Introduction
Obesity is a multifactorial disease, where interactions of genetics, metabolic, and environmental factors play an important role in its development. Reference Chan and Woo1 Nowadays, obesity has been described as a worldwide pandemic wherein poor diets and a reduction in physical activity have increased its prevalence. Reference Smith and Smith2 When obesity affects women during reproductive age, exposure of the fetus to the obesogenic intrauterine environment could cause changes in its organogenesis and generate permanent physiological responses. Reference Zambrano, Ibáñez, Martínez-Samayoa, Lomas-Soria, Durand-Carbajal and Rodríguez-González3 Previously, we demonstrated that the offspring of obese mothers also develop obesity. Reference De los Santos, Coral-Vázquez and Menjivar4
Several studies have shown that maternal obesity negatively impacts maternal health and offspring’s phenotype, predisposing the fetus to develop future chronic diseases, including obesity, insulin resistance, type 2 diabetes mellitus, metabolic syndrome, hypertension, and disorders in the cardiovascular system, among others. Reference Alfaradhi and Ozanne5–Reference Kotsis, Jordan and Micic8 Besides, it has been demonstrated that maternal obesity during pregnancy increases the risk of cardiovascular diseases in the offspring. Reference De Jong, Barrand and Wood-Bradley9,Reference Loche, Blackmore and Carpenter10
In a particular way, suboptimal exposure, such as overnutrition during pregnancy and lactation, can lead to cardiac hypertrophy (CH) in the offspring. Reference De Jong, Barrand and Wood-Bradley9,Reference Loche, Blackmore and Carpenter10 The CH could be initiated as an adaptive response to various physiological stimuli. Still, it could become pathological if the activation signal persists, leading at last to the remodeling of the extracellular matrix. Reference Heineke and Molkentin11–Reference Tham, Bernardo, Ooi, Weeks and McMullen13 Furthermore, pathological CH is accompanied by fibrosis, damaged contractility, heart failure, apoptosis, ischemia, overexpression of fetal genes, and disorders in myocardium structure, leading to heart dysfunction. Reference Tham, Bernardo, Ooi, Weeks and McMullen13 These effects are mediated by the activation of the pathological pathway via transcription factors GATA4, MEF2, and NFAT3, which induce expression of natriuretic peptides ANP and BNP, β-MHC isoform, ACTA1, and Col1a1—markers of cardiac damage. The upregulation of β-MHC, ANP, BNP, and Col1a1 is linked to both short- and long-term cardiac tissue damage, contributing to cardiovascular disease development. Reference Heineke and Molkentin11,Reference Tham, Bernardo, Ooi, Weeks and McMullen13
On the other hand, (-)-epicatechin (Epi), an abundant flavonoid in cocoa, Reference Taubert, Berkels, Roesen and Klaus14 has beneficial effects on health, lowering blood pressure, Reference Schroeter, Heiss and Balzer15 activating endothelial nitric oxide synthase, Reference Ramirez-Sanchez, Maya, Ceballos and Villarreal16 increasing resistance to fatigue and improving oxidative capacity Reference Nogueira, Ramirez-Sanchez and Perkins17 ; likewise, it promotes angiogenesis, Reference Ramirez-Sanchez, Aguilar, Ceballos and Villarreal18 and improves cardiac function. Reference De Los Santos, García-Pérez and Hernández-Reséndiz19
A previous study conducted by our research group showed that Epi induced the development of physiological CH in healthy mice, as well as an improvement of its cardiac function by activating the PI3K/Akt pathway and mimicking the exercise effects. Reference De Los Santos, García-Pérez and Hernández-Reséndiz19 Furthermore, another study by our group demonstrated that Epi could prevent the development of dilated cardiomyopathy of genetic origin. Reference De Los Santos, Palma-Flores, Zentella-Dehesa, Canto and Coral-Vázquez20
Taking into account the effects of maternal obesity on the offspring and the beneficial cardiac properties of Epi, this study aimed to investigate whether Epi can modify the expression of genes related to pathological CH, as well as the morphometric parameters of the heart in offspring descendant rats of a model of obesity by programing, previously characterized by our research group. Reference De los Santos, Coral-Vázquez and Menjivar4 We hypothesized that the treatment with (-)-epicatechin will decrease the expression of genes associated with pathological CH, as well as the morphometric parameters of the heart in offspring obese by programing in comparison with the offspring without treatment with this flavonoid.
Material and methods
Animal model
Before the start of the experimental animal protocol, it was approved by the Animal Experimentation Ethics Committee of the Salvador Zubirán National Institute of Medical Sciences and Nutrition (INCMNSZ) (CINVA: UIO-1892-17/19-1) and by the Ethics and Research from the National Autonomous University of Mexico.
