Introduction
Intramuscular fat (IMF) refers to the adipose tissue situated between skeletal muscle fibers (Liu et al. Reference Liu, Yu and Wei2019). Since its positive influence on determining meat nutritional value, flavor, and juiciness (Lee et al. Reference Lee, Maresh and Kraemer2010; Yu et al. Reference Yu, Ma and Luo2022), IMF content is commonly regarded as an important economic characteristic in livestock production (Font-i-furnols et al. Reference Font-i-furnols, Tous and Esteve-Garcia2012). Growing interest has been sparked in developing effective strategies to promote IMF deposition and subsequently elevate meat quality in animal husbandry. On the contrary, in human, excessive lipid accumulation within skeletal muscle is strongly associated with conditions such as obesity, insulin resistance, type 2 diabetes mellitus, and various metabolic diseases (Gao et al. Reference Gao, Guo and Fan2018; Goodpaster et al. Reference Goodpaster, Theriault and Watkins2000; Prior et al. Reference Prior, Joseph and Brandauer2007). Therefore, comprehending the molecular underpinnings governing lipid metabolism and adipogenesis in skeletal muscle is of great economic and medical value and may yield critical insights for enhancing meat quality or ameliorating metabolic dysregulation.
Dietary interventions are being explored to be an alternative to conventional breeding strategies for modulating IMF deposition recently. N,N,N-trimethylglycine, or normally named betaine, a neutral zwitterionic compound widely abundant in plants, animals, and microorganisms (Dobrijević et al. Reference Dobrijević, Pastor and Nastić2023; Zhao et al. Reference Zhao, He and Wu2018), holds considerable promise as a regulator for various physiological activities (Abd El-Ghany and Babazadeh Reference Abd El-Ghany and Babazadeh2022; Dobrijević et al. Reference Dobrijević, Pastor and Nastić2023; Idriss et al. Reference Idriss, Hu and Sun2017; Kathirvel et al. Reference Kathirvel, Morgan and Nandgiri2010; Li et al. Reference Li, Ge and Xu2015; Veskovic et al. Reference Veskovic, Mladenovic and Milenkovic2019; Zhao et al. Reference Zhao, He and Wu2018). Several animal model-based studies have reported its potential in affecting IMF content (Ma et al. Reference Ma, Yu and Liu2020; Madeira et al. Reference Madeira, Alfaia and Costa2014; Wu et al. Reference Wu, Wang and Xu2018b). However, the role of betaine in this context is still controversial (Madeira et al. Reference Madeira, Alfaia and Costa2015; Wu et al. Reference Wu, Wang and Xu2018b), and the precise mechanisms by which betaine operates necessitate further investigation as well.
N 6-methyladenosine (m6A), the most abundant mRNA modification in eukaryotes, exhibits rapid responsiveness in fine-tuning mRNA metabolism, including mRNA splicing, export, localization, translation, and stability (Chen and Wong Reference Chen and Wong2020; Wu and Wang Reference Wu and Wang2021). Our previous research has characterized a distinct variation in the m6A methylomes between high and low IMF content longissimus dorsi muscle samples (Gong et al. Reference Gong, Gong and Liu2023), implying the involvement of m6A modification in IMF deposition. What’s more, it’s well reported that betaine works as a methyl donor in the methionine cycle, participating in one-carbon metabolism to enhance the formation of S-adenosylmethionine (Wang et al. Reference Wang, Ma and Gong2021). These factors enable us to raise the conjecture of whether epigenetic modification regulation could be one of the mechanisms by which betaine alters IMF deposition.
Here, we aimed to provide a valuable resource from an epigenetics perspective to demonstrate that betaine could be a promising candidate for incorporation into dietary supplements to promote intramuscular adipogenesis, permitting us to better understand how to improve pork quality and offering a potential target for the therapy of metabolic diseases.
Materials and methods
Animal ethic statement
All animal experimental procedures were approved by the Committee on Animal Care and Use and the Committee on the Ethics of Animal Experiments of Zhejiang University (ZJU20240254) and were strictly carried out in accordance with the applicable regulations for animal experiments throughout the entire experimental period.
