Exclusion from Research Participation and Overreliance on Surrogate Consent
Precision medicine research (PMR) has potential to transform health by unearthing genetic and non-genetic disease mechanisms and developing tailored prevention, diagnostic, and treatment strategies that take into account individual variance in including genetic, environmental, and lifestyle factors. 1 Inclusion of adults with intellectual disability in PMR offers new opportunities to address their health needs,Reference Havercamp and Glidden 2 and is critical for benefits to accrue to this health disparities population.Reference Spong and Bianchi 3 Yet there is evidence that adults with intellectual disability are commonly excluded from health research and clinical trials, including PMR. For example, the national PMR initiative, the National Institutes of Health’s All of Us Research Program, began enrollment in 2018 but does not yet intentionally enroll adults with intellectual disability. 4
Informed consent, a requirement reflecting the fundamental values of human dignity and respect,Reference Pugh 5 is at the heart of research participation, yet evidence and consensus consistently suggest the over-exclusion of adults with intellectual disability therein. While autonomous consent remains the gold standard, 6 challenges associated with intellectual disability (e.g., understanding abstract concepts) and social dynamics (e.g., potential undue influence) foster erroneous beliefs that this population is unable to make decisions for themselves, thereby frequently leading to their exclusion from clinical researchReference Katherine, Schwartz and Sabatello 7 or inclusion only with proxy consent, regardless of consent capacity or consideration of the ethical, legal, or scientific justifications for either approach. Reference Goldsmith and Skirton 8
Exclusion from research and overreliance on surrogate consent emerged, in part, from prevailing assumptions in broader societal contexts. These include decades-long beliefs about inabilities alongside practices to protect adults with intellectual disability and restrict autonomy and legal rights to steward their lives. These have been done most notably through state guardianship laws for adults determined to lack legal competence to make decisions, though presumed incompetency often takes hold also on a group level, assuming that all adults with intellectual disability lack competency.Reference Plosky 9 Disability rights efforts challenge these beliefs and their associated structures that restrict autonomy, noting that discrimination on the basis of disability is a civil and human rights violation and that disability accommodations should be used to enable equitable access, including to exercise one’s right to autonomy. 10 The disability rights movement has created legal and social advances toward greater inclusion of people with disabilities in all aspects of life, emphasizing social forces that preclude full participation in society. Critically, it reinforced not only the recognition of the legal personality of people with disabilities as equal members of society, but also the right to enjoy — and exercise — one’s legal capacity on an equal basis with others in all aspects of life.Reference Melish, Sabatello and Schulze 11 These shifts carry critical implications for participation in PMR (and other health) research.
Beyond a shared commitment to and value for inclusion, the promise of translating scientific discoveries to improved health hinges on diverse representation in research cohorts. It has also been an instrumental effort since the launch of the All of Us Research Program in 2016, with the Program’s preliminary report highlighting that the cohort must reflect the diversity of the US population. In turn, it requires that individuals from all segments of society have the opportunity to consider participation and opt to participate. Efforts to promote cohort diversity cannot be achieved without study designs that are responsive to the values and needs of diverse communities. As research shows, these are bedrocks of trust, an essential precondition to enrollment in research — and subsequent perceptions that research is trustworthy.Reference Smirnoff 12 Emerging evidence indicates that adults with intellectual disability do not support their exclusion from research; rather, many want to be respectfully included in research, including PMR.Reference Sabatello 13 Their motivations to volunteer mirror those of the general public and include desires to learn new things and potentially derive other direct benefits,Reference Dinerstein, Powers and Holmes 14 such as to receive genetic and other results relevant for their health and identity, 15 help others, meet new people, and feel valued and respected.Reference Clark 16
Adults with intellectual disability also reject overreliance on surrogate decision-makers. Aligned with broader preferences for general decision-makingReference Sabatello and Appelbaum 17, and the focus on empowerment in PMR, 18 adults with intellectual disability emphasize that they should be in control of research participation decisions. Relatedly, they largely reject the idea that proxy consent is a desirable or appropriate protection.Reference McDonald 19 While upholding individual empowerment for adults with intellectual disability is vital for recognizing their equal worth as members of society, controlling these decisions is especially critical for PMR as such studies may involve potential benefits that require action to materialize (e.g., changing exercise regimen), unknown risks (e.g., returning findings of variants of unknown significance) and potential harm (e.g., genetic-based discrimination), and may have implications for personal and familial health information.Reference McGuire and Beskow 20 The latter in particular merit consideration given that family members may serve as guardians on some decisions of the individual, which may also introduce a conflict of interest when return of results from PMR is involved.