All animal experiments complied the ARRIVE guidelines and followed the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines; as well as, by the Laboratory Animal Resources Institute’s Guidelines for the Care and Use of Laboratory Animals (http://www.nal.usda.gov/awic/pubs/noawicpubs/careuse.htm) and by the Official Mexican Standard NOM-062-ZOO-1999.
Our research group previously described the Chow diet, the high-fat diet, the female phenotype standarization (F0) used to produce F1 study offspring, and the (-)-epicatechin intervention. Reference De los Santos, Coral-Vázquez and Menjivar4 Briefly, Female albino Wistar rats fed Chow diet, at 14–16 weeks of age (98–112 postnatal day) and weighing 200–240 g were randomly mated with males from other litters. Subsequently, the study was carried out in two stages. The first was carried out at weaning (day 21), where the F0 female pups from each litter were randomly assigned to the control group (C, n = 8, fed with Chow diet) or the maternal obesity group (MO, n = 8) fed a high-fat diet (HFD) according to what was previously reported. Reference De los Santos, Coral-Vázquez and Menjivar4
The eight F0 female rats were placed with proven male breeders on postnatal day 120 and conceived during the next cycle. Nursing mothers were maintained on the pregnancy diet (Supplementary Fig. 1). Besides, we documented the diet intervention of the control and maternal obesity mothers group from 21 postnatal day, in order to demonstrate that mother who received a HFD presented obesity during the pregnancy until the birth and breastfeeding of her offspring. Reference De Los Santos, Reyes-Castro, Coral-Vázquez, Mendez, Zambrano and Canto21
For stage two, the number of individuals per F1 litter and their weight were recorded at birth. The anogenital distance was used for the identification of females and males. All litters studied were adjusted to 10 puppies with equal males and females. Weaning was performed at 21 postnatal days, two rats were housed per cage and fed a Chow diet throughout the study. Only male offspring were studied to avoid potential cardioprotective bias reported in females. Reference Gardner, Brower, Voloshenyuk and Janicki22 Eight male offspring of C or MO mothers from different litters were randomly assigned to the control group (vehicle) or the (-)-epicatechin intervention groups (C + Epi and MO + Epi) (total male F1 rats = 32) (Supplementary Fig. 1).
(-)-Epicatechin intervention
Epi treatment from the 21st to the 110th postnatal day, male offspring were given vehicle (water) or Epi (1 mg/kg body weight) (Sigma-Aldrich, St Louis, MO, U.S.A.) by oral gavage twice daily (morning and afternoon), as previously described, the interventions were carried out at the same time and were carried out by the same people. The animals were always in the same place since the beginning of the experiment. Regarding to the food intake, this was done by measuring feed consumption daily during the intervention period and dividing the feed supplied by the amount of residual feed in each cage after 24 hours. An average of food consumed by a pair of rats housed in each cage was obtained. Reference De los Santos, Coral-Vázquez and Menjivar4
Retrieval organ and histological analysis
On postnatal day 110, the rats were fasted for 6 hours and anesthetized with isoflurane Stoelting Co., Cat. No. 50,207 l) and euthanized. The hearts of animals were obtained and weighed. We calculate the index of heart weight (HW) and body weight (BW) of the offspring (HW/BW ratio), by dividing the heart weight (g) between body weight (g) and analyzing, as previously described. Reference De Jong, Barrand and Wood-Bradley9,Reference Loche, Blackmore and Carpenter10
Heart tissues were stored at -80°C for subsequent analysis of gene expression and protein levels. For the histological analysis of the thickness of the ventricular walls and septal (SP) walls, the hearts were immediately fixed in 10% formalin, and embedded in paraffin blocks. Reference Kershaw, Heblinski, Lotz, Dirsch and Gruber23 Four-micrometer cross-sections were obtained 3 mm below the atrioventricular groove and stained with hematoxylin and eosin (H&E). The thickness of the right (RV) and left ventricle (LV) free walls and SP were measured at different sites: RV1, RV2, RV3, SP1, SP2, SP3, LV1, LV2, and LV3. Reference Kershaw, Heblinski, Lotz, Dirsch and Gruber23 Furthermore, the total area and perimeter of the heart, as well as the cavities, the area and the mass of the left ventricle, were analyzed.
Digital image analysis was performed using a Velab S4 stereoscope binocular with a 10X eyepiece with a magnification of 0.7X equipped with Tcapture Software (Meiji Techno Microscopes, Tucsen ISH1000), yielding images pixels that were transformed to millimeters. In addition, the distances and areas were quantified using the AxioVision software for digital imaging processing (Zeiss® copyright 2006–2010 Stuttgart-Germany).
