1.1 What Use Is Mineral Magnetism?
Before reading or dipping into such a massive tome, it is worth asking the question ‘what use is mineral magnetism?’ I hope that by the end of this chapter, you will be convinced that mineral magnetism is of outstanding importance not only in Earth and planetary science but also in environmental science, biomagnetism, technology, and biomedical science, and that this will stimulate you to continue to read the rest of this book.
1.1.1 Earth Science
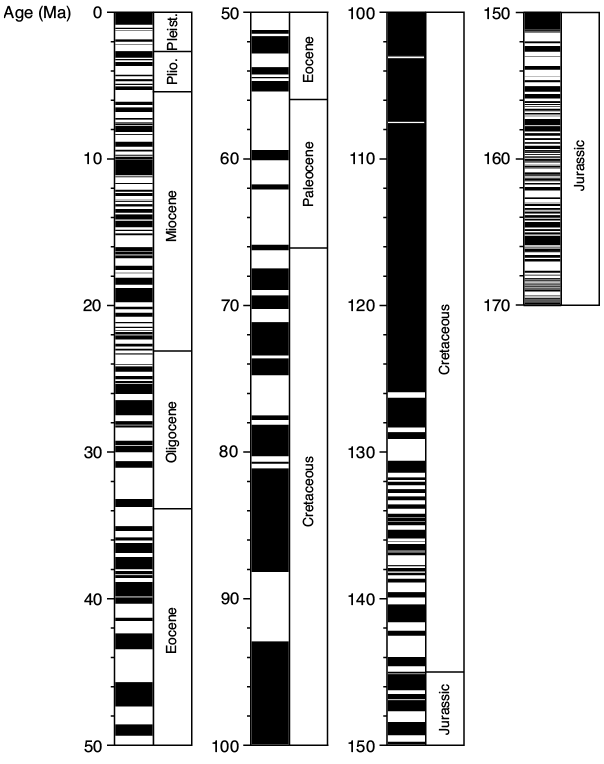
The ability of magnetic rock-forming minerals to record and retain magnetic information for long periods of geological time is fundamental to many areas of enquiry in Earth science. In particular, magnetic minerals provide the basis for the discipline of paleomagnetism, which has made fundamental contributions to understanding the Earth and solar system. For example, paleomagnetism underpins the global plate tectonic paradigm and provides the geomagnetic polarity timescale that is used to calibrate geological time by identification of the pattern of magnetic polarity reversals in geological sequences of sediments or lava flows (Fig. 1.1). Paleomagnetic analysis also provides an understanding of Earth’s magnetic field and the deep-Earth dynamo processes that generate the field and its variations through geological time, including providing knowledge of past geomagnetic polarity reversals, excursions, and secular variations. The geomagnetic field shields Earth from harmful cosmic radiation and early development of the field is argued to have contributed to conditions that allowed evolution of life on, or at least contributed to the habitability of, our planet (e.g., Tarduno et al., Reference Tarduno, Cottrell, Watkeys, Hoffman, Doubrovine, Mamajek, Liu, Sibeck, Neukirch and Usui2010, Reference Tarduno, Blackman and Mamajek2014). The magnetic information carried by minerals not only enables the reconstruction of ancient geomagnetic fields but also the tracking of continental movements, and the dating of rocks. Magnetic fields are ubiquitous in the cosmos, yet Earth remains the only planetary body for which reasonable knowledge of magnetic field history is available through paleomagnetic analysis of rocks that formed at different times in its geological history.

Figure 1.1 Geomagnetic polarity timescale (GPTS) for the last 170 Ma, which is defined largely using the marine magnetic anomaly record. Black = normal polarity; white = reversed polarity. Geological epochs are listed to the right of the GPTS; Pleist. = Pleistocene; Plio. = Pliocene. After Ogg (2012).
Figure 1.1Long description
The figure presents the geomagnetic polarity timescale (GPTS) for the last 170 million years. It displays alternating intervals of normal and reversed magnetic polarity in a vertical bar pattern. Geological epochs, including the Pleistocene and Pliocene, are listed on the right for reference. The timescale is based mainly on marine magnetic anomaly records. Adapted from Ogg (2012).
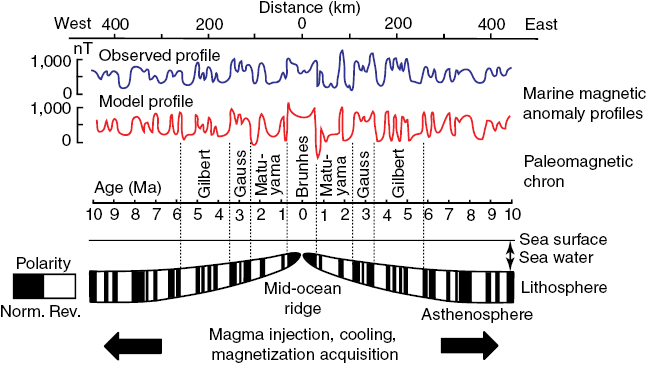
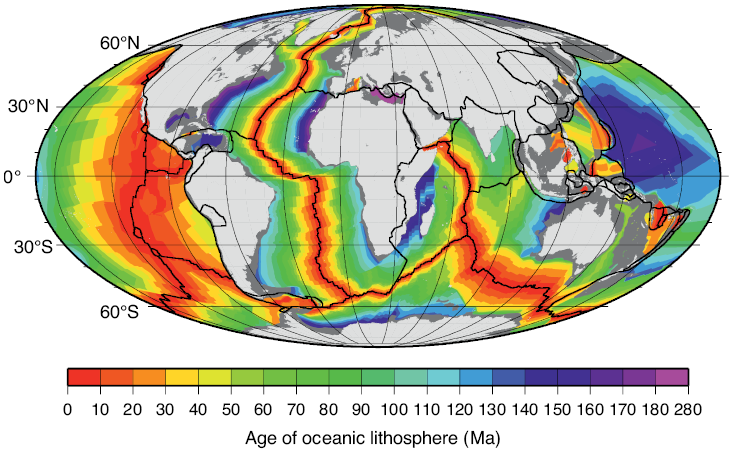
Many rocks record stable paleomagnetic signals that are carried by magnetic rock-forming minerals (e.g., magnetite, hematite, maghemite, goethite, pyrrhotite, and greigite). The reason these minerals record paleomagnetic signals that are so useful in Earth science is that they occur commonly in nature within the ideal stable single domain (SD) size range (for equi-dimensional magnetite (Fe3O4), this range is ~30 to 100 nm). The Nobel laureate, Louis Néel, demonstrated that these homogeneously magnetized particles can retain stable magnetizations for periods exceeding the age of the Earth (Néel, 1949, 1955). Néel theory provides the basis for the widespread usefulness of paleomagnetism in Earth science. For example, ocean crust, which makes up 65% of Earth’s surface (Moores and Twiss, 1995), carries a magnetization that is dominated by the ferrimagnetic mineral titanomagnetite (Fe3−xTixO4). Two principal factors are responsible for the exceptional magnetic recording fidelity of oceanic basalts, which makes them useful for understanding the history of seafloor spreading and of plate tectonics. First, mid-ocean ridge basalt (MORB) cools rapidly enough after eruption that magnetic anomalies are recorded in oceanic crust. The coherent magnetic structure of ocean crust is now understood to be more complex than the sharp block structure of early conceptions (Fig. 1.2), but it is still simple enough to give rise to clear recording of the sequence of geomagnetic polarity reversals over the last ~170 Myr (Fig. 1.1). Second, the titanomagnetite that crystallizes in MORB (and the titanomaghemite to which it often oxidizes) is fine-grained and occurs commonly in the magnetically stable SD state that enables high-quality paleomagnetic recording. Slower cooling that leads to crystallization of coarser particles, or subsequent reheating, would compromise the magnetic recording fidelity of these nanoparticles. The fact that seafloor magnetic anomalies can be so well defined and dated using the geomagnetic polarity timescale means that plate motions can be defined back to the age of the oldest oceanic crust at ~170 Ma (Cande and Kent, 1992, 1995; Müller et al., Reference Müller, Sdrolias, Gaina and Roest2008). Knowledge of plate motions enables quantitative plate reconstruction (Fig. 1.3) and is important for understanding Earth’s geological evolution and in exploring for the resources on which humanity has come to depend. Such knowledge would be much more difficult to obtain if it were not for the magnetic recording capabilities of these nanoparticles. Mineral magnetism, therefore, provides an important foundation for knowledge across Earth science.

