Introduction
Ceramics are widespread and abundant in the Holocene archaeological record. They can inform our understanding of past cultural, technological and culinary practices, and provide the typological framework for temporal and cultural phasing within a site or region. Traditionally, ceramics have been radiocarbon dated by association with food residues, plant remains or bone, with the quality of these chronologies determined by the strength of the association between sherd and dated sample. Direct dating of pottery has been attempted via organic temper but, with a few exceptions (Tóth et al. Reference Tóth, Petřík, Bickle, Adameková, Denis, Slavíček, Petr, Pokutta and Isaksson2023), dates produced are unreliable as the source of carbon is uncertain or bulk various sources (Hedges et al. Reference Hedges, Tiemei and Housley1992). Compound-specific approaches using gas chromatography (GC) (Eglinton et al. Reference Eglinton, Aluwihare, Bauer, Druffel and McNichol1996) applied to lipid residues absorbed by pottery walls during the processing/storage of plant/animal foods offer a viable alternative for radiocarbon dating (Casanova et al. Reference Casanova, Knowles, Williams, Crump and Evershed2018, Reference Casanova, Knowles, Bayliss, Dunne, Barański, Denaire, Lefranc, Di Lernia, Roffet-Salque and Smyth2020; Smyth et al. Reference Smyth, Berstan, Casanova, McCormick, Mulhall, Sikora, Synnott and Evershed2019; Stott et al. Reference Stott, Berstan, Evershed, Hedges, Ramsey and Humm2001). So far, this has mainly focused on saturated palmitic (C16:0) and stearic (C18:0) fatty acids (FAs), generally abundant in food residues, which result from the hydrolysis of triglycerides (fats and oils) (Casanova et al. Reference Casanova, Knowles, Bayliss, Dunne, Barański, Denaire, Lefranc, Di Lernia, Roffet-Salque and Smyth2020). However, these methods face noteworthy challenges. Mainly, the methodology is necessarily time-consuming and, thereby, low-throughput, and some potsherds simply do not contain sufficient lipids for analysis (Casanova et al. Reference Casanova, Knowles, Bayliss and Evershed2024)—lipid distribution and abundance has been shown to differ both between different vessel types and within a single vessel (Charters et al. Reference Charters, Evershed, Goad, Leyden, Blinkhorn and Denham1993). Moreover, contamination from exogenous materials on site, e.g., soil-derived lipids, and post excavation, e.g., solvents or phthalates from storage containers, can impact measurement accuracy, particularly if they elute closely with target compounds. Additionally, maintaining low and consistent backgrounds can be challenging, requiring careful consideration (Stott et al. Reference Stott, Berstan, Evershed, Hedges, Ramsey and Humm2001). The methylation process, for instance, introduces derivative carbon, necessitating post-analysis correction to ensure accurate age determination. Despite these obstacles, compound-specific lipid analysis remains a unique opportunity to directly date human activity by targeting animal and/or plant residues preserved in pottery. This study aims to develop a compound-specific lipid dating protocol at the Oxford Radiocarbon Accelerator Unit (ORAU), including testing methods to remove phthalate contamination from lipids extracted from archaeological pottery.
Materials
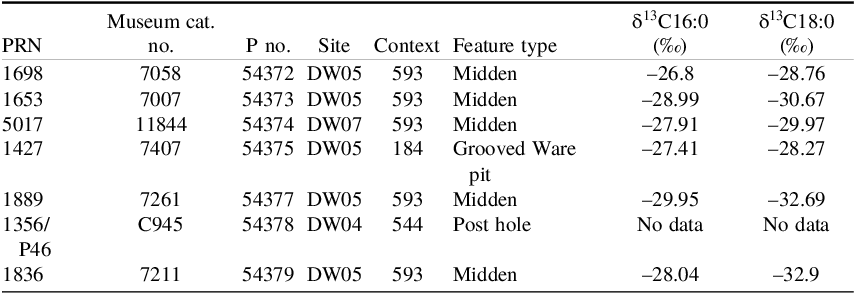
To assess the accuracy of the method, seven chronologically-constrained ceramics from Durrington Walls, Wiltshire, UK (Table 1) were analyzed. They were specifically selected given known, high total lipid yields, having been previously solvent-extracted and tested by a coauthor (OEC) at the University of York. Importantly, the selected samples yielded stable isotope values consistent with ruminant or dairy fats, indicating a terrestrial origin and thereby removing the need for any reservoir correction (Craig et al. Reference Craig, Shillito, Albarella, Viner-Daniels, Chan, Cleal, Ixer, Jay, Marshall, Simmons, Wright and Parker Pearson2015). The stable isotope analyses were previously performed on different sherds from the same vessels that were used for radiocarbon dating in this study.
