Introduction
Seabirds rely extensively on the marine environment for their life cycle and play an important role in marine food webs (Piatt and Sydeman Reference Piatt and Sydeman2007; Votier and Sherley Reference Votier and Sherley2017). Seabirds integrate processes occurring at lower trophic levels and are easy to observe when compared to other marine organisms. As such, seabirds are broadly used as indicators of ocean functioning and ocean health, notably in ecotoxicology, food web ecology, and conservation and fisheries sciences (Furness and Camphuysen Reference Furness and Camphuysen1997; Piatt and Sydeman Reference Piatt and Sydeman2007). As meso- to top predators with diverse diets, and the ability to migrate over distances ranging from short to intercontinental, seabirds are likely to encounter and disperse a wide range of trophically transmitted parasites from a variety of geographic regions (Gutiérrez et al. Reference Gutiérrez, Rakhimberdiev, Piersma and Thieltges2017; Mendes et al. Reference Mendes, Eira, Vingada, Miquel and Torres2013). However, there is a major knowledge gap on the parasite fauna of seabirds, notably for endoparasites such as helminths, which are mainly trophically transmitted and can disrupt host metabolism, fertility, and survival (Khan et al. Reference Khan, Provencher, Forbes, Mallory, Lebarbenchon and McCoy2019). These parasites are difficult to sample or observe with non-invasive techniques, thereby limiting their investigation in seabirds, which are one of the most endangered groups of birds globally (Dias et al. Reference Dias, Martin, Pearmain, Burfield, Small, Phillips, Yates, Lascelles, Borboroglu and Croxall2019). Moreover, the scarcity of available DNA sequences of seabird helminths (non-economically or medically relevant parasites are generally poorly represented in GenBank, Hupało et al. (Reference Hupało, Blasco-Costa, Trujillo-González, Leese, Smit and Sures2025)), as well as the intermittent excretion of helminth eggs or debris in faeces (Granroth-Wilding et al. Reference Granroth-Wilding, Daunt, Cunningham and Burthe2017), limits our capacity to rely solely on molecular non-invasive techniques to identify them. As such, necropsies of birds that died naturally are an essential step in the direct sampling and description of seabird helminth communities (e.g., Granroth-Wilding et al. Reference Granroth-Wilding, Daunt, Cunningham and Burthe2017; Mallory et al. Reference Mallory, McLaughlin and Forbes2007; Provencher et al. Reference Provencher, Bond, Hedd, Montevecchi, Muzaffar, Courchesne, Gilchrist, Jamieson, Merkel, Falk, Durinck and Mallory2014).
In the present study, we document the infection of ten seabird species from the North-East Atlantic and Antarctica with gastrointestinal helminths. Our aim was to assess the helminth diversity in different seabird host species and the association with host ecological characteristics (habitat preference, feeding strategies, and condition). We expected a higher diversity of helminths in coastal- than in oceanic-feeding seabirds, because several helminths rely on terrestrial, shoreline, and shallow water intermediate hosts to complete their life-cycle (Hoberg Reference Hoberg1996). We also expected potentially different infection levels in bycatch birds in good condition compared to stranded individuals that died due to a variety of causes. Given the heterogeneous nature of our opportunistic sampling and the partly low sample sizes, the results are interpreted with regard to the study’s limitations. Nevertheless, despite its exploratory character, this study provides valuable baseline information on the largely understudied helminth fauna of seabirds. The data presented here can serve as a reference point for future investigations, particularly in the context of ongoing environmental change. As shifting climatic and ecological conditions are likely to affect parasite distribution, transmission dynamics, and infection levels, establishing such baseline knowledge is crucial for tracking temporal trends and assessing future impacts on seabird populations and marine ecosystems.
