Introduction
Chile, a country renowned for its unique biogeographic isolation within South America, exhibits the highest level of generic endemism in the region and ranks among the highest globally (Moreira-Muñoz, Reference Moreira-Muñoz2011; Urbina-Casanova et al., Reference Urbina-Casanova, Saldivia and Scherson2015; Ramírez-Verdugo et al., Reference Ramírez-Verdugo, Tapia, Forest and Scherson2023). This exceptional endemism is primarily concentrated in the flora of central Chile, a region increasingly subjected to anthropogenic pressures on its natural resources (Miranda et al., Reference Miranda, Altamirano, Cayuela, Lara and González2017; Otavo and Echeverría, Reference Otavo and Echeverría2017; Noh et al., Reference Noh, Echeverría, Pauchard and Cuenca2019; Benavidez-Silva et al., Reference Benavidez-Silva, Jensen and Pliscoff2021; Braun et al., Reference Braun, Faßnacht, Valencia and Sepulveda2021). As a result, these ecosystems have been designated as one of the 35 global biodiversity hotspots, specifically the ‘Chilean Winter Rainfall-Valdivian Forest’ (Myers et al., Reference Myers, Mittermeier, Mittermeier, Da Fonseca and Kent2000; Arroyo et al., Reference Arroyo, Marquet, Marticorena, Simonetti, Cavieres, Squeo and Rozzi2004), making them a global conservation priority (Mittermeier et al., Reference Mittermeier, Turner, Larsen, Brooks, Gascon, Zachos and Habel2011). However, ongoing human activities have led to Chile being among the Latin American countries with the highest deforestation rates (Armenteras et al., Reference Armenteras, Espelta, Rodríguez and Retana2017). Consequently, both the hotspot and its rich biodiversity are under severe threat, with numerous endemic plant species currently facing varying degrees of extinction risk (Smith-Ramírez, Reference Smith-Ramírez2004; Otavo and Echeverría, Reference Otavo and Echeverría2017; Noh et al., Reference Noh, Echeverría, Pauchard and Cuenca2019). This scenario highlights the urgent need to explore alternative conservation strategies that extend beyond the boundaries of currently endangered areas.
As a complement to in situ conservation, ex situ strategies play a vital role in safeguarding threatened biodiversity outside its natural habitats (Cavender et al., Reference Cavender, Westwood, Bechtoldt, Donnelly, Oldfield, Gardner, Rae and McNamara2015). For spermatophytes, seeds are the preferred germplasm for collection due to their completeness as an organism and the cost-effectiveness of conserving genetic diversity – a process known as seed banking (Roberts, Reference Roberts1991; Jaisankar et al., Reference Jaisankar, Velmurugan, Sivaperuman, Sivaperuman, Velmurugan, Singh and Jainkar2018; Malhotra et al., Reference Malhotra, Panatu, Singh, Negi, Singh, Singh, Chandora and Singh2019; Walters and Pence, Reference Walters and Pence2021). However, not all species tolerate the seed banking process, which involves drying and storing seeds under cold conditions (O’Donnell and Sharrock, Reference O’Donnell and Sharrock2017). Species with seeds sensitive to desiccation or chilling – known as recalcitrant – pose significant challenges for genetic conservation in seed banks (Walters et al., Reference Walters, Berjak, Pammenter, Kennedy and Raven2013).
Although estimates vary depending on habitat and taxonomy (Dickie and Pritchard, Reference Dickie, Pritchard, Black and Pritchard2002; Tweddle et al., Reference Tweddle, Dickie, Baskin and Baskin2003; Wyse and Dickie, Reference Wyse and Dickie2017), it is generally accepted that approximately 20–30% of tree species worldwide produce recalcitrant seeds, making them the group with the highest incidence of this trait (Wyse et al., Reference Wyse, Dickie and Willis2018). In Chile, León-Lobos et al. (Reference León-Lobos, Sandoval, Bolados, Rosas, Stark and Gold2014) estimated that approximately 26% of native tree genera may produce recalcitrant seeds, suggesting limited suitability for long-term conservation through conventional seed banking. Their assessment, based primarily on expert knowledge and general seed biological traits, provided an important first approximation but highlighted the need for empirical validation and more refined predictive tools. Of the native tree species whose conservation status has been assessed by the Chilean Ministry of the Environment as of May 2024, 89% are classified as Vulnerable, Endangered or Critically Endangered (Ministerio del Medio Ambiente de Chile, 2024). Thus, seed recalcitrance and the limitations of conventional seed banking disproportionately affect the most threatened components of Chile’s native flora.
Despite its relevance to ex situ conservation, seed recalcitrance has been formally evaluated in only a limited number of plant species worldwide, leaving a critical knowledge gap regarding which rare, endemic or economically important species are unsuitable for seed banking (Lan et al., Reference Lan, Xia, Wang, Liu, Zhao and Tan2014; Wyse et al., Reference Wyse, Dickie and Willis2018). In Chile, apart from studies on Araucaria araucana (Molina) K. Koch (Tompsett, Reference Tompsett1984) and several Nothofagus species (León-Lobos and Ellis, Reference León-Lobos and Ellis2005), no formal assessments exist regarding seed storage behaviour or desiccation sensitivity in native trees. While some propagation studies infer seed storage behaviour secondarily, they often lack details on storage conditions, moisture content or seed viability before and after storage.