Animals study
(1) Duroc × Landrace × Yorkshire (DLY) crossed finishing pigs with an average body weight of 90.12 ± 2.20 kg were randomly divided into control group (CTL) and betaine group (BET) with three replicates per group (nine pigs per replicate). Pigs in the CTL were fed a basal diet, while those in the BET were fed a basal diet supplemented with 2500 mg/kg betaine (Table 1) (Fu et al. Reference Fu, Zhang and Chen2023; Turck et al. Reference Turck, Castenmiller and De Henauw2019), with ad libitum access to food and water. The entire experimental period lasted for 45 days (5 days pre-feeding period and 40 days formal test period). We recorded the initial weight, final weight, and daily feed intake to calculate the average daily gain and average daily feed intake. At the end of the feeding, six pigs (close to the average body weight) from each group were selected and slaughtered after fasting for 12 h. Within 20 min of slaughter, about 100 g samples of longissimus thoracis were obtained from the 3rd to 11th ribs on the right-side carcass rapidly, frozen in liquid nitrogen immediately, and subsequently stored at −80°C for further assay.
Ingredients and nutrient levels of experimental diets (%, air-dry basis)

1 Provided the following per kilogram of complete diet: vitamin A, 6400 IU; vitamin B1, 0.6 mg; vitamin B2, 10.4 mg; vitamin D3, 2200 IU; vitamin E, 10 mg; vitamin K3, 2 mg; biotin, 0.15 mg; D-pantothenic acid, 6.4 mg; nicotinic acid, 10 mg; Cu (as copper sulfate), 18 mg; Fe (as ferrous sulfate), 140 mg; Mn (as manganese sulfate), 36 mg; Zn (as zinc sulfate), 72 mg; I (as potassium iodide), 0.06 mg; Se (as sodium selenite), 0.44 mg.
(2) Six-week-old wild-type male C57BL/6J mice (n = 12) were purchased from the Shanghai Model Organisms Center, Inc. (Shanghai, China). They were housed (no more than four individuals per cage) in a pathogen-free animal facility under a 12-hour light/dark cycle, 22 ± 1℃ room temperature, and 55 ± 5% humidity with unrestricted access to food and tap water. Following a 1-week acclimatization period, mice were randomly allocated into two groups: standard laboratory normal chow diet (NCD, Jiangsu Xietong, XTCON50J) and NCD administered with a 2% (w/v) beet alkaline solution (Bet, Sigma-Aldrich, B2629) (Chen et al. Reference Chen, Zhang and Xu2021; Liu et al. Reference Liu, Liu and Chen2024; Lv et al. Reference Lv, Fan and Du2009). On the 42nd day, mice were injected with glycerin into skeletal muscle to induce fat infiltration (Mahdy Reference Mahdy2018; Xu et al. Reference Xu, You and Chen2021) and subsequently sacrificed for sampling 14 days later. Tissues were dissected, weighed, and either soaked in formalin for later morphological analysis or immediately snap-frozen in liquid nitrogen for further analysis.
Primary mouse cell isolation
Mouse primary hepatocytes were isolated from 8-week-old male C57BL/6J mice following two-step-perfusion method as described previously (Charni-Natan and Goldstein Reference Charni-Natan and Goldstein2020). The mice’s abdominal cavity was opened under a painless anesthesia condition. Subsequently, in situ liver perfusion was conducted using HBSS without Ca2+ and Mg2+ (Gibco™, C14175500BT) containing 25 mM HEPES (Sigma-Aldrich, H6903) and 0.5 mM EDTA (Gibco™, R1021), followed by the digestion of liver using HBSS with Ca2+ and Mg2+ (Biological Industries, 02-015-1ACS) containing 25 mM HEPES and 0.85 mg/ml collagenase type II (Gibco™, 17101015). After digestion, the liver was dissected and cells were released and filtered through a 70 μM cell strainer and centrifuged at 1100 g. Isolated primary hepatocytes were resuspended and then plated into 12-well plate coated with rat tail collagen I (Solarbio, C8062) at a density of 2 × 105 per well.
Fibro-adipogenic progenitors (FAPs) were isolated following previously reported protocol with minor adjustments (Kang et al. Reference Kang, Qian and Shi2024). Briefly, tibialis anterior (TA) was excised from 8-week-old male C57BL/6J mice, transferred into precooled phosphate-buffered saline (PBS), carefully removed non-muscle tissue, minced with scissors, and then digested with 1 mg/ml type II collagenase (Gibco™, 17101015) at 37℃ for 60 min. Muscle slurries were filtered through a cell strainer (70 µm) to remove large pieces and then centrifuged at 1100 g for 4 min. After being washed twice with PBS, the cell pellets were incubated with red blood cell lysis buffer (Beyotime Biotechnology, C3702) for 1 min, followed by centrifugation at 1100 g for 4 min. Isolated FAPs were resuspended and then plated into 10 cm dish. Floating cells were removed 4 h later, and fresh culture medium was added.