(a) Simple block model of uniformly magnetized blocks of ocean crust that give rise to measured marine magnetic anomalies.
Figure 1.2aLong description
Diagram shows a simplified block model of oceanic crust divided into adjacent, uniformly magnetized sections. The alternating magnetization directions of these blocks create variations in the magnetic field measured at the surface, producing the pattern of marine magnetic anomalies.
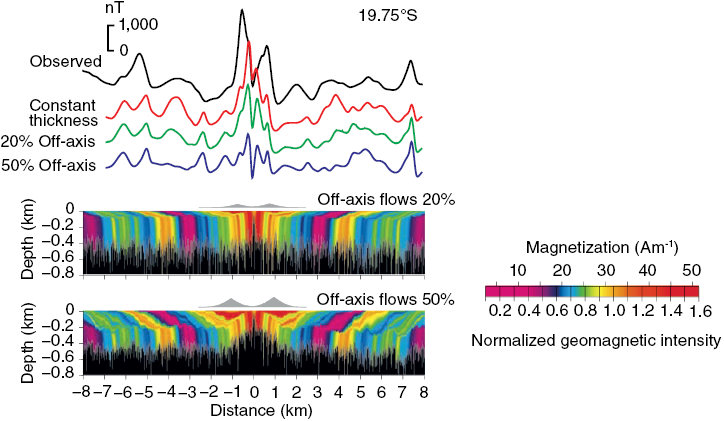
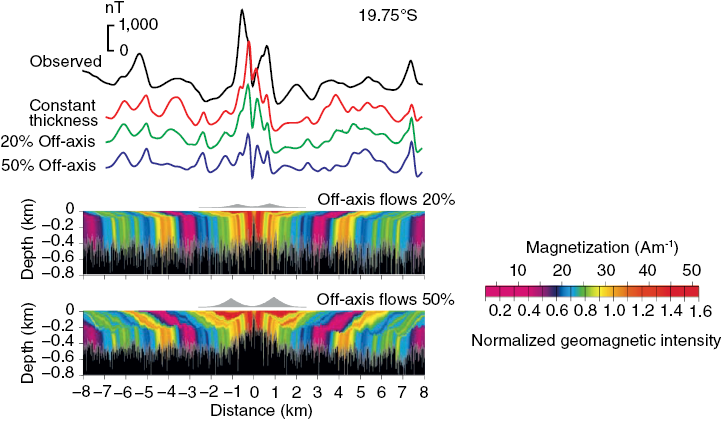
(b) More complex crustal magnetization structures with magnetic anomaly profiles from lava emplacement models compared to profiles for the East Pacific Rise. A bimodal emplacement model was used to simulate off-axis thickening of the extrusive layer as inferred from seismic observations. The upper model has smaller volumetric contributions from off-axis flows that are more consistent with the observed anomaly profile. Lava magnetization is proportional to geomagnetic field intensity. Colours correspond to isochrons in extrusives; dykes are indicated in black. Magnetic anomalies were calculated from multiple strips through the model. The anomaly for a constant thickness source (0.5 km) and magnetization proportional to geomagnetic intensity is shown in red.
Figure 1.2bLong description
Figure shows complex crustal magnetization structures and corresponding magnetic anomaly profiles generated from bimodal lava emplacement models. The models simulate off-axis thickening of the extrusive layer and compare these results with observed profiles from the East Pacific Rise. The upper model uses smaller off-axis flow volumes, aligning better with observations. Isochrons and dykes are indicated, and a reference anomaly from a constant-thickness source is included.
Figure 1.2 Models of ocean crust magnetizations.
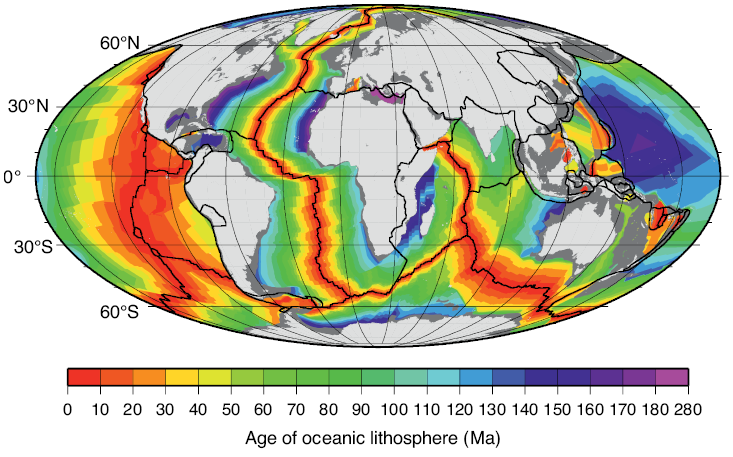
Figure 1.3 Age of ocean lithosphere based on marine magnetic anomalies. Light grey = continents; dark grey = continental margins; black lines = plate boundaries.