Material and methods
Sample collection
Two sets of fresh carcasses were used in this study. The first set (‘stranded birds’) included 38 individual North-East Atlantic seabirds belonging to eight species (Atlantic puffins Fratercula arctica, common guillemots Uria aalge, razorbills Alca torda, European shags Gulosus aristotelis, black-legged kittiwakes Rissa tridactyla, common gulls Larus canus, sandwich terns Thalasseus sandvicensis, and northern fulmars Fulmarus glacialis) that were collected by wildlife rescue centres between 2010 and 2018 in coastal areas of north-western France, and three individuals belonging to 2 Antarctic species (south polar skua Stercorarius maccormicki and Antarctic petrel Thalassoica antarctica) collected at Svarthamaren, Dronning Maud Land (Antarctica) in 2022. The cause of death of the first set of carcasses was unknown in several cases (52%), while known causes included starvation due to winter storms (18%), oil pollution (15%), and others (collisions with objects, bycatch, 15%). Most North-East Atlantic birds were adults and were found during the nonbreeding period from September to March (boreal autumn–winter). All European shags were juveniles (post-fledging chicks), and a few individuals of other species were juveniles or immatures (3/5 Atlantic puffins, 1/4 common gulls, 1/11 razorbills, 1/5 sandwich terns). The Antarctic birds were all adults and were collected during the breeding period in January (austral summer). The second set of carcasses (‘bycatch birds’) included five adult common guillemots and five adult razorbills from a single bycatch event. These birds were entangled in a single fishing net that ended up on a beach in northern France in December 2018. These wintering individuals were in very good condition and presumably healthy. The ‘bycatch’ common guillemots and razorbills were compared to ‘stranded birds’ of the same species to test potential differences in infection levels between birds in good and bad condition, respectively. The total number of sampled individuals was 52 and all species and their feeding ecology are presented in Table 1. Carcasses from North-East Atlantic seabirds were stored at −20°C until dissections at the Centre d’Etudes Biologiques de Chizé (CEBC), France (stranded birds), or at the Littoral Environnement et Sociétés (LIENSs), France (bycatch birds), in 2022. Carcasses from stranded Antarctic individuals were dissected on site in 2022, with digestive tracts being isolated and stored at −20°C until extraction of parasites at the CEBC or at the University of Duisburg-Essen, Germany, in 2022.
Bird species investigated including preferred habitat and feeding strategy, food source, and intensities of parasite infections. Intensity was quantified only for higher-level taxonomic groups of helminths (Acanthocephala, Cestoda, Nematoda, Trematoda). When possible, the number of cestode scolices was quantified

Abbreviations: L.i., Large intestine; S, stomach; S.i., small intestine.
References for diet (for Northern Hemisphere species, studies in the North-East Atlantic were prioritised): 1 Fayet et al. (Reference Fayet, Clucas, Anker-Nilssen, Syposz and Hansen2021); 2 St. John Glew et al. (Reference St John Glew, Wanless, Harris, Daunt, Erikstad, Strøm and Trueman2018); 3 St. John Glew et al. (Reference St John Glew, Wanless, Harris, Daunt, Erikstad, Strom, Speakman, Kurten and Trueman2019); 4 Anderson et al. (2014); 5 Lewis et al. (2001); 6 Markones et al. (Reference Markones, Guse, Hüppop and Garthe2009); 7 Lemesle et al. (Reference Lemesle, Jouanneau, Cherel, Legroux, Ward, Bustamante and Chastel2024); 8 Ojowski et al. (Reference Ojowski, Eidtmann, Furness and Garthe2001); 9 Darby et al. (Reference Darby, Dde Grissac, Arneill, Pirotta, Waggitt, Börger, Shepard, Cabot, Owen, Bolton, Edwards, Thompson, Quinn and Jessopp2021, Reference Darby, Clairbaux, Quinn, Thompson, Quinn, Cabot, Strom, Thorarinsson, Kempf and Jessopp2023); 10 Kubetzki et al. (Reference Kubetzki, Garthe and Hüppop1999); 11 Kubetzki and Garthe (Reference Kubetzki and Garthe2003); 12 Courtens et al. (Reference Courtens, Verstraete, Vanermen, Van de walle and Stienen2017); 13 Michelot et al. (Reference Michelot, Pinaud, Fortin, Maes, Callard, Leicher and Barbraud2017); 14 Howells et al. (Reference Howells, Burthe, Green, Harris, Newell, Butler, Wanless and Daunt2018); 15 Alfaro Garcia et al. (2022); 16 Brooke et al. (Reference Brooke, Keith and Rov1999); 17 Weimerskirch et al. (Reference Weimerskirch, Tarroux, Chastel, Delord, Cherel and Descamps2015); 18 Descamps et al. (Reference Descamps, Tarroux, Cherel, Delord, Godo, Kato, Krafft, Lorentsen, Ropert-Coudert, Skaret and Varpe2016); 19 Lorentsen et al. (Reference Lorentsen, Klages and Røv1998).
Extraction and identification of parasites from the gastrointestinal tract
Dissections were carried out using ethanol-sterilised instruments, beginning while the carcasses were still partially frozen. The digestive tract was separated from the carcass by ligaturing and transversally cutting the ends of the oesophagus and cloaca. Thereafter, different sections of the digestive tract were obtained: stomach, small intestine, and large intestine. The small and large intestines were then subdivided into three to 5 sections, with the number of sections varying depending on the species and the length of the intestine. The stomach and intestinal sections were cut open longitudinally and searched for parasites under a stereo-microscope (Leica MS5). Subsequently, the intestinal sections were washed in a beaker with a 400–500 mL physiological solution (0.9% NaCl). The contents of the beaker were then successively filtered using two filters (630 μm, 63 μm). The sieve residue of both filters was washed further with the physiological solution and then examined under the stereo-microscope. The parasites were extracted with tweezers and transferred to Petri dishes containing physiological solutions. For each bird host, the parasites were sorted according to the following taxonomic groups: acanthocephalans, cestodes, nematodes, and trematodes. Residual host digestive content was removed from the parasites by rinsing them several times with physiological solution. For molecular identification, parasites were washed twice in Milli-Q water to remove excess debris and salt residues. Thereafter, the parasites were transferred to a dish containing 70% ethanol, followed by full fixation in 99% ethanol.