Grooved Ware sherds from Durrington Walls sampled for radiocarbon dating. PRN = Pottery Reference Number. P no. = laboratory pretreatment number assigned at the ORAU. Stable isotope values (δ13C) of C16:0 and C18:0 fatty acids are published data from Craig et al. (Reference Craig, Shillito, Albarella, Viner-Daniels, Chan, Cleal, Ixer, Jay, Marshall, Simmons, Wright and Parker Pearson2015)

Durrington Walls is a late Neolithic henge monument located approximately 3km northeast of Stonehenge, Wiltshire, UK. Excavations from 2004-2007 revealed evidence of a settlement predating the henge, which included surviving house floors and hearths (Parker Pearson et al. Reference Parker Pearson, Cleal, Marshall, Needham, Pollard, Richards, Ruggles, Sheridan, Thomas, Tilley, Welham, Chamberlain, Chenery, Evans, Knüsel, Linford, Martin, Montgomery, Payne and Richards2007). Given the substantial quantities of feasting-related debris such as animal remains and Grooved Ware sherds excavated from associated middens and pits, it has been suggested that the settlement may have been inhabited by the builders of Stonehenge, given its proximity and contemporaneity with Stage 2 of the latter’s construction (Craig et al. Reference Craig, Shillito, Albarella, Viner-Daniels, Chan, Cleal, Ixer, Jay, Marshall, Simmons, Wright and Parker Pearson2015; Parker Pearson et al. Reference Parker Pearson, Cleal, Marshall, Needham, Pollard, Richards, Ruggles, Sheridan, Thomas, Tilley, Welham, Chamberlain, Chenery, Evans, Knüsel, Linford, Martin, Montgomery, Payne and Richards2007). Bayesian chronological modelling for the settlement dates the start and end of the occupation at Durrington Walls at 2525–2470 cal BCE and 2480–2440 cal BCE, respectively (Parker Pearson et al. Reference Parker Pearson, Pollard, Richards, Thomas, Welham, Albarella, Chan, Marshall, Viner, Aranda Jiménez, Montón-Subías and Sánchez Romero2011).
Methods
Lipid extraction
Prior to processing, pottery fragments were surface-cleaned using air abrasion with Al2O3 (29 µm powder; OEA Laboratories Ltd) to remove potential contamination from the burial environment (Hendy et al. Reference Hendy, Colonese, Franz, Fernandes, Fischer, Orton, Lucquin, Spindler, Anvari and Stroud2018). Bulk lipid extraction was performed using supercritical fluid extraction (SFE) with a Jasco system, comprising a high-pressure pump (PU-2086), an oven (CO-4065), a UV detector (UV-2077), a valve/event controller (LC-NetII/ADC), a back pressure regulator (BP-2080), and a fraction collector (FC-2088-30). Supercritical CO₂ serves as the main solvent, providing high dissolving power (Devièse et al. Reference Devièse, Van Ham-Meert, Hare, Lundy, Hommel, Ivanovich Bazaliiskii and Orton2018, Reference Devièse, Ribechini, Querci and Higham2019). To enhance extraction efficiency, a co-solvent mixture of 20% hexane [Sigma-Aldrich, ≥99% (GC), ACS reagent] and 80% ethanol [LiChrosolv, gradient grade for liquid chromatography] was used. The system operates at a flow rate of 2 mL/min, maintaining an 85:15 ratio between CO₂ and the co-solvent. The solvents are pressurized to 30 MPa, with the oven temperature set at 50 °C. Each sample underwent a 90-min extraction, followed by a 30-min line clean to prevent cross-contamination. Two blank extractions were performed between every five samples of unknown age to assess contamination. After extraction, samples were dried nearly to completion using a Genevac EZ2 at 45 °C before transfer to a 2 mL glass vial using a glass pipette and evaporated to dryness under N₂ flow at 35 °C.