Figure 1.3Long description
Figure presents a global map of oceanic lithosphere ages derived from marine magnetic anomalies. Continents and continental margins are outlined, and plate boundaries are marked with lines. The map uses a gradient to indicate relative ages, with younger crust near mid-ocean ridges and progressively older crust toward the continental margins.
1.1.2 Environmental Science
In addition to their capacity to record information about ancient magnetic fields, magnetic minerals are responsive to changing environmental conditions. The sub-discipline of environmental magnetism (Thompson et al., Reference Thompson, Bloemendal, Dearing, Oldfield, Rummery, Stober and Turner1980; Thompson and Oldfield, 1986; Verosub and Roberts, 1995; Maher and Thompson, 1999; Evans and Heller, 2003; Liu et al., Reference Liu, Roberts, Larrasoaña, Banerjee, Guyodo, Tauxe and Oldfield2012) exploits the sensitivity of magnetic minerals to chemical reduction–oxidation reactions at Earth’s surface, which drive transformation of ferrous to ferric iron, and vice versa, in magnetic minerals and facilitates understanding of environmental processes. Wide-ranging processes can be explored using environmental magnetism, including the transportation, deposition, biomineralization, and post-depositional modification of magnetic minerals, which can help to understand the climatic, tectonic, or other driving forces of environmental change. Rather than describing environmental magnetism in detail, a particularly clear example is provided of the use of sediment magnetic properties to better understand climate variability.
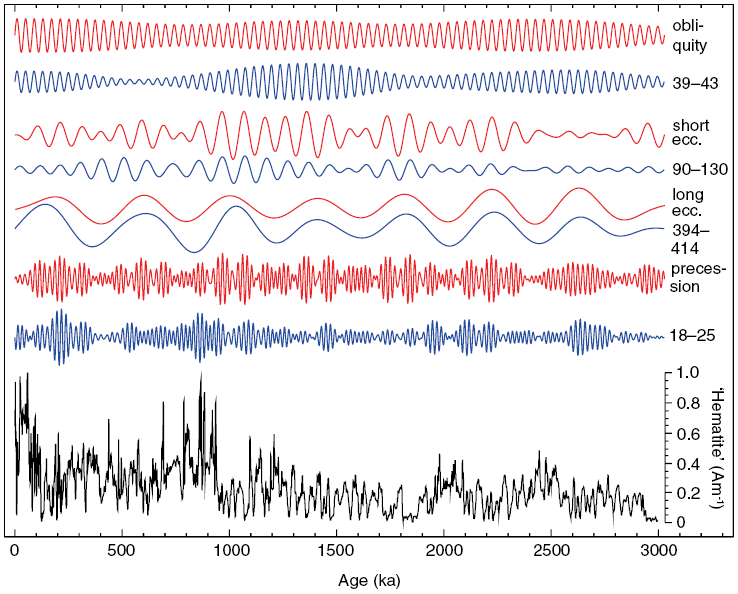
In hyper-arid Saharan environments, hematite ![]() forms via progressive dehydration and oxidation of iron-bearing precursor minerals. Regional aridity changes have driven variations in windblown (eolian) dust delivery from the northeastern Sahara to the Eastern Mediterranean Sea over at least the last 3 million years in response to African monsoon variations, for which hematite has proven to be an excellent proxy. A hematite-based environmental magnetic record of Saharan dust deposition into the Eastern Mediterranean Sea (Fig. 1.4), therefore, provides insights into the long-term functioning of the African monsoon and the history of the Sahara Desert (Larrasoaña et al., Reference Larrasoaña, Roberts, Rohling, Winklhofer and Wehausen2003). Monsoons are controlled by variations in Earth’s orbit that drive cyclical variations in the incident solar radiation (insolation) at Earth’s surface. The Eastern Mediterranean environmental magnetic dust record indicates that long-term African monsoon variations have driven cyclical climatic variations in North Africa, which change from hyper-arid desert conditions to more humid ‘green Sahara’ episodes (e.g., Larrasoaña et al., Reference Larrasoaña, Roberts, Rohling, Winklhofer and Wehausen2003, Reference Larrasoaña, Roberts and Rohling2013). Green Sahara episodes would have provided a landscape that was suitable for habitation and would have opened migration pathways for hominins and other organisms between Africa and Eurasia in contrast to the modern hyper-arid environment, which is a biogeographic barrier (Larrasoaña et al., Reference Larrasoaña, Roberts and Rohling2013). Orbital control of the monsoon is demonstrated when the hematite dust record is subjected to band-pass filtering (Fig. 1.4). Maximum dust production occurs during monsoon minima and vice versa. Dust variations occur in lockstep with Earth’s orbital variations (Fig. 1.4) (Larrasoaña et al., Reference Larrasoaña, Roberts, Rohling, Winklhofer and Wehausen2003). Further examples of the use of mineral magnetism for understanding environmental processes could be cited; such an immediate environmental response to orbital forcing demonstrates that mineral magnetism can provide powerful information about climate variability, monsoons, and desertification as described briefly here.
forms via progressive dehydration and oxidation of iron-bearing precursor minerals. Regional aridity changes have driven variations in windblown (eolian) dust delivery from the northeastern Sahara to the Eastern Mediterranean Sea over at least the last 3 million years in response to African monsoon variations, for which hematite has proven to be an excellent proxy. A hematite-based environmental magnetic record of Saharan dust deposition into the Eastern Mediterranean Sea (Fig. 1.4), therefore, provides insights into the long-term functioning of the African monsoon and the history of the Sahara Desert (Larrasoaña et al., Reference Larrasoaña, Roberts, Rohling, Winklhofer and Wehausen2003). Monsoons are controlled by variations in Earth’s orbit that drive cyclical variations in the incident solar radiation (insolation) at Earth’s surface. The Eastern Mediterranean environmental magnetic dust record indicates that long-term African monsoon variations have driven cyclical climatic variations in North Africa, which change from hyper-arid desert conditions to more humid ‘green Sahara’ episodes (e.g., Larrasoaña et al., Reference Larrasoaña, Roberts, Rohling, Winklhofer and Wehausen2003, Reference Larrasoaña, Roberts and Rohling2013). Green Sahara episodes would have provided a landscape that was suitable for habitation and would have opened migration pathways for hominins and other organisms between Africa and Eurasia in contrast to the modern hyper-arid environment, which is a biogeographic barrier (Larrasoaña et al., Reference Larrasoaña, Roberts and Rohling2013). Orbital control of the monsoon is demonstrated when the hematite dust record is subjected to band-pass filtering (Fig. 1.4). Maximum dust production occurs during monsoon minima and vice versa. Dust variations occur in lockstep with Earth’s orbital variations (Fig. 1.4) (Larrasoaña et al., Reference Larrasoaña, Roberts, Rohling, Winklhofer and Wehausen2003). Further examples of the use of mineral magnetism for understanding environmental processes could be cited; such an immediate environmental response to orbital forcing demonstrates that mineral magnetism can provide powerful information about climate variability, monsoons, and desertification as described briefly here.