Cell culture and differentiation
Mouse primary hepatocytes were cultured in Williams E media (Procell Life Science & Technology, PM151211) containing 10% fetal bovine serum (FBS, Gibco™, 10099141), 2 mM glutamine (Procell Life Science & Technology, PB180420), and 1% penicillin-streptomycin (NEST Biotechnology, 211092) in a humidified atmosphere at 37°C with 5% CO2.
FAPs were cultured in Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F12, Gibco™, 11320033) containing 15% FBS (Gibco™, 10099141) and 1% penicillin-streptomycin (Gibco™, 15140122) in a humidified atmosphere at 37°C with 5% CO2. To induce differentiation, after 2 days post-confluence, differentiation of FAPs was induced using an induction medium containing 1 μM dexamethasone (Sigma-Aldrich, D4902), 500 μM IBMX (Sigma-Aldrich, I5879), 5 µg/ml insulin (Solarbio, I8040), and 0.5 μmol/l rosiglitazone (Sigma-Aldrich, PHR2932) for 3 days, followed by a differentiation medium containing 5 µg/ml insulin. Fresh differentiation medium was replaced every 2 days until cells were ready for harvest, typically around day 8.
Cell transfection
For knockdown of Fto, the small interfering RNA (siRNA) was transfected using Lipofectamine RNAiMAX (Invitrogen, 13778030), according to the manufacturer’s instructions. The siRNAs were ordered from GenePharma and the sequences are as follows (5ʹ-3ʹ).
Negative control siRNA: UUCUCCGAACGUGUCACGUTT.
Mouse Fto siRNA: TTAAGGTCCACTTCATCATCGCAGG.
Bodipy staining
Cells were washed twice with PBS and stained with 1 μg/ml Bodipy 493/503 (Invitrogen, D3922) at 37°C in the dark for 30 min. Subsequently, cells were washed three times with PBS, counterstained with Hoechst 33342 (Yeasen, 40732ES03) at 37°C in the dark for 10 min and finally washed three times with PBS again. A fluorescence microscope was used to observe and photograph in a darkened microscopy room.
Quantitative real-time PCR (qPCR)
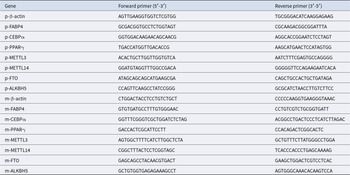
According to the instructions, RNA from tissues or cells was extracted using FreeZol Reagent (Vazyme, R71102) and then reverse transcribed into complementary DNA by SPARKscript II One Step RT-PCR Kit (Shandong Sparkjade Biotechnology Co., Ltd., AG0402). qPCRs were performed on the ABI Step-One PlusTM Real-Time PCR System (Applied Biosystems) using gene-specific primers (Table 2) and SYBR Green Fast qPCR Mix (ABclonal, RK21203). Relative mRNA abundance of gene was calculated by the 2−ΔΔCt method, with β-actin used as an internal control.
Primer sequences used in this study

Western blot
Protein in tissues or cells was extracted using the cell total protein extraction kit (Solarbio, BC3710). Quantification was performed using the bicinchoninic acid protein concentration assay kit (Solarbio, PC0020). Samples were separated on 10% SDS-PAGE gels and blotted onto PVDF membrane (Millipore, IPVH00010). After blocking with 5% skim milk (Solarbio, D8340) at room temperature for 1 h, the membranes were incubated with rabbit anti-fat mass and obesity-associated (FTO) (Proteintech, 27226-1-AP) at 4°C overnight and subsequently incubated with HRP-conjugated secondary antibodies (Biosharp, BL003A) at room temperature for 1 h. The membranes were visualized with Super ECL Plus (US EVERBRIGHT, S6009L).
mRNA isolation and m6A dot blot assays
mRNA was purified from total RNA using two rounds of polyA-tail purification with the Dynabeads® mRNA DIRECT™ kit (Thermo Fisher Scientific, 61012) and quantified with a Qubit Fluorometer (Thermo Fisher Scientific, Q33216).