Methylation
To enhance GC amenability of the target free fatty acids, prior to GC analysis, total lipid extracts were methylated (Sigma-Aldrich, 14% BF3 in methanol; 0.5 mL, 60 °C, 10 min). The reaction was quenched with ultrapure water (0.5 mL; Millipore Milli-Q), and the product fatty acid methyl esters (FAMEs) extracted into dichloromethane (DCM; Sigma-Aldrich, ACS reagent, ISO, ≥99.9% (GC); 3 × 0.5 mL). Finally, the combined extracts were dried over Na₂SO4 (Sigma-Aldrich, anhydrous for analysis EMSURE® ACS). The dried extracts were transferred to a clean vial and evaporated to dryness under N₂ at room temperature for storage prior to analysis.
Sample screening
GC/mass spectrometry (GC/MS) was used to identify compounds in the methylated lipid extracts and provide an indication of lipid concentration to identify suitable samples for radiocarbon dating. A GC (Agilent 8890)-mass spectrometer (MS; Agilent 5977B) system, with a cooled on-column inlet, was used to approximate the yield of methyl palmitate and stearate for each sample relative to a FAME standard [methyl palmitate (> 99%); methyl stearate (> 98.5%); both Sigma-Aldrich] calibration curve, with the aim of identifying samples that contained >100 µg C (either as individual FAMEs or by combining methyl palmitate and stearate). The GC was fitted with a Rxi-65TG GC capillary column (30 m × 0.25 mm internal diameter; 0.1 µm film thickness; Restek; stationary phase: Crossbond™ 65% diphenyl/35% dimethyl polysiloxane). The helium carrier gas flow rate was set to 1.2 mL/min. The temperature program consisted of an initial 2 min hold at 50 °C; then 40 °C/min to 160 °C (held for 1 min), followed by 10 °C/min to 220 °C (held for 0.5 min), and finally 40 °C/min to 280 °C (held 6.25 min; total run time was 20 min).
Collection
FAMEs were isolated using a GC (Agilent 7820A) equipped with a flame ionisation detector (FID; Agilent 7280). Samples were injected approximately 40 times, with each injection volume measuring 4.5 µL. Target fractions were collected using a Gerstel Preparative Fraction Collector (PFC) in conjunction with a Cooled Injection System (Gerstel CIS 4), with both systems operated under the control of the Gerstel C506 control unit. The GC was fitted with a Rxi-1ms GC capillary column (30 m × 0.53 mm internal diameter; 1.5 µm film thickness; Restek; stationary phase: Crossbond™ 100% dimethyl polysiloxane). The flow from the GC column was directed to a splitter plate, where it was split into two separate flow paths: a 0.1 mm inner diameter deactivated column with a length of ca. 20 cm was used for the FID, whilst a 0.32 mm inner diameter deactivated column with a length of ca. 1 m facilitated sample collection in the PFC. The injector was used in splitless mode with an initial temperature of 60 °C (held for 0.1 min) and then ramped at 12 °C/s to 300 °C. The helium carrier gas flow rate was set to 5 mL/min. The temperature program consisted of an initial 0.5 min hold at 70 °C, then 50 °C/min to 220 °C (held for 1.75 min), followed by 40 °C/min to 280 °C (held for 2 min), and finally 15 °C/min to 310 °C (held 6.25 min; total run time was 17 min). The optimisation involved adjusting column settings to reduce FID consumption and minimize the retention time offset, followed by using 0.6 µg/µL FAMEs to define the trapping windows. These settings directed over 90% of C16:0 and C18:0 FAMEs to the PFC, with an offset of approximately 2 s between FID detection and PFC elution.