Analysis of cardiac fibrosis
Paraffin-embedded cardiac cross-sections at 4 μm were stained by Masson’s Trichrome staining (ThermoFisher, UK). The ventricle fibrosis area was calculated dividing the total area of fibrosis by the total tissue area. Reference Chin, Holm, Duignan, Conn, Benjamin, Finch, Guerin, Nelson, Olshansky, Roth and Smiths24 All the pictures were performed with the microscope Olympus BX51 with the DP72 digital camera (Olympus Corporation, Tokyo, Japan) to capture high-resolution images, and were analyzed using color deconvolution plugin to ImageJ software v1.53 (Wayne Rasband and contributors National Institutes of Health, USA; http://imagej.nih.gov/ij).
Cardiac tissue RNA extraction
According to the manufacturer’s protocol, total RNA was isolated from 90 mg of cardiac tissue using the Trizol reagent (Invitrogen, California, U.S.A.). Spectrophotometry (Nanophotometer Implen GmBH, München, Germany) was used to determined RNA purity and concentrations. All samples were frozen and stored at −80 °C until use.
Gene expression analysis
RNA (100 ng) was used for DNA synthesis (cDNA) using the AgPath-ID™ One-Step RT-PCR Kit Reagents (Applied Biosystems™, Thermo Fisher Scientific) according to the provider’s instructions.
Gene expression of actin alpha 1 skeletal muscle (Acta1), β-myosin heavy chain isoform (Myh7), natriuretic peptide A (Anp), natriuretic peptide B (Bnp), and collagen type I alpha 1 chain (Col1a1) was determined in cardiac tissue by reverse transcription-quantitative polymerase chain reaction and TaqMan on Demand Gene Expression Probes from Applied Biosystem. Assay ID: Acta1 (Rn01426628_gl), Myh7 (Rn00568328_m1), Anp (Rn00561661_m1), Bnp (Rn00580641_m1), and Col1a1 (Rn01463848_m1). All samples were normalized using the expression of internal control β-actin (assay ID: Rn00667869_m1).
Reverse transcription-quantitative PCR was performed on a LightCycler® 480 Instrument (Roche Diagnostics Ltd, Switzerland), and relative mRNA concentrations were calculated with a LightCycler Relative Quantification analysis Software. All samples were analyzed in duplicate.
Western blotting
Between 150 and 180 mg of cardiac muscle were homogenized in RIPA lysis buffer (Santa Cruz Biotechnology, Inc. Dallas, U.S.A.) (sc-2494A) with protease and phosphatase inhibitors. Fifty µg of protein was separated in SDS-PAGE gel and transferred to a nitrocellulose membrane (BIO-RAD Laboratories, Cat. #1620115, Germany); after being blocked with 5% fat-free milk, the membranes were incubated with different primary antibodies. To evaluate the pathological CH pathway, we used anti-ACTA1 (ab28052, dilution 1:10000), anti-MYH7 (ab11083, dilution 1:10000), anti-MYH6 (ab207926, dilution 1:8000), anti-ANP (ab209232, dilution 1:14000), anti-BNP (ab239510, dilution 1:6000), anti-COL1A1 (ab90395; dilution 1:8000) (Abcam Cambridge, UK). For the physiological CH pathway, we used anti-AKT (sc-5298, dilution 1:2000), anti-phospho-AKT (sc-514032, dilution 1:2000) (Santa Cruz Biotechnology, Dallas, U.S.A.), anti-mTOR (2972S; dilution 1:15000), anti-phospho-mTOR (S2448; dilution 1:15000) (Cell Signaling Technology, Massachusetts, U.S.A.), and anti-GADPH (PA1-988; dilution 1:14000) (ThermoFisher Scientific, Rockford, U.S.A.) was used as a loading control.
Afterward, incubation with HRP-goat anti-rabbit IgG (111-095-003) and anti-mouse IgG (715-035-150) (Jackson Inmuno Research Laboratories, I.N.C.) as secondary antibodies. Detection of proteins was done with SuperSignal tm West Femto Maximum Sensitivity Substrate Kit (ThermoFisher Scientific, Rockford, U.S.A.). Band intensities were digitally quantified using LI-COR Image Studio software (http://www.licor.com/bio/products/software/image_studio_lite/), and ImageJ software (U. S. National Institutes of Health, Bethesda, Maryland, U.S.A.).
Statistical analysis
Data are expressed as means ± SEM. Statistical comparisons were made using two-way analysis of variance to estimate the effect of two variables (maternal obesity and epicatechin intervention), followed by Tukey’s multiple comparison test. P value < 0.05 was accepted as statistically significant. Statistical analysis and the graphics were performed with GraphPad Prism 10.1.2 software (GraphPad Software, San Diego, CA).