Inclusion and Autonomous Participation Decisions in PMR: The Legal Landscape
In addition to failing to garner support from adults with intellectual disability, historic exclusion and reliance on proxy consent are not aligned with guidelines that preclude categorical or diagnosis-based exclusion from research. 21 Existing statutes and policies, internationally and nationally, underscore the equally important goal of protecting the right to autonomy, and thus efforts to exclude or rely on proxy consent may deny rights without a legal basis.
The United Nations’ 2006 Convention on the Rights of Persons with Disabilities (CRPD) 22 emphasizes access to opportunity and autonomy, including specific provisions about research and scientific efforts. Article 4 on States’ general obligations, for example, explicitly require state parties to “undertake and promote” research that follows principles of universal design and that assures availability and use of new technologies for persons with disabilities. The US has signed but not ratified the Convention, indicating endorsement of and an expectation that it would follow its spirit. In addition, US federal laws set standards for accessibility, including reasonable accommodations, for inclusion of people with disabilities and prohibit disability-based discrimination, exclusion or denial of opportunities in programs and services that are publicly funded or receive federal financial assistance. 23 Both the CRPD and US disability laws, particularly section 504 of the 1973 Rehabilitation Act, the 1990 Americans with Disabilities Act, and 2010 Patient Protection and Affordable Care Act, further highlight that the absence of reasonable accommodation to enable exercise of individual rights, including to autonomy, is discriminatory in itself. 24
Of interest to research participation, Article 12 of the CRPD requires states parties to both uphold the legal personality of people with disability under the law and to recognize that persons with disabilities enjoy the right to exercise their legal capacity on an equal basis, which includes access to supports needed to exercise the right.Reference Dinerstein 25 Article 15 further includes that no one shall be subjected to medical or scientific experimentation without free consent. In the US, legal reform of state guardianship statutes is also shifting to reflect these human rights mandates. Historically, guardianship laws have directed courts to appoint another adult to make decisions for individuals determined to lack competence to decide for themselves. Depending on the jurisdiction, guardianship can be plenary (full) or limited with respect to the scope of decisions under the guardians’ purview, depending on the abilities of the adult with intellectual disability and why they were appointed a guardian. As a best practice followed in many jurisdictions, the scope of guardianship should be individually tailored, and each guardianship court order should specify the decisions a guardian is authorized to make. In practice, however, adults with intellectual disability subject to guardianship are often assumed incompetent to make most, perhaps all, decisions, including those not specified in the court order. 26 This is the case also for adults with intellectual disability who are subject to limited guardianship, which is, by definition, limited in the scope of power that is being granted to the guardians.