C16:0 and C18:0 FAMEs from archaeological samples were trapped within a 23 s window, which was experimentally established by injecting C16:0 and C18:0 FAME standards (0.6 µg/µL each in hexane; 4.5 µL) 40 times (total 180 µL), theoretically yielding 164 µg C. Actual yields were in excess of 125 µg C, corresponding to a collection efficiency close to 80%. Each eluting target compound from each ∼40 injection series per sample was condensed into the same glass tube packed with 0.3 - 0.4 mg silica wool (EMA, prebaked at 550 °C for 3 hr), after Casanova et al. (Reference Casanova, Knowles, Williams, Crump and Evershed2018). Following this method, a heat gun was applied for up to 5 s to evaporate residual condensed compounds from the end of the transfer capillaries connecting the switching valve to the traps.
The post-cleaning process of the GC-PFC is critical to minimize cross-contamination. Specifically, injection syringes were cleaned between samples by performing 10 sequential rinses of DCM, followed by 10 sequential rinses of ethyl acetate (HPLC Plus 99.9%, Sigma-Aldrich). Wash bottles were cleaned using ultrasonic baths, sequentially applying Milli-Q water, methanol, and hexane for 15 min each. Furthermore, the GC column, which exhibited carryover, was thoroughly conditioned between samples by running hexane solvent blanks until the FID signal indicated no detectable target-compound carryover, ensuring clean and reliable conditions for subsequent sample injections.
Radiocarbon dating
The silica wool and adsorbed FAME were combusted in a tin (Sn) capsule using a CHN elemental analyzer (Carlo-Erba NA 2000) coupled to a gas-source isotope ratio MS (Sercon 20/20).The resulting CO₂ was cryogenically collected and graphitized in the presence of hydrogen over an iron catalyst (Dee and Bronk Ramsey Reference Dee and Bronk Ramsey2000) for radiocarbon measurement using a MICADAS accelerator mass spectrometer (Ionplus AG). The carbon yield for radiocarbon dating was determined by measuring the volume of CO2 produced by the combustion of each sample using the elemental analyzer (Table S3). Radiocarbon dates were calculated following Stuiver and Polach (Reference Stuiver and Polach1977) and corrected for an AMS-derived δ13C. To monitor and control for contamination, blanks underwent the same lipid extraction, methylation, and collection processes as the samples. After collection, blanks were spiked with phthalic acid (F14C = 0) or IAEA-C7 [F14C = 0.4953 ± 0.0012; (Le Clercq et al. Reference Le Clercq, van der Plicht and Gröning1997)] to assess accuracy. Details of the background correction using the phthalic acid samples are provided in the SI.
Plasticizer contamination
Where present, dibutyl benzene-1,2-dicarboxylate (dibutyl phthalate; DBP), a plasticizer (Net et al. Reference Net, Sempéré, Delmont, Paluselli and Ouddane2015; Wormuth et al. Reference Wormuth, Scheringer, Vollenweider and Hungerbühler2006), eluted shortly after C16:0 under GC-PFC operating conditions (Figure 1). The 23-second trapping window covers the onset of DBP elution, resulting in the collection of both C16:0 FAME and DBP. Here, we evaluated two methods for its removal using the methyl palmitate and stearate standards noted in the “Sample screening” section, as well as a DBP standard (Sigma-Aldrich, 99%) for spiking. The first approach involved shortening the C16:0 FAME trapping window to 10s, timed to terminate just before the DBP peak enters the PFC. The second approach used argentation column chromatography (e.g., Morris Reference Morris1966) to separate FAMEs and DBP. Glass columns, packed with 0.75 g AgNO3/silica gel (10% w/w), were pre-rinsed with hexane until saturated before loading each sample onto the top of the column in 230 µL of hexane. 3.3 column volumes (5 mL) of 97:3 (v/v) hexane:ethyl acetate were used to elute saturated FAMEs.
FID chromatogram of organic residues extracted from sample 54379.4, showing the presence of various fatty acids alongside potential contaminants, including dibutyl benzene-1,2-dicarboxylate (DBP). Relevant compounds are noted above each peak.