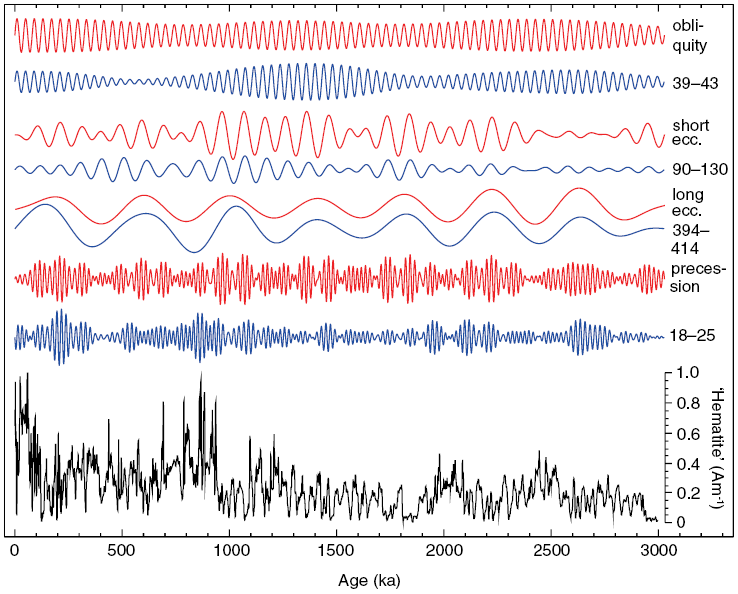
Figure 1.4 Illustration of climate forcing of Saharan dust deposition in marine sediments. The eolian hematite record (bottom, black) with band-pass filtered components of the dust record (blue; period bands are in kyr) from Larrasoaña et al. (Reference Larrasoaña, Roberts, Rohling, Winklhofer and Wehausen2003), which reflects an immediate response to insolation-driven African monsoon forcing at all major orbital periods (red) from Laskar et al. (Reference Laskar, Joutel and Boudin1993); band-pass filtering used the same band as the dust record with normalization by the respective standard deviation. The band-pass-filtered dust signal is scaled identically for all period bands and is multiplied by −1 because of anti-correlated dust production with insolation-driven monsoon response.
1.1.3 Biomagnetism
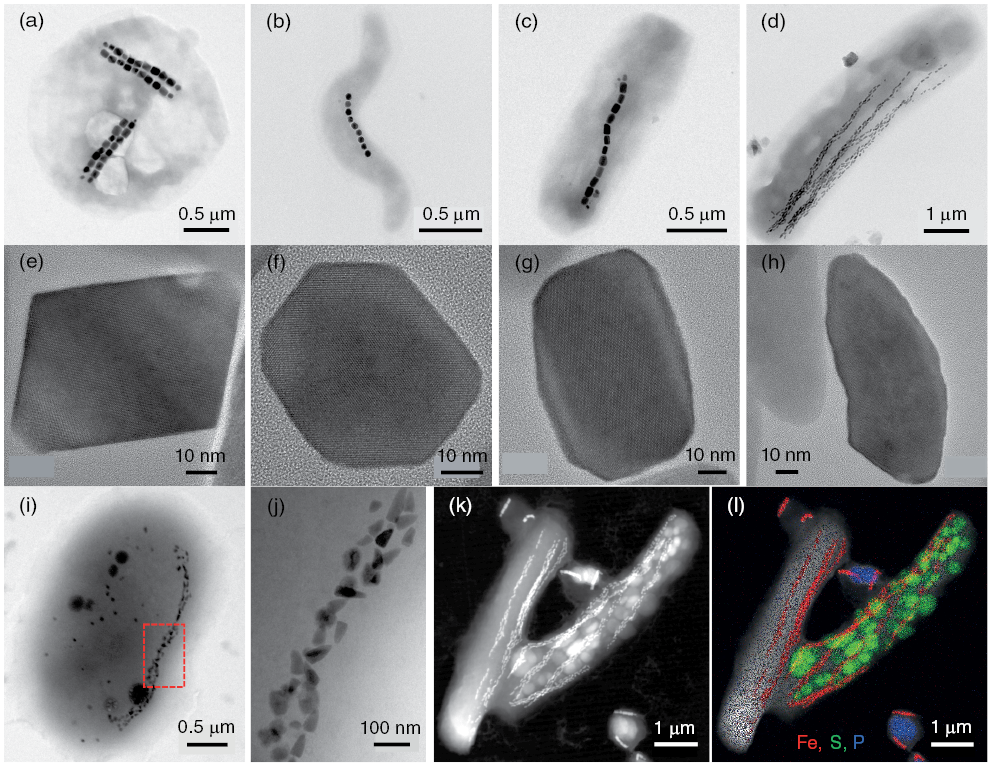
Many organisms biomineralize iron minerals, including magnetic nanoparticles (Frankel and Blakemore, 1991). It is argued that iron biomineral formation serves several purposes, including allowing organisms to accumulate iron for future metabolic needs while avoiding toxic intracellular Fe2+ concentrations, or, for magnetic biominerals, aiding navigation, or allowing exploitation of the hardness or density of these minerals (Frankel, 1991). Bacteria that biomineralize the ferrimagnetic minerals magnetite or greigite (Fe3S4), known as magnetotactic bacteria (Fig. 1.5), use magnetic minerals with highly controlled size, morphology, crystallinity, and stoichiometry to aid geomagnetic navigation. It is argued that geomagnetic navigation (magnetotaxis), along with chemotaxis, reduces the dimensionality of their search for ideal habitats in chemically stratified environments (e.g., Bazylinski and Frankel, 2004; Faivre and Schüler, 2008; Kopp and Kirschvink, 2008; Lefèvre and Bazylinski, 2013). Magnetotactic bacteria have been studied widely by microbiologists who make extensive use of mineral magnetic knowledge to understand the biophysical functions of magnetic biominerals. Likewise, preservation of the fossil remains of magnetotactic bacteria – known as magnetofossils – are of widespread importance for understanding sedimentary paleomagnetic signals (e.g., Kirschvink, 1982; Petersen et al., Reference Petersen, von Dobeneck and Vali1986; Stoltz et al., 1986; Vali et al., Reference Vali, Förster, Amarantidis and Petersen1987; Roberts et al., Reference Roberts, Chang, Heslop, Florindo and Larrasoaña2012). Constraining the nutrient and environmental conditions required for bacterial growth and for subsequent magnetofossil preservation are key to understanding sedimentary paleomagnetic and environmental magnetic records (e.g., Hesse, 1994; Roberts et al., Reference Roberts, Florindo, Villa, Chang, Jovane, Bohaty, Larrasoaña, Heslop and Fitz Gerald2011a; Yamazaki and Ikehara, 2012; Chang et al., Reference Chang, Vasiliev, van Baak, Krijgsman, Dekkers, Roberts, Fitz Gerald, van Hoesel and Winklhofer2014a, Reference Chang, Harrison, Zeng, Berndt, Roberts, Heslop and Zhao2018).