In PMR settings, this presumption of incompetence has significant negative impacts — and often without legal justification. Many PMR studies rely on secondary datasets and are presumed to be of no greater than minimum risk, such as joining a biobankReference Mikkelsen 27 that mostly raises informational risks, yet the binary approach of consent capacity may also preclude enrollment into such studies.Reference Hawkins 28 Concurrently, studies that pose additional risks, such as those involving the return of personal genetic results, are also intertwined with greater potential benefits 29 (e.g., the possibility of early identification of a genetic risk and acting on it). Yet the transfer of decision-making to a guardian (sometimes without clear legal authorization) may result in exclusion — and forgoing potential health benefits. Although guardianship orders rarely include explicit provisions on research decisions (some exceptions may exist regarding clinical trials with the prospect of direct benefit to the individual), researchers may not know the scope of the guardianship order or presume, perhaps inaccurately, that the guardian can and should make all decisions. Institutional Review Boards (IRBs) that aim to protect the rights and safety of research participants commonly include clinical experts and are similarly often unaware of these differences in scope of guardianship — even as determination of level of risks is integral to their work. When this unawareness occurs, an individual’s right to autonomous decision-making may be unjustly bypassed by a guardian’s decision. Individualized determinations of whether guardians may consent to participation in PMR reflect commitment to respecting and facilitating participant autonomy and represent a critical step toward mitigating potential negative effects.
This overview does not suggest that a guardian’s decision is always inappropriate regarding whether a person should participate in PMR. The history of research with human subjects, including incidents of mistreatment of people with intellectual disability, has demonstrated the need for protections of potential subjects. The potential informational harm, including genetic-based discrimination for individuals and their families, or concerns about community-level harms, are further relevant considerations.Reference Akyüz 30 However, these potential risks rarely result in outright exclusion as often occurs for adults with intellectual disability; the need for protections via guardianship must also be tailored to the individual and consider a complex web of circumstances: the study’s level of risk, duration, goals, and prospect of direct benefits; legal restrictions (e.g., scope of guardianship); and the measures employed to assess an individual’s understanding of consent disclosures and capacity to make research participation decisions, including who conducted the assessment and whether accommodations were provided throughout the process. Significantly, these concerns align with the guidance by the National Institutes of Health, 31 the largest funder of biomedical research in the world, including PMR.
Contrary to guidelines and contextual nuances, we found that the exclusion of adults with intellectual disability from clinical research is often nontransparent; rarely includes validated, structured consent capacity assessment; and heavily relies on poorly defined, highly subjective decisions of research staff.Reference McDonald 32 Significantly, many of these considerations have been raised regarding all research participants — without parallel protection measures being determined necessary. For example, it is well-known that research participants tend to conflate research and clinical treatment (therapeutic misconception)Reference Henderson 33 and that many have limited understanding of some key aspects of the studies in which they are enrolled.Reference Pietrzykowski and Smilowska 34 These limitations in the informed component of consent also exist in consent for biobanking, commonly considered a not greater than minimal risk research procedure, and in PMR studies more generally.Reference Kasperbauer 35 However, while there are disagreements as to the threshold of understanding that is required, there is evidence that researchers tend to accept participation decisions of participants from the general public, regardless of level of understanding.Reference Beskow and Weinfurt 36 Thus, even if balancing agency and protection may sometimes be a difficult act, it cannot — and should not — immediately translate to protections that restrict autonomy, including for adults with intellectual disability.
Given the limitations that guardianship legally, and often practically, imposes on individual rights, an increasing number of advocates, professionals, and policymakers have proposed alternatives to reduce reliance on this approach. Supported decision-making is a relatively recent legal concept (though has naturally existed as an informal practice for a long time), a growing practice, and a promising development with critical implications for consent to PMR. Supported decision-making aims to provide individually tailored supports to facilitate person-first consent while maintaining the person’s position as the decision-maker. The idea of supported decision-making has received increased legal recognition since the emphasis on supports for legal capacity in Article 12 of the CRPD, including in the US. 37 Our legal analysis indicates that 39 states have adopted laws that explicitly codify supported decision-making agreements or require the use of supported decision-making as the least restrictive alternative for guardianship in their guardianship laws, education law, or recognize this possibility in the context of organ transplantation. Significantly, some of these state laws explicitly connect supported decision-making agreements with guardianship. In Maryland, for example, the law explicitly allows an individual subjected to guardianship to enter into a supported decision-making agreement, prohibits a guardian from preventing the individual subjected to guardianship from entering into a supported decision-making agreement “without good cause” (e.g., direct conflict with their own responsibilities under the guardianship order), requires courts to consider the presence of such agreements before appointing a guardian, and allows courts to limit or remove guardianship due to the presence of such an agreement. 38 Such explicit provisions are further accompanied by the fact that, under most state laws, individuals are entitled to rely in good faith on supported decision-making agreements, unless undue influence is clearly present. 39 These legal reforms indicate substantial efforts to avoid guardianship in favor of formal supported decision-making, and other measures to protect individuals’ rights.