Chronological testing
Chronological testing was undertaken with OxCal v.4.4 (Ramsey Reference Ramsey2009). This involved (i) entering the estimated start and end boundaries for the Durrington Walls occupation presented in Parker Pearson et al. (Reference Parker Pearson, Pollard, Richards, Thomas, Welham, Albarella, Chan, Marshall, Viner, Aranda Jiménez, Montón-Subías and Sánchez Romero2011) as normal distributions—N(-2050, 30) and N(-2060, 20), respectively—to produce an age range using a Date function, and (ii) employing the Difference command to statistically compare this distribution to the measurements produced on the archaeological pottery. All age estimates are given at 95.4% confidence intervals (CI) and rounded to five years. The OxCal code can be found in the SI.
Results and discussion
Standards
Radiocarbon measurements for IAEA-C7 and phthalic acid samples can be found in Table S1. For the IAEA-C7 data (n = 10), one outlier (54146.0NRC10) is older than expected (Figure 2), indicating fossil carbon contamination equivalent to 8.22 μg C—most likely introduced from a 14C-depleted source other than column bleed (Casanova et al. Reference Casanova, Knowles, Bayliss and Evershed2024) and DBP (see SI for blanks FID chromatograms). Excluding this outlier, the weighted average F14C is 0.49165 ± 0.00277, which falls within the 1σ range of the standard value (0.4953 ± 0.0012), demonstrating consistency with expected results. For the phthalic acid data (n = 9), all but one sample (51645.0XB93), which shows modern carbon contamination equivalent to 2.92 μg C, yield values at or near zero (Figure 3). Separately, twenty untreated phthalic acid samples (Table S1) show modern carbon contamination equivalent to 0.43 ± 0.076 μg C (Figure S1). Taken together, the IAEA-C7 and phthalic acid dataset suggests that contamination introduced during laboratory processing is mixed in origin, involving both modern and fossil carbon. A larger sample size would allow for a more robust assessment.
Measured F14C value for blank samples spiked with IAEA-C7. The dotted lines represent the consensus value for IAEA-C7 (F14C = 0.4953) at 1 (0.0012) and 2σ (0.0024).

Measured F14C value for blank samples spiked with phthalic acid (F14C = 0).

Archaeological samples
Screening results for Durrington Wall samples are summarized in Table S2, with corresponding chromatograms in Figures S2–4. Sample 54378, as listed in Table 1, failed the screening process, resulting in insufficient carbon yield for radiocarbon dating (>100 μg C for graphite).
Radiocarbon measurements are shown in Table S3 and Figure 4. When no attempt was made to remove the dibutyl phthalate (DBP), ages were generally significantly older than expected. GC-MS showed that levels of DBP varied considerably and those containing the most DBP produced the oldest ages. Notably, 54379.4 contained the highest proportion of DBP (Figure 1) and gave an age millennia older than anticipated. Vessels 54373 and 54374 were each dated once—the limited remaining sample material precluded additional attempts using either DBP removal methods. Results indicate that DBP is made from a 14C-depleted source, likely attributable to the degradation of plastic sample bags. DBP was not detected in the blanks (see SI for FID chromatograms), excluding the possibility of introduction during laboratory processing.
Radiocarbon dates for the Durrington Walls vessels using different methods. The distribution in dark grey at the top (“DW Date”) represents the age of the Durrington Walls occupation (see Methods and SI for OxCal code). Measurements are split according to vessel, with each age obtained from a different sherd. Blue: dates with varying concentrations of DBP. Red: dates after adjusting the trapping window for C16:0 for DBP removal. Green: dates following treatment with argentation chromatography for DBP removal. Bars underneath each distribution denote the 95.4% confidence interval.