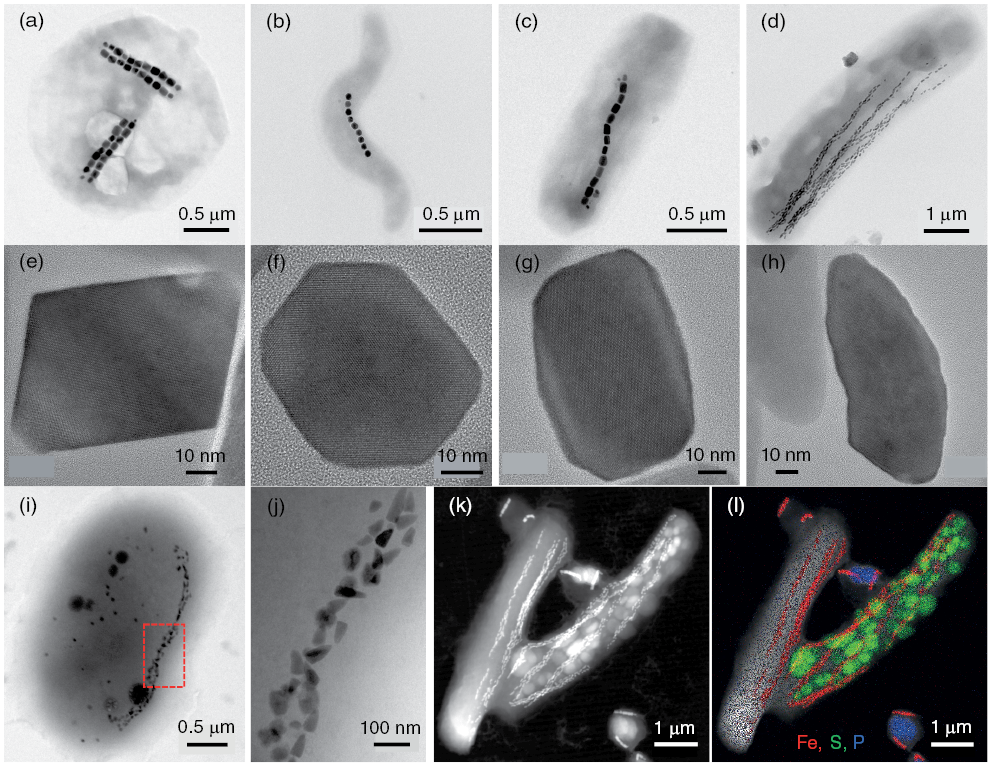
Figure 1.5 Illustration of the diversity of bacteria, chain types, and magnetic mineral morphologies in magnetotactic organisms. Bright-field transmission electron microscope (TEM) images of (a) magnetotactic coccus strain SHHC-1 (Zhang et al., Reference Zhang, Menguy, Wang, Benzerara, Leroy, Liu, Liu, Wang, Pan, Chen and Li2017), (b) magnetotactic spirillum MYS-1 (Li et al., Reference Li, Benzerara, Bernard and Beyssac2013), (c) magnetotactic bacterium strain SHHR-1 (Li et al., Reference Li, Wei, Zuo, Huang, Luo, An, Pelenovich, Mai and Zhang2017), and (d) Candidatus Magnetobacterium casensis strain MYR-1 (Li et al., Reference Li, Menguy, Gatel, Boureau, Snoeck, Patriarche, Leroy and Pan2015). (e–h) High-resolution TEM images of individual magnetite particles produced by (e) SHHC-1, (f) MYS-1, (g) SHHR-1, and (h) MYR-1, respectively. All particles were imaged along the [011] zone axis of magnetite to illustrate the (e) octahedral, (f) cubo-octahedral, (g) elongated prismatic, and (h) bullet-shaped morphologies of the respective magnetic particles. Bright-field TEM images of a rod-shaped bacterium that produces (i) bullet-shaped magnetite and (j) prismatic greigite in the same cell (Wang et al., Reference Wang, Lin, Li and Pan2013). (k) High-angle annular dark-field-scanning TEM (HAADF-STEM) image of two MYR-1 cells and three magnetotactic cocci. (l) HAADF-STEM image with energy-dispersive X-ray elemental mapping of the bacteria in (k) with sulphur-rich granules (green) and polyphosphate inclusions (blue) within magnetotactic bacteria in addition to iron-rich magnetite particles (red) (Li et al., Reference Li, Menguy, Gatel, Boureau, Snoeck, Patriarche, Leroy and Pan2015).
Figure 1.5Long description
Figure presents multiple electron microscopy images illustrating the diversity of magnetotactic bacteria and their magnetic mineral morphologies. Panels (a–d) show four strains with different bacterial shapes. Panels (e–h) display their magnetite crystals, including octahedral, cubo-octahedral, elongated prismatic, and bullet-shaped forms. Panels (i–j) show a rod-shaped bacterium producing two mineral types in one cell. Panels (k–l) present HAADF-STEM images, with elemental mapping revealing sulfur granules, polyphosphate inclusions, and magnetite within cells.