Fortunately, recalcitrant seeds are known to share specific physiological, morphological and ecological traits (Chin et al., Reference Chin, Krishnapillay and Stanwood1989; Farrant et al., Reference Farrant, Pammenter and Berjak1989; Hong et al., Reference Hong, Linington and Ellis1996; Tweddle et al., Reference Tweddle, Dickie, Baskin and Baskin2003; Daws et al., Reference Daws, Garwood and Pritchard2005; Probert et al., Reference Probert, Daws and Hay2009; Moothoo-Padayachie et al., Reference Moothoo-Padayachie, Varghese, Pammenter, Govender and Sershen2016; Wyse and Dickie, Reference Wyse and Dickie2018). These traits have been used to identify species likely to exhibit recalcitrant seed behaviour. While experimental determination of seed storage behaviour can be time-consuming and resource-intensive, predictive models based on correlated variables offer a viable alternative. Numerous studies have explored the relationship between seed traits, environmental variables and desiccation sensitivity across ecosystems. Daws et al. (Reference Daws, Garwood and Pritchard2006) identified seed mass (SM) and seed coat ratio (SCR) as key predictors in tropical forest species, with their model demonstrating effectiveness across continents, biomes and vegetation types. Subsequent research (Probert et al., Reference Probert, Daws and Hay2009; Hamilton et al., Reference Hamilton, Offord, Cuneo and Deseo2013; Pelissari et al., Reference Pelissari, José, Fontes, Matos, Pereira and Faria2018) confirmed the relevance of these and other traits – such as seed moisture content, embryo type and oil content – especially when used in combination.
More sophisticated predictive approaches, such as those developed by Wyse and Dickie (Reference Wyse and Dickie2018) and Mattana et al. (Reference Mattana, Peguero, Di Sacco, Agramonte, Encarnación Castillo, Jiménez, Clase, Pritchard, Gómez-Barreiro, Castillo-Lorenzo, Terrero Encarnación, Way, García and Ulian2020), integrated phylogenetic and habitat data with seed traits, significantly enhancing prediction accuracy. These studies highlighted that desiccation sensitivity is more prevalent in species from warm, humid and stable environments and tends to be phylogenetically conserved at lower taxonomic levels. Overall, combining species-specific, ecological and taxonomic data in predictive models improves seed classification and informs ex situ conservation strategies.
Building on the 26% estimate proposed by León-Lobos et al. (Reference León-Lobos, Sandoval, Bolados, Rosas, Stark and Gold2014) a decade ago, the present study combines experimental desiccation assays with probabilistic modelling to generate a more robust and data-driven evaluation of desiccation tolerance in native woody species of Chile. This study aimed to determine the desiccation tolerance of seeds from native woody species of Chile and to evaluate the predictive performance of probabilistic models incorporating seed morphological traits, habitat variables and taxonomy. Specifically, we compare the consistency of results obtained using the Seed Coat Ratio–Seed Mass model (SCR–SM; Daws et al., Reference Daws, Garwood and Pritchard2006) and a Boosted Regression Tree model (BRT; Wyse and Dickie, Reference Wyse and Dickie2018) with experimental data from native Chilean woody species. We hypothesize that predictive models of desiccation response will align with experimental results, consistent with the principles underlying these models.
Material and methods
Species selection and plant material
This experimental study focused on the 15 genera identified as probably recalcitrant in Chile, as reported by León-Lobos et al. (Reference León-Lobos, Sandoval, Bolados, Rosas, Stark and Gold2014), and included the genus Myrcianthes to encompass all Chilean Myrtaceous genera for which recalcitrance has been suggested (Table 1). Approximately 200 mature fruits per species were collected and transported to the laboratory in perforated or ventilated plastic bags placed inside cool boxes. Upon arrival, fruits were stored at 10 ± 1°C and processed as soon as possible, typically within 2 weeks. Prior to experimentation, seeds were extracted from the fruits, cleaned and visually inspected to discard any infested or abnormal seeds.
Families and genera of trees and arborescent species native to Chile that were included in this study and whose seeds are probably sensitive to desiccation

Evaluation of seed response to desiccation
The seed desiccation response was assessed using the 100-seed test, as proposed by Pritchard et al. (Reference Pritchard, Wood, Hodges and Vautier2004b) for the list of potentially recalcitrant genera (Table 1), and including a small group of species with recognized orthodox behaviour (Table 2). This method was selected due to the limited availability of seeds and the conservation status of several native species. Where seed availability permitted, the protocol was adapted by increasing sample sizes and the number of replicates. A schematic representation of the 100-seed test is shown in Fig. 1.
Schematic representation of the 100-seed test protocol proposed by Pritchard et al. (Reference Pritchard, Daws, Fletcher, Gaméné, Msanga and Omondi2004a), adapted from Gold and Hay (Reference Gold and Hay2022).

Seed storage behaviour, Viability Loss Index (VLI) and germination capacity according to moisture content based on the 100-seed test

Note: MC: moisture content; Germ: germination; DS: desiccation sensitivity; DT: desiccation tolerance; P: probably DT/DS; ND: no data. Marked with an asterisk is the response according to the tetrazolium test.
Equilibrium relative humidity (eRH) was measured immediately upon seed arrival at the laboratory using a Rotronic HygroPalm water activity monitor (Rotronic Ltd, UK), recording water activity for approximately 30 minutes or until a stable eRH value was obtained. Moisture content was determined gravimetrically by drying at 103–105°C for 17 ± 1 hours. Larger seeds were sectioned to facilitate drying. Depending on availability, this analysis was conducted on 2 or 3 replicate samples, each comprising 5–10 seeds.