To address DBP contamination, two removal strategies were tested and applied as described in the “Methods” section: (i) shortening the trapping window and (ii) argentation chromatography. The effectiveness of window shortening was first demonstrated using FAME and DBP standards (Figure S5), and similar improvements were observed in the archaeological samples—ages for DBP-contaminated specimens generally became younger, indicating significant removal of the contaminant. A second sherd from vessel 54377, which had previously shown insignificant DBP contamination (54377.1; Figure S2), showed higher DBP yield (Figure S3) and a marginally older age following window shortening (54377.3), indicative of uneven DBP distribution within the vessel and unsuccessful separation. Results for argentation chromatography showed that over 95% of DBP was effectively removed (Figures 5 and 6). Figure 5 shows the results using FAME and DBP standards, whilst Figure 6 illustrates the same for sample 54379 (which initially exhibited the highest DBP levels) as based on the GC peak area ratio of DBP to C18:0. A total yield loss across all compounds using this method was observed (<10%), however. Overall, except for vessel 54372—where window shortening yielded the youngest age estimate—ages on archaeological samples following argentation chromatography were younger than both the initial measurements and those obtained using a shortened trapping window. Relative to the Durrington Walls occupation—dated to 2535-2435 cal BCE (see Methods and SI for Oxcal code)—statistical analysis indicates that all window-shortened ages are statistically incomparable, whereas three of the four argentation chromatography measurements (vessel 54372 excluded) are statistically consistent (Figure S6). Thus, despite the small sample size, argentation chromatography achieved an accuracy of 75%.
GC-MS results before and after DBP removal using argentation column chromatography, with 30 ng each of C16:0 and C18:0 FAMEs and 20 ng DBP.

GC-MS chromatograms of sample 54379.2 before and after the application of argentation chromatography to remove dibutyl phthalate (DBP) contamination. (A) before the removal of DBP; (B) after the removal of DBP; (C) DBP released from argentation chromatography using 100% ethyl acetate. The elution time of DBP differs between GC-MS and GC-FID-PFC due to differences in the column’s stationary phase, such that DBP elutes shortly after C16:0 in the GC-FID-PFC method and appears after C18:0 in the GC-MS method.

Conclusion
This study presents a protocol for compound-specific radiocarbon dating of pottery lipids, with a focus on C16:0 and C18:0 fatty acids. The workflow integrates SFE, GC-MS and GC-FID-PFC, employing rigorous quality control measures to minimize contamination. The detection of DBP contamination, likely stemming from plastics, led to the development of two removal methods—shortening the trapping window and argentation chromatography—with the latter proving more effective. As a preventive measure, enclosing samples in aluminium foil prior to storage in plastic bags is recommended to reduce the risk of phthalate transfer. Notably, the refined protocol produced encouraging results for both standards and archaeological samples from the Durrington Walls site, offering significant potential for dating archaeological sites where pottery provides the sole basis for establishing a reliable chronology. Processing times are lengthy, however—five unknown-age samples take approximately two weeks to complete—so the protocol is necessarily low throughput, highly specialized, and costly. Future efforts at the ORAU will focus on maximising lipid recovery, including testing how acidified methanol extraction (Correa-Ascencio and Evershed Reference Correa-Ascencio and Evershed2014; Taché and Craig Reference Taché and Craig2015) compares with SFE—currently shown to achieve greater lipid yields than chloroform-methanol extraction (albeit with a modified solvent and derivatisation set-up; Devièse et al. Reference Devièse, Van Ham-Meert, Hare, Lundy, Hommel, Ivanovich Bazaliiskii and Orton2018)—as well as continuing optimisation of the latter.
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/RDC.2026.10192
Acknowledgments
We thank the John Fell Fund for financial support (grant reference 0011358), funding from the National Environmental Isotope Facility (NE/Y005449/1) and ORAU staff members for their assistance throughout this project. LBV acknowledges the Leverhulme Trust for supporting her research through an Early Career Fellowship (ECF-2022-532), during which time part of this work was undertaken. AI was used to assist in the R coding for Figures 2 and 3.
Author contributions
The manuscript was written by QM, with input from all coauthors. The project was initially conceptualized by LBV and BL. Funding was secured by LBV, BL, DC, CBR, RR, RH and RW. The experimental design was developed by QM, KL, LBV, BL, MB, DC, LB and AVH. Laboratory work was conducted by QM, KL, LBV, BL, and MB. Archaeological samples were provided by RC, BC, OC, TD, LF, AG, TK, ZL, and MPP.