However, whether and how laws (most notably on decision-making competence and rights to reasonable accommodations) and societal shifts apply to participation in PMR is not always clear, and there is a need for direct clarification to be integrated into research regulations, policies and practices. This need is especially important for adults with intellectual disability for whom even minor cognitive limitations are dwarfed by deep-seated biases about their abilities.
Identifying ways to maximize inclusion and self-determination is essential. 40 The integration of disability reasonable accommodations — which are a legal requirement in federally funded research pursuant to Section 504 of the Rehabilitation Act of 1973Reference Horner-Johnson and Bailey 41 — into PMR is strongly supported by adults with intellectual disability. They report that inaccessibility and difficulty communicating with researchers are significant barriers to their participation in PMR, and that having the right to make their own decisions requires that they receive accommodations to enhance their understanding of information and ability to communicate.Reference McDonald 42 These accommodations can include working with trusted people who provide them supports to understand and communicate their decisions about research participation. Unsurprisingly, many adults with intellectual disability report an interest in talking with someone of their choice as they make research participation decisions, 43 including both general research and PMR. 44
Supported Decision-Making: A Tool to Facilitate First-Person Consent Regarding Participation in PMR
Although further clarity about the scope of supported decision-making agreements could be helpful, there is a strong legal and ethical justification to leverage supported decision-making as a critical tool and reasonable accommodation to facilitate first-person consent for participation in PMR, and to establish it as a common practice. Adults with intellectual disability regularly use formal and informal supports for decision-makingReference Kohn and Blumenthal 45 including in clinical settings where having support present is routinely practiced.Reference Silverman 46 This approach can be used to support their responsible inclusion in PMR. In such studies, supported decision-making may be especially important as information about the study is often complicated and broad, involving unknown, abstract, and future-oriented risks related to genetic sampling, data sharing, biobanking, and return of personal health results. Consequently, study information and results may be poorly understood.Reference Sabatello 47 Critically, supported decision-making serves as an avenue toward first-person consent when concerns regarding consent capacity are present, or when a potential participant simply feels more confident or comfortable having a supporter there. Supported decision-making maximizes autonomy in research participation decisions through the relationship between a supporter and someone using support to enhance their understanding. Significantly, supported decision-making does not replace the decision-maker with the historical, and commonly used, substituted or surrogate decision-maker, and it entails limitations, namely: it expressly requires that the provided level of support is tailored, strictly necessary to the needs of the individual, and does not otherwise exceed the wishes and needs of the individual. 48
Project ENGAGE is an academic-community partnership comprised of people with a range of personal and professional expertise working on the responsible inclusion of adults with intellectual disability in PMR. We used our national survey with adults with intellectual disability, legal analysis, and expertise and experiences to develop recommendations for research protocols and a practical resource for using supported decision-making in PMR enrollment. Table 1 presents an overview of these recommendations. Integrating supported decision-making into PMR participation decisions is essential to protecting the rights and autonomy of adults with intellectual disability and encompasses a range of situations in which this may arise. These include when: (1) someone already has a formal supported decision-making agreement; (2) someone is subject to a guardianship order that does not apply to the research decision; (3) someone is not subject to guardianship and prefers to use supported decision-making; or (4) there are concerns about an individual’s consent capacity. Of note, supported decision-making must be paired with other efforts to maximize understanding and voluntariness, 49 key aspects of researchers’ responsibilities.