Seed viability was assessed via germination tests, based on physiological criteria (radicle emergence > 2 mm). Two or three replicates of 13–25 fresh seeds were used per species. Germination conditions were tailored to each species based on available literature. Small seeds were placed on agar-agar (0.7 g L−1) in Petri dishes, while larger seeds were placed on filter paper or directly on a peat-perlite (3:1) substrate within plastic containers. Germination was monitored periodically over a 120-day period. Seeds that did not germinate were evaluated using a tetrazolium test, following ISTA (2021) or a seed cut-test to determine viability and exclude dormancy as a cause of non-germination.
To compare desiccation responses, at least 64 seeds per species were used, divided equally between a drying treatment and a moist-stored control. Desiccation was performed in a desiccator with silica gel, using a minimum of 32 seeds. Simultaneously, at least 32 seeds were stored in sealed containers with moistened vermiculite to maintain high humidity. To prevent fungal growth, seeds were weighed, immersed for 10 minutes in a 1% sodium hypochlorite solution with Tween-20 and then dried on paper towels at room temperature until their original fresh weight was restored (Goveia et al., Reference Goveia, Kioko and Berjak2004; Berjak et al., Reference Berjak, Varghese and Pammenter2011). Prior to storage, seeds were treated with Captan 13 WP (active ingredient: N-(trichloromethylthio)cyclohex-4-ene-1,2-dicarboximide, 13% w/w; Anasac, Chile).
Seeds under the drying treatment were weighed periodically, and their eRH was monitored using the same water activity monitor. Drying was considered complete when eRH reached approximately 15 ± 3% and seed weight stabilized, indicating the absence of free water. Moist-stored seeds were removed when the drying treatment was finished. Moisture content was determined for at least six seeds from each treatment (drying and moist-stored) using the same gravimetric method described above, and viability was assessed for a minimum of 26 seeds per treatment using the same germination protocol applied initially.
The Viability Loss Index (VLI), as proposed by Mattana et al. (Reference Mattana, Peguero, Di Sacco, Agramonte, Encarnación Castillo, Jiménez, Clase, Pritchard, Gómez-Barreiro, Castillo-Lorenzo, Terrero Encarnación, Way, García and Ulian2020), was used to analyse the results:
VLI = [germination in fresh seeds (%) − germination in dry seeds (%)]/germination in fresh seeds (%).
VLI values range from 0 (desiccation-tolerant) to 1 (desiccation-sensitive). Negative values may occur when dried seeds germinate better than fresh ones. The classification is as follows:
• VLI > 0.95: desiccation-sensitive
• VLI > 0.5 but <0.95: potentially desiccation-sensitive
• VLI > 0.05 but <0.5: potentially desiccation-tolerant
• VLI < 0.05: desiccation-tolerant.
Evaluation of two predictive models of seed response to desiccation
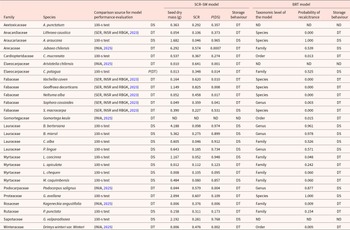
For a second set of 27 species (Table 3), which included both desiccation-sensitive and desiccation-tolerant taxa, the results of the 100-seed test were compared with predictions from the SCR–SM (Daws et al., Reference Daws, Garwood and Pritchard2006) and the BRT (Wyse and Dickie, Reference Wyse and Dickie2018) models.
Seed storage behaviour outputs of both models: SCR–SM and BRT for the species studied

Note: DT: desiccation-tolerant; DS: desiccation-sensitive; ND: no data were available to run the mo 100-s test: 100 seed tests.
For the SCR–SM model, seeds were extracted from the fruits and visually inspected to identify and discard those affected by fungal infection and/or predation damage. Following the methodology proposed by Daws et al. (Reference Daws, Garwood and Pritchard2006), at least 20 healthy seeds per species were dissected into two components: the endocarp and/or seed coat (or other structures surrounding the embryo/endosperm that may offer protection against predation; Grubb and Dfrp, Reference Grubb and Dfrp1998), and the embryo/endosperm. These components were dried separately in two or three replicates at 103–105°C for 17 ± 1 hours and weighed individually. The SCR was calculated as the proportion of the mass of the endocarp/coat component relative to the total SM.
The probability of desiccation sensitivity (P[DS]) for each species, according to the SCR–SM model, was determined using the following equation:
 \begin{equation*}P(DS)\, = \,\frac{{{e^{3.269 - 9.974a + 2.156b}}}}{{1 + {e^{3.269 - 9.974a + 2.156b}}}}\end{equation*}
\begin{equation*}P(DS)\, = \,\frac{{{e^{3.269 - 9.974a + 2.156b}}}}{{1 + {e^{3.269 - 9.974a + 2.156b}}}}\end{equation*}Where a is the SCR and b is the log₁₀ of SM in grams. The model is applicable for seeds with masses between 0.01 mg and 24 g, and SCR values between 0 and 1. A result of P(DS) > 0.5 indicates desiccation sensitivity, whereas P(DS) < 0.5 indicates desiccation tolerance (Daws et al., Reference Daws, Garwood and Pritchard2006).
The BRT models incorporate the following variables:
(a) Seed storage behaviour from the literature and taxonomic relationships between the target species and related taxa with known storage behaviour. This information is curated by the Society for Ecological Restoration (SER) in collaboration with the Royal Botanic Gardens, Kew (SER, INSR and RBGK, 2023).