Sample size and power were calculated according to the algorithm of the percentage of success and error test (by group/by trial): n = log B/log p; n = 8.39 per group/test:
B = power of statistical test = 1-B
where B is the power of the test = (0.05)
p = proportion of animals in the colony (expected event)
n= log (0.05)
log (0.70) =v8.39 per group/per test
Results
Body weight of the mothers/offsprings and weights of different fat pads to their offsprings
The F0 dams from the MO group were significantly heavier than the controls (p < 0.05, Fig. 1). Further, the F0 rats of the MO group were obese at the moment of conception and during gestation and lactation.
Body weight of the mothers. The F0 dams from the MO group were significantly heavier than the controls (five weeks after they start to consume the high fat diet).

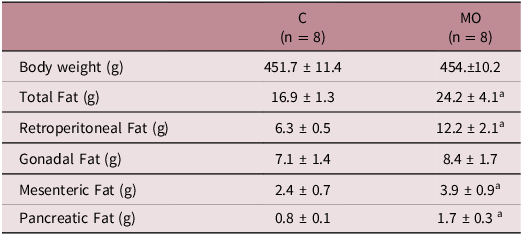
On the other had, we did not observe differences in the body weight of the offspring of the controls versus the offspring of the obese mothers group (Table 1). However, we observed a significantly higher total amount of the visceral fat tissue in the male offspring of the MO group when compared to the male offspring of the C group (p < 0.05), reflecting a higher adiposity index in the male offspring of the MO group. Besides, we found that the male offspring of the MO group had a greater amount of retroperitoneal, mesenteric and pancreatic fat tissue, when compared to the male offspring of C group (Table 1).
Body weight and weights of different fat pads to rats of the 110 postnatal day

n = number of animals; C = Control; MO = offspring of model obesity; a p < 0.05 vs C. Data are expressed as mean ± SD.
(-)-Epicatechin treatment increases the ratio of heart weight to body weight (HW/BW) in male offspring of obese Wistar rats
We found a significant increase of 9.4% (1.09-fold) of the HW/BW ratio in the male offspring of the C + Epi group vs. the control group (3.39 ± 0.17 mg/g and 3.10 ± 0.17 mg/g; p < 0.05, respectively) (Fig. 2, Panel A and D). Similarly, the male offspring of the MO + Epi increased the HW/BW ratio by 9.9% (1.10-fold) in comparison to the MO group (3.24 ± 0.26 mg/g and 3.56 ± 0.23mg/g; p < 0.05, respectively) (Fig. 2, Panel A and D).
Effect of maternal obesity and Epi administration in the HW/BW ratio of the four experimental groups. A) Representative histological images of heart sections below the atrioventricular groove in different study groups. Scale bars 1 mm. RV = right ventricle, SP = septum wall, and LV = left ventricle stereoscope binocular with a 10X eyepiece magnification of 0.7X. B) Body weight and C) Heart weight of the offspring, we did not observe significantly differences between the four experimental group. D) The HW/BW significantly differs between C vs. C + Epi and MO vs. MO + Epi groups. *p<0.05 vs. C + Epi, and *MO vs. MO + Epi; n = 8 male rats per group. C = male rat offspring of control mothers; C + Epi= male rat offspring of control mothers treated with Epi; MO = male rat descended from obese mothers; MO + Epi= male rat descended from obese mothers treated with Epi (all offspring in the four experimental groups were fed with control diet); SEM = standard error of the mean.

No significant differences in BW, heart weight (Fig. 2, Panel C and D) and food intake (data not shown) were observed between the study groups analyzed.
(-)-Epicatechin treatment decreases the content of collagen area of myocardial in male offspring of obese Wistar rats
Regarding the quantification of myocardial of the content of collagen area in both ventricles’ slices, we found a significant decrease (1.94-fold) of them in the male offspring of the C + Epi group vs. the control group (0.52 ± 0.03 % and 1.0 ± 0.06%; p < 0.0001, respectively) (Fig. 3, Panel A and B). Similarly, the Epi treatment (MO + Epi group) decreased the content of collagen area of 1.30-fold compared with the MO group (0.95 ± 0.05 % and 0.73 ± 0.04%; p < 0.05, respectively) (Fig. 3, Panel A and B).