Strategies for Using Supported Decision-Making to Facilitate First-Person Consent

Supported decision-making can exist prior to the invitation to participate in a research study (in a formal or informal agreement) or be initiated during the enrollment process, when there are indications that the potential participant desires involving another person or is having difficulty understanding information about the study. Given the importance of large and diverse cohorts for the generalizability of findings from PMR, researchers should proactively design studies that account for the potential involvement of support in the decision-making process. This approach should include being educated about the meaning and implementation of supported decision-making agreements, being skilled in raising this option with potential participants, planning for reasonable accommodations for discussion prior to consent, and ensuring that this strategy to enhance first-person consent is included in the IRB protocol. The approach also must include raising awareness about supported decision-making among IRB members who may be unaware of this possibility or concerned about its implications for human subject protection. As IRBs gain knowledge of the value of this approach, they can further educate other researchers on its availability as a strategy to enhance first-person consent.
Working in concert with any legal agreements or typical practices for decision-making, adults with intellectual disability should have the right to decide whether they want to use supported decision-making for PMR-related decisions, and, if so, to choose a support person(s) to help them understand consent disclosures, make a decision, and/or communicate their decision. For those who decline such support, if they are subsequently determined through validated means to lack consent capacity, supported decision-making can be reintroduced to enhance understanding and thus their potential for inclusion in the study through first-person consent — thereby avoiding automatic exclusion or inclusion through proxy consent. This exploration should become standard practice as part of the initial introduction to be in a research study.
Precision medicine researchers should communicate to potential participants that it is an option to have a trusted person(s) of their choice join them as they learn about the research study and make a participation decision. Leveraging supported decision-making requires researcher flexibility: some potential participants may already be familiar with or are using supported decision-making; others may be less familiar with or not use supported decision-making. A researcher should be prepared to work with potential participants as they explore possibilities and make choices about who provides them support. If a support provider is present in this early discussion, the researcher should also seek to speak privately to the potential participant to ascertain the voluntariness of their choice. Because some people elect to have different people support them with different types of decisions, researchers should convey key aspects of the research participation decision to enable the potential participant to select the most suitable supporter or discuss whether the study’s topic falls within the scope of an existing agreement and its use is desired by the potential participant. For example, an adult with intellectual disability who is invited to participate in a study involving reproductive health may chose a support person with whom they are comfortable discussing more private topics such as sexual history but look for other skill sets in support persons if the study is on cardiovascular health (e.g., a person who can help with changing one’s physical activity). This approach both respects individuals’ choices in light of their needs and highlights that consent capacity — also in its form of supported decision-making — is malleable and depends on tailored environmental supports. Once in place, supported decision-making can be used in both initial enrollment decisions and subsequent decisions that arise over the course of a study, such as return of personal genetic results.
Another aspect of leveraging the strengths of supported decision-making for research entails being prepared to interact with a potential participant and the person providing them support. This observation means giving careful attention to communication and adhering to their respective roles (e.g., maintaining the potential participant as the decision-maker), whether the support being provided facilitates first-person consent, and the possibility of undue influence or coercion. Researchers must look for indications that the support being provided aligns with the will and preferences of the potential participant. Indicators of good, effective support include: (1) the support person’s ability to effectively describe research study information to the potential participant, (2) the support person’s ability to understand the potential participant’s preferences and help to communicate the individual’s decision(s) to the researcher; and (3) the potential participant’s comfort level asking questions and expressing their opinions. Good support should facilitate first-person consent by translating consent disclosures using words or ideas that are more understandable, making connections to familiar things from personal experiences, helping the person feel comfortable asking questions, and assisting with interpretation of communication (including indicators of confusion, stress, discomfort, or dissent). Using supported decision-making also involves the researcher’s responsibility to ensure that the person providing support understands the potential research participant’s rights, including the right to withdraw from the study.