(b) Climate and elevation of the species’ geographic distribution, including annual and minimum monthly precipitation, mean annual temperature, diurnal temperature range and altitude. These data were obtained from the WorldClim database (Fick and Hijmans, Reference Fick and Hijmans2017).
(c) Woodiness, based on data from Zanne et al. (Reference Zanne, Tank, Cornwell, Eastman, Smith, Fitzjohn, McGlinn, O’Meara, Moles, Reich, Royer, Soltis, Stevens, Westoby, Wright, Aarssen, Bertin, Calaminus, Govaerts and Beaulieu2014), available via the Dryad data repository.
(d) SM and dispersal mode, sourced from the TRY Plant Trait Database (Kattge et al., Reference Kattge, Díaz, Lavorel, Prentice, Leadley, Bönisch, Garnier, Westoby, Reich, Wright, Cornelissen, Violle, Harrison, Van Bodegom, Reichstein, Enquist, Soudzilovskaia, Ackerly, Anand and Wirth2011), the Global Biodiversity Information Facility (GBIF.org, 2023) and SER in collaboration with Royal Botanic Gardens, Kew (SER, INSR and RBGK, 2023).
Wyse and Dickie (Reference Wyse and Dickie2018) proposed three BRT models based on the taxonomic proximity of the target species to those with known seed storage behaviour: (i) genus-level, (ii) family-level and (iii) order-level models. An example workflow for applying these models is provided in Supplementary Figure S1. The models are available as an online tool hosted by the Royal Botanic Gardens of Kew (2023), and as scripts to run in R software. The probability of recalcitrance ranges from 0 (likely tolerant) to 1 (likely sensitive), with a threshold of 0.5 used to distinguish between the two categories.
Comparison of predictive models and estimation of recalcitrance incidence
Model performance was evaluated using the F1-score, precision and recall. For each species, the response variable was obtained from three biological replicates, and the modelling pipeline did not incorporate repeated training–testing cycles or any resampling of the species set. Because no resampling procedures were applied, no sampling distribution of the F1-score was generated, precluding the estimation of confidence intervals; therefore, F1-scores are reported as point estimates.
• Precision (p) = TP/(TP + FP)
• Recall (r) = TP/(TP + FN)
• F1-score = 2× (p × r)/(p + r).
Where TP = true positives, FP = false positives and FN = false negatives.
The BRT model was refined using experimental data, a literature review and information from the National Catalogue of Plant Genetic Resources of the Agricultural Research Institute of Chile (INIA, 2025), under the assumption that species conserved in seed banks are, by definition, not recalcitrant. The corrected BRT model was then applied to estimate the incidence of seed recalcitrance among Chile’s native tree flora, using the species list provided by Rodriguez et al. (Reference Rodriguez, Marticorena, Alarcón, Baeza, Cavieres, Finot, Fuentes, Kiessling, Mihoc, Pauchard, Ruiz, Sanchez and Marticorena2018). These estimates were cross-referenced with the database of the Regulation for the Classification of Wild Species from the Chilean Ministry of the Environment (Ministerio del Medio Ambiente de Chile, 2024) to identify species that are both threatened and potentially possess recalcitrant seeds.
Results
Seeds’ response to desiccation
The only species excluded from the study was Blepharocalyx cruckshanksii (Hook. & Arn.) O. Berg, as no seeds were available from any population. Most of the species identified in the literature as potentially sensitive to desiccation were confirmed to be desiccation-sensitive (DS) (Table 2). Exceptions included Citronella mucronata (Ruiz & Pav.) D. Don, Crinodendron patagua Molina, Gevuina avellana Molina and Amomyrtus luma (Molina) D. Legrand & Kausel, which retained seed viability at moisture contents below 5% (Fig. 2).
Germination response of the 100-seed test for species previously identified in the literature as desiccation-tolerant, as well as those marked with an asterisk (*), denoting species previously classified as potentially desiccation-sensitive, yet demonstrating desiccation tolerance in the present study.

Citronella mucronata seeds exhibited dormancy and unknown germination requirements; therefore, viability was assessed using the tetrazolium test (Fig. 3). Crinodendron patagua showed identical germination rates in both the control and post-drying treatments (80.7%). Gevuina avellana demonstrated improved germination following drying (81.3%) compared to the control (72.9%). In contrast, A. luma exhibited a reduction in germination capacity from 50% in the fresh control to 38.5% after drying, with a VLI of 0.230, suggesting it should be classified as potentially desiccation-tolerant.
Seed of C. mucronata dyed with tetrazolium after desiccation treatment (15% eRH).

The same 100-seed test was applied to a smaller group of species with recognized orthodox behaviour: Sophora macrocarpa Sm., Eucryphia glutinosa (Poepp. & Endl.) Baill. and Laurelia sempervirens (Ruiz & Pav.) Tul. (Table 2). Figure 2 illustrates the variation in germination response to moisture treatments for these orthodox species. Sophora macrocarpa showed similar germination rates between the control (73%) and drying treatment (68%), as did E. glutinosa (23% and 21%, respectively). In contrast, L. sempervirens exhibited a marked decline in germination in both drying and moist storage treatments (12% and 10%, respectively), compared to the control (30%), indicating reduced viability not directly linked to moisture content.