Magnetic biominerals occur in many other organisms, including fish, birds, bats, insects, and molluscs. Magnetite has even been argued to occur in human brain tissue (Kirschvink et al., Reference Kirschvink, Kobayashi-Kirschvink and Woodford1992; Maher et al., Reference Maher, Ahmed, Karloukovski, MacLaren, Foulds, Allsop, Mann, Torres-Jardón and Calderon-Garciduenas2016). The key question in regard to biological magnetic mineral occurrences is whether they are involved in electrophysiological systems that enable an organism to detect magnetic fields, a sense known as magnetoreception (e.g., Wang et al., Reference Wang, Hilburn, Wu, Mizuhara, Couste, Abrahams, Bernstein, Matani, Shimojo and Kirschvink2019). It is beyond the scope of this chapter to explore the biological function of magnetic nanoparticles in different organisms, some of which are controversial, but one example is discussed here. A key issue that remains elusive in biomagnetism is to identify a sensory receptor and magnetic particles that contribute to magnetoreception. For example, Winklhofer et al. (Reference Winklhofer, Holtkamp-Rötzler, Hanzlik, Fleissner and Petersen2001) identified magnetite nanoparticles in homing pigeon beaks. These particles are too small to carry a stable permanent magnetization at ambient temperatures, but they respond clearly to magnetic field variations (for reasons why, see discussion of superparamagnetism in Chapter 3). Arrangement of magnetite nanoparticles adjacent to nerve material, and the fact that the particles respond to field variations, was interpreted to indicate a magnetic field receptor (Winklhofer et al., Reference Winklhofer, Holtkamp-Rötzler, Hanzlik, Fleissner and Petersen2001). Work of this type is necessary to establish the veracity of magnetoreception in organisms and is an active area of biomagnetic research. Wiltschko and Wiltschko (2013) reviewed evidence for avian magnetoreception and found that beak nerves mediate information on magnetic field changes that indicates genuine magnetoreception. Nevertheless, unresolved details remain about magnetoreception mechanisms, and it appears that magnetoreception is only one of multiple factors that contribute to navigational ‘map’ development by birds. The possibility of magnetoreception remains poorly constrained in most organisms.
Biomagnetism is a diverse research field. Detailed knowledge of the magnetic properties of magnetic minerals in organisms is fundamental to understanding their biological uses. Overlaps between the mineral magnetism and biomagnetism literatures are, therefore, significant and biomagnetic research often depends heavily on mineral magnetic information.
1.1.4 Industrial, Technological, and Medical Uses of Magnetic Minerals
While the main applications of the magnetic properties of minerals are in the Earth, planetary, and environmental sciences, and to a lesser extent in the biological sciences, there are important applications of mineral magnetism in industry, technology, and medical science. The magnetic properties of magnetic minerals have been exploited industrially, including through use of maghemite ![]() powders in early magnetic recording media and through ongoing use of finely milled magnetite powders in suspension in coal washing plants where magnetic properties are exploited to magnetically separate the magnetite for reuse. Ferrimagnetic semi-metallic minerals such as greigite have also been proposed to be useful in spintronics (e.g., Devey et al., Reference Devey, Grau-Crespo and de Leeuw2009), where giant magnetoresistance can be produced in alternating thin films of magnetic and non-magnetic layers with magnetization alignments that change from parallel to antiparallel to make the structure effectively immune to external magnetic fields (Wolf et al., Reference Wolf, Awschalom, Buhrman, Daughton, von Molnár, Roukes, Chtchelkanova and Treger2001). Interaction between electron spins and the magnetic properties of the material are exploited in such applications to develop improved semiconductor devices. In other cases, potential industrial uses of magnetic minerals such as in photovoltaic cells can be assessed effectively where the magnetic, electronic, and structural properties of minerals are well known (e.g., Zhang et al., Reference Zhang, Manno, Baruth, Johnson, Aydil and Leighton2013). A detailed understanding of the magnetic properties of minerals is, therefore, useful in such fields.
powders in early magnetic recording media and through ongoing use of finely milled magnetite powders in suspension in coal washing plants where magnetic properties are exploited to magnetically separate the magnetite for reuse. Ferrimagnetic semi-metallic minerals such as greigite have also been proposed to be useful in spintronics (e.g., Devey et al., Reference Devey, Grau-Crespo and de Leeuw2009), where giant magnetoresistance can be produced in alternating thin films of magnetic and non-magnetic layers with magnetization alignments that change from parallel to antiparallel to make the structure effectively immune to external magnetic fields (Wolf et al., Reference Wolf, Awschalom, Buhrman, Daughton, von Molnár, Roukes, Chtchelkanova and Treger2001). Interaction between electron spins and the magnetic properties of the material are exploited in such applications to develop improved semiconductor devices. In other cases, potential industrial uses of magnetic minerals such as in photovoltaic cells can be assessed effectively where the magnetic, electronic, and structural properties of minerals are well known (e.g., Zhang et al., Reference Zhang, Manno, Baruth, Johnson, Aydil and Leighton2013). A detailed understanding of the magnetic properties of minerals is, therefore, useful in such fields.
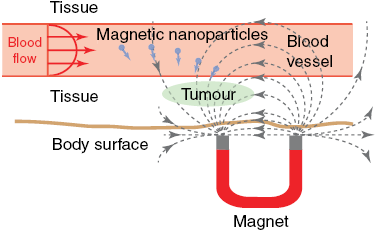
The magnetic properties of minerals are exploited in biomedical science and medical imaging applications. Magnetic nanoparticles are used as contrast agents for magnetic resonance imaging (MRI) and as carriers for drug delivery to specific sites in vivo (e.g., Pankhurst et al., Reference Pankhurst, Connolly, Jones and Dobson2003; Dobson, 2006; McBain et al., Reference McBain, Yiu and Dobson2008; Sun et al., Reference Sun, Lee and Zhang2008; Veiseh et al., Reference Veiseh, Gunn and Zhang2010). The principles by which magnetic nanoparticles are used for both purposes are simple and identical. Their small size means that they can penetrate human tissue and be directed by focussed application of magnetic fields to specific sites where they can interact with tissue and function at cellular and molecular levels (Fig. 1.6a). The superparamagnetic nanoparticles used in medical applications have diameters of tens of nanometres. Such particles are too small to retain a permanent magnetization, but, unlike paramagnetic particles, they have a strong magnetization when placed within a magnetic field (see Chapter 3 for explanations of these terms). These properties facilitate manipulation of particles by application of external magnetic fields to deliver them to sites of interest. When a field is removed, the magnetization is lost so that the particles do not aggregate as larger stable SD particles would.

(a) A colloidal suspension including drug-bearing magnetic nanoparticles is injected into the blood stream and directed to pathological tissue using focussed magnetic fields.