While supported decision-making can be a powerful tool to enhance autonomy and first-person decision-making, it can also raise unique ethical concerns for PMR. For example, when the person chosen for support is a biological relative, they may be directly affected by genetic results and have their own interests affected by their relative’s participation in the research.Reference Swaine 50 In such situations, it is important to ensure that the support person does not replace their own interests in personal health results or confidentiality with support for the adult with intellectual disability to participate in a study. While such an outcome may very well be unintentional, researchers ought to be aware that coercion, undue influence, or the person with intellectual disability’s desire to please may be a part of the decision-making process. Similarly, if a precision medicine researcher believes there is undue influence being placed on a participation decision, the researcher can take further action including a private conversation about the potential participant’s chosen supporter and their research decision(s), deploying strategies to decrease the support provider’s influence, consultation with colleagues, and, if needed, rejection of the potential participant’s decision if it is determined not to be voluntary. Policies and protocols to dampen undue influence or coercion may need to be further developed or refined.
The (potential or enrolled) participant should also be able to end the relationship with a supporter. Supported decision-making statutes uniformly provide that the person being supported can terminate the arrangement with the supporter at any time. 51 If supported decision-making remains desired or needed to achieve required consent capacity, the person can select a new supporter if they want to be (or continue) in the study. When further decisions are required during the study, researchers can verify that the supporter remains the choice of the participant and verify their continued willingness and availability to provide support.
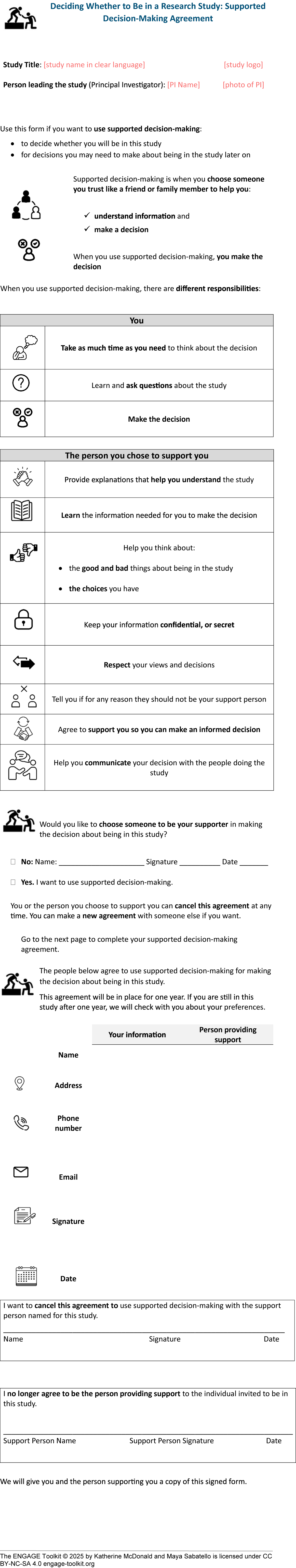
We recommend researchers work with each potential participant to consider whether they want to execute a supported decision-making agreement for the research participation decision. The documented agreement provides an opportunity to formalize the roles of each person, emphasizing that the potential participant retains their right to consent and delineating the responsibilities of the person providing support (including to maintain confidentiality). Figure 1 presents Project ENGAGE’s Supported Decision-Making Agreement: Deciding Whether to be in a Research Study.
Project ENGAGE’s Supported Decision-Making Agreement.

By encouraging researchers to develop study protocols that include supported decision-making as a desirable reasonable accommodation, we anticipate that more adults with intellectual disability will maintain their right to first-person consent and self-determination both for enrollment and subsequent decisions related to being in the study.