Effect of maternal obesity and Epi administration in the content of collagen, area and the perimeter cavity of left ventricle of the heart of the four experimental groups. A) Representative Masson’s trichrome staining of fibrosis area in the heart of the different study groups. Scale bars = 100 μm. B) Area of content of collagen shows a significant difference between Epi-treated rats compared to rats without treatment ****C vs. C + Epi and *MO vs. MO + Epi (****p<0.0001 and *p<0.05, respectively); n = 6 male rats per group. C) and D) A significant increase of area and the perimeter was observed of the cavities of the left ventricle of the offspring of maternal obesity compared to the control group. *p < 0.05 C vs. MO group; n = 8 male rats per group. Data are expressed as mean ± SEM and were analyzed by two-way ANOVA followed with Dunnett’s post hoc test for multiple comparison. C = male rat offspring of control mothers; C + Epi= male rat offspring of control mothers treated with Epi; MO = male rat descended from obese mothers; MO + Epi= male rat descended from obese mothers treated with Epi (all offspring in the four experimental groups were fed with control diet); SEM = standard error of the mean.

Effect of maternal obesity and (-)-epicatechin treatment in size of the left ventricle cavity and ventricular wall thickness measurement in male offspring of obese Wistar rats
The heart’s area and perimeter were measured to assess the size of the left ventricle cavity. We found a significant increase of 60.5% in the area (5.37 ± 1.83 mm2 and 8.62 ± 2.54 mm2; p < 0.05) and of 26.1% in the perimeter (13.42 ± 2.56 mm and 16.92 ± 1.83 mm; P < 0.05), only when comparing C versus MO group, respectively (Fig. 3, Panel C and D). Nevertheless, we did not find significant differences in the total area of the heart between the groups tested, as well as in the area and mass of the left ventricle (Supplementary Fig. 2, Panel A-to-C).
Furthermore, to evaluate the thickness of the walls, three positions were measured for the RV and LV walls and the septum. We did not observe significant differences in the ventricular walls or septum thickness in the male offspring of experimental groups (C, MO, C + Epi, and MO + Epi) (Supplementary Fig. 3, Panel A-to-D).
Effect of maternal obesity and (-)-epicatechin treatment in the mRNA expression of genes related to pathologic cardiac hypertrophy in male offspring of obese Wistar rats
Regarding the RNA expression, we found a significant increase of Myh7 (2.8-fold) and Anp (37.6-fold) in the C vs. MO group (p < 0.01 and p < 0.0001, respectively) (Fig. 4, Panel A and B). Further, the Epi treatment in the MO group (MO + Epi) induced a decrease in the expression of Anp (16.1-fold) in comparison to the MO group (p < 0.05, Panel B). We did not observe differences in Bnp, Acta1, and Col1a1 expression in any of the experimental groups (Fig. 4, Panel C-to-E).
Effect of maternal obesity and Epi in mRNA expression of A) Myh7, B) Anp, C) Bnp, D) Acta1 and E) Col1a1 in the cardiac tissue of the four experimental groups. A) and B) Maternal obesity increases the expression of markers of pathological CH; Myh7 and Anp, **p < 0.01, C vs. MO; ****p < 0.0001, C vs. MO, respectively. B) Effect of Epi significantly decreases the expression of Anp in the MO group. *p < 0.05, MO vs. MO + Epi; ; n = 7-to-8 male rats per group. Data are expressed as mean ± SEM and were analyzed by two-way ANOVA followed with Dunnett’s post hoc test for multiple comparison. C = male rat offspring of control mothers; C + Epi= male rat offspring of control mothers treated with Epi; MO = male rat descended from obese mothers; MO + Epi= male rat descended from obese mothers treated with Epi (all offspring in the four experimental groups were fed with control diet); SEM = standard error of the mean.

Effect of maternal obesity and (-)-epicatechin treatment in the expression of proteins related to pathologic cardiac hypertrophy in male offspring of obese Wistar rats
The Myh7 protein expression increased by 24.38% (1.32-fold) in offspring of C + Epi vs. the MO + Epi group (p < 0.05, Fig. 5, Panel A and C). Besides, the expression of Anp protein was decreased by 56.73% (2.3-fold) in the MO group in comparison to the C group (p < 0.05), albeit the Epi treatment increased it by 59.14% (2.4-fold) in the MO + Epi group compared to the MO group (p < 0.05; Fig. 5, Panel A and E). Further, a significant decrease in Col1a1 protein expression of 86.9% (7.6-fold) was found in the MO group against the C group (p < 0.05) (Fig. 5. Panel A and H); as well as the Epi treatment diminished the expression of this protein of 84.5% (6.4-fold) for C + Epi vs. C group and of 85.1% (6.6-fold) for MO + Epi front C group (p < 0.05 for both) (Fig. 5. Panel A and H).