Dot blot procedure was modified according to previously published researches (Huang et al. Reference Huang, Luo and Zeng2023; Shen et al. Reference Shen, Liang and Yu2017). About 3 μl samples containing mRNA (600 ng) were denatured by heating at 65°C for 5 min to disrupt secondary structures, spotted on Amersham Hybond-N + membrane (GE Healthcare, RPN203B), and then UV crosslinked (twice, 1200 J/cm2). After blocking with 5% skim milk (Solarbio, D8340) at room temperature for 1 h, the membrane was incubated with anti-m6A antibodies (Huabio, HA721152) at 4°C overnight and subsequently incubated with HRP-conjugated secondary antibodies (Biosharp, BL003A) at room temperature for 1 h. The membranes were visualized with Super ECL Plus (US EVERBRIGHT, S6009L). After exposure, the membrane was stained with 0.02% methylene blue dissolved in 0.3 M sodium acetate (Sigma-Aldrich, M9140) to verify the mRNA were loaded equally.
Total triglyceride (TG) detection
The TG levels of mouse TA samples were measured by using commercial kit (Nanjing Jiancheng, A110-1-1) according to the manufacture’s protocol.
Flow cytometry
Cells were harvested and washed with PBS, fixed in 70% cold ethanol, and stored at −20°C overnight (at least 18 h). Fixed cells were washed twice and incubated with 0.5 ml FxCycle™PI/RNase Solution. After incubation, cells were subjected to flow cytometer (Becton Dickinson). Data of cell cycle distribution were analyzed by ModFit LT 5.0 Software (Liao et al. Reference Liao, Liu and Chen2022).
Statistical analysis
Significance between groups was analyzed using unpaired Student’s t-test or one-way ANOVA after testing for homogeneity of variances with Levene’s test using GraphPad Prism 8. All data were presented as the mean ± standard deviation and P < 0.05 was considered statistically significant.
Results
Betaine promotes IMF deposition in vivo
To assess the role of betaine on IMF deposition in vivo, DLY crossbred finishing pigs were randomly assigned to two groups and fed either a basal diet (CTL) or a basal diet supplemented with 2500 mg/kg betaine (BET) for a duration of 40 days (Fig. 1A). Remarkably, the inclusion of betaine in diet dramatically elevated the IMF content in the longissimus dorsi muscle (Fig. 1D), despite comparable average daily weight gain (Fig. 1B) and carcass weight (Fig. 1C). To further determine the promoting effect of betaine on IMF deposition, male C57BL/6J mice were subjected to an NCD or an NCD plus 2% betaine (water supplementation, Bet) (Fig. 1E). As anticipated, dietary betaine treatment didn’t result in any notable impact on body weight (Fig. 1F–H) or daily average food consumption (Fig. 1I) of mice. However, it markedly increased IMF accumulation in the TA muscle of mice, as indicated by Bodipy staining (Fig. 1J). Consistently, the TG content (Fig. 1K) as well as the gene expression of lipid metabolism master regulators including fatty acid binding protein 4 (FABP4), CCAAT enhancer-binding protein alpha (CEBPα), and peroxisome proliferator activated receptor gamma (PPARγ) (Fig. 1L) were significantly upregulated owing to betaine intervention. All these findings demonstrate that betaine can lead to higher IMF deposition without exacerbating body weight in vivo.
Betaine promotes intramuscular fat deposition in vivo. (A) Schematic illustration of the experimental design for administering betaine to DLY pigs. (B) Average daily weight gain of pigs. (C) Carcass weight of pigs. (D) IMF content of longissimus dorsi muscle from pigs. (E) Schematic illustration of the experimental design for administering betaine to C57BL/6J mice. (F) Representative photographs of mice. (G) Dynamic changes in body weight of mice. (H) Relative body weight gain of mice at termination of study. (I) Food intake of mice. (J) Representative Bodipy staining of TA from mice (scale bar = 500 μm). (K) TG content of TA from mice. (L) mRNA expression levels of Fabp4, Cebpα, and Pparγ in TA of mice. *P < 0.05, **P < 0.01, and ***P < 0.001.

Betaine facilitates IMF formation after being metabolized through the liver
Subsequently, porcine intramuscular adipocytes (PIMF) and mouse FAPs were extracted as cellular models for further study in vitro. Nevertheless, it was noteworthy that intervening directly with betaine revealed no discernible impact on the polyester differentiation of PIMF (Fig. 2A) or FAPs (Fig. 2C). The mRNA expression levels of key adipogenic genes also cannot be significantly altered in such situation (Fig. 2B, D). As extensive research established, the liver is a major location for betaine metabolism, and the metabolic activation within the liver plays a vital role in enabling betaine carry out its physiological functions effectively (Ejaz et al. Reference Ejaz, Martinez-Guino and Goldfine2016). In light of this, we treated FAPs with cultural supernatants of betaine-treated hepatocytes (Fig. 2E) and surprisingly observed enhanced the differentiation of polyester into intramuscular adipocytes (Fig. 2F), along with increased mRNA expression levels of Fabp4, Cebpα, and Pparγ (Fig. 2G). Taken together, betaine does not directly facilitate IMF deposition; instead, its influence is manifested post-metabolism in liver. Further in-depth analysis is needed to understand how betaine promotes IMF deposition via liver metabolism.