Conclusion
The need to assure accessible materials and processes for inclusion of diverse participants in research endeavors is not new, and it has received growing attention with national efforts to promote engagement in PMR. Without diverse cohorts, research findings cannot be generalizable for the broader public, and inequities among communities that have long experienced health disparities are only likely to intensify. Accordingly, the past decade has seen a growth in efforts to assure cultural sensitivity and accessibility throughout study development and implementation.Reference Brett 52 Recent regulatory revisions highlighting accessibility, reasonable accommodations and universal design reflect this recognition and underscore researchers’ responsibility to enhance understanding in consent. 53
Although adults with intellectual disability may be recruited to participate in studies that focus on their disability, their inclusion in general PMR studies (i.e., studies that do not focus on the disability itself) has received considerably less attention to date. Yet their absence is increasingly being recognized, as highlighted, for example, in the 2022 report of the National Academies for Science, Engineering and Medicine 54 and the 2024 report from the National Council on Disability. 55 Because they are an underrepresented health disparities population, the inclusion of adults with intellectual disability is critical for improved prevention, diagnostic, and treatment options to accrue to them. The use of supported decision-making can substantially facilitate the exercise of first-person consent among adults with intellectual disability and serve as a critical tool in their responsible inclusion in PMR.
Implementation of supported decision-making for enrollment in PMR faces several challenges. First, the long history of protectionism in research needs to yield to alternative approaches for enhancing and determining consent capacity, at a time where researchers, and members of IRB who ultimately approve study protocols, may not understand the conceptual and legal construct of supported decision-making. The educational component is significant given that both groups are leading gatekeepers for research participation. 56 Second, since the US is only a signatory and not a ratifier of the CRPD, it is expected to follow its spirit but is not bound by the Convention nor subject to review by the CRPD’s monitoring committee. Although signing a treaty indicates a country’s intention to ratify the instrument, it is unlikely that ratification will be pursued any time soon. Notwithstanding the movement toward adopting supported decision-making statutes noted above, only some states have enacted specific laws to regulate the scope and obligations arising from a supported decision-making agreement. In states without such statutes, informal supported decision-making practices and agreements may still be applicable, but researchers may be less familiar with this option. In addition to the lost opportunity to augment autonomy, this limitation may also translate into greater — and often automatic — reliance on guardianship or exclusion from research. Both options undermine autonomy. Third, while supported decision-making can be informal, the combination of limited knowledge and protectionism may lead to concerns about inappropriate enrollment of individuals into research studies. Identifying where supported decision-making is regulated and explicitly permitted is a first step, though it must also be considered in light of the policies of each research entity. Clear criteria and information about supported decision-making could facilitate the use of such a process.
Supported decision-making also raises a host of ethical, social, and legal challenges that require attention and may be hard to resolve. One way of addressing this issue is by creating informational material and templates that can be easily used by researchers. Despite significant efforts in the past decade to create community-specific tailored study materials, such efforts have not yet been expanded to adults with intellectual disability. For adults with intellectual disability who do not have an existing supported decision-making agreement, researchers should offer the opportunity to execute one for their study. Project ENGAGE’s Supported Decision-Making Agreement allows tailoring of the support to the specific research participation decision and addresses this gap, providing new recommendations and a practical tool co-created with adults with intellectual disability.
Disclosures
Research reported in this work was supported by the Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD) and National Human Genome Research Institute (NHGRI) of the National Institutes of Health under Award Number R01HD108701 (MPIs: McDonald & Sabatello). Katherine McDonald was a co-lead on two projects for the Patient-Centered Outcomes Research Institute from 2021–2026, Research Ethics for All: Building Capacity via Topic Expansion and National Adoption and Research Ethics for All: Research Ethics Training for Patient Stakeholders with Intellectual and Developmental Disabilities, and was a member of the Subject Matter Expert Panel for NIH’s All of Us Disability Community Engagement Partner Project — Adults and the INCLUDE Down Syndrome Cohort Development Program, Consent/Assent Working Group. Robert Dinerstein was a member of the Subject Matter Expert Panel for NIH’s All of Us Disability Community Engagement Partner Project — Adults was a paid American Association on Health and Disability consultant on the All of Us Research Program Mark Cooley received consulting fees as a Program Consultant for the NIH INCLUDE Project and for serving on the IRB for the NIH All of Us Research Program. Maya Sabatello received consulting fees as a Program Consultant for the NIH INCLUDE Project and for serving on the IRB for the NIH All of Us Research Program.