Effect of maternal obesity and Epi treatment on markers of pathological CH in the cardiac tissue of the four experimental groups. A) representative immunoblotting of protein of Myh6, Myh7, Anp, Bnp, Acta1, and Col1a1. Gadpn was used as loading control. B-to-H) densitometry analysis of Myh6, Myh7, Myh7:Myh6 ratio, Anp, Bnp, Acta1, and Col1a1. C) Myh7 shows a higher protein level in offspring of C + Epi vs. MO + Epi group. E) maternal obesity decreased the expression of Anp and the Epi treatment increases it in the MO group. H) control group shows a higher protein level of Col1a1 with respect to C + Epi, MO and MO + Epi groups. *p < 0.05 vs. for all groups with significant differences; n = 6-to-8 male rats per group. Data are expressed as mean ± SEM and were analyzed by two-way ANOVA followed with Dunnett’s post hoc test for multiple comparison. CH = cardiac hypertrophy; C = male rat offspring of control mothers; C + Epi= male rat offspring of control mothers treated with Epi; MO = male rat descended from obese mothers; MO + Epi= male rat descended from obese mothers treated with Epi (all offspring in the four experimental groups were fed with control diet); SEM = standard error of the mean.

We did not observe differences in the expression of Myh6, Myh7:Myh6 ratio, Bnp, and Acta1 in any experimental group (Fig. 5, Panel A, B, D, F and G).
Maternal obesity and (-)-epicatechin treatment did not modify the Akt and mTOR pathway in male offspring of obese Wistar rats
We did not observe Epi treatment modifications in the phosphorylation level of Akt and mTOR in the male offspring of experimental groups (C, MO, C + Epi, and MO + Epi) (Supplementary Fig. 4, Panel A-to C).
Discussion
Studies in human and animal models have shown that maternal obesity has a negative impact on the fetus or neonate, increasing the risk of disease development in the short and/or long term in the offspring. Reference Zambrano, Ibáñez, Martínez-Samayoa, Lomas-Soria, Durand-Carbajal and Rodríguez-González3,Reference Godfrey, Reynolds and Prescott6,Reference Catalano and deMouzon25 Specifically, mothers fed a high-fat diet develop metabolic disorders and increase their offspring’s predisposition to developing obesity, Reference Lecoutre, Petrus, Rydén and Breton26 and CH. Reference De Jong, Barrand and Wood-Bradley9,Reference Loche, Blackmore and Carpenter10,Reference Kereliuk and Dolinsky27
On the other hand, it has been described that Epi has cardioprotective properties in animal models. Reference De Los Santos, Palma-Flores, Zentella-Dehesa, Canto and Coral-Vázquez20,Reference Jiménez, Duarte and Perez-Vizcaino28,Reference Galleano, Bernatova and Puzserova29 Herein, we evaluated in a model of obesity by programing, previously characterized by our research group, Reference De los Santos, Coral-Vázquez and Menjivar4 whether Epi chronic treatment (90 days) can reverse the deleterious effects of maternal obesity programing on the hearts of the offspring.
Prior studies have reported an increase in the HW/BW ratio in rats descended from obese mothers. Reference De Jong, Barrand and Wood-Bradley9,Reference Loche, Blackmore and Carpenter10 In contrast, our study found no significant changes in the HW/BW ratio or the levels of fibrosis in the hearts of offspring from obese mothers. However, consistent with the findings of Loche et al., Reference Loche, Blackmore and Carpenter10 and Basso et al., Reference Basso, Michaud and d’Amati30 our study revealed an increase in the area and perimeter of the left ventricular cavities. This observation suggests a remodeling of cardiac tissue characterized by eccentric growth. In this sense, it has been proposed that cardiac hypertrophy is triggered by volume and/or pressure overload, initially resulting in concentric CH. Over time, this condition can progress and evolve into eccentric and dilated forms of CH. Reference Basso, Michaud and d’Amati30,Reference Ahmed, Liang and Chi31 Our research group previously demonstrated that a 15-day treatment with Epi induces physiological CH in mice. This effect is characterized by increases in cardiac mass, septal thickness, and left ventricular wall thickness, accompanied by enhanced activity of the PI3K/Akt signaling pathway. Reference De Los Santos, Palma-Flores, Zentella-Dehesa, Canto and Coral-Vázquez20 The activation of adaptive CH occurs via this signaling pathway, Reference McMullen, Shioi and Zhang32 essential for normal myocardial growth and enhancing cardiac function under pathological conditions. Reference Shioi, Kang and Douglas33,Reference McMullen and Jennings34 Interestingly, while we observed that Epi increased the HW/BW ratio in both the C + Epi and MO + Epi groups, we found no differences in the proteins associated with the Akt/mTOR pathway, as reported by de los Santos et al., Reference De Los Santos, García-Pérez and Hernández-Reséndiz19 therefore, at least in the offspring of obese mothers, this change in heart mass could be related to pathological CH.