Betaine facilitates IMF formation after being metabolized through the liver. (A) Representative Bodipy staining of porcine intramuscular adipocytes (PIMF) treated with betaine or without (scale bar = 100 μm). (B) mRNA expression levels of FABP4, CEBPα, and PPARγ in PIMF treated with betaine or without. (C) Representative Bodipy staining of fibro-adipogenic progenitors (FAPs) treated with betaine or without (scale bar = 100 μm). (D) mRNA expression levels of Fabp4, Cebpα, and Pparγ in FAPs treated with betaine or without. (E) Experimental schematic for collecting cultural supernatants of betaine-treated hepatocytes (Bet-Sup) to treat FAPs, with cultural supernatants of betaine-untreated hepatocytes (CTL-Sup) as control. Schematic illustration of the experimental design for administering betaine to C57BL/6J mice. (F) Representative Bodipy staining of FAPs treated with CTL-Sup or Bet-Sup (scale bar = 100 μm). (G) Relative mRNA expression levels of Fabp4, Cebpα, and Pparγ in FAPs treated with CTL-Sup or Bet-Sup. *P < 0.05, **P < 0.01, and ***P < 0.001.

Betaine stimulates the production of NADPH in the liver, thereby elevating IMF content
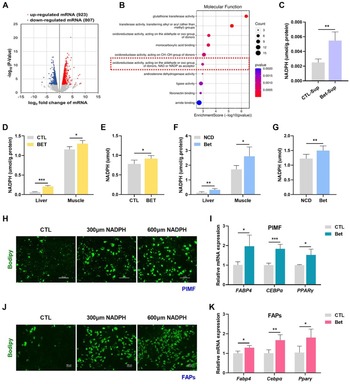
To investigate the alterations in hepatic metabolism following betaine administration, we analyzed the effects of betaine on liver transcriptome of wild-type mice, utilizing previously documented RNA sequencing (RNA-seq) data (Huang et al. Reference Huang, Yu and Pan2021). A total of 1730 genes exhibited significant changes due to betaine supplementation: 923 genes were distinctly upregulated, while 807 genes were notably downregulated in betaine-treated mice compared with the control group (Fig. 3A). Gene Ontology (GO) annotation classification analysis of upregulated genes clearly indicated a notable enrichment in pathways associated with nicotinamide adenine dinucleotide phosphate (NADPH) production (Fig. 3B). And it’s noteworthy that betaine could obviously increase the NADPH content in cultural supernatants of hepatocytes (Fig. 3C). According to this discovery, we postulated that betaine may enhance the production of NADPH in liver, subsequently leading to an increase in IMF accumulation. To explore this possibility, we detected NADPH content and found that betaine treatment obviously elevated the levels of NADPH in liver, serum, and skeletal muscle of both porcine (Fig. 3D, E) and murine subjects (Fig. 3F, G). What’s more, the differentiation of polyester into intramuscular adipocytes was enhanced in a dose-dependent manner with the exogenous addition of varying concentrations of NADPH (0, 300, 600 μM) to PIMF (Fig. 3H) or FAPs (Fig. 3J). The mRNA expression levels of critical adipocyte differentiation markers corroborated this finding (Fig. 3I, K). In summary, the data presented suggest that betaine promotes the formation of IMF by stimulating the production of NADPH.
Betaine stimulates the production of NADPH in the liver, thereby elevating IMF content. (A) Volcano plot displaying differentially expressed genes in the liver transcriptome between control and betaine-treated mice. (B) GO annotation classification analysis of upregulated genes in the liver transcriptome. (C) NADPH content in cultural supernatants of hepatocytes treated with betaine or without. (D) NADPH content in livers and longissimus dorsi muscle of pigs. (E) Serum levels of NADPH in pigs. (F) NADPH content in livers and TA of mice. (G) Serum levels of NADPH in mice. (H) Representative Bodipy staining of PIMF treated with various concentrations of NADPH (scale bar = 100 μm). (I) mRNA expression levels of FABP4, CEBPα, and PPARγ in PIMF treated with NADPH or without. (J) Representative Bodipy staining of FAPs treated with various concentrations of NADPH (scale bar = 100 μm). (K) Relative mRNA expression levels of Fabp4, Cebpα, and Pparγ in FAPs treated with NADPH or without. *P < 0.05, **P < 0.01, and ***P < 0.001.