Figure 1.6aLong description
Diagram illustrates targeted delivery of magnetic nanoparticles carrying drugs. After injection into the bloodstream, nanoparticles travel with blood flow toward a tumor or other pathological tissue. Focused magnetic field, generated by a magnet beneath the body surface, directs magnetic nanoparticles to accumulate at the targeted site for localized treatment.
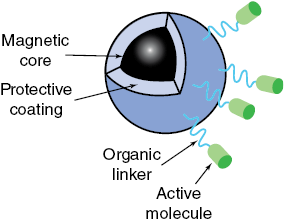
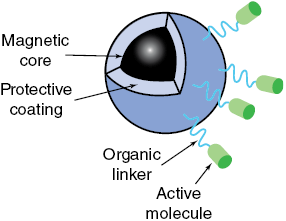
(b) Schematic detail of a magnetic nanoparticle designed for drug delivery. Magnetic nanoparticles have high surface area to volume ratios and are reactive, so they are covered with protective coatings, including polymers, silica or gold. Active therapeutic biomolecules are bound to the protective coating with organic linkers. See text for discussion.
Figure 1.6bLong description
Diagram shows a magnetic nanoparticle used for drug delivery. Magnetic core is coated with protective layers, including polymers, silica, or gold, to enhance stability and reactivity. Organic linkers connect active therapeutic biomolecules to the coating, enabling targeted delivery and controlled release within the body.
Figure 1.6 Schematic representation of the use of magnetic nanoparticles in therapeutic drug delivery to targeted sites in vivo.
Once magnetic nanoparticles are delivered to a desired location, different properties are exploited for imaging versus drug delivery. Non-invasive imaging with MRI makes use of proton precession around an applied magnetic field. When a pulsed field is applied, protons in body fluids are perturbed and then relax to their original state. Local variations in relaxation produce image contrasts that arise from proton density and from the chemical and physical nature of the surrounding tissue. Accumulation of magnetic nanoparticles within tissue provides an image contrast enhancement because the particles produce a nanoscopic field gradient when the field is applied, to which protons in the local vicinity respond directly. Magnetic nanoparticles are, therefore, useful contrast agents in MRI applications. Clinical imaging of cancerous tumours and metastases is capable of distinguishing lesions as small as 2–3 mm (Sun et al., Reference Sun, Lee and Zhang2008). Improving imaging technology using magnetic nanoparticles is an active research and development topic for other diagnostic purposes, including detection, diagnosis, and treatment of malignant tumours, cardiovascular disease, and neurological disease.
Drug delivery using magnetic nanoparticles provides a potentially transformational therapeutic development because it can overcome the side effects produced by non-specific chemical therapies on healthy tissue by delivering therapeutic agents directly to pathological tissue. The aim of direct delivery is to attach cytotoxic drugs or radionuclides to protective coatings that surround magnetic nanoparticles using an organic linker (Fig. 1.6b), place the particles into a colloidal suspension, and inject the suspension into veins or arteries, and guide the particles to a specific tumour site through the blood stream of the patient using high-gradient external magnetic fields (Fig. 1.6a). The drug can then be released from the magnetic carrier through various mechanisms, including enzymatic activity, changes in pH, osmosis, or temperature, to increase uptake of the drug by tumour cells at the target site. Application of alternating fields to magnetic nanoparticles can also elevate their temperature, which makes them hyperthermia agents that deliver toxic levels of thermal energy to tumours. Moderate degrees of tissue warming in the presence of chemotherapy or radiotherapy agents can also result in more effective destruction of malignant cells (Pankhurst et al., Reference Pankhurst, Connolly, Jones and Dobson2003). A further desirable property of iron oxide nanoparticles, including magnetite and maghemite, for biomedical applications is that they are biodegradable. Once metabolized, the solubilized iron is added to the body’s iron stores and is eventually incorporated into haemoglobin.
Use of magnetic nanoparticles in biomedical applications has enormous potential and is being advanced through improved nanotechnology, molecular cell biology, and imaging instrumentation. Nevertheless, significant challenges exist in up-scaling treatments from small animals in laboratory experiments to human subjects. In considering such applications of mineral magnetism, it should not be forgotten that some important magnetic parameters remain poorly constrained. For example, few precise estimates exist of the pre-exponential frequency factor (Néel, 1949) associated with the thermal relaxation process in superparamagnetic particles (e.g., Moskowitz et al., Reference Moskowitz, Frankel, Walton, Dickson, Wong, Douglas and Mann1997; Cao et al., Reference Cao, Tian, Liu, Liu, Chen and Pan2010; Berndt et al., Reference Berndt, Muxworthy and Paterson2015). Thus, important mineral magnetic research remains to be done that will be potentially useful in biotechnological applications.
1.2 Purpose of This Book
From the brief survey in Section 1.1, I hope that readers are convinced that the magnetic properties of rock-forming minerals are important in diverse applications in the Earth, planetary, environmental, biological, materials, and biomedical sciences. Rigorous understanding of the magnetic structure and mineral magnetic properties of magnetic minerals is needed to underpin interpretation of mineral magnetic signals in most relevant applications. Even in the cursory introductory treatment provided here, it should be clear that mineralogy, particle size, thermal history, and other factors are important for enabling magnetic particles to record useful magnetic information. The aim of this book is to provide an essential treatment of these details in a single source for each of the major terrestrial rock-forming magnetic minerals to help practitioners to access important information that is currently scattered across a large and diverse literature. By bringing a coherent structure to the presentation of this information, it is hoped that readers will be able to assess rapidly the key information and identify knowledge gaps where further research is needed.
Given the importance of magnetic minerals in diverse fields, it may be surprising that a comprehensive treatment of mineral magnetism has yet to be published. While important texts have been written on rock and environmental magnetism (e.g., O’Reilly, 1984; Dunlop and Özdemir, 1997; Maher and Thompson, 1999; Evans and Heller, 2003), none provides a comprehensive presentation of the mineral magnetic properties for the range of magnetic minerals discussed here. In the modern international research environment where diverse studies are conducted on numerous magnetic minerals, it is increasingly important for such work to rest on a solid foundation. Lack of knowledge of important magnetic parameters in this context is just as important as what is known. A key aim of the systematic treatment provided in this book is to point to major knowledge gaps in relation to the mineral magnetic properties of each mineral discussed so that concerted future effort can address these gaps to build an ever more secure foundation for interpretations that hinge on mineral magnetic knowledge.
1.3 Structure and Scope of This Book
Mineral Magnetism has three parts. In Part I, foundational issues are outlined, including the broader importance of mineral magnetism (Chapter 1), key aspects of mineralogy as it relates to magnetic structures and the recording of magnetic information (Chapter 2), and the fundamentals of magnetism and the underlying physics of how minerals record magnetic information (Chapter 3). In Chapter 4, key magnetic and mineralogical techniques that are used to determine the most important intrinsic magnetic properties of minerals are described. This treatment should help readers to understand what is required to determine such properties.