Examples of germination responses to desiccation are shown in Fig. 4 for Myrceugenia obtusa (DC.) O. Berg and in Fig. 5 for Myrcianthes coquimbensis (Barnéoud) Landrum & Grifo. Myrceugenia obtusa exhibited high germination (86.6%) when sown fresh at a moisture content of approximately 49.4%. However, germination dropped sharply to 34.4% at 28.3% moisture content, and nearly lost all viability (2.2%) at 23.5% moisture (Fig. 4). Similarly, M. coquimbensis showed 100% germination when fresh, but lost all viability (0%) after partial drying to 18% moisture. In contrast, seeds stored under moist conditions during the same period retained a germination rate of 76.6%, comparable to the control (Fig. 5).
Moisture sorption isotherm (lines) and germinative response (bars) of seeds of the recalcitrant species M. obtusa.

Cumulative germination of the recalcitrant species M. coquimbensis for control (fresh), moist-stored and partially dried seeds of the 100-seed test stored for 36 days. Note: During the moist-stored treatment, 16% of the seeds germinated during storage.

Comparison of predictive models
Of the 27 species listed in Table 3, the SCR–SM model was successfully implemented for 26 species, whereas application of the BRT model was feasible for 24 species. Desiccation-sensitive species exhibited a mean SCR of 0.157 ± 0.1 (min: 0.046; max: 0.311), while desiccation-tolerant species showed a significantly higher mean SCR of 0.468 ± 0.18 (min: 0.106; max: 0.825). Supplementary Figure S2 presents cross-sections of recalcitrant seeds, highlighting the proportion of seed coat relative to other tissues – an essential component of the SCR–SM model.
In terms of model performance, the SCR–SM model achieved F1-scores of 0.761 and 0.838 for identifying DS and DT seeds, respectively (Table 4). These scores are based on consistency with the 100-seed test for recalcitrant species and literature review for orthodox species. The BRT model was also applied to the same species set (Table 3), yielding F1-scores of 0.526 and 0.689 for DS and DT classification, respectively (Table 4).
Evaluation of precision, recall and F1-score for models used to predict seed desiccation sensitivity

Note: SCR–SM: Seed Coat Ratio – Seed Mass mo BRT: Boosted Regression Tree mo DT: desiccation-tolerant; DS: desiccation-sensitive.
A comparison of F1-scores indicates that the SCR–SM model outperforms the BRT model in predicting DS and DT species. However, the BRT model’s performance varies depending on the taxonomic level used. When applied at the genus level, its accuracy improved to 80%, whereas performance dropped significantly to 22% when applied at the family level.
Estimation of recalcitrance incidence
Application of the BRT model to a dataset of 70 Chilean tree species – representing 95.8% of the native and endemic trees of continental Chile (n = 73) – suggests a recalcitrance incidence of 18% (n = 11), with outputs for 61 species. After adjusting predictions using experimental data, literature sources and information from INIA’s gene bank system, the estimated incidence of recalcitrance among Chile’s native tree flora is approximately 19% (n = 11), based on 58 species (Supplementary Table S1). This estimate excludes shrub and small tree species identified as recalcitrant in this study, such as Gayella valparadisaea (Molina) Pierre, Luma chequen (Molina) A. Gray, Legrandia concinna (Phil.) Kausel, M. coquimbensis and Myrceugenia spp.
Among the 17 tree and shrub species confirmed as recalcitrant in this study, 12 are endemic to Chile (70.6%), and 9 are listed under threat categories (Critically Endangered, Endangered or Vulnerable; 53%) (Table 5).
Endemism and conservation status of Chilean woody species with confirmed recalcitrance

Note: EN: endangered; VU: vulnerable; NT: near threatened; LC: least concern; NE: not evaluated; RCE: Regulation for the Classification of Wild Species; DS: Supreme Decree; IUCN: International Union for Conservation of Nature; MMA: Ministry of the Environment; MINSEGPRES: Ministry of the General Secretariat of the Presidency.
Discussion
Seeds’ response to desiccation
Except for C. mucronata, G. avellana, A. luma and C. patagua, Chilean tree species included in this study and identified in the literature as potentially desiccation-sensitive seeded species exhibited this trait. Particular attention should be given to C. mucronata, as effective dormancy-breaking and germination protocols could not be established during the study period. Given the absence of prior information regarding tetrazolium staining patterns for this species and the lack of studies on dormancy and germination requirements, the classification of its desiccation response and potential tolerance status remains provisional. Further research is needed to develop reliable dormancy-release procedures, characterize tetrazolium staining patterns, refine viability assessments and evaluate its desiccation response and storage behaviour. Moreover, the genus Citronella, which comprises 22 accepted species, has not been previously studied in terms of desiccation tolerance or storage response.
Seeds that are sensitive to desiccation typically exhibit active metabolic processes and rapid germination, often resulting in low dormancy prevalence (Tweddle et al., Reference Tweddle, Dickie, Baskin and Baskin2003). Some researchers have explored the relationship between desiccation sensitivity and dormancy, suggesting that these traits are frequently mutually exclusive (Jaganathan, Reference Jaganathan2021) or that desiccation tolerance and dormancy may have co-evolved through analogous mechanisms (Costa et al., Reference Costa, Farrant, Oliver, Ligterink, Buitink and Hilhorst2016). In certain cases, a causal relationship has been proposed, whereby desiccation induces dormancy (Wood et al., Reference Wood, Pritchard and Amritphale2000). Consequently, the deep dormancy observed in C. mucronata seeds suggests desiccation tolerance. However, recent work by Jaganathan and Phartyal (Reference Jaganathan and Phartyal2025) has shown that desiccation-sensitive species – particularly within the Fagaceae – may also exhibit physiological or epicotyl physiological dormancy, indicating that desiccation sensitivity and dormancy can coexist more often than previously assumed.