NADPH promotes the cloning and proliferation process of intramuscular adipocytes
Elucidating the potential mechanisms through which NADPH enhances IMF content is the next question we attempted to decipher. In this regard, according to available RNA-seq data, 1702 genes showed differential expression in FAPs induced by adipogenesis compared with the control group (Fig. 4A). Through Reactome analysis (Fig. 4B), Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis (Fig. 4C), and GO annotation classification analysis (Fig. 4D), we noted that upregulated genes were dramatically enriched in mitotic clonal expansion (MCE)-related processes, such as DNA replication and cell cycle. MCE serves as a critical prerequisite for the differentiation of 3T3-L1 preadipocytes into mature adipocytes, marking it as the most significant event in the early stages of the program (Chang and Kim Reference Chang and Kim2019; Wu et al. Reference Wu, Yao and Jiang2018a). Whether NADPH modulates IMF deposition through regulating the MCE process? We then analyzed the cell cycle distribution of control and betaine-treated PIMF (Fig. 4E, F) or FAPs (Fig. 4G, H) via flow cytometry to substantiate this hypothesis. The results revealed that NADPH could effectively decrease the relative proportion of cells in G0–G1 phase, suggesting that betaine-induced NADPH facilitates the cloning and proliferation process of intramuscular adipocytes to further enhance IMF deposition, whereas, so far, our understanding of the precise mechanisms underlying the promotion of NADPH on the cell cycle process is largely unknown.
NADPH promotes the cloning and proliferation process of intramuscular adipocytes. (A) Volcano plot displaying differentially expressed genes in the transcriptome of mouse intramuscular adipocytes induced by adipogenesis. (B–D) Reactome analysis (B), KEGG analysis (C), and GO annotation classification analysis (D) of differentially expressed genes. (E–F) Flow cytometry analysis of cell cycle progression in PIMF treated with NADPH or without. (G–H) Flow cytometry analysis of cell cycle progression in FAPs treated with NADPH or without.

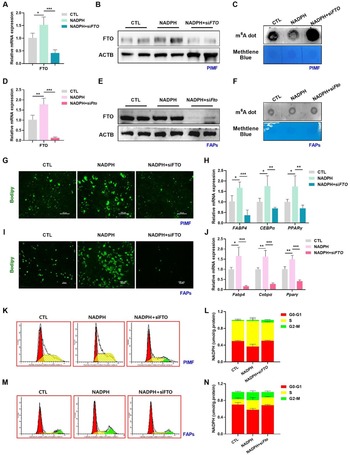
Betaine and NADPH reduce m6A modification via accelerating FTO expression
m6A represents the most widespread and abundant mRNA modification in eukaryotic organisms. It serves as a key regulatory pathway for various biological events, encompassing cell differentiation and metabolic reprogramming (Li et al. Reference Li, Wang and Huang2020). What’s more, our previous work, along with that of others, has provided insight into the crucial role of m6A modification in the processes of adipogenesis in liver and inguinal white adipose tissue (Chen et al. Reference Chen, Wu and Chen2021; Huang et al. Reference Huang, Luo and Zeng2023; Wang et al. Reference Wang, Wu and Liu2020; Wu et al. Reference Wu, Yao and Jiang2018a). These pieces of evidence collectively prompted us to hypothesize whether the promoter function of betaine-induced NADPH on the cell cycle process in intramuscular adipocytes may be linked to m6A methylation as well. To determine this possibility, total m6A-modified mRNA levels were detected using m6A dot blot. Attractively, betaine intervention resulted in a dramatical reduction of overall m6A levels in skeletal muscle, irrespective of whether the subjects were pigs (Fig. 5A) or mice (Fig. 5B). Coinciding with our in vivo data, m6A levels were also lower in PIMF (Fig. 5C) or FAPs (Fig. 5D) following NADPH treatment compared with the control. Owing to the abundance of m6A on mRNA being determined by the dynamic interplay between demethylases (erasers, FTO, and ALKBH5) and methyltransferases (writers, METTL3, and METTL14) enzyme families, we subsequently examined and identified FTO as the key m6A regulator contributes to the mitigated m6A levels (Fig. 5E–H). Intervening directly with betaine didn’t cause any significant alteration on overall m6A levels of PIMF (Fig. 5I, K) or FAPs (Fig. 5J, L). To sum up, betaine and NADPH can downregulate FTO-mediated m6A methylation in vivo and in vitro.