Details of the magnetic properties of the major terrestrial rock-forming magnetic minerals are presented in Part II of Mineral Magnetism. These minerals include the iron oxides (Chapters 5–10 and 14), the iron oxyhydroxides (Chapters 11 and 14), and the iron sulphides (Chapters 12–14). The aim for each mineral is to present key information, and a guide to the primary literature, without providing an exhaustive treatise. Even with this aim, individual chapters in Part II are vastly longer than any treatment of the magnetism of these minerals in existing texts.
Chapters in Part II of Mineral Magnetism focus on terrestrial rock-forming minerals. The magnetism of extraterrestrial bodies is the subject of intensive modern investigation through analysis of meteoritic material. While common terrestrial iron oxide minerals such as magnetite and titanomagnetite, and sometimes hematite on the surface of Mars (e.g., Rochette et al., Reference Rochette, Gattacceca, Chevrier, Mathé and Menvielle2006; Jiang et al., Reference Jiang, Liu, Roberts, Dekkers, Barrón, Torrent and Li2022), and pyrrhotite (e.g., Rochette et al., Reference Rochette, Gattacceca, Bonal, Bourot-Denise, Chevrier, Clerc, Consolmagno, Folco, Gounelle, Kohout, Pesonen, Quirico, Sagnotti and Skripnik2008), have been reported from extraterrestrial materials, the oxidation state of most extraterrestrial parent bodies generally did not enable formation and preservation of pure Fe3+-bearing phases and the mixed valence magnetite is generally the most oxidized phase present (Rochette et al., Reference Rochette, Weiss and Gattacceca2009). As is the case on Earth, titanomagnetite also occurs in the extraterrestrial equivalent of basaltic rocks (angrites) and in Martian meteorites (Rochette et al., Reference Rochette, Gattacceca, Chevrier, Mathé and Menvielle2006, Reference Rochette, Gattacceca, Bonal, Bourot-Denise, Chevrier, Clerc, Consolmagno, Folco, Gounelle, Kohout, Pesonen, Quirico, Sagnotti and Skripnik2008, Reference Rochette, Weiss and Gattacceca2009; Weiss et al., Reference Weiss, Berdahl, Elkins-Tanton, Stanley, Lima and Carporzen2008). Overall, however, magnetic signals in extraterrestrial materials tend to be carried by a diverse range of magnetic minerals that are not present in crustal terrestrial rocks (e.g., metallic iron, or iron–nickel alloys, such as kamacite, taenite, tetrataenite, and awaruite). In the most chemically reduced meteorites, magnetic minerals with poorly understood magnetic properties have been reported, such as suessite ((Fe, Ni)3Si), schreibersite ((Fe, Ni)3P), and cohenite ((Fe, Ni)3C). Some extraterrestrial magnetic minerals have been well studied, but the state of knowledge of many of these minerals is generally at a much lower level than for the terrestrial rock-forming minerals discussed in this book. While extraterrestrial magnetic minerals are becoming of increasing interest, they are beyond the scope of the present book. Interested readers are directed to the following relatively recent papers that either present results for extraterrestrial magnetic minerals or that contain valuable reviews that point to the relevant literature (e.g., Wasilewski et al., Reference Wasilewski, Acuña and Kletetschka2002; Gattacceca et al., Reference Gattacceca, Rochette and Bourot-Denise2003; Rochette et al., Reference Rochette, Gattacceca, Chevrier, Mathé and Menvielle2006, Reference Rochette, Gattacceca, Bonal, Bourot-Denise, Chevrier, Clerc, Consolmagno, Folco, Gounelle, Kohout, Pesonen, Quirico, Sagnotti and Skripnik2008, Reference Rochette, Weiss and Gattacceca2009; Acton et al., Reference Acton, Yin, Verosub, Jovane, Roth, Jacobsen and Ebel2007; Weiss et al., Reference Weiss, Berdahl, Elkins-Tanton, Stanley, Lima and Carporzen2008, Reference Weiss, Gattacceca, Stanley, Rochette and Christensen2010; Lappe et al., Reference Lappe, Church, Kasama, Bastos da Silva Fanta, Bromiley, Dunin-Borkowski, Feinberg, Russell and Harrison2011, Reference Lappe, Feinberg, Muxworthy and Harrison2013; Bryson et al., Reference Bryson, Herrero-Albillos, Kronast, Ghidini, Redfern, van der Laan and Harrison2014; Gattacceca et al., Reference Gattacceca, Suavet, Rochette, Weiss, Winklhofer, Uehara and Friedrich2014a; Einsle et al., Reference Einsle, Harrison, Kasama, Conbhui, Fabian, Williams, Woodland, Fu, Weiss and Midgley2016, Reference Einsle, Eggeman, Martineau, Saghi, Collins, Blukis, Bagot, Midgley and Harrison2018; Nichols et al., Reference Nichols, Bryson, Herrero-Albillos, Kronast, Nimmo and Harrison2016, Reference Nichols, Bryson, Blukis, Herrero-Albillos, Kronast, Rüffer, Chumakov and Harrison2020; Strauss et al., Reference Strauss, Feinberg and Johnson2016; Harrison et al., Reference Harrison, Bryson, Nicholls, Weiss, Elkins-Tanton and Weiss2017; Shah et al., Reference Shah, Williams, Almeida, Nagy, Muxworthy, Kovács, Valdez-Grijalva, Fabian, Russell, Genge and Dunin-Borkowski2018; Fu et al., Reference Fu, Kehayias, Weiss, Schrader, Bai and Simon2020).
Part III of Mineral Magnetism contains a single chapter. Chapter 15 is a synthesis of the final part of each chapter in Part II of Mineral Magnetism that lists important magnetic parameters that remain to be determined robustly. The aim of this final chapter is to stimulate development of future PhD and other systematic research projects that will place the field of mineral magnetism onto a more mature footing. When many of the unknown parameters outlined are determined precisely, it will be time to write a second, and much more definitive, edition of this book.
Explaining and describing the subject of mineral magnetism involves use of many parameters and a veritable alphabet soup of abbreviations. To avoid repetition, these parameters and abbreviations are not redefined in each chapter. To help readers avoid constant backward checking to seek definitions, symbols used in the text are listed and defined in Appendix 1 (alphabetical followed by Greek symbols), and abbreviations used in the text are defined in Appendix 2.