In contrast, the findings for G. avellana diverge from those of Donoso (Reference Donoso2006), who reported that seeds of this species lack tolerance to storage and must be sown immediately to avoid desiccation-related viability loss. While our results support a desiccation-tolerant response, they do not imply orthodox storage behaviour. Rather, an intermediate response remains a plausible possibility, as tolerance to drying could coexist with sensitivity to storage conditions. Further research is therefore needed to determine whether the seeds of this species have an inherently short lifespan or are particularly vulnerable to low temperatures.
As G. avellana is a monotypic genus (Moya-Moraga and Pérez-Ruíz, Reference Moya-Moraga and Pérez-Ruíz2022), comparative analyses are limited. Nevertheless, related species within the subfamily Grevilleoideae (Proteaceae) display a range of storage behaviours, including intermediate forms such as Macadamia integrifolia Maiden & Betche (Kholibrina et al., Reference Kholibrina, Susilowati, Kusuma and Aswandi2019) and orthodox forms like Banksia ashbyi Baker f. (Merritt et al., Reference Merritt, Touchell, Dixon, Plummer and Turner2000). Among native Chilean species closely related to G. avellana, members of the genera Lomatia, Orites and Embothrium – also within Grevilleoideae – are preserved in the INIA Base Seed Bank and classified as orthodox (INIA, 2025), which supports the findings for G. avellana in this study.
The results also reveal contrasting desiccation responses between the two species of the genus Amomyrtus: A. luma is tolerant to desiccation, whereas A. meli (Phil.) D. Legrand & Kausel is sensitive. Such divergence within a single genus is relatively uncommon (Wyse and Dickie, Reference Wyse and Dickie2018). As these are the only known representatives of Amomyrtus, further research is warranted to elucidate the mechanisms underlying these differences. A similar case is observed with C. patagua, which demonstrated desiccation tolerance. Crinodendron hookerianum Gay, also native and endemic to Chile, was not included in the experimental evaluation. Therefore, assessing its desiccation response would be valuable to determine whether it behaves similarly to C. patagua or exhibits intra-generic variation akin to that observed in Amomyrtus.
The desiccation responses of the remaining species were consistent with the potential sensitivity to drying proposed by León-Lobos et al. (Reference León-Lobos, Sandoval, Bolados, Rosas, Stark and Gold2014), which was inferred from traits associated with recalcitrance, such as fruit type and size, dispersal syndrome and morphological characteristics.
Performance of models for predicting seed desiccation sensitivity
The investigation of Chilean woody species supported the hypothesis regarding the reliability of predictive models based on morphological, ecological and taxonomic traits in determining responses to desiccation, albeit with varying degrees of accuracy. Among the species analysed, the SCR–SM model outperformed the BRT model in predicting both desiccation sensitivity and tolerance. This finding is consistent with the theory that desiccation-sensitive species tend to possess thinner seed coats, which represent a smaller proportion of the total SM compared to desiccation-tolerant species (Pritchard et al., Reference Pritchard, Daws, Fletcher, Gaméné, Msanga and Omondi2004b; Daws et al., Reference Daws, Garwood and Pritchard2006). In this study, the mean SCR was 0.157 for DS species and 0.468 for DT species, closely aligning with values reported by Daws et al. (Reference Daws, Garwood and Pritchard2005) and Lan et al. (Reference Lan, Xia, Wang, Liu, Zhao and Tan2014), who documented average SCRs of 0.209 and 0.21 for DS species, and 0.509 and 0.47 for DT species, respectively.
The SCR–SM model is based on the theoretical premise that larger seeds, which accumulate greater reserves, provide a degree of independence during early establishment (Pritchard et al., Reference Pritchard, Daws, Fletcher, Gaméné, Msanga and Omondi2004b; Marques et al., Reference Marques, Buijs, Ligterink and Hilhorst2018). Additionally, larger seeds tend to lose water more slowly from the embryo, a critical factor given the embryo’s vulnerability to desiccation (Jaganathan, Reference Jaganathan2021). Literature suggests that larger seeds generally possess thinner seed coats or shells (i.e., lower SCR), a trait hypothesized to be linked to shorter time interval preceding germination, reducing exposure to predation and the need for substantial investment in physical defence structures (Daws et al., Reference Daws, Garwood and Pritchard2005). Additionally, in recent work on Fabaceae species from northern India, lower SCR values were strongly associated with the absence of physical dormancy (Jayasuriya and Phartyal, Reference Jayasuriya and Phartyal2024).
In the present study, the SCR–SM model correctly classified 80.7% of the 26 species analysed, including both DS and DT species. While this accuracy is slightly lower than the 88% reported by Lan et al. (Reference Lan, Xia, Wang, Liu, Zhao and Tan2014) and the 87% by Daws et al. (Reference Daws, Garwood and Pritchard2005), it exceeds the 70% predictability reported by Samarasinghe et al. (Reference Samarasinghe, Jayasuriya, Gunaratne, Dixon and Senanayaka2024). Furthermore, our findings indicate that the SCR–SM model is more effective in identifying desiccation tolerance than sensitivity, with F1-scores of 0.838 for DT seeds and 0.761 for DS seeds.