Betaine and NADPH reduce m6A modification via accelerating FTO expression. (A) mRNA m6A modification levels in longissimus dorsi muscle of pigs. Methylene blue staining was used as a loading control. (B) mRNA m6A modification levels in TA of mice. Methylene blue staining was used as a loading control. (C–D) mRNA m6A modification levels in PIMF (C) and FAPs (D) treated with NADPH or without. Methylene blue staining was used as a loading control. (E–F) mRNA expression levels of METTL3, METTL14, FTO, and ALKBH5 in PIMF (E) and FAPs (F) treated with NADPH or without. (G–H) Protein expression levels of FTO in PIMF (G) and FAPs (H) treated with NADPH or without. (I–J) mRNA m6A modification levels in PIMF (I) and FAPs (J) treated with betaine or without. (K–L) mRNA expression levels of METTL3, METTL14, FTO, and ALKBH5 in PIMF (K) and FAPs (L) treated with betaine or without. *P < 0.05, **P < 0.01, and ***P < 0.001.

FTO is essential for NADPH-promoted MCE process and further IMF deposition
So, whether the decreased m6A modification was a contributing factor that further affects the MCE process and ultimately the IMF content? To explore this, we then conducted loss-of-function studies by perturbing FTO to assess the mediator of m6A modification. Quantitative PCR and Western blot analyses demonstrated that silencing of FTO markedly attenuated the elevated mRNA and protein abundance of FTO in NADPH-treated PIMF (Fig. 6A, B) or FAPs (Fig. 6D, E). Consistently, exogenous addition of NADPH had no significant impact on cellular m6A levels with the depletion of FTO (Fig. 6C, F). Regarding the final IMF deposition, it was discovered that interfering with FTO expression effectively impede the role of NADPH in promoting adipogenic differentiation of intramuscular adipocytes (Fig. 6G, I) and significantly reduced the mRNA expression of adipocyte differentiation markers FABP4, C/EBPα, and PPARγ (Fig. 6H, J). Additionally, the NADPH-facilitated cell cycle process of PIMF (Fig. 6K, L) and FAPs (Fig. 6M, N) were also inhibited in the absence of FTO. These observations collectively substantiate that FTO acts as a vital mediator in NADPH-promoted MCE process and subsequent IMF accumulation within intramuscular adipocytes.
FTO is essential for NADPH-promoted MCE process and further IMF deposition. (A) mRNA expression levels of Fto in Fto-silenced or NADPH-treated PIMF. (B) Protein expression levels of FTO in Fto-silenced or NADPH-treated PIMF. (C) mRNA m6A modification levels in Fto-silenced or NADPH-treated PIMF. Methylene blue staining was used as a loading control. (D) mRNA expression levels of Fto in Fto-silenced or NADPH-treated FAPs. (E) Protein expression levels of FTO in Fto-silenced or NADPH-treated FAPs. (F) mRNA m6A modification levels in Fto-silenced or NADPH-treated FAPs. Methylene blue staining was used as a loading control. (G) Representative Bodipy staining of Fto-silenced or NADPH-treated PIMF (scale bar = 100 μm). (H) mRNA expression levels of FABP4, CEBPα, and PPARγ in Fto-silenced or NADPH-treated PIMF. (I) Representative Bodipy staining of Fto-silenced or NADPH-treated FAPs (scale bar = 100 μm). (J) mRNA expression levels of FABP4, CEBPα, and PPARγ in Fto-silenced or NADPH-treated FAPs. (K–L) Flow cytometry analysis of cell cycle progression in Fto-silenced or NADPH-treated PIMF. (M–N) Flow cytometry analysis of cell cycle progression in Fto-silenced or NADPH-treated FAPs. *P < 0.05, **P < 0.01, and ***P < 0.001.

Discussion
Appropriate deposition of IMF is the most vital factor determining pork quality in livestock production and also holds critical medical implications for human health. Betaine, one of non-essential amino acids, has demonstrated considerable potential as a nutritional strategy for regulating intramuscular adipogenesis, although its exact efficacy and underlying mechanisms still remain subjects of ongoing debate and investigation. Here in this study, we clarify the promoting effect of betaine on IMF deposition and further delineate a novel NADPH-m6A-MCE axis that enhances the adipogenic differentiation of intramuscular adipocytes.