Morphological and molecular identification of parasites was carried out at the laboratory of the Aquatic Ecology department at the University of Duisburg-Essen, Germany. For the morphological identification, parasites were first photographed under a stereo-microscope (Olympus SZ51) equipped with a Moticam 2300, and with a light microscope (Olympus BX51), and then identified to the lowest taxonomic level possible based on the relevant literature (Hoberg & Soudachanh Reference Hoberg and Soudachanh2020; Munro et al. Reference Munro, Crompton and Stoddart1995; Nagasawa et al. Reference Nagasawa, Baruš and Ogi1998; Rocka Reference Rocka2019). As the examined bird carcasses varied in their degree of preservation due to extended storage at −20°C, morphological identification at high taxonomic resolution was not always possible. For example, parasites in poor condition (e.g., small fragments or heavily degraded specimens) were excluded from morphological identification. Therefore, molecular identification using DNA barcoding was considered a more reliable approach for taxonomic identification. As amplification success as well as representation in GenBank can differ for the parasite taxa investigated, different primer pairs were used to amplify partial sequences of the internal transcribed spacer (ITS), cytochrome oxidase I (COI), and the 18S rRNA and 28S rRNA gene, via polymerase chain reaction (PCR) (for primer sequences and references as well as the respective PCR conditions see Supplementary Table S1). The parasite samples were homogenized in 1.5-mL reaction tubes with 600 μL TNES buffer (Tris, NaCl, EDTA, SDS) using a micropestle (Eppendorf), and the DNA extraction was performed with a salt precipitation according to Grabner et al. (Reference Grabner, Weigand, Leese, Winking, Hering, Tollrian and Sures2015). Amplification with the primer pairs was carried out in a 20 μL reaction volume containing 10 μL Accustart II PCR ToughMix with 0.35 μL loading dye (Quantabio), 0.5 μmol of each primer, and 1 μL DNA. The mixture was then brought to a volume of 20 μL with distilled water. The PCR protocols for each primer pair are listed in Supplementary Table S1. The positive PCR products were combined with 10 μL ultra-pure water, and 12 μL of each sample was sent for Sanger sequencing using the respective forward and reverse primers (Microsynth Seqlab, Germany). The obtained sequences were assigned to the lowest possible taxonomic level using BLAST search (https://blast.ncbi.nlm.nih.gov/Blast.cgi). The sequences were edited, and alignments were created in GENEIOUS (version 2022.0.2, Biomatters Ltd., Auckland, New Zealand).
Data analysis
Parasite prevalence was calculated for each parasite taxon according to Bush et al. (Reference Bush, Lafferty, Lotz and Shostak1997). In order to evaluate the similarity in parasite species composition between host species, the Sørensen similarity index was considered, with bird species grouped according to their habitat and feeding strategy as follows: oceanic/diving: Atlantic puffin, Common guillemot, Razorbill; oceanic/surface: Black-legged kittiwake, Northern fulmar; Coastal: Common gull, Sandwich tern, European shag. Antarctic species were excluded from this analysis because of the small sample size and different ecological context.
Results
Molecular data in combination with morphological features were used to confirm the assignment to a particular parasite taxon. While non-economically or medically important parasites are generally underrepresented in GenBank (Hupało et al. Reference Hupało, Blasco-Costa, Trujillo-González, Leese, Smit and Sures2025), there is also a big difference among the gene regions that are used for barcoding. While 18S and 28S rRNA sequences are comparatively well represented, they mostly allow distinction of isolates only at higher taxonomic levels. In turn, ITS or COI allow distinction down to the species level (e.g., Contracaecum sp.), but hardly any parasite sequences are available. The primer pair/gene region that provided the best classification, as well as the related GenBank accession numbers, is shown in Supplementary Table S2. Only in a few cases, no sequence result was obtained, and identification in these cases was based purely on morphology (Supplementary Table S2).