The BRT model demonstrated a lower overall performance, correctly identifying desiccation responses in 62.5% of the 24 species studied. Notably, within the Myrtaceae family, the BRT model classified Chilean species as desiccation-tolerant when applied at the family level. However, predictive accuracy at this taxonomic scale was considerably reduced. Previous studies and our own findings indicate that many Chilean Myrtaceae species are, in fact, desiccation sensitive. Nonetheless, on a global scale, other genera and species within Myrtaceae exhibit desiccation-tolerant seed behaviour (SER, INSR and RBGK, 2023). This misclassification reveals an important limitation of the model: its predictive reliability is not uniform across taxonomic groups. Even when predictions are generated at the same taxonomic resolution (e.g., family level), the accuracy of the model can vary substantially depending on the biological attributes, ecological characteristics and biogeographic contexts of the families involved.
This finding contrasts with the performance reported by the model’s authors, who achieved over 79% accuracy for desiccation-sensitive species at both family and genus levels in cross-validation studies based on floras from Britain, Ireland and Ecuador (Wyse and Dickie, Reference Wyse and Dickie2018). The divergence may reflect intrinsic features of the family, including its evolutionary history, ecological strategies, morphological seed traits or the prevalence of desiccation sensitivity in certain lineages, all of which can differ markedly from patterns observed in other regions.
Recognizing this limitation is essential for correctly interpreting the model’s outputs and for informing its application to other floras. It underscores that predictive models, although valuable tools, may require regional calibration or the incorporation of family-specific empirical data to achieve reliable performance across diverse biogeographic contexts.
The application of the BRT model to Chilean woody species demonstrates its effectiveness when implemented at the genus level. This is consistent with the model’s theoretical basis, which posits that seed desiccation responses are relatively conserved at lower taxonomic levels. Consequently, trait values from closely related species may serve as reliable indicators for predicting the desiccation response of a target species (Wyse and Dickie, Reference Wyse and Dickie2018). According to Wyse and Dickie (Reference Wyse and Dickie2018), when applied at the genus level, the proportion of congeners producing desiccation-sensitive seeds accounts for 91% of the model’s classification accuracy, making it the most influential variable. SM is the next most significant variable, contributing an additional 2.7% to the model’s predictive capacity. In this study, the contrasting desiccation responses of the two Amomyrtus species represent an exception to the generally conserved pattern of recalcitrance observed at lower taxonomic levels.
Estimation of recalcitrance incidence
The estimated incidence of recalcitrance among Chile’s arboreal flora, at 19%, is consistent with the proportion of species bearing desiccation-sensitive seeds reported by Tweddle et al. (Reference Tweddle, Dickie, Baskin and Baskin2003), who found that 17.6% of trees and shrubs across 25 study regions exhibited this trait. Similarly, this proportion aligns with recalcitrance incidence patterns observed across vegetation zones. Although the Chilean flora analysed in this study was selected based on the likelihood of recalcitrance rather than geographical distribution, most species fall within the subtropical and temperate rainforest vegetation zones – regions where recalcitrance incidence is estimated at approximately 20% (Tweddle et al., Reference Tweddle, Dickie, Baskin and Baskin2003; Wyse and Dickie, Reference Wyse and Dickie2017).
Given the restricted number of species evaluated in the model comparison (n = 27), caution is warranted in generalizing these results across the full Chilean woody flora. Expanding the dataset of experimentally assessed species was not possible within the current study, yet these findings provide a valuable foundation. Future research should build upon this work by increasing taxonomic coverage and applying the standardized methodological approach proposed here, thereby enhancing the robustness and predictive power of desiccation-response assessments.
The variation in the proportion of species with recalcitrant seeds across vegetational and climatic zones – particularly the higher frequency in humid environments with moderate temperatures – reflects the evolutionary development of this trait in response to specific ecological conditions (Dickie and Pritchard, Reference Dickie, Pritchard, Black and Pritchard2002). In this context, it is critical to highlight the conservation risks posed by climate change to species with recalcitrant seeds. Projections indicate, with high confidence, that extreme drought events – both in frequency and intensity – are expected to increase globally (Arias et al., Reference Arias, Bellouin, Coppola, Jones, Krinner, Marotzke, Naik, Palmer, Plattner, Rogelj, Rojas, Sillmann, Storelvmo, Thorne, Trewin, Achuta Rao, Adhikary, Allan, Armour and Zickfeld2021). This trend poses a significant threat to the long-term survival of recalcitrant species, whose prevalence declines as habitats become drier (Tweddle et al., Reference Tweddle, Dickie, Baskin and Baskin2003; Probert et al., Reference Probert, Daws and Hay2009; Fernández et al., Reference Fernández, León-Lobos, Contreras, Ovalle, Sershen and Ballesteros2023).
Among the tree and shrub species confirmed as recalcitrant in this study (excluding those predicted but not yet experimentally validated), 70.5% (n = 12) are endemic to Chile, and 53% (n = 9) are classified as threatened. The integrated assessment of seed recalcitrance, endemism and conservation status enables the prioritization of at least six endemic woody species for urgent ex situ conservation efforts. These include one species classified as Vulnerable: Beilschmiedia miersii (Gay) Kosterm.; and five classified as Endangered: A. araucana, Beilschmiedia berteroana (Gay) Kosterm., G. valparadisaea, L. concinna, M. coquimbensis and Pitavia punctata Molina. These species urgently require tailored conservation strategies to ensure long-term preservation and maintain adequate levels of genetic diversity. A similar approach, incorporating desiccation sensitivity as a criterion for prioritizing collections and guiding conservation strategies, has been proposed by Ley-López et al. (Reference Ley-López, Wawrzyniak, Chacón-Madrigal and Chmielarz2023).