Early in 2002, Lawrence et al. reported that feeding diets with 1.25 g/kg supplemental betaine had no statistically significant effect on IMF content in finishing swine (Lawrence et al. Reference Lawrence, Schinckel and Adeola2002), whereas, different from this notion, Albuquerque et al. discovered that long-term 1 g/kg betaine intervention can selectively promote muscle lipogenesis while not affecting other physical and chemical characteristics in pigs (Albuquerque et al. Reference Albuquerque, Neves and Redondeiro2017). Our prior research published in 2018 also indicated that 10 mM betaine markedly facilitates cellular lipid accumulation in C2C12 (C2 mouse myoblast cell line) through inhibition of ERK1/2 signaling on the late adipogenic stage (Wu et al. Reference Wu, Wang and Xu2018b). To thoroughly and comprehensively examine this controversial issue, we undertook new experimental investigations based on porcine, murine, and cellular models, which confirmed the promoting effect of betaine on intramuscular adipogenesis. However, our available findings are not yet able to sufficiently elucidate the discrepancies among existing studies. Differences in experimental models and designs, such as variations in treatment dosage or duration, may serve as potential explanations.
Another important finding of our work, to the best of our knowledge, is the first demonstration showing that betaine cannot directly modulate IMF deposition but rather does so in a liver-dependent manner. Liver is a major site for betaine metabolism, bridging the dynamic cross-talk between betaine and peripheral target tissues. Here we innovatively found that intervening with culture supernatants derived from betaine-treated hepatocytes, rather than the direct use of betaine, results in higher cellular IMF content through regulating pathways associated with NADPH production. How betaine elevates NADPH levels is the upstream regulatory mechanism that is not addressed in this study but merits further in-depth investigation. As for this matter, it is noteworthy that betaine has been recognized as a participant in one-carbon metabolism (Wang et al. Reference Wang, Ma and Gong2021). The folate cycle, which is part of one-carbon metabolism, is an important source of NADPH in mammals, accounting for 40% to its overall production (Fan et al. Reference Fan, Ye and Kamphorst2014). Exploring from this perspective perhaps provide insights into the fundamental mechanisms involved.
Previous publications, including our own, have established that MCE functions as an essential prerequisite for 3T3-L1 preadipocytes to differentiate into mature adipocytes (Chang and Kim Reference Chang and Kim2019; Huang et al. Reference Huang, Luo and Zeng2023; Kim et al. Reference Kim, Kang and Lee2014; Liao et al. Reference Liao, Liu and Chen2022). Considering this, we direct our attention to the cell cycle and present compelling evidence indicating that NADPH can significantly promote the cloning and proliferation process in intramuscular adipocytes to facilitate IMF formation. What’s more, our results substantiate the important function of FTO-mediated m6A modification in this process. CCNA2 and CDK2 are key regulators of early mitotic events and play vital roles in cell cycle regulation and the G1/S phase transition (Tang et al. Reference Tang, Otto and Lane2003). Therefore, subsequent studies also should be conducted to screen out which gene(s) involved in NADPH-altered MCE process.
Conclusion
In summary, we show that betaine enhances intramuscular adipogenesis by promoting MCE process in an NADPH-FTO-m6A coordinated manner. Betaine supplementation elevates NADPH levels in liver, circulation, and skeletal muscle, which subsequently diminishes FTO-mediated m6A modification in intramuscular adipocytes, thereby facilitating the cloning and proliferation of intramuscular adipocytes and the differentiation of polyester in intramuscular adipocytes. Our work expands our understanding of the regulatory function of betaine in IMF deposition and sheds light on the molecular mechanisms underlying its modulation.
Data availability statement
All public RNA profile data analyzed during the study could acquire from the Gene Expression Omnibus (GEO, http://www.ncbi.nlm.nih.gov/geo/). The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
This work was supported by the National Key R&D Program of China (2023YFD1301303), the National Natural Science Foundation of China (U21A20249), the Science and technology innovation leading talent project of Zhejiang Province (2022R52023), and the Science and Technology Major Project of Jinhua, Zhejiang Province (2021-2-011).
Author contributions
Jiaqi Liu, Tongyudan Yang, Yuxi Liu, Youhua Liu, Yidan Zhu, and Chaoqun Huang performed experimental work under the supervision of Xinxia Wang. Jiaqi Liu, Tongyudan Yang, and Yuxi Liu were the main contributors in the conduct of study, data collection and analysis, and data interpretation. Jiaqi Liu and Tongyudan Yang wrote and revised the manuscript. Jiaqi Liu and Tongyudan Yang contributed equally to this work. All the authors have read and approved the published version of the manuscript.
Conflict of interest
The authors declare that they have no competing interests.