A total of 52 individual seabirds belonging to 10 species were examined. Parasite load of acanthocephalans was highest in European shags, and most cestode scolices were retrieved from northern fulmars (Table 1). European shags and northern fulmars were also most heavily infected with nematodes, while trematode loads were highest in common gulls (Table 1). Overall parasite prevalence ranged from zero in Atlantic puffins and common guillemots up to 80% in European shags (Table 2; prevalence reached 100% in Antarctic petrels but this was based on a single individual host). In total, we identified 13 helminth taxa including 1 acanthocephalan of the genus Andracantha (likely two species due to sequence divergence of 2.8% for 28S rRNA in a sequence of 911 bp and 15.2% for COI in a sequence of 424 bp), 2 cestodes (Cyclophyllidea, and potentially 5 Tetrabothrius spp. based on divergence of 2.3–5.1% in a 1,037 bp 18S rRNA sequence), 6 nematodes (Anisakis sp., Contracaecum rudolphii and C. septentrionale, Cosmocephalus sp., Stegophorus sp., and three isolates of Spirurina based on divergence of 11.9–22.2% for COI in a sequence of 433 bp), and 4 trematode isolates (Cardiocephaloides longicollis, Cryptocotyle lingua, Himasthlidae, and Microphallidae) (Table 2). The largest diversity of parasite species was identified in northern fulmars (9 species), European shags (5 species), razorbills (4 species), and black-legged kittiwakes (4 species) (Table 2). Tetrabothrius sp. and C. septentrionale were found in most host species (n = 4), and no parasite taxon was present in all infected birds (Table 2). Co-infections with multiple parasite species in one host individual were common. The largest number of 5 parasite species was found in 1 northern fulmar, while co-infections with 2 parasite species were the most common overall (7 cases). Single infections were found in 5 birds, 2 parasite species in 8, 3 parasite species in 4, and 4 parasite species in 2 (Table 2). Only Anisakis sp. nematodes and microphallid trematodes were found in single host species, the northern fulmar and the black-legged kittiwake, respectively. Among North-East Atlantic seabirds, all four parasite groups were found in both coastal and oceanic surface-feeding birds (Table 3). Razorbills were infected exclusively by nematodes, while Sandwich terns were infected with a single trematode species (Cryptocotyle lingua). Antarctic species were dominated by acanthocephalans and cestodes (Table 2).
Parasites detected and their prevalence in ten seabird species from the North-East Atlantic and Antarctica. The identity (ID) of the infected individuals is shown to illustrate co-infections. ‘Types’ for Andracantha, Tetrabothrius, and Spirurina denote different sequence isolates that are likely indicating different species. Cyclophyllidea were also likely 5–6 different species according to sequences, but as isolates were obtained from different primer pairs, direct comparison was not possible (see Supplementary Table S2). Individuals underlined were identified based on morphology only, as no usable sequence was retrieved

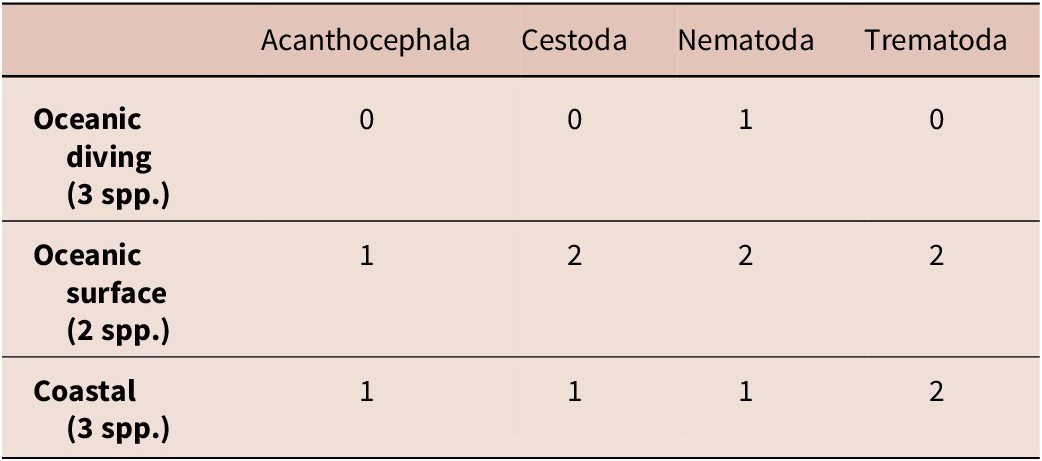
Number of seabird species infected with the respective parasite group according to habitat preference for North-East Atlantic seabirds

Among North-East Atlantic seabirds, the oceanic diving species were found to be the most parasite-poor in our study, both in terms of diversity (only nematodes) and number of infected bird species, i.e., only one (razorbills), while Atlantic puffins and common guillemots did not have any parasites (Table 3). Although the number of samples was limited, a pattern emerged with respect to habitat-related feeding preferences of bird species. The highest faunistic similarity in parasite species composition (Sørensen’s index = 0.66) was observed between the oceanic surface-feeding and coastal birds. In contrast, both groups showed markedly lower similarity to bird species feeding in the water column (oceanic divers), with Sørensen’s index values of 0.42 for the oceanic surface-feeding group and 0.33 for the coastal group. Due to the limited sample size of Antarctic bird species, they were excluded from the analysis.