Protocols for assessing seed desiccation responses typically require substantial experimental time and often involve destructive techniques to evaluate post-drying viability (Pelissari et al., Reference Pelissari, José, Fontes, Matos, Pereira and Faria2018). These constraints pose challenges for species with limited seed availability, those that are endangered, or populations that are difficult to access. Moreover, viability assessments depend on prior knowledge of species-specific dormancy-breaking and germination requirements (Mattana et al., Reference Mattana, Peguero, Di Sacco, Agramonte, Encarnación Castillo, Jiménez, Clase, Pritchard, Gómez-Barreiro, Castillo-Lorenzo, Terrero Encarnación, Way, García and Ulian2020), as exemplified by C. mucronata in this study. These considerations underscore the need to develop methodologies that enable indirect determination of storage behaviour or drying tolerance through predictive models based on trait correlations.
In this study, some species previously considered potentially desiccation-sensitive were experimentally classified as desiccation-tolerant. This finding warrants further investigation and caution. Species identified as tolerant to desiccation should not be classified as orthodox until their tolerance to low temperatures and viability under long-term conventional seed bank conditions are confirmed. Without such evidence, the potential for misclassification remains, which could have important implications for conservation planning and ex situ seed management. Future studies should therefore incorporate controlled cold storage trials and monitor viability over extended periods to validate storage behaviour and inform appropriate conservation strategies. Additionally, it would be pertinent to assess the impact of desiccation on seed vigour, as this study used germination as the sole indicator of desiccation tolerance.
Species identified as desiccation-sensitive also merit further investigation into the debated existence of species-specific critical moisture content thresholds. This would allow the establishment of intra- and interspecific sensitivity gradients. The concept of a critical moisture threshold is controversial, as it depends on methodological factors and may vary among populations (Stavrinides et al., Reference Stavrinides, Dussert, Combes, Fock-Bastide, Severac, Minier, Bastos-Siqueira, Demolombe, Hem, Lashermes and Joët2020; Chen et al., Reference Chen, Visscher, Ai, Yang, Pritchard and Li2023). To advance this line of inquiry, it is essential to define standardized methodologies for the quantitative assessment of desiccation response, including drying techniques, clear definitions of severe damage, viability assessment protocols and consideration of interpopulation variability (Pammenter et al., Reference Pammenter, Naidoo, Berjak, Nicolás, Bradford, D and Pritchard2002), as well as the specific seed structures assessed versus the desiccation response of excised embryos.
This study represents a pioneering effort to investigate desiccation tolerance and sensitivity across a broad range of Chilean woody species, while also evaluating the performance of predictive models for this critical trait. The findings provide a robust classification of species based on their storage behaviour, establishing a foundation for identifying candidates suitable for seed banking. Furthermore, the study highlights species that require alternative ex situ conservation strategies, emphasising the need for targeted scientific efforts in biotechnological preservation. To consolidate this argument, it is important to consider that the effective ex situ management of desiccation-sensitive or recalcitrant species necessitates the incorporation of alternative technologies beyond conventional seed banking. Methods such as cryopreservation and in vitro conservation, along with advanced biotechnological interventions – widely endorsed within integrated conservation frameworks (Pence et al., Reference Pence, Meyer, Linsky, Gratzfeld, Pritchard, Westwood and Bruns2022) – constitute critical avenues for ensuring the persistence of these taxa.
Importantly, six endemic and threatened species identified in this study require urgent conservation action. Their prioritization in national biological conservation plans and programs is critical, not only for safeguarding Chile’s unique biodiversity but also for informing policy and resource allocation for conservation efforts. By emphasizing the policy relevance of these findings, this work provides both a scientific foundation and a practical roadmap for preserving species most at risk.
Supplementary materials
The supplementary material for this article can be found at https://doi.org/10.1017/S0960258526100087.
Acknowledgements
We gratefully acknowledge the valuable support provided during seed collection by Manuel Gutiérrez and Paulo Guarda (INIA), Mónica Musalem and Claudia González (Vivero y Jardín Pumahuida), David Muñoz (Department of Protected Wild Areas, CONAF Maule), as well as park rangers Carlos Reyes (Los Queules National Reserve), Cristian Alegría (Bellotos del Melado National Reserve) and Lorenzo Pardo (Radal Siete Tazas National Park). We also thank resident student José Miguel Lucero for his assistance with field and laboratory work.
Author contributions
Conceptualisation, A.F., P.L. and S.C.; methodology, A.F., P.L. and S.C.; validation, A.F., P.L. and S.C.; formal analysis, A.F.; investigation, A.F. and L.A.; resources, A.F., P.L. and S.C.; data curation, A.F.; writing – original draft preparation, A.F.; writing – review and editing, A.F., P.L. and S.C.; visualization, A.F.; supervision, P.L. and S.C.; project administration, A.F., P.L. and S.C.; and funding acquisition, A.F., P.L. and S.C. All authors have read and agreed to the published version of the manuscript.
Funding statement
This research was funded by ANID Chile, Advanced Human Capital Training Program, National Doctoral Scholarship, grant number 21211942 (A.F.), The INIA-ANID Science Program, grant number 503861-71 and the Native Forest Research Fund, National Forest Corporation (CONAF), Ministry of Agriculture, Chile ID 015/2023.
Competing interests
The authors declare no conflicts of interest.
Data availability
All data are available in the article and supplementary materials.