10). Iron incorporation in erythroid cells is also normally seen in scattered erythroblasts that usually demonstrate one or two siderotic granules adjacent to the nucleus (sideroblasts). A minimum of seven particles must be examined to establish the absence of stainable iron and a minimum of nine particles must be reviewed to see the maximum iron stores in 100% of samples and therefore make a valid judgement of whether iron stores are reduced, normal or increased [Reference Hughes, Stuart-Smith and Bain40]. Iron stores must be graded (see Chapter 2) and the presence of ring sideroblasts recorded [Reference Gale, Torrance and Bothwell43].
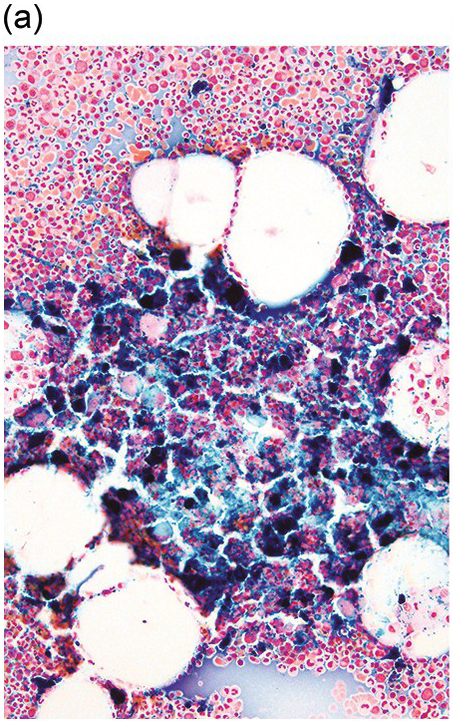
(a) An iron stain on an aspirate smear shows a markedly increased amount of stainable iron.
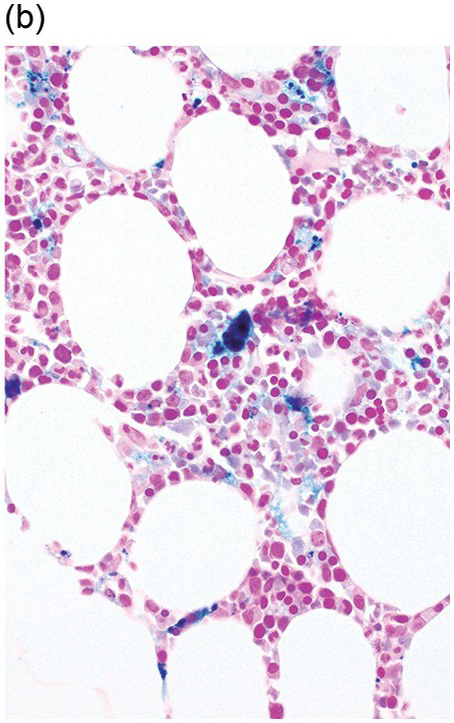
(b) Iron staining on a bone marrow core biopsy, which typically under-represents the amount of iron present. This is due to the iron solubilization effect that may occur during histologic tissue processing. The loss can be partially prevented by using non-acid decalcification methods.
The International Working Group on Morphology of Myelodysplastic Syndrome (IWGM-MDS) recommended that ring sideroblasts be defined as having at least five granules in a perinuclear distribution; that these granules could either surround the entire nucleus, be localized to portions of the perinuclear area or cover at least one third of the nucleus (Figure 1.11) [Reference Mufti, Bennett and Goasguen41]. Ring sideroblasts are found exclusively in pathological BMs and should not be confused with ferritin sideroblasts, which are present in normal BM. These are normal erythroblasts that, after Perls’ staining, show a few blue granules scattered in the cytoplasm, representing endosomes filled with excess iron not utilized for haeme synthesis (siderosomes) in contrast to the iron-laden mitochondria found in ring sideroblasts [Reference Cazzola and Invernizzi42]. Their detection is important in the diagnosis of hereditary and acquired sideroblastic anaemias, including the recently defined entity refractory anaemia with ring sideroblasts and thrombocytosis.
Bone marrow core biopsy decalcification removes iron, depending on the method used. Decalcified sections therefore underestimate iron stores and may give the misleading impression of iron deficiency. Caution must therefore be exercised when grading low quantities of iron in decalcified BMBs. Iron stores are more accurately reflected in undecalcified plastic-embedded sections and in clot section preparations, if adequate BM particles are present. Haemosiderin may also be visible in H&E-stained sections of BM as coarse golden-brown granules in macrophages.
The extracellular matrix is often neglected, especially in normal BMs, but it forms an important component of many BM diseases both in diagnosis and prognosis and must be included in the BM report. The topic is dealt with in detail in Chapters 3 and 10.