A comparison of the parasites found in stranded and bycatch birds revealed a similar diversity. Specifically, no parasites were observed in either the stranded or bycatch individuals of the common guillemot. Among razorbills, two stranded birds (ID4 and ID6) and two bycatch birds (ID7 and ID9) hosted parasites of the species Contracaecum septentrionale (Table 2). One of the stranded razorbills (ID4) was also infected with three other species of nematodes (Table 2).
Discussion
The present study is one of the few to examine helminth occurrence in seabird hosts, especially in such a large number of seabird species simultaneously (n = 10), with different habitat preferences and feeding strategies, from both the North-East Atlantic and Antarctica. A total of 13 parasite taxa were identified, which most likely comprise more species, as genetic determination was not always possible down to the species level. To the best of our knowledge, this study is the first record of Andracantha sp. in northern fulmars and south polar skuas, of C. septentrionale in northern fulmars and black-legged kittiwakes, of a species of Microphallidae in black-legged kittiwakes, of Cardiocephaloides longicollis in European shags, of Cryptocotyle lingua in Sandwich terns, and of a cyclophyllidean species in south polar skuas and Antarctic petrels. Given the limited number of individuals examined for each seabird species (n between 1 and 6), we focus on the comparison of the parasite diversity between the studied species and the literature, without considering infection intensities, and provide a qualitative assessment of parasite diversity variation depending on host feeding strategy and habitat. Table 4 gives an overview of the available life cycle parameters of the parasites detected in the present study.
Information on life cycles of the parasites encountered in the present study in seabirds

Helminth diversity
Acanthocephala
Acanthocephalans of the genus Andracantha could be identified, with divergence of 28S rRNA and COI indicating the presence of at least two species. Due to a lack of matching sequences, these isolates could not be compared to existing molecular phylogenies (e.g., Garcia-Varela et al. Reference Garcia-Varela, Hernandez-Orts, Lopez-Jimenez and Gonzalez-Garcia2023). The genus Andracantha is common in species of the family Phalacrocoracidae (cormorant and shag species), Gaviidae (loon species), and in some penguins (Spheniscidae), but several species have also been described in other aquatic birds (Bennett et al. Reference Bennett, Presswell and Poulin2023; Garcia-Varela et al. Reference Garcia-Varela, Perez-Ponce de Leon, Aznar and Nadler2009; Reference Garcia-Varela, Hernandez-Orts, Lopez-Jimenez and Gonzalez-Garcia2023; Presswell et al. Reference Presswell, Garcia-Varela and Smales2018). To the best of our knowledge, we present the first record of Andracatha sp. in northern fulmars. The type found in northern fulmars was the same as the one infesting European shags in our sample (‘type A’). European shags were previously found to be infected with Andracantha tunitae (Munro et al. Reference Munro, Crompton and Stoddart1995). This is also the first record of Andracantha sp. (‘type B’) in south polar skuas, or in any skua species, to the best of our knowledge. However, the molecular sequences obtained showed insufficient discriminatory power between the Andracantha species, so it cannot be determined with certainty whether type A represents A. tunitae or another Andracantha species.
Cestoda
Both cestode groups found (the cyclophyllidean species and Tetrabothrius spp.) are common in seabirds with a number of known species (e.g., Barbosa & Palacios Reference Barbosa and Palacios2009; Hoberg Reference Hoberg1996; Hoberg and Soudachanh Reference Hoberg and Soudachanh2020; Rocka Reference Rocka2019; Schwantes Reference Schwantes2023). Again, divergence of the obtained 18S rRNA/COI sequence sections indicates the presence of different species of both groups, but the lack of reference sequences in GenBank prevented a more precise identification. Tetrabothrius sp. were found in four seabird species, including both Antarctic seabirds (each hosting a different species ‘type’). Interestingly, two Tetrabothrius sp. individuals, each sampled from a different northern fulmar, had different morphological characteristics, indicating that at least 2 different species of Tetrabothrius infected northern fulmars. In a previous study, Tetrabothrius minor was the most prevalent species in northern fulmars (Kuklin et al. Reference Kuklin, Kuklina and Ezhov2019), suggesting it as a potential candidate for the species recorded here. South polar skuas were previously found to be infected with Tetrabothrius (Culmenamniculus) cylindraceus at the South Shetlands Islands (Rocka Reference Rocka2019). Antarctic petrels were also already found to host four species of Tetrabothrius, although their identification is not available (Jones Reference Jones1988). In contrast, Cyclophyllidae cestodes were not previously described in the 2 Antarctic species targeted, being more common in subarctic birds feeding in terrestrial/coastal environments (kelp gull Larus dominicanus, Snowy sheathbill Chionis alba, Barbosa and Palacios Reference Barbosa and Palacios2009).