Conversely, the molecular mechanisms underlying pathological cardiac hypertrophy involve the upregulation of fetal gene programs, characterized by increased expression of Myh7, Anp, Bnp, and Acta1, alongside the downregulation of Myh6. Reference Bernardo, Weeks, Pretorius and McMullen35 Consequently, this study examined the expression of these pathological hypertrophy markers at both the mRNA and protein levels.
De Jung et al., Reference De Jong, Barrand and Wood-Bradley9 observed an increase in the expression of Myh7, Anp, and Bnp mRNA, along with elevated Col1a1 mRNA an established marker of fibrosis in younger rat offspring of obese mothers. These findings suggest maternal obesity induces transcriptional reprograming of fetal genes associated with CH. Notably, the authors analyzed these pathological hypertrophy markers at postnatal days 1 and 10, coinciding with the period immediately following normal maturation of cardiac energy metabolism. In contrast, Loche et al., Reference Loche, Blackmore and Carpenter10 found that offspring of obese mothers exhibited an overexpression of Myh7 mRNA, while the expression levels of Anp, Bnp, and Col1a1 mRNA remained unchanged. They proposed that exposure to maternal obesity during gestation is as harmful to cardiac function in offspring as being obese in adulthood. Likewise, consistent with De Jong et al., Reference De Jong, Barrand and Wood-Bradley9 and Loche et al., Reference Loche, Blackmore and Carpenter10 we observed a significant increase in Myh7 mRNA expression in the offspring of obese mothers compared to the control group. However, no differences were noted at the protein level.
Importantly, Myh7 is crucial in facilitating slow heart contractions Reference Tardiff, Hewett, Factor, Vikstrom, Robbins and Leinwand36 by reducing the actin and myosin cross-bridge formation rate. Reference Gupta37 Consequently, during the initial stages of injury, the upregulation of Myh7 in the offspring of obese mothers may enhance cardiac performance. Reference Sadoshima and Izumo38 Supporting this notion, De Jong et al., Reference De Jong, Barrand and Wood-Bradley9 described, in other pathways, early changes at the transcriptional level but not at the protein level during the stage of compensated hypertrophy in rodent models of obesity and type 2 diabetes mellitus.
In our analysis of Anp, we observed an increase in mRNA expression in the offspring of obese mothers; however, Epi treatment reduced this expression within that group. Notably, Anp plays a critical role in regulating blood volume and vascular tone, Reference Baron, Lofton, Newman and Currie39,Reference Chopra, Cherian, Verghese and Jacob40 generating compensatory hemodynamic effects that help delay the progression of heart failure and the dilation of the ventricular walls. Reference Chopra, Cherian, Verghese and Jacob40 Interestingly, we observed an inverse phenomenon at the protein level, with the offspring of obese mothers exhibiting decreased Anp levels compared to the control group (C vs MO). This finding correlates with the observed increase in left ventricular cavity size and the development of eccentric CH. We hypothesize that this reduction in Anp protein levels could represent a compensatory mechanism in response to the cardiac stress associated with maternal obesity. Despite the cardioprotective effects of Epi treatment, Reference De Los Santos, Palma-Flores, Zentella-Dehesa, Canto and Coral-Vázquez20,Reference Jiménez, Duarte and Perez-Vizcaino28,Reference Galleano, Bernatova and Puzserova29 our results revealed an increase in Anp levels in the MO + Epi group compared to the MO group. One possible explanation for this discrepancy is that previous studies employed shorter durations of Epi treatment, Reference De Los Santos, Palma-Flores, Zentella-Dehesa, Canto and Coral-Vázquez20,Reference Jiménez, Duarte and Perez-Vizcaino28,Reference Galleano, Bernatova and Puzserova29,Reference Yamazaki, Romero-Perez and Barraza-Hidalgo41 ranging from 6 to 28 days, while our study administered Epi for an extended period of 90 days. This prolonged exposure may have different physiological implications. In this context, Epi has been proposed as an exercise mimetic, capable of inducing mitochondrial biogenesis in vitro Reference Moreno-Ulloa, Cid, Rubio-Gayosso, Ceballos, Villarreal and Ramirez-Sanchez42 and in vivo. Reference Nogueira, Ramirez-Sanchez and Perkins17 Therefore, the extended duration of Epi treatment could mimic the effects of high-performance exercise, potentially leading to the development of CH. Regarding Col1a1 protein levels, we observed a significant decrease in the MO group compared to the control group. This reduction may be attributed to COL1A1’s role in mediating histological changes in the heart through remodeling the extracellular matrix, Reference Kong, Christia and Frangogiannis45 which involves thickening fibers and providing tensile strength during the contraction process. Reference Fan, Takawale, Lee