Immunohistochemical and Molecular Techniques Useful in Bone Marrow Evaluation
The assessment of the abnormal BM biopsy is now dependent on IHC in most cases and increasingly molecular investigations are essential, especially when adequate material is not available from the peripheral blood or BM aspirates or, in the case of lymphoid neoplasia, from an original lymph node biopsy. Processing of the specimen in a way that optimizes these tests has been dealt with here and the diagnostic applications are emphasized throughout the book, while details of antibodies and cytogenetic abnormalities are found in Chapters 19 and 20.
The Bone Marrow Report
Table 1.3 lists the suggested elements to include in the BM report, which should fulfil ICSH guidelines [Reference Lee, Erber, Porwit, Tomonaga and Peterson10]. Many elements of the report, as well as which tests are performed, are determined by the initial morphologic findings and clinical situation; the clinical indication for the BM examination should therefore be included. Available complete blood cell count data should also be included as well as review of the peripheral blood smear. Adequacy or inadequacy of the sample should also be commented upon and the distinction between a negative biopsy and an inadequate biopsy clarified, such as may be an issue for lymphoid infiltrates and other focal lesions.
Table 1.3 Elements to include in a bone marrow report.
| Clinical indication for bone marrow study |
| Peripheral blood examination |
| Complete blood count data including reticulocyte count |
| Red blood cell changes |
| Red blood cell number and size |
| Hypochromasia or increased polychromasia if present |
| Description of anisocytosis and poikilocytosis and description of granules or inclusions, if present |
| White blood cell changes |
| White blood cell number and description of cell types with relative hyperplasias, cytopaenias, or left shift |
| Description of dysplastic or toxic changes |
| Presence or absence of blast cells, atypical or neoplastic lymphoid cells, or other abnormal cell populations |
| Platelet changes |
| Platelet number and morphology, including granulation and size |
| Bone marrow aspirate smear or imprint |
| Adequacy and relative cellularity (hypo, moderate, hypercellular) |
| Myeloid-to-erythroid ratio |
| Red blood cell precursors |
| Relative percentage and normal maturation versus left shift |
| Dysplastic changes |
| Granulocyte precursors |
| Relative percentage and normal maturation versus left shift |
| Dysplastic changes |
| Percentage of blasts and description of blasts if evaluated |
| Megakaryocytes |
| Relative number |
| Description of morphology, including size and nuclear features |
| Description of other cell types, including evaluations of lymphocytes, plasma cells, monocytes, mast cells, eosinophils, and basophils |
| Differential cell count (typically 300 or 500 cell), if indicated |
| Bone marrow trephine or clot biopsy |
| Adequacy and marrow cellularity, including percentage and comparison to normal cellularity for age |
| Proportions of myeloid to erythroid cells and relative number of megakaryocytes |
| Features of bone trabeculae |
| Degree of fibrosis, if present |
| Abnormal cellular infiltrates, including granulomas, lymphoid cell infiltrates, plasma cells, and metastatic tumour |
| Location and pattern (particularly neoplastic lymphoid infiltrates) |
| Cell type (lymphoma cell type or differentiation of plasma cell infiltrate) |
| Percentage of marrow involvement |
| Cytochemistry results, if indicated |
| Iron stain results for reticuloendothelial stores and red blood cell incorporation, including presence and percentage of ring sideroblasts |
| Cytochemical studies for acute leukemia or hairy cell leukemia, including stains performed and results |
| Immunophenotyping results, if indicated |
| Method used (e.g., flow cytometry, immunocytochemistry, IHC) |
| Antibodies and antigens studied |
| Cell population studies (e.g., blast cells, lymphoid cells, plasma cells) |
| Specimen type studies (peripheral blood, bone marrow aspirate, bone marrow biopsy) |
| Results for each antibody and antigen and interpretation of total findings |
| Molecular genetic and cytogenetic studies, if indicated |
| Method used (e.g., karyotype analysis, polymerase chain reaction, fluorescence in situ hybridization, etc) |
| Test performed (e.g., BCR/ABL1, RT-PCR, or FISH, immunoglobulin heavy chain gene rearrangement PCR) |
| Interpretation of results |
| Diagnosis |
| Tissue examined |
| Diagnostic interpretation, incorporating all results |
| Classification system used clearly indicated in the diagnosis (if in question) |
The BM aspirate and biopsy examinations describe the three main cell lineages of the BM and, as appropriate, other cellular elements. In the case of infiltrative lesions in the BM, the cell type and percentage of involvement are included. Results of all ancillary studies should be included in the final report and correlated with the final diagnosis. This may require amending a preliminary or interim report, which must be clearly identified as such, when ancillary study results that are not available at the time of the initial report, such as molecular genetic or cytogenetic results, become available. Ideally the synoptic report should only be signed off after discussion with all the clinical and laboratory teams involved [Reference Sever, Abbott and Baca6, Reference Ramsay, Pomplun and Wilkins8]. This multistep approach allows for a final ‘comprehensive’ diagnostic interpretation that is based on all available data and addresses all findings, including seemingly contradictory data that may be easily explained when all of the information is considered together.