Nematoda
The most common nematode taxa found belonged to the genus Contracaecum, with Contracaecum rudolphii reaching the highest intensities in European shags. Both C. rudolphii and C. septentrionale have previously been recorded in European shags (Abollo et al. Reference Abollo, Gestal and Pascual2005). C. septentrionale was found to be less abundant than C. rudolphii both in the study of Abollo et al. (Reference Abollo, Gestal and Pascual2005) and in the present study. Similar to our study, C. septentrionale was found previously in razorbills (Li et al. Reference Li, D’Amelio, Paggi, He, Gasser, Lun, Abollo, Turchetto and Zhu2005), while it was, to our knowledge, not reported from black-legged kittiwakes and northern fulmars. Larval Anisakis sp. were also found in northern fulmars, as previously shown (e.g., Kuklin et al. Reference Kuklin, Kuklina and Ezhov2019; Mallory et al. Reference Mallory, McLaughlin and Forbes2007). Stegophorus sp. was found in northern fulmars and razorbills in the present study. The most commonly mentioned species of this genus in the literature occurring in the Northern Hemisphere is S. stellaepolaris, which was originally described from northern fulmars (Parona Reference Parona1901), but also found in a number of other seabird hosts (Nagasawa et al. Reference Nagasawa, Baruš and Ogi1998; Threlfall Reference Threlfall1971). Reference Nagasawa, Baruš and Ogi1998). This seemingly generalist parasite might be a species complex, as slightly divergent morphological variants were found in individuals parasitising Brünnich’s guillemots Uria lomvia (Nagasawa et al. Reference Nagasawa, Baruš and Ogi1998). The nematode Cosmocephalus sp. is also worldwide distributed and is found in various seabird hosts (e.g., Fusaro et al. Reference Fusaro, Capasso, Lorenti, Panisse, Fuentes, Fornillo, Libertelli, Ansaldo and Diaz2024; Mutafchiev et al. Reference Mutafchiev, Halajian and Georgiev2010; Shutler et al. Reference Shutler, Alisauskas and Daniel McLaughlin2012; Threlfall Reference Threlfall1971). Here, Cosmocephalus sp. was detected only in a single razorbill individual. Other species of nematodes were detected in razorbills, European shags, and northern fulmars, but could only be classified to a very general level (infraorder Spirurina), due to the lack of reference sequences in GenBank. Nevertheless, they seemed to be 3 different species based on the comparison of the sequences.
Trematoda
The most common trematode was Cryptocotyle lingua, with 3 seabird species being infected, specifically sandwich terns (first record, and the only helminth found in this seabird species in the present study), black-legged kittiwakes, and northern fulmars, as previously found (reviewed in Schwantes Reference Schwantes2023). This is consistent with C. lingua metacercariae being common in North-East Atlantic fish (Duflot et al. Reference Duflot, Gay, Midelet, Kania and Buchmann2021; 2023). Cardiocephaloides longicollis was the only trematode found in European shags and the first record for the species. Species of Himasthlidae and Microphallidae are commonly found in coastal ecosystems of the North-East Atlantic (Lauckner Reference Lauckner1984; Richard et al. Reference Richard, Maire, Daffe, Magalhaes and de Montaudouin2022), having gastropods as first intermediate hosts and reaching maturity in seabirds, notably in species of Laridae.