and Kassiri46 We hypothesize that the downregulation of COL1A1 could represent a compensatory mechanism facilitating these histological adaptations. Furthermore, Epi treatment in both the C + Epi and MO + Epi groups significantly reduced COL1A1 protein expression compared to their counterparts (C and MO). This effect was also observed by Luo et al., Reference Luo, Lu and Wang47 who demonstrated that Epi treatment significantly reduced the cardiac fibrosis biomarkers, including COL1A1. Their results imply that Epi may inhibit the transformation of myofibroblasts and the synthesis of collagen, thereby contributing to a pathway that prevents the development of cardiac fibrosis. Epi may play a crucial role in preserving cardiac structure and function by mitigating these processes, particularly in contexts associated with increased fibrosis. Finally, we did not observe significant differences in the Myh7:Myh6 ratio or in the expression levels of Myh7, Myh6, Acta1, and Bnp across any of the experimental groups. This lack of variation could be attributed to the young age of the animals (110 days). Previous studies have indicated that the deregulation of critical genes and transcription factors in the heart can lead to late-onset heart disease. Reference Ahmed, Liang and Chi31 Consequently, the adverse effects of maternal obesity are associated with a higher risk of developing cardiovascular disease during adulthood. Reference Gaillard48,Reference Kankowski, Ardissino and McCracken49 Therefore, it is possible that our animals may develop cardiac disorders later in life as a consequence of these early-life exposures.
In conclusion, our study indicates that maternal obesity promotes the activation of pathological markers of CH through the reactivation of fetal cardiac genes associated with the histological changes observed in cardiac tissue. Likewise, chronic treatment with (-)-epicatechin appears to mitigate the expression of specific fetal cardiac genes involved in the pathological CH pathway in the offspring of obese mothers while also decreased the content of collagen area. Therefore, by identifying the potential therapeutic effects of (-)-epicatechin, our research proposes this flavonoid as a promising possible intervention that requires further investigation. Additionally, further research is needed to elucidate the long-term effects of maternal obesity on cardiac function in offspring, particularly as they transition into adulthood.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S204017442510010X
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
This work was submitted in partial fulfillment of the requirements for the PhD degree of Leticia Orozco-Arguelles at the Programa de Doctorado en Ciencias Bioquímicas, Universidad Nacional Autónoma de México. L. Orozco-Arguelles was supported by a Consejo Nacional de Ciencia y Tecnología (CONACyT), México, fellowship award. Sergio De los Santos received a postdoctoral fellowship award from the Programa de Becas Posdoctorales de la Dirección General de Asuntos del Personal Académico, División de Investigación, Facultad de Medicina, Universidad Nacional Autónoma de México. We thank Sebastián De la Rosa, María Elena Tejeda and Jorge Uribe, from the Unidad de Investigación en Obesidad, Facultad de Medicina, Universidad Nacional Autónoma de México (S.D.R. and M.E.T) and from the Departamento de Biología de Reproducción, Instituto Nacional de Ciencias Médicas y Nutrición “Salvador Zubirán” (J.U.), for their technical assistance in this study.
Authors contribution
Orozco-Arguellez L and De los Santos S: Conceptualization; Data curation; Formal analysis; Investigation; Visualization; Manuscript elaboration. Coral-Vázquez RM: Conceptualization; Formal analysis; Investigation; Visualization; Manuscript elaboration. Zambrano E: Conceptualization; Formal analysis; Visualization; Funding acquisition; Manuscript elaboration. Vega-García CC: Data curation; Histopathological analysis; Investigation. Canto P: Conceptualization; Formal analysis; Funding acquisition; Investigation; Project administration; Supervision; Manuscript elaboration. All authors reviewed the manuscript.
Financial support
This work was partially supported by the Fondo Sectorial de Investigación para la Educación SEP-CONACYT, Convocatoria de Investigación Científica Básica 2017-2018, CONACyT México, under Grant: A1-S-9740, and by the Newton Fund RCUK- CONACYT-2015 (Research Councils UK–CONACyT).
Competing interests
None.
Ethical standard
The animal research protocol followed in the present study was approved by the Animal Experimentation Ethics Committee of the Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán (INCMNSZ) (CINVA: UIO-1892-17/19-1) and by the Research and Ethics Commissions of the Facultad de Medicina, Universidad Nacional Autónoma de México (FM/DI/001/2017).
Artificial intelligence
The authors do not use artificial intelligence (AI) or AI-assisted technologies in the writing process.