Qualitative differences in helminth diversity depending on host species feeding strategies
The targeted seabird species present contrasting feeding strategies and habitats. In the North-East Atlantic, all 3 species of auks (Atlantic puffins, common guillemots, and razorbills) are pursuit-diving, specialised piscivorous seabirds that feed primarily on small schooling fish, such as sandeels, clupeids, and sprat (Anderson et al. Reference Anderson, Evans, Potts, Harris, Wanless and Pichegru2013; Fayet et al. Reference Fayet, Clucas, Anker-Nilssen, Syposz and Hansen2021; St John Glew et al. Reference St John Glew, Wanless, Harris, Daunt, Erikstad, Strøm and Trueman2018; Reference St John Glew, Wanless, Harris, Daunt, Erikstad, Strom, Speakman, Kurten and Trueman2019). Chick-rearing adults forage in both oceanic and neritic waters, minimising distance to the colonies (Fayet et al. Reference Fayet, Clucas, Anker-Nilssen, Syposz and Hansen2021), while during the nonbreeding period, all three species feed mainly in oceanic waters (Buckingham et al. Reference Buckingham, Daunt, Bogdanova, Furness, Bennett, Duckworth, Dunn, Wanless, Harris, Jardine, Newell, Ward, Weston and Green2023; Fayet et al. Reference Fayet, Freeman, Anker-Nilssen, Diamond, Erikstad, Fifield, Fitzsimmons, Hansen, Harris, Jessopp, Kouwenberg, Kress, Mowat, Perrins, Petersen, Petersen, Reiertsen, Robertson, Shannon, Sigurethsson, Shoji, Wanless and Guilford2017). Black-legged kittiwakes and northern fulmars feed at the surface in oceanic waters both during the breeding and nonbreeding periods, and have a more diverse diet, relying primarily on fish (Lemesle et al. Reference Lemesle, Jouanneau, Cherel, Legroux, Ward, Bustamante and Chastel2024; Markones et al. Reference Markones, Guse, Hüppop and Garthe2009; Ojowski et al. Reference Ojowski, Eidtmann, Furness and Garthe2001). In particular, northern fulmars are highly mobile generalist surface feeders that exploit extensively and increasingly fisheries discards, both during and outside breeding (Darby et al. Reference Darby, Dde Grissac, Arneill, Pirotta, Waggitt, Börger, Shepard, Cabot, Owen, Bolton, Edwards, Thompson, Quinn and Jessopp2021; Reference Darby, Clairbaux, Quinn, Thompson, Quinn, Cabot, Strom, Thorarinsson, Kempf and Jessopp2023). Sandwich terns and common gulls are restricted to coastal environments and feed at the surface, both during and outside the breeding period (Garthe and Flore Reference Garthe and Flore2007; Pedersen et al. Reference Pedersen, Fritze and Kharitonov2000). Sandwich terns are fish-specialists (Courtens et al. Reference Courtens, Verstraete, Vanermen, Van de walle and Stienen2017), while common gulls have a generalist diet, with prey captured in marine or terrestrial environments, including during the nonbreeding period (Kubetzki et al. Reference Kubetzki, Garthe and Hüppop1999; Kubetzki and Garthe Reference Kubetzki and Garthe2003). European shags are also restricted to coastal environments and are piscivorous specialists feeding on benthic and demersal fish captured by pursuit-diving, both during and outside breeding (Howells et al. Reference Howells, Burthe, Green, Harris, Newell, Butler, Wanless and Daunt2018; Michelot et al. Reference Michelot, Pinaud, Fortin, Maes, Callard, Leicher and Barbraud2017). The Antarctic seabird species targeted breed in an inland location in Dronning Maud Land, approximately 200 km from the open sea. During breeding, south polar skuas feed exclusively on eggs, chicks, and sometimes adults of the Antarctic petrel and snow petrel Pagodroma nivea, and never forage at sea (Brooke et al. Reference Brooke, Keith and Rov1999; Busdieker et al. Reference Busdieker, Patrick, Trevail and Descamps2020), while they migrate to oceanic waters of the Indian and Atlantic oceans during the non-breeding season (Weimerskirch et al. Reference Weimerskirch, Tarroux, Chastel, Delord, Cherel and Descamps2015). The Antarctic petrel is a pagophilic species foraging in dense pack ice but also open Antarctic waters, feeding primarily on Antarctic krill Euphasia superba (Descamps et al. Reference Descamps, Tarroux, Cherel, Delord, Godo, Kato, Krafft, Lorentsen, Ropert-Coudert, Skaret and Varpe2016; Lorentsen et al. Reference Lorentsen, Klages and Røv1998).
Previous research has demonstrated that diet plays a crucial role in the parasite diversity of birds. Notably, dietary generalist feeding birds have a higher diversity of parasites than dietary specialist birds (Gutiérrez et al. Reference Gutiérrez, Rakhimberdiev, Piersma and Thieltges2017; Thieltges et al. Reference Thieltges, Amundsen, Hechinger, Johnson, Lafferty, Mouritsen, Preston, Reise, Zander and Poulin2013). Our results are partially in agreement with this. Fish-feeding specialists such as the auks were less parasitised than generalist species such as the northern fulmar. However, the generalist common gull was infected with only two parasite taxa. In contrast, European shags, which are fish specialists, had high parasite diversity and prevalence. Parasite diversity has also been shown to vary with habitat, being, for example, greater in coastal/neritic marine mammal hosts than in their oceanic counterparts (Raga et al. Reference Raga, Fernández, Balbuena, Aznar, Würsig and Thewissen2009), because of higher availability and abundance of intermediate hosts at the interface between coastal and terrestrial environments, favouring complex parasite life cycles. Based on our dataset, feeding in coastal or oceanic waters did not seem to be a clear driver of variation in parasite diversity. Instead, oceanic surface-feeding seabirds had comparable parasite diversity to coastal surface-feeding seabirds, as shown by a relatively high Sørensen’s similarity index between these 2 groups. In addition, both oceanic surface-feeders and coastal seabirds had higher parasite diversity than the oceanic diving species (the auks). This suggests that oceanic birds feeding at the surface are more prone to parasite infection than birds that feed deeper in the water column. One possible explanation for this could be the manipulation of intermediate hosts by the parasites themselves. While this behaviour has not been investigated in open-sea marine fish, studies on estuarine and freshwater fish species infected with trematodes have revealed that parasitised fish spend more time at the surface of the water or display more conspicuously, making them more susceptible to predation by birds foraging at the surface (Lafferty & Morris Reference Lafferty and Morris1996; Schwelm et al. Reference Schwelm, Georgieva, Grabner, Kostadinova and Sures2021). The European shag was an exception, being parasite-rich and a diving species. The high parasite diversity of European shags could stem from feeding in coastal benthic environments, which have been shown to be parasite-rich (Hoberg Reference Hoberg1996). In addition, and in contrast to the other bird species, all European shags were juveniles, suggesting a potential age-class effect on parasite richness that should be confirmed by further studies.
Interestingly, several trematode parasites were present in the oceanic black-legged kittiwakes and northern fulmars. This observation is consistent with previous records of Cryptocotyle lingua in these seabird species (Kuklin et al. Reference Kuklin, Kuklina and Ezhov2019; Schwantes Reference Schwantes2023). Although trematode life cycles are typically associated with coastal environments due to the shallow-water distribution of their molluscan intermediate hosts, infection of oceanic seabirds does not necessarily imply recent coastal feeding. Instead, these birds may have acquired infections through the consumption of infected fish acting as second intermediate hosts, many of which are known to migrate from coastal nursery habitats into offshore waters (Duflot et al. Reference Duflot, Cresson, Julien, Chartier, Bourgau, Palomba, Mattiucci, Midelet and Gay2023). Such host movements likely facilitate the transport of metacercariae far from the coast and provide an effective mechanism for offshore transmission of coastal trematode parasites (Marcogliese Reference Marcogliese2007).
The only seabird species that were completely free from helminthic gastrointestinal parasites in the present study were common guillemots and Atlantic puffins, even though these species were previously found to harbour various helminths (Hoberg Reference Hoberg1996; Kuklin et al. Reference Kuklin, Kuklina and Ezhov2025; Muzaffar and Jones Reference Muzaffar and Jones2004; Hoberg and Soudachanh Reference Hoberg and Soudachanh2020; Schwantes Reference Schwantes2023). This probably stems from the small sample size, the body condition of the birds, or other unknown factors. Stranded and bycatch razorbills and common guillemots harboured broadly comparable parasite communities, and no obvious differences in infection patterns were detected between the two groups. This suggests that variation in parasite occurrence may be more strongly influenced by extrinsic factors, such as diet or seasonal effects, rather than intrinsic factors like body condition, although this cannot be conclusively assessed with the present data. Given the limited sample size, these observations should be interpreted with caution. Nevertheless, the results indicate that stranded birds can provide useful material for the assessment of helminth diversity in seabirds.
Finally, it is noteworthy that parasite infection in south polar skuas during the breeding period could originate directly from their prey (Antarctic and snow petrels) through post-cyclic transmission, or, more likely, from ingestion of the petrels’ stomach contents (primarily krill). Snow petrel parasite diversity, and a larger sample size of south polar skuas and Antarctic petrels, should be analysed to unravel this.
Conclusions
Several novel host-parasite associations (n = 9) were reported in the present study, contributing to the knowledge of seabird helminth fauna. The small size and heterogeneous nature of the sample prevented us from statistically testing differences in infection characteristics between groups. Our observation of a higher helminth diversity in oceanic surface-feeding versus diving seabirds merits further investigation, including a larger sample size. This could be achieved by using molecular, non-destructive techniques such as eDNA metabarcoding, and should account for potentially confounding factors such as host sex, age, migratory destinations, season, and geographic region. Nevertheless, this study provides an overview of the distribution of helminth parasites among a subset of understudied seabird species.
Supplementary material
The supplementary material for this article can be found at http://doi.org/10.1017/S0022149X26101163.
Acknowledgements
The authors thank Ewan Wakefield, Joanna Sulich, and Ceslav Czyz for assistance in the field at Svarthamaren and at Troll Station, as well as Frédéric Angelier, Charles-André Bost, Paco Bustamante, Charline Parenteau, and Gauthier Poiriez for support during dissections at the CEBC and LIENSs. Further, the authors are grateful to the French wildlife rescue centres of the Nantes Veterinary School (CVFSE/ONIRIS), the Ille Grande Station (Ligue de Protection des Oiseaux), and the Association ‘Volée de Piafs’ for providing carcasses of birds.
Financial support
Excellence Chair ECOMM funded by the Region Nouvelle Aquitaine (France). The work at Svarthamaren in the 2021/2022 breeding season was funded by the Antarctic Programme of the Norwegian Polar Institute.
Competing interest
The authors declare none.